Improvement of electronic structure calculations for the interpretation of the emission spectrum for NdIII complexes†
Yolimar
Gil
 a,
María José
Maldonado
a,
María José
Maldonado
 a,
Ricardo Costa
de Santana
a,
Ricardo Costa
de Santana
 b,
Andrés
Vega
b,
Andrés
Vega
 c,
Pablo
Fuentealba
c,
Pablo
Fuentealba
 *a and
Daniel
Aravena
*a and
Daniel
Aravena
 *d
*d
aFacultad de Ciencias Químicas y Farmacéuticas, Universidad de Chile, Olivos 1007, 8380544 Santiago, Chile. E-mail: pfuentealbacastro@ciq.uchile.cl
bInstituto de Física, Universidade Federal de Goiás, Goiania 74690-900, GO, Brazil
cFacultad de Ciencias Exactas, Dpto. de Ciencias Químicas, Universidad Andrés Bello, Viña del Mar, Chile
dDepartamento de Química de los Materiales, Facultad de Química y Biología, Universidad de Santiago de Chile, Casilla 40, Correo 33, Santiago, Chile. E-mail: daniel.aravena.p@usach.cl
First published on 10th April 2025
Abstract
A protocol to correct ab initio calculated luminescence spectra of NdIII complexes is proposed. The emission spectrum of [NdIII(bipy)(tta)3] was measured to calibrate the optimal correction for the Racah parameters on top of a CASSCF calculation to attain the best energetic placement of the 4F3/2 → 4I13/2−9/2 emission lines. As interelectronic repulsion is the most important source of error in this calculation, this straightforward correction results in an accurate placement of transitions, allowing the assignment of a complex spectral shape in terms of its underlying transitions. Finally, the correction derived for [NdIII(bipy)(tta)3] was directly applied to a different NdIII complex, demonstrating the broad use of this approach.
Introduction
Electronic structure calculations are extensively employed in many fields of chemistry to augment the information extractable from experimental measurements. For instance, reactivity studies can often identify products and determine information about reaction rates purely from experimental data, while detailed mechanistic proposals are often based on electronic structure calculations.1 Similar approaches are found in other fields such as magnetism, where the overall magnetic moment can be probed experimentally but the microscopic magnetic interactions responsible for that response are studied computationally.2 In the field of spectroscopy, ab initio calculations are often exploited to aid in the assignment of spectra ranging from magnetic resonances to high-energy techniques, such as X-ray experiments.3UV/vis absorption and emission spectroscopy studies routinely profit from this experimental and theoretical synergy. The most common computational method in this area is time dependent density functional theory (TD-DFT),4 which provides excitation energies and oscillator strengths at an affordable computational cost for molecular systems up to a couple hundred of atoms at the present time. In many cases, the deviations between experimental and TD-DFT predicted spectra are systematic and fixed energy shifts are used to force a better match between experiment and theory.5 Such corrections, although empirical, have proven to be useful enough to become common practice nowadays, especially for closed shell, organic molecules.
Inorganic systems, particularly lanthanides, have their own caveats which complicate the computational production of spectra that can accurately match experiments, even employing simple empirical corrections. The most fundamental problem is the inadequacy of single reference methods, such as DFT, to provide spectroscopically consistent wave functions.6 This can be solved using multireference methods such as complete active space self-consistent field (CASSCF), confirmed by the generalized use of this method in lanthanide magnetism.7–15 However, CASSCF calculations suffer from additional problems to be accurate in optics. The most important is their lack of dynamical correlation, which leads to the overestimation of interelectronic repulsion, resulting in poor relative energies between 4f multiplets.16 This effect manifests in strong misplacements of f–f absorption and emission band wavelengths, which can be as large as hundreds of nanometres. Even worse, the relative energy of each pair of multiplets will be displaced by a different magnitude in a complicated way, preventing the use of a fixed energy shift to correct the experiment. Interestingly, ligand field and spin–orbit effects are better described in lanthanide CASSCF calculations,16 so the correction of just interelectronic repulsion can yield a dramatic correction of f–f transition energies.
Recent advances in this field indicate that a good description of lanthanide luminescence requires the calculation of larger active spaces, including additional orbital shells of the lanthanide and key ligand orbitals.17,18 Perturbative corrections are also beneficial for a better agreement with experimental data.19 The main drawback of this path is the increased computational cost of these more sophisticated approaches and the need for assessing the proper active space in a case to case basis.
In this article, we provide a simple procedure to correct CASSCF calculations to obtain NIR emission spectra with a good placement of the f–f bands in NdIII complexes. Thus, this protocol helps in the assignment of the main features of the spectra without increasing the computational cost beyond the one associated with CASSCF. The procedure does not rely on any symmetry idealization of the coordination environment and can be thus applied to high- and low-symmetry systems at an equal footing. This correction is general for any NdIII system and was calibrated using the emission spectrum of tris(thenoyltrifluoroacetonato)2,2′-dipyridylneodymium(III) ([NdIII(bipy)(tta)3]), which was synthesized using literature methods20 and measured in our laboratory. CASSCF calculations were performed using the crystallographic structure deposited in the CCDC database21 (see Fig. 1). The faint nephelauxetic effect present in lanthanides suggests that the correction of Racah parameters derived for [NdIII(bipy)(tta)3] will be useful for other NdIII systems without the need of recalibrating them in a case-to-case fashion. To prove this point, we synthesized a new NdIII complex with a completely different coordination environment ([NdIIIL1(NCS)3]) (Fig. 1). Then, we measured its emission spectrum at room temperature and applied the corrected CASSCF protocol, achieving a good agreement between experimental and theoretical data.
 | ||
| Fig. 1 Molecular structures of the [NdIII(bipy)tta3] (top) and [NdIIIL1(NCS)3] (bottom). Colour code: Nd (light yellow), S (yellow), F (light green), O (red), N (blue), C (grey) and H (white). | ||
Energy correction procedure
The procedure to generate the corrected spectrum considers three steps: (i) perform a standard CASSCF(3,7) calculation considering all quartet (35) and doublet (112) roots, where the active space includes the 4f shell of the NdIII ion, (ii) obtain the corrected energies using orca_lft with the corrected Racah parameters provided in this article and (iii) perform a new CASSCF calculation using the corrected energies. The last calculation can be run on top of the converged wavefunction of the first step, so the simulation will mark convergence at the first step and proceed to the spin–orbit coupling part swiftly. A detailed example of the procedure is provided as ESI.†The emission spectrum is simulated assuming the following formula for the intensity (I):22–24
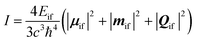 | (1) |
To perform the statistical fit of the corrected Racah parameters, we synthesized a simple, yet representative emissive complex, [NdIII(bipy)(tta)3]. The choice of NdIII is intended to provide a NIR emission spectrum with several discernible bands to fit (4F3/2 → 4IJ (J = 9/2–13/2)). The function to optimize was the cross-correlation between the experimental and calculated emission curves. As the NdIII spectrum shows three relevant NIR bands with different intensities, the cross-correlation was calculated for each peak separately, where experimental and calculated data were normalised to present a maximum of 1. Thus, the optimization function to maximize was the sum of squares of the three cross-correlations. In this way, the optimization procedure will not be biased towards adjusting the most intense peak at the expense of the other two. The maximum of the cross-correlation function was searched using the LIPO algorithm.26 The curve broadening was chosen to maximize the objective function at each point of the optimization, so the final Racah parameters are not affected by the broadening.
Results and discussion
The molecular structures of [NdIII(bipy)(tta)3] and [NdIIIL1(NCS)3] are presented in Fig. 1, which evidences the differing coordination environment observed for these two compounds. Starting from the nature of the donor atoms, [NdIII(bipy)(tta)3] exhibits an O6N2 motif from the three acetonates and the bipyridine ligand. In contrast, [NdIIIL1(NCS)3] shows a first coordination sphere composed exclusively by nitrogen atoms.In terms of geometrical conformation, [NdIII(bipy)(tta)3] is an octacoordinated complex, which can be better described as a distorted square antiprism (with a Shape measure of 0.651). On the other hand, [NdIIIL1(NCS)3] is a nonacoordinate system close to the muffin and the hula-hoop conformation (3.591 and 3.823, respectively). As discussed later, these differences were sufficient for obtaining two clearly distinguishable emission profiles for the most intense band (4F3/2 → 4I11/2). In this way, we can test if the proposed correction can provide spectral shapes, which include meaningful features of the coordination environment besides the bare energy correction of the bands.
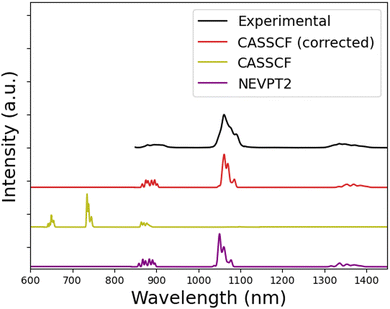
Fig. 2 presents the experimental and calculated emission spectra for [NdIII(bipy)(tta)3], which served to calibrate the corrected Racah parameters, as described in the previous section. As expected, the CASSCF calculation (olive line) depicts a strong overestimation of the energy difference between the 4F3/2 and the 4I multiplets, resulting in a general blueshift of the bands, which is around 250–400 nm, depending on the band. The NEVPT2 corrected results (violet) shift closer to the experiment, with a slight difference for the maximum of the 4F3/2 → 4I11/2 transition. The CASSCF corrected spectrum (red) shows an accurate placement of the bands, consistent with the optimization procedure. The shape and relative intensity of the three bands is reasonably captured by all methods. In the case of the most intense band (4F3/2 → 4I11/2), there are three discernible peaks which descend in intensity at longer wavelength. This pattern is captured in all calculations.
Table 1 shows the comparison between the CASSCF, NEVPT2, CASSCF optimized, and experimental Racah parameters. CASSCF calculations yield a large overestimation of interelectronic repulsion, as expected by the lack of dynamical correlation characteristic of this method.16 Perturbative approaches, such as NEVPT2 can correct for this deviation. In this case, almost matching the reference values. However, the computational burden of NEVPT2 can significantly extend the computation time of the bare CASSCF since all quartet (35) and doublet (112) roots must be corrected. In this case, the total execution time of the CASSCF run was 2 h while the NEVPT2 calculation took 237 h. The difference in execution time between NEVPT2 and CASSCF depends strongly on the number of roots to calculate. In this sense, other emissive lanthanides have an even larger number of roots (as EuIII or TbIII), so the computational time of NEVPT2 can be even longer. On the other side, ions like PrIII and TmIII have fewer roots to correct so NEVPT2 should be more accessible.
Corrected CASSCF calculations provide an accurate placement of the emission bands, close to the one obtained from NEVTP2. Thus, the statistical fit of the emission curve of [NdIII(bipy)(tta)3] produced effective Hamiltonian parameters, which are close to the ones adjusted to lanthanide elpasolites in ref. 27 and 28 in accordance with the inner nature and relative insensitivity of the 4f shell with respect to the coordination environment.
After establishing the procedure and its comparison with experiments, we employed the information derived from the calculation to describe the main features of the emission spectrum for [NdIII(bipy)(tta)3]. Focusing on the most intense peak (4F3/2 → 4I11/2), we can decompose the contributions from each Kramers’ doublet to the total emission, either from the initial (4F3/2) or final state (4I11/2). The maximum splitting of 4F3/2 and 4I11/2 multiplets is two and six, respectively; thus, the 4F3/2 → 4I11/2 band can be decomposed in twelve transitions. Fig. 3 shows the decomposition of the emission stemming from the low (blue bands) and high (red bands) energy Kramers’ doublets of the 4F3/2 multiplet. Clearly, the three peak pattern is generated by the transitions from the lower energy doublet of 4F3/2 to the six components of the 4I11/2 multiplet, which are distributed in three main blocks involving one (1), two (2, 3) and three (4, 5, 6) Kramers’ doublets (see Dieke diagram in Fig. 3).
The contributions from the upper doublet of 4F3/2 mainly give intensity to the two more intense peaks and result in a weak signal at the left end of the band. In the literature, these peaks are known as hot bands,29,30 and are indeed assigned to emission of high energy states of the emissive doublet.
To check the accuracy of the method and its applicability to other systems, we synthesized the NdIII analog of a complex previously published by our group ([NdIIIL1(NCS)3], according to the nomenclature of ref. 31). As mentioned earlier, this complex presents a different coordination number and therefore coordination geometry than [NdIII(bipy)(tta)3], and the first coordination sphere is formed by only nitrogen atoms. Fig. 4 shows the satisfactory performance of the method in this new system, which even captures the different pattern observed for the main peak, with two clearly discernible maxima for [NdIIIL1(NCS)3] instead of the three peaks observed for [NdIII(bipy)(tta)3].
As evidenced by the decomposition of the 4F3/2 → 4I11/2 band from [NdIIIL1(NCS)3], the origin of the two-peak patterns relates to a different pattern in the transition intensities in comparison to the one observed for [NdIII(bipy)(tta)3] (see Fig. 5). In the case of [NdIIIL1(NCS)3], there is a very intense transition stemming from the higher emissive Kramers’ doublet, which dominates the peak at shorter wavelength (indicated as 4′ in Fig. 5). The other main peak is exclusively explained by one transition from the lower emissive state (transition 6 in Fig. 5). Hence, we can expect a different behaviour of these two peaks with respect to temperature, following the change in population of their respective emissive states. This contrasts with the situation for [NdIII(bipy)(tta)3], where the three peak pattern was mainly sustained by transitions associated with the lower emissive doublet.
We conclude this discussion with some general remarks about the extension of this correction for other lanthanide ions. In principle, there is no limitation to applying this same optimization procedure to spectra for other lanthanides. However, the optimization of the spectral shape requires that the source CASSCF calculation can produce transition intensities that resemble the experimental pattern to provide a meaningful fit. This is the case for [NdIII(bipy)(tta)3] and [NdIIIL1(NCS)3], where the three and two peak patterns for the most intense transition are reproduced in the bare CASSCF calculations, even if they are strongly misplaced in energy. This situation is not expected to be generally extensible for other lanthanide ions, especially for transitions with mixed electric and magnetic dipole mechanisms. In such situations, CASSCF will probably capture the intensity of the magnetic term properly, but will strongly underestimate the electric term. As both contributions follow different patterns, the calculated shape will not follow the observed trend. Thus, it is necessary to develop a method to correct this bias, either by exploring more sophisticated ab initio methods or introducing ad hoc corrections.
Conclusions
In sum, we optimized Racah parameters to correct CASSCF(3,7) calculations, obtaining a better placement of the emission bands of NdIII complexes. The corrected spectrum was in close agreement with reference experimental data and with NEVPT2, which is a more sophisticated and computationally demanding approach. Furthermore, the shape of the most intense peak was satisfactorily reproduced for the two studied systems. This level of agreement allowed the decomposition of the band shape into their individual contributions and the discovery of the transitions responsible for the observed pattern. In this way, the three-maxima shape for the most intense peak of [NdIII(bipy)(tta)3] is related with transitions originating from the lower emissive doublet of the 4F3/2 multiplet, while the pattern of [NdIIIL1(NCS)3] mixes contributions from both doublets belonging to 4F3/2. This analysis suggests a different thermal dependence of both spectroscopic signals. This kind of analysis could be useful for luminescence thermometry studies.Experimental
Synthesis of [NdIII(bipy)tta3]
Reagent grade chemicals and HPLC quality solvents were used as received. The synthesis of [NdIII(bipy)(tta)3] was performed by using an Anton Paar microwave reactor MonoWave 200, following a reported procedure with some modifications,20 which is described as follows: 2-thenoyltrifluoroacetone (267 mg, 1.2 mmol) and triethylamine (168 μL, 1.2 mmol) were added to a 30-mL volume microwave vial containing 15 mL of methanol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethanol mixture (1
ethanol mixture (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Then, Nd(NO3)3·6H2O (175 mg, 0.4 mmol) was added and the mixture was stirred for 15 min. Finally, a solution of 2,2′-bipyridine (bipy) (63.2 mg, 0.4 mmol) in 5 mL of the same solvent mixture was added, and the resulting solution was irradiated in the microwave reactor for 15 min at 100 °C. Purple rod crystals were obtained by slow evaporation of the reaction solution after 4 days with a yield of 265 mg (69%).
1). Then, Nd(NO3)3·6H2O (175 mg, 0.4 mmol) was added and the mixture was stirred for 15 min. Finally, a solution of 2,2′-bipyridine (bipy) (63.2 mg, 0.4 mmol) in 5 mL of the same solvent mixture was added, and the resulting solution was irradiated in the microwave reactor for 15 min at 100 °C. Purple rod crystals were obtained by slow evaporation of the reaction solution after 4 days with a yield of 265 mg (69%).
Synthesis of [NdIIIL1(NCS)3]
To obtain the mononuclear NdIII complex, the template reaction was done by reproducing a procedure that was previously published by our group,31 which is briefly described as follows: an ethanolic solution of the lanthanide triflate salt (0.5 mmol/30 mL) was added to a solution of 2,6-pyridinedicarboxaldehyde (1 mmol/50 mL acetonitrile), under constant stirring at room temperature. To close the macrocyclic ligand, ethylenediamine was added dropwise (1 mmol/50 mL acetonitrile). Finally, to incorporate the thiocyanate anions, Bu4NSCN was added (1.5 mmol/20 mL ethanol). The final solution was stirred for four hours, obtaining a homogeneous solution. Single crystals were formed from the mother liquor after two or three weeks (42.7% yield).Structural and spectroscopic characterization
Powder X-ray diffraction (PXRD) patterns were collected at room temperature using Bruker D-8 ADVANCE equipment, with CuKα1 radiation. This pattern was compared with the calculated PXRD obtained from the crystal structure32 (Fig. S1, ESI†). The agreement of these patterns supports the pure state of the bulk. The solid-state emission spectrum was recorded on a spectrofluorimeter (Fluorolog-3, Horiba Scientific) with a diode laser (Crystal Laser LC) of 804 nm.Computational details
All calculations were carried out using the ORCA 5.0.4 software.33,34 As the crystal structure of [NdIII(bipy)(tta)3] from the CCDC database21 lacked hydrogen atoms and presented disorder at the thiophene groups, a geometry optimization of the peripheral atoms was performed prior to the CASSCF calculations. To avoid disrupting the coordination environment of the NdIII ion, the metal position, all coordinating atoms and carbon atoms forming the chelate bite were kept frozen at their crystallographic positions. To avoid convergence problems, the NdIII ion was replaced by YIII. The structural relaxation was done by a density functional theory calculation using the BP86 functional35,36 and the Def2-TZVP basis set,37 including a 28 electron ECP for the Y atom.38 The crystallographic structure of [NdL1(NCS)3] was employed without further optimization. CASSCF(3,7)39 calculations were run on top of the optimized structure, where the active space was composed of the 4f electrons. 35 quartets and 112 doublets were considered to saturate the excitation space. The Def2-TZVP basis set was selected for light elements and the SARC2-DKH-QZVP40 basis set was chosen for NdIII. NEVPT219,41,42 correction was applied on top of the CASSCF converged wave functions. Racah, spin–orbit coupling and ligand field parameters were obtained by means of the ab initio ligand field theory approach.43 Scalar relativistic effects were accounted for using the DKH Hamiltonian.44,45 Spin–orbit coupling was described by means of the Quasi-degenerate perturbation theory. Calculations were accelerated by using the RI approximation for the integral transformation step.46Magnetic dipole and electric quadrupole contributions were calculated using the “quad true” keyword although they were later discarded from the analysis since their magnitude was negligible in comparison to the electric dipole term. The emission spectrum was calculated by extracting the oscillator strength from the absorption spectrum and weighting the population of the emissive 4F3/2 multiplet by their Boltzmann population. The intensity is calculated as in eqn (1).22–24
Author contributions
Yolimar Gil: investigation, data curation, writing; María José Maldonado: investigation, data curation.; Ricardo Costa de Santana: resources, writing; Andrés Vega: investigation, data curation, writing; Pablo Fuentealba: investigation, data curation, writing; Daniel Aravena: conceptualization, investigation, formal analysis, writing.Data availability
The data supporting this article have been included as part of the ESI.† The crystal structure of [NdIIIL1(NCS)3] was deposited with the CCDC number 2421205.Conflicts of interest
There are no conflicts to declare.Acknowledgements
D. A. and P. F thank FONDECYT Regular projects 1210325 and 1241928 for financial support. Y. G. thanks ANID FONDECYT Postdoctoral Project 3220200. Powered@NLHPC: This research was partially supported by the supercomputing infrastructure of the NLHPC (CCSS210001). RCS thanks the CNPq research fellowship 310307/2021-0 and FAPEG (202310267000236).References
- M. Breugst, Enabling Tools and Techniques for Organic Synthesis, Wiley, 2023, pp. 259–311 Search PubMed.
- G. Rajaraman, Computational Modelling of Molecular Nanomagnets, Springer International Publishing, Cham, 2023, vol. 34 Search PubMed.
- V. Barone, S. Alessandrini, M. Biczysko, J. R. Cheeseman, D. C. Clary, A. B. McCoy, R. J. DiRisio, F. Neese, M. Melosso and C. Puzzarini, Computational molecular spectroscopy, Nat. Rev. Methods Primers, 2021, 1, 38 CrossRef CAS.
- E. Runge and E. K. U. Gross, Density-Functional Theory for Time-Dependent Systems, Phys. Rev. Lett., 1984, 52, 997–1000 CrossRef CAS.
- A. Ali, M. I. Rafiq, Z. Zhang, J. Cao, R. Geng, B. Zhou and W. Tang, TD-DFT benchmark for UV-visible spectra of fused-ring electron acceptors using global and range-separated hybrids, Phys. Chem. Chem. Phys., 2020, 22, 7864–7874 RSC.
- F. Neese, Prediction of molecular properties and molecular spectroscopy with density functional theory: From fundamental theory to exchange-coupling, Coord. Chem. Rev., 2009, 253, 526–563 CrossRef CAS.
- Y. Gil and D. Aravena, Understanding Single-Molecule Magnet properties of lanthanide complexes from 4f orbital splitting, Dalton Trans., 2024, 53, 2207–2217 RSC.
- J. Torrent, C. Puigjaner, R. Herchel and J. Mayans, Cerium-Based Metal–Organic Frameworks: Unveiling the Role of Terahertz Vibrations in the Spin Relaxation Dynamics, Inorg. Chem., 2025, 64, 3735–3746 CrossRef PubMed.
- V. Briganti and A. Lunghi, A machine-learning framework for accelerating spin-lattice relaxation simulations, NPJ Comput. Mater., 2025, 11, 62 CrossRef PubMed.
- X. Gou, Y. Wu, H. Wen, L. Li, W. Lan, N. Liu, S. Zhang, L. Ungur, P. Cheng and W. Shi, Modulating Static and Dynamic Magnetizations of Ytterbium(III) Coordination Polymers by Light-Induced Radicals, CCS, Chemistry, 2025, 1–9 Search PubMed.
- V. Vieru, S. Gómez-Coca, E. Ruiz and L. F. Chibotaru, Increasing the Magnetic Blocking Temperature of Single-Molecule Magnets, Angew. Chem., Int. Ed. Engl., 2024, 63, e202303146 CrossRef CAS PubMed.
- À. Tubau, S. Gómez-Coca, S. Speed, M. Font-Bardía and R. Vicente, New series of mononuclear β-diketonate cerium(III) field induced single-molecule magnets, Dalton Trans., 2024, 53, 9387–9405 RSC.
- S. C. Corner, G. K. Gransbury, I. J. Vitorica-Yrezabal, G. F. S. Whitehead, N. F. Chilton and D. P. Mills, Halobenzene Adducts of a Dysprosocenium Single-Molecule Magnet, Inorg. Chem., 2024, 63, 9552–9561 CrossRef CAS PubMed.
- A. Swain, Y. L. Whyatt, D. Wielechowski, S. Muthu, S. L. Benjamin, K. S. Murray, G. Rajaraman and S. K. Langley, Enhancing blocking temperatures in {CrIII2DyIII2} butterfly SMMs: deciphering the role of exchange interactions and developing magneto-structural maps, Inorg. Chem. Front., 2025, 12, 1059–1079 RSC.
- N. Monni, S. Dey, V. García-López, M. Oggianu, J. J. Baldoví, M. L. Mercuri, M. Clemente-León and E. Coronado, Tunable SIM properties in a family of 3D anilato-based lanthanide-MOFs, Inorg. Chem. Front., 2024, 11, 5913–5923 RSC.
- D. Aravena, M. Atanasov and F. Neese, Periodic Trends in Lanthanide Compounds through the Eyes of Multireference ab Initio Theory, Inorg. Chem., 2016, 55, 4457–4469 CrossRef CAS PubMed.
- V. R. M. Nielsen, M. Grasser, S. S. Mortensen, B. Le Guennic and T. J. Sørensen, Electronic Structure of a Neodymium(III) Tris(oxidiacetate) Complex from Luminescence Data and Ab Initio Calculations, Inorg. Chem., 2024, 63, 18596–18607 CrossRef CAS PubMed.
- M. Grasser and B. Le Guennic, Ab initio investigations of circularly polarised luminescence in Samarium(III)-based complexes, Phys. Chem. Chem. Phys., 2024, 26, 7203–7210 RSC.
- C. Angeli, R. Cimiraglia and J.-P. Malrieu, N-electron valence state perturbation theory: a fast implementation of the strongly contracted variant, Chem. Phys. Lett., 2001, 350, 297–305 CrossRef CAS.
- H.-Y. Wong, W.-S. Lo, W. T. K. Chan and G.-L. Law, Mechanistic Investigation of Inducing Triboluminescence in Lanthanide(III) β-Diketonate Complexes, Inorg. Chem., 2017, 56, 5135–5140 CrossRef CAS PubMed.
- C. R. Groom, I. J. Bruno, M. P. Lightfoot and S. C. Ward, The Cambridge Structural Database, Acta Crystallogr., Sect. B: Struct. Sci., Cryst. Eng. Mater., 2016, 72, 171–179 CrossRef CAS PubMed.
- F. Gendron, B. Moore, O. Cador, F. Pointillart, J. Autschbach and B. Le Guennic, Ab Initio Study of Circular Dichroism and Circularly Polarized Luminescence of Spin-Allowed and Spin-Forbidden Transitions: From Organic Ketones to Lanthanide Complexes, J. Chem. Theory Comput., 2019, 15, 4140–4155 CrossRef CAS PubMed.
- B. Pritchard and J. Autschbach, Calculation of the Vibrationally Resolved, Circularly Polarized Luminescence of D-Camphorquinone and (S,S)-trans-β-Hydrindanone, Chem. Phys. Chem., 2010, 11, 2409–2415 CrossRef CAS PubMed.
- G. Longhi, E. Castiglioni, S. Abbate, F. Lebon and D. A. Lightner, Experimental and Calculated CPL Spectra and Related Spectroscopic Data of Camphor and Other Simple Chiral Bicyclic Ketones, Chirality, 2013, 25, 589–599 CrossRef CAS PubMed.
- F. S. Richardson, Selection rules for lanthanide optical activity, Inorg. Chem., 1980, 19, 2806–2812 CrossRef CAS.
- C. Malherbe and N. Vayatis, Proceedings of the 34th International Conference on Machine Learning, 2017, vol. 70, pp. 2314–2323.
- F. S. Richardson, M. F. Reid, J. J. Dallara and R. D. Smith, Energy levels of lanthanide ions in the cubic Cs2NaLnCl6 and Cs2NaYCl6:Ln3+ (doped) systems, J. Chem. Phys., 1985, 83, 3813–3830 CrossRef CAS.
- M. F. Reid and F. S. Richardson, Free-ion, crystal-field, and spin-correlated crystal-field parameters for lanthanide ions in Cs2NaLnCl6 and Cs2NaYCl6:Ln3+ systems, J. Chem. Phys., 1985, 83, 3831–3836 CrossRef CAS.
- W. Yang, M. Rosenkranz, G. Velkos, F. Ziegs, V. Dubrovin, S. Schiemenz, L. Spree, M. F. de Souza Barbosa, C. Guillemard, M. Valvidares, B. Büchner, F. Liu, S. M. Avdoshenko and A. A. Popov, Covalency versus magnetic axiality in Nd molecular magnets: Nd-photoluminescence, strong ligand-field, and unprecedented nephelauxetic effect in fullerenes NdM2N@C80 (M = Sc, Lu, Y), Chem. Sci., 2024, 15, 2141–2157 RSC.
- D. K. Sardar and R. M. Yow, Stark components of 4F3/2, 4I9/2 and 4I11/2 manifold energy levels and effects of temperature on the laser transition of Nd3+ in YVO4, Opt. Mater., 2000, 14, 5–11 CrossRef CAS.
- Y. Gil, P. Fuentealba, A. Vega, E. Spodine and D. Aravena, Control of magnetic anisotropy by macrocyclic ligand distortion in a family of DyIII and ErIII single molecule magnets, Dalton Trans., 2020, 49, 17709–17718 Search PubMed.
- J. G. Leipoldt, L. D. C. Bok, S. S. Basson and A. E. Laubscher, The crystal structure of tris(thenoyltrifluoroacetonato)2,2′-dipyridylneodymium(III), J. Inorg. Nucl. Chem., 1976, 38, 1477–1479 CrossRef CAS.
- F. Neese, Software update: The ORCA program system—Version 5.0, WIREs Comput. Mol. Sci., 2022, 12, e1606 Search PubMed.
- F. Neese, The SHARK integral generation and digestion system, J. Comput. Chem., 2023, 44, 381–396 CrossRef CAS PubMed.
- A. D. Becke, Density-functional exchange-energy approximation with correct asymptotic behavior, Phys. Rev. A, 1988, 38, 3098–3100 CrossRef CAS PubMed.
- J. P. Perdew, Density-functional approximation for the correlation energy of the inhomogeneous electron gas, Phys. Rev. B: Condens. Matter Mater. Phys., 1986, 33, 8822–8824 CrossRef PubMed.
- F. Weigend and R. Ahlrichs, Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy, Phys. Chem. Chem. Phys., 2005, 7, 3297 RSC.
- D. Andrae, U. Häussermann, M. Dolg, H. Stoll and H. Preuss, Energy-adjustedab initio pseudopotentials for the second and third row transition elements, Theor. Chim. Acta, 1990, 77, 123–141 CrossRef CAS.
- P.-Å. Malmqvist and B. O. Roos, The CASSCF state interaction method, Chem. Phys. Lett., 1989, 155, 189–194 CrossRef CAS.
- D. Aravena, F. Neese and D. A. Pantazis, Improved Segmented All-Electron Relativistically Contracted Basis Sets for the Lanthanides, J. Chem. Theory Comput., 2016, 12, 1148–1156 CrossRef CAS PubMed.
- Y. Guo, K. Sivalingam, E. F. Valeev and F. Neese, SparseMaps—A systematic infrastructure for reduced-scaling electronic structure methods. III. Linear-scaling multireference domain-based pair natural orbital N-electron valence perturbation theory, J. Chem. Phys., 2016, 144, 094111 CrossRef PubMed.
- C. Angeli, R. Cimiraglia, S. Evangelisti, T. Leininger and J.-P. Malrieu, Introduction of n-electron valence states for multireference perturbation theory, J. Chem. Phys., 2001, 114, 10252–10264 CrossRef CAS.
- M. Atanasov, D. Ganyushin, K. Sivalingam and F. Neese, A Modern First-Principles View on Ligand Field Theory Through the Eyes of Correlated Multireference Wavefunctions, Molecular Electronic Structures of Transition Metal Complexes II, Structure and Bonding, Springer, 2011, vol. 143, pp. 149–220 DOI:10.1007/430_2011_57.
- M. Reiher, Douglas–Kroll–Hess Theory: a relativistic electrons-only theory for chemistry, Theor. Chem. Acc., 2006, 116, 241–252 Search PubMed.
- T. Nakajima and K. Hirao, The Douglas–Kroll–Hess Approach, Chem. Rev., 2012, 112, 385–402 CrossRef CAS PubMed.
- G. L. Stoychev, A. A. Auer and F. Neese, Automatic Generation of Auxiliary Basis Sets, J. Chem. Theory Comput., 2017, 13, 554–562 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Additional structural and spectroscopic data, and sample input files. CCDC 2421205. For ESI and crystallographic data in CIF or other electronic format see DOI: https://doi.org/10.1039/d5cp00508f |
| This journal is © the Owner Societies 2025 |