Synergistic cyclic cooperativity governs the strength of chalcogen, pnictogen and tetrel bonds in microhydrated clusters†
Ayush
Shivhare
 a,
Chandan Kumar
Lenka
a,
Chandan Kumar
Lenka
 a,
Bharti
Dehariya
a,
Bharti
Dehariya
 a and
Milind M.
Deshmukh
a and
Milind M.
Deshmukh
 *b
*b
aDepartment of Chemistry, Dr Harisingh Gour Vishwavidyalaya (A Central University), Sagar 470003, India. E-mail: ayushshivharepanna@gmail.com; chandankumarlenka80@gmail.com; dehariyabharti29@gmail.com
bDepartment of Chemistry, University of Delhi, 110007, Delhi, India. E-mail: milind.deshmukh@gmail.com; mdeshmukh@dhsgsu.edu.in
First published on 4th July 2025
Abstract
It has been demonstrated that the sum of the cooperativity contributions (CCs) of the cyclic structures (common to a referenced hydrogen bond (HB)) when added to the energy of the HB in the respective dimer provides an accurate estimation of its energy in an actual molecular cluster. For this purpose, a molecular tailoring approach (MTA)-based method was utilized to estimate the CCs of these cycles. The HB energies calculated in this fashion (ESynergeticHB) were in excellent agreement with those of their actual cluster counterparts (EClusterHB). In this study, the generality of this methodology for estimating the energy of other non-covalent bonds (NCBs), viz. tetrel (TBs), chalcogen (CBs) and pnictogen bonds (PBs), in microhydrated clusters is tested. For this purpose, the microhydrated clusters of carbon dioxide (CO2), nitrous (N2O) oxide and sulphur dioxide (SO2) are employed. These microhydrated clusters exhibit TBs, PBs and CBs with the surrounding water molecules. The energies of these NCBs calculated using the synergetic cyclic cooperativity (ESynergeticNCB) approach were found to be in excellent agreement with those of their full cluster counterparts (EClusterNCB) calculated using the MTA-based method. The difference between the two values was found to be less than 0.6 kcal mol−1. It is emphasized here that the variation in the strength of these different NCBs can be nicely explained in terms of the interplay of the nature of the cooperativity exhibited by the cyclic structures common to them.
Introduction
For the past two decades, there has been growing interest in exploring the understanding of non-covalent bonds (NCBs) other than the hydrogen bonds (HBs).1–8 Among these, halogen bonding (XB) has been quite extensively explored.1 In addition, pnictogen (PB), chalcogen (CBs) and tetrel bonds (TBs) have recently attracted significant attention.2–7 Molecular electrostatic potential (MESP) analysis is often used in interaction studies to recognize the preferential sites of donors and acceptors.8–10 These terminologies (TB, PB and CB) comprise the atoms of groups 14, 15 and 16 of a molecule, in which a positive electrostatic potential (MESP) is often found to interact with the suitable electron donor species. Depending on the position of positive MESP to be along or perpendicular to the molecular plane, the concepts of σ and π-holes have been used.11,12 Although research on these NCB complexes involving TB, PB and CB is increasing daily, it is limited to the study of the interaction present in the dimeric complex. There are few examples reported recently wherein the effect of a third molecule on the strength of these NCBs has been investigated.13–15 The presence of a third molecule either enhances or diminishes the strength of a referenced NCB.16 The enchantment or decrement in the strength of a referenced NCB due to the presence of surrounding molecules is called cooperativity or anti-cooperativity, respectively.Cooperativity is an important feature in modulating the strength of NCBs. This phenomenon is popular and recognized in the context of HBs. For instance, the cooperativity corresponding to the interconnected networks of HBs has been extensively studied in molecular clusters of water (H2O)n,17 hydrogen fluoride (HF)n,18 and ammonia (NH3)n19 as well as in intramolecular hydrogen-bonded systems.20,21 The HB cooperativity is found to be responsible for the properties of liquids,22,23 hydration of ions,24,25 and the structure of large supramolecular aggregates and biomolecules.26,27 Cooperativity is observed when the direction of all the HBs (donor to acceptor) is same either in a linear or cyclic structure; such a cycle is called a homodromic cycle. However, anti-cooperativity prevails when this directionality is broken at two or more neighboring HBs, resulting in the formation of an antidromic cycle.28 It has been reported that a large three-dimensional (3D) structure of a molecular cluster is made up of small cyclic structures with a different number of HBs.29 The cooperativity and anti-cooperativity of these homodromic or antidromic cycles are crucial to determine the strength of the HBs, which are present at the interface of these cyclic structures. However, the methods available in the literature30–38 cannot be employed to understand the individual HB energy and cooperativity contribution of neighboring molecules towards them in molecular clusters. To understand the nature of cooperativity in a given 3D molecular cluster, the quantitative estimates of the individual HB strength and the cooperativity contribution of other HBs towards them are essential.
With this understanding, Deshmukh and Gadre developed a molecular tailoring approach (MTA)-based method for direct, reliable estimation of the energy of individual HBs in various molecules,38–44 molecular clusters45–49 and crystals.50 In a large 3D molecular cluster, it has been found that an HB may be present at the intersection of two or more cyclic structures of the interconnected HB networks.29 Shivhare and Deshmukh demonstrated that the energy of such an X–H⋯Y HB in molecular clusters can be accurately determined in terms of cooperativity contributions (CCs) of these cyclic structures.51,52 For instance, the MTA-based method was employed to calculate the CCs of these individual cycles. The CCs of all these cyclic structures were added to the energy of this HB in the corresponding dimer chopped out of the 3D molecular cluster.51,52 The energy of an HB calculated in this manner (in terms of synergetic effects of the cooperativity of cyclic structures) is denoted by ESynergeticHB. The ESynergeticHB values were found to be in excellent agreement with their full calculation counterparts obtained by applying the MTA-based method using the actual cluster.
It is emphasized here that there are only few investigations reported in the literature directed towards the understanding of the cooperativity effect on the strength of TBs, PBs and CBs along with HBs. For instance, one of the early studies investigated a ring-shaped molecular complex formed by CH3, COX2 or CSX2 (X = F, Cl) and HY (Y = CN, NC), which are connected via two HBs and a single TB.53 It was concluded that the contribution of three-body terms (measure of cooperativity) to the total interaction energy significantly depends on the nature of the substituents (X and Y), which is largest and smallest for the CH3⋯CSF2⋯HNC and CH3⋯CSCl2⋯HNC complexes, respectively.53 Another interesting study was conducted by Li and coworkers54 who investigated the ternary complexes of PhTY3⋯4-X-pyridine⋯N-base (N-base = HCN, NH3, NHNH2, and NH2CH3; T = C, Si, and Ge; Y = H, F and Cl; and X = bromo or iodo).54 The 4-Halopyridine with σ-hole on the halogen atom leads to the formation of halogen bond (XB), while the electron-deficient tetrel (T) atom in PhTY3 is involved in the formation of a TB. The strengths of both XBs and TBs are reported to be enhanced owing to positive cooperativity, whereas the strengths of both HB and XB in PhCF3⋯4-iodopyridine⋯NH3 are weakened as a result of negative cooperativity.54 A similar study involving F−⋯(CH3)2X⋯YF (X = O, S, Se; Y = F, H) triads was reported by McDowell,55 wherein a significant enhancement of the X⋯Y interactions relative to the neutral (CH3)2X⋯YF dyads was reported. Scheiner and coworkers13 also performed ab initio calculations of dimers and trimers formed by imidazole (IM) and F2TO (T = C, Si, Ge) molecules. A certain trimeric complex with negative cooperative TB between the central F2TO molecule and two IM molecules was reported. The total interaction energy of the ternary complex is smaller than the sum of the two separate TBs in the IM⋯F2TO dimer. Alternate configurational geometries stabilized by positive cooperativity was also reported in which the second IM formed an HB through the NH group of the first IM. The formation of such a positive cooperative ternary complex is also reflected in terms of shorter intermolecular distances. Deshmukh and coworkers investigated the geometries and NCB energies in microhydrated complexes of nitric oxide (NO).56 It was found that neutral NO can form HB and PBs with water depending on the orientation; however, such HBs and PBs are weaker compared to the water⋯water HBs. This was attributed a smaller value of cooperativity contributions (−0.66 to 1.31 kcal mol−1) found for NO···water and a moderate cooperativity contribution for water⋯water HBs (−0.20 to 4.38 kcal mol−1). However, NO+ species form stronger PBs with significant positive cooperativity contributions (5.08 to 17.84 kcal mol−1). The strength of water⋯water HBs in NO+ microhydrated clusters was weak owing to significantly negative cooperativity contributions (−0.05 to −1.69 kcal mol−1).
In summary, although the studies reported in the literature are useful to understand the cooperative effect of neighboring molecules on the strength of other NCBs (such as TBs, PBs and CBs), a thorough investigation to modulate the effect of homodromic (cooperative) and antidromic (anti-cooperative) cooperative cycles involving TBs, PBs and CBs along with HBs in microhydrated clusters is indispensable. Important questions to be answered here are as follows: (i) whether the synergetic cyclic cooperativity effect investigated earlier in the context of HBs is also applicable to TBs, PBs and CBs or not? (ii) In other words, is synergetic cyclic cooperativity a general phenomenon? and (iii) Is it possible to accurately determine the energies of these NCBs (other than HBs) in terms of synergetic cooperativity effects of cyclic structures to which they are common? To answer these important questions, in this study, the microhydrated clusters of carbon dioxide (CO2), nitrous oxide (N2O) and sulphur dioxide (SO2) are investigated. The primary focus of the present investigation is to study these TBs, PBs and CBs, which are at the interface of two or more cyclic structures formed by the interconnected NCBs between CO2, N2O and SO2 molecules and water molecules in their microhydrated clusters.
Method and computational details
The geometries of microhydrated clusters viz. (CO2)(H2O)n, (N2O)(H2O)n, and (SO2)(H2O)n with n = 1, 5 water molecules were optimized at the MP2/6-311++G(d,p) level of theory. Frequency calculations were performed at the same level of theory to confirm the local minimum nature of the optimized structures. Single point (SP) energy calculations at the MP2/aug-cc-pVTZ level is performed on the optimized structures and their fragments of the MTA-based method. The SP energies are then used to calculate the energies and CCs to TB, PB and CBs in these microhydrated clusters. All the calculations were carried out using the Gaussian 16 package.57Results and discussion
Types of cyclic cooperativity experienced by non-covalent bonds
To characterize the direction of flow of electron density from nucleophile to electron-deficient atoms of groups 14 (tetrels), 15 (pnictogens) and 16 (chalcogens), the molecular electrostatic potential (MESP) is useful. Fig. 1 illustrates the MESP mapped on the iso-density surface (value = 0.001 a.u.) along with the respective values of the MESP surface minima (Vmin) and maxima (Vmax) for various monomers and their respective dimeric complexes.The positions of these Vmin and Vmax values correspond to the site's electrophilic and nucleophilic attack, respectively. The electron-deficient regions on the tetrel (carbon), pnictogen (nitrogen) and chalcogen (sulphur) atoms are clearly evident from the appearance of surface maximum around the region perpendicular to the N2O (Vmax = 21.5 kcal mol−1) or CO2 (Vmax = 25.9 kcal mol−1) bond axis. Additionally, in the case of SO2 (Vmax = 32.3 kcal mol−1), it is observed over the S-atom. However, in the H2O molecule, the surface minima appear around the oxygen atom (Vmin = −32.2 kcal mol−1). For other molecules, it appears near the terminal hetero (N or S) atoms. Upon complex formation, the Vmin value on the O-atom of the water molecule is substantially increased (becoming less negative) in all the dimeric complexes. This is nicely complemented by the decrease (becoming less positive) in Vmax values, indicating the transfer of electron density from the water molecule to the counter-molecule, which establishes the NCBs, viz. PB, TB or CB.
With this understanding, one can identify the nature of the cyclic structure that may be observed in the microhydrated clusters of these molecules. Herein, instead of using the direction of the bond, the flow of electron density between pairs of atoms is considered. For TB, PB and CB, the flow of electron density is from the O of water to the C or N or S atoms of CO2, N2O and SO2, respectively. For X–H⋯Y HB, it is opposite the direction of the bond. In a cyclic structure resulting from the interconnected network of NCBs, a reference NCB may experience different types of cooperativity depending on the flow of electron density around it. For instance, consider the situations of an O⋯C TB between water and CO2 molecule within the cyclic trimer and tetramer (cf.Scheme 1) involving CO2 and 2 or 3 water molecules. The respective water molecules are labelled with their O atoms as O1, O2, and O3. In Scheme 1, the flow of electron density is shown in blue arrows. Depending upon the directions of these arrows (flow of electron density), a referenced TB experiences different types of cyclic cooperativity.
For example, in a cyclic trimer (cf.Scheme 1(a)), the direction of the flow of electron density between the three NCBs is the same (unidirectional). Therefore, the referenced TB between the C-atom CO2 and O2 of water experiences a nice flow of electron density (as also shown by the full blue dotted circle) around this TB. In other words, the referenced TB experiences a full cyclic cooperativity (FCC). A similar FCC is also revealed by a TB in a cyclic tetramer, as shown in Scheme 1(c). These TBs are observed to have a positive cooperativity owing to the formation of an interconnection of a network of NCBs with adjacent molecules. However, in the cyclic trimer shown in Scheme 1(b), the directions of the flow of electron density between C1⋯O2 TB and O2⋯O1 HB are opposite. A similar situation of flow of electron density is found in cyclic tetramers shown in Scheme 1(e). Note that in these structures, the flow of electron density is broken between the adjacent TB and HB. Therefore, the referenced TB (between C1 and O2) in these structures experiences anti-cooperativity (AC). The reference TB in these cyclic structures would have a negative cooperativity contribution. The intermediate situation is shown in Scheme 1(d). The directions of flow of electron density between O1⋯O2, O2⋯C1, and O4⋯O5 are all the same (unidirectional). However, this unidirectionality is broken at the HB between O1 and O5 (cf.Scheme 1(d)). In other words, the unidirectionality of the referenced TB between O2 and C1 is partially conserved. The TB in this cyclic tetramer (Scheme 1(d)) is said to experience positive partial cyclic cooperativity (PCC). Similarly, different types (FCC, PCC or AC) of cyclic cooperativity may be experienced by the pnictogen and chalcogen bonds between the O atom of a water molecule (electron donor) and the N- or S-atom (electron acceptor) of N2O or SO2. These situations of cyclic cooperativity corresponding to PB and CB are shown in Schemes S1 and S2 in the ESI.† The discussion of these is skipped herein for brevity.
Synergetic energies of tetrel bonds in microhydrated clusters of carbon dioxide
The MP2/6-311++G(d,p) optimized geometries of CO2(H2O)n, n = 3–5 clusters, are displayed in Fig. 2. Herein, the molecular clusters are labelled according to the number of water molecules and the cluster stability. For instance, in the label CWnA or CWnB, ‘C’ and ‘W’, respectively, represent carbon dioxide and water molecules, and ‘n’ is the number of water molecules in the given cluster. Label A corresponds to the energetically most stable cluster; the next one is denoted by label B and so on. Note that only geometries in which at least one TB bond is found to be common to two or more cyclic structures formed by the interconnected networks of NCBs are considered. To discuss the synergetic effect of the cyclic cooperativity, other geometries are not useful; hence, our discussion is restricted to the CO2(H2O)n, n = 3–5 clusters, as shown in Fig. 2. As depicted in Fig. 2, in large 3D molecular clusters, a TB common to two or more cyclic structures (labelled as ‘a’, ‘b’, ‘c’ and ‘d’ cycles) may experience FCC, PCC, or AC. Consider the geometry of the CW3B cluster (cf.Fig. 2 and Scheme S3 in ESI†). In this geometry, a C⋯O TB1 between CO2 and water molecule is common to two trimeric cycles a and b (Scheme S3 in ESI†). Both of these cyclic trimers exhibit the full cyclic cooperativity (FCC) towards TB1 common to them. It is postulated here that the strength of TB1 is determined solely by the total CCs of the two cyclic trimers common to TB1. In other words, the numerical addition of the CC of these cyclic trimers when added to the energy of TB1 in the respective isolated CO2⋯ H2O dimer (chopped out of CW3B cluster) gives the energy of TB1 in the CW3B cluster. To establish this, the CC towards TB1 from these two cycles is calculated using the MTA-based method.45Scheme 2 illustrates the fragmentation procedure of the MTA-based method for the estimation of energy and CCs towards TB1 in cyclic trimer a, consisting of CO2, and two water molecules denoted by O2 and O3. Recall that the TB1 corresponds to the C1⋯O2 interaction between CO2 and the water molecule O2. Hence, cyclic trimer a is divided into two primary fragments: F1 (obtained by removing water O2) and F2 (obtained by removing the CO2 molecule).
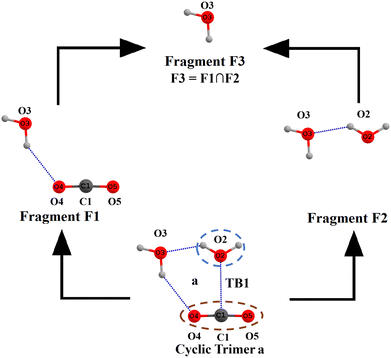 | ||
| Scheme 2 Fragmentation procedure of the MTA-based method for calculating the energy of TB1 in the cyclic trimer a of the CW3B cluster. | ||
Putting fragments F1 and F2 together, one may regenerate the geometry of this cyclic trimer a. However, such regenerated geometry reveals two features: (i) the TB1 between C1 and O2 present in cyclic trimer a is missed out, and (ii) there is a double counting of the common structural part (viz. water molecule O3) between F1 and F2 (cf.Scheme 2). This water molecule O3 is denoted as fragment F3. Upon the addition of SP energies of fragments F1 and F2 and subtraction of the energy of fragment F3, the energy of cyclic trimer a may be obtained. However, this energy of cyclic trimer a misses out on the TB1 energy (ETB1). Therefore, the TB1 energy in this cyclic trimer a is calculated as Ecyclic![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) trimer
trimer![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) aTB1 = (EF1 + EF2 − EF3) − Ecyclic
aTB1 = (EF1 + EF2 − EF3) − Ecyclic![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) trimer
trimer![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a = 3.28 kcal mol−1 at the MP2/aug-cc-pVTZ level. For EF1, EF2, EF3, and Ecyclic
a = 3.28 kcal mol−1 at the MP2/aug-cc-pVTZ level. For EF1, EF2, EF3, and Ecyclic![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) trimer a, see Table S1 in ESI.† Note that
trimer a, see Table S1 in ESI.† Note that  includes the CC coming from the interconnected network of HBs with TB1 formed by an additional water O3 molecule in this cyclic trimer a. Therefore, this
includes the CC coming from the interconnected network of HBs with TB1 formed by an additional water O3 molecule in this cyclic trimer a. Therefore, this 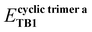 value is different from the TB1 energy in the respective isolated CO2⋯water O2 dimer (
value is different from the TB1 energy in the respective isolated CO2⋯water O2 dimer ( ). The latter is obtained by applying a supermolecular approach as follows:
). The latter is obtained by applying a supermolecular approach as follows:  . For EdimerC1⋯O2,
. For EdimerC1⋯O2,  , and Edimerwater
, and Edimerwater![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) O2, see Table S1 in ESI.† This
O2, see Table S1 in ESI.† This  value does not have a CC. The difference between
value does not have a CC. The difference between  and
and  gives CC to the TB1 in this cyclic trimer a, i.e.
gives CC to the TB1 in this cyclic trimer a, i.e. . Note that in this cyclic trimer a, the directions of electron density flow for all the three bonds (one TB and two HBs) are the same (unidirectional), which agrees with the calculated positive value of CC.
. Note that in this cyclic trimer a, the directions of electron density flow for all the three bonds (one TB and two HBs) are the same (unidirectional), which agrees with the calculated positive value of CC.
Similarly, the energy of TB1 and the CC in the cyclic trimer b (cf.Fig. 2) is calculated using the MTA-based method to be Ecyclic![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) trimer
trimer![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) bTB1 = 3.29 and
bTB1 = 3.29 and  , respectively (cf. Schemes S3 and S4 in ESI†). Recall that this cyclic trimer b also exhibits FCC, which agrees with the calculated positive value of CC. Therefore, the total CC of cyclic trimers a and b towards TB1 is
, respectively (cf. Schemes S3 and S4 in ESI†). Recall that this cyclic trimer b also exhibits FCC, which agrees with the calculated positive value of CC. Therefore, the total CC of cyclic trimers a and b towards TB1 is  . This total CC when added to the TB1 energy in the isolated dimer gives the synergetic energy of TB1 in the CW3B cluster, i.e.
. This total CC when added to the TB1 energy in the isolated dimer gives the synergetic energy of TB1 in the CW3B cluster, i.e. . The TB1 energy in the actual CW3B cluster by applying the MTA-based method is also evaluated; see Scheme S5 in ESI† and discussion below it. The TB1 energy obtained by applying the MTA-based method employing the actual CW3B cluster (
. The TB1 energy in the actual CW3B cluster by applying the MTA-based method is also evaluated; see Scheme S5 in ESI† and discussion below it. The TB1 energy obtained by applying the MTA-based method employing the actual CW3B cluster ( ) instead of cyclic trimers is very similar to the
) instead of cyclic trimers is very similar to the  value. The absolute difference (|ΔETB|) is small (0.07 kcal mol−1). This small |ΔETB| value suggests that the TB1 strength in the CW3B cluster is accurately calculated using the proposed method of synergetic cyclic cooperativity.
value. The absolute difference (|ΔETB|) is small (0.07 kcal mol−1). This small |ΔETB| value suggests that the TB1 strength in the CW3B cluster is accurately calculated using the proposed method of synergetic cyclic cooperativity.
With this understanding, the energies of tetrel bonds in various geometries of CO2(H2O)n, n = 3 to 5 clusters (cf.Fig. 2) by the present synergetic cyclic cooperativity approach and also by the MTA-based method employing actual clusters are evaluated. These TB energies in CO2(H2O)n, n = 3 to 5 clusters, calculated by applying these two methods are compared in Table 1.
| TB | E DimerTB | E Cycle1coop. | E Cycle2coop. | E SynergeticTB | E Actual ClusterTB | |ΔETB| |
|---|---|---|---|---|---|---|
| a Partially optimized. | ||||||
| CW3A | ||||||
| TB1 | 1.21 | −0.21 | −0.10 | 0.90 | 0.84 | 0.06 |
| TB2 | 2.42 | 0.40 | −0.10 | 2.72 | 2.66 | 0.06 |
| CW3B | ||||||
| TB1 | 2.72 | 0.56 | 0.57 | 3.85 | 3.78 | 0.07 |
| CW4A | ||||||
| TB1 | 2.09 | −0.24 | 0.43 | 2.28 | 2.08 | 0.20 |
| CW4B | ||||||
| TB1 | 1.12 | −0.23 | −0.13 | 0.76 | 0.69 | 0.07 |
| TB2 | 2.44 | −0.13 | 0.81 | 3.11 | 3.05 | 0.05 |
| CW4D | ||||||
| TB1 | 1.24 | −0.05 | −0.23 | 0.96 | 0.88 | 0.08 |
| TB2 | 2.16 | −0.05 | 0.41 | 2.52 | 2.34 | 0.18 |
| CW4E | ||||||
| TB1 | 2.90 | 0.85 | 0.21 | 3.96 | 3.90 | 0.06 |
| CW5A | ||||||
| TB1 | 2.43 | 0.64 | −0.21 | 2.86 | 2.52 | 0.34 |
| CW5B | ||||||
| TB1 | 1.93 | −0.01 | −0.01 | 1.90 | 1.91 | 0.01 |
| TB2 | 1.88 | −0.01 | −0.26 | 1.61 | 1.67 | 0.07 |
| CW5D | ||||||
| TB1 | 1.83 | 0.40 | −0.25 | 1.98 | 1.68 | 0.30 |
| CW5E | ||||||
| TB1 | 1.21 | −0.08 | −0.20 | 0.94 | 1.01 | 0.07 |
| TB2 | 2.21 | −0.08 | 0.40 | 2.53 | 2.31 | 0.22 |
| CW5F | ||||||
| TB1 | 2.94 | 1.02 | 1.02 | 4.98 | 4.90 | 0.08 |
| CW5G | ||||||
| TB1 | 2.88 | 0.85 | 0.45 | 4.18 | 4.14 | 0.04 |
| CW5H | ||||||
| TB1 | 2.93 | 0.44 | 0.44 | 3.80 | 3.78 | 0.02 |
| CW5I | ||||||
| TB1 | 1.85 | −0.41 | 0.88 | 2.32 | 2.35 | 0.03 |
| CW5J | ||||||
| TB1 | 0.49 | −0.56 | −0.15 | −0.21 | −0.18 | 0.03 |
As shown in Table 1 and Fig. 2, two TBs (TB1 and TB2) are present in the CW3A cluster. The TB1 bond is common to two trimeric cycles a (consisting of O1, O2 and CO2) and b (consisting of O1, O3 and CO2), both exhibiting AC as also reflected from their negative CCs (−0.21 and −0.1 kcal mol−1, respectively). The TB2 is also common to two trimeric cycles, i.e.cycle a (consisting of O1, O2 and CO2) and cycle d (consisting of O2, O3 and CO2). As discussed, trimeric cycle a exhibits AC. However, cycle d presents an FCC in which the flow of electron density between bonds is unidirectional. This FCC nature of cycle d is also evident from the positive value of CC (0.40 kcal mol−1). Therefore, the calculated energy of TB2 is (2.72 kcal mol−1) larger than that of TB1 (0.90 kcal mol−1). Importantly, these calculated values of TB1 and TB2 by applying the method of synergetic cyclic cooperativity are in excellent agreement with their actual cluster counterparts (0.84 and 2.66 kcal mol−1) calculated using the MTA-based method.
In the CW4A cluster, TB1 is common to two trimeric cycles a and b. Here, cycle a bears AC; the direction of the flow of electron density between O3⋯O1 and O3⋯C1 is opposite to each other. This AC nature of cycle a is also evident in the negative value of CC (−0.24 kcal mol−1) to TB1. However, trimeric cycle b exhibits FCC (cf.Fig. 2) with positive CC (0.43 kcal mol−1) to TB1. By adding the total CC of both of these cycles to TB1 energy in the isolated dimers, the synergetic TB1 energy is obtained to be 2.28 kcal mol−1. This TB1 energy is in good agreement with its energy (2.08 kcal mol−1) in the actual CW4A cluster calculated by applying the MTA-based method. The structure of CW4B is very similar to that of CW3A, except that there is an extra water molecule, O4, between O3 and the CO2 molecule. The TB1 of CW4B is common to one trimeric AC (CC = −0.13 kcal mol−1) cycle a (consisting of two water molecules O1, O2 and CO2 molecule) and one tetrameric cycle b (consisting of three water molecules O1, O3, O4 and CO2 molecule), which also exhibits AC (CC = −0.23 kcal mol−1) (Scheme S6 in ESI†). The TB2 of CW4B is common to one trimeric AC (CC = −0.13 kcal mol−1) cycle a and one tetrameric cycle d (consisting of three water molecules O2, O3, and O4 and a CO2 molecule), which exhibits FCC (CC = 0.81 kcal mol−1). Because of this FCC nature of cycle d, the TB2 (3.11 kcal mol−1) is stronger than TB1 (0.76 kcal mol−1). In the optimized structure of CW4C, there is only one tetrameric cycle, which consists of TB1. Therefore, its geometry for the discussion of its energy evaluation by the synergetic cooperativity evaluation is not considered. The next important cluster is the CW4D cluster, consisting of three tetrel bonds, viz.TB1, TB2 and TB3. Of these TBs, TB3 is not a part of any cyclic structure; therefore, its discussion is skipped herein, and its energy can be evaluated using the direct MTA-based method. The TB1 is common to two trimeric cycles, a (consisting of water molecules O1, O2, and CO2) and b (consisting of two water molecules O1, O3, and CO2), both bearing AC with CCs of −0.05 and −0.23 kcal mol−1, respectively. The TB2 is also common to AC trimeric cycle a and cycle d (consisting of water molecules O2, O3, and CO2), which bears an FCC (CC = 0.41 kcal mol−1). Therefore, the synergetic energy of TB2 (2.52 kcal mol−1) is larger than that of TB1 (0.96 kcal mol−1). The CW4E is an interesting cluster. Here, the tetrel bond TB1 is common to one trimeric cycle a (consisting of O3 and O4 water molecules and CO2) bearing FCC and another tetrameric cycle b (consisting of O3, O5, and O6 water molecules and CO2), which exhibits partial cyclic cooperativity (PCC). Because of these positive FCC (0.85 kcal mol−1) and PCC (0.21 kcal mol−1) contributions of these cyclic structures, synergistic TB1 energy (3.96 kcal mol−1) is the largest among all CW4 clusters. This energy calculated by the synergistic effect of cyclic cooperativity is in good agreement with its actual cluster counterpart (3.90 kcal mol−1) calculated by applying the MTA-based method.
In the CW5A cluster (cf.Fig. 2), TB1 is common to AC trimeric cycle b and FCC tetrameric cycle a, with CC contributions of −0.21 and 0.64 kcal mol−1, respectively. The calculated synergetic energy of TB1 is 2.86 kcal mol−1, which is in good agreement with the one calculated by applying the MTA-based method with the actual CW5A cluster (2.52 kcal mol−1). In the CW5B cluster, TB1 is common to two tetrameric cycles a (consisting of O1, O4, and O7 water molecules and CO2) and b (consisting of O1, O2, and O3 water molecules and CO2), both bearing AC (CC = −0.01 kcal mol−1 for both cycles); see Scheme S6 in ESI.† Because of these two AC cycles, the energy of TB1 is very small, 1.9 kcal mol−1. The energy of TB2 in this CW5B cluster is even smaller (1.61 kcal mol−1), which may be attributed to the fact that TB2 is common to one tetrameric cycle a and trimeric cycle c (consisting of O3 and O7 water molecules and CO2), both bearing AC (cf. Scheme S6 in ESI†) with CC values of −0.01 and −0.24 kcal mol−1, respectively. These synergistic energies of TB1 and TB2 are in excellent agreement with their actual cluster counterparts (cf.Table 1). In the CW5D cluster, TB1 is common to two trimeric cycles: a (consisting of O3 and O4 water molecules and CO2) and b (consisting of O3, O1 water molecules and CO2). Former cycle a bears FCC with a CC of 0.40 kcal mol−1. However, trimeric cycle b exhibits AC with a CC of −0.25 kcal mol−1. The calculated synergetic energy of TB1 is 1.98 kcal mol−1 is in good agreement with the one calculated by applying the MTA-based method employing the actual CW5D cluster (1.68 kcal mol−1). The structure of the CW5E cluster is similar to that of the CW4D cluster, except that the additional water molecule forms a cyclic structure involving TB3. The calculated synergistic energies of TB1 (0.94 kcal mol−1) and TB2 (2.53 kcal mol−1) in the CW5E cluster are similar to those in the CW4D (0.96 and 2.52 kcal mol−1) cluster. The energies of TB1 in CW5F, CW5G, CW5H, CW5I, and CW5J clusters, wherein TB1 is common to two tetrameric cycles, are discussed. Although these structures of clusters look similar, they differ in terms of cooperativity exhibited by the two tetrameric cycles towards TB1. For instance, two tetrameric cycles (a and b) exhibit FCC in the CW5F cluster (cf.Fig. 2), as is evident from positive CCs (1.02 kcal mol−1) for both the cycles (cf.Table 1). Because of these positive CCs of both cycles, the TB1 energy (4.98 kcal mol−1) is largest among all the CWn (n = 3–5) clusters studied herein. The energy of TB1 is slightly smaller (4.18 kcal mol−1) in the CW5G cluster compared to the CW5F cluster. This may be because in the CW5G cluster, tetrameric cycle a exhibits FCC (CC = 0.85 kcal mol−1) to TB1, while cycle b bears partial cyclic cooperativity (PCC) with smaller but positive CC (0.45 kcal mol−1). However, since both the cycles (a and b) in the CW5H cluster (cf.Fig. 2) exhibit PCC with CC of each being 0.44 kcal mol−1. The energy of TB1 is smaller (3.80 kcal mol−1) than its value in CW5F and CW5G clusters. A partially optimized CW5I cluster is also considered, wherein TB1 is at the interface of two cyclic tetrameric structures, of which one exhibits AC (cycle a) and the other shows FCC (cycle b) (Fig. 2). The CC of cycle a is −0.41 kcal mol−1 and that of cycle b is 0.88 kcal mol−1, which is consistent with the nature of these cyclic structures. Another partially optimized cluster is CW5J, wherein TB1 is at the interface of two tetrameric cycles a and b, both exhibiting AC. The CC of cycle a is −0.56 kcal mol−1 and that of cycle b is −0.15 kcal mol−1, which are consistent with their AC nature. Although the TB1 energy in the isolated dimer is small and positive, the energy of TB1 in the actual cluster is small and negative (−0.21 kcal mol−1). The energy of TB1 calculated by the synergistic cooperative effects of the two cycles is in excellent agreement with its value calculated by full calculation using the MTA-based method (−0.18 kcal mol−1). The negative value indicates that this bond is unstable in the cluster compared to when it is isolated as a dimer. Note that the structures exhibiting one or two AC cycles are partially optimized (with frozen atoms). These structures cannot be fully optimized in the present form. The reason could be the instability of these structures because of the formation of AC cycle(s). It is emphasized here that the interplay of the nature of cyclic structures common to a TB in all the CWn (n = 3–5) clusters studied in this work is nicely reflected in the calculated synergistic energy of such TBs. This suggests the robustness of the proposed methodology. Importantly, all the calculated synergistic energies of TBs in various CWn (n = 3–5) clusters are in excellent agreement with their full-calculation counterparts. The maximum absolute difference between two values (|ΔETB|) is less than 0.4 kcal mol−1, suggesting that the present approach of synergistic cooperativity provides accurate estimates of the energy of various tetrel bonds in CO2(H2O)n clusters.
Synergetic energies of pnictogen bonds in microhydrated clusters of nitrous oxide
Similarly, various pnictogen bonds (PB), which are at the interface of two or more cyclic structures formed owing to interconnected networks of NCBs in microhydrated clusters of nitrous oxide (N2O), are investigated. The MP2/6-311++G(d,p) optimized geometries of various microhydrated clusters N2O, viz. the N2O(H2O)n, n = 3–5 clusters, are shown in Fig. 3.Herein, these clusters are labelled similar to CO2(H2O)n clusters, i.e. according to the number of water molecules and the cluster stability. For instance, in the label NWnA or NWnB, ‘N’ and W represent nitrous oxide and water molecule, respectively. ‘n’ is the number of water molecules in a given cluster. Label A corresponds to the energetically most stable cluster; the next one is denoted by label B and so on. Further, only geometries wherein at least one PB bond is found to be common to two or more cyclic structures are considered (Fig. 3). As depicted in Fig. 3, similar to a TB, a PB is found to be common to two or more cyclic structures and may experience an FCC, PCC, or AC in these microhydrated N2O clusters. For instance, consider the geometry of the NW3C cluster shown in Fig. 3. In this geometry, an N⋯O pnictogen bond (PB1) between N2O and a water molecule is common to two cyclic trimers a and b. Both of these cyclic trimers exhibit the FCC to PB1. Therefore, the energy of this PB1 can be determined by the total CCs of these two cyclic trimers. The CCs towards PB1 of both cycles are estimated by applying the MTA-based method. For details of fragmentation procedures for the calculation of PB1 energy and the respective CCs in cycles a and b, see Schemes S7 and S8 in ESI.† The PB1 energy along with the respective CCs of each of these trimeric cycles is reported in Table 2. As shown in Table 2, the cooperativity contributions of the two cycles are 0.41 and 0.30 kcal mol−1, respectively. The calculated positive CCs are consistent with the FCC nature of these two cycles. When these two CCs are added to the energy of PB1 in the respective dimer, the synergetic energy of PB1 in the NW3C cluster is obtained to be 3.05 kcal mol−1. This calculated energy of PB1 is in excellent agreement with its value in the actual cluster counterparts (2.95 kcal mol−1) calculated by applying the MTA-based method; see Scheme S9 in ESI† for the fragmentation procedure. The difference between the energy of PB1 calculated by these two approaches is negligibly small (0.1 kcal mol−1), indicating that the proposed method of synergetic cooperativity also provides an accurate estimation of the energy of PB. It is mentioned here that this is the only tri-hydrated N2O cluster structure wherein the PB1 is found to be common to two trimeric cyclic structures. The other lower energy conformers, viz.NW3A and NW3B clusters, have PB1 as part of only one cycle (Scheme S10 in ESI†).
| PB | E DimerPB | E Cycle1coop. | E Cycle2coop. | E SynergeticPB | E actualPB | |ΔEPB| |
|---|---|---|---|---|---|---|
| a Partially optimized. | ||||||
| NW3C | ||||||
| PB1 | 2.34 | 0.41 | 0.30 | 3.05 | 2.95 | 0.10 |
| NW4A | ||||||
| PB1 | 2.10 | −0.20 | 0.40 | 2.30 | 2.00 | 0.30 |
| NW4B | ||||||
| PB1 | 1.25 | −0.10 | −0.24 | 0.91 | 0.83 | 0.08 |
| PB2 | 2.31 | −0.10 | 0.73 | 2.94 | 2.87 | 0.07 |
| NW4D | ||||||
| PB1 | 2.12 | 0.22 | −0.12 | 2.22 | 2.16 | 0.06 |
| NW4E | ||||||
| PB1 | 1.32 | −0.08 | −0.21 | 1.03 | 1.00 | 0.03 |
| PB2 | 2.21 | −0.06 | 0.37 | 2.52 | 2.32 | 0.20 |
| NW4G | ||||||
| PB1 | 1.39 | −0.10 | −0.17 | 1.12 | 1.26 | 0.14 |
| PB2 | 2.28 | −0.10 | 0.30 | 2.48 | 2.40 | 0.08 |
| NW5A | ||||||
| PB1 | 2.33 | 0.51 | −0.16 | 2.68 | 2.32 | 0.36 |
| NW5B | ||||||
| PB1 | 2.02 | 0.17 | 0.77 | 2.96 | 2.86 | 0.10 |
| NW5C | ||||||
| PB1 | 2.51 | −0.07 | 0.73 | 3.17 | 3.02 | 0.15 |
| PB2 | 1.14 | −0.08 | −0.34 | 0.72 | 0.48 | 0.24 |
| NW5D | ||||||
| PB1 | 2.44 | 0.83 | 0.11 | 3.38 | 3.31 | 0.07 |
| NW5E | ||||||
| PB1 | 2.63 | 0.18 | 0.37 | 3.18 | 3.15 | 0.02 |
| NW5F | ||||||
| PB1 | 1.97 | 0.38 | −0.23 | 2.12 | 1.77 | 0.35 |
| NW5G | ||||||
| PB1 | 1.34 | −0.08 | −0.15 | 1.11 | 1.21 | 0.10 |
| PB2 | 2.29 | −0.09 | 0.33 | 2.53 | 2.28 | 0.25 |
| NW5H | ||||||
| PB1 | 1.39 | −0.19 | 0.65 | 1.85 | 1.85 | 0.00 |
| NW5I | ||||||
| PB1 | 1.03 | −0.07 | −0.17 | 0.79 | 0.88 | 0.09 |
With this understanding of the method of the synergetic cyclic cooperativity predicting the accurate energy of PB1 in the NW3C cluster, this method is applied to determine the energy of various PBs in higher order (n = 4 and 5) microhydrated clusters of N2O. For instance, in the NW4A cluster, the N2O molecule seems to interact with the water tetramer, forming a PB1 bond common to two trimeric cycles a and b. Herein, cycle a bears an AC, while cycle b bears an FCC. The AC and FCC natures of these cycles are nicely reflected in terms of the calculated CCs being −0.20 and 0.40 kcal mol−1, respectively. By adding the total CCs of these cycles to PB1 energy in the isolated dimers, the synergetic PB1 energy is obtained (2.30 kcal mol−1), which is in good agreement with its value (2.00 kcal mol−1) in the actual NW4A cluster calculated by applying the MTA-based method. The structure of NW4B is somewhat different from that of NW4A. In NW4B, a cyclic water trimer (denoted as d) interacts with the N2O molecule, forming two bifurcated pnictogen bonds: PB1 and PB2. The fourth water molecule (denoted by O2) forms two HBs: one with the terminal N of N2O and the other with the H of water O2 (cf.Fig. 3). The PB1 is at the intersection of one trimeric cycle b (consisting of N2O, O1 and O5 water molecules) and one tetrameric cycle a (consisting of N2O, and three water molecules O1, O2, and O3), both exhibiting AC to PB1 (Scheme S11 in ESI†). The AC nature of these two cyclic structures common to PB1 is nicely reflected in their negative CCs to PB1 (−0.10 and −0.24 kcal mol−1, respectively). Thus, the calculated synergetic PB1 energy is very small (0.91 kcal mol−1). However, PB2 is common to one trimeric AC cycle b (also common to PB1) and a tetrameric cycle c (consisting of N2O, and three water molecules O3, O2 and O5) bearing FCC to PB2 (cf. Scheme S11 in ESI†). The CC of FCC cycle c is 0.73 kcal mol−1. This is the reason that the synergetic energy of PB2 is larger (2.94 kcal mol−1) than that of PB1. The energy of PB1 and PB2 calculated by the method of synergetic cyclic cooperativity is in excellent agreement with their actual cluster counterparts calculated by applying the MTA-based method (cf.Table 2). The difference in the energy calculated by applying the two methods is less than 0.08 kcal mol−1. The structure of the NW4D cluster is similar to that of the NW4B cluster except that the PB2 of NW4B is converted to HB between water and the terminal O-atom of N2O. Therefore, there is only a PB1 bond, which is at the interface of one trimeric cycle a bearing an FCC and the other tetrameric cycle b bearing an AC (cf.Fig. 3). The respective CCs of these two cycles are 0.22 and −0.12 kcal mol−1. The synergetic energy of PB1 is 2.22 kcal mol−1, which is in excellent agreement with its actual cluster counterpart (2.16 kcal mol−1). In NW4E, there are three pnictogen bonds (PB1, PB2 and PB3). The energy of PB1 and PB2 can be obtained using the method of synergetic cyclic cooperativity. The PB1 is at the intersection of two AC trimeric cycles a (consisting of N2O and two water O1 and O3 molecules) and b (consisting of N2O and two water molecules O1 and O2). The AC nature of these cycles is also reflected in the respective negative CCs (−0.08 and −0.21 kcal mol−1), resulting in a weak (ESynergeticPB1 = 1.03 kcal mol−1 and EactualPB1 = 1.00 kcal mol−1) PB1 bond. AC cycle b is also common to PB2 along with FCC trimeric cycle d (consisting of N2O and two water molecules O2 and O3). The positive CCs (0.37 kcal mol−1) of FCC cycle d along with the small AC of cycle b (−0.06 kcal mol−1) make PB2 stronger (ESynergeticPB2 = 2.52 kcal mol−1 and EactualPB2 = 2.32 kcal mol−1) than PB1. The structure of NW4G is similar to that of NW4E, except that the fourth water molecule forms HB with the O-atom of N2O instead of PB3. The smaller synergistic energy of PB1 (1.12 kcal mol−1) than that of PB2 (2.48 kcal mol−1) in the NW4G cluster may be similarly explained. Importantly, these energies of PB1 and PB2 are in excellent agreement with those calculated by employing the actual NW4G cluster in the MTA-based method.
Similar to a tetra-hydrated NW4A cluster, the most stable NW5A cluster has a cyclic water pentamer interacting with N2O, forming a PB1 bond common to one tetrameric FCC cycle a and one trimeric AC cycle b. The FCC and AC natures of these cyclic structures are nicely reflected in the calculated positive (0.51 kcal mol−1) and negative (−0.16) CCs. Upon adding these CCs to the PB1 energy in the isolated ON2⋯water dimer, synergetic PB1 energy (2.68 kcal mol−1) is obtained. This synergetic PB1 energy is in good agreement with its value (2.32 kcal mol−1) obtained using the MTA-based method with the actual NW5A cluster. In the higher energy penta-hydrated clusters, the N2O molecule interacts with cyclic water tetramer (NW5C and NW5F) and cyclic water trimer (NW5G). In the NW5C cluster, there are two PBs (PB1 and PB2). The PB1 is common to one AC trimeric cycle b (consisting of N2O and water molecules O3 and O5) and an FCC pentameric cycle a (consisting of N2O and O1, O2, O6, and O5 water molecules); see Scheme S11 in ESI.† The AC and FCC natures of these cycles result in negative (−0.07 kcal mol−1) and positive (0.73 kcal mol−1) CCs to PB1, respectively. The synergetic energy of PB1 is 3.17 kcal mol−1, which is in good agreement with its value (3.02 kcal mol−1) in the actual NW5C cluster. However, the energy of PB2 is much smaller (0.72 kcal mol−1) compared to that of PB1. This may be attributed to the fact that PB2 resides at the interface of two AC cycles, viz. trimeric cycle b (also common to PB1) and tetrameric cycle c (consisting of N2O and O1, O2, and O3 water molecules). The negative CCs (−0.08 and −0.34 kcal mol−1) of these cycles make PB2 very weak. In the NW5F cluster, PB1 is at the intersection of one FCC (cycle a) and another AC (cycle b) trimeric cycle with their respective CCs being 0.38 and −0.23 kcal mol−1, respectively. The synergetic energy of PB1 is 2.12 kcal mol−1, and that in the actual NW5F cluster is very close to it (1.77 kcal mol−1). The structure of NW5G is similar to that of NW4G discussed above, except that the 5th water molecule (denoted as O5) forms HB with the 4th water molecule (O4) along with an additional PB (PB3). This PB3 is part of only one cyclic structure; therefore, its energy can be evaluated using the standard MTA-based method employing the NW5G cluster. The PB1 is at the intersection of two AC trimeric cycles a and b (cf. Scheme S11 in ESI†), with their respective CCs being −0.08 and −0.15 kcal mol−1. Because of these two negative CCs, the energy of PB1 is small (ESynergeticPB1 = 1.11 kcal mol−1 and EactualPB1 = 1.21 kcal mol−1, respectively). However, the PB2 is common to one AC trimeric cycle a (also common to PB1) and another trimeric FCC cycle c (consisting of N2O and O2 and O3 water molecules). The positive CC (0.33 kcal mol−1) of this FCC cycle c and the small negative CC (−0.09 kcal mol−1) of cycle a make PB2 (ESynergeticPB2 = 2.53 kcal mol−1 and EactualPB2 = 2.28 kcal mol−1) stronger than PB1.
The structure of NW5D is interesting as it bears the PB1 at the intersection of one FCC tetrameric cycle a (consisting of N2O and O2, O4, and O6 water molecules) and another PCC tetrameric cycle b (consisting of N2O and O2, O3, and O5 water molecules) (Fig. 3). The CCs of these cycles are 0.83 and 0.11 kcal mol−1, respectively, leading to one of the strongest PB1 bond (ESynergeticPB1 = 3.38 kcal mol−1 and EactualPB1 = 3.31 kcal mol−1, respectively). Starting from NW5D, various structures with a PB1 bond at the intersection of two cycles (similar to NW5D) bearing different combinations of types of cooperativities are generated. For instance, in NW5E, both tetrameric cycles exhibit PCC to PB1 with their respective CCs being 0.18 and 0.37 kcal mol−1, making PB1 slightly weaker (ESynergeticPB1 = 3.18 kcal mol−1 and EactualPB1 = 3.15 kcal mol−1) in NW5E compared to PB1 in NW5D. In NW5H, tetrameric cycle a is AC (CC = −0.19 kcal mol−1), while tetrameric cycle b is FCC (CC = 0.65 kcal mol−1) with synergetic energy of PB1 to be 1.85 kcal mol−1, which is in good agreement with its value in the actual cluster (EactualPB1 = 1.85 kcal mol−1). It should be noted here that the NW5H structure is partially optimized. On full optimization, this combination of cyclic structures is not retained. Similarly, a partially optimized structure with both (cycles a and b) AC cycles (CCs: −0.07 and −0.17 kcal mol−1, respectively) leads to the weakest PB1 bond (ESynergeticPB1 = 0.79 kcal mol−1 and EactualPB1 = 0.88 kcal mol−1) among all the clusters in this class. It is emphasized here that the interplay of the nature of cyclic structures common to PBs in all NWn (n = 3–5) clusters is nicely reflected in their calculated synergistic energies, once again highlighting the robustness of the proposed approach. Importantly, all the calculated synergistic PB energies are in excellent agreement with their full-calculation counterpart with |ΔEPB| falling between 0.00 and 0.36 kcal mol−1.
Synergetic energies of chalcogen bonds in microhydrated clusters of sulphur dioxide
With the above understanding of the method of synergetic cyclic cooperativity for evaluating the accurate energy of TBs and PBs, the applicability of this method is tested for estimating the energy of chalcogen bonds (CBs) in microhydrated clusters of SO2. Recall that the electron-deficient region around the S-atom of SO2 interacts with the electron-rich O-atom of water to form a dimeric complex stabilized by a CB. The MP2/6-311++G(d,p) optimized geometries of various microhydrated clusters of SO2, viz. SO2(H2O)n, n = 3–5, are shown in Fig. 4. The nomenclature SWn of SO2(H2O)n clusters is similar to those of CO2(H2O)n and N2O(H2O)n clusters. As depicted in Fig. 4, a CB is also found to be common to two or more cyclic structures formed owing to the interconnected networks of HBs (HO–H⋯O(H2) and HO–H⋯O(SO)) and a CB ((H2)O⋯SO2). For instance, in the SW3B cluster, the CB1 bond is common to two trimeric cycles: a and b. Therefore, the energy of this CB1 can be calculated by adding the CCs of these cycles to its energy in the respective isolated dimer chopped out of the SW3B cluster. The CCs of these cycles were evaluated by applying the MTA-based method (Schemes S12–S14 in ESI†). The discussion of these schemes is very similar to those in Scheme 1 and Schemes S4, S5 (ESI†) for TB and Schemes S7–S9 (ESI†) for PB; therefore, we skipped it for brevity. As shown in Schemes S12 and S13 in ESI,† the CCs of these two trimeric cycles are 2.70 and 2.69 kcal mol−1, which are consistent with the fact that both of these cycles exhibit positive FCC to CB1 in the SW3B cluster. On adding the total CCs to the energy of CB1 in the isolated H2O⋯SO2 dimer (3.45 kcal mol−1), the synergetic energy of CB1 is obtained (8.84 kcal mol−1), which is in good agreement with its value in the actual SW3B cluster (8.91 kcal mol−1; see Scheme S14, ESI†), indicating that the proposed method also works for the CB.With this understanding of the accurate evaluation of CB energy in the SW3B cluster, the synergetic cyclic cooperativity method is applied to different CBs in microhydrated SWn clusters. For example, in the lowest energy SW3A cluster, two bifurcated CBs are formed. The CB1 is common to two trimeric cycles a and b, of which cycle a exhibits FCC (CC = 1.28 kcal mol−1) while cycle b exhibits AC (CC = −0.38 kcal mol−1); see Scheme S15 in ESI.†Cycle b is also common to CB2 with the same value of CC. The other cycle c exerts AC to CB2 (CC = −0.42 kcal mol−1). Therefore, CB2 is much weaker (ESynergeticCB2 = −0.16 kcal mol−1 and EactualCB2 = −0.27 kcal mol−1) than CB1 (ESynergeticCB1 = 5.01 kcal mol−1 and EactualCB1 = 4.90 kcal mol−1) in the SW3A cluster. The negative CB2 energy indicates the destabilization of this interaction in the SW3A cluster compared to the isolated dimer (EDimerCB2 = 0.65 kcal mol−1).
In the lowest energy SW4A cluster, the CB1 bond is at the intersection of two trimeric cycles, of which one is AC (cycle a, CC = −0.76 kcal mol−1) and the other is FCC (cycle b, CC = 1.80 kcal mol−1). Because of these AC and FCC combinations of cyclic structures, the CB1 in the SW4A cluster is of moderate strength (ESynergeticCB1 = 4.78 kcal mol−1 and EactualCB1 = 4.43 kcal mol−1). However, the CB1 in the SW4B cluster is common to one FCC trimeric cycle a (CC = 1.75 kcal mol−1) and another AC tetrameric cycle b (CC = 0.09 kcal mol−1). Upon adding these CCs to CB1 energy in the respective dimer, the obtained synergetic CB1 energy (5.42 kcal mol−1) is in excellent agreement with its value in the actual SW4B cluster (5.39 kcal mol−1) calculated by applying the MTA-based method (Table 3). In the SW4C cluster, there are two CBs at the interface of the two cyclic structures. The CB1 is common to trimeric cycle b (consisting of SO2 and O1 and O4 water molecules) and tetrameric cycle a (consisting of SO2 and O1, O2, and O3 water molecules), both exhibiting AC (Scheme S15 in ESI†). The respective negative CCs (−0.30 and −0.46 kcal mol−1) make the CB1 bond weak (ESynergeticCB1 = 0.12 kcal mol−1 and EactualCB1 = −0.003 kcal mol−1). However, CB2 is at the intersection of trimeric cycle b (also common to CB1), exhibiting AC (CC = −0.46 kcal mol−1) and tetrameric cycle c (consisting of SO2 and O4, O2, and O3 water molecules), which shows FCC (CC = 1.82 kcal mol−1). Because one of the cycles has a positive CC, the energy of CB2 is moderate (ESynergeticCB1 = 4.90 kcal mol−1 and EactualCB1 = 4.78 kcal mol−1). In the SW4D cluster, the CB1 bond is common to one PCC tetrameric cycle a (CC = 1.10 kcal mol−1) and one FCC trimeric cycle b (CC = 2.38 kcal mol−1). This makes CB1 in the SW4D cluster one of the strongest (ESynergeticCB1 = 7.35 kcal mol−1 and EactualCB1 = 7.37 kcal mol−1) bond among all the tetra-hydrated complexes of SO2.
| CB | E DimerCB | E Cycle1coop. | E Cycle2coop. | E SynergeticCB | E actualCB | |ΔECB| |
|---|---|---|---|---|---|---|
| a Partially optimized. | ||||||
| SW3A | ||||||
| CB1 | 4.11 | 1.28 | −0.38 | 5.01 | 4.90 | 0.11 |
| CB2 | 0.65 | −0.42 | −0.38 | −0.16 | −0.27 | 0.11 |
| SW3B | ||||||
| CB1 | 3.45 | 2.70 | 2.69 | 8.84 | 8.91 | 0.07 |
| SW4A | ||||||
| CB1 | 3.74 | −0.76 | 1.80 | 4.78 | 4.43 | 0.35 |
| SW4B | ||||||
| CB1 | 3.59 | 1.75 | 0.09 | 5.42 | 5.39 | 0.03 |
| SW4C | ||||||
| CB1 | 0.88 | −0.30 | −0.46 | 0.12 | −0.003 | 0.12 |
| CB2 | 3.52 | −0.46 | 1.84 | 4.90 | 4.78 | 0.12 |
| SW4D | ||||||
| CB1 | 3.87 | 1.10 | 2.38 | 7.35 | 7.37 | 0.02 |
| SW5A | ||||||
| CB1 | 3.91 | 2.68 | 0.73 | 7.32 | 7.33 | 0.01 |
| SW5B | ||||||
| CB1 | 4.13 | −0.86 | 1.73 | 5.00 | 4.69 | 0.31 |
| CB2 | 1.40 | −0.71 | −0.23 | 0.46 | 0.15 | 0.31 |
| SW5C | ||||||
| CB1 | 1.77 | 4.97 | 4.80 | 11.54 | 12.13 | 0.59 |
| SW5D | ||||||
| CB1 | 3.28 | 1.93 | −0.51 | 4.70 | 4.48 | 0.22 |
| CB2 | 1.02 | −0.51 | −0.26 | 0.25 | 0.08 | 0.17 |
| SW5E | ||||||
| CB1 | 0.53 | 4.26 | 4.27 | 9.06 | 9.43 | 0.37 |
| SW5F | ||||||
| CB1 | 2.53 | 1.24 | 3.00 | 6.77 | 6.81 | 0.04 |
| SW5G | ||||||
| CB1 | 2.78 | 1.46 | 1.49 | 5.73 | 5.74 | 0.01 |
| SW5H | ||||||
| CB1 | 3.08 | 3.23 | −1.26 | 5.05 | 4.82 | 0.23 |
| SW5I | ||||||
| CB1 | 3.57 | −0.68 | −0.62 | 2.27 | 2.38 | 0.11 |
In the lowest energy penta-hydrated SW5A cluster, the CB1 bond is at the interface of two tetrameric cycles, of which one possesses FCC (cycle a) and the other possesses PCC (cycle b). The respective cooperativity contributions are 2.68 and 0.73 kcal mol−1, making CB1 one of the strongest bonds (ESynergeticCB1 = 7.32 kcal mol−1 and EactualCB1 = 7.33 kcal mol−1). In the SW5B cluster, the S atom is involved in the formation of two bifurcated CBs. Herein, CB1 is common to two tetrameric cycles a (consisting of SO2 and O1, O2, and O5 water molecules) and b (consisting of SO2 and O1, O3, and O4 water molecules) (Scheme S15 in ESI†). Cycle a possesses an AC (CC = −0.86 kcal mol−1), while cycle b possesses an FCC (CC = 1.73 kcal mol−1) towards CB1, leading to its synergetic energy being 5.00 kcal mol−1, which is fairly in good agreement with its value (4.69 kcal mol−1) in the actual SW5B cluster. However, the CB2 is also common to AC cycle a (CC = −0.71 kcal mol−1) and AC cycle c (consisting of SO2 and O4, O5, and O6 water molecules) with a small negative CC (−0.86 kcal mol−1). Because of these two AC contributions, CB2 is the weakest (ESynergeticCB1 = 0.46 kcal mol−1 and EactualCB1 = 0.15 kcal mol−1) bond among all the CBs in the various penta-hydrated clusters studied here. In contrast, the CB1 bond in the SW5C cluster nicely resides at the interface of two FCC tetrameric cycles: a (consisting of SO2 and O1, O4, and O6 water molecules) and b (consisting of SO2 and O3, O5, and O7 water molecules). The cooperativity contributions of the two cycles are very large (4.97 and 4.80 kcal mol−1, respectively), making CB1 the strongest bond (ESynergeticCB1 = 11.54 kcal mol−1 and EactualCB1 = 12.13 kcal mol−1) among all the CBs in the various penta-hydrated clusters studied here. The structure of the SW5D cluster is very similar to that of the SW4C cluster. The strengths of CB1 and CB2 may be similarly explained.
To further understand the interplay of cyclic cooperativity on the strength of CB, the specially optimized clusters, viz.SW5E, SW5F, and SW5G, were considered, wherein CB is common to two tetrameric cycles a and b, with different combinations of the nature of cyclic cooperativity. In SW5E clusters, these cycles exhibit FCC with CCs being 4.26 and 4.27 kcal mol−1, making CB1 the strongest bond (ESynergeticCB1 = 9.06 kcal mol−1 and EactualCB1 = 9.43 kcal mol−1) in this class of structures. In SW5F, one of these cycles (cycle a) is replaced with the one having PCC (CC = 1.24 kcal mol−1), and the strength of CB1 is moderately decreased (ESynergeticCB1 = 6.77 kcal mol−1 and EactualCB1 = 6.81 kcal mol−1). However, CB1 strength further weakens (ESynergeticCB1 = 5.73 kcal mol−1 and EactualCB1 = 5.74 kcal mol−1) in the SW5G cluster when both cycles exhibit PCC (CCs = 1.46 and 1.49 kcal mol−1). A combination of two cyclic structures in which cycle a bears FCC (CC = 3.23 kcal mol−1) and cycle b exhibits AC (−1.26 kcal mol−1) leads to further weakening of the CB1 (ESynergeticCB1 = 5.05 kcal mol−1 and EactualCB1 = 4.82 kcal mol−1) as in SW5H. When both cycles a and b exhibit AC (CC = −0.68 and −0.62 kcal mol−1, respectively), the energy of CB1 is the smallest in this class (ESynergeticCB1 = 2.27 kcal mol−1 and EactualCB1 = 2.38 kcal mol−1) as in SW5I. Note that NW5G, NW5H, and NW5I are partially optimized structures (with certain frozen atoms). Upon full optimization, these structures are not local minima, leading to other structures.
In summary, the proposed method of synergetic cyclic cooperativity can accurately determine the energies of TB, PB, and CBs in various microhydrated clusters of CO2, N2O and SO2, respectively. Fig. 5 shows a correlation plot between the ESynergeticTB/PB/CBversus EactualTB/PB/CB values for all the TBs, PBs and CBs present in various microhydrated clusters of CO2, N2O and SO2 studied in the present work (60 data points). It reveals an excellent linear correlation (R2 = 0.9963) between these two variables, with the root-mean-square and standard deviation of 0.186 and 0.189 kcal mol−1, respectively. The mean absolute error and maximum absolute errors are 0.11 and 0.59 kcal mol−1, respectively.
 | ||
| Fig. 5 Correlation plot between the energy of TB/PB/CB calculated by synergetic cooperativity effects and its value in the actual microhydrated clusters calculated using the MTA-based method. | ||
Concluding remarks
In this work, the energy of individual non-covalent bonds (NCBs), viz. tetrel (TB), pnictogen (PB) and chalcogen (CBs), which are at the interface of two or more cyclic structures, is estimated using the cooperativity contributions (CCs) of these cycles. The CCs of individual cycles are estimated using the molecular tailoring approach-based (MTA-based) method. The total CCs of all the cyclic structures common to a referenced NCB are added to the energy of this NCB present in a corresponding dimer. Thus, the calculated energy is referred to as the synergetic energy (ESynergeticNCB) of NCB. To demonstrate this approach, the microhydrated clusters (up to 5 water molecules) of atmospherically important gases, viz. CO2, N2O and SO2 were taken. In the optimized geometries of these clusters, the water molecule forms TB, PB and CB with C of CO2, N of N2O, and S of SO2, respectively. The energy of such TB, PB and CB common to two or more cyclic structures formed owing to the interconnection of NCBs is evaluated using the proposed method of the synergetic cyclic cooperativity. It has been found that the proposed method also provides very accurate energies of all these NCBs in their respective microhydrated clusters. For instance, the calculated ESynergeticTB values in microhydrated CO2 clusters fall between −0.21 and 4.98 kcal mol−1, while that for ESynergeticPB values in microhydrated N2O clusters falls between 0.72 and 3.38 kcal mol−1. The calculated values of ESynergeticCB suggests that the CB bonds in microhydrated clysters of SO2 could be of very weak to very strong strength (−0.16 to 11.54 kcal mol−1). The qualitative and quantitative strengths of these weak, moderate and strong NCBs are nicely explained in terms of the nature of the cyclic structures to which they are common. For instance, a referenced NCB is common to two or more cyclic structures, both exhibiting full-cyclic cooperativity (FCC), and is found to be the strongest. An NCB is of moderate strength if it is present at the interface of two cyclic structures, of which one exhibits FCC and the other shows partial cyclic cooperativity (PCC). However, NCBs are the weakest if both cyclic structures common to them bear anti-cooperativity (AC).Most importantly, the calculated energies of these different NCBs (TBs, PBs and CBs) using the method of synergetic cyclic cooperativity are in excellent agreement with the one calculated using the MTA-based method employing the actual microhydrated cluster of these gases (EactualTB/PB/CB). For instance, there exists a nice correlation (R2 = 0.9963) between the ESynergeticTB/PB/CBversus EactualTB/PB/CB values for all the NCBs present in various microhydrated clusters of CO2, N2O and SO2. Furthermore, the root-mean-square and standard deviations are 0.186 and 0.189 kcal mol−1, respectively. The mean absolute error and maximum absolute error is 0.11 and 0.59 kcal mol−1, respectively.
In conclusion, the proposed method of the synergetic cyclic cooperativity can accurately estimate the energy of different NCBs, such as HBs, TBs, PBs and CBs, in various microhydrated clusters of atmospherically important gases, thereby proving the general nature of this methodology. In the near future, the applicability of this methodology will be tested on various other NCBs, such as halogen and triel bonds. Currently, the automation of this procedure is underway. Such an automated code in conjunction with our hydrogen bond energy estimation (H-BEE) code49 would be really useful for rapid evaluation of the energy of NCBs in any large 3D molecular cluster.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the ESI.†Acknowledgements
The authors dedicate this article to Professor Dr Narayanasami Sathyamurthy on the occasion of his 75th birthday. It is part of the special issue of Physical Chemistry Chemical Physics in his honor. His pioneering contributions to theoretical and computational chemistry continue to inspire generations of researchers in the field. AS thanks the Department of Science and Technology (DST) for the INSPIRE fellowship (No. DST/INSPIRE Fellowship/IF210132). BD is thankful to Dr Harisingh Gour Vishwavidyalaya (a Central University), Sagar, 470003, India, for the UGC Non-NET fellowship. MMD acknowledges the high performance computational facility at the Department of Chemistry, University of Delhi, Delhi.References
- G. Cavallo, P. Metrangolo, R. Milani, T. Pilati, A. Priimagi, G. Resnati and G. Terraneo, Chem. Rev., 2016, 116, 2478–2601, DOI:10.1021/acs.chemrev.5b00484.
- A. C. Legon, Phys. Chem. Chem. Phys., 2017, 19, 14884–14896, 10.1039/C7CP02518A.
- S. Scheiner, J. Phys. Chem. A, 2021, 125, 10419–10427, DOI:10.1021/acs.jpca.1c09213.
- A. Varadwaj, P. R. Varadwaj, H. M. Marques and K. Yamashita, Inorganics, 2022, 10, 149, DOI:10.3390/inorganics10100149.
- L. Vogel, P. Wonner and S. M. Huber, Angew. Chem., Int. Ed., 2019, 58, 1880–1891, DOI:10.1002/anie.201809432.
- S. Scheiner, Phys. Chem. Chem. Phys., 2021, 23, 5702–5717, 10.1039/D1CP00242B.
- P. R. Varadwaj, A. Varadwaj, H. M. Marques and K. Yamashita, CrystEngComm, 2023, 25, 1411–1423, 10.1039/D2CE01621D.
- C. H. Suresh, G. S. Remya and P. K. Anjalikrishna, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2022, 12, e1601, DOI:10.1002/wcms.1601.
- S. Chandra, B. Suryaprasad, N. Ramanathan and K. Sundararajan, Phys. Chem. Chem. Phys., 2021, 23, 6286–6297, 10.1039/D0CP06273A.
- A. Bauzá, R. Ramis and A. Frontera, J. Phys. Chem. A, 2014, 118, 2827–2834, DOI:10.1021/jp502301n.
- S. Scheiner, CrystEngComm, 2025, 27, 921–930, 10.1039/D4CE01194E.
- P. Politzer, J. S. Murray and T. Clark, Phys. Chem. Chem. Phys., 2021, 23, 16458–16468, 10.1039/D1CP02602J.
- X. Wang, Q. Li and S. Scheiner, Phys. Chem. Chem. Phys., 2023, 25, 29738–29746, 10.1039/d3cp04430k.
- R. Gupta, S. Singha and D. Mani, J. Phys. Chem. A, 2024, 128, 4605–4622, DOI:10.1021/acs.jpca.4c00911.
- N. Liu, Q. Wu, Q. Li and S. Scheiner, Phys. Chem. Chem. Phys., 2022, 24, 1113–1119, 10.1039/D1CP05323J.
- Q. Wu, X. Xie, Q. Li and S. Scheiner, Phys. Chem. Chem. Phys., 2022, 24, 25895–25903, 10.1039/D2CP04194D.
- M. B. Ahirwar, S. R. Gadre and M. M. Deshmukh, J. Phys. Chem. A, 2023, 127, 4394–4406, DOI:10.1021/acs.jpca.3c00359.
- D. Patkar, M. B. Ahirwar, S. R. Gadre and M. M. Deshmukh, J. Phys. Chem. A, 2021, 125, 8836–8845, DOI:10.1021/acs.jpca.1c06478.
- M. B. Ahirwar, D. Patkar, I. Yadav and M. M. Deshmukh, Phys. Chem. Chem. Phys., 2021, 23, 17224–17231, 10.1039/D1CP02839A.
- M. M. Deshmukh, L. J. Bartolotti and S. R. Gadre, J. Phys. Chem. A, 2008, 112, 312–321, DOI:10.1021/jp076316b.
- M. B. Ahirwar, S. R. Gadre and M. M. Deshmukh, J. Phys. Chem. A, 2024, 128, 6099–6115, DOI:10.1021/acs.jpca.4c01176.
- K. Stokely, M. G. Mazza, H. E. Stanley and G. Franzese, Proc. Natl. Acad. Sci. U. S. A., 2010, 107, 1301–1306, DOI:10.1073/pnas.0912756107.
- S. Blanco, P. Pinacho and J. C. López, Angew. Chem., Int. Ed., 2016, 128, 9477–9481, DOI:10.1002/ange.201603319.
- A. Sauza de la Vega, T. Rocha-Rinza and J. M. Guevara-Vela, ChemPhysChem, 2021, 22, 1269–1285, DOI:10.1002/cphc.202000981.
- K. J. Tielrooij, N. Garcia-Araez, M. Bonn and H. J. Bakker, Science, 2010, 328, 1006–1009, DOI:10.1126/science.1183512.
- R. P. Sheridan, R. H. Lee, N. Peters and L. C. Allen, Biopolymers, 1979, 18, 2451–2458, DOI:10.1002/bip.1979.360181006.
- J. Li, Y. Wang, J. Chen, Z. Liu, A. Bax and L. Yao, J. Am. Chem. Soc., 2016, 138, 1824–1827, DOI:10.1021/jacs.5b13140.
- J. M. Guevara-Vela, E. Romero-Montalvo, V. A. M. Gòmez, R. Chávez-Calvillo, M. García-Revilla, E. Francisco, Á. Martín Pendás and T. Rocha-Rinza, Phys. Chem. Chem. Phys., 2016, 18, 19557–19566, 10.1039/C6CP00763E.
- F. Weinhold, Molecules, 2022, 27, 4218, DOI:10.3390/molecules27134218.
- C. Pérez, D. P. Zaleski, N. A. Seifert, B. Temelso, G. C. Shields, Z. Kisiel and B. H. Pate, Angew. Chem., Int. Ed., 2014, 53, 14368–14372, DOI:10.1002/anie.201407447.
- O. Mo, M. Yáñez and J. Elguero, J. Chem. Phys., 1992, 97, 6628–6638, DOI:10.1063/1.463666.
- N. Kobko and J. J. Dannenberg, J. Phys. Chem. A, 2003, 107, 10389–10395, DOI:10.1021/jp0365209.
- A. S. Mahadevi, Y. I. Neela and G. N. Sastry, Phys. Chem. Chem. Phys., 2011, 13, 15211–15220, 10.1039/C1CP21346F.
- L. Rincon, R. Almeida and D. G. Aldea, Int. J. Quantum Chem., 2005, 102, 443–453, DOI:10.1002/qua.20401.
- R. F. W. Bader, Atoms in Molecules: A Quantum Theory, Clarendon Press, Oxford, 1990 Search PubMed.
- E. Espinosa, E. Molins and C. Lecomte, Chem. Phys. Lett., 1998, 285, 170–173, DOI:10.1016/S0009-2614(98)00036-0.
- J. M. Guevara-Vela, F. Francisco, T. Rocha-Rinza and A. M. Pendás, Molecules, 2020, 25, 4028, DOI:10.3390/molecules25174028.
- M. M. Deshmukh, S. R. Gadre and L. J. Bartolotti, J. Phys. Chem. A, 2006, 110, 12519–12523, DOI:10.1021/jp065836o.
- M. M. Deshmukh and C. H. Suresh, and S. R, J. Phys. Chem. A, 2007, 111, 6472–6480, DOI:10.1021/jp071337r.
- M. M. Deshmukh and S. R. Gadre, J. Phys. Chem. A, 2009, 113, 7927–7932, DOI:10.1021/jp9031207.
- M. M. Deshmukh, S. R. Gadre and E. M. Cocinero, New J. Chem., 2015, 39, 9006–9018, 10.1039/C5NJ02106E.
- N. Siddiqui, V. Singh, M. M. Deshmukh and R. Gurunath, Phys. Chem. Chem. Phys., 2015, 17, 18514–18523, 10.1039/C5CP02690C.
- V. Singh, I. Ibnusaud, S. R. Gadre and M. M. Deshmukh, New J. Chem., 2020, 44, 5841–5849, 10.1039/D0NJ00304B.
- M. M. Deshmukh and S. R. Gadre, Molecules, 2021, 26, 2928, DOI:10.3390/molecules26102928.
- M. B. Ahirwar, S. R. Gadre and M. M. Deshmukh, J. Phys. Chem. A, 2020, 124, 6699–6706, DOI:10.1021/acs.jpca.0c05631.
- M. B. Ahirwar, N. D. Gurav, S. R. Gadre and M. M. Deshmukh, Phys. Chem. Chem. Phys., 2022, 24, 15462–15473, 10.1039/D2CP01663J.
- M. B. Ahirwar and M. M. Deshmukh, J. Phys. Chem. A, 2023, 127, 1219–1232, DOI:10.1021/acs.jpca.2c08087.
- M. B. Ahirwar and M. M. Deshmukh, J. Comput. Chem., 2023, 44, 1861–1874, DOI:10.1002/jcc.27133.
- M. B. Ahirwar, S. S. Khire, S. R. Gadre and M. M. Deshmukh, J. Comput. Chem., 2024, 45, 274–283, DOI:10.1002/jcc.27237.
- B. Dehariya, M. B. Ahirwar, A. Shivhare and M. M. Deshmukh, J. Comput. Chem., 2025, 46, e70008, DOI:10.1002/jcc.70008.
- A. Shivhare, B. Dehariya, S. R. Gadre and M. M. Deshmukh, Phys. Chem. Chem. Phys., 2024, 26, 21332–21336, 10.1039/D4CP02580F.
- A. Shivhare, B. Dehariya, S. R. Gadre and M. M. Deshmukh, Phys. Chem. Chem. Phys., 2025, 27, 3661–3672, 10.1039/D4CP04741A.
- Z. Rezaei, M. Solimannejad and M. D. Esrafili, Comput. Theor. Chem., 2015, 1074, 101–106, DOI:10.1016/j.comptc.2015.10.015.
- H. Xu, J. Cheng, X. Yang, Z. Liu, X. Boa and Q. Li, RSC Adv., 2017, 7, 21713–21720, 10.1039/C7RA02068F.
- S. A. C. McDowell, Phys. Chem. Chem. Phys., 2018, 20, 18420–18428, 10.1039/c8cp03641a.
- T. M. Ismail, D. Patkar, P. K. Sajith and M. M. Deshmukh, J. Phys. Chem. A, 2023, 127, 10360–10374, DOI:10.1021/acs.jpca.3c04181.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson and H. Nakatsuji, et al., Gaussian 16, Revision A.03, Gaussian, Inc., Wallingford CT, 2016 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Molecular energies (in a.u.) of various species of TB1 energy evaluation in cyclic trimers a and b of CW3B cluster using the MTA-based method in Tables S1 and S2, respectively; molecular energies (in a.u.) of various species of TB1 energy evaluation in the actual CW3B cluster by the MTA-based method in Table S3; representation of a PB and CB in cyclic trimers in Schemes S1 and S2; The MP2/aug-cc-pVDZ geometry of CW3B clusters exhibiting a TB common to two cyclic structures (a) and (b), both exhibiting full cyclic cooperativity in Scheme S3. The fragmentation procedure of the MTA-based method for calculating the energy of TB1 in the cyclic trimer b of CW3B cluster in Scheme S4; the fragmentation procedure of the MTA-based method for calculating the energy of TB1 in CW3B cluster in Scheme S5; the structure of the CO2(H2O)n clusters showing different types of cyclic structures exhibiting FCC, PCC or AC towards TBs common to these cycles in Scheme S6; the fragmentation procedure of MTA-based method for calculating the energy of PB1 in cyclic trimers a and b of NW3C clusters in Schemes S7 and S8; the fragmentation procedure of MTA-based method for calculating the energy of PB1 in the NW3C cluster in Scheme S9. Optimized geometries of lowest energy conformers of N2O(H2O)3 in Scheme S10; the structure of the N2O(H2O)n clusters showing different types of cyclic structures exhibiting FCC, PCC or AC towards PBs in Scheme S11; the fragmentation procedure of MTA-based method for calculating the energy of CB1 in the cyclic trimers a and b of SW3A cluster in Schemes S12 and S13; the fragmentation procedure of MTA-based method for calculating the energy of CB1 in the SW3A cluster in Scheme S14; and the structure of the SO2(H2O)n clusters showing different types of cyclic structures exhibiting FCC, PCC or AC towards CBs in Scheme S15. See DOI: https://doi.org/10.1039/d5cp02070k |
| This journal is © the Owner Societies 2025 |





