Influence of hydration, hydroxylation, and doping of TiO2(110) on the adsorption and decomposition of sarin: a density functional theory investigation†
Yenny
Cardona Quintero‡
and
Ramanathan
Nagarajan
 *
*
US Army Combat Capabilities Development Command Soldier Center, 15 General Greene Avenue, Natick, MA 01760, USA. E-mail: yenny.p.cardonaquintero.civ@us.navy.mil; Ramanathan.nagarajan.civ@army.mil
First published on 29th July 2025
Abstract
Metal oxides are promising candidates for the adsorption and decomposition of chemical warfare agents (CWA) and can be the foundations of novel CWA destruction technologies. In this work, we use density functional theory (DFT) to explore how dry, wet and doped states of the metal oxide TiO2(110), influence the adsorption and chemical dissociation of the nerve agent, sarin. The DFT calculations show that the dissociative adsorption of sarin is more energetically favored than the molecular adsorption for all dry, wet and doped states of TiO2(110). The calculated energy barrier for the adsorption of sarin on dry TiO2(110) showed that sarin is initially adsorbed with a molecular configuration, followed by the dissociative process. For the adsorption of sarin on wet TiO2, under both hydrated and hydroxylated states, the 0.5 H2O monolayer (ML) showed the lowest adsorption energy compared to the dry and other wetness levels explored. Finally, the adsorption of sarin on TiO2(110) doped with Hf exhibited lower adsorption energy and higher charge transfer compared with TiO2(110) doped with Zr and Ge, as well as the undoped systems. These results demonstrate how hydration, hydroxylation, and doping of TiO2(110) significantly influence the adsorption and decomposition of sarin on this metal oxide.
1. Introduction
Nerve agents are highly toxic chemicals that continue to pose a threat to military and civilian personnel.1 The nerve agents are organophosphate (OP) compounds, and their lethality comes from their ability to bind to the acetylcholinesterase (AChE) enzyme, preventing the enzymatic breakdown and regulation of the neurotransmitter acetylcholine (ACh). The resulting accumulation of acetylcholine causes continuous nerve impulses and muscle contractions, and this neurotoxicity can be acute and fatal.1,2 Sarin (isopropyl methyl phosphono fluoridate, commonly designated as agent GB), is one example of a nerve agent which has been used in terrorist attacks and armed conflicts including the Syrian war, resulting in many fatalities and acute health effects for vast populations. Apart from such weaponization, the residual, undeclared, stockpiles of chemical weapons held by nations, that have not been destroyed despite it being 28 years since the Chemical Weapons Convention,3 pose an increasing environmental threat to land, water and civilian populations. Therefore, to protect soldier and civilian populations and to decontaminate equipment and infrastructure in the event of a chemical attack and to ensure remediation of environments affected by residual stockpiles of chemical agents, there is an imperative need to develop novel materials that can adsorb and dissociate nerve agents such as sarin into harmless chemicals.Among the materials considered for the adsorption and dissociation of nerve agents are various metal oxides, which exhibit high stability, large numbers of reactive sites and good selectivity for applications in sensing and decontamination of nerve agents.4–8 Titanium dioxide has proven to be the leading material for photocatalyst applications.9–13 Furthermore, metal doping of TiO2 has been shown to be an effective way to tune electronic and optical properties including decreasing the band gap and extending the absorption into the visible region, which is expected to improve the photocatalytic activity for degradation of chemical warfare agents.14–17 Cation doping of TiO2 is also considered a strategy for delaying the deactivation of TiO2.
Experimental investigations of adsorption and dissociation of nerve agents on metal oxides are necessarily limited due to their high toxicity. To circumvent handling these toxic chemicals, one approach is to conduct experiments using simulants, which are molecules with structures and molecular features similar to the nerve agents but with lower toxicity.18 Many experimental studies have investigated the adsorption and dissociation of simulants for nerve agents, dimethyl methyl phosphonate (DMMP) being one of the most frequently selected simulants.19–25 The other approach is to conduct theoretical studies on nerve agents, since this obviously does not involve any toxicity risk and allows one to explore the fundamentals of adsorption and decomposition of nerve agents and other toxic chemicals on different decontaminating material surfaces.
Among theoretical methods, density functional theory (DFT) is probably the most important and widely used tool that allows the exploration of how agent molecules interact with different types of potential decontaminating surfaces. DFT-based first principles methods are essential for exploring novel materials, enabling the theoretical calculation of materials properties and the simulation of interaction mechanisms. DFT computations provide fundamental insights complementing experimental findings by offering physical understanding and guiding experimental design, exploration and screening efforts. Some recent examples of diverse applications for DFT computations include calculations of electronic properties of perylene diimide derivatives to determine the response of electromechanical sensors,26 the binding energy of amino acids on transition metal dichalcogenides for chemical sensor applications,27 or the NO adsorption energy on metal chelates for controlling environmental NOx emissions.28 Using DFT, one can determine the structure, stability and electronic properties of nerve agents29–38 and simulants39–51 on metal oxide surfaces, at atomic and molecular scales.
While the experimental and theoretical studies in the literature cited above have provided a wealth of information, there are still important unanswered questions as to how surface impurities such as dopants and factors such as the environmental conditions impact the adsorption and dissociation process of nerve agents on metal oxide surfaces. We address this knowledge gap in this work, selecting the metal oxide TiO2(110) rutile, which is the most thermodynamically stable phase of TiO2.52,53 Using DFT methods, we can provide important insights into the ability of TiO2 to adsorb and decompose the chemical warfare agent sarin. More importantly we can study the local structure modification through doping titania with different elements and the influence of the environmental conditions including H2O adsorption on the surface and determine their potential impact on the catalytic activity of TiO2 for the adsorption and decomposition of sarin.
Dopants can influence the surface properties of TiO2 because the number of their valency electrons may be different from that of Ti4+, the steric effect related to the different size of the dopant compared to Ti4+ or the orbital effect due to the energy levels associated to the dopant not being present on the pristine TiO2 surface. We selected isovalent dopants (eliminating influence of different numbers of valency electrons) that are of a size similar to Ti, so that the geometry perturbations on titania will be minimal when substitutional doping of a Ti atom with the dopant atom is done. The isovalent dopant elements Hf, Zr and Ge can adopt the same oxidation state to the one of Ti on the TiO2 structure (4+) and possess a similar atomic radius54 (155 pm for Hf and Zr and 125 pm for Ge) compared to Ti (140 pm). They have also been used in experimental studies.16,17
We investigated the molecular and dissociative adsorption of sarin on dry, wet and doped TiO2(110), its stability and structure. The molecular and dissociative adsorption energy of sarin on TiO2 was calculated using density functional theory, while the nudge elastic band method was used to calculate the energy barrier for the dissociation of sarin on dry TiO2. The charge transfer was calculated using the Bader charge analysis. We determined the effect of water presence and concentration on the TiO2 surface for the adsorption and dissociation of sarin. By doping TiO2 with different elements, we were able to determine the variation in the adsorption energy and charge transfer due to the presence of these dopants on the TiO2 surface for the adsorption and dissociation of sarin. We found that the hydration, hydroxylation and doping of the TiO2(110) surface cause significant variations in the adsorption and decomposition behavior of sarin on this metal oxide.
2. Computational methods
Density functional theory calculations were developed using the Perdew–Burke–Ernzerhof generalized gradient approximation (PBE-GGA)55 and the projected-augmented wave (PAW)56 approach as implemented in the Quantum Espresso software.57 van der Waals interactions were also considered through the DFT-D2 method of Grimme.58 The cutoff kinetic energy for the charge density and wave functions were 350 and 30 Ry, respectively (1 Ry = 1312.75 kJ mol−1), as suggested in the pseudopotentials for the different atom types used in this research.The charge transfer for the sarin doped TiO2 system was calculated using the Bader charge analysis.59 In Bader's approach, space is divided into regions by surfaces that run through minima in the charge density. Specifically, at a point on a dividing surface the gradient of the electron density has no component normal to the surface. The regions bounded by such dividing surfaces are referred to as Bader regions. Each Bader region often contains one nucleus, but this is not necessarily so, sometimes no nucleus is found within a Bader region. By integrating the electronic density within the Bader region where an atom's nucleus is located and possibly adding the electronic charge in nearby regions that do not include a nucleus, the total charge on an atom can be estimated. The computations are carried out using the algorithm developed by Henkelman.60,61 Using this analysis, the charge for the Sarin molecule, the doped TiO2 surface, and the doped interface were calculated, with the subsequent charge transfer determined.
2.1. Sarin isolated molecule and TiO2 slab
To determine the most stable structure of the isolated sarin (C4H10FO2P) molecule, three initial conformations were selected based on the geometries reported in previous investigations as the most stable structures of the molecule62–66 and verifying the geometric parameters for the different conformations. We used a 20 × 20 × 20 Å3 supercell, relaxing the structures until the parameters reached the convergence criteria to obtain the most stable configuration, while the energy was calculated simultaneously.The TiO2 rutile bulk system was modeled as a tetragonal crystal lattice with a mesh of 8 × 8 × 10 k-points. The final a and c lattice parameters for the bulk rutile were obtained as 4.6408 Å and 2.9683 Å, respectively, in agreement with previous theoretical and experimental studies.39,64 The TiO2(110) rutile slab structure was built with a surface area of 2 × 4 (13.126 Å × 11.873 Å) with 5 TiO2 perpendicular layers (corresponding to 80 TiO2 groups or 240 total atoms) and a 15 Å vacuum region and is hereafter referred to as a 2 × 4 × 5 supercell. The TiO2(110) rutile structure has been identified as the most thermodynamically stable among the TiO2 phases with a large number of reactive sites.67 The dimensions of the 2 × 4 × 5 supercell structure of TiO2 selected in this study allow us to model the adsorption of the sarin molecule. Such large slab systems prevent modification of the molecular geometry associated with large intermolecular interaction effects and provide better accuracy compared to cluster systems.
2.2. Sarin adsorbed on dry TiO2(110)
The adsorption of sarin on the TiO2(110) surface was modeled with a 0.125 monolayer (ML) coverage or 1 molecule of sarin per 8 available Ti atoms. The sarin molecule was placed on top of the TiO2(110) surface forming an asymmetrical configuration for which a dipole moment correction was required to account for the artificial field in the system. A vacuum region of 15 Å was selected after testing different vacuum lengths and corroborating the stability in the geometry and energy of the sarin–TiO2 system. The initial adsorption geometries of the sarin molecule on the TiO2(110) surface were built based on information from previous theoretical and experimental studies that reported the possible structure and geometry of the sarin–TiO2 system.13,29,62,64,68 For dissociative adsorption of sarin, molecular groups generated from P–F and P–O bond breakages were considered. In addition, parameters such as bond angles, bond lengths, distance of the molecule to the surface, and the possible formation of monodentate configurations, bidentate configurations or secondary bonds were taken into consideration to model the initial structure (see the ESI† Section A, Fig. S1–S3 for examples of the initial sarin conformations considered).2.3. Sarin adsorbed on wet TiO2(110)
The adsorption of sarin on wet TiO2(110) was explored for hydrated and hydroxylated systems with different water coverages. In the hydrated system, the O of the H2O molecule bonds with a Ti atom on the surface, while in the hydroxylated system the oxygen atom of an OH− group bonds with a Ti atom and the remaining H+ interacts with an O on the TiO2 surface. The geometries of the TiO2 hydrated and hydroxylated surfaces were selected to have three different water coverages 0.25, 0.5 and 0.75 H2O ML, corresponding to 2, 4 and 6 water molecules per 8 available Ti atoms on the top TiO2 layer. The adsorption of the sarin molecule on the hydrated and hydroxylated surfaces with different water coverage was modeled and then relaxed to obtain the most stable conformation. The initial geometries for the adsorption of sarin on the hydrated and hydroxylated TiO2 were considered based on the results reported elsewhere62 for the adsorption of sarin on 0.5 H2O ML coverage for the hydrated and hydroxylated TiO2 (see the ESI† Section A, Fig. S4 for examples of the initial sarin conformations considered).2.4. Sarin adsorbed on doped TiO2(110)
The adsorption of sarin on TiO2 doped with Hf, Zr and Ge as a single point dopant was evaluated. One Ti atom from TiO2 was substituted by a single dopant atom of the elements mentioned. In the atomic position substituted in the first layer, the Ti atom is bonding with the sarin molecule. The single dopant atom was substituted in each one of the five TiO2 layers at the Ti atom with the same or closer to the xy position to the Ti atom of the first layer. This procedure was repeated for each one of the three dopant elements, Hf, Zr and Ge. Additional individual dopant positions in the first TiO2 layer were also considered with the Hf element substituting the Ti atoms closer to the sarin molecule. The initial conformations for molecular and dissociated sarin were selected as those for the dopant-free system and then subjected to optimization.3. Results and discussion
3.1. Isolated sarin molecule and TiO2(110) slab
Three different geometries of the isolated sarin molecule were studied in this work as initial conformations. These geometries were selected based on previous studies which described them as the stable configuration of sarin,62–66 with some discrepancies about bond angles, bond lengths and in general atomic arrangement of the most stable configuration in the molecule. For the geometries of sarin molecule with conformation 1,62–64 conformation 2,65 and conformation 3,66 the final energies were calculated. The final structures and geometrical parameters for the different conformations of sarin are shown in Fig. 1. The lowest total energy for the sarin molecule was obtained for the conformation 1. Conformation 2 and conformation 3 had a higher total energy with respect to conformation 1, of 0.0709 and 0.0388 eV, respectively. Based on the analysis of the total energy, conformation 1 of the sarin molecule has the highest stability.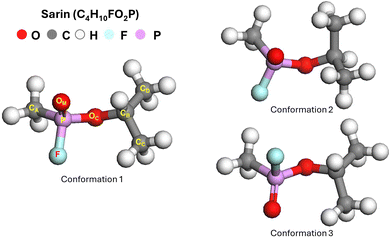 | ||
| Fig. 1 Final geometry and structures of isolated sarin molecules for three starting conformations. Conformation 1 is determined to be the most stable structure. | ||
In the most preferable conformation of sarin, the P atom is a chiral center, while the F atom and the CCH3 group are close to a perpendicular direction with respect to the P–OC–CB plane. The F–P–OC and CC–CB–OC bond angles were obtained as 103° and 109°, respectively, and the P–F, P–OC and P–OM bond lengths were 1.59, 1.47 and 1.59 Å, respectively. The final geometric parameters of the most stable conformation 1 of isolated sarin molecules, including some bond lengths and bond angles are reported in Table 1.
| Isolated sarin molecule | TiO2(110) 2 × 4 × 5 supercell | ||
|---|---|---|---|
| Bond lengths (Å) | Bond lengths (Å) | ||
| P–F | 1.59 | Ti5fc–O3fc | 1.96 |
| P–OM | 1.47 | Ti6fc–O2fc | 1.83 |
| P–CA | 1.80 | Ti6fc–O3fc-1 | 2.11 |
| P–OC | 1.59 | ||
| OC–CB | 1.48 | ||
| CB–CC, CB–CD | 1.52 | ||
| Bond angles (°) | Bond angles (°) | ||
|---|---|---|---|
| F–P–OM | 112 | Ti6fc–O2fc–Ti6fc | 108 |
| F–P–OC | 103 | Ti6fc–O3fc-1–Ti6fc | 89 |
| OM–P–CA | 118 | Ti5fc–O3fc–Ti5fc | 98 |
| CA–P–OC | 103 | ||
| P–OC–CB | 121 | ||
| CC–CB–OC | 109 | ||
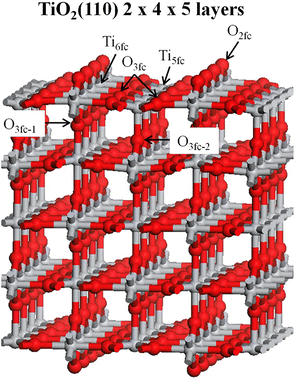
For the TiO2(110) 2 × 4 × 5 supercell, the two bottom layers of the metal oxide supercell were fixed at the TiO2 bulk position while the three top layers were allowed to relax. The geometrical parameters of the TiO2(110) 2 × 4 × 5 supercell are outlined in Table 1 and the corresponding labels are shown in Fig. 2.
In the TiO2(110) surface, the top and bottom layers have two Ti atom types, the Ti five-fold coordinated (Ti5fc) and the Ti six-fold coordinated (Ti6fc), as well as two O atom types, the O two-fold coordinated (O2fc) and the O three-fold coordinated (O3fc). In Ti5fc and Ti6fc the Ti atom bonds with 5 and 6 O atoms, respectively, while in O2fc and O3fc, the O atom bonds with 2 and 3 Ti atoms, respectively.
3.2. Adsorption and dissociation of sarin on dry TiO2
The molecular and dissociative adsorption of sarin on the TiO2(110) 2 × 4 × 5 supercell structure was initially investigated for the clean surface (without impurities or water adsorption). The initial structures were selected based on the information reported in a similar work62 for the adsorption of sarin on TiO2. The molecular and dissociative adsorptions of sarin on TiO2(110) were studied with different initial possible configurations of sarin on rutile TiO2(110) in which the sarin molecule interacts with the TiO2 surface by the formation of a monodentate configuration with Ti–O or F–Ti bonds or a bidentate configuration with the Ti–O and F–Ti bonds. We determined the preferable configuration of the sarin-TiO2 system through calculation of the adsorption energy for the different conformations explored. The adsorption energy (Eads) for the sarin molecule adsorbed on TiO2(110) is defined as:| Eads = ESarin-TiO2(110) − ETiO2(110) − ESarin | (1) |
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group from sarin and the Ti5fc atom from the TiO2(110) surface with the formation of several secondary bonds between sarin and the O2fc surface atom of TiO2(110). The Eads and structure of the most stable configuration for the molecular adsorption of sarin on TiO2(110) is shown on Fig. 3.
O group from sarin and the Ti5fc atom from the TiO2(110) surface with the formation of several secondary bonds between sarin and the O2fc surface atom of TiO2(110). The Eads and structure of the most stable configuration for the molecular adsorption of sarin on TiO2(110) is shown on Fig. 3.
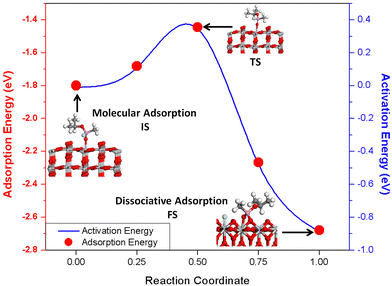
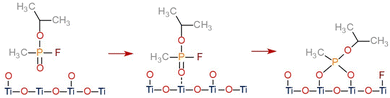
We also considered the dissociative adsorption of sarin on TiO2(110) with different dissociative products, which was also described in previous reported work.62 The Eads for the most stable dissociative adsorption of sarin on TiO2(110) was obtained as −2.70 eV. The final geometry for the dissociative adsorption of the sarin–TiO2 system was obtained as the dissociation of the F atom from the surface and its adsorption on the TiO2 surface by the formation of a F–Ti bond. The remaining C4H10O2P+ molecule interacts with an O2fc atom on the surface forming a bidentate configuration. This final geometry, as well as the Eads for the dissociative adsorption of sarin on TiO2 are also shown on Fig. 3. The comparison between the Eads values obtained for the most stable configurations of the molecular and dissociative adsorption of sarin on TiO2 indicates that the dissociative adsorption of sarin is preferred over molecular adsorption. These results, along with the final structures obtained, could indicate the possible mechanism being an initial molecular adsorption of sarin on TiO2 followed by the dissociation of the molecule, as also suggested by previous experimental studies.12,13
To clearly establish the adsorption and dissociative process of sarin on TiO2(110), the energy barrier between the molecular and dissociative adsorption of the molecule was calculated using the nudge elastic band method. The final geometry for the molecular adsorption of sarin on TiO2 is considered as the initial state (IS) while the dissociative adsorption configuration of sarin on TiO2 was identified as the final state (FS). We found that the dissociation of sarin on TiO2(110) is exothermic with an energy barrier of 0.35 eV at the transition state (TS). The energy barrier and the structure of IS, TS and FS obtained for the adsorption process of sarin on TiO2 are shown in Fig. 3. The rotation and orientation of the isopropyl group and the F atom in the sarin molecule from the initial state to the transition state, before the dissociation of F, are the main contributions to the energy barrier for sarin dissociation on TiO2(110). Given the small energy barrier obtained for the dissociation of sarin on TiO2 and its exothermic nature, we suggest the adsorption mechanism of sarin on TiO2 as an initial molecular adsorption (observed in IS) with a subsequent dissociation of the F atom and the formation of a bidentate configuration by the remaining molecule as observed in FS in Fig. 3. The adsorption followed by the dissociation of sarin on TiO2(110) is shown in Fig. 4.
Our computational results agree with previous theoretical and experimental studies on the adsorption of sarin on different surfaces. The theoretical investigations on the adsorption of sarin on the anatase TiO2(001) and Al2O3 clusters determined the Eads values to be −2.00 eV,66 and −2.23 eV,29 respectively. A combined theoretical and experimental work on the adsorption of sarin on SiO2 also reported values for Eads of −0.82 and −0.95 eV from the experiments and calculations, respectively.69 A previous experimental work68 investigating the adsorption of sarin on hydroxylated TiO2 found that dissociative adsorption of the molecule is preferable with the F atom from sarin interacting with the hydroxyl groups from the surface, facilitating the final dissociation of the fluorine atom and the subsequent formation of a bidentate configuration from the remaining sarin molecule.
3.3. Adsorption and dissociation of sarin on wet TiO2 with different H2O ML coverage
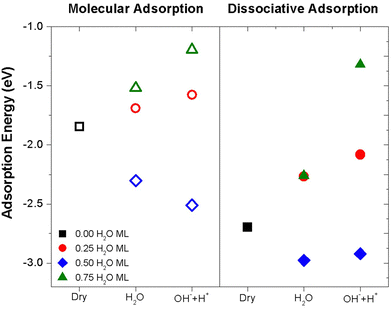
The results obtained for the adsorption of sarin on dry TiO2 indicate that theoretically, the dissociation of the molecule on a dry surface is possible. However, a clean and dry TiO2 surface is difficult to obtain in practice, under common experimental conditions. Previous experimental study68 reported the adsorption of sarin on a hydroxylated TiO2(110) as an initial molecular adsorption process followed by the dissociation of the sarin molecule on the hydroxylated TiO2(110) surface. Based on a previous investigation62 on the hydration and hydroxylation of sarin on TiO2(110) with 0.5 H2O ML coverage, we selected the most stable molecular and dissociative configurations of sarin on a TiO2 surface and considered additional levels of H2O coverage.To determine the effect of water content on the TiO2 surface for the adsorption and dissociation of sarin, we compared the Eads for the molecular and dissociative adsorption of sarin on a hydrated and hydroxylated TiO2(110) surface for three H2O coverages: 0.25 ML, 0.5ML and 0.75 ML (corresponding to 2, 4 and 6 H2O molecules of water, respectively, per 8 available Ti5fc atoms on top of the TiO2 slab). The molecular and dissociative adsorption of sarin on the 0.5 H2O ML for the hydrated and hydroxylated TiO2 had the lowest Eads values among the different H2O coverages, as shown in Fig. 5. In addition, Eads for the dissociative adsorption of sarin on both the hydrated and hydroxylated TiO2 surfaces were lower compared to Eads for the molecular adsorption, showing a similar tendency to that observed for the adsorption of sarin on dry TiO2. The comparison of Eads on dry and wet sarin–TiO2(110) systems showed that the dissociative adsorption of sarin on 0.5 H2O ML TiO2, specifically a hydrated surface is the preferable configuration, is in general agreement with the suggestion proposed in a previous experimental study.68
The final geometries for the most stable molecular and dissociative adsorption of sarin on wet (hydrated and hydroxylated) TiO2 systems are shown on Fig. 6 and these are similar to the ones observed for the dry systems. A schematic of the mechanism of adsorption of sarin on dry and wet TiO2 for hydrated (H2O) and hydroxylated (OH + H+) surfaces is provided in the ESI† Section B, Fig. S5.
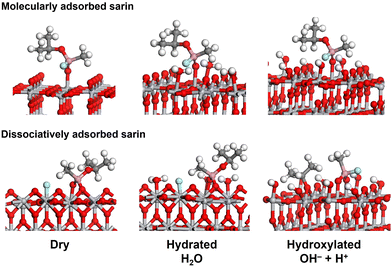 | ||
| Fig. 6 Structure and geometry of molecularly and dissociatively adsorbed sarin on dry, hydrated (H2O) and hydroxylated (OH− + H+) TiO2(110). | ||
For the molecular adsorbed structure, the formation of a P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond with the TiO2 surface was more stable, while in the dissociatively adsorbed structure, the F atom dissociates from sarin and is adsorbed on the TiO2 surface while the remaining molecule forms a bidentate configuration with a O2fc from TiO2. Additional secondary bonds formed between sarin and the water molecules were observed for the hydrated and hydroxylated systems. The formation of these secondary bonds could have a relevant impact on the stability of the molecular and dissociative adsorption of sarin on wet TiO2.
O bond with the TiO2 surface was more stable, while in the dissociatively adsorbed structure, the F atom dissociates from sarin and is adsorbed on the TiO2 surface while the remaining molecule forms a bidentate configuration with a O2fc from TiO2. Additional secondary bonds formed between sarin and the water molecules were observed for the hydrated and hydroxylated systems. The formation of these secondary bonds could have a relevant impact on the stability of the molecular and dissociative adsorption of sarin on wet TiO2.
A comparison of the number of secondary bonds formed between the different H2O ML suggests that in the case of the lower H2O coverage (0.25 ML) the number of secondary bonds between sarin and H2O or OH groups decreased compared to the 0.5 H2O ML. In contrast, in the case of higher H2O coverage (0.75 ML) the presence of additional H2O or OH groups saturate the Ti5fc available positions preventing the bond formation between Ti and the sarin molecule.
At 0.25 H2O ML, the chemisorption of sarin on TiO2 is observed with weak interatomic and intermolecular interactions occurring between sarin and H2O. At higher degrees of hydration, specifically 0.5 H2O ML, sarin still exhibits chemisorption on TiO2 with strong bonds formed between the molecule and the surface. Additionally, there are interatomic interactions occurring between H2O and the sarin molecules, which could be mostly identified as van der Waals interactions or hydrogen bonding, resulting in lower Eads. As the degree of hydration increases for 0.75 H2O ML, the H2O molecules start to compete with sarin for adsorption sites on the titania surface with sarin showing now weaker interaction and mostly physisorption on the TiO2 surface and consequently higher Eads.
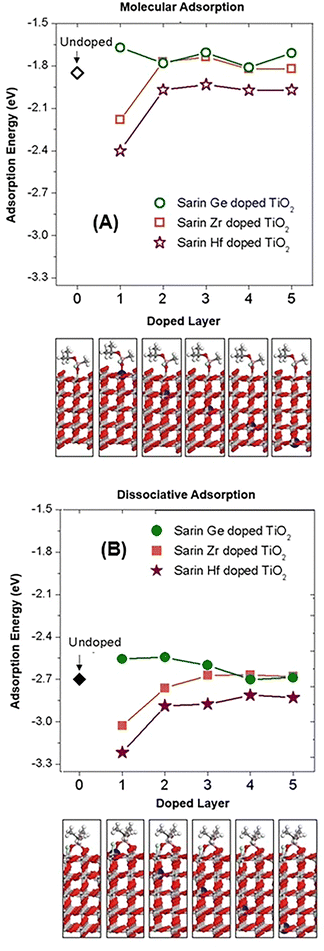
3.4. Adsorption and dissociation of sarin on substitutionally doped TiO2 with X (X = Hf, Zr or Ge)
Previous experimental studies4,70–72 have suggested that doping TiO2 with elements such as Hf, Zr and Ge can increase the degradation rate of chemical warfare agents including soman, VX and sulfur mustard. In the case of sarin, there are no available theoretical or experimental studies, to the best of our knowledge, on the adsorption of sarin on doped TiO2. To understand the influence of doping TiO2 on the adsorption and decomposition of sarin, we selected three dopant elements Hf, Zr and Ge to act as substitutional dopants. These elements can adopt the same oxidation state to the one of Ti on the TiO2 structure (4+) and possess a similar atomic radius52 (155 pm for Hf and Zr and 125 pm for Ge) compared to Ti (140 pm).Different orientations of sarin on the TiO2 surface were considered to account for the possible adsorption configurations of the molecule both in the molecular and dissociative configuration (see the ESI† Section A, Fig. S1–S3 for examples of initial sarin conformations considered, also for the doped TiO2 system). The specific configurations were selected based on different geometrical parameters for the interface including bond angle, bond length, possible secondary bonds formed, as well as distance and orientation of the molecule respect to the surface.62 The selection of the most stable adsorption configuration was determined based on the adsorption energy (Eads) values calculated for each system.
The Eads values for the molecular and dissociative adsorption of sarin on doped TiO2 with the dopants Hf, Zr and Ge were calculated with a similar relationship to eqn (1) and are shown in Fig. 7. The final Eads values are lower when the dopants Hf and Zr are at the first layer of the TiO2 surface forming a bond with the sarin molecule. The Eads values for the molecular and dissociative adsorption of sarin on Hf-doped TiO2 were −2.40 and −3.22 eV, respectively. For the case of sarin on Zr-doped TiO2 the molecular and dissociative adsorption had values of −2.18 and −3.03 eV, respectively, as shown in Fig. 7. When these dopants are positioned at the other TiO2 layers, the Eads values increase (between 0.3 and 0.5 eV larger) and in some cases are even higher than the undoped systems. In the case of the molecular and dissociative adsorption of sarin on Ge-doped TiO2, the Eads shows in general a constant tendency with higher values than those of sarin on Hf and Zr-doped TiO2, as well as the undoped systems.
In terms of the different types of adsorption, the Eads values calculated for the dissociative adsorption of sarin on X-doped TiO2 are lower than for the molecular adsorption, for all the cases considered. The comparison between the doped and undoped systems showed that the adsorption of sarin on Hf and Zr-doped TiO2 where the dopant is placed at the first layer is more stable compared with the undoped systems, for the cases of both molecular and dissociative adsorption.
In addition, as the dopant position moves farther away from the surface, and from the sarin molecule, the Eads values are closer to those of the undoped system, indicating that the effect of the dopant becomes more tenuous, as can be expected. For the case of Hf as the dopant, the asymptotic adsorption energy for both molecular and dissociative adsorption, as the layer depth increases is still a little lower than the adsorption energy for the undoped TiO2. While dopants located deeply within the titania might have a less direct impact compared to surface dopants, they can still influence catalytic activity through electronic and strain effects that extend to the surface. The extent of this influence depends on the material properties, dopant characteristics, and the depth of the dopant within the titania structure. Considering the ionic radius, lattice strain and electronegativity of the different dopant elements Hf, Zr and Ge, compared with Ti in the rutile structure, the combined properties of Hf (small radius and electronegativity) can lead to a more stable structure when doping the rutile TiO2 material and therefore be a better candidate for the adsorption of sarin compared to the other doped materials. The comparison in the direction of the Eads observed for the three dopant elements indicate that in the case of Hf a surface reconstruction or strain can be present which could lead to variation in the properties compared to the other dopants.
The charge transfer (ΔQ) was calculated for each atom of the sarin X-doped TiO2 systems (X = Hf, Zr or Ge), using the following expression:
ΔQ = QSarin-X-doped![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) TiO2 − QX-doped TiO2 − QX-doped![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) TiO2 − QSarin TiO2 − QSarin | (2) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) TiO2 is the charge of the sarin X-doped TiO2 system, QX-doped
TiO2 is the charge of the sarin X-doped TiO2 system, QX-doped![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) TiO2 is the charge of the X-doped TiO2 surface with X = Hf, Zr or Ge being at the same position as for the sarin X-doped TiO2 system and QSarin is the charge of the sarin molecule. All these quantities are evaluated at each atom of the specific system. Fig. 8 shows ΔQ at some relevant atoms for the molecularly and dissociatively adsorbed sarin on doped TiO2 systems for the Hf, Zr and Ge dopants. The atomic positions and labels are depicted at the sarin on the doped TiO2 structure.
TiO2 is the charge of the X-doped TiO2 surface with X = Hf, Zr or Ge being at the same position as for the sarin X-doped TiO2 system and QSarin is the charge of the sarin molecule. All these quantities are evaluated at each atom of the specific system. Fig. 8 shows ΔQ at some relevant atoms for the molecularly and dissociatively adsorbed sarin on doped TiO2 systems for the Hf, Zr and Ge dopants. The atomic positions and labels are depicted at the sarin on the doped TiO2 structure.
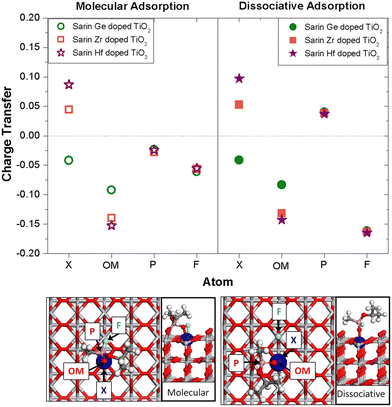 | ||
| Fig. 8 Charge transfer calculated at different atoms of the molecularly and dissociatively adsorbed sarin on X-doped TiO2 systems for X = Hf, Zr or Ge. The dopant is on the surface layer. | ||
Among the ΔQ values obtained for the different atoms, the results for the dopants (X) and OM draw special attention. The ΔQ values calculated at the dopant atom showed a higher value for sarin on Hf-doped TiO2 compared with the systems with Zr or Ge. These results apply to both the molecular as well as the dissociative adsorption. In addition, there is a clear difference in the charge transfer direction for the sarin on X-doped TiO2, when the system is doped with Ge compared to Hf or Zr. In the case of sarin on Hf and Zr-doped TiO2 the charge transfer is positive, while the same property displays a negative value for sarin on Ge-doped TiO2.
These results can be interpreted as due to the difference in electronegativities between the different dopants. The electronegativities of Zr and Hf (1.33 and 1.3, respectively) are lower than the electronegativity of Ti (1.54), while the opposite is true for Ge (2.01). The Ge dopant atom has the tendency to attract more electrons when it substitutes the Ti atom of the TiO2 surface, while Hf and Zr show the opposite effect. A similar comparison for the ΔQ values at the OM atom of sarin on TiO2 doped with the different elements show that ΔQ is more negative for the system doped with Hf compared to Zr and Ge. In the X–OM bond, the charge has a preferred direction toward O due to its higher electronegativity3,50 and becomes even more negative when the TiO2 surface is doped with Hf compared to Zr and Ge. Once more, the difference in electronegativity between Ti and the dopant atoms play a role in the charge transfer direction. In addition, the analysis of ΔQ for the P atom shows a different charge transfer direction for the molecular and dissociative configurations with positive and negative values, respectively.
In the molecularly adsorbed configuration, the F atom forms a P–F bond in the sarin molecule, while in the dissociatively adsorbed configuration the F atom dissociates and bonds with a Ti atom at the surface. The breakage of the P–F bond causes a variation not only in the ΔQ value but also in the charge transfer direction at the P atom.
The comparison for sarin on X-doped TiO2 between the different dopants indicates that the trend observed for the adsorption energy in terms of increasing stability is also observed qualitatively for the charge difference. Sarin on the Hf-doped TiO2 system with the Hf dopant at the first layer had the lowest Eads (the highest stability) and showed a higher charge transfer, while the Eads had the lowest value for sarin on the Ge-doped TiO2 system with charge transfer having a lower value and also an opposite direction compared with the Hf-doped system, which suggest that the trend observed for the Eads could have the same origin in the electronic properties of sarin on doped systems.
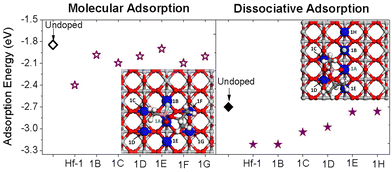 | ||
| Fig. 9 Calculated adsorption energies for molecular and dissociative adsorption of sarin on Hf-doped TiO2(110) with Hf at different positions on the first TiO2 layer (Hf atoms in blue). | ||
In the case of the molecular adsorption of sarin on Hf-doped TiO2, we observe that the Eads values for the additional dopant positions considered (1B, 1C, 1D, 1E, 1F and 1G) are between −1.90 and −2.10 eV which are similar to the Eads of the sarin-TiO2 undoped system (−1.85 eV) and higher than the value of −2.4 eV for the dopant position Hf-1 in which case sarin bonds with the dopant Hf. In the case of the dissociative adsorption of sarin on Hf-doped TiO2, the Eads values range between −3.20 and −2.80 eV with 2 systems being more stable and having the same Eads value (−3.20 eV). These Eads are obtained for sarin on Hf-doped TiO2 systems when Hf is bonding with the OM atom (Hf-1, as also described previously) and when Hf is bonding with the F atom dissociated from sarin, corresponding to 1B in Fig. 9.
We calculated the charge transfer at different atomic positions and when the Hf dopant is at the 1B position, we found that ΔQ at Hf is small (0.02 e), while ΔQ at the F atom is negative with a value close to −0.11. In the Hf–F bond, the charge could go in the direction toward the F atom which could be the reason for the small ΔQ value obtained at the Hf atom. The values for ΔQ at OM and P are around −0.16 and 0.04 eV with the positive charge difference in P due to the dissociation of F from sarin and the charge rearrangement in the remaining molecule. The additional dopant positions also have a lower Eads compared with the undoped system (−2.70 eV), with two positions having values close to −3.0 eV. In these two positions, the O2fc atom bonding with the remaining sarin molecule after dissociation is also forms X–O bonds with the dopant, corresponding to positions 1C and 1D in Fig. 9. The results described here show that not only the dopant element but also the dopant position plays a decisive role in the stability and dissociation of the sarin–TiO2 system.
4. Conclusions
The molecular and dissociative adsorption of sarin on dry, wet and doped TiO2(110) surfaces was studied using density functional theory. We found that(i) For the different cases considered here, the dissociative adsorption of sarin had lower adsorption energy compared against the molecular adsorption, with the F atom dissociating from the sarin molecule and forming a bond with a Ti5fc atom from the surface.
(ii) The energy barrier for the dissociation process of sarin on dry TiO2 was obtained as 0.35 eV in which an initial molecular adsorption was followed by a dissociative adsorption of sarin on the dry TiO2 surface.
(iii) The calculation of the adsorption energy for sarin on wet TiO2 surfaces showed that the dissociative adsorption of sarin on a hydrated TiO2 with 0.5 H2O ML coverage was the preferred configuration among the different wet and dry systems considered.
(iv) The adsorption and dissociation of sarin on doped TiO2 with Hf, Zr and Ge as single point dopants at different layers of the TiO2 slab was also studied and the calculations showed higher stability for the dissociative adsorption of sarin on Hf-doped TiO2 compared to Zr and Ge doped systems. The dopant position also had an effect on the stability of sarin on doped TiO2 systems.
(v) The calculation of the charge transfer at the dopant atom and the sarin molecule for sarin on doped TiO2 showed the highest value for the system with the highest stability, namely, sarin on Hf-doped TiO2.
The results obtained in this work suggest that methods such as the hydration, hydroxylation and doping of TiO2 could represent effective ways to improve the adsorption and dissociation of sarin on this metal oxide and more importantly could facilitate the decontamination process of this highly toxic molecule.
Author contributions
Conceptualization, Y. C. Q., R. N.; methodology and investigation, Y. C. Q.; formal analysis, Y. C. Q., R. N.; writing – original draft preparation, Y. C. Q.; writing – review and editing, Y. C. Q., R. N.; project administration, R. N. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
The authors declare no conflict of interest.Abbreviations
| Ach | Acetylcholine |
| AChE | Acetylcholine esterase |
| CWA | Chemical warfare agent |
| DFT | Density functional theory |
| DMMP | Dimethyl methyl phosphonate |
| GB | Sarin, Isopropyl methyl phosphono fluoridate |
| ML | Monolayer |
| OP | Organophosphate |
| PAW | Projected augmented wave |
| PBE-GGA | Perdew–Burke–Ernzerhof generalized gradient approximation |
Data availability
All original data from this study are included in the article itself and further inquiries can be directed to the corresponding author. Example of an input file for the sarin–TiO2 system with molecular adsorption is included in the ESI† Section C.Acknowledgements
This research was conducted as part of the core research at the U.S. Army Combat Capabilities Development Command Soldier Center. The authors acknowledge computational support through the DoD High-Performance Computing Modernization Program. Yenny Cardona Quintero was a National Research Council Postdoctoral Fellow during this project.References
- Y. J. Jang, K. Kim, O. G. Tsay, D. A. Atwood and D. G. Churchill, Update 1 of: Destruction and detection of chemical warfare agents, Chem. Rev., 2015, 115, PR1–PR76 CrossRef CAS PubMed.
- R. Stone, How to defeat a nerve agent, Science, 2018, 359, 23 CrossRef CAS PubMed.
- Chemical Weapons Convention, Organization for the Prohibition of Chemical Weapons, 2025, https://www.opcw.org/chemical-weapons-convention.
- V. Štengl, J. Henych, P. Janoš and M. Skoumal, Nanostructured metal oxides for stoichiometric degradation of chemical warfare agents, Rev. Environ. Contam. Toxicol., 2016, 236, 239–258 Search PubMed.
- N. Sharma and R. Kakkar, Recent Advancements on warfare agents/metal oxides surface chemistry and their simulation study, Adv. Mater. Lett., 2013, 4, 508–521 CrossRef.
- E. D. Davis, W. O. Gordon, A. R. Wilmsmeyer, D. Troya and J. R. Morris, Chemical warfare agent surface adsorption: Hydrogen bonding of sarin and soman to amorphous silica, J. Phys. Chem. Lett., 2014, 5, 1393–1399 CrossRef CAS PubMed.
- G. W. Wagner, O. B. Koper, E. Lucas, S. Decker and K. J. Klabunde, Reactions of VX, GD, and HD with nanosize CaO: Autocatalytic dehydrohalogenation of HD, J. Phys. Chem. C, 2000, 104, 5118–5123 CrossRef CAS.
- W. Zhang, A. M. Asiri, D. Liu, D. Du and Y. Lin, Nanomaterial-based biosensors for environmental and biological monitoring of organophosphorus pesticides, Trends Anal. Chem., 2014, 54, 1–10 CrossRef CAS.
- T. Li, R. Tsyshevsky, M. McEntee, E. M. Durke, C. Karwacki, E. E. Rodriguez and M. M. Kuklja, Titania nanomaterials for sarin decomposition: Understanding fundamentals, ACS Appl. Nano Mater., 2022, 5, 6659–6670 CrossRef CAS.
- C. Tesvara, C. J. Karwacki and P. Sautet, Decomposition of the toxic nerve agent sarin on oxygen vacancy sites of rutile TiO2(110), J. Phys. Chem. C, 2023, 127, 8006–8015 CrossRef CAS.
- G. Bazargan, I. V. Schweigert and D. Gunlycke, Adsorption of organophosphate nerve agent VX on the (101) surface of anatase titanium dioxide, Surf. Sci., 2022, 716, 121957 CrossRef CAS.
- T. Hirakawa, K. Sato, A. Komano, S. Kishi, C. K. Nishimoto, N. Mera, M. Kugishima, T. Sano, H. Ichinose, N. Negishi, Y. Seto and K. Takeuchi, Experimental study on adsorption and photocatalytic decomposition of isopropyl methylphosphonofluoridate at surface of TiO2 photocatalyst, J. Phys. Chem., 2010, 114, 2305–2314 CAS.
- K. Sato, T. Hirakawa, A. Komano, S. Kishi, C. K. Nishimoto, N. Mera, M. Kugishima, T. Sano, H. Ichinose, N. Negishi, Y. Seto and K. Takeuchi, Titanium dioxide photocatalysis to decompose isopropyl methylphosphonofluoridate (GB) in gas phase, Appl. Catal., B, 2011, 106, 316–322 CrossRef CAS.
- D. R. Sengele, N. Keller, V. Keller, A. Herissan and C. Colbeau-Justin, Ta-doped TiO2 as photocatalyst for UV-A activated elimination of chemical warfare agent simulant, J. Catal., 2016, 334, 129–141 CrossRef.
- C. S. Cao, W. Liu, A. Ma, X. Jiao, Y. Yang, J. Li and F. Fu, Single-atom Pt-decorated TiO2 nanotubes for boosted photocatalytic degradation of chemical warfare agents, ACS Sustainable Chem. Eng., 2025, 13, 6379–6387 CrossRef CAS.
- V. Štengl, T. M. Grygar, F. Opluštil and T. Němec, Ge4+ doped TiO2 for stoichiometric degradation of warfare agents, J. Hazard. Mater., 2012, 227, 62–67 CrossRef PubMed.
- Z. Shen, J. Y. Zhong, N. N. Chai, X. He, J. Z. Zang, H. Xu, X. Y. Han and P. Zhang, Evolution of microstructural defects of TiO2 nanocrystals by Zr4+ or/and Ge4+ doping lead to high disinfection efficiency for CWAs, Chem. Phys. Lett., 2017, 678, 146–152 CrossRef CAS.
- J. Lavoie, R. Srinivasan and R. Nagarajan, Using cheminformatics to find simulants for chemical warfare agents, J. Hazard. Mater., 2011, 194, 85–91 CrossRef CAS PubMed.
- C. N. Rusu and J. T. Yates, Adsorption and decomposition of dimethyl methylphosphonate on TiO2, J. Phys. Chem. B, 2000, 104, 12292–12298 CrossRef CAS.
- D. A. Panayotov and J. R. Morris, Uptake of a chemical warfare agent simulant (DMMP) on TiO2: Reactive adsorption and active site poisoning, Langmuir, 2009, 25, 3652–3658 CrossRef CAS PubMed.
- D. A. Panayotov and J. R. Morris, Thermal decomposition of a chemical warfare agent simulant (DMMP) on TiO2: Adsorbate reactions with lattice oxygen as studied by infrared spectroscopy, J. Phys. Chem. C, 2009, 113, 15684–15691 CrossRef CAS.
- J. Zhou, S. Ma, Y. C. Kang and D. A. Chen, Dimethyl methylphosphonate decomposition on titania-supported Ni clusters and films: A comparison of chemical activity on different Ni surfaces, J. Phys. Chem. B, 2004, 108, 11633–11644 CrossRef CAS.
- J. Zhou, K. Varazo, J. E. Reddic, M. L. Myrick and D. A. Chen, Decomposition of dimethyl methylphosphonate on TiO2 (110): Principal component analysis applied to X-ray photoelectron spectroscopy, Anal. Chim. Acta, 2003, 496, 289–300 CrossRef CAS.
- A. R. Wilmsmeyer, W. O. Gordon, E. D. Davis, D. Troya, B. A. Mantooth, T. A. Lalain and J. R. Morris, Infrared spectra and binding energies of chemical warfare nerve agent simulants on the surface of amorphous silica, J. Phys. Chem. C, 2013, 117, 15685–15697 CrossRef CAS.
- P. Janos, P. Kuran, M. Kormunda, V. Stengl, T. M. Grygar, M. Dosek, M. Stastny, J. Ederer, V. Pilarova and L. Vrtoch, Cerium dioxide as a new reactive sorbent for fast degradation of parathion methyl and some other organophosphates, J. Rare Earths, 2014, 32, 360–370 CrossRef CAS.
- A. Tiwari, V. Adepu, R. S. Fernandes, N. Dey, P. Sahatiya and S. Kanungo, Perylene diimide architecture-based electromechanical sensors: a systematic experimental and theoretical framework for the comparative analysis and study of the transduction mechanism, Mater. Adv., 2024, 5, 9243–9258 RSC.
- A. Tiwari, H. V. Barkale, N. Harshini, S. Chennareddy, N. Dey and S. Kanungo, Phenylalanine and tyrosine interactions with the WSe2 nanosheet: A comparative analysis based on DFT calculation and experimental confirmation, J. Phys. Chem. C, 2025, 129, 1769–1778 CrossRef CAS.
- H. J. Cho, S. H. Kim, J. Kang, K. Cho, H. Y. Lee, H. Kim, H. K. Ju, J. W. Choi, S. G. Kim, S. G. Oh, C. W. Lee and H. C. Yoon, Experimental and DFT studies on the equilibrium properties, kinetics, and mechanism of nitric oxide removal using metal-EDTA and ferrous thiochelates, Chem. Eng. J., 2022, 431, 134010 CrossRef CAS.
- V. M. Bermudez, Computational study of environmental effects in the adsorption of DMMP, Sarin, and VX on γ-Al2O3: Photolysis and surface hydroxylation, J. Phys. Chem. C, 2009, 113, 1917–1930 CrossRef CAS.
- A. Michalkova, M. Ilchenko, L. Gorb and J. Leszczynski, Theoretical study of the adsorption and decomposition of sarin on magnesium oxide, J. Phys. Chem. B, 2004, 108, 5294–5303 CrossRef CAS.
- A. Michalkova and J. Leszczynski, Interactions of nerve agents with model surfaces: Computational approach, J. Vac. Sci. Technol., A, 2010, 28, 1010–1017 CrossRef CAS.
- B. N. Papas, I. D. Petsalakis, G. Theodorakopoulos and J. L. Whitten, CI and DFT studies of the adsorption of the nerve agent sarin on surfaces, J. Phys. Chem. C, 2014, 118, 23042–23048 CrossRef CAS.
- R. Wang, B. Wang, A. S. Abdullahi and H. Fan, Understanding the prototype catalyst TiO2 surface with the help of density functional theory calculation, Comput. Mol. Sci., 2024, 14, e1686 CrossRef CAS.
- P. Janos, J. Henych, O. Pelant, V. Pilarova, L. Vrtoch, M. Kormunda, K. Mazanec and V. Stengl, Cerium oxide for the destruction of chemical warfare agents: A comparison of synthetic routes, J. Hazard. Mater., 2016, 304, 259–268 CrossRef CAS PubMed.
- V. S. Vaiss, I. Borges and A. A. Leit, Sarin degradation using brucite, J. Phys. Chem. C, 2011, 115, 24937–24944 CrossRef CAS.
- A. Michalkova, J. Martinez, O. A. Zhikol, L. Gorb, O. V. Shishkin, D. Leszczynska and J. Leszczynski, Theoretical study of adsorption of sarin and soman on tetrahedral edge clay mineral fragments, J. Phys. Chem. B, 2006, 110, 21175–21183 CrossRef CAS PubMed.
- A. Michalkova, Y. Paukku, D. Majumdar and J. Leszczynski, Theoretical study of adsorption of tabun on calcium oxide clusters, Chem. Phys. Lett., 2007, 438, 72–77 CrossRef CAS.
- L. Yang, D. Tunega, L. Xu, N. Govind, R. Sun, R. Taylor, H. Lischka, W. A. Dejong and W. L. Hase, Comparison of cluster, slab, and analytic potential models for the dimethyl methylphosphonate (DMMP)/TiO2(110) intermolecular interaction, J. Phys. Chem. C, 2013, 117, 17613–17622 CrossRef CAS.
- V. M. Bermudez, Ab initio study of the interaction of dimethyl methylphosphonate with rutile (110) and anatase (101) TiO2 surfaces, J. Phys. Chem. C, 2010, 114, 3063–3074 CrossRef CAS.
- V. M. Bermudez, First-principles study of adsorption of dimethyl methylphosphonate on the TiO2 anatase (001) surface: Formation of a stable titanyl (Ti = O) site, J. Phys. Chem. C, 2011, 115, 6741–6747 CrossRef CAS.
- V. M. Bermudez, Computational study of the adsorption of dimethyl methylphosphonate (DMMP) on the (010) surface of anatase TiO2 with and without faceting, Surf. Sci., 2010, 604, 706–712 CrossRef CAS.
- Y. Paukku, A. Michalkova and J. Leszczynski, Adsorption of dimethyl methylphosphonate and trimethyl phosphate on calcium oxide: An ab initio study, Struct. Chem., 2008, 19, 307–320 CrossRef CAS.
- J. Quenneville, R. S. Taylor and A. C. T. van Duin, Reactive molecular dynamics studies of DMMP adsorption and reactivity on amorphous silica surfaces, J. Phys. Chem. C, 2010, 114, 18894–18902 CrossRef CAS.
- M. R. Housaindokht and N. Zamand, A DFT study of associative and dissociative chemical adsorption of DMMP onto SnO2(110) surface nanocluster, Struct. Chem., 2015, 26, 87–96 CrossRef CAS.
- D. A. Trubitsyn and A. V. Vorontsov, Molecular and reactive adsorption of dimethyl methylphosphonate over (001) and (100) anatase clusters, Comput. Theor. Chem., 2013, 1020, 63–71 CrossRef CAS.
- A. Kiselev, A. Mattson, M. Andersson, A. E. C. Palmqvist and L. Osterlund, Adsorption and photocatalytic degradation of diisopropyl fluorophosphate and dimethyl methylphosphonate over dry and wet rutile TiO2, J. Photochem. Photobiol., A, 2006, 184, 125–134 CrossRef CAS.
- M. B. Mitchell, V. N. Sheinker and E. A. Mintz, Adsorption and decomposition of dimethyl methylphosphonate on metal oxides, J. Phys. Chem. B, 1997, 101, 11192–11203 CrossRef CAS.
- A. R. Head, R. Tsyshevsky, L. Trotochaud, Y. Yu, L. Kyhl, O. Karslıoǧlu, M. M. Kuklja and H. Bluhm, Adsorption of dimethyl methylphosphonate on MoO3: The role of oxygen vacancies, J. Phys. Chem. C, 2016, 120, 29077–29088 CrossRef CAS.
- L. Trotochaud, R. Tsyshevsky, S. Holdren, K. Fears, A. R. Head, Y. Yu, S. Pletincx, B. Eichhorn, M. Zachariah and M. M. Kuklja, et al., Spectroscopic and computational investigation of room-temperature decomposition of a chemical warfare agent simulant on polycrystalline cupric oxide, Chem. Mater., 2017, 29, 7483–7496 CrossRef CAS.
- I. V. Schweigert and D. Gunlycke, Hydrolysis of dimethyl methylphosphonate by the cyclic tetramer of zirconium hydroxide, J. Phys. Chem. A, 2017, 121, 7690–7696 CrossRef CAS PubMed.
- G. Wang, C. Sharp, A. M. Plonka, Q. Wang, A. I. Frenkel, W. Guo, C. Hill, C. Smith, J. Kollar, D. Troya and J. R. Morris, Mechanism and kinetics for reaction of the chemical warfare agent simulant, DMMP(g), with Zirconium (IV) MOFs: An ultrahigh-vacuum and DFT study, J. Phys. Chem. C, 2017, 121, 11261–11272 CrossRef CAS.
- S. J. Smith, R. Stevens, S. Liu, G. Li, A. Navrotsky, J. Boerio-Goates and B. F. Woodfield, Heat capacities and thermodynamic functions of TiO2 anatase and rutile: Analysis of phase stability, Am. Mineral., 2009, 94, 236–243 CrossRef CAS.
- Z. H. Cui, F. Wu and H. Jiang, First-principles study of relative stability of rutile and anatase TiO2 using the random phase approximation, Phys. Chem. Chem. Phys., 2016, 18, 29914–29922 RSC.
- J. C. Slater, Atomic radii in crystals, J. Chem. Phys., 1964, 41, 3199–3205 CrossRef CAS.
- J. Perdew, J. Chevary, S. Vosko, K. Jackson, M. Pederson, D. Singh and C. Fiolhais, Atoms, molecules, solids, and surfaces: Applications of the generalized gradient approximation for exchange and correlation, Phys. Rev. B: Condens. Matter Mater. Phys., 1992, 46, 6671–6687 CrossRef CAS PubMed.
- P. E. Blöchl, Projector augmented-wave method, Phys. Rev. B: Condens. Matter Mater. Phys., 1994, 50, 17953–17979 CrossRef.
- P. Giannozzi, S. Baroni, N. Bonini, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, G. L. Chiarotti, M. Cococcioni and I. Dabo, et al., QUANTUM ESPRESSO: A modular and open-source software project for quantum simulations of materials, J. Phys.: Condens. Matter, 2009, 21, 395502 CrossRef PubMed.
- S. Grimme, Semiempirical GGA-type density functional constructed with a long-range dispersion correction, J. Comput. Chem., 2006, 27, 1787–1799 CrossRef CAS PubMed.
- R. F. W. Bader, Atoms in Molecules: A Quantum Theory, Clarendon Press, Oxford, 1994 Search PubMed.
- G. Henkelman, A. Arnaldsson and H. Jónsson, A fast and robust algorithm for Bader decomposition of charge density, Comput. Mater. Sci., 2006, 36, 354–360 CrossRef.
- Henkelman Group Code for Bader Charge Analysis, 2023. https://theory.cm.utexas.edu/henkelman/code/bader/.
- Y. Cardona Quintero and R. Nagarajan, Molecular and dissociative adsorption of DMMP, sarin and soman on dry and wet TiO2(110) using density functional theory, Surf. Sci., 2018, 675, 26–35 CrossRef.
- A. Kaczmarek, L. Gorb, A. J. Sadlej and J. Leszczynski, Sarin and soman: Structure and properties, Struct. Chem., 2004, 15, 517–525 CrossRef CAS.
- X. Shan, J. C. Vincent, S. Kirkpatrick, M. D. Walker, M. R. Sambrook and D. C. Clary, A combined theoretical and experimental study of sarin (GB), J. Phys. Chem. A, 2017, 121, 6200–6210 CrossRef CAS PubMed.
- T. Ash, T. Debnath, T. Banu and A. K. Das, Exploration of unimolecular gas-phase detoxication pathways of sarin and soman: A computational study from the perspective of reaction energetics and kinetics, Chem. Res. Toxicol., 2016, 29, 1439–1457 Search PubMed.
- N. Q. Le, C. E. Ekuma, B. I. Dunlap and D. Gunlycke, First-principles calculations of sarin adsorption on anatase surfaces, J. Phys. Chem. C, 2018, 122, 2832–2839 CrossRef CAS.
- S. Tan and B. Wang, Active Sites for Adsorption and reaction of molecules on rutile TiO2(110) and anatase TiO2(001) surfaces, Chin. J. Chem. Phys., 2015, 28, 383–395 CrossRef CAS.
- C. J. Howard, T. M. Sabine and F. Dickson, Structural and thermal parameters for rutile and anatase, Acta Crystallogr., Sect. B, 1991, 47, 462–468 CrossRef.
- D. C. Taylor, K. Runge, M. G. Cory, D. S. Burns, J. L. Vasey, J. D. Hearn, K. Griffith and M. V. Henley, Surface binding of organophosphates on silica: Comparing experiment and theory, J. Phys. Chem. C, 2013, 117, 2699–2708 CrossRef CAS.
- V. Štengl, S. Bakardjieva, N. Murafa and F. Opluštil, Zirconium doped titania: Destruction of warfare agents and photocatalytic degradation of orange 2 dye, Open Process Chem. J., 2008, 1, 1–7 CrossRef.
- V. Štengl, S. Bakardjieva, N. Murafa, F. Opluštil, L. Österlund, A. Mattsson and P. O. Andersson, Warfare agents degradation on zirconium doped titania, Microsc. Microanal., 2009, 15, 1038–1039 CrossRef.
- V. Štengl, T. M. Grygar, F. Opluštil and T. Nemec, Ge4+ doped TiO2 for stoichiometric degradation of warfare agents, J. Hazard. Mater., 2012, 227–228, 62–67 CrossRef PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d5cp01092f |
| ‡ Current address: Naval Undersea Warfare Center, Division Newport, Newport, RI 02841, USA. |
| This journal is © the Owner Societies 2025 |