Open Access Article
Open Access ArticleDirect experimental verification of negative deviations from ideal mixing in an archetypal deep eutectic solvent
Francesco I.
Travaglini
a,
Lorenza
Romagnoli
 *a,
Angelika M.
Czerska
a,
Matteo
Busato
*a,
Angelika M.
Czerska
a,
Matteo
Busato
 a,
Giorgia
Mannucci
a,
Giorgia
Mannucci
 a,
Stefano
Vecchio Ciprioti
a,
Stefano
Vecchio Ciprioti
 b,
Paola
D’Angelo
b,
Paola
D’Angelo
 a and
Andrea
Ciccioli
a and
Andrea
Ciccioli
 *a
*a
aDipartimento di Chimica, Sapienza Università di Roma, P.le Aldo Moro 5, I-00185 Rome, Italy. E-mail: andrea.ciccioli@uniroma1.it; lorenza.romagnoli@uniroma1.it
bDipartimento S.B.A.I., Sapienza Università di Roma, via del Castro Laurenziano 7, 00161 Rome, Italy
First published on 6th November 2025
Abstract
The thermodynamic activity of urea in the liquid solution of the urea–choline chloride DES at the eutectic composition was measured for the first time through direct tensimetric experiments using Knudsen effusion mass spectrometry. In the explored temperature range (319–343 K), the urea activity coefficients (with respect to pure liquid urea) were found to lie in the 0.25–0.44 range, thus indicating a clear negative deviation from the Raoultian behavior. From the temperature dependence of activity, evidence for negative values of partial enthalpy and entropy of mixing was found, indicating strong attractive interactions between urea and choline chloride, with a significant ordering of urea molecules compared to pure liquid urea.
Negative deviation of the liquid phase from the Raoultian ideal behavior has been claimed as the fundamental property leading to the phenomenon of deep eutectic temperatures in the so-called DESs (deep eutectic solvents), which in the last few years have received an outburst of interest in view of their many potential technological applications. Coutinho et al.1 pointed out the key role played by extra-stabilization of the liquid phase in distinguishing DESs from ordinary eutectic systems. However, despite the inherently thermodynamic origin of DES behavior, the investigation of this extra-stabilization in terms of classic experimental thermodynamics has surprisingly received little attention. This phenomenon has been investigated at a molecular level by computational or spectroscopic approaches, focusing on hydrogen bonds and intermolecular forces in the liquid,2–6 rather than by macroscopic thermodynamic measurements. In a recent paper,7 M. Costa-Gomez stated that “the link between the microscopic and macroscopic description of the liquid phase of DESs is missing […]. To date, no experimental thermodynamics studies quantifying the balance between molecular interactions and liquid disorder were reported, while they could provide the missing link for a rational design of DESs for target applications”. In the most recent literature, a few calorimetric papers have appeared, focusing on the enthalpic portion of the Gibbs energy of mixing,8–10 and activity values have been estimated from the experimentally measured liquidus curves.9 However, small inaccuracies in the shape of the liquidus can lead to serious errors in the estimation of activities. Furthermore, this procedure requires accurate values of the melting properties of the pure components, which is not always the case, as shown, for example, by the accurate re-determination of the enthalpy of fusion of choline chloride.11 As for the experimental determination of thermodynamic activity, a grave lack of information emerges from analysis of the existing literature. The limited data currently available concern the activities of water12 or other molecular solutes in binary DESs (generally for extraction purposes) determined using chromatographic techniques.13 Headspace gas chromatography mass spectrometry has also been used to determine the activity coefficients of components in hydrophobic DES systems.14
The most direct method to determine the deviation from ideal behavior for each component is to measure its pressure in the vapors equilibrated with the mixture (pi) and with the pure phase 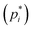 of that component, respectively, thus obtaining the activity
of that component, respectively, thus obtaining the activity 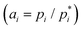 . To the best of our knowledge, no direct measurement of activity has been reported for any DES, including the most archetypal one formed by urea and choline chloride (choline chloride = ChCl in the following). In this communication, we report the results of tensimetric measurements using the Knudsen Effusion Mass Spectrometry (KEMS) technique15,16 on the urea–ChCl liquid mixture at the eutectic composition (urea mole fraction xurea = 0.67) and on pure urea in the temperature range of 319–343 K, well above the invariant temperature (305 K).17 Experimental details are reported in the SI. Here, we note that the use of an effusion cell is expected to assure the attainment of a close-to-equilibrium condition of the heterogeneous liquid–vapor system, unlike open pan (free evaporation) conditions often used in thermal analysis experiments. Furthermore, KEMS identifies individual species in the vapor phase, and in particular water, which is known to markedly affect DES properties18 and can lead to serious errors in the vapor pressures obtained with mass-loss-based techniques. Finally, the high sensitivity of the technique permits the evaporation experiments to be performed at relatively low temperatures, where thermal decomposition is unlikely to occur and the loss of the volatile component is low enough to limit the compositional change of the liquid.
. To the best of our knowledge, no direct measurement of activity has been reported for any DES, including the most archetypal one formed by urea and choline chloride (choline chloride = ChCl in the following). In this communication, we report the results of tensimetric measurements using the Knudsen Effusion Mass Spectrometry (KEMS) technique15,16 on the urea–ChCl liquid mixture at the eutectic composition (urea mole fraction xurea = 0.67) and on pure urea in the temperature range of 319–343 K, well above the invariant temperature (305 K).17 Experimental details are reported in the SI. Here, we note that the use of an effusion cell is expected to assure the attainment of a close-to-equilibrium condition of the heterogeneous liquid–vapor system, unlike open pan (free evaporation) conditions often used in thermal analysis experiments. Furthermore, KEMS identifies individual species in the vapor phase, and in particular water, which is known to markedly affect DES properties18 and can lead to serious errors in the vapor pressures obtained with mass-loss-based techniques. Finally, the high sensitivity of the technique permits the evaporation experiments to be performed at relatively low temperatures, where thermal decomposition is unlikely to occur and the loss of the volatile component is low enough to limit the compositional change of the liquid.
Ideal liquid solutions are known to be characterized by the following properties for all the components at all temperatures and pressures: ΔmixHi = 0, ΔmixSi = −R![[thin space (1/6-em)]](https://www.rsc.org/images/entities/i_char_2009.gif) ln
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) xi (independent from mole fractions other than that of i), ai = xi (for all components), and ΔmixGi = RT
xi (independent from mole fractions other than that of i), ai = xi (for all components), and ΔmixGi = RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) xi < 0 (mixing driven only by entropy). In this case, the liquidus curves and their intersection at the eutectic composition and temperature are entirely determined by the melting properties of the pure components. Conversely, in solutions displaying deviations from Raoultian behavior, ΔmixHi ≠ 0, ΔmixSi ≠ −R
xi < 0 (mixing driven only by entropy). In this case, the liquidus curves and their intersection at the eutectic composition and temperature are entirely determined by the melting properties of the pure components. Conversely, in solutions displaying deviations from Raoultian behavior, ΔmixHi ≠ 0, ΔmixSi ≠ −R![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) xi and the activity differs from the molar fraction, ai = γixi, where γi is the activity coefficient, which depends on composition, temperature and pressure, with γi < 1 and γi > 1 for negative (ΔmixGexcessi = RT
xi and the activity differs from the molar fraction, ai = γixi, where γi is the activity coefficient, which depends on composition, temperature and pressure, with γi < 1 and γi > 1 for negative (ΔmixGexcessi = RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) γi < 0) and positive (ΔmixGexcessi > 0) deviations of the given component, respectively. Negative deviations indicate that the component i has an extra-stabilization as compared to the purely entropic random mixing term. Generally speaking, this stabilization can be due to enthalpic and/or entropic effects. While negative deviations in DESs are often ascribed to energetic/enthalpic effects,1 a significant role of entropy is currently under examination in the theoretical and experimental literature.9,19–22
γi < 0) and positive (ΔmixGexcessi > 0) deviations of the given component, respectively. Negative deviations indicate that the component i has an extra-stabilization as compared to the purely entropic random mixing term. Generally speaking, this stabilization can be due to enthalpic and/or entropic effects. While negative deviations in DESs are often ascribed to energetic/enthalpic effects,1 a significant role of entropy is currently under examination in the theoretical and experimental literature.9,19–22
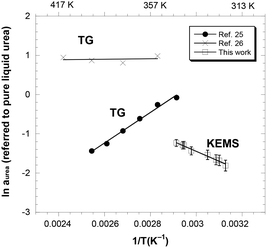
In this work, a series of alternate KEMS experiments was conducted to evaluate the activity of urea by measuring the saturated pressure of pure solid urea and the liquid urea–ChCl mixture at its eutectic composition, in the temperature range of 319–343 K. As detailed in the SI, the only species observed in the mass spectrum was gaseous urea,23 and the ratio of mass spectrometric ion intensities at m/z = 60 in the two experiments provided a direct measure of the activity of urea in the mixture, with pure solid urea taken as the reference phase. The values thus obtained are reported in the second column of Table 1. From these values, we could estimate the saturation vapor pressures of the solution by using the vapor pressures of pure solid urea given in ref. 24 [ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) p(pure urea)/Pa = 29.919 − 10922/T(K)] in the temperature range of our experiments. The resulting values are reported in the last column of Table 1 and are compared in Fig. 1 with the two series of values available in the literature,25,26 both measured by isothermal thermogravimetry (TG) under free evaporation conditions, with a computational value,27 and with the vapor pressure of pure urea.24 While the temperature range covered in our experiments is lower than that explored by other authors, it seems evident that both the absolute pressure values and the temperature trend of the three series of data are not in agreement. Note that the slopes of the regression lines calculated from the data in ref. 25 and 26 (46.9 ± 1.5 kJ mol−1 and 79.0 ± 2.7 kJ mol−1, at T = 367 K and 382 K, respectively) are at variance with each other and are significantly lower compared to our value (96.3 ± 1.6 kJ mol−1 at 331 K) and to the sublimation enthalpy of pure urea24 (94.9 kJ mol−1 ± 0.9 kJ mol−1 at 369 K). However, at the only temperature where our range overlaps with that of ref. 25, the KEMS pressures are lower by a factor of 3, revealing an order-of-magnitude consistency. Possible sources of inaccuracies in TG measurements include water impurities and uncertainties in the mixture composition, as stated by the authors themselves.26 Meanwhile, KEMS results are sensitive to imperfect reproduction of the experimental conditions in the consecutive experiments on urea and DES and to possible uncertainties in the final composition of the mixture.
p(pure urea)/Pa = 29.919 − 10922/T(K)] in the temperature range of our experiments. The resulting values are reported in the last column of Table 1 and are compared in Fig. 1 with the two series of values available in the literature,25,26 both measured by isothermal thermogravimetry (TG) under free evaporation conditions, with a computational value,27 and with the vapor pressure of pure urea.24 While the temperature range covered in our experiments is lower than that explored by other authors, it seems evident that both the absolute pressure values and the temperature trend of the three series of data are not in agreement. Note that the slopes of the regression lines calculated from the data in ref. 25 and 26 (46.9 ± 1.5 kJ mol−1 and 79.0 ± 2.7 kJ mol−1, at T = 367 K and 382 K, respectively) are at variance with each other and are significantly lower compared to our value (96.3 ± 1.6 kJ mol−1 at 331 K) and to the sublimation enthalpy of pure urea24 (94.9 kJ mol−1 ± 0.9 kJ mol−1 at 369 K). However, at the only temperature where our range overlaps with that of ref. 25, the KEMS pressures are lower by a factor of 3, revealing an order-of-magnitude consistency. Possible sources of inaccuracies in TG measurements include water impurities and uncertainties in the mixture composition, as stated by the authors themselves.26 Meanwhile, KEMS results are sensitive to imperfect reproduction of the experimental conditions in the consecutive experiments on urea and DES and to possible uncertainties in the final composition of the mixture.
| T/K | a urea (DES, wrt (s)) | P sat,urea(DES)/Pa |
|---|---|---|
| 319.7 | 0.60 | 0.0086 |
| 322.5 | 0.64 | 0.012 |
| 324.0 | 0.63 | 0.014 |
| 327.9 | 0.66 | 0.022 |
| 335.7 | 0.64 | 0.047 |
| 339.4 | 0.69 | 0.072 |
| 340.0 | 0.68 | 0.075 |
| 343.3 | 0.69 | 0.10 |
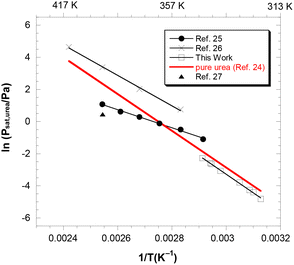 | ||
| Fig. 1 Vapor pressure of urea in the liquid urea–choline chloride mixture vapors (xurea = 0.67) from the present work and the literature (ref. 25 and 26: isothermal thermogravimetry; ref. 27: Monte Carlo simulations). The red line is the vapor pressure of pure solid urea.24 Vertical axis: ln (Psat,urea in Pa); bottom horizontal axis: reciprocal temperature in K−1; top horizontal axis: temperature in K. | ||
In order to properly evaluate the deviation from Raoultian behavior, the activity values in Table 1 were recalculated by shifting the reference phase from pure solid urea to pure liquid urea by the factor exp (−ΔlcrG/RT), where ΔlcrG is the (positive) Gibbs energy of melting of urea28,29 at the experimental temperature. The procedure used to estimate this property is detailed in the SI. The resulting activity and activity coefficient values with respect to liquid urea are reported in the second and third columns of Table 2, respectively, and show a clear, albeit not large, negative deviation from the ideal Raoultian behavior. The partial Gibbs energies of mixing derived from the activities are also listed in Table 2.
| T/K | a urea (DES, wrt (l)) | γ urea (DES, wrt (l)) | ΔmixGureaa (J mol−1) |
|---|---|---|---|
| a Uncertainty estimated as discussed in the SI. | |||
| 319.7 | 0.16 ± 0.023 | 0.25 ± 0.023 | −4811 ± 370 |
| 322.5 | 0.19 ± 0.024 | 0.28 ± 0.024 | −4499 ± 350 |
| 324.0 | 0.19 ± 0.024 | 0.28 ± 0.024 | −4477 ± 340 |
| 327.9 | 0.22 ± 0.026 | 0.32 ± 0.026 | −4185 ± 330 |
| 335.7 | 0.24 ± 0.025 | 0.34 ± 0.025 | −4000 ± 290 |
| 339.4 | 0.28 ± 0.027 | 0.41 ± 0.027 | – 3637 ± 270 |
| 340.0 | 0.28 ± 0.026 | 0.41 ± 0.026 | −3649 ± 270 |
| 343.3 | 0.29 ± 0.026 | 0.44 ± 0.026 | −3515 ± 260 |
Deviations from ideal behavior are known to arise from enthalpic factors, entropic factors or both. In particular, negative enthalpy of mixing or positive entropy of mixing exceeding the random mixing term −R![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) xi can drive towards this behavior. To dissect the two contributions, the trend of activity with temperature was analyzed, according to the equations:
xi can drive towards this behavior. To dissect the two contributions, the trend of activity with temperature was analyzed, according to the equations:
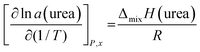 | (1) |
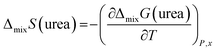 | (2) |
The first equation implies a negative enthalpy of mixing for a decreasing ln a vs. 1/T trend. The activity values determined by KEMS (see Table 2) are graphically displayed in this form in Fig. 2 (open squares), indicating a negative partial enthalpy of mixing of urea in the temperature range covered in our experiments (average temperature T = 331 K, taken as the harmonic mean of individual temperatures). The physical meaning of this result is that the state of urea inside the liquid solution is enthalpically favored, compared to pure liquid urea at the same temperature. This can be interpreted on the molecular scale as due to the more effective interactions established between urea and ChCl, compared to urea–urea interactions at play in the pure liquid. This result is consistent both with the more widely accepted view that strong urea⋯Cl contacts via the urea amino groups dominate the enthalpic stabilization of the liquid phase, and with alternative models suggesting that Ch⋯urea interactions may play a competitive role.6,30–32 Indeed, both scenarios imply ChCl⋯urea association as more favorable than weaker urea⋯urea interactions occurring in the pure liquid. Many other hydrogen bonds deriving from Ch⋯urea, Ch⋯Ch, and urea⋯urea contacts are also known to exist in the liquid mixture.6,33 While a weaker ChCl⋯urea interaction would hinder component mixing, the role of these soft contacts has been interpreted as making the hydrogen bond network less rigid, thereby preventing the components from organizing into a co-crystal.30–32 Our overall outcome is still consistent with the picture of DES formation as governed by a delicate balance between these opposite effects.
The enthalpy-driven deviation from ideality resulting from our experiments seems to confirm the usual view on the enthalpic origin of type-III DESs.20 Interestingly, recently available calorimetric measurements on water–ChCl, ethylene glycol–ChCl, and 1,3-propanediol-ChCl DESs point towards an enthalpy-dominated Gibbs energy excess for the first system, and an entropy-dominated one for the other two. While the markedly decreasing temperature trend of activity in Fig. 2 provides clear evidence for the occurrence of a negative partial enthalpy of mixing, the low number of data points and the narrow temperature range make the quantitative evaluation of ΔmixHurea questionable. Unfortunately, the temperature range could not be extended due to (i) detection limits at the lower end and (ii) breakdown of regular evaporation/effusion behavior at the highest one. Despite these limitations, we performed a tentative weighted least-squares fitting of the data, leading to a provisional value of ΔmixHurea = −21 ± 4 kJ mol−1, where the uncertainty was estimated as detailed in the SI.
In Fig. 2, the activity values calculated by us from the aforementioned vapor pressure data retrieved from the literature25,26 are also reported (to this end, the same procedure used to calculate the activity from KEMS vapor pressure data, detailed in the SI, was applied). Incidentally, it is worth noting that this figure synoptically displays the huge degree of uncertainty from which the current knowledge of such a crucial property of typical DESs suffers. As expected from the vapor pressure data displayed in Fig. 1, our results are in disagreement with those of ref. 26, which would provide activity values close to unity with a practically flat temperature trend. As for the data of ref. 25, the activities would imply a much less marked negative deviation as compared to the KEMS results and, more importantly, the increasing temperature trend would suggest a pronounced positive partial mixing enthalpy of urea (exceeding 30 kJ mol−1). Obviously, this positive enthalpy would imply a highly positive partial entropic term accounting for negative values of partial mixing Gibbs energies.
Finally, by considering the temperature dependence of the partial Gibbs energies of mixing of urea (Table 2), an attempt to estimate the partial entropy of mixing (eqn (2)) was also made, resulting in a tentative value of ΔmixSurea = −51 ± 12 J K−1 mol−1. By subtracting the partial contribution due to random mixing (−R![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.67), an excess entropy of −54 ± 12 J K−1 mol−1 is found, which suggests a significant ordering of urea molecules with respect to pure liquid urea, driven by the coordination to the ionic component, with the latter acting as a “structure-making” solute, to borrow the expression commonly adopted for ions in aqueous solutions.34 We note that a pronounced ordering effect associated with mixing urea and ChCl has been clearly documented by neutron scattering and atomistic modeling, and attributed to a reduction in the configurational freedom of urea molecules upon strong association with ChCl compared to the pure liquid state.6 The present work seems to provide, for the first time, an experimental thermodynamic counterpart of this picture. In comparing the outcomes of our work with theoretical results, it must be taken into account that the present results refer only to the partial contribution of urea. Obviously, the overall mixing properties are also affected by the partial contribution of ChCl, which is not accessible by the experiments reported here, because the partial pressure of ChCl is much below the lower detection limit at the temperatures explored in these experiments. Moreover, at temperatures where the ChCl vapor pressure would become measurable, the urea partial pressure would definitely be too high for the molecular effusion regime to be maintained. In general, the simultaneous determination of the activities of both components by tensimetric techniques is hardly feasible for type-III DESs, due to the expected much lower volatility of the ionic component. However, an attempt may be made to speculate on the impact of ChCl on the overall mixing entropy. Previous inelastic neutron scattering studies have shown that chloride anions in the urea–ChCl mixture are displaced from the preferred positions atop the Ch methyl groups, as found in crystalline ChCl.30 In addition, reports highlighting the role of cation asymmetry and torsional movement of the C–C bond in deviations from ideality,20,30 suggest a positive contribution of ChCl to the entropy of mixing. This would contrast the negative ΔmixSurea, which reflects the reduced configurational freedom of urea molecules in the mixed state. Nevertheless, the lack of experimental information on the activity of ChCl makes this interpretation tentative.
0.67), an excess entropy of −54 ± 12 J K−1 mol−1 is found, which suggests a significant ordering of urea molecules with respect to pure liquid urea, driven by the coordination to the ionic component, with the latter acting as a “structure-making” solute, to borrow the expression commonly adopted for ions in aqueous solutions.34 We note that a pronounced ordering effect associated with mixing urea and ChCl has been clearly documented by neutron scattering and atomistic modeling, and attributed to a reduction in the configurational freedom of urea molecules upon strong association with ChCl compared to the pure liquid state.6 The present work seems to provide, for the first time, an experimental thermodynamic counterpart of this picture. In comparing the outcomes of our work with theoretical results, it must be taken into account that the present results refer only to the partial contribution of urea. Obviously, the overall mixing properties are also affected by the partial contribution of ChCl, which is not accessible by the experiments reported here, because the partial pressure of ChCl is much below the lower detection limit at the temperatures explored in these experiments. Moreover, at temperatures where the ChCl vapor pressure would become measurable, the urea partial pressure would definitely be too high for the molecular effusion regime to be maintained. In general, the simultaneous determination of the activities of both components by tensimetric techniques is hardly feasible for type-III DESs, due to the expected much lower volatility of the ionic component. However, an attempt may be made to speculate on the impact of ChCl on the overall mixing entropy. Previous inelastic neutron scattering studies have shown that chloride anions in the urea–ChCl mixture are displaced from the preferred positions atop the Ch methyl groups, as found in crystalline ChCl.30 In addition, reports highlighting the role of cation asymmetry and torsional movement of the C–C bond in deviations from ideality,20,30 suggest a positive contribution of ChCl to the entropy of mixing. This would contrast the negative ΔmixSurea, which reflects the reduced configurational freedom of urea molecules in the mixed state. Nevertheless, the lack of experimental information on the activity of ChCl makes this interpretation tentative.
Conclusions
The partial mixing properties of urea in the urea–ChCl liquid mixture at the eutectic composition xurea = 0.67 were determined by direct tensimetric measurements of urea activity for the first time. The results showed an unambiguous negative deviation of urea from random mixing Raoultian behavior, with activity coefficients ranging in the interval 0.25–0.44 in the 319–343 K temperature range. From the temperature trend of the activity, provisional evidence was found for a pronounced enthalpic contribution to non-ideality and for a negative entropic term mitigating the enthalpic factor. Further measurements will be carried out in the future to confirm the latter findings and to achieve a full thermodynamic description of the urea mixing properties in this and other type-III DESs, mapping both the temperature and the composition dependence of the activity coefficient in the liquid phase. This work is intended to pave the way for a connection between the still-detached molecular-level and macroscopic-level descriptions of DESs.Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.Supplementary information: experimental details, shifting of the reference state and estimate of the uncertainties.
Acknowledgements
This research was financially supported by Sapienza, University of Rome, under the project “Thermodynamic study of deep eutectic solvents for green chemistry technologies by means of vapor pressure measurements”, grant number AR22419078906E7A (Recipient: Lorenza Romagnoli, Supervision: Andrea Ciccioli).References
- D. O. Abranches and J. A. P. Coutinho, Annu. Rev. Chem. Biomol. Eng., 2023, 14, 141 CrossRef CAS PubMed.
- S.-H. Pan, H.-T. Yeh, E. C. Lin, J.-C. Jiang and T.-S. Yang, J. Mol. Liq., 2025, 436, 128229 CrossRef CAS.
- S. Kaur, A. Malik and H. K. Kashyap, J. Phys. Chem. B, 2019, 123, 8291 CrossRef CAS PubMed.
- C. F. Araujo, A. O. Abranches, J. A. P. Coutinho, P. D. Vaz, P. Ribeiro-Claro and M. Nolasco, Appl. Spectrosc. Rev., 2025, 60, 137 CrossRef CAS.
- P. Han, W. Nie, G. Zhao and P. Gao, J. Mol. Liq., 2022, 366, 120243 CrossRef CAS.
- O. S. Hammond, D. T. Bowron and K. J. Edler, Green Chem., 2016, 18, 2736 RSC.
- A. van den Bruinhorst and M. Costa Gomes, Curr. Opin. Green Sustainable Chem., 2022, 37, 100659 CrossRef CAS.
- Y. Wang, C. Ma, C. Liu, X. Lu, X. Feng and X. Ji, J. Chem. Eng. Data, 2020, 65, 2446 CrossRef CAS.
- A. van den Bruinhorst, C. Corsini, G. Depraetère, N. Cam, A. Pádua and M. Costa Gomes, Faraday Discuss., 2024, 253, 273 RSC.
- L. A. Souza, B. R. de Moraes, R. M. de Souza, G. A. L. Porto, A. den Bruinhorst, M. Costa Gomes, M. C. C. Ribeiro and R. A. Ando, Phys. Chem. Chem. Phys., 2025, 27, 9123 RSC.
- A. van den Bruinhorst, J. Avila, M. Rosenthal, A. Pellegrino, M. Burghamme and M. Costa Gomes, Nat. Commun., 2023, 14, 6684 CrossRef CAS PubMed.
- P. J. Smith, C. B. Arroyo, F. Lopez Hernandez and J. C. Goeltz, J. Phys. Chem. B, 2019, 123, 5302 CrossRef CAS PubMed.
- B. Kabane and G. G. Redhi, J. Mol. Liq., 2020, 311, 113216 CrossRef CAS.
- K. Xin, I. Roghair, F. Gallucci and M. van Sint Annaland, J. Mol. Liq., 2021, 325, 115227 CrossRef CAS.
- N. S. Jacobson, J.-Y. Colle, V. Stolyarova, T. Markus and I. Nuta, Rapid Commun. Mass Spectrom., 2024, 38, e9744 CrossRef CAS PubMed.
- M. Pesci, L. Romagnoli, B. Brunetti, S. Vecchio Ciprioti, A. Ciccioli and A. Latini, J. Phys. Chem. C, 2025, 129, 9291 CrossRef CAS PubMed.
- M. Gilmore, M. Swadzba-Kwasny and J. D. Holbrey, J. Chem. Eng. Data, 2019, 64, 5248 CrossRef CAS.
- R. Khodabandeh and A. R. Zolghadr, Phys. Chem. Chem. Phys., 2025, 27, 18358 RSC.
- R. N. Mukherjee, P. K. Ghorai and R. Biswas, J. Phys. Chem. B, 2025, 129, 4410 CrossRef CAS PubMed.
- C. F. Araujo, P. Ribeiro-Claro, P. D. Vaz, S. Rudic, R. A. F. Srrano, L. P. Silva, J. A. P. Coutinho and M. M. Nolasco, Phys. Chem. Chem. Phys., 2024, 26, 5969 RSC.
- Y. Marcus, The Entropy of Deep Eutectic Solvent Formation, Entropy, 2018, 20, 524 CrossRef PubMed.
- L. J. B. M. Kollau, M. Vis, A. van den Bruinhorst, R. Tuinier and G. de With, J. Mol. Liq., 2020, 302, 112155 CrossRef CAS.
- https://webbook.nist.gov/cgi/cbook.cgi? ID = C57136&Mask = 200 retrieved the 3rd August, 2025.
- D. Zaitsau, G. J. Kabo, A. A. Kozyro and V. M. Sevruk, Thermochim. Acta, 2003, 406, 17 CrossRef CAS.
- K. Shahbaz, F. S. Mjalli, G. Vakili-Nezhaad, I. M. Ainashef, A. Asadov and M. M. Farida, J. Mol. Liq., 2016, 222, 61 CrossRef CAS.
- S. Ravula, N. E. Larm, M. A. Mottaleb, M. P. Heitz and G. A. Baker, ChemEngineering, 2019, 3, 42 CrossRef CAS.
- H. S. Salehi, H. Mert Polat, F. de Meyer, C. Houriez, C. Coquelet, T. J. H. Vlugt and O. A. Moultos, J. Chem. Phys., 2021, 155, 114504 CrossRef CAS PubMed.
- W. Acree Jr. and J. S. Chickos, J. Phys. Chem. Ref. Data, 2016, 45, 033101 CrossRef.
- M. Gambino and J. P. Bros, Thermochim. Acta, 1988, 127, 223 CrossRef CAS.
- C. Araujo, J. A. P. Coutinho, M. M. Nolasco, S. F. Parker, P. J. A. Ribeiro-Claro, S. Rudic, B. I. G. Soares and P. D. Vaz, Phys. Chem. Chem. Phys., 2017, 19, 17998 RSC.
- L. P. Silva, C. F. Araujo, D. O. Abranches, M. Melle-Franco, M. A. R. Martins, M. M. Nolasco, P. J. A. Ribeiro Claro, S. P. Pinho and J. A. P. Coutinho, Phys. Chem. Chem. Phys., 2019, 21, 18278 RSC.
- C. R. Ashworth, R. P. Matthews, T. Welton and P. A. Hunt, Phys. Chem. Chem. Phys., 2016, 18, 18145 RSC.
- V. Migliorati, F. Sessa and P. D’Angelo, Chem. Phys. Lett., 2019, 737, 100001 CrossRef.
- Y. Marcus, Chem. Rev., 2009, 109, 1346 CrossRef CAS PubMed.
| This journal is © the Owner Societies 2025 |