Open Access Article
Open Access ArticleKinetic modeling of ammonia and hydrogen dissociative co-adsorption on iron surface and its effect on hydrogen embrittlement
Nan
Zhang
*ac,
Kentaro
Wada
a,
Ryuoske
Komoda
b,
Aleksandar
Staykov
 a and
Masanobu
Kubota
*a
a and
Masanobu
Kubota
*a
aInternational Institute for Carbon Neutral Research, Kyushu University, Fukuoka, Japan
bDepartment of Mechanical and Control Engineering, Kyushu Institute of Technology, Kitakyushu, Japan
cDepartment of Mechanical Engineering, Nagaoka University of Technology, Nagaoka, Japan. E-mail: nanzhang@vos.nagaokaut.ac.jp
First published on 28th October 2025
Abstract
We study the mitigation of environmental hydrogen embrittlement of iron by ammonia impurities in the hydrogen gas using combined theoretical and experimental methods. The competitive and dissociative co-adsorption of gaseous ammonia and gaseous hydrogen on the iron surface was investigated using density functional theory. The surface adsorption and decomposition of ammonia, as well as the ammonia partial pressure, were considered as influential factors contributing to the control of atomistic hydrogen uptake into the material. To elucidate the mechanism of ammonia competing with hydrogen on the iron surface and of ammonia mitigating hydrogen embrittlement, we develop kinetic modeling that can estimate the reaction rate and the dynamic surface coverage of different adsorbed species on the iron surface. The reaction rates for hydrogen and ammonia co-adsorption and dissociation were calculated using transition state theory combined with the Langmuir adsorption model, and a fracture toughness test was conducted to validate theoretical results. The adsorption rate of ammonia on iron is significantly higher compared to hydrogen, thus, ammonia hinders the hydrogen adsorption on the iron surface. However, partial pressure dependent ammonia decomposition also provides hydrogen atoms, which induce hydrogen embrittlement. The theoretical results of this study were supported well by experimental fracture toughness test results.
1. Introduction
The combination of environmental and economic factors led to the wider adoption of hydrogen as an energy source and energy storage compound. The abundance of renewable energy sources combined with steam electrolyzes could successfully convert renewable energy into chemical fuel (H2) that can be stored and transported over long distances. Hydrogen can be utilized in the chemical industry, in fuel cells for stationary and mobile generation of electricity, and in combustion turbines on its own or in mixtures with hydrocarbons. However, hydrogen causes degradation of material strength of metal components such as storage containers and pipelines, which is generally known as hydrogen embrittlement (HE) of metals. HE is caused by the diffusion of hydrogen atoms into the metal lattice, leading to the formation of brittle hydride phases,1 weakening of metal bonds,2 enhancement of localized plasticity,3 and so on resulting in lower resistance to fracture, which can cause sudden and catastrophic failure. HE manifests as ductility loss in the tensile tests, fatigue crack growth acceleration, and fracture toughness reduction.4–8 In the gaseous hydrogen environment, hydrogen uptake from the environment into the material is the first step for HE of iron (Fe) and steel. In the process of hydrogen uptake at room temperature, the dissociation of hydrogen molecules into hydrogen atoms is assisted by catalysis of the iron surface.9 In this context, if the catalysis of the Fe surface is suppressed by surface catalytic poisons the HE could be significantly delayed.10–16 The strategy of adding ppm level gaseous impurities to the hydrogen gas to mitigate the HE is attracting more attention because the impurity mitigation strategy allows us to use the existing steel pipelines and storage vessels utilized for natural gas without the construction of expensive, hydrogen resistant infrastructure. For instance, the well-established natural gas pipeline could be employed for hydrogen gas transportation. The impurity mitigation strategy has the potential to accelerate the adoption of hydrogen energy at reduced cost. Nagao et al. experimentally demonstrated that exposure of the SCM435 low-alloy steel, whose surface is covered by a natural oxide layer, to 120 MPa H2 gas at room temperature caused no increase in the hydrogen concentration in the material.17 It implies that hydrogen uptake from H2 gas into the steel at room temperature requires fresh iron surface, which can act as a catalytic site for the dissociation of molecular hydrogen into atomic hydrogen. Fresh iron surface is created by crack propagation initially through the native oxide layer.18 Staykov et al. explained the role of the iron surface as a catalyst for the dissociation of H2 molecules by density functional theory (DFT) calculations and elucidated the mechanism of O2 mitigation effect on HE.19 The iron surface should be clean in order to act as a catalyst for the H2 dissociation. Preadsorbed oxygen on the iron surface hinders the catalytic activity by localization of the electron density. Somerday et al. considered that the hydrogen uptake from hydrogen gas occurs at the newly created surfaces during crack propagation. They experimentally confirmed both the hydrogen uptake through the surfaces created during crack propagation and the blocking of hydrogen uptake by preadsorbed oxygen based on their crack propagation tests of X52 steel.20In addition to oxygen, carbon monoxide (CO) is well-studied as a mitigator of HE.21,22 Komoda et al. clarified the mitigation effects of CO and O2 on the HE is focusing on their adsorption rates on the iron surface. Staykov et al. theoretically calculated the ratio of the impurity-occupied sites to free sites on the surface in order to interpret the result of the fracture toughness tests.21 They showed that O2 coverage is 100%, while that of CO is less than 75%.21,22 These results are useful for understanding the different HE mitigation properties of O2 and CO impurities. While both O2 and CO show excellent HE mitigation effects, there are some limitations to their industrial applications. For example, O2 forms explosive mixtures with H2 at higher concentrations, thus mixing O2 and H2 is considered a safety issue.23 CO is a catalytic poison for platinum nanoparticles in polymer electrolyte fuel cell and is an undesired impurity for industrial applications.24 CO mitigation effect is also limited to low pressure and short time intervals owing to the incomplete surface coverage. Thus, the mitigation effects of these impurities should be utilized in their respective suitable contexts. Therefore, to industrially use the mitigation effects of impurities more broadly, it is necessary to comprehensively elucidate the influences of other impurities besides O2 and CO.
In this context, we focused on ammonia (NH3) because it does not imply industrial safety issues and does not have a poisoning effect on the catalytic activity of platinum. However, the number of studies on the effect of NH3 on HE is very limited. Srikrishnan et al. reported that NH3 mitigated hydrogen-assisted crack growth of iron.25 They carried out the crack growth experiment of 4340 steel under H2 gas and H2 gas mixed with 50 ppm in volume (vppm) NH3 at 330 Pa of gas pressure, the result showed that the 50 vppm NH3 significantly reduced the crack growth speed compared with that in the pure H2. On the other hand, NH3 induces stress corrosion cracking, which is one of the forms of hydrogen embrittlement.26 Also, degradation of the tensile strength properties is induced in the NH3 environment.27 Thus, the effect of NH3 on HE is complicated as it can both mitigate and induce HE. The mitigation mechanism of NH3 on HE remains unclear.
In addition to first-principles energetics, a kinetic framework is needed to translate elementary adsorption and dissociation steps into observable surface coverages and rates under realistic gas compositions. A mean-field (Langmuir-type) microkinetic treatment provides this bridge by mapping DFT-derived barriers and adsorption energies to competitive and dissociative co-adsorption of NH3 and H2, thereby rationalizing how partial pressures and temperature control the instantaneous availability of catalytic sites and the net hydrogen supply to iron. Such models are widely used as a pragmatic baseline for trend-level comparisons and for screening mitigation strategies, especially when experiments alone cannot resolve fast interfacial steps. Here we adopt this framework to quantify NH3–H2 competition on Fe(110) and to connect surface reaction kinetics to hydrogen-embrittlement outcomes. The bcc Fe(110) facet is the thermodynamically most stable (lowest surface free energy) over the stability range of bcc iron, and thus a commonly exposed and extensively studied model surface. It is routinely prepared and characterized as a clean single-crystal surface in UHV studies.
In this work, we use first-principle methods to investigate the co-adsorption of NH3 and H2 on the iron Fe(110) surface, including NH3 molecular adsorption, NH3 decomposition, and its competition with H2 dissociation. We investigated the NH3 adsorption rate and decomposition rate based on the transition state theory. Furthermore, we investigate the effect of preadsorbed NH3 on the H2 dissociation. Those processes are significant factors that control the NH3 and H coverage on iron surface. We established a kinetic model to calculate the coverage of NH3, NH3-derived species, and H2-derived H atoms on the Fe surface and investigated the NH3 partial pressure effect on the NH3-derived H atom coverage. In addition, we performed a fracture toughness test of JIS SCM440 Cr–Mo steel in H2, NH3 and H2 mixture, NH3 and N2 mixture, and N2 gases under gas pressure were 0.1 MPa in order to validate the calculation results. This material suffered from hydrogen embrittlement.
2. Methods of investigation
2.1 Theory
Periodic DFT was performed using Numerical Atomic Orbitals (NAO) implemented in Quantum ATK.28 The method is based on quantum mechanics principles and can be used to investigate the structural and electronic properties of materials.29 The GGA Perdew–Burke–Ernzerhof (PBE) exchange–correlation functional was applied using Fritz-Haber-Institute (FHI) pseudopotentials. Electron energies were converged to 10−6 eV using double zeta polarized (DZP) basis set for all valence electrons. All calculations were spin polarized. The calculations were performed with Monkhorst–Pack k-point mesh of 8 × 8 × 8 (properly describe the properties of one unit cell for the BCC bulk iron lattice) and 4 × 4 × 1 for the Fe(110) terminated iron slab. The geometry relaxation was performed until the forces converged to values below 0.05 eV Å−1. For bulk systems, relaxation was performed for the cell volume, cell shape, and atomic positions. For slab models, relaxation was performed for the atomic positions only. Slabs were constructed using 6 alternating iron layers. The coordinates of the atoms in the middle two layers were fixed, while the coordinates of the top two and bottom two were fully relaxed. Activation barriers for various reaction mechanisms were obtained using the climbing image-nudged elastic band method (CI-NEB). In the process of NEB calculations, eight to ten images were used between the starting and the ending geometries of reactions. Adsorption energies and activation barriers are reported as 0 K DFT total energies without free-energy corrections. This pragmatic approximation is widely used in surface reaction studies30 (Exploring CO2 hydrogenation to methanol at a CuZn–ZrO2 interface via DFT calculations). These assumptions may not strictly hold for NH3/H co-adsorption on Fe(110); however, within the coverage range examined (11–33%), this approximation is widely used and adequate for trend-level comparisons. Moreover, dissociative H2 adsorption is treated with the standard √p(H2) dependence to reflect its two-site requirement. Coverage-dependent DFT energetics (Tables 3 and 5) partially account for lateral interactions by parameterizing rate/equilibrium constants at explicit coverages. Potential refinements (e.g., Fowler–Guggenheim/Temkin isotherm) are discussed in the SI-S1.In this study, the vibrational frequencies of normal modes of the decomposition reaction of NH3 and dissociation reaction of H2 were calculated by combining the dynamical matrix and vibrational mode calculation of Quantum ATK. Initial and transition geometry were converged to 10−8 eV to ensure that the vibrational frequency calculation yields proper results because vibrational frequency calculation requires well-converged electron density. Each ion in the cell is then displaced by ±0.01 Å in all directions of the cartesian vector, then a Hessian matrix was constructed from the force which shows how atoms react to displacement. Force derivative tolerance is set to 10−3 eV Å−1. Once the normal mode vibrational frequencies of the system were obtained, all data was (excluding the imaginary vibration of the transition state) divided by the product of the data from the initial geometry to obtain the attempt frequency.
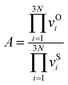 | (1) |
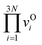 is multiplying all vibrational frequency at initial configuration,
is multiplying all vibrational frequency at initial configuration, 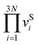 is multiplying all vibrational frequency at transition state configuration (has one more imaginary frequency).
is multiplying all vibrational frequency at transition state configuration (has one more imaginary frequency).
2.2 Experiment
The material used in this study was JIS SCM440 Cr–Mo low-alloy steel. The chemical composition is shown in Table 1. The mechanical properties after the heat treatments are shown in Table 2. The microstructure was tempered martensite with BCC lattice. The fracture toughness tests were carried out in accordance with the ASTM E 1820 standard. The NH3 partial pressure was 1000 ppm and 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ppm. The loading rate was 2.0 × 10−3 and 2.0 × 10−5 mm s−1. The temperature was 293 K. A detailed description of the fracture toughness test system and compact tension (CT) specimen is provided in the SI-S3. The authors have used the same test system to investigate the impurity mitigation effect on HE.
000 ppm. The loading rate was 2.0 × 10−3 and 2.0 × 10−5 mm s−1. The temperature was 293 K. A detailed description of the fracture toughness test system and compact tension (CT) specimen is provided in the SI-S3. The authors have used the same test system to investigate the impurity mitigation effect on HE.
| Fe | C | Si | Mn | P | S | Cu | Ni | Cr | Mo |
|---|---|---|---|---|---|---|---|---|---|
| Bal. | 0.40 | 0.22 | 0.63 | 0.008 | 0.004 | 0.02 | 0.02 | 0.94 | 0.15 |
| 0.2% proof strength (MPa) | Ultimate tensile strength (MPa) | Elongation | Reduction of area | Vickers hardness |
|---|---|---|---|---|
| 841 | 971 | 20% | 54% | HV329 |
3. Result and discussion
3.1 Theory
We optimize the lattice of BCC iron and obtain a lattice constant of 2.82 Å, which agrees with previous reports in the literature.30 The surface optimization yields a slightly shorter distance between the first and second layers and a slightly longer distance between the second and third layers. First, we investigate the surface geometry and surface coverage by NH3 molecules. The Fe(110) surface provides three distinct adsorption positions for NH3 molecules. Those positions include the top, bridge, and hollow sites. All initial geometries in the process of geometry optimization converge to top site of the Fe(110) surface as shown in Fig. 1. We used different quantities of NH3 molecules to cover the Fe surface, representing different coverage as shown in Fig. 2. We examined the adsorption energy of NH3 on Fe(110) to determine the effect of different surface coverages and it is summarized in Table 3. The adsorption energy of NH3 on the Fe(110) surface decreases with the increase of NH3 coverage. This phenomenon is based on two reasons: (1) the pre-adsorbed NH3 withdrew and localized electron density of the Fe surface, thus the electron density of the adjacent sites is no longer sufficient for the additional NH3 adsorption; (2) the repulsive interactions between the NH3 molecules. The most stable surface geometry is NH3 molecules occupying top sites with coverage in the range of 11–33%.| Coverage | 11% NH3 adsorbed on Fe(110) | 25% NH3 adsorbed on Fe(110) | 33% NH3 adsorbed on Fe(110) |
|---|---|---|---|
| Adsorption energy (eV) | −1.01 | −0.94 | −0.93 |
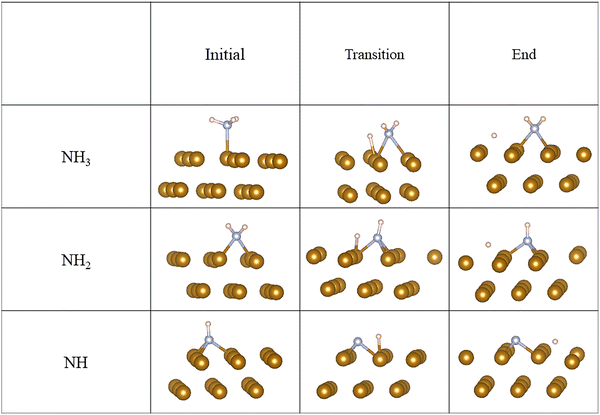
To fully understand the effect of NH3 on HE we investigated the decomposition of NH3 with a relatively low NH3 coverage (11%) on Fe(110) surface as shown in Fig. 3 This NH3 decomposition process consists of three distinct steps. During each step, NH3, NH2, and NH will release a hydrogen atom on the iron surface. The products of the NH3 and hydrogen decomposition occupied different sites on the surface and consequently affected the surface coverage as shown in Table 4. When gas molecules interact with surfaces, they establish equilibrium with the surface products which is governed by the adsorption law, the reaction kinetics, and the partial pressure of the gas components. An increase in the gas partial pressure leads to an increase in the coverage of the adsorption products on the solid surface at a constant temperature.31–33 Therefore, to understand the effect of NH3 partial pressure, a higher NH3 coverage (25%) on the Fe(110) surface as shown in Fig. 4 (NH3 decomposition on the iron surface at elevated coverage) was investigated. The summary of the activation energy barriers (Ea) of NH3 decomposition in each step (initial geometry, transition geometry, and end geometry) is shown in Table 5. The Ea for the decomposition of NH3 on the Fe(110) surface at 25% coverage is compared with that at 11% coverage. The first step of ammonia decomposition catalyzed by the Fe surface is affected by an increase in the surface coverage of ammonia. Additionally, as ammonia molecules only interact with individual Fe atoms, the Fe surface provides sufficient electrons to catalyze the decomposition of ammonia. Consequently, the Ea for the first step decreases with increased NH3 coverage. However, as NH3 decomposes into NH2 and NH, NH2 and NH interact with two and three Fe atoms, respectively. This leads to the withdrawal and localization of more electrons, thereby reducing the available free electrons for catalysis. Therefore, both the second step (NH2 → NH + H) and the third step (NH → N + H) of NH3 decomposition exhibit significant changes with increased NH3 coverage. The activation energy for the second decomposition step at 25% NH3 coverage nearly doubles compared to that at 11% NH3 coverage. Additionally, the reaction for the third decomposition step undergoes a transformation from an exothermic to an endothermic process as NH3 coverage increases. Consequently, the higher NH3 surface coverage mitigated NH3 decomposition better.
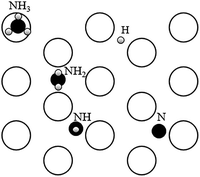
| Adsorbed species | Occupied site |
|---|---|
| NH3 | Top |
| NH2 | Bridge |
| H atom derived by NH3 | Bridge |
| NH | Hollow |
| H atom derived by NH2 | Bridge |
| N | Hollow |
| H atom derived by NH | Bridge |
| Decomposition activation barrier (11%) | Decomposition activation barrier (25%) | Desorption activation barrier (11%) | Desorption activation barrier (25%) | |
|---|---|---|---|---|
| Exo = exothermic reaction. Endo = endothermic reaction. | ||||
| NH3 → NH2 + H | 0.72 eV (Exo) | 0.68 eV (Exo) | 1.43 eV (Endo) | 1.39 eV (Endo) |
| NH2 → NH + H | 0.45 eV (Exo) | 0.84 eV (Exo) | 1.36 eV (Endo) | 1.70 eV (Endo) |
| NH → N + H | 0.99 eV (Exo) | 1.05 eV (Endo) | 1.18 eV (Endo) | 0.98 eV (Exo) |
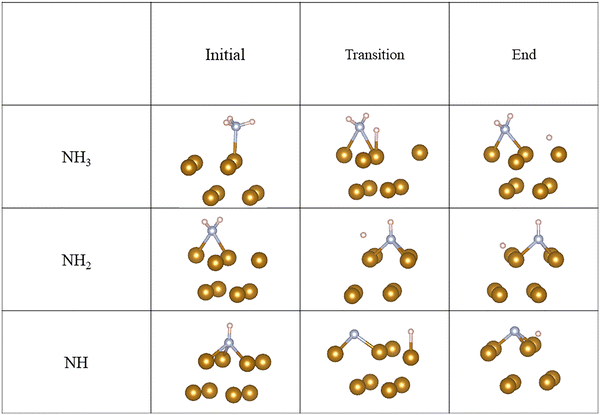
We further investigate the H2 dissociation process on the pure Fe(110) surface and Fe(110) surfaces pre-treated by NH3 with different coverage. The geometries are summarized in Fig. 5. The activation energy barrier and dissociation energy of H2 on the Fe(110) surface is shown in Table 6. Without NH3 pre-adsorbed on the Fe(110) surface, the H2 spontaneously dissociates on Fe(110) surface without an activation barrier. The dissociation energy of H2 is −1.56 eV. At 11% coverage, the activation energy barrier for H2 dissociation on Fe(110) surface is 0.04 eV, and the dissociation energy of H2 is −1.54 eV. The dissociation energy slightly increased from 0.00 eV to 0.04 eV, which indicates that even at low coverage NH3 can hinder H2 dissociation. With the increase of NH3 coverage on the Fe(110) surface from 11% to 33%, the activation barrier of H2 dissociation increased up to 0.7 eV and the dissociation energy of H2 decreased to −1.17 eV. The activation energy barrier would reduce the H2 dissociation reaction rate. Decreasing the dissociation energy of H2 would lead to easier H2 molecule desorption. As a result, an increased activation energy barrier and decreased dissociation energy would lead to a decrease in the hydrogen atom concentration on the surface, thus, reducing the probability for hydrogen to permeate to Fe.
| Coverage | 0% NH3 pre-adsorb Fe(110) | 11% NH3 pre-adsorb Fe(110) | 25% NH3 pre-adsorb Fe(110) | 33% NH3 pre-adsorb Fe(110) |
|---|---|---|---|---|
| Activation energy (eV) | 0.00 | 0.04 | 0.06 | 0.07 |
| Dissociation energy (eV) | −1.56 | −1.54 | −1.22 | −1.17 |
Chemical equilibrium is established by the mass fluxes between the gas phase and the surface equalize.34 For molecules in contact with a solid surface at a fixed temperature, the Langmuir isotherm,35 describes the partitioning between the gas phase and adsorbed species as a function of applied pressure. The basic assumption of this theory is: (1) assume the surface is homogeneous (no corrugations); (2) all sites are energetically equivalent, and the energy of adsorption is equal for all sites; (3) the gas adsorbs into an immobile state; (4) mono-layer coverage only; (5) no interactions between adsorbate molecules on adjacent sites.36,37 The Langmuir theory consisted of different adsorption models: adsorption, competitive adsorption, dissociative adsorption, and competitive and dissociative adsorption. In this study, we investigate the competitive and dissociative adsorption of NH3 and H2 on the Fe(110) surface. Because NH3–H2 co-adsorption shows coverage effects, we benchmarked our conclusions against an interaction-inclusive isotherm following Christensen et al.;38 details and sensitivity results are in SI-S1.
For the single adsorbate case, the model assumes adsorption and desorption as being elementary processes, where the rate of adsorption rad and the rate of desorption rd are given by
| rad = kadpA[S] | (2) |
| rd = kd[Aad] | (3) |
At equilibrium, the rate of adsorption is equal to the rate of desorption. Setting rad = rd, thus, the equilibrium constant
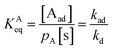 | (4) |
In eqn (4), the rate is given by the Arrhenius equation.39 The equilibrium constant is independent of the pressure of the system or of the concentration of the reacting species. If the entire surface is covered by adsorbed species A and free sites, and we apply eqn (4) we can derive the Langmuir adsorption isotherm,
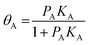 | (5) |
For competitive adsorption, the surface is covered by A, B, and free sites, therefore, the expressions for adsorbed species of A and B in surface coverage θA and θB are as follows:
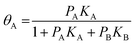 | (6) |
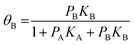 | (7) |
However, in the case of dissociation adsorption, the 1/2 power on pD2 arises because one gas phase molecule produces two adsorbed species.
In this study, we have performed surface coverage estimation based on the Langmuir theory. The Langmuir model neglects explicit adsorbate–adsorbate interactions and spatial correlations; such effects can matter when co-adsorbates interact or when pair-site requirements apply, and more detailed treatments (Fowler–Guggenheim/Temkin, cluster/Monte-Carlo or pair-site LH variants) may be used when needed. Here our objective is to rationalize relative trends; under these conditions, mean-field microkinetics remains a standard and defensible baseline. The H2 molecule dissociates into two H atoms and can be solved by the 1/2 power on pH2. The complete NH3 decomposition on the Fe surface is into an N atom and three H atoms. The kinetic derivation of NH3 complete decomposition on the Fe surface, and complete NH3 decomposition competitive with H2 dissociation on the Fe surface are studied after considering NH3 adsorbed on Fe(110) surface as a single molecule.
3.2 NH3 mitigation effect on HE
To investigate the mechanism of NH3 mitigation effect on HE, we first considered the NH3 adsorbed on the Fe surface as a molecule (without decomposition) and performed the Langmuir adsorption model, which assumes the adsorbate behaves as an ideal gas under isothermal conditions.39–41 We apply the Langmuir competitive adsorption model in eqn (6) and (7), and further extend it to competitive and dissociative model due to H2 is dissociative adsorption on the Fe surface. The 1/2 power on H2 arises because hydrogen atoms occupy two sites on the surface. We used the data in Tables 5 and 6 to modify the Langmuir competitive adsorption model for the NH3 and H2 competitive and dissociative co-adsorption on the Fe(110) surface. The surface coverage equation for NH3 adsorption and H2 dissociative adsorption is given below (eqn (8) and (9), respectively). In those equations, θNH3 and θH are denoted by the fractional occupancy on the surface by NH3 or atomic hydrogen, respectively. The activation barriers for different NH3 coverage are taken from the DFT calculations, the temperature is set to 293 K, and high-purity H2 was considered with 1000 ppm and 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ppm partial pressure of NH3 to match the experiment conditions. We used two different coverage models to represent the different NH3 partial pressure on H2 gas because increased partial pressure leads to higher surface coverage.42,43 Thus, 1 NH3 on the 9 surface Fe model represents the lower partial pressure of NH3 in H2, and 1 NH3 on the 4 surface Fe model represents the higher partial pressure of NH3 in H2.
000 ppm partial pressure of NH3 to match the experiment conditions. We used two different coverage models to represent the different NH3 partial pressure on H2 gas because increased partial pressure leads to higher surface coverage.42,43 Thus, 1 NH3 on the 9 surface Fe model represents the lower partial pressure of NH3 in H2, and 1 NH3 on the 4 surface Fe model represents the higher partial pressure of NH3 in H2.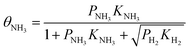 | (8) |
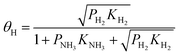 | (9) |
In eqn (8) and (9)KNH3 and KH2 denote the NH3 and H2 adsorption/desorption equilibrium constants for activation barriers listed in Tables 5 and 6, which were calculated by DFT. PNH3 and PH2 denote the partial pressure of NH3 and H2, respectively. The rate constants were estimated using the Arrhenius equation, where A is a preexponential factor, R is the gas constant, T is the temperature, and Ea is the activation barrier. The equilibrium constants are computed as the ratio between the adsorption and the desorption rate constants. The preexponential factor is computed as the reaction attempt frequency which is given by the product of the vibration frequencies at the resting site divided by the product of vibration frequencies in the transition state (the single imaginary frequency is excluded). It is important to note that the preexponential factor differs for the forward and reversed reactions. While the vibration frequencies at transition state are the same, they differ for the starting and end point of the reaction (product and reactant). The vibration frequencies are obtained through Hessian deionization in the DFT calculations.
From the cooperative adsorption/desorption/dissociation model in eqn (8) and (9), we calculated the NH3 and atomic hydrogen coverage on the Fe surface with different NH3 partial pressure, the result is shown in Table 7. The hydrogen atom coverage on the Fe(110) surface is decreasing with the increase in NH3 coverage, which is achieved by increased NH3 partial pressure. There is no direct study related to the ratio of atomistic hydrogen surface coverage on iron surface and initiation of crack growth induced by HE. We have reported that hydrogen atom coverage on the iron surface is 0.6% with CO 75% coverage.16 Experimental results show that CO could mitigate the HE.16 For NH3 25% coverage on Fe(110) surface the atomic hydrogen coverage is 0.01%, which is below 0.60%. Thus, we can expect that NH3 could possibly mitigate HE if it could have a faster adsorption rate compared to H2.
| Condition | H2 + 1000 ppm NH3 | H2 + 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ppm NH3 000 ppm NH3 |
|---|---|---|
| θ NH3 | 10% | 25% |
| θ H | 1.70% | 0.01% |
Surface reactions can be classified into two generic types. The first includes reactions between two adsorbed species or between an adsorbed species and a vacant site (Langmuir–Hinshelwood process), which is fitting for our consideration of coadsorption between NH3 and H2. The other reaction is called the Eley–Rideal process, which is considering that adsorbed species form a product.
Based on the Langmuir–Hinshelwood process, for randomly distributed adsorbates on a surface in the absence of adsorbate-adsorbate interactions, the rate of reaction is given by eqn (10).
| rr = kθAθv | (10) |
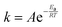 | (11) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ppm to estimate the hydrogen and NH3 reaction rates in Table 8.
000 ppm to estimate the hydrogen and NH3 reaction rates in Table 8.
| NH3 | H2 | |
|---|---|---|
| A (Hz) | 3.18 × 1020 | 3.90 × 1011 |
| k (s−1) | 3.14 × 1020 | 3.92 × 1011 |
| r r(11%NH3) (s−1) | 2.77 × 1019 | 5.85 × 109 |
| r r(25%NH3) (s−1) | 5.88 × 1019 | 2.92 × 107 |
With increasing the coverage of NH3 from 11% to 25%, the reaction rate of H2 with the Fe(110) surface decreases from 5.85 × 109 s−1 to 2.92 × 107 s−1. Thus, increasing NH3 partial pressure reduces hydrogen dissociation rate on Fe(110) surface. On the other hand, the adsorption rate of NH3 is significantly faster than H2, which indicates that NH3 preferentially adsorbs on the Fe(110) surface compared to H2.
3.3 Effect of NH3 decomposition on its mitigation effect on HE (while competing with H2 dissociation)
To fully understand the effect of NH3 competing with H2 with respect to HE, we divided the process of NH3 decomposition into four steps, and simultaneously considered the dissociation of H2. The decomposition mechanism of NH3 includes the chemisorption of NH3 on the catalyst surface,40,43–46 stepwise dehydrogenation of NH3 to NH2 + H, NH + H, N + H. The adsorption of all the species follows the Langmuir isotherm model, therefore it is assumed that the surface sites are energetically equivalent, and each site can hold at most one adsorbed species. The mechanism can be described according to the following sequence of elementary reactions as shown in (R1)–(R4):| NH3 + * ⇌ NH3* | (R1) |
| NH3* + * ⇌ NH2* + H* | (R2) |
| NH2* + * ⇌ NH* + H* | (R3) |
| NH* + * ⇌ N* + H* | (R4) |
To establish the kinetic model of competitive and dissociative co-adsorption between NH3 and H2 on the Fe(110) surface we must consider the H2 dissociation. The reaction of H2 dissociation on the Fe(110) surface can be expressed by the chemical reaction (R5) below:
| H2 + 2* ⇌ 2H* | (R5) |
The term * denotes a vacant site, and NH3*, NH2*, NH*, N*, and H* are the adsorbed species. NH3 and H2 represent the free gaseous state. If all the above reactions are equilibrium that the constants of equilibrium (K1, K2, K3, K4, and K5) are given by eqn (3) for each step. The reaction rate coefficient k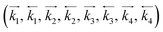 in each step is given by Arrhenius equation (eqn (11)).
in each step is given by Arrhenius equation (eqn (11)).
Then, we investigate the hydrogen atom surface coverage from the NH3 decomposition on the Fe(110) surface step by step and compare the competing H2 dissociation reaction. We assume that the reaction stops at each step and reaches equilibrium. Therefore, the whole process is divided into three cases denoted with 1, 2, and 3.
In order to keep the main text concise, we only summarize the setup of the staged NH3 decomposition while competing with H2 dissociation. We analyze three quasi-equilibrated stopping cases—Case 1 (NH3* ⇌ NH2* + H*), Case 2 (up to NH* + H*), and Case 3 (up to N* + H*)—with competitive dissociative H2 adsorption (H2 + 2* ⇌ 2H*). The corresponding equilibrium relations (K1–K5), Arrhenius parameterization, and the site balance are solved self-consistently at 293 K under the stated gas compositions. The full mass-balance equations, algebraic solution, and numerical procedure are provided in SI-S2, and the resulting coverages are reported in Table 9.
| H coverage 1000 vppm NH3 + H2 | H coverage 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 vppm NH3 + H2 000 vppm NH3 + H2 |
|
|---|---|---|
| Case 1 | 2.96% | 4.37% |
| Case 2 | 8.42% | 15.20% |
| Case 3 | 8.42% | 15.20% |
The hydrogen coverage of each case is summarized in Table 9. From Case 1 to Case 3, owing to the NH3 decomposition, the hydrogen atom coverage on Fe(110) surface significantly increased from 2.96% to 8.42% (1000 vppm NH3), 4.73% to 15.20% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 vppm NH3). The hydrogen atom coverage on Fe(110) surface increased with the NH3 decomposition and the NH3 partial pressure. The final step of NH3 decomposition to N and H has almost no contribution to H atom coverage on the Fe surface. However, the HE mitigation effect is not only affected by coverage, but also time dependent through the reaction rate. Therefore, we investigate the decomposition rate of NH3 at each step.
000 vppm NH3). The hydrogen atom coverage on Fe(110) surface increased with the NH3 decomposition and the NH3 partial pressure. The final step of NH3 decomposition to N and H has almost no contribution to H atom coverage on the Fe surface. However, the HE mitigation effect is not only affected by coverage, but also time dependent through the reaction rate. Therefore, we investigate the decomposition rate of NH3 at each step.
We use the Arrhenius equation (eqn (11)) where the pre-exponential factor A is taken from Table 8, the Ea is taken from Table 5, R is the universal gas constant, and T is 293 K for fitting with the fracture toughness test condition. The reaction rate coefficient of NH3 decomposition in each step can be calculated as shown in Table 10. Under the same NH3 partial pressure, the reaction rate coefficient of NH3, NH2, and NH is decreasing. For lower NH3 partial pressure (1000 vppm) we find a reduction in the reaction rate coefficient from NH2 to NH, which demonstrates that the last step of NH3 decomposition (NH → N + H) is the slowest step. However, the first two decomposition steps (NH3 → NH2 + H; NH2 → NH + H) are characterized with high reaction rate coefficients. When the NH3 partial pressure is increased to 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 vppm we find a reduction in the reaction rate coefficient from NH3 to NH2, which indicates that the NH3 decomposition is not likely to occur on the experimental time scale. Thus, during the experimental time scale, increased NH3 partial pressure will decrease the NH3 decomposition rate constant, and in a limited time scale, before the equilibrium is reached, it might successfully kinetically mitigate HE.
000 vppm we find a reduction in the reaction rate coefficient from NH3 to NH2, which indicates that the NH3 decomposition is not likely to occur on the experimental time scale. Thus, during the experimental time scale, increased NH3 partial pressure will decrease the NH3 decomposition rate constant, and in a limited time scale, before the equilibrium is reached, it might successfully kinetically mitigate HE.
| NH3 | NH2 | NH | |
|---|---|---|---|
| k (1000 vppm NH3) (s−1) | 1.368 × 108 | 7.284 × 105 | 2.130 × 10−1 |
k (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 vppm NH3) (s−1) 000 vppm NH3) (s−1) |
6.656 × 108 | 1.459 × 10−1 | 1.986 × 10−2 |
Our theoretical results at equilibrium show that the hydrogen coverage on Fe(110) surface increases with the increase in NH3 partial pressure. However, the NH3 reaction kinetics suggests that at short time intervals NH3 will decompose to NH2, NH and H at low partial pressure (1000 vppm) and only to NH2 and H at high partial pressure (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 vppm). Thus, the H surface coverage will be kinetically locked to 8.42% at 1000 vppm NH3 partial pressure and 4.37% at 10
000 vppm). Thus, the H surface coverage will be kinetically locked to 8.42% at 1000 vppm NH3 partial pressure and 4.37% at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 vppm NH3 partial pressure. As a result, increasing the NH3 partial pressure would have a mitigating effect on HE.
000 vppm NH3 partial pressure. As a result, increasing the NH3 partial pressure would have a mitigating effect on HE.
4. Fracture toughness test results
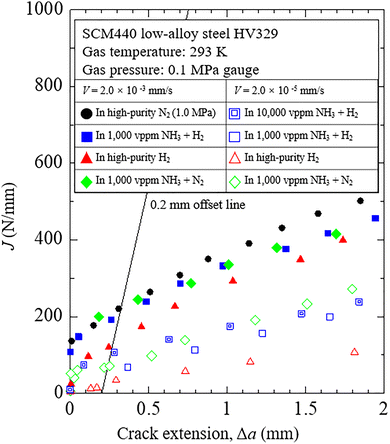
Hydrogen embrittlement is a phenomenon that includes a series of processes: surface adsorption/desorption, molecular dissociation, surface/sub-surface migration, hydrogen dissolution, defects trapping, grain boundaries segregation, hydrogen concentration ahead of the crack tip, etc. In this study, we designed an experiment where we kept all conditions the same and attempted to only relate the crack growth resistance ability of the material to the ammonia partial pressure and loading rate.The fracture toughness test results, shown in Fig. 6, illustrate the relationship between the J-integral value and crack extension (Δa). The fracture toughness of the material, determined by the J-integral value at the intersection of the J–Δa curve and the 0.2 mm offset line, is evaluated in accordance with the ASTM E1820 standard. This value represents the material's resistance to the onset of crack extension. The data obtained in high-purity N2 serves as the reference.
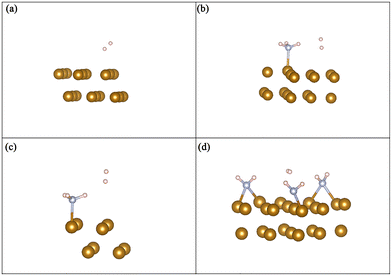 | ||
| Fig. 6 Molecular hydrogen dissociation on the Fe(110) surface with (a) 0%, (b) 11%, (c) 25%, and (d) 33% NH3 coverage. | ||
At the loading rate was 2.0 × 10−3 mm s−1, there was a reduction of fracture toughness of material in H2 gas. This result indicates that hydrogen embrittlement occurred. However, when 1000 ppm NH3 is added to H2 gas, the reduction is recovered. It was confirmed that NH3 has a mitigating effect on hydrogen embrittlement. The NH3 mitigation effect increased with increased NH3 partial pressure.
Additionally, we conducted fracture toughness tests in nitrogen gas with added NH3. If NH3 has neither a detrimental nor an improving effect, it is expected that the J–Δa curve would match that obtained in pure N2. During the fracture toughness test with 1000 ppm NH3 at a loading rate of 2.0 × 10−3 mm s−1, no reduction in fracture toughness was observed. However, surprisingly, when the loading rate was reduced to 1/100 (i.e., 2.0 × 10−5 mm s−1), the J–Δa curve for nitrogen gas with 1000 ppm NH3 shifted significantly downward, resulting in a marked decrease in fracture toughness.
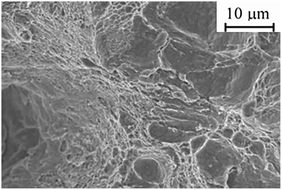
As shown in Fig. 7, the fracture surface obtained in nitrogen gas with 1000 ppm NH3 exhibited a quasi-cleavage fracture morphology, resembling the fracture surface obtained in the fracture toughness test in hydrogen. This observation suggests that hydrogen embrittlement occurred despite the absence of H2 gas in the test environment. A possible mechanism is the production of hydrogen atoms due to the decomposition of NH3 on the iron surface, facilitated by the catalytic action of the Fe surface (Fig. 8).
In summary, NH3 exhibits both mitigating and inducing effects on hydrogen embrittlement, depending on the loading rate. If this assumption is correct, the loading rate dependence of NH3's mitigating effect in H2 + NH3 environments can be clearly explained. As shown in Fig. 7, the addition of 1000 ppm NH3 significantly mitigated HE at a loading rate of 2 × 10−3 mm s−1. However, this mitigating effect drastically diminished when the loading rate was reduced to 2 × 10−5 mm s−1. According to the DFT simulation results shown in Table 8 and 10, the adsorption rate of NH3 on the Fe surface is significantly higher than its decomposition rate. Therefore, at a relatively high loading rate, crack propagation occurs while the Fe surface is predominantly covered by NH3 molecules, suppressing hydrogen uptake into the material. In contrast, at a sufficiently low loading rate, the slower crack propagation allows NH3 to decompose into H and NH2. The hydrogen produced from NH3 decomposition then becomes a source of hydrogen embrittlement, negating the mitigating effect of NH3.
However, Fig. 7 still presents a puzzling result. Specifically, in the fracture toughness test conducted at a loading rate of 2.0 × 10−5 mm s−1, the fracture toughness in the 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ppm NH3 + H2 environment was higher than that in the 1000 ppm NH3 + H2 environment. If the previous discussion—that NH3 decomposition produces hydrogen atoms at relatively low loading rates—is correct, it would be expected that increasing the NH3 concentration would further reduce fracture toughness due to an increase in hydrogen atom production. However, the observed result is the opposite.
000 ppm NH3 + H2 environment was higher than that in the 1000 ppm NH3 + H2 environment. If the previous discussion—that NH3 decomposition produces hydrogen atoms at relatively low loading rates—is correct, it would be expected that increasing the NH3 concentration would further reduce fracture toughness due to an increase in hydrogen atom production. However, the observed result is the opposite.
This anomaly can be explained by the results of the DFT calculations shown in Table 10. As shown in the figure, the NH3 decomposition rate (NH2 → NH + H) dramatically decreases with increasing NH3 partial pressure. This is because NH3 decomposition requires a vacant site adjacent to the adsorbed NH3 on the Fe surface. An increase in NH3 concentration leads to higher surface coverage of NH3 on the Fe surface, thereby reducing the availability of vacant sites. Consequently, the adsorbed NH3 loses the opportunity to decompose as the NH3 concentration increases.
5. Conclusion
NH3 was preferentially adsorbed on the Fe(110) surface because the adsorption rate of NH3 on the Fe(110) surface is significantly higher than H2. When the coverage of NH3 on the Fe(110) surface was 25%, the atomic hydrogen coverage on Fe(110) surface would be 0.01%. In conclusion, preferentially adsorbed NH3 mitigates the HE in gaseous hydrogen by hindering hydrogen uptake. The NH3 decomposition reaction has a strong time dependence because the adsorption rate of NH3 is much higher than its decomposition rate. Therefore, NH3 decomposition occurs only when reaction time is sufficient. The NH3 decomposition rate (NH2 → NH + H) decreased as NH3 partial pressure increased resulting in hindering NH3-derived hydrogen supply. As a result, insufficient time for NH3 further decomposition leads to lower H atom coverage on the Fe surface even at high NH3 partial pressures. Therefore, increased NH3 partial pressure could successfully mitigate HE through a kinetic mechanism. Our fracture toughness test results supported the theoretical results well. The test duration was controlled by the loading rate. Under the relatively higher loading rate, NH3 mitigates HE. However, as we reduced the loading rate, the NH3 mitigation effect on HE decreased due to its decomposition. Under the relatively slower loading rate, the NH3 mitigation effect increased with increased NH3 partial pressure. This kinetic modeling highlights two practical control parameters for hydrogen entry: NH3 surface coverage (via partial pressure) and exposure time scale (loading rate). At high coverage and short times, rapid NH3 adsorption suppresses H2 dissociation; over longer times, slow NH3 decomposition can supply H to the surface. These findings suggest pressure–time operating windows to limit hydrogen ingress and offer a transferable protocol for HE mitigation strategy.Conflicts of interest
There are no conflicts to declare.Data availability
The manuscript contains the computed results and detailed explanation how the simulations can be repeated and verified. In addition, supplementary information (SI) file is provided containing description of the experimental setup. In case further information is needed, it can be obtained via email from the corresponding author. Supplementary information is available. See DOI: https://doi.org/10.1039/d5cp02423d.Acknowledgements
This study was supported by JST SPRING, Grant Number JPMJSP2136. This study was supported by the World Premier International Research Center Initiative (WPI), MEXT, Japan. The International Institute for Carbon-Neutral Energy Research (WPI-I2CNER) is supported by the World Premier International Research Center Initiative (WPI), MEXT, Japan. Kyushu University platform of Inter-transdisciplinary Energy Research (Q-PIT), Japan.References
- D. Hardie and P. McIntyre, The low-temperature embrittlement of niobium and vanadium by both dissolved and precipitated hydrogen, Metall. Trans. A, 1973, 4, 1247–1254 CrossRef CAS.
- A. R. Troiano, The role of hydrogen and other interstitials in the mechanical behavior of metals, Trans. Am. Soc. Met., 1960, 52, 54–80 Search PubMed.
- H. K. Birnbaum and P. Sofronis, Hydrogen-enhanced localized plasticity—a mechanism for hydrogen-related fracture, Mater. Sci. Eng., A, 1994, 176, 191–202 CrossRef CAS.
- C. D. Beachem, A new model for hydrogen-assisted cracking (hydrogen “embrittlement”), Metall. Trans., 1972, 3, 441–455 CrossRef.
- S. Suresh and R. O. Ritchie, Mechanistic dissimilarities between environmentally influenced fatigue-crack propagation at near-threshold and higher growth rates in lower strength steels, Mater. Sci., 1982, 16, 529–538 CAS.
- D. Hardie and S. Liu, The effect of stress concentration on hydrogen embrittlement of a low alloy steel, Corros. Sci., 1996, 38, 721–733 CrossRef CAS.
- H. E. Townsend, Effects of zinc coatings on the stress corrosion cracking and hydrogen embrittlement of low-alloy steel, Metall. Mater. Trans. A, 1975, 6, 877–883 CrossRef.
- P. S. Lam, R. L. Sindelar, A. J. Duncan and T. M. Adams, Literature Survey of Gaseous Hydrogen Effects on the Mechanical Properties of Carbon and Low Alloy Steels, J. Press Vessel. Technol., 2009, 131, 1–14 Search PubMed.
- R. P. Gangloff, Gaseous hydrogen embrittlement of high strength steels, Metall. Mater. Trans. A, 1977, 8, 1043–1053 CrossRef.
- B. P. Somerday, P. Sofronis and K. A. Nibur, et al., Elucidating the variables affecting accelerated fatigue crack growth of steel in hydrogen gas with low oxygen concentrations, Acta Mater., 2013, 61, 6153–6170 CrossRef CAS.
- R. Komoda, K. Yamada and M. Kubota, et al., The inhibitory effect of carbon monoxide contained in hydrogen gas environment on hydrogen-accelerated fatigue crack growth and its loading frequency dependency, Int. J. Hydrogen Energy, 2019, 44, 29007–29016 CrossRef CAS.
- A. Staykov, J. Yamabe and B. P. Somerday, Effect of hydrogen gas impurities on the hydrogen dissociation on iron surface, Int. J. Quantum Chem., 2014, 114, 626–635 CrossRef CAS.
- A. Staykov, R. Komoda and M. Kubota, et al., Coadsorption of CO and H2 on an Iron Surface and Its Implication on the Hydrogen Embrittlement of Iron, J. Phys. Chem. C, 2019, 123, 30265–30273 CrossRef CAS.
- R. Komoda, M. Kubota and S. Yoshida, et al., Inhibition of Hydrogen Embrittlement of Cr-Mo Steel by the Addition of Impurities to Hydrogen Environment and the Effect of Material Strength. Paper presented at the The 28th International Ocean and Polar Engineering Conference, Sapporo, Japan, June 2018.
- R. Komoda, M. Kubota and A. Staykov, et al., Inhibitory effect of oxygen on hydrogen-induced fracture of A333 pipe steel, FFEMS, 2019, 42, 1387–1401 CAS.
- J. H. Holbrook, H. J. Cialone and E. W. Collings, et al., Control of hydrogen embrittlement of metals by chemical inhibitors and coatings, in Woodhead Publishing Series in Metals and Surface Engineering, Gaseous Hydrogen Embrittlement of Materials in Energy Technologies, ed. R. P. Gangloff and B. P. Somerday, Woodhead Publishing, 2012, ch. 5, vol. 1, pp. 129–153 Search PubMed.
- A. Nagao, J. Koga, S. Takagi, H. Ono and N. Ishikawa, Experimental and computational studies on hydrogen entry into steel exposed to high-pressure hydrogen gas, ISIJ conference, Tokyo, Japan, 2018 Search PubMed.
- F. Bakhshi and N. Farhadian, Co-doped graphene sheets as a novel adsorbent for hydrogen storage: DFT and DFT-D3 correction dispersion study, Int. J. Hydrogen Energy, 2018, 43, 8355–8364 CrossRef CAS.
- A. Staykov, J. Yamabe and B. P. Somerday, Effect of hydrogen gas impurities on the hydrogen dissociation on iron surface, Int. J. Quantum Chem., 2014, 114, 626–635 CrossRef CAS.
- B. P. Somerday, P. Sofronis and K. A. Nibur, et al., Elucidating the variables affecting accelerated fatigue crack growth of steel in hydrogen gas with low oxygen concentrations, Acta Mater., 2013, 61, 6153–6170 CrossRef CAS.
- A. Staykov, R. Komoda and M. Kubota, et al., Coadsorption of CO and H2 on an Iron Surface and Its Implication on the Hydrogen Embrittlement of Iron, J. Phys. Chem. C, 2019, 123, 30265–30273 CrossRef CAS.
- G. T. Gunasooriya, et al., CO Adsorption on Pt(111): From Isolated Molecules to Ordered High-Coverage Structures, ACS Catal., 2018, 8, 10225–10233 CrossRef CAS.
- C. Samanta, Direct synthesis of hydrogen peroxide from hydrogen and oxygen: An overview of recent developments in the process, Appl. Catal., A, 2008, 350, 133–149 CrossRef CAS.
- A. Manasilp and E. Gulari, Selective CO oxidation over Pt/alumina catalysts for fuel cell applications, Appl. Catal., B, 2002, 37, 17–25 CrossRef CAS.
- V. Srikrishnan, Selective adsorption and hydrogen embrittlement, Metall. Mater. Trans. A, 1976, 11, 1669–1675 CrossRef.
- G. T. Gunasooriya, et al., CO Adsorption on Pt(111): From Isolated Molecules to Ordered High-Coverage Structures, ACS Catal., 2018, 8, 10225–10233 CrossRef CAS.
- M. R. Louthan and R. E. Swanson, Material defects, gas purity and hydrogen embrittlement, Int. J. Hydrogen Energy, 1985, 10, 551–554 CrossRef CAS.
- S. Smidstrup and T. Markussen, et al., Quantum ATK: an integrated platform of electronic and atomic-scale modelling tools, J. Phys. Condens., 2019, 32, 1–36 Search PubMed.
- J. R. Chelikowsky and S. G. Louie, First-principles linear combination of atomic orbitals method for the cohesive and structural properties of solids: application to diamond, Phys. Rev. B: Condens. Matter Mater. Phys., 1984, 29, 6–15 CrossRef.
- J. A. Yan, C. Y. Wang and S. Y. Wang, Generalized-stacking-fault energy and dislocation properties in bcc Fe: a first-principles study, Phys. Rev. B: Condens. Matter Mater. Phys., 2000, 70, 1–5 Search PubMed.
- G. M. Hermse and F. Frechard, et al., Combining density-functional calculations with kinetic models: NO/Rh(111), J. Chem. Phys., 2003, 118, 7081–7089 CrossRef.
- F. Mafuné, J. M. Bakker and S. Kudoh, Dissociative adsorption of NO introduces flexibility in gas phase Rh6+ clusters leading to a rich isomeric distribution, Chem. Phys. Lett., 2021, 780, 138937 CrossRef.
- M. H. John and T. Ziegler, Density Functional Theory and Kinetic Studies of Methanation on Iron Surface, J. Phys. Chem. C, 2007, 111, 11012–11025 CrossRef.
- M. W. Lindauer, The evolution of the concept of chemical equilibrium from 1775 to 1923, J. Chem. Educ., 1962, 39, 384–390 CrossRef CAS.
- I. Langmuir, The Adsorption of Gases on Plane Surface of Glass, Mica and Platinum, J. Am. Chem. Soc., 1918, 40, 1361–1402 CrossRef CAS.
- I. Langmuir, “Part I” The Research Laboratory of the General Electric Company, 1916, p. 2221 Search PubMed.
- I. Langmuir, “Part II” The Research Laboratory of the General Electric Company, 1918, p. 1848 Search PubMed.
- S. Hansen, An Adsorption Isotherm That Includes the Interactions Between Adsorbates, J. Phys. Chem. C, 2025, 129, 5393–5407 CrossRef CAS.
- S. Arrhenius, Quantitative relationship between the rate a reaction proceed and its temperature, J. Phys. Chem., 1889, 4, 226–248 Search PubMed.
- A. Staykov, R. Komoda and M. Kubota, et al., Coadsorption of CO and H2 on an Iron Surface and Its Implication on the Hydrogen Embrittlement of Iron, J. Phys. Chem. C, 2019, 123, 30265–30273 CrossRef CAS.
- A. Staykov, S. Fukumori and K. Yoshizawa, et al., Interaction of SrO-terminated SrTiO3 surface with oxygen, carbon dioxide, and water, J. Mater. Chem. A, 2018, 6, 22662–22672 RSC.
- G. Pekridis, K. Kalimeri and N. Kaklidis, et al., Study of the reverse water gas shift (RWGS) reaction over Pt in a solid oxide fuel cell (SOFC) operating under open and closed-circuit conditions, Catal. Today, 2007, 127, 337–346 CrossRef CAS.
- W. Tsai and W. H. Weinberg, Steady-State Decomposition of Ammonia on the Ru(001) Surface, J. Phys. Chem., 1987, 91, 5302–5307 CrossRef CAS.
- G. Ertl, Surface Science and Catalysis—Studies on the Mechanism of Ammonia Synthesis: The P. H. Emmett Award Address, Chem. Rev., 1980, 21, 201–223 CAS.
- Z. Kowalczyk, J. Sentek, S. Jodzis, M. Muhler and O. Hinrichsen, Effect of Potassium on the Kinetics of Ammonia Synthesis and Decomposition over Fused Iron Catalyst at Atmospheric Pressure, J. Catal., 1997, 167, 407–414 CrossRef.
- G. Centi and S. Perathoner, Adsorption and Reactivity of No on Copper-on-Alumina Catalysts: II. Adsorbed Species and Competitive Pathways in the Reaction of No with NH3 and O2, J. Catal., 1995, 152, 93–102 CrossRef CAS.
| This journal is © the Owner Societies 2025 |