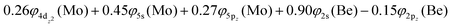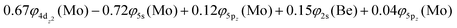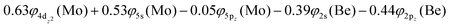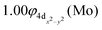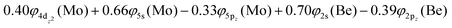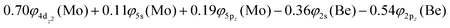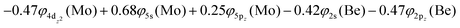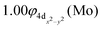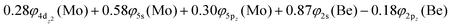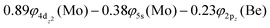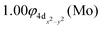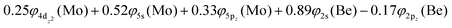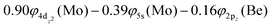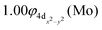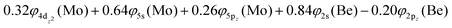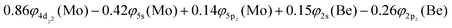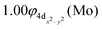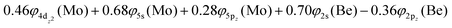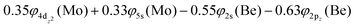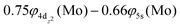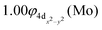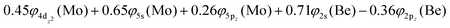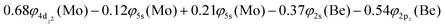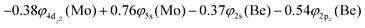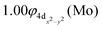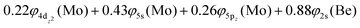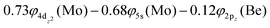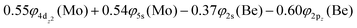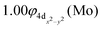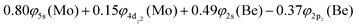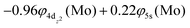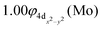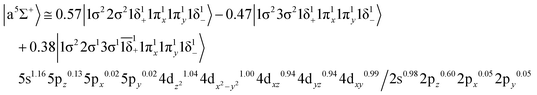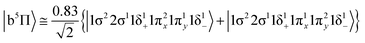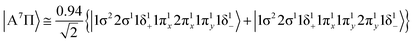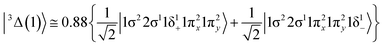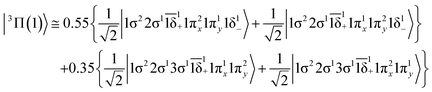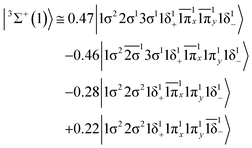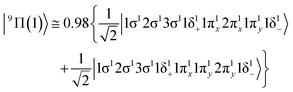Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Electronic structure and chemical bonding of the MoBe molecule
Constantinos
Demetriou
a and
Demeter
Tzeli
 *ab
*ab
aLaboratory of Physical Chemistry, Department of Chemistry, National and Kapodistrian University of Athens, Athens, Greece. E-mail: tzeli@chem.uoa.gr
bTheoretical and Physical Chemistry Institute, National Hellenic Research Foundation, Athens, Greece
First published on 15th October 2025
Abstract
Molybdenum beryllium materials are being researched and applied in cutting-edge technologies. It has been found that beryllium, even though it has a full 2s subshell, can form a variety of bonds with specific atoms. Here, the simple building block, the MoBe molecule, is investigated to shed light in their bonding. Specifically, forty-three low-lying states of MoBe have been investigated via complete active space self-consistent field (CASSCF) and multi-reference configuration interaction (MRCISD(+Q)) using the aug-cc-pV5Z(-PP) basis set. Dissociation energies (De), dipole moments, and various spectroscopic constants are calculated, while potential energy curves are plotted. A variety of bonding is formed in MoBe, i.e., half bonds up to the formation of triple bonds, while in most cases, Be atoms are excited at the Be(3P) state. The ground state, X7Σ+, is well separated from the excited ones, i.e., the first excited state, a5Σ+, is lying 15.0 kcal mol−1 above. The adiabatic De of calculated states range from 2.5 (9Σ+(1), van der Waals interaction) to 57.7 kcal mol−1 (b5Π). The b5Π, 3Δ(1), and 3Π(1) have triple bonds, while their diabatic De values are 86.7, 92.0 and 88.0 kcal mol−1. The MRCISD+Q bond distances range from 2.047 (3Δ(1)) to 2.787 Å (9Σ+(1)), while dipole moments range from 1.51 to 3.28 D. Overall, the present work highlights the exceptional ability of beryllium atoms to participate in a variety of bonding schemes, and it could provide the opening gate for further investigation of this species or associated materials and complexes.
1 Introduction
Materials containing rare metallic elements, such as molybdenum (Mo), have been under extensive investigation and research due to the exceptional properties they present, such as high thermal and corrosion resistance, and good mechanical performance. Alloying Mo with other elements can further enhance these properties, making it suitable for demanding applications. For example, this element is used in high-temperature environments, such as in aerospace and nuclear technology, i.e. rocket nozzles and components in nuclear reactors.1–4 Furthermore, it is employed in applications that require improved strength and is suitable for situations demanding high mechanical performance.5,6 Incidentally, molybdenum beryllium materials, containing Mo–Be bonds, are being researched and applied across these industries, due to the unique physical and chemical properties of both elements,7–11 as well as they have been implemented in surface science, where they are being studied especially for their reflective properties as coated mirrors.12–14 Despite this, the molecular and electronic structure of the ground and excited states of the molybdenum beryllium diatomic molecule have not been investigated, while their bonding has not been explored.It has been extensively stressed that the elucidation of such bonding schemes inside the simplest building blocks of such compounds, which are their diatomic molecules, contributes greatly to the understanding of the bonding arising inside more complex systems comprised of them.15 While this feature is bountiful, their accurate description on a theoretical level poses a demanding task, due to their computational complexity rising from their high density of states and the high space-spin angular momentum of the contained transition metal atoms. Therefore, the understanding of the bonding schemes between a transition metal element and a main group element is not an easy task.16 A thorough inspection into the simplest building blocks of such compounds is the steppingstone towards the investigation of more complex systems. For instance, in the case of the MoS2 2D material, it was found that the low-lying septet states of the diatomic MoS17 and triatomic MoS2 molecules are involved in the material as a building block, explaining the variety of its morphologies.18
Efforts in the exploration of the electronic structure of molybdenum compounds are constantly ongoing, owing to the key role they present in molecular biology, namely in nitrogen fixation and oxidation catalysis.19,20 Incidentally, molybdenum (Mo) presents quite a unique electronic configuration as an element, that is, all of its valence subshells are half filled; Mo: [Kr] 4d55s1, which in turn provides it with a bonding profile that allows it to participate in a multitude of bonding schemes, with some of them resulting in significant properties in the forming compounds.21–25
Conversely, beryllium (Be) presents an electronic configuration of a completely closed and occupied valence subshell orbital; Be: [He] 2s2. For this reason, one would expect beryllium to be highly inert, like its brethren helium (He), but in reality, Be has been found to form many different compounds uer a lot of bonding schemes,26–40 where beryllium forms from single bonds26–33 to multiple bonds,34–40 for instance delocalized bonds,36 double,37 triple bonds,38 and even quadruple bonds.39,40 Quadruple bonds are observed in the case of the PdBe molecule and PbBe− anion, specifically four datives Pd–Be bonds are formed: (4dxz → 2px)2, (4dyz → 2py)2, (4dz2 → 2pz)2, and (5s0 ← 2s)2 in the X1Σ+ state of PbBe39 and (4dxz → 2px)2, (4dyz → 2py)2, (4dz2 → 2pz)2, and (5pz ← 2s)2 in the X2Σ+ state of the PdBe− anion.40 The corresponding dissociation energies are 52.8 kcal mol−1 and 56.7 kcal mol−1 respectively at MRCISD+Q/aug-cc-pV5Z(-PP).39,40 Recently, the electronic spectra for CuBe, which is isovalent with the PdBe− anion, were recorded using resonantly enhanced one-color, two-photon ionization and calculated via CCSD(T) and DFT methodology and it was found that the ground state, X2Σ+, has a dissociation energy (De) of 26.7 kcal mol−1 at RCCSD(T)+DKH2/cc-pwCVQZ-DK.41
Overall, most diatomic molecules of molybdenum have been studied extensively, but this does not hold for the MoBe molecule. To the best of our knowledge, there exist a complete lack of theoretical examination on the MoBe diatomic, while only recently has there been any theoretical investigation regarding it, carried out by our team, where the ground state of the MoX molecules, where X = Li, Be, B, C, O, N, F via multireference and coupled cluster methodologies with the inclusion of correlation energy calculations of both core (4s24p6 of Mo and 1s2 of X) + valence electrons having been carried out.42,43 Here, we have shed light into the electronic spectrum of MoBe for the first time. Specifically, we have investigated the ground state and 42 low-lying states of the MoBe molecule via complete active space self-consistent field (CASSCF), while nine (9) of them were further investigated via multi-reference configuration interaction (MRCISD and MRCISD+Q). The present study aims to deliver a more detailed discussion of the properties of this molecule, with high precision calculations and to fill the gap in the study of the diatomic molecules. Furthermore, the selected calculated states are correlated with material or complexes where the Mo–Be bonds are included in their structure.
2 Computational details
The electronic structure of the MoBe molecule was studying using the state-average complete active space self-consistent field (SA-CASSCF),44,45 CASSCF (without employing any state average technique),44 and multireference configuration interaction plus single and double excitations (CASSCF + single + double replacements = MRCISD)46 methodologies. All calculations were carried out using the augmented correlation consistent polarized valence basis sets of quintuple-ζ quality, i.e., Mo: aug-cc-pV5Z-PP47 and Be: aug-cc-pV5Z.48 For the Mo atom, the accurate core relativistic ECP28MDF pseudopotential developed from the Stuttgart/Köln group49 has been used, which equates to 28 core electrons i.e., 1s22s22p63s23p63d10 electrons. Thus, the 4s24p64d55s1 electrons of Mo were treated via ab initio calculations using the contracted basis of [8s8p7d5f4g3h2i] and 1s22s2 electrons of Be were treated using the contracted [7s6p5d4f3g2h] basis set. Instead of using a relativistic pseudopotential on Mo (as in the present calculations), scalar relativistic effects (mass–velocity and Darwin terms) can be included via an all-electron basis set appropriate for the second-order Douglas–Kroll–Hess (DKH2) approach. Previous calculations from our group on the MoC molecule21 showed that including scalar relativistic effects via DKH2 resulted in bond distances and dissociation energies very similar to those obtained using accurate core-relativistic pseudopotentials of the aug-cc-pV5Z-PP basis set.21 Therefore, we expect that both methodological approaches will yield similar results for the MoBe molecule. Accordingly, in the present work we use only the approach based on basis sets with accurate relativistic pseudopotentials.Finally, regarding the inclusion of spin–orbit coupling for the lowest states, it is found that the ground and first excited states are both Σ states, i.e., X7Σ+ and a5Σ+. Their energy difference is 15.5 kcal mol−1 at the MRCISD level, which is a relatively large separation; thus, inclusion of spin–orbit coupling is not expected to significantly affect their relative energy ordering.
At first, 43 states were studied via SA-CASSCF/aug-cc-pV5Z(-PP) method.45,47,48 The SA-CASSCF reference wavefunctions are obtained by distributing eight active electrons [Mo (4d55s1) + Be (2s2)] to ten orbital functions, one “5s” and five “4d” orbitals on Mo, one ”2s” and three “2p” orbitals on Be. All calculations were carried out under C2v symmetry constraints. The CASSCF wavefunctions have the correct axial angular momentum symmetry, i.e., |Λ| = 0 (Σ+, Σ−), 1 (Π), 2 (Δ), 3 (Φ), 4 (Γ), 5 (H), 6 (I). Thus, Σ+ corresponds to A1 symmetry, Σ− corresponds to A2; Δ, Γ, and I are linear combinations of A1 and A2 symmetries, while Π, Φ, and H are linear combinations of B1 and B2 symmetries. The SA-CASSCF was applied for states within the same multiplicity of spin, while two groups were obtained for each multiplicity of spin, i.e., the first one contains four states of the A1 and four states of A2 symmetries, which includes Σ+, Σ−, Δ, Γ, and I, and the second one contains four states of B1 and four states of B2 symmetries, which includes Π, Φ, and H. All states at the SA-CASSCF/aug-cc-pV5Z(-PP) method are optimized with weight 1. The thresholds used are: 10−5 for the orbital gradient and 10−8 for change of the total energy.
Then nine states out of forty-three, i.e., the lowest in energy states and some selected ones were further studied via CASSCF/aug-cc-pV5Z(-PP)44,47,48 followed by the MRCISD/aug-cc-pV5Z(-PP)46–48 method. Note that MRCISD wavefunctions may not display pure spatial angular momentum symmetry, however, in the present calculations, their CASSCF wavefunctions have the correct spatial angular momentum symmetry and thus the corresponding MRCISD wavefunctions retain the same axial angular momentum symmetry.45
The CASSF reference spaces consist of 5154 (3Σ+), 5220 (3Π), 5196 (3Δ), 2070 (5Σ+), 2060 (5Π), 303 (7Σ+), 292 (7Π), 9 (9Σ+), and 12 (9Π) configuration state functions (CSFs). The corresponding MRCISD spaces consist of 2 × 108 (3Σ+, 3Π, 3Δ), 1 × 108 (5Σ+, 5Π), 3 × 107 (X7Σ+, 7Π), 2 × 106 (9Σ+, 9Π) CSFs. The thresholds used are: 10−5 for the orbital gradient and 10−8 for the change of the total energy. In all multireference calculations, the internal contraction scheme was used,50 where the multireference CSFs were reduced by about two orders of magnitude. Since this scheme is used for all multireference calculations, the notation MRCISD will be used for simplicity. Finally, the Davidson correction for unlinked quadruples (+Q)51 was employed, which minimizes the size non-extensivity problems.
For all calculated states, bond distances, dissociation energies, and other spectroscopic constants have been computed at all levels of theory. The corresponding potential energy curves (PECs) have been plotted. Additionally, for some states where avoided crossings were observed, their diabatic PECs are also plotted. Furthermore, spectroscopic constants were obtained via a Dunham analysis.52 Moreover, the chemical bonding is analyzed. In each case, the bonding has been plotted via 2-D valence bond Lewis (vbL) diagrams and via the 3D plot of the valence molecular orbitals. Mulliken population analysis53 is also included using a small correlation consistent polarized valence basis set of triple-ζ quality basis set, cc-pVTZ(-PP), since Mulliken population analysis is basis set dependent, and it provides better data with simple non-augmented basis sets.54 It is generally accepted that population analysis, either Mulliken or natural population analysis, helps in comparing similar states of the same molecule.54 All computations were carried out using the MOLPRO55 code.
3 Results & discussion
3.1 SA-CASSCF
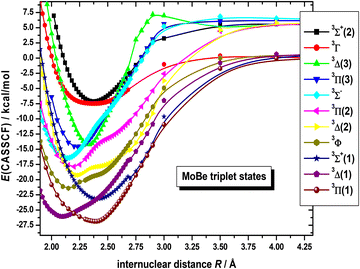
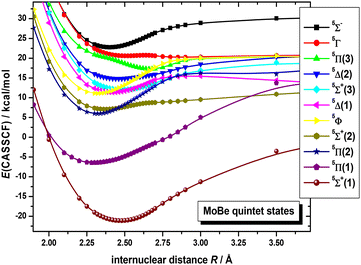
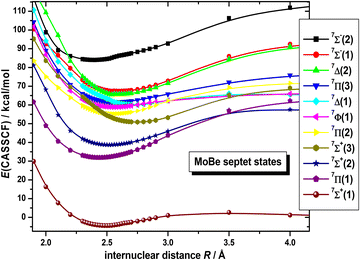
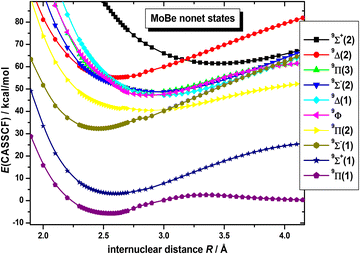
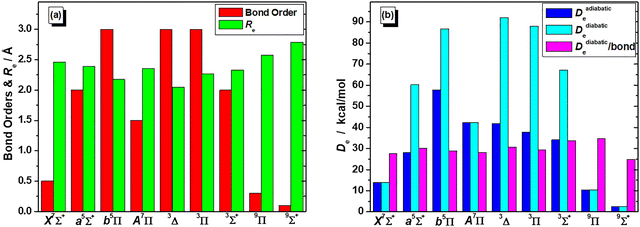
On the basis that a complete lack of any previous background, on either experimental or theoretical grounds regarding the picture on the electronic spectra of MoBe, as a starting point exploratory SA-CASSCF calculations were carried out to determine 43 electronic states and set the scene. Specifically, 10 nonet states, i.e.9(Σ+ [2], Σ− [2], Π [3], Δ [2], Φ, Γ), 11 septet states, i.e.7(Σ+ [3], Σ− [2], Π [3], Δ [2], Φ), 11 quintet states, i.e.5(Σ+ [3], Σ−, Π [3], Δ [2], Φ, Γ), and 11 triplet states, i.e.3(Σ+ [2], Σ−, Π [3], Δ [3], Φ, Γ), with the numbers in the square brackets i.e., Π[3], denoting that three Π states have been calculated. The calculated states are correlated to Mo(a7S, a5S, a5D, a5G, a5P, a3P, a3D, a3G) + Be(1S, 3P), adiabatically. It is worth noting that the triplet excited states of Mo, i.e., a3P, a3D, and a3G, are very close lying. Additionally, regarding which of their j term lies lowest in energy, a3P0 lies lower than a3D1, which in turn lies lower than a3G3, meaning that their ordering is exactly like that of a3P, a3D, and a3G. However, because the calculation that were carried out considered their j-average term, the a3G term will be regarded as the lowest one, and this is the reason why the triplet states of MoBe correlate adiabatically to the a3G term.The SA-CASSCF PECs for all calculated molecular states are plotted in Fig. 1–4, and, additionally, their bond distances as well as their relative energy differences are also provided in Table 1. Due to having retrieved a large number of calculated spin states with the same angular momentum symmetries, for clarity regarding their energetic succession we have included the ordering inside parenthesis, i.e., 5Σ+(1) is the first 5Σ+ state, while 5Σ+(2) is the second 5Σ+ state. The bond distances range from 2.096 Å (in 3Δ(1)) up to 3.459 Å (in 9Σ+(2)). Interestingly, the shortest bond distances are observed in triplet and quintet states, i.e., 3Δ(1), 3Σ−(1), 3Φ(1), 3Π(2), 3Δ(2), 3Π(3), 5Π(1), see Table 1, which, at the MRCISD/aug-cc-pV5Z level of theory, showcase identical bonding schemes, see discussion bellow.
| State | R e | T e | State | R e | T e | State | R e | T e |
|---|---|---|---|---|---|---|---|---|
| X 7Σ+ | 2.497 | 0 | 5Γ | 2.505 | 53.0 | 7Π(3) | 2.574 | 65.9 |
| 5Σ+(1) | 2.474 | 11.3 | 3Φ | 2.151 | 53.8 | 3Γ | 2.379 | 67.6 |
| 5Π(1) | 2.293 | 26.0 | 5Σ− | 2.401 | 55.2 | 3Σ+(2) | 2.375 | 67.9 |
| 7Π(1) | 2.438 | 36.3 | 7Σ+(3) | 2.746 | 55.3 | 7Δ(2) | 2.699 | 70.1 |
| 5Π(2) | 2.329 | 38.4 | 9Π(1)a | 2.556 | 55.4 | 7Σ−(1) | 2.603 | 71.8 |
| 5Σ+(2) | 2.373 | 39.5 | 3Δ(2) | 2.226 | 55.8 | 7Σ−(2) | 2.395 | 88.4 |
| 7Σ+(2) | 2.521 | 43.1 | 3Π(2) | 2.179 | 57.1 | 9Σ−(1) | 2.461 | 93.4 |
| 5Φ | 2.321 | 43.6 | 3Σ− | 2.136 | 58.6 | 9Π(2) | 2.889 | 101.6 |
| 5Δ(1) | 2.419 | 43.8 | 7Π(2) | 2.569 | 59.9 | 9Φ | 2.875 | 108.3 |
| 5Σ+(3) | 2.485 | 44.4 | 3Π(3) | 2.234 | 60.4 | 9Δ(1) | 2.979 | 108.3 |
| 5Δ(2) | 2.456 | 47.2 | 3Δ(3) | 2.339 | 60.8 | 9Σ−(2) | 2.957 | 109.8 |
| 3Π(1) | 2.399 | 48.2 | 7Φ | 2.569 | 63.3 | 9Π(3) | 2.903 | 109.8 |
| 3Δ(1) | 2.096 | 49.0 | 9Σ+(1) | 2.597 | 64.3 | 9Δ(2) | 2.607 | 116.3 |
| 5Π(3) | 2.671 | 49.7 | 7Δ(1) | 2.735 | 64.5 | 9Σ+(2) | 3.459 | 122.5 |
| 3Σ+(1) | 2.409 | 51.9 |
The PECs of the triplet states of MoBe are plotted in Fig. 1. They are lying about 48 kcal mol−1 (≈1.7 eV) above the ground state and they present bond distances around 2.1–2.2 Å, see Table 1. As seen in Fig. 1, at around 2.3 Å, an avoided crossing between the 3Π(2) with the 3Π(3) state occurs, where the corresponding states interexchange configurations. Similarly, the 3Δ(1) state appears to be coupled to the 3Δ(2) state, where an avoided crossing occurs at around 2.3 Å. Additionally, the 3Δ(2) state presents a coupling to the 3Δ(3) state, where an avoided crossing occurs at around 2.3 Å, too. Furthermore, the 3Δ(3) state presents, another coupling to some higher in energy state, that has not been calculated, however their crossing is easily observed in their configurations, as depicted in Fig. 1, at around 2.9 Å. Lastly, the 3Φ(1), and 3Σ−(1) states showcase avoided crossing with some higher states, cooccurring at around 2.4 Å.
Quintet states are lying about 10 kcal mol−1 (≈0.4 eV) above the ground state, placing them adjacent to it, in reality intermediating between the ground and the rest of the septet states. Their PECs are plotted in Fig. 2, where an observant reader can discern an avoided crossing between the 5Π(1) with the 5Π(2) state, at around 2.5 Å, and another one, between the 5Π(2) with the 5Π(3) state, at around 2.75 Å, as well as between the 5Δ(1) with the 5Δ(2) state, at around 2.75 Å, as well. While not easily noticed in the quintet PECs, the 5Σ+(2) couples with the 5Σ+(3), as well, at around 2.6 Å. This was examined at the MRCISD level too, where the same avoided crossing occurs, albeit we won’t be discussing it any further, as it goes beyond the scope of the present study. Lastly, the 5Φ(1), and 5Γ(1) states showcase avoided crossing with some higher states, at around 2.5 Å, and 2.6 Å, respectively.
In Fig. 3 we have collected the eleven lowest in energy septet states. It is observed that the X7Σ+(1) state, standing well separated from the bunch of the rest of the septet states., i.e., it lies about 36 kcal mol−1 (≈1.6 eV) below them, and thus it is undoubtedly the ground state of MoBe. An avoided crossing is observed between 7Δ(1) and 7Δ(2) at about 2.5 Å and they interchange their character, see Fig. 3.
Finally, the nonet states are plotted in Fig. 4, lying at about 55 kcal mol−1 (≈2.4 eV) above the ground state. While SA-CASSCF calculations predict that the 9Π(1) and 9Σ+(1) correlate to entirely different adiabatic products at their atomic limits, in the CASSCF, MRCISD, and MRCISD+Q calculations, that proceeded, it was revealed that they corelate to the exact same products, (a7S) + Be(3P). Moreover, while at the SA-CASSCF/aug-cc-pV5Z computational level, the PECs of 9Π(1) and 9Π(2) give the impression of a coupling being involved between the states, even though they are clearly separated, at the CASSCF, MRCISD, and MRCISD+Q/aug-cc-pV5Z(-PP) levels it's demonstrated that such coupling doesn’t occur, and no avoided crossing between those two states takes place, as 9Π(1) is found to be a single-reference state.
3.2 MRCISD(+Q) methods
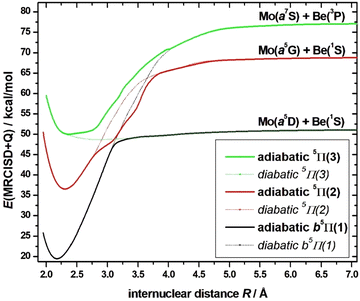
The four low-lying electronic states, 7Σ+(1), 5Σ+(1), 5Π(1), and 7Π(1), as well as the bunch of the three lowest lying triplet states, i.e.3Δ(1), 3Π(1), 3Σ+(1), which are closely packed together, along with the two lowest nonet states, 9Π(1), and 9Σ+(1), were further investigated at the MRCISD(+Q) level of theory. Their PECs were calculated, see Fig. 5, while their bonding across their PECs were analyzed. It should be mentioned, as it will matter later on, that, while the 3Δ(1) lies higher in energy than the 3Π(1) state at the SA-CASSCF and CASSCF levels of theory, at the MRCISD(+Q) computational levels the successive order of this pair is reversed, see Tables 1 and 2. Moreover, it should be noted that CASSCF and SA-CASSCF present in general the same relative energy ordering of the states, with the one exception being the high spin state 9Σ+(1). Additionally, the ground molecular state of MoBe, 7Σ+(1), along with the three lowest-lying excited states, them being 5Σ+(1), 5Π(1), and 7Π(1), are clearly separated for the remaining excited states across all used methods and their ordering is retained in all methods.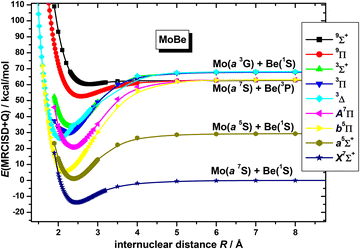 | ||
| Fig. 5 PECs of the selected states of MoBe at the MRCISD+Q/aug-cc-pV5Z(-PP) computational level. The Mo(a7S) + Be(1S) limit, to which the X7Σ+ state correlates, is used to define the zero of energy. | ||
| State | Methodology | R e | D e | ω e | ω e x e | μ FF | 〈μ〉a | T e |
|---|---|---|---|---|---|---|---|---|
| a μ FF: dipole moment via finite field; 〈μ〉: dipole moment calculated as an expectation value. b At the CASSCF/aug-cc-pV5Z(-PP) level, the X7Σ+ state is nearly bound. At the SA-CASSCF/aug-cc-pV5Z(-PP) level, state-average of the X7Σ+(1), 7Σ+(2), 7Σ+(3), 7Δ(1), 7Δ(2), 7Σ−(1), and 7Σ−(2) states, the X7Σ+ state is bound. c Ref. 42; RCCSD(T)/aug-cc-pV5Z(-PP), the valence electrons (8 electrons) are correlated. d Ref. 42; C-MRCISD+Q/aug-cc-pwCV5Z(-PP), C-RCCSD(T)/aug-cc-pwCV5Z(-PP), where C- stands for core. All 18 electrons treated via ab initio calculations are correlated. e Adiabatic De with respect to Mo(a5S; 4d55s1) + Be(1S); diabatic De with respect to Mo(a7S; 4d55s1) + Be(3P) in parenthesis. f Adiabatic De with respect to Mo(a5D; 4d45s2) + Be(1S); diabatic De with respect to Mo(a7S; 4d55s1) + Be(3P) in parenthesis. g Adiabatic De with respect to Mo(a3G) + Be(1S); diabatic De with respect to Mo(a5G) + Be(3P) in parenthesis. h Adiabatic De with respect to Mo(a3G) + Be(1S); diabatic De with respect to Mo (a5S) + Be (3P) in parenthesis. | ||||||||
| X 7Σ+ | SA-CASSCFb | 2.497 | 4.39 | 0.0 | ||||
| MRCISD | 2.481 | 11.42 | 330.7 | 6.26 | 1.41 | 0.94 | 0.0 | |
| MRCISD+Q | 2.462 | 13.85 | 351.1 | 5.66 | 1.51 | 0.0 | ||
| RCCSD(T)c | 2.481 | 13.51 | 340.3 | 8.22 | 1.28 | 0.0 | ||
| C-MRCISD+Qd | 2.431 | 13.20 | 383.4 | 5.93 | 1.65 | 0.0 | ||
| C-RCCSD(T)d | 2.452 | 14.44 | 349.4 | 11.63 | 1.24 | 0.0 | ||
| a 5Σ+ | CASSCF | 2.488 | 16.79 (48.84)e | 383.5 | 2.16 | 4.07 | 4.07 | 6.1 |
| MRCISD | 2.400 | 27.20 (59.25)e | 406.8 | 2.22 | 3.02 | 4.13 | 15.5 | |
| MRCISD+Q | 2.392 | 28.20 (60.25)e | 407.3 | 2.26 | 2.76 | 15.0 | ||
| b 5Π | CASSCF | 2.286 | 33.22 (62.23)f | 412.5 | 2.71 | 3.39 | 3.39 | 17.6 |
| MRCISD | 2.181 | 54.58 (83.59)f | 505.8 | 3.26 | 2.60 | 2.61 | 20.4 | |
| MRCISD+Q | 2.177 | 57.72 (86.73)f | 511.2 | 3.27 | 2.51 | 19.2 | ||
| A 7Π | CASSCF | 2.464 | 21.61 | 384.2 | 2.21 | 3.25 | 3.25 | 29.3 |
| MRCISD | 2.365 | 38.81 | 435.7 | 2.18 | 1.96 | 2.14 | 36.2 | |
| MRCISD+Q | 2.359 | 42.32 | 436.9 | 2.32 | 1.77 | 34.6 | ||
| 3Δ(1) | CASSCF | 2.087 | 27.23 (77.40)g | 443.1 | 2.13 | 3.22 | 3.22 | 48.3 |
| MRCISD | 2.045 | 40.66 (90.83)g | 571.1 | 1.19 | 2.27 | 2.40 | 42.4 | |
| MRCISD+Q | 2.047 | 41.84 (92.01)g | 576.7 | 3.53 | 2.14 | 40.2 | ||
| 3Π(1) | CASSCF | 2.317 | 27.12 (77.29)g | 449.5 | 9.10 | 4.23 | 4.23 | 46.4 |
| MRCISD | 2.264 | 36.86 (87.03)g | 446.9 | 24.52 | 2.76 | 3.49 | 45.8 | |
| MRCISD+Q | 2.266 | 37.81 (87.98)g | 456.0 | 24.32 | 2.31 | 43.9 | ||
| 3Σ+(1) | CASSCF | 2.404 | 22.55 (55.63)h | 445.6 | 4.14 | 3.72 | 3.72 | 50.0 |
| MRCISD | 2.338 | 32.61 (65.69)h | 459.7 | 9.70 | 2.43 | 2.88 | 50.0 | |
| MRCISD+Q | 2.333 | 34.12 (67.20)h | 449.8 | 6.10 | 2.15 | 47.7 | ||
| 9Π(1) | CASSCF | 2.661 | 1.96 | 220.2 | 11.71 | 3.91 | 3.91 | 48.9 |
| MRCISD | 2.581 | 8.88 | 273.5 | 5.38 | 2.72 | 2.98 | 66.1 | |
| MRCISD+Q | 2.579 | 10.43 | 278.5 | 4.54 | 2.41 | 66.5 | ||
| 9Σ+(1) | CASSCF | 2.851 | 2.07 | 141.2 | 21.00 | 3.74 | 3.74 | 50.6 |
| MRCISD | 2.752 | 1.77 | 198.9 | 9.00 | 3.36 | 3.67 | 72.7 | |
| MRCISD+Q | 2.787 | 2.48 | 203.4 | 8.12 | 3.28 | 74.1 | ||
To sum up, our best results are obtained at the MRCISD+Q level; the states are named based on their MRCISD+Q energies as X7Σ+, a5Σ+, b5Π, Α7Π, 3Δ(1), 3Π(1), 3Σ+(1), 9Π(1), and 9Σ+(1). For these nine electronic states, their usual spectroscopic parameters, such as bond distances, dissociation energies, dipole moments, harmonic and anharmonic corrections, relative adiabatic energies at CASSCF, MRCISD and MRCISD+Q/aug-cc-pV5Z(-PP) are provided in Table 2 and their leading equilibrium CASSCF configuration accompanied by its corresponding molecular orbitals are given in Table 3. Over the following paragraphs we further discuss in detail the bonding of these selected electronic states.
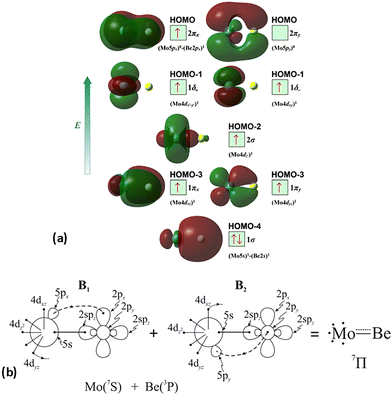
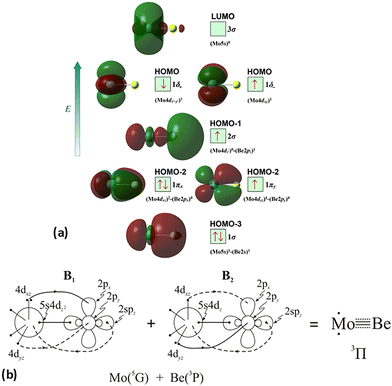
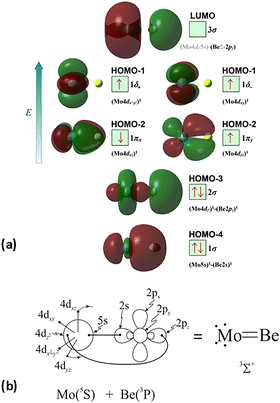
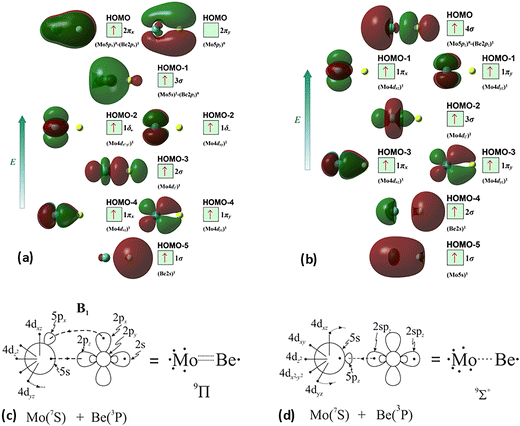
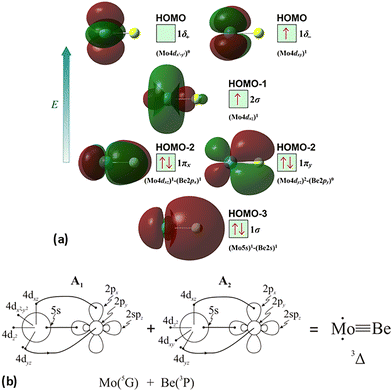
The molecular orbitals (MOs) of the valence electrons are plotted in Scheme 1a and the formed bond is summarized in the vbL diagram in Scheme 1b. MOs show that there exists an evident 4dz25s5pz hybridization of orbitals on the Mo atom, while the Be atom exhibits 2s2pz hybridization, see Table 3. Based on the MOs, it is found that a σ dative-type bond formed between the 4dz2 single electron of Mo and the vacant 2pz orbital of Be, resulting in a σ half-bond with a length of 2.462 Å, and a dissociation energy of 13.85 kcal mol−1, at the computational level MRCISD+Q/aug-cc-pV5Z(-PP), as shown in Table 2. Also, the 1π1x and 1π1y orbitals show that a small electron density is transferred to the empty 2px and 2py orbitals of the Be atom. Mulliken and natural population analysis at the MRCISD/aug-cc-pV5Z(-PP) levels are given in SI. NPA shows also the existence of a 2s2pz hybridization on Be. All populations show that the Mo atom is in the ground state. Of course, there are some differences in the charge distributions between population analysis methods; nevertheless, it is generally accepted that both Mulliken and natural population analysis are useful for comparing similar states of the same molecule within a consistent methodological framework.54
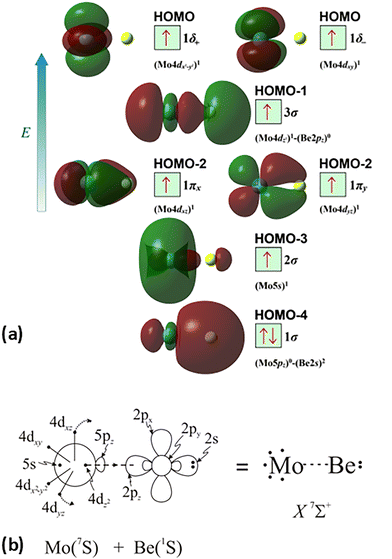 | ||
| Scheme 1 (a) MOs diagrams and (b) valence bond Lewis diagram of the ground state X7Σ+ of MoBe at the CASSCF/aug-cc-pV5Z(-PP) level. | ||
The X7Σ+ state has been studied via coupled-cluster theory in ref. 42, where the ground states of the MoX molecules, where X = Li, Be, B, C, N, O, and F, have been calculated. The X7Σ+ state of the MoBe molecule has been calculated via the restricted open shell CCSD(T)56 using the aug-cc-pV5Z(-PP) basis set45 when the valence [2s2(Be) + 5s14d5(Mo)] electrons are correlated and using the aug-cc-pwCV5Z(-PP) basis set45 when all 18 electrons treated via ab initio calculations [1s22s2(Be) + 4s24p65s14d5(Mo)] are correlated. For reason of simplicity and in accordance with ref. 42, the methods are denoted as RCCSD(T)/aug-cc-pV5Z(-PP) and C-RCCSD(T)/aug-cc-pwCV5Z(-PP), where C- stands for core. The coupled cluster dissociation energy was calculated at 13.51 kcal mol−1via the RCCSD(T)/aug-cc-pV5Z(-PP) method and 14.44 kcal mol−1via C-RCCSD(T)/aug-cc-pwCV5Z(-PP).42 The values are similar to the MRCISD+Q/aug-cc-pV5Z(-PP) value of 13.85 kcal mol−1. Finally, it should be added that the correlation of the 4s24p6 electrons of Mo and 1s2 electrons of Be leads to a shorter bond distance in the ground state of about 0.03 Å, see Table 2, as is expected, either at multireference level of theory or coupled cluster level of theory. To sum up, the C-RCCSD(T)/aug-cc-pwCV5Z(-PP) method provides the best De and re values, i.e., De = 14.44 kcal mol−1 and re = 2.452 Å, for the ground state, since the X7Σ+ state is a single reference state. Finally, as mentioned in the computational details section, our previous calculations on the MoC molecule,21 using either a relativistic pseudopotential on Mo (as in the present study) or an all-electron basis set to correlate core electrons including relativistic effects, showed that applying the second-order Douglas–Kroll–Hess (DKH2) approach resulted in the same bond distances and similar dissociation energies compared to those obtained with accurate core-relativistic pseudopotentials of the aug-cc-pV5Z-PP basis set.21 Therefore, we conclude that the use of accurate core-relativistic pseudopotentials of quintuple-ζ quality effectively accounts for scalar relativistic effects.
Based on the above representation, we can assert that the a5Σ+ state exhibits an intent multi-reference character, at the MRCISD/aug-cc-pV5Z(-PP) level, as indicated by the relatively low reference weight (c0 = 0.57) of the leading configuration, which reflects significant configuration mixing. Although this state correlates adiabatically to Mo(a5S) + Be(1S) at dissociation, its electronic structure near equilibrium is predominantly described by configurations corresponding to either Mo(a5D) + Be(1S) or Mo(a7S) + Be(3P). It was not possible to clarify which one is the predominant since both correspond to the same double σ bond (1σ22σ2). However, the origin of the electrons forms the double bond differ, see Scheme 2b. In the first scenario, molybdenum, residing in its excited Mo(a5D) state, forms with beryllium, in its ground state, Be(1S), two dative σ bonds, owing to the Be atom acting as an electron density donor, transferring its 2s electron lone pair inside Mo's vacant 4dz2 orbital, forming a dative σ bond, while the Mo atom responds by forming an additional dative σ bond, by transferring its own 5s electron lone pair inside the vacant 2pz orbital of Be, resulting in a double bond. In the alternative, but equally plausible case, Mo atom is in its ground state, Mo(a7S), whilst Be atom resides in its first excited state, Be(3P). In this combination, two σ covalent bonds can form, one arising from the spin coupling between a 5s single electron of Mo with a 2s single electron of Be, and one from the spin coupling between a 4dz2 single electron of Mo with a 2pz single electron of Be, giving rise to a double bond, as well. A superposition between these two configurations exists at equilibrium. Population analysis shows that about 0.4e− is transferred from Be to Mo in the σ frame, i.e., the Mo/Be is: 5s1.165pz0.134dz21.04/2s0.982pz0.60 while, it is evident that a 4dz25s5pz hybridization exists on the Mo atom, and a 2s2pz hybridization on Be atom. However, it can be regarded as a more likely scenario the Mo(a7S) + Be(3P) combination, since two covalent bonds may be more favorable than two dative or because in the case of two dative bonds it is not likely to be transferred one electron to the empty 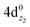 of Mo. Finally, a small electron density is transferred to the empty 2px and 2py orbitals of the Be atom from the 1π1x and 1π1y orbitals and as a result a total of about 0.26e are transferred from Be to Mo.
of Mo. Finally, a small electron density is transferred to the empty 2px and 2py orbitals of the Be atom from the 1π1x and 1π1y orbitals and as a result a total of about 0.26e are transferred from Be to Mo.
 | ||
| Scheme 2 (a) MOs diagrams and (b) valence bond Lewis diagram of the a5Σ+(1) state of MoBe at the CASSCF/aug-cc-pV5Z(-PP) level. | ||
At the MRCISD+Q/aug-cc-pV5Z(-PP) level of theory, the a5Σ+ state has a bond length of 2.393 Å, and an adiabatic dissociation energy of 28.20 kcal mol−1. The diabatic dissociation energy can be estimated by adjusting the adiabatic dissociation energy: specifically, by subtracting the energy required for Mo(a5S) to relax back to its ground state, Mo(a7S), and adding the excitation energy needed for Be(1S) to transition to its triplet first excited state, Be(3P). This yields a diabatic De of 60.25 kcal mol−1 = {28.20–30.79[Mo(a7S) ← Mo(a5S)] + 62.84[Be(1S) → Be(3P)]} kcal mol−1.57,58 Interestingly, when referenced to the Mo(a5D) + Be(1S) atomic limit, the diabatic dissociation energy is of the same order, of 62.03 kcal mol−1 {= 28.00 kcal mol−1, +33.83 Mo(a5S) → Mo(a5D)}, reinforcing the equivalence between the two alternative configurations. Based on the dissociation energies, a relatively strong covalent bond must form between the two atoms, as expected from the bond order.
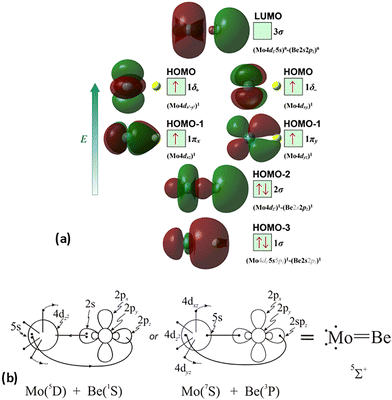
At the equilibrium minimum of the b5Π state, the leading equilibrium CASSCF configuration, followed by the Mulliken atomic distributions (Mo/Be) are:
| 5s0.945pz0.195px0.045py0.014dz20.954dx2−y21.024dxz1.334dyz0.914dxy0.99/2s0.812pz0.602px0.602py0.10 B1 |
| 5s0.945pz0.195px0.015py0.044dz20.954dx2−y21.024dxz0.914dyz1.334dxy0.99/2s0.812pz0.602px0.102py0.60 B2 |
 | ||
| Scheme 3 (a) MOs diagrams and (b) valence bond Lewis diagram of the b5Π(1) state of MoBe at the CASSCF/aug-cc-pV5Z(-PP) level. | ||
Thus, a bond of order 3, σ2σ1π2π1, with a bond length of 2.177 Å is formed with an adiabatic dissociation energy of 57.72 kcal mol−1, at the MRCISD+Q/aug-cc-pV5Z(-PP) level of theory. The diabatic dissociation energy, i.e., the energy with respect to the in situ atoms, can be estimated by adjusting the adiabatic dissociation energy, by subtracting the energy required for Mo(a5D) to relax back to its ground state, Mo(a7S), and adding the excitation energy needed for Be(1S) to transition to its triplet first excited state, Be(3P); De = 86.73 kcal mol−1 = {57.72–33.83[Mo(a7S) ← Mo(a5D)] + 62.84[Be(1S) → Be(3P)]}.57,58 Based on this dissociation energy, we conclude that a strong covalent bond is formed between the two atoms, as expected from the high bond order.
| 5s0.785pz0.205px0.385py0.014dz20.964dx2−y21.004dxz0.974dyz0.954dxy0.99/2s0.942pz0.072px0.642py0.06 B1 |
| 5s0.785pz0.205px0.015py0.384dz20.964dx2−y21.004dxz0.954dyz0.974dxy0.99/2s0.942pz0.072px0.062py0.64 B2 |
The bonding in this state is summarized in the vbL diagram of Scheme 4. Namely, a σ2 covalent bond is formed, 5s1(Mo)–2s1(Be), and a π1 half-bond either a 5p0x(Mo)–2p1x(Be) or 5p0y(Mo)–2p1y(Be). Likewise, a 4dz25s5pz hybridization is observed at the Mo atom. Overall, a total electron charge of about 0.25e is transferred from the Be atom to the Mo metal. The resulting σ2π1 bond has an order of 1.5, a bond length of 2.359 Å, and a dissociation energy of 42.3 kcal mol−1, at the MRCISD+Q/aug-cc-pV5Z(-PP) level of theory.
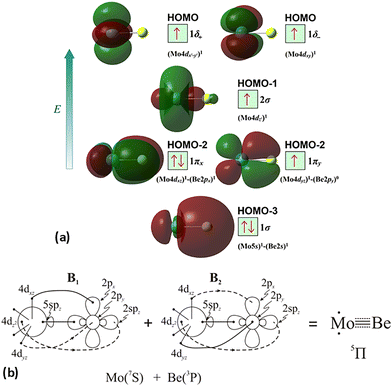
At the equilibrium minimum of the 3Δ(1) state, the leading equilibrium CASSCF configuration, followed by the Mulliken atomic distributions (Mo/Be) are:
| 5s1.145pz0.185px0.035py0.034dz20.954dx2−y21.004dxz1.534dyz1.534dxy0.07/2s0.652pz0.062px0.372py0.37 A1 |
| 5s1.145pz0.185px0.035py0.034dz20.954dx2−y20.074dxz1.534dyz1.534dxy1.00/2s0.652pz0.062px0.372py0.37 A2 |
 | ||
| Scheme 5 (a) MOs diagrams and (b) valence bond Lewis diagram of the 3Δ(1) state of MoBe at the CASSCF/aug-cc-pV5Z(-PP) level. | ||
This state presents the shortest bond distance and the largest diabatic dissociation energy among the calculated states. The bond distance is 2.047 Å, the adiabatic De value is 41.84 kcal mol−1, at the MRCISD+Q/aug-cc-pV5Z(-PP) level of theory, whilst its diabatic dissociation energy is 92.01 kcal mol−1 {= 41.84–12.67[Mo(a5G) ← Mo(a3G)] + 62.84[Be(1S) → Be(3P)]} kcal mol−1.57,58
| 5s1.105pz0.165px0.035py0.024dz20.784dx2−y20.794dxz1.484dyz1.084dxy0.80/2s0.892pz0.492px0.212py0.10 B1 |
| 5s1.105pz0.165px0.025py0.034dz20.784dx2−y20.794dxz1.084dyz1.484dxy0.80/2s0.892pz0.492px0.102py0.21 B2 |
The 3Π state has a strong multi-reference character as indicated by the relatively small coefficient (c0 = 0.55) of the leading configuration. This change in asymptotic character occurs due to the internally mixed nature of the state. The bonding in this state is summarized in Scheme 6. The bonding consists of a σ2 covalent bond 5s1(Mo)–2s1(Be), a σ1 dative bond 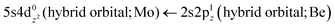 , and two dative π2π1 bonds, either d2xz(Mo) → 2p0x(Be) and 4d0yz(Mo) ← 2p1y(Be) or d2yz(Mo) → 2p0y(Be) and 4d0xz(Mo) ← 2p1x(Be). About 0.26e are transferred from the Be atom to the Mo metal. The resulting σ2σ1π2π1 bond has an order of 3, a bond length of 2.266 Å, and a dissociation energy of 37.81 kcal mol−1, at the MRCISD+Q/aug-cc-pV5Z(-PP) level of theory, whilst its diabatic dissociation is 87.98 kcal mol−1 {= 37.81–12.67[Mo(a5G) ← Mo(a3G)] + 62.84[Be(1S) → Be(3P)]}.57,58
, and two dative π2π1 bonds, either d2xz(Mo) → 2p0x(Be) and 4d0yz(Mo) ← 2p1y(Be) or d2yz(Mo) → 2p0y(Be) and 4d0xz(Mo) ← 2p1x(Be). About 0.26e are transferred from the Be atom to the Mo metal. The resulting σ2σ1π2π1 bond has an order of 3, a bond length of 2.266 Å, and a dissociation energy of 37.81 kcal mol−1, at the MRCISD+Q/aug-cc-pV5Z(-PP) level of theory, whilst its diabatic dissociation is 87.98 kcal mol−1 {= 37.81–12.67[Mo(a5G) ← Mo(a3G)] + 62.84[Be(1S) → Be(3P)]}.57,58
| 5s1.235pz0.165px0.025py0.024dz21.044dx2−y20.994dxz0.884dyz0.884dxy0.99/2s0.972pz0.522px0.102py0.10 A1 |
The 3Σ+(1) has a strongly multi-reference character indicating the presence of configuration mixing. Although this state correlates adiabatically to Mo(a3G) + Be(1S) at dissociation, its electronic structure near equilibrium is predominantly described by configurations corresponding to Mo(a5S) + Be(3P). Two covalent σ2σ2 bonds are formed, i.e., 5s1(Mo)–2s1(Be) and 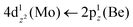 and ∼0.26e are transferred from Be to Mo, see Scheme 7. The bond distance is 2.333 Å and the adiabatic De values is 34.12 kcal mol−1 at MRCISD+Q/aug-cc-pV5Z(-PP). The diabatic dissociation energy is De (diabatic) = 67.20 kcal mol−1 {= 34.12–29.76[Mo(a5S) ← Mo(a3G) + 62.84[Be(1S) → Be(3P)]}.
and ∼0.26e are transferred from Be to Mo, see Scheme 7. The bond distance is 2.333 Å and the adiabatic De values is 34.12 kcal mol−1 at MRCISD+Q/aug-cc-pV5Z(-PP). The diabatic dissociation energy is De (diabatic) = 67.20 kcal mol−1 {= 34.12–29.76[Mo(a5S) ← Mo(a3G) + 62.84[Be(1S) → Be(3P)]}.
| 5s0.785pz0.105px0.255py0.014dz20.894dx2−y20.984dxz0.984dyz0.944dxy0.99/2s0.822pz0.492px0.752py0.04 B1 |
| 5s0.785pz0.105px0.015py0.254dz20.894dx2−y20.994dxz0.944dyz0.984dxy0.99/2s0.822pz0.492px0.042py0.75 B2 |
| and |9Σ+(1)〉 ≅ 0.97|1σ12σ13σ14σ11δ1+1π1x1π1y1δ1−〉 |
| 5s0.975pz0.425px0.025py0.024dz20.944dx2−y20.994dxz0.924dyz0.924dxy0.99/2s0.872pz0.752px0.052py0.05 A1 |
In the 9Π(1) state, two weak dative interactions are formed, i.e., σ1: 5s1(Mo) → 2p0z(Be) and π1: 5p0x(Mo)⋯2p1x(Be) or 5p0y(Mo)⋯2p1y(Be), see Scheme 8. Note that a 4dz25s5pz hybridization exists on the Mo atom and a 2s2pz on Be. The calculated re is 2.579 Å and the De is 10.43 kcal mol−1 at the MRCISD+Q/aug-cc-pV5Z(-PP) method. The 9Σ+(1) state is clearly a van der Waals state, there is a 5p0z(Mo)⋯2s2p1z(Be) interaction resulting in a bond distance of 2.787 Å and a dissociation energy of 2.48 kcal mol−1 at the MRCISD+Q/aug-cc-pV5Z(-PP) level of theory.
Table 4 lists the nine MRCISD(+Q) calculated states along with their equilibrium products and asymptotic limits. Only four out of nine, i.e. X5Σ+, A7Π, 9Π(1), and 9Σ+(1) are single-reference states, while their equilibrium products coincide with their dissociation atomic limits. The remaining five states, a5Σ+, b5Π, 3Π(1), 3Σ+(1), and 3Δ(1), exhibit avoided crossing; thus equilibrium products differ from their adiabatic dissociation atomic limits, see Table 4 and they present a multireference character. Specifically, the a5Σ+, 3Π(1), and 3Σ+(1) states present a strong multi-reference character, i.e., the coefficient of their main CSF is 0.57, 0.55 and 0.47, respectively.
| State | D e (Ddiabatice) | Bonding | Equilibrium products | Asymptotic products |
|---|---|---|---|---|
| a van der Waals interaction, the symmetry of the interaction is given parenthesis. | ||||
| X7Σ+ | 13.9 | σ1 | Mo(a7S) + Be(1S) | Mo(a7S) + Be(1S) |
| a5Σ+ | 28.2 (60.3) | σ2σ2 | Mo(a7S) + Be(3P) | Mo(a5S) + Be(1S) |
| b5Π | 57.7 (86.7) | σ2σ1π2π1 | Mo(a7S) + Be(3P) | Mo(a5D) + Be(1S) |
| A7Π | 42.3 | σ2π1 | Mo(a7S) + Be(3P) | Mo(a7S) + Be(3P) |
| 3Δ(1) | 41.8 (92.0) | σ2π2π2 | Mo(a5G) + Be(3P) | Mo(a3G) + Be(1S) |
| 3Π(1) | 37.8 (88.0) | σ2σ1π2π1 | Mo(a5G) + Be(3P) | Mo(a3G) + Be(1S) |
| 3Σ+(1) | 34.1 (67.2) | σ2σ2 | Mo(a5S) + Be(3P) | Mo(a3G) + Be(1S) |
| 9Π(1) | 10.4 | vdW: (σ π)a | Mo(a7S) + Be(3P) | Mo(a7S) + Be(3P) |
| 9Σ+(1) | 2.5 | vdW: (σ)a | Mo(a7S) + Be(3P) | Mo(a7S) + Be(3P) |
Regarding the calculation of the dissociation energy, the adiabatic De of calculated states range from 2.5 kcal mol−1 to 57.7 kcal mol−1. The bonding ranges from van der Waals interactions to triple bonds. Note that the three states, namely b5Π, 3Δ(1), and 3Π(1), which have triple bonds, present avoided crossings and thus their diabatic De values, which are directly related with the strength of the bonding, are 86.7, 92.0 and 88.0 kcal mol−1, respectively. Interestingly, the bonding schemes behind the triplet and quintet states appear to be indistinguishable (apart from spins); i.e. the bonding scheme of a5Σ+ is identical to that of 3Σ+(1), and the bonding scheme of b5Π is identical to that of 3Π(1). Generally, in most states, electron charge is transferred from Be to Mo via the bonding, since the Be atom is the electropositive participating element. Finally, the corresponding bond distances range from 2.047 Å [3Δ(1)] to 2.787 Å [9Σ+(1)] in accordance with the size of the bond order.
Overall, the correlation between bond distances, bond order and dissociation energies are depicted in Fig. 9 and specifically, how the bonding affects them. While the Be atom possesses a closed shell valence configuration, it can participate in a multitude of bond schemes, i.e., offering and receiving half bonds, offering and receiving dative bonds, and forming covalent bonds. More specifically, in its ground state (1S), it can receive electron density in its vacant 2p valence orbitals, or offer its 2s electron lone pair density, and in its first excited state (3P), it can use its 2s, and possibly 2p, electron's spin for coupling, or have its 2p valence orbitals available for reception of electron density. In the MoBe molecule, it was found that it forms from weak interactions such as van der Waals (in 9Σ+(1)) and half-bond (in X7Σ+) or as high as triple bonds (in b5Π(1), 3Δ(1), and 3Π(1) states), see Fig. 9. It's noteworthy that, in most cases, it is found that the Be atom forms bonds in its first excited state; Be(3P). In most states, a 2s2pz hybridization is assumed on Be, while a 4dz25s5pz hybridization is assumed on Mo, and, in some cases, where the Be atom assumes no hybridization, Mo still showcases the same 4dz25s5pz hybridization, by itself (i.e. b5Π and A7Π). The bond distances of the calculated states range from as low as 2.047 Å, in 3Δ(1), as far as 2.787 Å, in 9Σ+(1), see Table 2, and Fig. 9. Generally, it is observed that the excited Be(3P) atom forms the shortest bonds, partially because its 2s orbital can accept easily electron density from the 4dz25s5pz orbitals of Mo, and thus form strong σ bonds, and partially owing to its capability of forming bonds both along the molecular axis, as well as laterally to it, resulting in strong bonds of high orders.
The adiabatic dissociation energies are depicted in Fig. 9. Additionally, the dissociation energy per bond, which corresponds to the bond strength, is obtained if the diabatic (related to the contributing towards bonding atomic configurations) dissociation energy is divided by the bond order 0.5 (half bond σ1 or π1 to 3). Regarding nonet 9Π(1) and 9Σ+(1) states, the 9Σ+(1) is clearly a van der Waals state, while in the 9Π(1) there is a weak interaction via σ and π frame. We can assume that each interaction is ∼10% the strength of typical Mo–Be covalent or dative bonds. Generally, it is considered that, the upper bound of a vdW interaction [alkyl⋯alkyl] is 5% of a typical covalent bond (e.g. C–H bond) and 7–20% of a typical dative bond (e.g. N→B bond), while the upper bound of a hydrogen bond (H⋯O) is 20% of the covalent bond;59 however, these % values correspond in bonds from different two atoms and for the same atoms like in our case. Thus, in the Mo–Be diatomic molecule, where the vdW interaction stronger than a typical alkyl⋯alkyl interaction, the estimation of each interaction as the ∼10% the strength of typical Mo–Be two electron bond is reasonable. It is interesting that in all calculated states, on average, the dissociation energy per bond of the seven calculated states is 29.8 kcal mol−1. Moreover, the corresponding value of the nine states, i.e. including also the two nonet states, is again 29.8 kcal mol−1, see Fig. 9. To sum up, we have concluded that the final value for a two-electron Mo–Be bond is 30 kcal mol−1.
Compounds containing interacting molybdenum and beryllium components in the bulk phase have been the focus of ongoing research for some time now, whether they be alloys, co-joint materials or interfaces and absorbed layers, aiming to identify their physical properties.7–14 Despite this, thorough inspection of the bonding taking place in these cases has been overlooked up until now, and there are no points of reference in order to identify bonding schemes or even predict them in such materials. Thus, shedding some light into the details of the interactions between molybdenum and beryllium in solid-state systems and diffuse of either Mo or Be in a bulk material can provide insights into their potential applications in optics, catalysis, catalysis, lithography, aerospace, thermal applications, and electronics. Overall, the present study can add physical insight into Mo–Be bonding, and enhance our understanding of these interactions in materials, offering valuable information for possible uses in the mentioned technologies.
4 Conclusions & final remarks
In the present study, we have explored the electronic structure and bonding of the ground and 42 low-lying states of the MoBe molecule, employing the (SA-)CASSCF and MRCISD(+Q) methodologies in conjunction with aug-cc-PV5Z(-PP) basis set. Bond distances, dissociation energies, dipole moments as well as common spectroscopic constants have been calculated while the bonding of selected calculated states is analyzed, and the variety of bonding schemes is further discussed.The four lowest in energy states, X7Σ+, a5Σ+, b5Π, A7Π, are clearly separated from the remaining excited states in all used methods. The first excited state a5Σ+ is lying 15.0 kcal mol−1 above the ground one. Regarding the X7Σ+ state, it was found that a σ1 dative bond is formed between the 4dz2 single electron of Mo and the vacant 2pz orbital of Be with a length of 2.462 Å. The dissociation energy was calculated at 13.85 kcal mol−1 at the MRCISD+Q/aug-cc-pV5Z(-PP) method considering the correlation of the valence electron.
The adiabatic De values of calculated states, considering the correlation of all ab initio treated electron, range from 2.5 (9Σ+(1)) to 57.7 kcal mol−1 (b5Π). Three states, namely b5Π, 3Δ(1), and 3Π(1) have triple bonds, and their diabatic De values, which are directly related to the bond strength, are 86.7, 92.0 and 88.0 kcal mol−1. The calculated bond distances range from 2.047 Å, in 3Δ(1), up to 2.787 Å, in 9Σ+(1) while, the dipole moment values range from −1.51 D, in X7Σ+, “up” to 3.28 D, in 9Σ+(1).
The present work analyzes the bonding of the MoBe molecule. The Be atom participates actively in the formation of bonds by contributing its single valence electrons of its ground or first excited atomic state to Mo and/or passively become the recipient of electron density in its vacant orbitals, despite their high energy. The arising bonds vary from van der Waals interactions (9Σ+(1)), and half-bonds (X7Σ+), to triple bonds (b5Π, 3Δ(1), and 3Π(1)). In most states, it is found a 2s2pz hybridization on Be and a 4dz25s5pz hybridization on Mo, while electron charge is transferred from Be to Mo. As far as we know, the electronic spectrum of MoBe has never been studied before experimentally or theoretically. The binding schemes of all calculated states are presented for the first time filling the gap in the literature regarding diatomic molecules. It was found that on average, a single covalent Mo–Be bond is about 30 kcal mol−1.
Overall, the present work highlights the exceptional ability of beryllium atoms to participate in a variety of bonding schemes, explaining experimental observations of chemically absorbed Be layers on Mo surfaces. Therefore, this study could provide an opening gate for further investigation of this species or associated material and complexes.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: energetics of all MoBe calculated states at the CASSCF, SA-CASSCF, MRCISD, MRCISD+Q/aug-cc-pV5Z(-PP) methods and potential energy curves of the ground and excited states of MoBe at MRCISD level of theory. See DOI: https://doi.org/10.1039/d5cp03025k.Acknowledgements
DT acknowledge computational time granted from the Greek Research & Technology Network (GRNET) in the National HPC facility ARIS under project ID pr015035-TrMeCo.References
- D. M. Dimiduk and J. H. Perepezko, MRS Bull., 2003, 28, 639–645 CrossRef.
- A. Stevanovic, I. Cvijovic-Alagic and N. Mitrovic, E3S Web Conf., 2024, 356, 02016 Search PubMed.
- A. N. Shmelev, G. G. Kulikov, B. K. Kozhahmet, E. G. Kulikov and V. A. Apse, Kerntechnik, 2016, 81, 596–598 CrossRef.
- B. Cheng, P. Chou and Y.-J. Kim, EPJ Nuclear Sci. Technol., 2016, 2, 5 CrossRef.
- G. Liu, G. J. Zhang, F. Jiang, X. D. Ding, Y. J. Sun, J. Sun and E. Ma, Nat. Mater., 2013, 12, 344–350 CrossRef PubMed.
- P. Uranga, C. J. Shang, T. Senuma and H. Mohrbacher, Adv. Manuf., 2020, 8, 15–34 CrossRef.
- S. G. Gordon, J. A. McGurty and G. E. Klein, et al. , JOM, 1951, 3, 637–638 CrossRef.
- C. R. Watts, Trans. Metall. Soc. AIME, 1969, 245, 329–333 Search PubMed.
- A. S. Panov and M. M. Rysina, J. Less-Common Met., 1977, 54, 1–8 CrossRef.
- T. Iwadachi, US Pat., 6176418B1, 2001 Search PubMed.
- A. Eftekhari, Appl. Surf. Sci., 2003, 220, 343–348 CrossRef.
- K. M. Skulina, C. S. Alford, R. M. Bionta, D. M. Makowiecki, E. M. Gullikson, R. Soufli, J. B. Kortright and J. H. Underwood, Appl. Opt., 1995, 34, 3727–3730 CrossRef PubMed.
- S. Bajt, J. Vac. Sci. Technol., A, 2000, 18, 557–559 CrossRef.
- T. V. Afanasieva, A. G. Fedorus, A. G. Naumovets and D. V. Rumiantsev, Surf. Sci., 2019, 682, 14–24 CrossRef.
- E. I. Stiefel and K. Othmer, Encyclopedia of Chemical Technology, Wiley, New York, 2001, p. 871 Search PubMed.
- J. F. Harrison, Chem. Rev., 2000, 100, 679–716 CrossRef PubMed.
- D. Tzeli, I. Karapetsas, D. M. Merriles, J. C. Ewigleben and M. D. Morse, J. Phys. Chem. A, 2022, 126, 1168–1181 CrossRef CAS PubMed.
- M. A. Mermigki, I. Karapetsas and D. Tzeli, ChemPhysChem, 2023, 24, e202300365 CrossRef CAS.
- B. M. Hoffman, D. Lukoyanov, Z.-Y. Yang, D. R. Dean and L. C. Seefeldt, Chem. Rev., 2014, 114, 4041–4062 CrossRef CAS PubMed.
- M. V. White, E. E. Claveau, E. Miliordos and K. D. Vogiatzis, J. Phys. Chem. A, 2024, 128, 2038–2048 CrossRef CAS.
- A. Androutsopoulos, D. Tzeli, K. H. Tomchak and M. D. Morse, J. Chem. Phys., 2024, 160, 234304 CrossRef CAS.
- T. Depastas, A. Androutsopoulos and D. Tzeli, J. Chem. Phys., 2022, 157, 054302 CrossRef CAS PubMed.
- L. Zhang, W. Zou, Y. Yu, D. Zhao, X. Maa and J. Yang, J. Quant. Spectrosc. Radiat. Transfer, 2021, 269, 107690 CrossRef CAS.
- J. J. Sorensen, E. Tieu, A. Sevy, D. M. Merriles, C. Nielson, J. C. Ewigleben and M. D. Morse, J. Chem. Phys., 2020, 153, 074303 CrossRef CAS.
- C. Demetriou, C. E. Tzeliou, A. Androutsopoulos and D. Tzeli, Molecules, 2023, 28, 8016 CrossRef PubMed.
- J. M. Merritt, V. E. Bondybey and M. C. Heaven, Science, 2009, 324, 1548–1551 CrossRef.
- J. T. Boronski, A. E. Crumpton, L. L. Wales and S. Aldridge, Science, 2023, 380, 1147–1149 CrossRef; J. Q. Nguyen, A. Rajabi, A. J. Wang, J. W. Ziller, F. Furche and W. J. Evans, Organometallics, 2023, 42, 2185–2188 CrossRef.
- G. Wang, J. E. Walley, D. A. Dickie, S. Pan, G. Frenking and R. J. Gilliard Jr., J. Am. Chem. Soc., 2020, 142, 4560–4564 CrossRef PubMed; C. Berthold, J. Maurer, J. Langer, M. Morasch, S. Harder and G. Frenking, Angew. Chem., Int. Ed., 2024, 63, e202408422 CrossRef PubMed; M. A. Gosch, S. Pan, C. Poggel, N. Spang, M. Müller, G. Frenking and J. Sundermeyer, Chem. – Eur. J., 2024, 30, e202402118 CrossRef PubMed.
- M. Lesiuk, M. Przybytek, J. G. Balcerzak, M. Musiał and R. Moszynski, J. Chem. Phys., 2018, 148, 224305 Search PubMed; A. V. Mitin, A. A. Gusev, O. Chuluunbaatar, S. I. Vinitsky, V. L. Derbov and L. Le Hai, J. Chem. Phys., 2023, 159, 124307 CrossRef PubMed.
- N. Fröhlich, U. Pidun, M. Stahl and G. Frenking, Organometallics, 1997, 16, 528–537 CrossRef; M. Yáñez, P. Sanz, O. Mó, I. Alkorta and J. Elguero, J. Chem. Theory Comput., 2009, 5, 2763–2771 CrossRef PubMed.
- K. Eskandari, J. Mol. Model., 2012, 18, 3481–3487 CrossRef PubMed; E. Fernández Villanueva, O. Mó and M. Yáñez, Phys. Chem. Chem. Phys., 2014, 16, 17531–17536 RSC.
- K. Iversen, S. Couchman, D. Wilson and J. L. Dutton, Coord. Chem. Rev., 2015, 297–298, 40–48 CrossRef; M. E. Alikhani, J. Mol. Model., 2020, 26, 94 CrossRef PubMed.
- J. Brandejs, L. Veis, S. Szalay, G. Barcza, J. Pittner and Ö. Legeza, J. Chem. Theory Comput., 2023, 19, 1112–1126 Search PubMed.
- L. Wang, S. Pan, G. Wang, X. Zeng, M. Zhou and G. Frenking, Chem. Commun., 2022, 58, 8532–8535 RSC; R. Liu, L. Qin, Z. Zhang, L. Zhao, F. Sagan, M. Mitoraj and G. Frenking, Chem. Sci., 2023, 14, 4872–4887 RSC; M. R. Buchner, S. Pan, C. Poggel, N. Spang, M. Müller, G. Frenking and J. Sundermeyer, Chem. – Eur. J., 2024, 30, e202400966 CrossRef CAS.
- D. Naglav, A. Neumann, D. Bläser, C. Wölper, R. Haack, G. Jansen and S. Schulz, Chem. Commun., 2015, 51, 3889–3891 RSC; G. Wang, J. E. Walley, D. A. Dickie, A. Molino, D. J. D. Wilson and R. J. Gilliard Jr., Angew. Chem., Int. Ed., 2021, 60, 9407–9411 CrossRef CAS; C. Helling, D. J. D. Wilson and C. Jones, J. Am. Chem. Soc., 2025, 147, 16620–16629 CrossRef.
- B. Jin, X.-L. Guan, M. Yan, Y.-J. Wang and Y.-B. Wu, Chem. – Eur. J., 2023, 29, e202302672 CrossRef CAS.
- X. Liu, M. Zhang, S. Yu, Y. Geng, X. Zhang, Y. Ding and Z. Su, Phys. Chem. Chem. Phys., 2018, 20, 23898–23902 RSC.
- H. Rzepa, Henry Rzepa's Blog, 2022 DOI:10.14469/hpc/10125.
- D. Tzeli and I. N. Karapetsas, J. Phys. Chem. A, 2020, 124, 6667–6681 CrossRef CAS PubMed.
- D. Tzeli, J. Comput. Chem., 2021, 42, 1126–1137 CrossRef CAS PubMed.
- A. Rodriguez, S. Vadachkoria, F. A. Evangelista and M. C. Heaven, J. Phys. Chem. A, 2025, 129, 2041–2048 CrossRef CAS PubMed.
- A. Androutsopoulos and D. Tzeli, ACS Omega, 2025, 10, 40174–40189 CrossRef CAS.
- C. Demetriou and D. Tzeli, Molecules, 2025, 30, 2874 CrossRef CAS PubMed.
- O. Roos, P. R. Taylor and P. E. M. Siegbahn, Chem. Phys., 1980, 48, 157–173 CrossRef.
- H.-J. Werner and E. A. Reinsch, J. Chem. Phys., 1982, 76, 3144 CrossRef CAS; P. J. Knowles and H.-J. Werner, Chem. Phys. Lett., 1988, 145, 514 CrossRef.
- H.-J. Werner and P. J. Knowles, J. Chem. Phys., 1988, 89, 5803–5814 CrossRef CAS.
- K. A. Peterson, D. Figgen, M. Dolg and H. Stoll, J. Chem. Phys., 2007, 126, 124101 CrossRef PubMed.
- B. P. Prascher, D. E. Woon, K. A. Peterson, T. H. Dunning Jr and A. K. Wilson, Theor. Chem. Acc., 2011, 128, 69–82 Search PubMed.
- M. Dolg, H. Stoll, H. Preuß and P. Schwerdtfeger, Theor. Chem. Acc., 1997, 97, 191–199 Search PubMed.
- H.-J. Werner and P. J. Knowles, J. Chem. Phys., 1988, 89, 5803 CrossRef CAS.
- S. R. Langhoff and E. R. Davidson, Int. J. Quantum Chem., 1974, 8, 61 CrossRef CAS; M. R. A. Blomberg and P. E. M. Siegbahn, J. Chem. Phys., 1983, 78, 5682–5692 CrossRef.
- J. L. Dunham, Phys. Rev., 1932, 41, 713 CrossRef.
- R. S. Mulliken, J. Chem. Phys., 1955, 23, 1833–1840 CrossRef.
- A. E. Reed, R. B. Weinstock and F. Weinhold, J. Chem. Phys., 1985, 83, 735–746 CrossRef; S. Ishikawa, G. Madjarova and T. Yamabe, J. Phys. Chem. B, 2001, 105, 11986–11993 CrossRef; D. Tzeli, I. Petsalakis and G. Theodorakopoulos, J. Phys. Chem. A, 2011, 115, 11749–11760 CrossRef PubMed.
- P. J. Knowles, G. Knizia, F. R. Manby, M. Schütz, P. Celani, W. Györffy, D. Kats, T. Korona, R. Lindhet al., MOLPRO 2015.1 is a package of ab initio programs written by H.-J. Werner.
- F. Coester and H. Kiimmel, Nucl. Phys., 1960, 17, 477–485 CrossRef; K. Raghavachari, G. W. Trucks, J. A. Pople and M. Head-Gordon, Chem. Phys. Lett., 1989, 157, 479–483 CrossRef.
- A. Kramida and W. C. Martin, J. Phys. Chem. Ref. Data, 1977, 26, 1185–1194 CrossRef.
- J. Sugar and A. Musgrove, J. Phys. Chem. Ref. Data, 1988, 17, 155–239 CrossRef; A. Kramida, Y. Ralchenko, J. Reader, K. Olsen and R. Ibacache, NIST Standard Reference Database 78, Version 5.12 DOI:10.18434/T4W30F.
- F. Biedermann and H.-J. Schneider, Chem. Rev., 2016, 116, 5216–5300 CrossRef PubMed.
| This journal is © the Owner Societies 2025 |