Photochemistry of dibromine peroxide, BrOOBr
Wiem
Chebbi
 *ab,
Najoua
Derbel
*ab,
Najoua
Derbel
 ac,
Thibaud
Cours
ac,
Thibaud
Cours
 b and
Alexander
Alijah
b and
Alexander
Alijah
 b
b
aLSAMA, Laboratoire de Spectroscopie Atomique Moléculaire et Applications, Department of Physics, University Tunis – El Manar, 1060 Tunis, Tunisia
bGSMA, Groupe de Spectrométrie Moléculaire et Atmosphérique, UMR CNRS 7331, University of Reims Champagne-Ardenne, 51100 Reims, France. E-mail: wiem.chebbi@univ-reims.fr
cFSB, Faculty of Sciences of Bizerte, University of Carthage, 7021 Jarzouna – Bizerte, Tunisia
First published on 15th July 2025
Abstract
The photochemistry of dibromine peroxide was investigated by means of mixed quantum–classical trajectory calculations on seven coupled potential energy surfaces. Photoexcited BrOOBr disintegrates rapidly, at a rate comparable with thermal decomposition. The rate constant is J = 9.7 × 10−2 s−1. The mechanism was investigated, and the final photolysis products are shown to be mainly 2Br + O2, though there is a minor channel leading to 2BrO. Their formation involves an unusual intermolecular vibronic energy redistribution process. A hitherto unknown isomer of BrOOBr, the van der Waals complex O2(a1Δg)·Br2(X1Σ+g), was found, which is more stable than the peroxide by 1.2 mEh.
1. Introduction
Free halogen atoms and halogen oxides are mainly responsible for the depletion of stratospheric ozone by converting it into dioxygen. Evidence for a catalytic cycle involving chlorine atoms, which are created in the stratosphere by photochemical decomposition of halogenated aliphatic hydrocarbons, was first provided by Molina and Rowland.1 Chlorine oxide appears as an intermediate in this cycle. Chlorine oxide also contributes to other catalytic cycles, such as to the ClO dimer cycle:2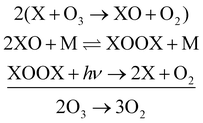 | (1) |
Understanding the photochemistry of halogen oxides, and in particular bromine oxides, is of great importance given their ozone-destroying capacity. In the present work, we examine the photochemistry of dibromine peroxide, BrOOBr, which is formed in the BrO dimer cycle. The kinetics of the BrO + BrO reaction was studied experimentally by a large number of groups20–36 since the 1970s. In the experiments, BrO was produced by flash photolysis37 of a mixture of Br2, O2 and O3 and the products were identified by methods such as UV spectroscopy and molecular beam mass spectrometry. The reaction is believed to proceed via the following mechanism, first suggested by Sander and Watson,22 and involves the following three channels:
channel a:
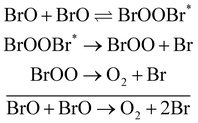 | (2) |
channel b:
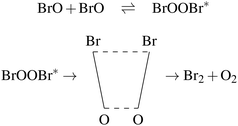 | (3) |
channel c:
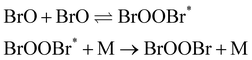 | (4) |
The JPL-recommended38 rate constants for the two bimolecular reactions are k1a = 2.4 × 10−12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(40/T) cm3 molecule−1 s−1 and k1b = 2.8 × 10−14
exp(40/T) cm3 molecule−1 s−1 and k1b = 2.8 × 10−14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(860/T) cm3 molecule−1 s−1. An estimate exists33 for the termolecular rate constant, k1c. In fact, there are two experimental papers that provide evidence for a short-lived intermediate species. The first is that of Mauldin et al.27 who, in 1993, studied this reaction at 220 K. A species bound by at least 5 kcal mol−1, attributed to BrOOBr, was observed at 312 nm. Five years later, Harwood and colleagues33 again studied this reaction. At low temperatures, 222 K and 235 K, they also observed the presence of an UV absorber other than BrO, O3 or Br2, which was identified as BrOOBr. Simulations of the overall BrO loss reaction showed that inclusion of the formation and thermal decomposition of BrOOBr accurately described the observed data. The best fit was obtained with a Br2O2 absorption cross-section of 1.2 × 10−17 cm2 molecule−1 at 320 nm. Based on this value, the UV spectrum was scaled accordingly. It compares well with the residual UV spectrum obtained by Mauldin et al. The rate constants for the formation and thermal decomposition of BrOOBr show that this species can only exist at low temperatures, below 250 K. The other proposed intermediates, bromine peroxide, BrOO, and the trapezoidal Br2O2 complex were not observed.
exp(860/T) cm3 molecule−1 s−1. An estimate exists33 for the termolecular rate constant, k1c. In fact, there are two experimental papers that provide evidence for a short-lived intermediate species. The first is that of Mauldin et al.27 who, in 1993, studied this reaction at 220 K. A species bound by at least 5 kcal mol−1, attributed to BrOOBr, was observed at 312 nm. Five years later, Harwood and colleagues33 again studied this reaction. At low temperatures, 222 K and 235 K, they also observed the presence of an UV absorber other than BrO, O3 or Br2, which was identified as BrOOBr. Simulations of the overall BrO loss reaction showed that inclusion of the formation and thermal decomposition of BrOOBr accurately described the observed data. The best fit was obtained with a Br2O2 absorption cross-section of 1.2 × 10−17 cm2 molecule−1 at 320 nm. Based on this value, the UV spectrum was scaled accordingly. It compares well with the residual UV spectrum obtained by Mauldin et al. The rate constants for the formation and thermal decomposition of BrOOBr show that this species can only exist at low temperatures, below 250 K. The other proposed intermediates, bromine peroxide, BrOO, and the trapezoidal Br2O2 complex were not observed.
The structures and stability of dibromine peroxide, BrOOBr, and its isomers were investigated computationally39–46 at both DFT and ab initio levels of theory. It was found that three isomers with the formula Br2O2 exist, which are, in order of decreasing stability, dibromine peroxide, BrOOBr, bromyl bromide, BrBrO(O), and bromine bromite, BrOBrO. All structures are more stable than two separated BrO molecules and could in principle be formed by the BrO + BrO reaction. They are less stable than O2 + Br2 in their electronic ground states. It is interesting to note here that the highly unstable isomers bromyl bromide and bromine bromite were observed for the first time in 2006 by Grothe and coworkers44 in an argon matrix by means of IR spectroscopy. They were produced by passing a mixture of Br2/O2/Ar through a microwave discharge. Dibromine peroxide was not observed. As to computational work, Guha and Francisco,39 in 1997, published a profound analysis of these molecules based on DFT calculations. Two years later, Gomez and Pácios40,41 investigated them using MP2 ab initio calculations with a spin-averaged relativistic effective core potential, AREP. Papayannis and coworkers42 used MP2 theory, with the 6-311+G(2d) basis set, and constructed a full, six-dimensional potential energy surface. The reaction dynamics was investigated by quasi-classical trajectory calculations and the rate constants k1a, k1b were computed. They agree well with the experimental results of Mauldin et al.27 and of Harwood et al.33 The dominant pathway leads to O2 + 2Br (channel a), followed by Br2 + O2 (channel b). Formation of BrOOBr (channel c) was not found. In a second publication, Papayannis and Kosmas47 studied the effect of initial vibrational excitation of BrO, which they found is minor. Furthermore, they suggested that the excited complex on the passage to the channel-b products is the planar cis-conformer of BrOOBr. Z. Li and Jeong43 reported ab initio results using the QCISD(T)/6-311+G(2d) method. Y. Li and Vo45 examined stationary points on the electronic ground state surface at the CCSD(T)/aug-cc-pVTZ level of theory and also reported vertical excitation energies to singlet and triplet excited states using CASSCF and MRCI. Finally, Dixon and coworkers46 investigated a significant number of halogen–oxygen compounds at a very high level of ab initio theory, CCSD(T) with extrapolation to the complete basis set limit, and reported accurate thermochemical data.
To conclude, the thermochemistry of chemical reactions involving bromine oxides is now well understood. There are some open questions concerning the formation of BrOOBr and possible intermediates during the BrO + BrO self-reaction at low temperatures and pressures. The photochemistry of dibromine peroxide is completely unknown and will be investigated in the present work. We have also found a metastable van der Waals complex, Br2·O2, that might be a candidate for the four-centre intermediate postulated by Sander and Watson.22
2. The ground state of BrOOBr
Dibromine peroxide has a twisted equilibrium structure similar to dihydrogen peroxide. There are two enantiomeric forms, separated by torsional barriers denoted as cis and trans barriers. A detailed analysis of the torsional motion can be found in a publication by Pacios and Gómez.40 Although these three stationary points on the electronic ground state surface were investigated by a number of groups as summarised in the introduction, consistent high-level CCSD(T)/AVTZ data, i.e. structures and harmonic frequencies, are not available for all of them. Furthermore, some of the data reported by Li and Vo45 differ from those of other researchers. Consolidating calculations were thus performed in the present work, and the energies and frequencies are shown in Tables 1 and 2 to make this work self-contained. DFT(M06-2X and B3PW91)/AVTZ results are included for comparison, as DFT methods were used in the course of this work to investigate photodynamic processes.| Structure | Method | r(BrO) | r(OO) | ∠(BrOO) | τ(BrOOBr) | Energies (Eh) | ZPE (mEh) |
|---|---|---|---|---|---|---|---|
| Twisted | M06-2X | 1.839 | 1.378 | 110.70 | 83.12 | −5298.738854 | 6.860 |
| B3PW91 | 1.896 | 1.339 | 112.71 | 84.18 | −5298.687352 | 6.202 | |
| CCSD(T) | 1.877 | 1.402 | 110.32 | 83.80 | −5295.382758 | 5.947 | |
| cis | M06-2X | 1.802 | 1.456 | 118.60 | 0.0 | −5298.721637 | 6.258 |
| B3PW91 | 1.800 | 1.501 | 117.80 | 0.0 | −5298.669302 | 5.641 | |
| CCSD(T) | 1.812 | 1.548 | 116.88 | 0.0 | -5295.367089 | 5.285 | |
| trans | M06-2X | 1.808 | 1.449 | 104.61 | 180.0 | −5298.731389 | 6.575 |
| B3PW91 | 1.815 | 1.476 | 104.43 | 180.0 | −5298.676993 | 5.880 | |
| CCSD(T) | 1.827 | 1.503 | 103.44 | 180.0 | −5295.375609 | 5.653 | |
| Structure | Method | ν 1 | ν 2 | ν 3 | ν 4 | ν 5 | ν 6 |
|---|---|---|---|---|---|---|---|
| Twisted | M06-2X | 88.60 | 298.08 | 411.35 | 639.56 | 667.59 | 905.90 |
| B3PW91 | 80.22 | 271.91 | 370.75 | 534.44 | 585.85 | 879.02 | |
| CCSD(T) | 78.35 | 264.05 | 364.55 | 544.35 | 589.10 | 770.15 | |
| cis | M06-2X | 238.96i | 148.28 | 395.80 | 608.03 | 780.34 | 814.39 |
| B3PW91 | 232.10i | 133.33 | 384.64 | 585.25 | 617.51 | 755.19 | |
| CCSD(T) | 203.21i | 136.23 | 359.51 | 508.48 | 592.83 | 722.97 | |
| trans | M06-2X | 53.60 | 209.59 | 226.39 | 686.50 | 780.58 | 929.32 |
| B3PW91 | 38.26i | 182.73 | 213.06 | 644.09 | 703.10 | 838.18 | |
| CCSD(T) | 25.09i | 181.64 | 205.69 | 627.04 | 696.78 | 770.32 | |
We identified another minimum that has not been reported previously. It corresponds to a van der Waals complex, O2(a1Δg)·Br2(X1Σ+g), located 1.2 mEh below the twisted structure. Description of the van der Waals complex requires the use of at least a triple-zeta basis set and inclusion of dynamic correlation. Without this, the computed energy will be above that of the twisted structure. The results obtained with the CCSD(T) method are presented in Table 3. Inclusion of scalar relativistic effects within the Douglas–Kroll approximation48 do not change the results significantly.
| Structure | CCSD(T) | CCSD(T)-DK | ||
|---|---|---|---|---|
| Energy | Relative energy | Energy | Relative energy | |
| cis | −5295.367090 | 0.016921 | 5359.575030 | 0.017340 |
| trans | −5295.375610 | 0.008401 | −5359.583601 | 0.008769 |
| Twisted | −5295.382758 | 0.001253 | −5359.591318 | 0.001061 |
| van der Waals | −5295.384011 | 0.0 | −5359.592372 | 0.0 |
3. UV-vis excitation of BrOOBr
BrOOBr possesses a large number of excited electronic states that can be reached from the electronic ground state by absorption of radiation with λ > 300 nm, which is the wavelength range relevant for atmospheric photochemistry. These states can be constructed by recombination of two BrO molecules in their electronic ground states X2Π or with one of them in the first excited state BrO (A2Π), which is located only about 27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 or 0.125 = 3.41 eV above the ground state.49,50 Relativistic effects are not important for the discussion. The combination of two 2Π states gives rise to the following electronic states of BrOOBr: two 1,3Σ and 1,3Δ, in the linear configuration. Upon bending, the degenerate D states decompose into states with symmetry A⊕B in the C2 point group. There are thus as many as four electronic states, 3A + B, with singlet multiplicity with asymptotes BrO(X2Π) + BrO(X2Π) or BrO(X2Π) + BrO(A2Π) and the same number of triplet states. All excited states are repulsive.
500 or 0.125 = 3.41 eV above the ground state.49,50 Relativistic effects are not important for the discussion. The combination of two 2Π states gives rise to the following electronic states of BrOOBr: two 1,3Σ and 1,3Δ, in the linear configuration. Upon bending, the degenerate D states decompose into states with symmetry A⊕B in the C2 point group. There are thus as many as four electronic states, 3A + B, with singlet multiplicity with asymptotes BrO(X2Π) + BrO(X2Π) or BrO(X2Π) + BrO(A2Π) and the same number of triplet states. All excited states are repulsive.
In the electronic ground state, the 86 electrons of dibromine peroxide occupy 22 orbitals of symmetry a and 21 orbitals of symmetry b. The 7 lowest excited states are generated by excitations from the three orbitals HOMO (21b), HOMO−1 (22a) and HOMO−2 (21a) to the orbitals LUMO (23a) and LUMO+1 (22b), which are shown in Fig. 1. Of these orbitals, the HOMO, HOMO−2 and HOMO−3 have major atomic orbital contributions from the bromine atoms and thus have nonbonding character. The remaining orbitals in the figure are antibonding. The electronic configurations of the excited states, generated by the promotion of electrons from non-bonding to antibonding orbitals as detailed in Table 4, indicate that these states are indeed unbound.
| Transition | Orbitals | Amplitudes |
|---|---|---|
| S0(X1A) → S1(11B) | 22a → 22b | 51.5 |
| 21b → 23a | 47.9 | |
| S0(X1A) → S2(11A) | 22a → 23a | 48.6 |
| 21b → 22b | 50.8 | |
| S0(X1A) → S3(21A) | 22a → 23a | 49.5 |
| 21b → 22b | −48.4 | |
| S0(X1A) → S4(21B) | 21a → 22b | 50.5 |
| 22a → 22b | −33.9 | |
| 21b → 23a | 33.8 | |
| S0(X1A) → S5(31A) | 20b → 22b | 39.3 |
| 21a → 23a | 58.5 | |
| S0(X1A) → S6(31B) | 20b → 23a | 57.0 |
| 21a → 22b | 22.8 | |
| 22a → 22b | 23.5 | |
| 21b → 23a | −24.9 | |
| S0(X1A) → S7(41A) | 20b → 22b | 58.2 |
| 21a → 23a | −39.1 |
The high density of electronic states makes the investigation of the photodynamics a formidable task, and the choice of the quantum-chemical method is not evident a priori. Both ab initio and density functional methods were tested, with basis sets of augmented triple-zeta quality. Table 5 gives a comparison of results obtained with DFT and ab initio methods without consideration of relativistic corrections. The effect of the latter is minor, as can be seen from the results in Table 6, obtained at the MCSCF and MRCI levels of theory using the Douglas–Kroll48,51 approach.† Inclusion of dynamic correlation is more important as it lowers the transition energies by about 0.2 eV (see Table 7). Li and Vo,45 who investigated the excited electronic states previously, arrived at the same conclusion though some of their numerical data differ from ours. These differences are due to small differences in the reference configuration of the nuclei. BrOOBr is an extremely floppy molecule, and small variations of the nuclear geometry parameters lead to notable changes in the vertical excitation energies.
| Excited state | B3PW91(TDA) | B3PW91(TDDFT) | CASSCF | |
|---|---|---|---|---|
| Active space (26e, 16o) | Active space (26e, 18o) | |||
| S1(11A) | 3.133 | 3.056 | 3.631 | 3.571 |
| S2(11B) | 3.134 | 3.060 | 3.678 | 3.941 |
| S3(21A) | 3.702 | 3.642 | 4.476 | 4.452 |
| S4(21B) | 3.927 | 3.848 | 4.495 | 4.593 |
| S5(31A) | 3.983 | 3.899 | 4.546 | 4.628 |
| S6(31B) | 4.045 | 3.959 | 5.182 | 5.226 |
| S7(41A) | 4.344 | 4.329 | 5.328 | 5.337 |
| Excited state | MCSCF | MRCI | EOM–CCSD | |||
|---|---|---|---|---|---|---|
| MCSCF | MCSCFDK | MCSCFECP | MRCI | MRCIDK | ||
| S1(11A) | 3.631 | 3.594 | 3.595 | 3.436 | 3.399 | 3.274 |
| S2(11B) | 3.678 | 3.637 | 3.637 | 3.470 | 3.433 | 3.305 |
| S3(21A) | 4.476 | 4.421 | 4.423 | 4.204 | 4.158 | 4.147 |
| S4(21B) | 4.495 | 4.438 | 4.441 | 4.266 | 4.216 | 4.155 |
| S5(31A) | 4.546 | 4.495 | 4.495 | 4.277 | 4.228 | 4.802 |
| S6(31B) | 5.182 | 5.128 | 5.128 | 5.007 | 4.972 | 4.909 |
| S7(41A) | 5.328 | 5.271 | 5.271 | 5.240 | 5.187 | 5.445 |
| Excited | B3PW91(TDA) | MCSCF, CAS(26e, 16o) | MRCI | ||||||
|---|---|---|---|---|---|---|---|---|---|
| E (eV) | λ (nm) | f | E (eV) | λ (nm) | f | E (eV) | λ (nm) | f | |
| S1(11A) | 3.133 | 395.70 | 0.0001 | 3.631 | 341.46 | 0.0008 | 3.436 | 360.79 | 0.0000 |
| S2(11B) | 3.134 | 395.60 | 0.0000 | 3.678 | 337.46 | 0.0010 | 3.470 | 357.26 | 0.0001 |
| S3(21A) | 3.702 | 334.88 | 0.0077 | 4.476 | 277.18 | 0.0050 | 4.204 | 294.91 | 0.0064 |
| S4(21B) | 3.927 | 315.72 | 0.0440 | 4.495 | 275.94 | 0.0017 | 4.266 | 290.59 | 0.0003 |
| S5(31A) | 3.983 | 311.21 | 0.0000 | 4.546 | 272.97 | 0.0028 | 4.277 | 289.85 | 0.0003 |
| S6(31B) | 4.045 | 306.45 | 0.0324 | 5.182 | 239.49 | 0.0459 | 5.007 | 247.61 | 0.0466 |
| S7(41A) | 4.344 | 285.42 | 0.0005 | 5.328 | 232.92 | 0.0013 | 5.240 | 236.59 | 0.0006 |
The tables demonstrate that the outcomes from different methods differ in particular for excitation to higher electronic states. It should be noted, however, that the vertical transition energies and oscillator strengths can only give a crude approximation to the UV spectrum of the title molecule, as this can depart significantly from the equilibrium nuclear configurations due to large-amplitude torsional motion. For a more realistic simulation of the UV spectrum, displacement of the nuclei from their equilibrium positions needs to be taken into account. Such a simulation was performed using the Newton-X package52 coupled to Gaussian G1653 or to Molpro.‡![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 54 Within Newton-X, the photoabsorption cross section is computed as
54 Within Newton-X, the photoabsorption cross section is computed as
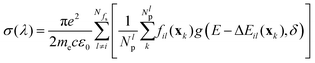 | (5) |
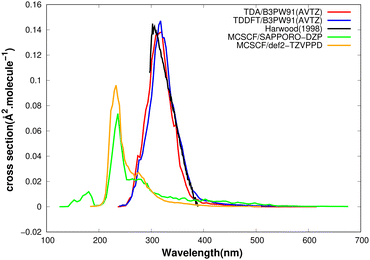
Using Newton-X software, the UV-vis spectrum was computed employing a variety of quantum chemical methods, with the aim to determine the most adequate procedure for the investigation of photodynamics. The strategy adopted previously57 by our group is that the method that reproduces the experimental spectrum best should also be reliable away from the Franck–Condon region and thus appropriate for photodynamics. Both MCSCF and TDDFT methods are computationally feasible for use in photodynamics and were tested here, and some of the results are presented in Fig. 2 where they are compared with the experimental spectrum.27,33 It can be seen that the signal obtained with MCSCF is quite displaced from the experimental one, whereas TDDFT/B3PW91 leads to almost perfect agreement, as far as both position and intensity are concerned.
The decomposition of the computed UV spectrum in terms of electronic states is presented in Fig. 3. Contributions are due to excitations to the seven lowest singlet states, but that from the first excited state is negligible over the entire range of wavelengths. An important contribution comes from the fifth excited state, though the oscillator strength corresponding to vertical excitation from the ground state equilibrium nuclear configuration is zero. This is a manifestation of large-amplitude motion and demonstrates the importance of adequate sampling of nuclear configurations.
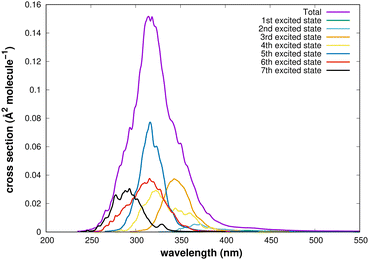 | ||
| Fig. 3 UV spectrum computed with the B3PW91(TDA)/AVTZ method and its contributions from individual electronic states. | ||
The same spectrum computed at the MCSCF/AVTZ level (Fig. 4) is blue-shifted from the DFT spectrum. Furthermore, there are differences in the excited state contributions, mainly that there is now a strong absorption to the S7 state rather than to the S5 state of the DFT calculation, which we attribute to differences in the orbital energies. The MCSCF spectrum could have been improved by inclusion of dynamic correlation by means of a MRCI calculation. Such a treatment is expected to shift the signal by about 0.2 eV towards the red, judging from the results presented in Table 6, but it would still be displaced from the experimental spectrum. As the DFT result is very satisfying, we did not compute the spectrum at the MRCI level of theory.
 | ||
| Fig. 4 UV spectrum computed with the MCSCF/AVTZ method and its contributions from individual electronic states. | ||
4. Photodecomposition of excited BrOOBr
4.1. Product channels
The photodecomposition of dibromine peroxide was studied by running classical trajectories using the Newton-X package. For each of the excited electronic states, 150 trajectories were followed up to a simulation time of tmax = 1000 fs with step size Δt = 0.5 fs. The initial conditions for the trajectories were obtained by projecting a sample of nuclear configurations from the ground state surface to that of an excited state and computing their initial velocities from the derivatives of the potential energy surface. Electronic energies, gradients and non-adiabatic coupling terms were computed on the fly using the Gaussian G16 quantum chemistry code. The TDDFT-TDA/B3PW91 approach was used with the AVTZ basis set.Three product channels can be distinguished, leading to 2BrO (channel a), BrOO + Br (channel b) or 2Br + O2 (channel c). BrOO was observed experimentally58,59 but is highly unstable60 and, if initially formed photochemically, would likely decompose into Br + O2. However, it may also be produced by thermal decomposition of ground state BrOOBr at low temperatures. The excited states of BrOO are repulsive. Table 8 provides the possible exit channels for each of the excited electronic states, including S1 though it is not populated, and an estimate of their asymptotic energies. The latter were computed using the data from ref. 46 and 49.
| States | Products | E (eV) | Products | E (eV) |
|---|---|---|---|---|
| Channel BrOOBr→2BrO | ||||
| S0–S3 | 2BrO (X2Π) | 0.950 | ||
| S4–S7 | BrO (X2Π) + BrO (A2Π) | 4.360 | ||
| Channel BrOOBr → BrOO + Br → 2Br + O2 | ||||
| S0–S2 | BrOO (X2A′′) + Br (2P) | 0.690 | 2Br (2P) + O2 (X3Σ−g) | 0.600 |
| S3–S6 | BrOO (A2A′) + Br (2P) | — | 2Br (2P) + O2 (X3Σ−g) | 0.600 |
| S7–S10 | BrOO (B2B′) + Br (2P) | — | 2Br (2P) + O2 (X1Δ) | 1.577 |
The results of the trajectory calculations are presented in Table 9. For the automatic identification of the fragments, we consider a bond as broken when its internuclear distance exceeds the initial value by more than 25%. The procedure was adopted in a previous work on photodynamics.61 The formation probabilities p and error bounds were obtained by a statistical analysis as p ± zp(1 − p)/n, with p being the fraction of trajectories leading to a particular fragment channel and n the total number of trajectories. z = 1.96 for the 95% confidence interval. The error bounds for the 2BrO channel are as large as the formation probabilities as only very few trajectories lead to these products. One trajectory is analysed in detail in Fig. 5 and the explanatory text in Section 4.2.
| State | BrO + BrO | 2Br + O2 | Br + BrOO |
|---|---|---|---|
| S2 | 0.01 ± 0.01 | 0.40 ± 0.07 | 0.59 ± 0.07 |
| S3 | 0.02 ± 0.02 | 0.69 ± 0.07 | 0.29 ± 0.07 |
| S4 | 0.00 | 0.81 ± 0.06 | 0.19 ± 0.06 |
| S5 | 0.00 | 0.79 ± 0.06 | 0.16 ± 0.05 |
| S6 | 0.00 | 0.82 ± 0.06 | 0.18 ± 0.06 |
| S7 | 0.00 | 0.96 ± 0.03 | 0.04 ± 0.03 |
Performing the trajectory calculations for dibromine peroxide is challenging even on a supercomputer. It is computationally prohibitive to follow the trajectories over a sufficient time interval for the formation of the final fragments as the non-adiabatic dynamics of BrOOBr is slow because of the heavy bromine nuclei. What is more, the need to include up to seven electronic states in the simulation requires small time steps to assure numerical stability. The Tamm–Dancoff approximation to the time-dependent density functional approach was used here as it performs favourably well, particularly in the asymptotic region where the electronic states are dense and avoided crossings are common.62 However, not all trajectories could be propagated up to the end due to numerical instabilities. In those cases, the dissociation products were analysed manually at the last point of the propagation.
4.2. Mechanisms of photodecomposition
The principal reaction channel is the one leading to 2Br + O2 in their electronic ground states. Two distinct mechanisms are present: concerted rupture of the two Br–O bonds or sequential rupture. Concerted rupture occurs mainly on highly excited electronic state surfaces, S3 or higher. On the lower electronic states, S0 to S2, sequential rupture is favoured as the BrOO (X2A′′) fragment is weakly bound. The second bond then breaks up within 50 fs after the first bond. The fragment distribution in Table 9 reflects the result of a competition between fast breakup followed by non-adiabatic stabilisation and the opposite, i.e. rapid non-adiabatic relaxation and breakup on a lower electronic surface.There is also a certain probability of separation into two BrO radicals. This is a rare event as the O–O bond is much stronger than the Br–O bond. The mechanism for such a breakup is quite remarkable. It can be identified from Fig. 5 where the time evolution of the internuclear distances is shown for the case of BrOOBr initially placed on the S2 electronic surface. Fast oscillations of the O–O bond are observed along with slow oscillations, with a period of about 400 fs, corresponding to a frequency of 76, which is due to large-amplitude torsional motion. During the first period, the Br–O bonds vibrate in phase. Thus, the Br–O bond lengths augment significantly, up to 2.8 Å. It is quite remarkable that the bromine atoms do not fly apart but rather reapproach their oxygen atoms. During the second period, between 400 fs and 800 fs, the Br–O bonds oscillate out of phase. It can be noted that, at the beginning of this phase, the bromine atom labelled Br(1) arrives at a distance of about 3.5 Å from oxygen O(1), violet line in the figure, and then keeps a distance of about 3 Å over 300 fs, while the remaining triatomic fragment undergoes several rearrangements, with intermediate triatomic structures resembling bromine peroxide BrOO and bromine dioxide OBrO. Then, suddenly, Br(1) approaches its oxygen atom and at about 800 fs the O–O bond breaks up. The two BrO fragments separate. During this second period, near 760 fs, a non-adiabatic transition from S2 to S1 takes place and electronic energy is converted to vibrational energy. Another non-adiabatic transition occurs at 880 fs where the system arrives on the electronic ground state.
The breakup of the O–O bond is thus a result of energy transfer from electronic to nuclear motion followed by intramolecular vibrational energy redistribution towards the O–O vibration, though it appears more accurate to classify this as intermolecular vibrational energy redistribution, given that the bromine atoms are at large distances while the dioxygen keeps vibrating. The dissociation process is slow as it passes through a complex vibronic resonance.
4.3. Dissociation rate constant
The unimolecular photodecomposition rate constant J can be computed as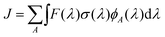 | (6) |
| J = 9.7 × 10−2 s−1 | (7) |
This constant may be compared with the rate constant of thermal decomposition reported by Harwood et al.33 at temperatures of T1 = 222 K and T2 = 235 K. For the second-order decomposition
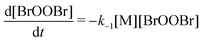 | (8) |
| kuni−1 ≈ 10−1 s−1 | (9) |
The density of potential collider molecules near the tropopause is of the same order, so that in view of atmospheric chemistry the two decay mechanisms are equally important.
5. Conclusions
Dibromine peroxide is a weakly stable molecule whose very existence and formation in a termolecular reaction, i.e. recombination of two BrO radicals in the presence of a third body, M, was proved indirectly by the well-known experiments conducted by Mauldin27 and Harwood33 and their colleagues some time ago. They have particularly shown that this molecule is stable at temperatures below 250 K, which are typically found in the Earth's atmosphere near the tropopause. Rate constants for its formation and thermal decomposition are well reported in the literature, as outlined in the introduction. In contrast, the photochemistry of dibromine peroxide has remained unexplored, despite the importance of photochemical reactions in the atmosphere in general. We have shown in the present work, by means of mixed quantum–classical trajectory calculations on seven coupled electronic potential energy surfaces, that photoexcited dibromine peroxide decomposes rapidly (τ1/2 ≈ 7 s), eventually leading, via different unstable intermediates, to 2Br + O2, which are also the principal thermal decomposition products. The rate constant for photodecomposition under atmospheric conditions was estimated as J = 9.7 × 10−2 s−1 and is comparable to the thermal decomposition rate constant. Therefore, photolysis should be taken into account in atmospheric modelling as it likely affects the Br/BrO ratio. We finally conclude that bromine containing radicals (Br and BrO) are not neutralised by the BrOOBr channel and remain active species in the atmosphere.The mechanism leading to the breakup into two BrO radicals following vibronic energy redistribution is most uncommon, and we are not aware of similar processes in molecular physics. We therefore hope that our results will lead to new experiments aiming at a better understanding of the formation and decomposition of dibromine peroxide, particularly since the two less stable isomers of Br2O2 were observed experimentally in 2006 by Grothe and coworkers.44
Author contributions
All authors contributed equally to this work. W. C. is a PhD student supervised by the other three authors.Conflicts of interest
There are no conflicts to declare.Data availability
All data are reported in the article. The following computational codes were used: 1. Gaussian G16, 2. Molpro and 3. Newton-X. They were all cited in the article.Acknowledgements
We are grateful for the financial support from the “PHC Utique” programme of the French Ministry of Foreign Affairs and Ministry of Higher Education and Research and the Tunisian Ministry of Higher Education and Scientific Research (project number 24G1301). W. C. acknowledges a PhD studentship from the Tunisian Ministry of Higher Education and Scientific Research. The supercomputer time was provided by the ROMEO HPC Center at the University of Reims Champagne-Ardenne, by CRIANN (Centre des Ressources Informatiques et Applications Numériques de Normandie) and by GENCI-IDRIS (Grant 2024-103918).Notes and references
- M. J. Molina and F. S. Rowland, Nature, 1974, 249, 810–812 CrossRef CAS.
- L. T. Molina and M. J. Molina, J. Phys. Chem., 1987, 91, 433–436 CrossRef CAS.
- T. von Clarmann, Atmosfera, 2013, 26, 415–458 CrossRef CAS.
- D. J. Lary, J. Geophys. Res., 1997, 102, 21515–21526 CrossRef CAS.
- J. J. Lin, A. F. Chen and Y. T. Lee, Chem. – Asian J., 2011, 6, 1664–1678 CrossRef CAS PubMed.
- J. B. Burkholder, R. A. Cox and A. R. Ravishankara, Chem. Rev., 2015, 115, 3704–3759 CrossRef CAS PubMed.
- J. S. Daniel, S. Solomon, R. W. Portmann and R. R. Garcia, J. Geophys. Res., 1999, 104, 23871–23880 CrossRef CAS.
- B.-M. Sinnhuber, N. Sheode, M. Sinnhuber, M. P. Chipperfield and W. Feng, Atmos. Chem. Phys., 2009, 9, 2863–2871 CrossRef CAS.
- J. E. Klobas, D. K. Weisenstein, R. J. Salawitch and D. M. Wilmouth, Atmos. Chem. Phys., 2020, 20, 9459–9471 CrossRef CAS.
- G. Le Bras and U. Platt, Geophys. Res. Lett., 1995, 22, 599–602 CrossRef CAS.
- Y. Bedjanian and G. Poulet, Chem. Rev., 2003, 103, 4639–4656 CrossRef CAS PubMed.
- R. J. Salawitch, M. B. McElroy, J. H. Yatteau, S. C. Wofsy, M. R. Schoeberl, L. R. Lait, P. A. Newman, K. R. Chan, M. Loewenstein, J. R. Podolske, S. E. Strahan and M. H. Proffitt, Geophys. Res. Lett., 1990, 17, 561–564 CrossRef.
- J. G. Anderson, D. Toohey and W. H. Brune, Science, 1991, 251, 39–46 CrossRef CAS PubMed.
- M. B. McElroy, R. J. Salawitch, S. C. Wofsy and J. A. Logan, Nature, 1986, 321, 759–762 CrossRef CAS.
- J. McConnell, G. Henderson, L. Barrie, J. Bottenheim, H. Niki, C. Langford and E. Templeton, Nature, 1992, 355, 150–152 CrossRef CAS.
- T. K. Koenig, S. Baidar, P. Campuzano-Jost, C. A. Cuevas, B. Dix, R. P. Fernandez, H. Guo, S. R. Hall, D. Kinnison, B. A. Nault, K. Ullmann, J. L. Jimenez, A. Saiz-Lopez and R. Volkamer, Proc. Natl. Acad. Sci. U. S. A., 2020, 117, 1860–1866 CrossRef CAS PubMed.
- C. A. Cuevas, R. P. Fernandez, D. E. Kinnison, Q. Li, J.-F. Lamarque, T. Trabelsi, J. S. Francisco, S. Solomon and A. Saiz-Lopez, Proc. Natl. Acad. Sci. U. S. A., 2022, 119, e2110864119 CrossRef CAS PubMed.
- P. Ricaud and F. Lefèvre, Advances in Fluorine Science, in Fluorine and the Environment, ed. A. Tressaud, Elsevier, 2006, vol. 1, pp. 1–32 Search PubMed.
- W. H. Schlesinger, E. M. Klein and A. Vengosh, Global Biogeochemical Cycles, 2020, 34, e2020GB006722 CrossRef CAS.
- M. A. Clyne and R. T. Watson, J. Chem. Soc., Faraday Trans. 1, 1975, 71, 336–350 RSC.
- S. Jaffe and W. Mainquist, J. Phys. Chem., 1980, 84, 3277–3280 CrossRef CAS.
- S. P. Sander and R. T. Watson, J. Phys. Chem., 1981, 85, 4000–4007 CrossRef CAS.
- R. Cox, D. Sheppard and M. Stevens, J. Photochem., 1982, 19, 189–207 CrossRef CAS.
- A. A. Turnipseed, J. W. Birks and J. G. Calvert, J. Phys. Chem., 1990, 94, 7477–7482 CrossRef CAS.
- I. Barnes, V. Bastian, K. Becker and R. Overath, Int. J. Chem. Kinet., 1991, 23, 579–591 CrossRef CAS.
- I. Lancar, G. Laverdet, G. L. Bras and G. Poulet, Int. J. Chem. Kinet., 1991, 23, 37–45 CrossRef CAS.
- R. Mauldin III, A. Wahner and A. Ravishankara, J. Phys. Chem., 1993, 97, 7585–7596 CrossRef.
- I. Bridier, B. Veyret and R. Lesclaux, Chem. Phys. Lett., 1993, 201, 563–568 CrossRef CAS.
- D. M. Rowley, M. H. Harwood, R. A. Freshwater and R. L. Jones, J. Phys. Chem., 1996, 100, 3020–3029 CrossRef CAS.
- M. K. Gilles, A. A. Turnipseed, J. B. Burkholder, A. Ravishankara and S. Solomon, J. Phys. Chem. A, 1997, 101, 5526–5534 CrossRef CAS.
- B. Laszlo, R. E. Huie, M. J. Kurylo and A. W. Miziolek, J. Geophys. Res., 1997, 102, 1523–1532 CrossRef CAS.
- Y. Bedjanian, G. Le Bras and G. Poulet, J. Phys. Chem. A, 1998, 102, 10501–10511 CrossRef CAS.
- M. H. Harwood, D. M. Rowley, R. A. Cox and R. L. Jones, J. Phys. Chem. A, 1998, 102, 1790–1802 CrossRef CAS.
- V. Ferracci, K. Hino and D. M. Rowley, Phys. Chem. Chem. Phys., 2011, 13, 7997–8007 RSC.
- M. K. M. Ward and D. M. Rowley, Phys. Chem. Chem. Phys., 2017, 19, 23345–23356 RSC.
- Y. Bedjanian, Int. J. Chem. Kinet., 2020, 52, 319–328 CrossRef CAS.
- G.-N. Porter, Proc. R. Soc. London, Ser. A, 1950, 200, 284–300 CAS.
- J. B. Burkholder, S. P. Sander, J. Abbatt, J. R. Barker, et al., Chemical Kinetics and Photochemical Data for Use in Atmospheric Studies, Evaluation No. 19, Publication 19-5, Pasadena, ca: Jet propulsion laboratory, national aeronautics and space technical report, 2019.
- S. Guha and J. S. Francisco, J. Phys. Chem. A, 1997, 101, 5347–5359 CrossRef CAS.
- L. Pacios and P. Gómez, THEOCHEM, 1999, 467, 223–231 CrossRef CAS.
- P. C. Gómez and L. F. Pacios, J. Phys. Chem. A, 1999, 103, 739–743 CrossRef.
- D. Papayannis, A. M. Kosmas and V. S. Melissas, Chem. Phys., 1999, 243, 249–262 CrossRef CAS.
- Z. Li and G.-R. Jeong, Chem. Phys. Lett., 2001, 340, 194–204 CrossRef CAS.
- O. Gálvez, A. Zoermer, A. Loewenschuss and H. Grothe, J. Phys. Chem. A, 2006, 110, 6472–6481 CrossRef PubMed.
- Y. Li and C. K. Vo, J. Chem. Phys., 2006, 124, 204309 CrossRef PubMed.
- D. J. Grant, E. B. I. Garner, M. H. Matus, M. T. Nguyen, K. A. Peterson, J. S. Francisco and D. A. Dixon, J. Phys. Chem. A, 2010, 114, 4254–4265 CrossRef CAS PubMed.
- D. K. Papayannis and A. M. Kosmas, Can. J. Chem., 2001, 79, 1940–1945 CrossRef CAS.
- M. Douglas and N. M. Kroll, Ann. Phys., 1974, 82, 89–155 CAS.
- Y. Li, J. S. Francisco and K. A. Peterson, J. Chem. Phys., 2000, 113, 8556–8560 CrossRef CAS.
- D. Zhou, D. Shi, J. Sun and Z. Zhu, Comput. Theor. Chem., 2017, 1112, 94–103 CrossRef CAS.
- T. Nakajima and K. Hirao, Chem. Rev., 2012, 112, 385–402 CrossRef CAS PubMed.
- M. Barbatti, M. Ruckenbauer, F. Plasser, J. Pittner, G. Granucci, M. Persico and H. Lischka, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2014, 4, 26–33 CAS.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria and M. A. Robb, et al., Gaussian 16 Revision C.01, Gaussian Inc., Wallingford CT, 2016 Search PubMed.
- H.-J. Werner, P. J. Knowles, G. Knizia, F. R. Manby, M. Schütz, et al., MOLPRO, version 2021.1, a package of ab initio programs, 2021, see https://www.molpro.net Search PubMed.
- Z. Wang, PhD thesis, Department of Chemistry, Supervisor Alex Brown, 2019.
- M. Barbatti, A. J. A. Aquino and H. Lischka, Phys. Chem. Chem. Phys., 2010, 12, 4959–4967 RSC.
- O. Ferchichi, N. Derbel, T. Cours and A. Alijah, Phys. Chem. Chem. Phys., 2020, 22, 4059–4071 RSC.
- J. Kölm, A. Engdahl, O. Schrems and B. Nelander, Chem. Phys., 1997, 214, 313–319 CrossRef.
- K. Suma, Y. Sumiyoshi and Y. Endo, J. Chem. Phys., 2005, 123, 024312 CrossRef PubMed.
- L. F. Pacios and P. C. Gómez, J. Phys. Chem. A, 1997, 101, 1767–1773 CrossRef CAS.
- W. Chebbi, N. Derbel, A. Alijah and T. Cours, Phys. Chem. Chem. Phys., 2024, 26, 123–129 RSC.
- S. Hirata and M. Head-Gordon, Chem. Phys. Lett., 1999, 314, 291–299 CrossRef CAS.
Footnotes |
| † Molpro-recommended option DKHO = 8 with exponential parametrisation for unitary transformations. |
| ‡ A Molpro–Newton-X interface was provided by Alex Brown and Zhibo Wang,55 and extended by the present authors. |
| § The National Center for Atmospheric Research in Boulder (https://www.acom.ucar.edu/Models/TUV/Interactive_TUV/) |
| This journal is © the Owner Societies 2025 |