Imidazo[1,5]carbachlorin as a hybrid ligand
Kinga
Szydełko
 ,
Sebastian
Koniarz
,
Sebastian
Koniarz
 ,
Michał J.
Białek
,
Michał J.
Białek
 and
Piotr J.
Chmielewski
and
Piotr J.
Chmielewski
 *
*
Department of Chemistry, University of Wrocław, F. Joliot-Curie 14, 50 383 Wrocław, Poland. E-mail: piotr.chmielewski@uwr.edu.pl
First published on 16th October 2025
Abstract
A family of novel complexes of imidazo[1,5]carbachlorin has been synthesized and characterized by spectroscopic, X-ray diffraction, and electrochemical methods. In most of these compounds, both the macrocyclic crevice and the fused external imidazole are involved in the coordination of the metal ion, thus defining imidazocarbachlorin as a hybrid ligand system consisting of NNNC- and N-donor sets. The effect of the external coordination on the shape of the macrocyclic ring has been studied and discussed.
Introduction
N-confused porphyrin (NCP), i.e., 2-aza-21-carbaporphyrin (Fig. 1), is an aromatic tetrapyrrole of distinct coordinating properties related to the structural features of the macrocyclic ring.1–3 Apart from the internal NNNC core that may adopt several coordination modes and geometries and stabilize uncommon oxidation states,4–31 there is an external nitrogenous donor site located on the peripheries of the macrocycle. Several monomeric and oligomeric transition metal complexes of NCP and its derivatives have been reported, where the macrocyclic core and monodentate external N-donor of the confused pyrrole are both involved in coordination.11,14,18,20,32–38 In several of such complexes, ligation of the external nitrogen N2 is supported by ortho-metalation of an aryl located at the adjacent meso-position C20,12,38–41 which is accompanied in some instances with a severe distortion of the porphyrinoid geometry. Obviously, such an external ligation of the regular porphyrin Por (Fig. 1) requires an additional donor atom or donor set localized at the macrocyclic perimeter, which is well-exposed and points towards the potential coordination center. In this regard, the most interesting seem to be imidazole-fused porphyrins,42–52 which can also be converted from a potentially N-donating species into the N-heterocyclic carbene ligand (NHC) by alkylation of both imidazole nitrogens.44–50,53 Owing to the [2,3] ortho-fusion mode of the imidazole-pyrrole moiety and the steric factors imposed by the aryls at the meso-positions C5 and C20, these derivatives appear to be effective NHC ligands rather than N-donors. In fact, to date, there has been no report regarding N-coordination of porphyrin-fused imidazole. Conversely, in the readily obtainable imidazo[1,5]carbachlorin (ICC),54 the well-exposed nitrogen of the ortho-fused imidazole constitutes a promising donor site. However, also for this system, only NHC-type gold(I) extraannular coordination of the N-methylated derivative of silver(III) complex of ICC (AgICCMe) has been reported until now (Fig. 1). We thus decided to explore the coordination of the external nitrogen of the ICC free base as well as of its complexes with inert d8 transition metals, as well as boron(III). Special attention will be given to the structural alterations of the porphyrinoid ring and changes in the redox properties of the complexes caused by the external coordination.Results and discussion
Synthesis and structure
Although the coordination cores of NCP and ICC seem to be analogous, there is an essential distinction between these ligands that is related to the presence of a basic center N2 at the perimeter of the macrocycle in the former. Thus, the NCP macrocyclic core can act as a mono-, di-, or tris(anionic) ligand forming neutral complexes with divalent or trivalent metal cations, with 2-NH compensating an excess of the negative charge in the case of M2+-NCP systems. On the other hand, the imidazole imide-like nitrogen N3′ in ICC appears to be unable to hold the proton under neutral conditions. Hence, ICC is invariantly a tris(anionic) ligand if the macrocyclic coordination core is concerned, stabilizing trivalent metals and concurrently preserving basic nucleophilic properties of N3′. Thus, we chose a readily obtainable silver(III) complex as a workhorse and common platform in our study of the coordination of the imidazole nitrogen of ICC. However, in some instances, we observed here the coordination of the N3′ site in the systems not involving the macrocyclic crevice in ligation. This allowed us to discuss the influence of the macrocyclic ring rigidity, imposed by the coordination of the inert d8 metal ion, on the overall structure of the bis(metallic) system.The syntheses were carried out under relatively mild conditions, and the products were characterized by spectroscopic methods, including 1D NMR and 2D homo- and heteronuclear correlation techniques, single crystal X-ray diffraction analyses (XRD), optical spectroscopy, and electrochemical methods (cyclic and differential pulse voltammetry).
![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) as a racemate.
as a racemate.
 | ||
| Fig. 2 Molecular structures of the triphenylboroxine adduct of ICC based on XRD. The two symmetry-independent molecules are presented separately as molecule A and molecule B. In front views (top, 50% probability displacement ellipsoids), all hydrogens were removed. In the stick side views (mid), the meso-phenyls were removed for clarity. In the bottom line, the atom displacements from the porphyrin mean plane calculated67 from XRD data are presented for both molecules. The error of the distance estimations is ≤0.01 Å. | ||
| Compound | Doming [Å] | Saddling [Å] | Ruffling [Å] | WavingX [Å] | WavingY [Å] | Propellering [Å] | D oop [Å] | DH1b [°] | DH2c [°] | d ext |
|---|---|---|---|---|---|---|---|---|---|---|
| a Total out-of-plane distortion, ±0.01 Å. b Dihedral angle (±0.5°) between the mean plane defined by all non-hydrogen atoms of the macrocyclic ring except C21, N2, and C3, and the mean plane of the confused pyrrole. c Dihedral angle (±0.5°) between the mean planes of the confused pyrrole and fused imidazole rings. d Distance between coordinated boron and the mean plane defined by C21, N22, N23, and N24. e X-ray data taken from ref. 2. f X-ray data taken from ref. 74. | ||||||||||
| NCP | 0.196 | 1.360 | −0.070 | 0.259 | −0.055 | 0.040 | 1.465 | 27.0 | — | — |
| ICC | 0.000 | 0.000 | 0.000 | −0.147 | 0.369 | −0.001 | 0.457 | 16.6 | 7.5 | — |
| ICC(PhBO)3A | −0.441 | −1.620 | 0.576 | −0.155 | 0.042 | 0.004 | 1.817 | 31.8 | 3.3 | 3.116 |
| ICC(PhBO)3B | 0.025 | 1.185 | 0.260 | 0.224 | 0.025 | 0.020 | 1.289 | 21.2 | 1.2 | 2.188 |
The 1H NMR study indicated persistence of the adduct in solution and fluxional character of ICC(PhBO)3. Pyrrole β-proton signals were downfield shifted by about 0.1 ppm with respect to the ICC free base. At the same time, imidazole H2′ and H4′ resonated at, respectively, 0.32 and 0.23 ppm higher chemical shifts than in the parent ligand. On the other hand, the signal of 21-CH appeared at δ −5.76 ppm in the adduct, indicating it was upfield shifted by 0.23 ppm compared to the free ICC. Apart from the changes in the chemical shifts observed for the signals of the porphyrinoid, there were two broad resonances at δ 7.40 (ortho-H) and 7.96 ppm (meta- and para-H) with a total relative intensity of 15H, diagnostic for the presence of (PhBO)3 in solution. However, the observed lack of differentiation of the phenyl signals placed in the magnetically distinct sites, i.e., at tetrahedral and trigonal borons, strongly suggested a chemical exchange that is fast in the NMR time scale above 250 K (Fig. 3). Similar dynamics in solution has been observed and analyzed for (PhBO)3(7-azaindole)71 and for the adducts of other tris(aryl)boroxine with amines.73 The averaging of the chemical shifts indicated a fast change of the boron atom involved in the coordination of the imidazole nitrogen, and thus, a highly labile character of the complex. Moreover, upon crystal growing of ICC(PhBO)3, we obtained from the mother liquor (DCM/hexane) another crystal that was colorless and was identified by XRD as that of free (PhBO)3 occurring by a decomposition of the adduct, in line with its labile character.
 | ||
| Fig. 4 Low-field regions of the 1H NMR spectra (300 K, CDCl3) of AuICCAuCl3 (A), AgICC (B), and AgICCAuCl (C). s = a residual CHCl3 signal. | ||
The XRD analysis confirmed the structure of the complex, revealing the presence of two gold ions: one internally coordinated by the ICC macrocyclic core, and the other bound to the imidazole nitrogen and three chlorides (Fig. 5).
 | ||
| Fig. 5 Upper box: Molecular structures based on XRD analyses (50% displacement ellipsoids) of AuICCAuCl3 (top, left column) and AuNCPAuCl3 (right column). All hydrogens are omitted. In the ball-and-sticks representations of the molecule side views (mid), all meso phenyls are also removed for clarity. The bottom line presents part of the packing diagrams showing Cl-Cl intermolecular contacts in both crystals. Lower box: the atom displacements from the porphyrin mean plane calculated67 from XRD data of AuICCAuCl3 (black sticks) and AuNCPAuCl3 (red sticks). The error of the distance estimations is ≤0.01 Å. | ||
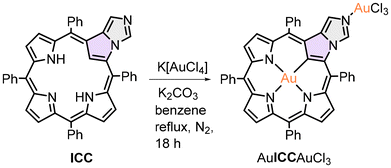
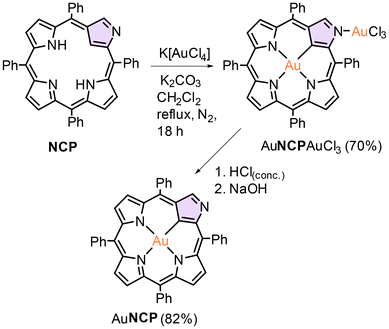
The high efficiency of the gold insertion into the coordination core of the carbaporphyrinoid upon application of K[AuCl4] prompted us to repeat that synthetic approach for unaltered NCP. Although AuNCP was obtained in 2008 by Furuta and co-workers,23 the gold(III) insertion by means of AuI(Me2S)Cl as a metal source requires activation of the porphyrin by bromination of the inner carbon C21. That, in turn, results in a partial inversion of the confused pyrrole and formation of the N-fused porphyrin,75 reducing the metalation yield. In our hands, the reaction of NCP with potassium tetrachloroaurate(III) proceeded smoothly in refluxing dichloromethane in the presence of a proton scavenger (solid K2CO3), giving rise to the product containing two gold(III) ions (Scheme 3). XRD and NMR (Fig. 5 and 6) verified the formation and composition of the complex. Furthermore, the bis(gold(III)) complex AuNCPAuCl3 was shown to undergo facile demetallation at the external coordination site, efficiently giving rise to AuNCP, containing gold(III) only in the porphyrin core (Scheme 3). That species was identical to that obtained by Furuta et al., as we established based on 1H NMR (Fig. 6). Interestingly, despite rather harsh conditions applied for the demetallation, i.e., concentrated hydrochloric acid, the carbaporphyrinoid gold(III) complex AuNCP remained intact, which indicated a high thermodynamic and kinetic stability of this species. Conversely, the analogous silver(III) NCP complex can be easily demetallated under such conditions. On the other hand, the ICC bis[gold(III)] complex, AuICCAuCl3, appeared to be less robust than its NCP counterpart and decomposed into many unidentified products upon attempted external demetallation with HCl.
 | ||
| Fig. 6 Low-field regions of the 1H NMR spectra (300 K, CDCl3) of AuNCPAuCl3 (bottom), and AuNCP (top). | ||
Both bis[gold(III)] complexes indicated similar structural features of the macrocyclic and external chromophores. For AuICCAuCl3 the inner Au1–N bond lengths were 2.030(5), 2.059(5), and 2.036(5) Å while the Au1–C21 distance was 2.004(6) Å. For the external Au2, the distances with chorines were 2.270(2), 2.262(2), and 2.270(2) Å, and the unique Au2–N was 2.0254(5) Å. For AuNCPAuCl3, due to two-fold disorder, only a rough estimation of the average inner Au1–N/C bond length of 2.04 Å was available, and the external Au2–Cl lengths were 2.29(1), 2.201(8), and 2.31(1) Å with an Au2–N distance of 2.00(1) Å. The average displacement of the donor atoms from the Au1CNNN mean plane was about 0.06 with the metal lying 0.01 Å over that plane in AuICCAuCl3. In comparison, the respective displacements were estimated as 0.05 Å and 0 Å in AuNCPAuCl3, indicating effective planarity of the metal environment in both macrocyclic complexes. Also, Au2ClClClN chromophores were essentially planar in both species, with average displacement of chlorines and nitrogen in AuICCAuCl3 of 0.07 but 0.03 and 0.01 Å, respectively, in AuNCPAuCl3. The tetrapyrrole ring in both complexes was saddle-shaped (Fig. 5, Table 2). However, a total deviation from the mean plane Doop was slightly higher for AuNCPAuCl3, and so were the individual displacements from the mean plane of the macrocyclic atoms (Fig. 5). Likely, the shorter distance between the macrocycle and the external AuCl3 moiety coordinating directly to the confused pyrrole may be responsible for the higher deformation of the ring. It is definitely more space for such a moiety in AuICC, which may be inferred from the lower value of the dihedral angle between the mean planes of chromophores Au1CNNN and Au2ClClClN in AuICCAuCl3 (37.8°) than in AuNCPAuCl3 (67.7°). The internal and external chromophores cannot be coplanar with each other due to the repulsion between the chlorides and the imidazole carbons and hydrogens (C2′H and C4′H) in AuICCAuCl3. However, a stronger repulsion is observed for the analogous NCP complex. The presence of an aryl substituent at the adjacent meso position (C20) in AuNCPAuCl3 pushes the gold(III)–trichloride plane in a position that is almost perpendicular to the plane of the macrocycle. For both complexes, intermolecular interaction in the solid state was inferred from an appropriate orientation of the interacting molecules as well as from the Cl–Cl distances observed in both crystal structures (Fig. 5) that were shorter than the sum of the chlorine van der Waals radii (3.5 Å).
| Compound | Doming [Å] | Saddling [Å] | Ruffling [Å] | WavingX [Å] | WavingY [Å] | Propellering [Å] | D oop [Å] | DH1b [°] | DH2c [°] | d ext [Å] | ANexte |
|---|---|---|---|---|---|---|---|---|---|---|---|
| a Total out-of-plane distortion. The error of the distance estimations is ≤0.01 Å. b Dihedral angle (±0.5°) between the mean plane defined by all non-hydrogen atoms of the macrocyclic ring except C21, N2, and C3, and the mean plane of the confused pyrrole. c Dihedral angle (±0.5°) between the mean planes of the confused pyrrole and the fused imidazole rings. d Distance between the externally coordinated metal atom and the mean plane defined by internal chromophore MC21N22N23N24. e An angle between C21–Next and Next–M directions. f X-ray data taken from ref. 74. | |||||||||||
| AuNCPAuCl3 | 0.000 | 1.173 | −0.001 | −0.107 | 0.103 | −0.017 | 1.193 | 8.8 | — | 1.03 | 160.2(8) |
| AuICCAuCl3 | −0.019 | −1.059 | −0.069 | −0.085 | 0.077 | 0.003 | 1.072 | 9.7 | 2.5 | 0.99 | 178.3(3) |
| AgICCTAuCl | 0.080 | −1.214 | 0.267 | 0.031 | –0.033 | 0.007 | 1.252 | 11.1 | 1.7 | 0.61 | 172.7(3) |
| AgICCIrCp*Cl2 | 0.149 | 0.757 | −0.063 | 0.056 | –0.110 | 0.011 | 0.810 | 12.0 | 4.3 | 2.143 | 162.5(1) |
| AgICCMeAuClf | −0.055 | −1.417 | −0.513 | 0.053 | –0.140 | −0.067 | 1.528 | 14.5 | 5.4 | 2.078 | |
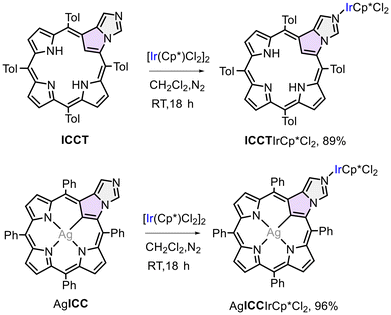
Another gold complex was obtained in the reaction of silver(III) ICCT complex with [AuCl(Me2S)]. The mix-metal complex containing trivalent silver and monovalent gold was formed under mild conditions (Scheme 4). The structure of this species was evaluated based on the XRD analysis (Fig. 7). The Au1–N and Au1–Cl1 bond lengths (1.993(7) and 2.244(2) Å, respectively) are slightly shorter than those observed for AuICCAuCl3 or AuNCPAuCl3. 1H NMR spectrum indicated the presence of a silver atom in the porphyrin coordination core by showing split pyrrole proton signals due to H–Ag spin–spin coupling (4JAgH = 1.5 Hz).† In the crystal lattice of AgICCTAuCl, formation of a dimer was observed with mutual interaction of the gold ion belonging to one of the monomers with the β-pyrrole carbons C12 and C13 of the other monomer (Fig. 7A). The distances 3.185(7), 3.139(8), and 3.089 for Au1–C12, Au1–C13, and the Au–(C12![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C13) centroid, respectively are lower than the sum of the van der Waals radii and strongly suggest the presence of a non-covalent interaction of the electron-rich metal ion and ethylene-like moiety in ICC. Deviation from planarity of the macrocycle was significantly more substantial in AgICCTAuCl than that observed for bis[gold(III)] systems (Table 2), involving doming and ruffling of the ring apart from a more pronounced saddling. That may be the result of the crystal packing forces, mainly the dimer formation in the crystal lattice of AgICCTAuCl. On the other hand, in all these complexes comprising externally coordinated gold(I/III) ion, the macrocycle displayed less pronounced deformation than that of the previously reported system,74 where gold(I) chloride is bound to C2′ of N-methylated imidazole in AgICCMe acting as N-heterocyclic carbene ligand (Fig. 1, 7C and Table 2). It may be a result of the steric congestion in the region of external metal coordination in AgICCMeAgCl, causing deformation of the ring due to repulsion between the AuCl moiety and the aryl substituent at the meso-position C5. No such overcrowding occurs in the molecules consisting of N-coordinating imidazole of AgICC or AuICC.
C13) centroid, respectively are lower than the sum of the van der Waals radii and strongly suggest the presence of a non-covalent interaction of the electron-rich metal ion and ethylene-like moiety in ICC. Deviation from planarity of the macrocycle was significantly more substantial in AgICCTAuCl than that observed for bis[gold(III)] systems (Table 2), involving doming and ruffling of the ring apart from a more pronounced saddling. That may be the result of the crystal packing forces, mainly the dimer formation in the crystal lattice of AgICCTAuCl. On the other hand, in all these complexes comprising externally coordinated gold(I/III) ion, the macrocycle displayed less pronounced deformation than that of the previously reported system,74 where gold(I) chloride is bound to C2′ of N-methylated imidazole in AgICCMe acting as N-heterocyclic carbene ligand (Fig. 1, 7C and Table 2). It may be a result of the steric congestion in the region of external metal coordination in AgICCMeAgCl, causing deformation of the ring due to repulsion between the AuCl moiety and the aryl substituent at the meso-position C5. No such overcrowding occurs in the molecules consisting of N-coordinating imidazole of AgICC or AuICC.
 | ||
| Fig. 7 Panel A: Molecular structure based on XRD analyses (50% displacement ellipsoids) of AgICCTAuCl (top), ball-and-sticks representation of side view (mid), and two views of the fragment of crystal packing with gold and interacting carbon atoms depicted with space-filling representations (bottom). Panel B: Molecular structure based on XRD analyses (50% displacement ellipsoids) of AgICCIrCp*Cl2 (top), ball-and-sticks representation of side view (mid), and a fragment of crystal packing with interacting carbon atoms depicted with space-filling representations (bottom). All hydrogens are omitted. In the ball-and-sticks representations of the molecule, all meso-phenyls are also removed for clarity. Panel C: The atom displacements from the porphyrin mean plane calculated67 from XRD data of AgICCTAuCl (black sticks), AgICCIrCp*Cl2 (blue sticks), and AgICCMeAuCl (red sticks).74 The error of the distance estimations is ≤0.01 Å. | ||
Electronic and redox properties
| System | E Red1 [V] | E Red2 [V] | E Ox1 [V] | E Ox2 [V] | HLG [eV] |
|---|---|---|---|---|---|
| a Data from ref. 74. b All processes irreversible. c Ligand with p-tolyl instead of phenyl meso-substituents. Data from ref. 74. d Irreversible process. e Data from ref. 23. | |||||
| ICC | −1.74 | −2.23 | 0.25 | 0.49 | 1.99 |
ICC(BOPh)3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b b |
−1.76 | −2.25 | 0.49 | 0.64 | 2.25 |
AgICC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a a |
−1.31 | 0.36 | 0.48 | 1.67 | |
AgICCT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
−1.32 | 0.35 | 0.49 | 1.67 | |
| AgICCAuCl | −1.23 | −1.36 | 0.56 | 0.90d | 1.79 |
| AgICCIrCp*Cl | −1.21 | 0.49 | 0.58 | 1.71 | |
| AuICCAuCl3 | −0.43d | −1.89 | 0.70 | 1.11 | 1.14 |
| AuNCPAuCl3 | −0.49d | −1.40 | 0.86 | 1.35 | |
AuNCP![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e e |
−1.54d | 0.53 | 2.07 | ||
Conclusions
In conclusion, we have shown here that imidazocarbachlorin can serve as a monodentate ligand in both free-base and internally metallated forms. Coordination by the externally located imidazole nitrogen affects to some extent the structure of the ligand, and the changes of its shape are much more pronounced for the system with no metal inside the porphyrin core. The fused imidazole is shown to be a part of the coordination sphere of linear, tetrahedral, square pyramidal, and piano-stool geometries. We anticipate that the exposition of the imidazole nitrogen out of the sterically congested region of adjacent meso-aryls enables coordination of more than one molecule of ICC to the metal ions, thus allowing formation of coordinative dimers or trimers. External coordination influences the optical properties of the porphyrinoid, resulting in a hypsochromic shift of the Soret and Q-bands. Strong absorption of the metalloligands MICC and their redox properties, along with coordination abilities of the external metal centers, may find application in the construction of various catalytic and photocatalytic systems tailored by judicious choice of the internal and external central atoms. Further study on these subjects is underway in our laboratory.Experimental
Methods and instrumentation
Commercial reagents were used without further purification. Solvents were freshly distilled from the appropriate drying agents or purified under nitrogen with the mBraun MBSPS-800 before use. Column chromatography was performed using silica gel 60 (200–300 mesh ASTM). The NMR spectra were recorded on a Bruker Avance III spectrometer, operating at 500 MHz for 1H and 125 MHz for 13C, or a Bruker Avance III spectrometer operating at 600 MHz for 1H and 150 MHz for 13C. TMS was used as an internal reference for 1H and 13C chemical shifts, and CDCl3 was used as the solvent. Standard pulse programs from the Bruker library were used for homo- and heteronuclear 2D experiments. Mass spectrometry measurements were conducted by using the electrospray ionization technique on a Bruker Daltonics microTOF-Q. Absorption UV/Vis/NIR spectra were recorded by using a Varian Cary 60 and Jasco V-770 spectrophotometers. Electrochemical measurements were performed by means of an Autolab (Metrohm) potentiostat/galvanostat system for dichloromethane solutions with a glassy carbon, a platinum wire, and Ag/AgCl as the working, auxiliary, and pseudoreference electrodes, respectively. Tetrabutylammonium hexafluorophosphate was used as a supporting electrolyte. The potentials were referenced with the ferrocene/ferrocenium couple used as an internal standard. The X-ray diffraction was measured using either XtaLAB Synergy R or Xcalibur, Onyx diffractometers using a copper source of radiation (λ = 1.54184 Å), and collected with a CCD camera. The standard temperature of the measurement was 100 K. The structures were solved using direct methods with SHELXT79 and refined by the full-matrix least-squares method on all F2 data by using the SHELXL80,81 incorporated in the OLEX2 program.81 All hydrogen atoms, including those located in the difference density map, were placed in calculated positions and refined as the riding model. The SQUEEZE procedure,82 was applied for disordered lattice solvent molecules in AgICCAuCl, AuNCPAuCl3, and ICC(BOPh)3. Crystallographic details are collected in Tables S1–S5 (SI). Disordered solvent molecules masked with OLEX2 were included in final crystal compositions (structures AuNCPAuCl3, AgICCAuCl, and ICC(BOPh)3). Due to partial reduction of the externally attached AuCln and consecutive demetallation at 3′, which likely occurred during crystal growing, these fragments were refined with <100% occupancy in structures of AuICCAuCl3 and AgICCAuCl. In the structure of AuNCPAuCl3, maximum around Au1 was assigned as a small disordered gold contribution. Its occupancy was refined to be ca. 4%, then was fixed, and the displacement ellipsoid was constrained with EADP to Au1. CCDC 2394665, 2394666, 2394670, 2394671, 2394677 contain supplementary crystallographic data for this paper.Syntheses and characterizations
Selected data for ICC(BOPh)3: 1H NMR (600 MHz, CDCl3, 300 K): δ −5.76 (s, 1H, –CH (21)), −1.84 (s, 2H, –NH), 6.51 (s, 1H, (4′)), 7.21 (s, 1H, (2′)), 7.43 (m, 10H, –Ph(B)), 7.77 (m, 12H, (m-Ph, p-Ph, ArH)), 7.99 (m, 5H, –Ph(B)), 8.05 (d, 4H, 3JHH = 7.64 (o-Ph, ArH)), 8.15 (d, 4H, 3JHH = 7.57 (o-Ph, ArH)), 8.59 (m, 2H, (pyrrH)), 8.63 (m, 2H, (pyrrH)), 8.67 (AB, 1H, 3JHH = 4.88, (pyrrH)), 8.72 (AB, 1H, 3JHH = 4.95, (pyrrH)). 13C NMR (150 MHz, CDCl3, 300 K): δ 155.7, 141.6, 140.1, 138.2, 138.1, 138.0, 137.8, 136.9, 134.5, 134.4, 133.3, 132.7, 130.4, 130.3, 130.2, 129.3, 128.9, 128.2, 127.4, 127.3, 127.1, 126.9, 126.6, 126.3, 125.9, 121.1, 119.4, 114.5, 111.8, 103.7. UV-vis (CH2Cl2) λ [nm] (ε/104 [M−1 cm−1]) = 334 (1.08), 350 (sh), 368 (1.19), 380 (sh), 448 (7.98), 509 (sh), 534 (1.09), 562 (sh), 577 (0.32), 617 (sh), 640 (0.24), 673 (sh).
Selected data for AgICCIrCp*Cl2: 1H NMR (600 MHz, CDCl3, 300 K): δ 1.56 (s, 15H, Cp*), 6.44 (s, 1H, (4′)), 7.21 (s, 1H, (2′)), 7.76 (m, 6H, ArH), 7.82 (m, 3H, ArH), 7.91 (m, 3H, ArH), 7.97 (m, 2H, ArH), 8.00 (m, 2H, ArH), 8.13 (d, 3JHH = 7.8 Hz, 4H, ArH), 8.67 (AB, 3JHH = 4.8 Hz, 1H, pyrrH), 8.67 (AB, 3JHH = 4.8 Hz, 1H, pyrrH), 8.73 (d, 4JAgH = 1.2 Hz, 2H, pyrrH), 8.75 (3JHH = 4.9 Hz, 4JAgH = 0.8 Hz, 1H, pyrrH), 8.83 (dd, 3JHH = 5.0 Hz, 4JAgH = 1.5 Hz, 1H, pyrrH). 13C NMR (150 MHz, CDCl3, 300 K): δ 141.6, 141.5, 140.8, 139.2, 139.0, 138.2, 138.1, 137.5, 137.3, 137.1, 133.9, 133.8, 131.7, 131.2, 130.4, 130.2, 130.1, 129.8, 129.3, 129.2, 128.8, 128.8, 128.3, 127.3, 127.0, 126.8, 124.1, 122.9, 122.5, 122.0, 121.8, 117.2, 115.3, 85.1, 77.4, 77.2, 76.9, 8.9. UV-vis (CH2Cl2) λ [nm] (ε/104 [M−1 cm−1]) = 264 (5.51), 327 (sh), 388 (2.23), 402 (sh), 449 (22.40), 502 (sh), 526 (2.16), 547(sh), 556 (1.17), 561 (sh), 569 (1.36), 600 (sh), 617 (0.46).
Selected data for ICCTIrCp*Cl2: 1H NMR (600 MHz, CDCl3, 300 K): δ −5.71 (s, 1H, 21-CH), 0.79–(−3.71) (2H, 22-NH, 24-NH), 1.58 (s, 15H, Cp*), 2.68 (m, 9H, (–CH3)), 2.72 (s, 3H, (–CH3)), 6.38 (s, 1H, (4′)), 7.16 (s, 1H, (2′)), 7.55 (AA′ XX′, 4H, (m-tol, ArH)), 7.63 (AA′ XX′, 2H, (m-tol, ArH)), 7.71 (AA′ XX′, 2H, (m-tol, ArH)), 8.03 (m, 8H, (o-tol, ArH)), 8.57 (m, 2H, pyrrH), 8.60 (AB, 3JHH = 4.7 Hz, 1H, pyrrH), 8.62 (AB, 3JHH = 5.1 Hz, 1H, pyrrH), 8.67 (AB, 3JHH = 4.7 Hz, 1H, pyrrH), 8.69 (AB, 3JHH = 5.1 Hz, 1H, pyrrH). 13C NMR (150 MHz, CDCl3, 300 K): δ 155.4, 140.0, 138.9, 138.7, 138.4, 138.3, 137.9, 137.7, 137.1, 135.4, 134.5, 134.1, 134.0, 133.5, 132.7, 132.5, 131.8, 130.9, 129.5, 127.8, 127.1, 126.9, 126.7, 125.9, 125.7, 121.9, 120.8, 118.7, 111.5, 103.0, 102.9, 100.0, 94.5, 88.3, 86.4, 85.0, 32.1, 29.8, 29.5, 22.8, 21.8, 21.6, 14.2, 9.5, 9.2, 8.7. HRMS (ESI): m/z 1072.3677 [M − Cl], calcd for C60H54ClIrN5 1072.3688. UV-vis (CH2Cl2) λ [nm] (ε/104 [M−1 cm−1]) = 341 (1.80), 357 (sh), 450 (11.76), 510 (sh), 533 (1.57), 564 (sh), 574 (0.64), 615 (sh), 636 (0.40), 700 (0.22).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH2Cl2 – 40
CH2Cl2 – 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 60). The collected fraction was evaporated to dryness and recrystallized from the dichloromethane/n-hexane system. Yield 4.3 mg (31%).
60). The collected fraction was evaporated to dryness and recrystallized from the dichloromethane/n-hexane system. Yield 4.3 mg (31%).
Selected data for AgICCTAuCl: 1H NMR (600 MHz, CDCl3, 300 K): δ 2.70 (s, 6H, (–CH3)), 2.75 (s, 3H, (–CH3)), 2.76 (s, 3H, (–CH3)), 6.33 (s, 1H, (4′)), 7.10 (s, 1H, (2′)), 7.56 (AA′ XX′, 3JHH = 7.75 Hz, 4H, (m-tol, ArH)), 7.62 (AA′ XX′, 3JHH = 7.9 Hz, 2H, (m-tol, ArH)), 7.70 (AA′ XX′, 3JHH = 7.8 Hz, 2H, (m-tol, ArH)), 7.83 (3JHH = 8.0 Hz, 4H, (o-tol, ArH)), 8.00 (AA′ XX′, 3JHH = 7.2 Hz, 4H, (m-tol, ArH)), 8.67 (AB, 3JHH = 4.7 Hz, 4JAgH = 1.4 Hz, 1H, pyrrH), 8.72 (m, 2H, pyrrH), 8.76 (AB, 3JHH = 4.8 Hz, 1H, pyrrH), 8.79 (m, 1H, pyrrH), 8.82 (AB, 3JHH = 4.8 Hz, 4JAgH = 1.5 Hz, 1H, pyrrH). 13C NMR (150 MHz, CDCl3, 300 K): δ 140.4, 139.5, 139.2, 139.1, 139.0, 138.6, 138.5, 138.4, 138.3, 138.2, 138.1, 137.9, 137.7, 137.5, 135.1, 133.8, 133.8, 133.7, 132.6, 131.3, 131.1, 130.9, 130.7, 130.5, 130.1, 129.8, 129.7, 129.6, 129.3, 128.1, 128.0, 127.3, 127.0, 123.4, 122.8, 122.5, 121.6, 116.0, 115.4, 32.1, 29.8, 29.5, 22.8, 22.0, 21.9, 21.7. UV-vis (CH2Cl2) λ [nm] (ε/104 [M−1 cm−1]) = 373 (1.17) 393 (sh), 428(14.59), 480 (sh), 514 (1.06), 544 (sh), 557 (0.81), 595 (sh).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH2Cl2 – 30
CH2Cl2 – 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70). The collected fraction was evaporated to dryness and recrystallized from the dichloromethane/n-hexane solvent system. Yield 63 mg (71%).
70). The collected fraction was evaporated to dryness and recrystallized from the dichloromethane/n-hexane solvent system. Yield 63 mg (71%).
Selected data for AuICCAuCl3: 1H NMR (600 MHz, CDCl3, 300 K): δ1H NMR (600 MHz, CDCl3) δ (ppm): 6.66 (s, 1H, (4′)), 7.38 (s, 1H, (2′)), 7.79 (m, 5H, ArH), 7.86 (m, 3H, ArH), 7.94 (m, 4H, ArH), 7.98 (m, 4H, ArH), 8.13 (d, 3JHH = 7.3 Hz, 4H, ArH), 8.82 (m, 3H, pyrrH), 8.83 (m, 2H, pyrrH), 8.91 (AB, 3JHH = 5.1 Hz, 1H, pyrrH). 13C NMR (150 MHz, CDCl3, 300 K): δ 140.8, 140.2, 138.6, 138.3, 137.9, 137.6, 137.5, 137.2, 136.3, 135.8, 133.8, 131.3, 131.1, 130.8, 130.7, 130.5, 130.1, 129.8, 129.6, 129.5, 129.0, 128.8, 128.6, 127.6, 127.5, 127.4, 124.1, 124.0, 122.3, 122.0, 118.6, 117.5, 116.5, 102.7. UV-vis (CH2Cl2) λ [nm] (ε/104 [M−1 cm−1]) = 428 (18.31), 493 (sh), 514 (1.47), 534 (sh), 557 (1.17).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH2Cl2 – 30
CH2Cl2 – 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70). The collected fraction was evaporated to dryness and recrystallized from the dichloromethane/n-hexane solvent system. Yield 78 mg (71%).
70). The collected fraction was evaporated to dryness and recrystallized from the dichloromethane/n-hexane solvent system. Yield 78 mg (71%).
Selected data for AuNCPAuCl3: 1H NMR (500 MHz, CDCl3, 300 K): δ 7.71–7.80 (om, 9H, ArH), 7.83 (om, 2H, ArH), 7.90 (om, 1H, ArH), 8.01 (om, 4H, ArH), 8.06 (om, 2H, ArH), (om, 1H, ArH), 8.15 (om, 2H, ArH), 8.50 (AB, 3JHH = 5.1 Hz, 1H, pyrrH), 8.53 (AB, 3JHH = 5.1 Hz, 1H, pyrrH), 8.55 (AB, 3JHH = 5.1 Hz, 1H, pyrrH), 8.56 (AB, 3JHH = 5.1 Hz, 1H, pyrrH), 8.64 (AB, 3JHH = 5.1 Hz, 1H, pyrrH), 8.83 (AB, 3JHH = 5.1 Hz, 1H, pyrrH), 9.36 (s, 1H, pyrrH). 13C NMR (150 MHz, CDCl3, 300 K): δ 160.7, 142.7, 142.6, 141.1, 141.0, 139.7, 139.4, 137.9, 136.4, 135.9, 135.7, 134.4, 133.65, 133.61, 133.58, 133.0, 132.8, 132.5, 132.1, 131.8, 130.9, 130.0, 129.5, 129.1, 128.8, 128.7, 128.0, 127.9, 127.7, 127.61, 127.56, 122.1, 121.1, 120.6, 103.0. UV-vis (CH2Cl2) λ [nm] (ε/104 [M−1 cm−1]) = 292 (sh), 316 (2.18), 384 (sh), 438 (16.65), 496 (sh), 517 (1.28), 541(sh), 558 (0.92), 590 (sh), 619 (0.81), 640 (sh), 668 (1.29).
Crystallographic data
AgICCAuCl (CCDC 2394666): C54H45Ag0.94Au0.9Cl2.84N5, Mr = 1143.29, monoclinic, P21/n, a = 16.4860(2) Å, b = 9.15880(12) Å, c = 30.2897(4) Å, β = 92.7667(12)°, Z = 4, R = 0.0623 [I ≧ 2σ(I)], Rw = 0.1578 (all data), GOF = 1.037.AuICCAuCl3 (CCDC 2394670): C47H30Au1.9Cl4.7N5, Mr = 1205.61, triclinic, P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) , a = 9.41147(18) Å, b = 14.0313(3) Å, c = 16.8034(3) Å, α = 110.8961(17)°, β = 94.6483(15)°, γ = 100.4507(16)°, Z = 2, R = 0.0372 [I ≧ 2σ(I)], Rw = 0.0921 (all data), GOF = 1.059.
, a = 9.41147(18) Å, b = 14.0313(3) Å, c = 16.8034(3) Å, α = 110.8961(17)°, β = 94.6483(15)°, γ = 100.4507(16)°, Z = 2, R = 0.0372 [I ≧ 2σ(I)], Rw = 0.0921 (all data), GOF = 1.059.
AuNCPAuCl3 (CCDC 2394677): C45.5H30Au2Cl6N4, Mr = 1239.37, monoclinic, C2/c, a = 19.8854(4) Å, b = 27.5952(6) Å, c = 8.02367(19) Å, β = 96.673(2)°, Z = 4, R = 0.0850 [I ≧ 2σ(I)], Rw = 0.2318 (all data), GOF = 1.119.
AgICCIrCp*Cl2 (CCDC 2394665): C71H58AgCl2IrN5, Mr = 1352.19, triclinic, P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) , a = 12.6740(2) Å, b = 15.7863(2) Å, c = 16.6020(2) Å, α = 113.853(3)°, β = 99.635(2)°, γ = 101.492(2)°, Z = 2, R = 0.0230 [I ≧ 2σ(I)], Rw = 0.0593 (all data), GOF = 1.046.
, a = 12.6740(2) Å, b = 15.7863(2) Å, c = 16.6020(2) Å, α = 113.853(3)°, β = 99.635(2)°, γ = 101.492(2)°, Z = 2, R = 0.0230 [I ≧ 2σ(I)], Rw = 0.0593 (all data), GOF = 1.046.
ICC(BOPh)3 (CCDC 2394671): C130H95B6N10O6Cl4, Mr = 2099.81, triclinic, P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) , a = 17.8280(3) Å, b = 17.84840(18) Å, c = 18.3446(2) Å, α = 73.5280(10)°, β = 71.5735(12)°, γ = 88.9914(11)°, Z = 2, R = 0.0499 [I ≧ 2σ(I)], Rw = 0.1376 (all data), GOF = 1.028.
, a = 17.8280(3) Å, b = 17.84840(18) Å, c = 18.3446(2) Å, α = 73.5280(10)°, β = 71.5735(12)°, γ = 88.9914(11)°, Z = 2, R = 0.0499 [I ≧ 2σ(I)], Rw = 0.1376 (all data), GOF = 1.028.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: additional spectroscopic, mass spectrometric, crystallographic, and electrochemical data. See DOI: https://doi.org/10.1039/d4dt03034f.CCDC 2394665, 2394666, 2394670, 2394671 and 2394677 contain the supplementary crystallographic data for this paper.83a–e
Acknowledgements
This work was supported by the Polish National Science Center (2022/45/B/ST4/01229).References
- P. J. Chmielewski, L. Latos-Grażyński, K. Rachlewicz and T. Głowiak, Angew. Chem., Int. Ed. Engl., 1994, 33, 779–781 CrossRef.
- H. Furuta, T. Asano and T. Ogawa, J. Am. Chem. Soc., 1994, 116, 767–768 CrossRef CAS.
- G. R. Geier III, D. M. Haynes and J. S. Lindsey, Org. Lett., 1999, 1, 1455–1458 CrossRef PubMed.
- L. Latos-Grażyński and P. J. Chmielewski, New J. Chem., 1997, 21, 691–700 Search PubMed.
- M. Toganoh and H. Furuta, Chem. Rev., 2022, 122, 8313–8437 CrossRef CAS PubMed.
- M. J. Białek, K. Hurej, H. Furuta and L. Latos-Grażyński, Chem. Soc. Rev., 2023, 52, 2083–2144 RSC.
- J. D. Harvey and C. J. Ziegler, Coord. Chem. Rev., 2003, 247, 1–19 CrossRef CAS.
- P. J. Chmielewski and L. Latos-Grażyński, Coord. Chem. Rev., 2005, 249, 2510–2533 CrossRef CAS.
- H. Furuta, H. Maeda and A. Osuka, Chem. Commun., 2002, 1795–1804 RSC.
- H. Furuta, T. Ishizuka, A. Osuka, Y. Uwatoko and Y. Ishikawa, Angew. Chem., Int. Ed., 2001, 40, 2323–2325 CrossRef CAS PubMed.
- A. Srinivasan, H. Furuta and A. Osuka, Chem. Commun., 2001, 1666–1667 RSC.
- H. Furuta, K. Youfu, H. Maeda and A. Osuka, Angew. Chem., Int. Ed., 2003, 42, 2186–2188 CrossRef CAS PubMed.
- H. Maeda, Y. Ishikawa, H. Matsueda, A. Osuka and H. Furuta, J. Am. Chem. Soc., 2003, 125, 11822–11823 CrossRef CAS PubMed.
- H. Furuta, T. Morimoto and A. Osuka, Inorg. Chem., 2004, 43, 1618–1624 CrossRef CAS PubMed.
- J.-C. Liu, T. Ishizuka, A. Osuka and H. Furuta, Chem. Commun., 2004, 1908–1909 Search PubMed.
- M. Toganoh, T. Ishizuka and H. Furuta, Chem. Commun., 2004, 2464–2465 RSC.
- T. Morimoto, S. Taniguchi, A. Osuka and H. Furuta, Eur. J. Org. Chem., 2005, 3887–3890 CrossRef CAS.
- M. Toganoh, J. Konagawa and H. Furuta, Inorg. Chem., 2006, 45, 3852–3854 CrossRef CAS PubMed.
- T. Niino, M. Toganoh, B. Andrioletti and H. Furuta, Chem. Commun., 2006, 4335–4337 RSC.
- M. Toganoh, T. Niino, H. Maeda, B. Andrioletti and H. Furuta, Inorg. Chem., 2006, 45, 10428–10430 CrossRef CAS PubMed.
- Y. Xie, T. Morimoto and H. Furuta, Angew. Chem., Int. Ed., 2006, 45, 6907–6910 CrossRef CAS PubMed.
- A. Srinivasan, M. Toganoh, T. Niino, A. Osuka and H. Furuta, Inorg. Chem., 2008, 47, 11305–11313 CrossRef CAS PubMed.
- M. Toganoh, T. Niino and H. Furuta, Chem. Commun., 2008, 4070–4072 RSC.
- D.-H. Won, M. Toganoh, H. Uno and H. Furuta, Dalton Trans., 2009, 6151–6158 RSC.
- T. Yamamoto, M. Toganoh and H. Furuta, Dalton Trans., 2012, 41, 9154–9157 RSC.
- T. Yamamoto, M. Toganoh, S. Mori, H. Uno and H. Furuta, Chem. Sci., 2012, 3, 3241–3248 RSC.
- T. Miyazaki, T. Yamamot, S. Mashita, Y. Deguchi, K. Fukuyama, M. Ishida, S. Mori and H. Furuta, Eur. J. Inorg. Chem., 2018, 203–207 CrossRef CAS.
- T. Yamamoto, K. Mitsuno, S. Mori, S. Itoyama, Y. Shiota, K. Yoshizawa, M. Ishida and H. Furuta, Chem. – Eur. J., 2018, 24, 6742–6746 CrossRef CAS PubMed.
- T. Miyazaki, K. Fukuyama, S. Mashita, Y. Deguchi, T. Yamamot, M. Ishida, S. Mori and H. Furuta, ChemPlusChem, 2019, 84, 603–607 CrossRef CAS PubMed.
- O. Iwanaga, K. Fukuyama, S. Mori, J. T. Song, T. Ishihara, T. Miyazaki, M. Ishida and H. Furuta, RSC Adv., 2021, 11, 24575–24579 RSC.
- H. Furuta, T. Ogawa, Y. Uwatoko and K. Araki, Inorg. Chem., 1999, 38, 2676–2682 CrossRef CAS.
- H. Furuta, T. Ishizuka and A. Osuka, J. Am. Chem. Soc., 2002, 124, 5622–5623 CrossRef CAS PubMed.
- T. Morimoto, H. Uno and H. Furuta, Angew. Chem., Int. Ed., 2007, 46, 3672–3675 CrossRef CAS PubMed.
- J. D. Harvey and C. J. Ziegler, Chem. Commun., 2002, 1942–1943 RSC.
- C.-H. Hung, W.-C. Chen, G.-H. Lee and S.-M. Peng, Chem. Commun., 2002, 1516–1517 RSC.
- J. Wojaczyński, J. Maciołek and P. J. Chmielewski, Chem. – Asian J., 2017, 12, 643–647 CrossRef PubMed.
- P. J. Chmielewski, Angew. Chem., Int. Ed., 2005, 44, 6417–6420 CrossRef CAS PubMed.
- H. Furuta, N. Kubo, H. Maeda, T. Ishizuka, A. Osuka, H. Nanami and T. Ogawa, Inorg. Chem., 2000, 39, 5424–5425 CrossRef CAS PubMed.
- P. J. Chmielewski and I. Schmidt, Inorg. Chem., 2004, 43, 1885–1894 CrossRef CAS PubMed.
- P. J. Chmielewski, B. Durlej, M. Siczek and L. Szterenberg, Angew. Chem., Int. Ed., 2009, 48, 8736–8739 CrossRef CAS PubMed.
- S. Koniarz, K. Szydełko, M. J. Białek, K. Hurej and P. J. Chmielewski, Adv. Sci., 2024, 11, 2306696/1–2306696/15 Search PubMed.
- J.-F. Longevial, S. Clément, J. A. Wytko, R. Ruppert, J. Weiss and S. Richeter, Chem. – Eur. J., 2018, 24, 15442–15460 CrossRef CAS PubMed.
- M. J. Crossley and J. A. McDonald, J. Chem. Soc., Perkin Trans. 1, 1999, 2429–2431 RSC.
- J.-F. Lefebvre, D. Leclercq, J.-P. Gisselbrecht and S. Richeter, Eur. J. Org. Chem., 2010, 1912–1920 CrossRef CAS.
- M. Lo, J.-F. Lefebvre, D. Leclerq, A. van der Lee and S. Richeter, Org. Lett., 2011, 13, 3110–3113 CrossRef CAS PubMed.
- J.-F. Lefebvre, M. Lo, D. Leclercq and S. Richeter, Chem. Commun., 2011, 14, 2976–2978 RSC.
- J.-F. Lefebvre, M. Lo, J.-P. Gisselbrecht, O. Coulembier, S. Clément and S. Richeter, Chem. – Eur. J., 2013, 19, 15652–15660 CrossRef CAS PubMed.
- M. Abdelhameed, P.-L. Karsenti, A. Langlois, J.-F. Lefebvre, S. Richeter, R. Ruppert and P. D. Harvey, Chem. – Eur. J., 2014, 20, 12988–13001 CrossRef CAS PubMed.
- J.-F. Longevial, A. Langlois, A. Buisson, C. H. Devillers, S. Clément, A. van der Lee, P. D. Harvey and S. Richeter, Organometallics, 2016, 35, 663–672 CrossRef CAS.
- J.-F. Lefebvre, J.-F. Longevial, K. Molvinger, S. Clément and S. Richeter, C. R. Chim., 2016, 19, 94–102 CrossRef CAS.
- Y. Kashiwagi, K. Ohkubo, J. A. McDonald, I. M. Blake, M. J. Crossley, Y. Araki, O. Ito, H. Imahor and S. Fukuzumi, Org. Lett., 2003, 5, 2719–2721 CrossRef CAS PubMed.
- G. B. Bodedla, H. Wang, S. Chang, S. Chen, T. Chen, J. Zhao, W.-K. Wong and X. Zhu, ChemistrySelect, 2018, 3, 2558–2564 CrossRef CAS.
- S. Richeter, A. Hadj-Aissa, C. Taffin, A. van der Lee and D. Leclercq, Chem. Commun., 2007, 2148–2150 RSC.
- S. Shimizu, Chem. Rev., 2017, 117, 2730–2784 CrossRef CAS PubMed.
- Z. Li, L. Zhang, Q. Wu, H. Li, Z. Kang, C. Yu, E. Hao and L. Jiao, J. Am. Chem. Soc., 2022, 144, 6692–6697 CrossRef CAS PubMed.
- W. J. Belcher, M. Breede, P. J. Brothers and C. E. F. Rickard, Angew. Chem., Int. Ed., 1998, 37, 1112 CrossRef CAS PubMed.
- A. Weiss, H. Pritzkow, P. J. Brothers and W. Siebert, Angew. Chem., Int. Ed., 2001, 40, 4182–4184 CrossRef CAS PubMed.
- P. J. Brothers, Inorg. Chem., 2011, 50, 12374–12386 CrossRef CAS PubMed.
- A. M. Albrett, K. E. Thomas, S. Maslek, A. Młodzianowska, J. Conradie, C. M. Beavers, A. Ghosh and P. J. Brothers, Inorg. Chem., 2014, 53, 5486–5493 CrossRef CAS PubMed.
- W. J. Belcher, M. C. Hodgson, K. Sumida, A. Torvisco, K. Ruhlandt-Senge, D. C. Ware, P. D. W. Boyd and P. J. Brothers, Dalton Trans., 2008, 1602–1614 RSC.
- M. R. Rao and M. Ravikanth, J. Org. Chem., 2011, 76, 3582–3587 CrossRef PubMed.
- T. K. Khan, M. Broering, S. Manthur and M. Ravikanth, Coord. Chem. Rev., 2013, 257, 2348–2387 CrossRef CAS.
- A. Młodzianowska, L. Latos-Grażyński, L. Szterenberg and M. Stępień, Inorg. Chem., 2007, 46, 6950–6957 CrossRef PubMed.
- J. Beckmann, D. Dakternieks, A. Duthie, A. E. K. Lim and E. R. T. Tiekink, J. Organomet. Chem., 2001, 633, 149–156 CrossRef CAS.
- M. C. Haberecht, M. Bolte, M. Wagner and H.-W. Lerner, J. Chem. Crystallogr., 2005, 35, 657–665 CrossRef CAS.
- C. P. Brock, R. P. Minton and K. Niedenzu, Acta Crystallogr., Sect. C: Cryst. Struct. Commun., 1987, 43, 1775–1779 CrossRef.
- J. Krumsieck and M. Bröring, Chem. – Eur. J., 2021, 27, 11580–11588 CrossRef CAS PubMed.
- W. Jentzen, X.-Z. Song and J. A. Shelnutt, J. Phys. Chem. B, 1997, 101, 1684–1699 CrossRef CAS.
- W. Jentzen, J.-G. Ma and J. A. Shelnutt, Biophys. J., 1998, 74, 753–763 CrossRef CAS PubMed.
- M. A. Beckett, G. C. Strickland, K. S. Varma, D. E. Hibbs, M. B. Hursthouse and K. M. A. Malik, J. Organomet. Chem., 1997, 535, 33–41 CrossRef CAS.
- Q. G. Wu, G. Wu, L. Brancaleon and S. Wang, Organometallics, 1999, 18, 2553–2556 CrossRef CAS.
- Y.-J. Wang, A.-Q. Jia, X.-S. Chen, H.-T. Shi and Q.-F. Zhang, J. Chem. Crystallogr., 2015, 45, 284–289 CrossRef CAS.
- M. A. Beckett, D. E. Hibbs, M. B. Hursthouse, P. Owen, K. M. A. Malik and K. S. Varma, Main Group Chem., 1998, 2, 251–258 CrossRef CAS.
- D. Ren, S. Koniarz, X. Li and P. J. Chmielewski, Chem. Commun., 2020, 56, 4856–4839 RSC.
- H. Furuta, T. Ishizuka, A. Osuka and T. Ogawa, J. Am. Chem. Soc., 1999, 121, 2945–2946 CrossRef CAS.
- M. R. Churchill and S. A. Julis, Inorg. Chem., 1977, 16, 1488–1494 CrossRef CAS.
- K. M. Kadish, W. E, Z. Ou, J. Shao, P. J. Sintic, K. Ohkubo, S. Fukuzumi and M. J. Crossley, Chem. Commun., 2002, 356–357 RSC.
- E. Rabinovich, I. Goldberg and Z. Gross, Chem. – Eur. J., 2011, 11, 12294–12301 CrossRef PubMed.
- G. M. Sheldrick, Acta Crystallogr., Sect. A:Found. Adv., 2015, 71, 3–8 CrossRef PubMed.
- G. M. Sheldrick, Acta Crystallogr., Sect. C: Struct. Chem., 2015, 71, 3–8 Search PubMed.
- O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard and H. Puschmann, J. Appl. Crystallogr., 2009, 42, 339–341 CrossRef CAS.
- A. L. Spek, Acta Crystallogr., Sect. C: Struct. Chem., 2015, 71, 9–18 CrossRef CAS PubMed.
- (a) CCDC 2394665: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2lcv83; (b) CCDC 2394666: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2lcv94; (c) CCDC 2394670: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2lcvf8; (d) CCDC 2394671: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2lcvg9; (e) CCDC 2394677: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2lcvnh.
Footnote |
| † 1H NMR spectra of certain fractions collected upon the column chromatography of AgICCAuCl comprised, apart from the resonances of the silver(III) complex, another set of the weaker signals (about 20% of the integral intensity of the major component) that may be assigned to the AuICCAuCl or AuICC. |
| This journal is © The Royal Society of Chemistry 2025 |