Implications of magnetic dilution of PrFeO3 with Bi3+ on its dielectric and magnetic properties†
Shivangi
Rao
 a,
Subhajit
Sau
a,
Subhajit
Sau
 b,
V.
Kanchana
b,
V.
Kanchana
 b,
G.
Vaitheeswaran
b,
G.
Vaitheeswaran
 *c and
Rajamani
Nagarajan
*c and
Rajamani
Nagarajan
 *a
*a
aMaterials Chemistry Group, Department of Chemistry, University of Delhi, Delhi-110007, India. E-mail: 18darklotus@gmail.com; rnagarajan@chemistry.du.ac.in
bDepartment of Physics, Indian Institute of Technology Hyderabad, Kandi, Medak-502285, Telangana, India. E-mail: ph21resch01001@iith.ac.in; kanchana@phy.iith.ac.in
cSchool of Physics, University of Hyderabad, Prof. C. R. Rao Road, Gachibowli, Hyderabad 500046, Telangana, India. E-mail: vaithee@uohyd.ac.in
First published on 28th April 2025
Abstract
Research on functional oxide ceramics has tremendous translational potential for technological applications. Despite sharing a similar formula, BiFeO3 and REFeO3 (RE = rare earth) perovskites differ widely in structure and properties. The potential of substituting non-magnetic Bi3+ with a stereochemically active 6s2 lone pair in rare-earth ferrites remains largely unexplored. In this work, the consequences of replacing Pr3+ with Bi3+ on the dielectric and magnetic properties of PrFeO3 were investigated by synthesizing the samples using a solution combustion method. The inclusion of bismuth led to local site disorder and promoted the reduction of more amounts of Fe3+ to Fe2+, as verified by Raman and XPS measurements. The higher concentration of Fe2+ resulted in the formation of oxygen vacancies. The band gaps of the pure and Bi-substituted PrFeO3 samples were in the range of 1.90–2.08 eV. The field and temperature-dependent magnetic measurements of Pr1−xBixFeO3 confirmed the magnetic dilution. ZFC and FC measurements at low fields revealed a spin reorientation transition at 101 K in the case of Pr0.70Bi0.30FeO3, which supported the negative exchange bias effect at room temperature. The dielectric constant increased with an increase in bismuth content. Electronic structure calculations with charge density plots revealed the induction of polarization by the electric field in the Bi3+-containing samples. This was also verified through PUND measurements, which showed the existence of intrinsic and switchable polarization (0.17 μC cm−2). The random distribution of the stereochemically active 6s2 lone pair on Bi3+ has been proposed as the reason for the observed polarization.
1 Introduction
Solids with a perovskite structural arrangement are the prototype for demonstrating any application among the materials science community.1–4 The perovskite structure's flexibility to accommodate multiple substitutions with suitable cations, exhibiting diverse symmetries, allows researchers to tailor their functions based on optical, electrical, magnetic, and catalytic properties.1–4 The halide-based perovskites deserve special mention as they offer better possibilities for a renewable, sustainable energy conversion source.5 Although the perovskite structure is ideally cubic, structural deviations from distortion can lower its symmetry to tetragonal, orthorhombic, rhombohedral, monoclinic, and triclinic.2–4 The distortion in the perovskite structure in compounds made under ambient pressure and temperature conditions has been correlated to the Goldschmidt tolerance factor (t) involving the ionic radii of the constituent ions.6 Structurally distorted perovskite oxides occur when ‘t’ is less than unity, modifying their electrical and magnetic properties. The most common distortion is the tilting/rotation of the rigid BO6 octahedra imposed by the small size of the A-cation while maintaining their corner-sharing connectivity.6 Despite sharing a similar formula, BiFeO3 and REFeO3 (RE = rare earth) perovskites differ widely in their structure and properties.7–11 While BiFeO3 has a rhombohedrally distorted structure, REFeO3 exhibits orthorhombic distortion. BiFeO3 crystallizes in the non-centrosymmetric space group (R3c (#161)), where the cuboctahedral and octahedral positions of the perovskite unit cell are occupied by Bi3+ and Fe3+ ions, respectively. Displacement of Bi3+ ions from their center of symmetry along the [111]c direction and antiphase-rotated FeO6 units along the rhombohedral axis (Glazer notation a−a−a−) are present in the structure.1–4 Due to the stereo-active lone pair of electrons (6s2), the Bi3+ ions have a more significant displacement compared to Fe3+ ions, resulting in ferroelectric ordering (TC ∼ 830 °C) and antiferromagnetic (AFM) ordering (TN ∼ 370 °C).7–9 The magnetic moments of Fe3+ in BiFeO3 show a G-type antiferromagnetic (AFM) arrangement.7–9 On the other hand, rare-earth orthoferrites (REFeO3; RE = Pr–Lu) show orthorhombic distortion of the perovskite structure crystallizing in the non-polar Pnma (#62) space group. Its symmetry is described by an in-phase tilt of FeO6 octahedra in the a-direction and out-of-phase tilt of FeO6 octahedra in the b- and c-directions (Glazer notation a+b−b−).10 While the Bi3+ ion in BiFeO3 has an eight-fold coordination with oxygen, the RE3+ ion in REFeO3 is a pseudo-twelve-coordinated polyhedron with eight first-nearest oxygen atoms and four second-nearest oxygen atoms. Of the two magnetic sublattices in REFeO3, Fe3+–Fe3+ exchange interactions dominate over those of Fe3+–RE3+ and RE3+–RE3+, which causes the spins to order near 650–700 K with a slightly canted G-type AFM arrangement.In the search for materials exhibiting a large electrical polarization along with the magnetic ordering, researchers consider rare-earth orthoferrites as an alternative to BiFeO3, Aurivillius phases, PbZrO3, and PbTiO3.12–20 NdFeO3, SmFeO3, GdFeO3, DyFeO3, and HoFeO3, despite crystallizing in centrosymmetric space groups (Pnma/Pbnm) showed ferroelectricity facilitated by the exchange striction caused by the spin-exchange interaction between the 4f- and 3d-ion-containing sublattices.14–19 In the case of YFeO3 (containing a non-magnetic Y3+ ion), the origin of ferroelectricity is attributed to the antisite defects, creating a net local dipole moment.12 Hence, taking a magnetic orthoferrite and diluting the A-site with a non-magnetic trivalent ion assumes greater significance as it can bring in more clarity on the origin of ferroelectricity in rare-earth orthoferrites.
The Bi3+ ion has a stereochemically active 6s2 lone pair, and introducing such a metal ion in rare-earth orthoferrites can perform the dual functions of applying chemical pressure and introducing possible polarization under applied electric fields. It is worth noting that theoretical simulations have demonstrated a large enough epitaxial misfit strain (compressive and tensile) to induce improper ferroelectricity in REFeO3 thin films.21–23 The consequences of including non-magnetic Bi3+ in place of magnetic RE3+ or non-magnetic Y3+ in the REFeO3 system are quite limited.24–28 The results from these studies are divergent, yielding fewer answers but many questions. In the case of LaFeO3, the bismuth inclusion caused the reduction of Fe3+ to Fe2+, leading to oxygen vacancies, whereas, in Bi0.50Nd0.50FeO3, the exclusive presence of Fe3+ was deduced.24–27 While the weak ferromagnetic behavior became stronger with bismuth substitution in LaFeO3, further weakening in the ferromagnetic behavior was noticed in another report.25,26 Importantly, ferromagnetic and antiferromagnetic coupling leads to an exchange bias effect in bismuth-substituted LaFeO3 samples.26 Quite a few reports have dealt with the outcome of Bi-substitution in PrFeO3 and none has investigated the physical property changes introduced by Bi3+. The existence of two redox couplets (Pr4+/3+ and Fe3+/2+) may be a possible reason for the availability of less reports as they may hinder their synthesis and such an investigation can contribute to an understanding of the role of bismuth in modulating its properties.29 From such a study, one can derive crucial information that can link the observations reported earlier in Bi-substituted LaFeO3 and NdFeO3 systems. To address this gap, we investigated the changes in the structure and optical, magnetic, and dielectric properties of PrFeO3 after substituting it with progressive amounts of Bi3+ (up to 50 mol%) by preparing the samples employing the combustion method. The results from these experiments have been discussed in detail. The encouraging results from the density functional theory (DFT) calculations about the possibility of electric field polarization in Bi3+-containing PrFeO3 samples have been verified from experiments.
2 Experimental details
2.1 Synthesis
The synthesis of Pr1−xBixFeO3 (x = 0.00, 0.10, 0.20, 0.30, 0.40, and 0.50) was carried out following an auto-combustion process using Pr2O3 (99.9%, Sigma Aldrich), Bi2O3 (99.999%, Sigma Aldrich), iron metal powder (98.5%, Central Drug House), citric acid (99%, Central Drug House), and HNO3 (68% v/v, Merck).3 The amounts of reactants used in the synthesis are tabulated in Table S1 (ESI†). Double-distilled water was used for the entire synthetic procedure. Stoichiometric quantities of Pr2O3 and Bi2O3 were dissolved separately in a minimum amount of dilute HNO3 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v)). The required amounts of iron metal powder were dissolved separately in a minimum quantity of concentrated HNO3 by heating (∼200 °C) to get a clear orange solution. These solutions were mixed thoroughly with the help of a magnetic stirrer, to which fuel (citric acid solution in water), in the fuel-to-oxidizer ratio of 0.30, was added. Intense brown-colored flaky ashes resulted after the ignition process when the solution was evaporated to dryness at 200 °C. The samples with x up to 0.20 were calcined in the air at 800 °C for 12 h, followed by natural cooling to room temperature (RT). For stoichiometries of x > 0.20, the samples were calcined in a flowing argon atmosphere at 800 °C for 12 h.
1 (v/v)). The required amounts of iron metal powder were dissolved separately in a minimum quantity of concentrated HNO3 by heating (∼200 °C) to get a clear orange solution. These solutions were mixed thoroughly with the help of a magnetic stirrer, to which fuel (citric acid solution in water), in the fuel-to-oxidizer ratio of 0.30, was added. Intense brown-colored flaky ashes resulted after the ignition process when the solution was evaporated to dryness at 200 °C. The samples with x up to 0.20 were calcined in the air at 800 °C for 12 h, followed by natural cooling to room temperature (RT). For stoichiometries of x > 0.20, the samples were calcined in a flowing argon atmosphere at 800 °C for 12 h.
2.2 Characterization details
A high-resolution Rigaku X-ray diffractometer (MiniFlex600), equipped with a xenon proportional detector engaging Cu Kα radiation (λ = 1.5418 Å), was employed for collecting the sample's powder X-ray diffraction (PXRD) pattern over a 2θ range of 10–100° with a step size of 0.01° and a scan rate of 0.16 seconds per step at RT (25 °C). The lattice and structural refinements of the PXRD patterns were carried out using the Le Bail and Rietveld methods with the help of the GSAS + EXPGUI program.30 The DIAMOND (Version 3.0) program was employed to construct the crystal structures. The sample's scanning electron microscopy (SEM) image and information about the constituent elements were obtained with the help of a JEOL 6610LV SEM 500 microscope equipped with an EDS attachment. With the help of an FEI Tecnai G2 F30 microscope (operating at 300 kV), the sample's high-resolution transmission electron microscopy (HRTEM) images and selected area electron diffraction (SAED) patterns were gathered. The sample's global and local site symmetries were analyzed with the help of Raman spectroscopy using a Renishaw inVia™ microscope system operating with an Ar+ laser (λ = 532 nm with a resolution of 1 cm−1). Repeated scans were performed on thin pellets of the prepared ceramics and averaged out to get the final spectrum. A PHI 5000 VersaProbe II, FEI Inc., X-ray photoelectron spectrophotometer was used to perform the XPS analysis of the samples. The spectrophotometer employed an Ar+ ion and C60 sputter gun and was operated at 10−9 Torr pressure. The core-level spectra were recorded using Al Kα radiation at a pass energy of 50 eV, an electron take-off angle of 90°, and a resolution of 0.1 eV. The chemically distinct species were resolved using a Gaussian distribution fitting procedure using the XPSPEAK41 software. Both the charge and peak-position corrections were carried out using the C 1s peak at 284.8 eV as a reference. The samples’ diffuse reflectance spectra (DRS) were recorded using a PerkinElmer Lambda 35 scanning double-beam spectrometer equipped with a 50 mm integrating sphere. The solid samples’ electron spin resonance spectra were recorded at RT using a JEOL Model JES-X320 ESR spectrometer operating at an X-band frequency of 8.75–9.65 GHz. Field- and temperature-dependent measurements were conducted using the magnetic property measurement system (MPMS) Excel model manufactured by Quantum Design USA with an applied field of 0.005, 0.01, 0.02 and 0.1 T. Magnetic-field-dependent measurements (±5 T) were performed at 298 and 2 K. The dielectric and ferroelectric measurements were conducted on sintered pellets (sintering done at 800 °C for 6 h) with a diameter of 10 mm. For sintering, the powders were mixed with polyvinyl alcohol (PVA) and pelletized at 10 MPa pressure for 5 min using a hydraulic pellet press. The samples had relative densities of ∼90–95% (measured using the Archimedes principle). For measurements, both sides of the pellets were coated with a conducting silver paste (SPI supplies, USA: SPI#05002-AB). The dielectric properties of the samples (thickness > 1 mm) were measured using a Novocontrol impedance analyzer in a wide range of frequencies (10−1–106 Hz) at RT. For ferroelectric measurements, the samples with thickness (∼ 0.6 mm) were poled electrically in silicon oil at RT under a DC electric field of 1 kV mm−1 for 2 h before measuring the polarization and electric field (P–E) curves using a computer-based automatic P–E loop tracer system (Model: 20PE 1 kHz 0.1N, Marine India, India) at RT up to an electric field of 20 kV cm−1 and 50 Hz frequency. To carry out the capacitance–voltage (C–V) and positive-up negative-down (PUND) measurement at RT, we employed an Advance P–E Loop Ferroelectric Test System (Marine India, Model: 0.03PE 2MHZ 1F).2.3 Computational details
In this study, calculations were carried out within the framework of density functional theory (DFT)31,32 using the projector-augmented plane-wave (PAW)33 method as implemented in the VASP package (version 5.4.4).34,35 We adopted the generalized gradient approximation (GGA) for the exchange–correlation functional with PBESol flavor.36 The atomic pseudopotentials used in the calculations incorporate the valence electronic configurations of the elements as follows: Pr (4f36s2, version 07Jan2002), Fe (3d64s2, version 03Mar1998), O (2s22p4, version 05Jan2001), Bi (5d106s26p3, version 10Feb1998). The d and f electrons were corrected through the DFT+U approximation within the Dudarev formalism.37 We used Hubbard parameter UPr = 2.8 eV and UFe = 4 eV for the pristine crystal structure. For the 50% Bi-doped structure we used UPr = 4.2 eV to match the experimental band gaps measured in this study. We have chosen UFe = 4 eV, as used in the first-principles calculations for a similar orthoferrite structure with an [FeO6] octahedral environment.38,39 The same value was used in rare-earth doped BiFeO3, which is the most frequent value used in the first-principles calculations.40 Therefore, in this study, we maintain a fixed U value for Fe as well while varying the U value for Pr in the theoretical calculations in accordance with its changing molar concentration in the experiment. All calculations were performed with a plane-wave energy cutoff of 700 eV, Monkhorst–Pack41k-mesh 8 × 6 × 8, and the energy convergence criterion was chosen to be 10−06 eV. All DFT calculations were carried out using the experimentally determined crystal structure. Band structures were plotted using the Sumo toolkit.42 The spontaneous polarization was evaluated using the Berry-phase method43,44 as implemented in VASP. The polarization calculation was performed utilizing both the PBE+U and PBESol+U exchange functionals to explore their impact.3 Results and discussion
The PXRD patterns of the calcined precursors with the compositions of Pr1−xBixFeO3 (x = 0.00–0.50) in air and under a flowing argon atmosphere are presented in Fig. 1(a) and Fig. S1 (ESI†). For the samples with x up to 0.20, diffraction peaks related to orthorhombic PrFeO3 (ICSD No: 98-002-7274) are observed in the PXRD patterns when calcined in the air. No other diffraction peaks are observed, signifying the absence of other crystalline secondary/impurity phases within the detection limit of the diffractometer. When the concentration of Bi(x) in Pr1−xBixFeO3 is 0.30, peaks of tetragonal Bi2O2.5 (ICSD No: 98-001-0470) and h-Pr2O3 (ICSD No: 98-006-1179) are seen in addition to the peaks of PrFeO3 (Fig. S1, ESI†). As both Pr and Fe can exist in mixed oxidation states, the atmosphere during the synthesis of these samples may be critical to avoid generating secondary phases. Hence, the ashes have been calcined in flowing argon for the compositions with x = 0.30–0.50 in Pr1−xBixFeO3, and in their PXRD patterns, peaks of the orthorhombic structure are present exclusively (Fig. 1(a)). The line profiles of h-Pr2O3 and various polymorphic forms of Bi2O3 are compared with the observed PXRD pattern of Pr0.50Bi0.50FeO3 in Fig. S2 (ESI†), from which we can rule out their presence in our samples. This reinforces the purity of our samples within the detection limit of the used diffractometer.The diffracted peaks shift towards lower 2θ values in the PXRD patterns of the samples with increased bismuth content, suggestive of the unit cell expansion caused by the higher ionic radius of Bi3+ (1.17 Å VIII-fold coordination) than Pr3+ (1.126 Å VIII-fold coordination) (Fig. 1(b)). The increase in the lattice parameters becomes more prominent beyond x = 0.20, hinting at the possible contribution from Fe2+ having a larger ionic radius.
For the refinement of the PXRD patterns, Pr3+ and Bi3+ ions were considered to occupy the 4c site, Fe3+ ions at the central 4a site at (0,0,0), and O2− ions at the 4c and 8d sites. While the isothermal parameters were refined for both the cations and anions, occupancies were refined for Pr/Bi at the 4c site. The positional parameters of the cations and anions were refined except for the parent composition and Fe3+ at the 4a site, as it was a special position in the structure. The experimental and theoretically simulated diffraction patterns converge successfully with satisfactory agreement parameters (Fig. 2). The absence of any unaccounted diffraction peak is verified from the expanded 2θ range shown in the insets of Fig. 2. The atomic parameters from the final cycle of refinements are collected in Table S2 (ESI†). The refinement data show that the concentration of the A-site with Bi in PrFeO3 increases the unit cell constants, with a concomitant increment in the unit cell volume. The increase in the a and b lattice parameters is quite substantial, while the increase in the c parameter is minimal (Fig. 1(c)). The diffracted peaks broaden with increasing bismuth content, indicating local structural changes. The refinement data shows that the Fe–O bond lengths expand as the A-site is concentrated with Bi (Table S3, ESI†). The two Fe–O–Fe angles are not equal to 180° due to the FeO6 octahedral tilt in the pseudocubic [111] direction. The bond angles gradually increase, preserving the tilted FeO6 octahedra, implying that the distortion extent decreases with increased Bi content. Still, the distortion is maintained due to the repulsions generated by stereochemically active lone pair of electrons in bismuth. The octahedral tilt angle (ϕ subtracted from the ideal 180°) decreases as the bismuth content increases, maintaining the distortion as shown pictorially in Fig. S3 (ESI†).28,45
A porous microstructure morphology with dense crystallites is observed in the SEM images of the samples of Pr1−xBixFeO3 series (x = 0.00–0.50) (Fig. 3(a) and (b) and Fig. S4, ESI†). The average crystallite size distribution of each sample, obtained using the Image J software analyzer, increases from 204–422 nm with the increase in bismuth content in PrFeO3. The EDS spectra of these samples confirm the qualitative presence of Pr, Bi, Fe, and O (Fig. S5, ESI†).
The PrFeO3 and Pr0.70Bi0.30FeO3 samples have also been analyzed using HRTEM and SAED techniques. The HRTEM images support the excellent crystallinity of the as-prepared samples. Both the PrFeO3 and Pr0.70Bi0.30FeO3 samples exhibit a well-resolved lattice fringe in the HRTEM images with the interplanar spacings of 3.49 nm and 2.28 nm corresponding to the (111) and (220) hkl planes of the orthorhombic structure (Fig. 3(c) and (e)). The SAED patterns of PrFeO3 and Pr0.70Bi0.30FeO3 show intense spots in a regular array, indicating that the particles are distinctly single crystalline (Fig. 3(d) and (f)). In the SAED patterns of the PrFeO3 and Pr0.70Bi0.30FeO3 samples, the distances between spots correspond to the (331), (301), (004), and (232) hkl planes.
The structural and microstructural changes arising from the Bi-substitution in PrFeO3 have been analyzed with the help of Raman spectroscopy. Fig. 4 shows the comparison of the Raman spectra of these samples at RT. Orthorhombic PrFeO3 (Pnma) can give rise to 24 Raman-active modes (7Ag + 5B1g + 7B2g + 5B3g) and 25 IR modes (9B1u + 7B2u + 9B3u).46 The phonon modes below 200 cm−1 are majorly sensitive to displacements of the rare-earth ions (Pr in this case) concerning the FeO6 octahedra, as highlighted in Fig. 4. The modes between 200 and 300 cm−1 relate to the vibrations involving both rare-earth and oxygen ions. The Raman modes above 300 cm−1 predominantly correspond to light oxygen ions (involving Fe–O bond lengths and Fe–O6 rotation and bending). As the four equivalent iron ions occupy the center of inversion in the Pnma lattice, one phonon vibration involving them is Raman-inactive.46,47 The Raman modes below 200 cm−1 (2Ag and B2g) and above 400 cm−1 (2B1g and B3g) are greatly affected due to Bi's inclusion at the Pr site, among other modes. A considerable shift in the peak positions towards lower wavenumbers, noted for the phonon modes below 200 cm−1 and above 400 cm−1 (2B1g and B3g), can be due to the substitution of bigger and heavier Bi3+ in place of lighter and smaller Pr3+. B2g and B3g modes (at ∼300 cm−1) and B1g and Ag modes (at ∼450 cm−1) merge for higher Bi concentrations (majorly commencing from x = 0.30). The transition from the sharp features to broad modes with increased bismuth content indicates the introduction of local site disorder. The local distortions include the changes in the preferred coordination number of Bi (pseudo-eight) over Pr (eight) and the deformation of FeO6 octahedra.48,49 Such local-site-induced disorder has also been reported in Bi1−xPrxFeO3.50 In the Raman spectra of all the Bi-substituted PrFeO3 samples, no additional band arising from any secondary/impurity phase is seen, confirming their microscopic purity.
As a change in synthesis atmosphere was required for making bismuth-containing samples beyond x = 0.20 in Pr1−xBixFeO3, we were curious to determine the valence states of the constituents of the PrFeO3 and Pr0.70Bi0.30FeO3 samples. Fig. 5(a) shows the XPS survey spectra of the pristine PrFeO3 and Pr0.70Bi0.30FeO3 samples, from which the presence of Pr, Fe, Bi, and O is deduced. The Pr-3d spectrum shows a common line shape characteristic of Pr3+, which, on deconvolution, consists of four peaks at 935.5, 939.5, 956.7, and 960.3 eV (Fig. 5(b)). The peaks with binding energies of 939.5 and 960.3 eV are related to Pr3+ (3d5/2) and Pr3+ (3d3/2), and the peaks at 935.5 and 956.7 eV correspond to shakedown satellite peaks arising from well-screened 4f3 final states, respectively.51 The spin–orbit splitting of 3d5/2 and 3d3/2 orbitals of praseodymium was found to be ∼20.8 eV, consistent with the literature.52 The mixed oxidation states of Fe in PrFeO3 were revealed from the Fe-2p spectral analysis. The peaks with binding energies of 715.5 (2p3/2) and 728.6 (2p1/2) eV are due to Fe2+ ions, whereas peaks with binding energies of 717.7 (2p3/2), 724.5 (satellite peak), and 731.3 (2p1/2) eV correspond to Fe3+ ions (Fig. 5(c)). The ratio of the peak area of Fe 2p3/2 compared to that of 2p1/2 is 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, consistent by considering the spin–orbit coupling.53 From the area under the curve analysis, 43 and 57% of iron exist as Fe2+ and Fe3+, respectively. The lattice oxygen appeared at a binding energy of 535.3 eV in the O-1s spectrum (Fig. 5(d)). The Pr-3d spectrum of the Pr0.70Bi0.30FeO3 sample contains peaks with binding energy values of 935.6 and 955.8 eV due to Pr3+ (3d5/2) and Pr3+ (3d3/2), and at 931.6 and 951.8 eV due to shakedown satellite peaks, respectively (Fig. 5(e)). The exclusive presence of Bi3+ was inferred from the Bi-4f spectrum, showing two peaks at 164.3 (4f7/2) and 169.8 eV (4f5/2) (Fig. 5(f)). Fe also existed in mixed oxidation states in the Bi-substituted sample, as inferred from the Fe-2p spectrum (Fig. 5(g)). The peaks around 712.7 (2p3/2) and 726.1 (2p1/2) eV arise from Fe2+ ions, and the peaks at the binding energy values of 714.2 (2p3/2), 720.6 (satellite peak), and 727.7 (2p1/2) eV are related to Fe3+ ions. Here again, the areas for 2p3/2 and 2p1/2 of Fe-2p confirm the expected ratio of 2
1, consistent by considering the spin–orbit coupling.53 From the area under the curve analysis, 43 and 57% of iron exist as Fe2+ and Fe3+, respectively. The lattice oxygen appeared at a binding energy of 535.3 eV in the O-1s spectrum (Fig. 5(d)). The Pr-3d spectrum of the Pr0.70Bi0.30FeO3 sample contains peaks with binding energy values of 935.6 and 955.8 eV due to Pr3+ (3d5/2) and Pr3+ (3d3/2), and at 931.6 and 951.8 eV due to shakedown satellite peaks, respectively (Fig. 5(e)). The exclusive presence of Bi3+ was inferred from the Bi-4f spectrum, showing two peaks at 164.3 (4f7/2) and 169.8 eV (4f5/2) (Fig. 5(f)). Fe also existed in mixed oxidation states in the Bi-substituted sample, as inferred from the Fe-2p spectrum (Fig. 5(g)). The peaks around 712.7 (2p3/2) and 726.1 (2p1/2) eV arise from Fe2+ ions, and the peaks at the binding energy values of 714.2 (2p3/2), 720.6 (satellite peak), and 727.7 (2p1/2) eV are related to Fe3+ ions. Here again, the areas for 2p3/2 and 2p1/2 of Fe-2p confirm the expected ratio of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The Pr0.70Bi0.30FeO3 sample contains 48 and 52% of Fe2+ and Fe3+, respectively. A nearly 5% rise in Fe2+-concentration was observed in the Pr0.70Bi0.30FeO3 sample, compared to pristine PrFeO3, promoted by the reducing atmosphere employed for the synthesis. Our observation matches the earlier report in which Fe2+ creation due to Bi's inclusion in LaFeO3 is noted.26 Two peaks at binding energies of 543.4 and 545.3 eV in the O-1s spectrum are attributed to the lattice oxygen (low binding energy) and the oxygen vacancies in the sample. Notably, the peak with the binding energy of 545.3 eV has been attributed to surface-adsorbed hydroxide groups or carbonates (Fig. 5(h)).54 However, we assign this peak due to oxygen vacancies as we do not observe such a feature in the O-1s spectrum of pure PrFeO3, and an increase in the concentration of Fe2+ emerges after substituting with bismuth (30 mol%).55 The oxygen vacancies are not observed in the pristine sample due to passivation during annealing in air. Further proof of oxygen vacancies in the Bi-substituted sample was gathered from EPR experiments at RT. For comparison, the EPR spectrum of PrFeO3 has also been recorded. A weak EPR signal at a g value of ∼2.24 is noticed in the case of PrFeO3, signifying octahedrally coordinated Fe3+ ions (Fig. S6, ESI†).56 The g value keeps decreasing with increasing Bi content and decreases to 2.17 in the case of Pr0.50Bi0.50FeO3, with a subsequent increase in the intensity of the EPR signal. This decrease in the g-value and increase in the EPR signal intensity indicates a gradual increase of localized oxygen vacancies and enhanced Fe–O hybridization (Fig. S6, ESI†).56,57 Although the increment in linewidth is not uniform, for compositions from x = 0.30–0.50 in Pr1−xBixFeO3, the ΔH is comparatively more compared to the air-annealed samples, corroborating well with the increment in oxygen vacancies, caused possibly by the argon annealing.58
1. The Pr0.70Bi0.30FeO3 sample contains 48 and 52% of Fe2+ and Fe3+, respectively. A nearly 5% rise in Fe2+-concentration was observed in the Pr0.70Bi0.30FeO3 sample, compared to pristine PrFeO3, promoted by the reducing atmosphere employed for the synthesis. Our observation matches the earlier report in which Fe2+ creation due to Bi's inclusion in LaFeO3 is noted.26 Two peaks at binding energies of 543.4 and 545.3 eV in the O-1s spectrum are attributed to the lattice oxygen (low binding energy) and the oxygen vacancies in the sample. Notably, the peak with the binding energy of 545.3 eV has been attributed to surface-adsorbed hydroxide groups or carbonates (Fig. 5(h)).54 However, we assign this peak due to oxygen vacancies as we do not observe such a feature in the O-1s spectrum of pure PrFeO3, and an increase in the concentration of Fe2+ emerges after substituting with bismuth (30 mol%).55 The oxygen vacancies are not observed in the pristine sample due to passivation during annealing in air. Further proof of oxygen vacancies in the Bi-substituted sample was gathered from EPR experiments at RT. For comparison, the EPR spectrum of PrFeO3 has also been recorded. A weak EPR signal at a g value of ∼2.24 is noticed in the case of PrFeO3, signifying octahedrally coordinated Fe3+ ions (Fig. S6, ESI†).56 The g value keeps decreasing with increasing Bi content and decreases to 2.17 in the case of Pr0.50Bi0.50FeO3, with a subsequent increase in the intensity of the EPR signal. This decrease in the g-value and increase in the EPR signal intensity indicates a gradual increase of localized oxygen vacancies and enhanced Fe–O hybridization (Fig. S6, ESI†).56,57 Although the increment in linewidth is not uniform, for compositions from x = 0.30–0.50 in Pr1−xBixFeO3, the ΔH is comparatively more compared to the air-annealed samples, corroborating well with the increment in oxygen vacancies, caused possibly by the argon annealing.58
 | ||
| Fig. 5 (a) Survey spectra, (b)–(d) and (e)–(h) core-level spectra of Bi 4f, Pr 3d, Fe 2p, and O 1s of the PrFeO3 and Pr0.70Bi0.30FeO3 samples, respectively. The letter ‘s’ denotes satellite peaks. | ||
The optical absorbance spectra of the Pr1−xBixFeO3 series (x = 0.00–0.50) were recorded to understand the electronic structural changes brought about by bismuth substitution. The maximum absorption edge for PrFeO3 lies at 628 nm (Fig. S7, ESI†). With the introduction of Bi, a gradual red-shift can be seen in the DRS data. A reddish tinge dominates the color of the sample with increasing Bi inclusion. The O–Bi charge transfer transitions appear in the UV region at around 240 and 390 nm. The band in the visible region (at around 485 nm) may be attributed to the charge transfer from the O2− ion to the Fe ion.28 The bands arising from d–d transitions of octahedrally coordinated Fe3+ ions appear as broad transitions in the 705–680 nm range. The broad and diffused transition at around 975–995 nm signifies the 5T2g → 5Eg transition of Fe2+.59 Both the direct and indirect band gap values estimated using the Kubelka–Munk function analysis decrease marginally with the inclusion of bismuth in PrFeO3 (Fig. 6). The estimated band gap value of PrFeO3 (Edirect = 2.08 eV; Eindirect = 1.99 eV) matches well with the values observed earlier.60 A similar trend is reported earlier in Bi-substituted GdFeO3.28,29
 | ||
| Fig. 6 (a)–(f) Normalized (Kubelka–Munk × energy)n (n = 2 and 1/2) plots of Pr1−xBixFeO3 (x = 0.00–0.50) samples used for determining the direct and indirect band gaps. | ||
We further performed DFT calculations to ascertain the electronic structural changes. To commence with, an examination of the ferromagnetic (FM) and nine additional antiferromagnetic (AFM) spin configurations was done to identify the ground state energy to model the magnetic structure for the pristine composition computationally (Fig. S8, ESI†). These nine AFM configurations included A-, C-, and G-AFM arrangements of Fe. They also account for the spin orientations of Pr atoms positioned within planes at y = 0.25 and y = 0.75, each plane containing 2 Pr atoms. The ground state spin configuration was derived with the Fe atoms arranged in a G-type pattern, whereas the Pr atoms were arranged antiferromagnetically within both of the planes (y = 0.25 and 0.75) but ferromagnetically between the planes y = 0.25 and 0.75 (Fig. S8(h), ESI†). The same configuration gives the ground state structure for GdFeO3 as well.61 Fig. S9 (ESI†) shows the simulated crystal structures of 50% Bi-substituted PrFeO3, generated by adopting a special quasi-random structure (SQS) approach. Since there are 4 Pr atoms in the unit cell, the desired substitution level can be achieved by replacing 2 of them. This substitution results in three possible combinations of atoms. Furthermore, an additional nine possibilities arise considering the spin arrangements for both Pr and Fe atoms. Among these spin configurations, the lowest energy is achieved when Fe is arranged in a G-AFM configuration with both Pr atoms having an up spin (Fig. S9(b), ESI†). With the aforementioned ground state structure, we calculated electronic structure properties for both pristine and Bi-substituted structures while incorporating spin–orbit coupling (SOC), as shown in Fig. 7.
SOC inclusion becomes important when a heavy element such as Bi is present in the system. For the pristine PrFeO3, the valence band maximum (VBM) consists of major contribution from the Pr-f orbitals, and the conduction band minimum (CBM) is occupied by the Fe-d orbitals with moderate contributions from the O-p orbitals in both the VBM and CBM. On the other hand, a Bi-s contribution arises along with the contributions from the Pr-f and O-p orbitals in the doped scenario in the VBM. The band-structure plots reveal the indirect band gaps of pristine and 50% Bi-substituted PrFeO3 are 1.99 eV (between the S–T point) and 1.92 eV (between the X–T point), respectively. A similar decrement in the direct and indirect band gaps is observed experimentally.
The bismuth inclusion has created mixed valence states of Fe (2+ and 3+) and oxygen vacancies, which can significantly modify the superexchange and magnetic interactions, thereby altering the magnetic properties of PrFeO3. Hence, we measured the magnetic properties of pristine PrFeO3 and Bi-substituted samples. The magnetization of Pr1−xBixFeO3 samples (x = 0.00–0.50) under magnetic fields of ±5 T at 298 and 2 K are shown in Fig. 8(a)–(c) and Fig. S10 (ESI†). At RT, PrFeO3 exhibits a hysteresis loop with considerable ferromagnetic character, characterized by Mr = 0.12 emu g−1 and Hc = 0.2 T, aligning well with the earlier report.62 The anisotropy of the Fe sublattice can contribute to the observed Hc at RT. For the Pr0.70Bi0.30FeO3 sample, the magnetization did not pass through the origin and is horizontally shifted (Fig. 8(b)). This indicates a negative exchange bias resulting from the coupling of ferromagnetic and antiferromagnetic interactions in this sample. We observe only weak ferromagnetism at low temperatures (2 K) for PrFeO3 and Bi-substituted samples (x = 0.10–0.50). The unsaturation, accompanied by reduced magnetization, in all these samples at 2 K suggests the presence of competing weak ferromagnetic and antiferromagnetic interactions.63 This decrease in magnetization values with Bi3+ substitution in PrFeO3 at both RT and 2 K can be attributed to the random distribution of Fe3+, Fe2+, and oxygen vacancies, which causes a slight disruption in the antiferromagnetic superexchange interactions, thereby reducing the overall magnetization. The Mr and Hc values again change with increased Bi content in Pr1−xBixFeO3 at 2 K (Table S4, ESI†). The net magnetization in PrFeO3 and the samples substituted with 10, 20, and 30 mol% Bi at 2 K may originate from spin canting due to tilted FeO6 octahedra with varying Fe–O–Fe bond angles, altering the Dzyaloshinsky–Moriya interaction.28 The variation with temperature of the molar susceptibilities of the Pr1−xBixFeO3 samples (x = 0.00–0.50) measured in zero-field-cooled (ZFC) and field-cooled (FC) modes is shown in Fig. 8(d)–(f) and Fig. S11 (ESI†). When these measurements are conducted with a field of 0.1 T, the molar susceptibilities decrease as the temperature increases. Also, the ZFC and FC plots overlap, indicating a typical paramagnetic behavior. The reciprocal of susceptibility (1/χm) under FC conditions at H = 0.1 T for pristine and Bi-substituted PrFeO3 in the 2–300 K temperature range is plotted in Fig. S12 (ESI†). All the samples exhibit Curie–Weiss behavior in the 150–300 K range, and below 150 K, antiferromagnetic interactions become dominant. As a significant negative exchange bias is observed in the Pr0.70Bi0.30FeO3 sample, ZFC and FC measurements at lower fields of 0.02, 0.01, and 0.005 T have been conducted, which can provide insight into the spin reorientation transition (SRT) arising from the anisotropy between the RE3+ and Fe3+ sublattices.45,62 The ZFC and FC data appear bifurcated during these measurements till room temperature (RT). The creation of Fe2+ due to bismuth inclusion and associated oxygen vacancies can bring down the Néel temperature below 700 K.22 A small hump in the ZFC plot at ∼101 K is observed consistently, which can be attributed to the spin reorientation transition temperature (TSR), and it is lower than the TSR reported for PrFeO3 (∼140 K).22
The effect of bismuth inclusion in place of Pr on the electrical properties of PrFeO3 was investigated by first recording the frequency-dependent (0.1 Hz–0.5 MHz range) dielectric responses at RT. Fig. 9(a) shows the dielectric constants of the Pr1−xBixFeO3 (x = 0.10–0.50) samples as function of frequency. The dielectric constant for all the samples attains a high value (at a low-frequency regime) but decreases as the frequency increases and shows frequency-independent behavior (104 Hz onwards). This type of behavior is a general observation of typical dielectrics and is consistent with the combined response of orientational relaxation of dipoles and conduction of charge carriers. At low frequencies, the dipoles get enough time to follow the alternating field, resulting in a higher value of ε′, whereas, at a higher frequency, dipoles cannot follow the field, resulting in a decrease in the value of ε′. Usually, the dipolar and interfacial polarizations contribute to the dielectric constant at low frequencies, whereas the electronic and ionic polarizations are dominant contributors at higher frequencies. It is worth noting that the dielectric constant value increases as the Bi content increases in the sample. This observation matches the trend observed in the Bi-substituted LaFeO3 system.64 Pr0.50Bi0.50FeO3 exhibits the highest value of ε′ (1159) among the Bi-containing samples, which indicates the weakening of the Pr–Fe sublattice interaction, matching well with the trend noticed for Sm0.50Bi0.50FeO3.65
 | ||
Fig. 9 Frequency dependence of (a) the real dielectric component (ε′) and (b) the dielectric loss tangent (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ) of the Pr1−xBixFeO3 samples (x = 0.10–0.50) at RT. δ) of the Pr1−xBixFeO3 samples (x = 0.10–0.50) at RT. | ||
To understand whether the dielectric response of the samples is due to non-linear dielectric/intrinsic nature or by some artefact/extrinsic effect, the collected data are analyzed as per the universal dielectric response (UDR) model.66 According to the UDR model, the relation between conductivity σ′(f) and dielectric constant ε′ can be expressed as:
| σ′(f) = σdc + σ0fs, | (1) |
ε′ = (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ(sπ/2)σ0(T)fs−1)/ε0 = A(T)fs−1, δ(sπ/2)σ0(T)fs−1)/ε0 = A(T)fs−1, | (2) |
| fε′ = A(T)fs, | (3) |
A(T) = (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ(sπ/2)σ0(T)fs−1)/ε0. δ(sπ/2)σ0(T)fs−1)/ε0. |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) f plots. It can be seen that the UDR model is followed in the entire frequency range only by Pr0.90Bi0.10FeO3. For x = 0.20–0.50, the UDR model fits from 10 Hz to 0.5 MHz and deviates at low frequencies. In all cases, the slope lies within the range of 0.06–0.2, indicating close correlation with the UDR model.66 It appears that at very low frequencies, the localized charge carriers accumulate and give rise to Maxwell–Wagner-type interfacial polarization. In conclusion, the substantially high dielectric constant observed in Pr1−xBixFeO3 systems upon incremental bismuth substitution may arise primarily from electron hopping between Fe2+ and Fe3+ leading to dipolar polarization and the oxygen vacancies (acting as charge carriers) existing in the grain boundary regions contributing to the space charge polarizations near/across the boundary regions.67–69Fig. 9(b) shows the variation of tan
f plots. It can be seen that the UDR model is followed in the entire frequency range only by Pr0.90Bi0.10FeO3. For x = 0.20–0.50, the UDR model fits from 10 Hz to 0.5 MHz and deviates at low frequencies. In all cases, the slope lies within the range of 0.06–0.2, indicating close correlation with the UDR model.66 It appears that at very low frequencies, the localized charge carriers accumulate and give rise to Maxwell–Wagner-type interfacial polarization. In conclusion, the substantially high dielectric constant observed in Pr1−xBixFeO3 systems upon incremental bismuth substitution may arise primarily from electron hopping between Fe2+ and Fe3+ leading to dipolar polarization and the oxygen vacancies (acting as charge carriers) existing in the grain boundary regions contributing to the space charge polarizations near/across the boundary regions.67–69Fig. 9(b) shows the variation of tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ as a function of frequency at RT for Pr1−xBixFeO3 (x = 0.00–0.50). The continuous decrease of dielectric loss with an increase in frequency is a general trend noted in the case of rare-earth ferrites.70 The samples with x > 0.20 show a peak-like behavior between 1 and 100 Hz, reflecting the occurrence of relaxation processes.65,70 In the low-frequency region, the increased values of tan
δ as a function of frequency at RT for Pr1−xBixFeO3 (x = 0.00–0.50). The continuous decrease of dielectric loss with an increase in frequency is a general trend noted in the case of rare-earth ferrites.70 The samples with x > 0.20 show a peak-like behavior between 1 and 100 Hz, reflecting the occurrence of relaxation processes.65,70 In the low-frequency region, the increased values of tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ can be due to the conductivity and accumulation of electric charge on the inter-crystalline boundaries and structural defects within the Maxwell–Wagner conduction mechanism framework.71
δ can be due to the conductivity and accumulation of electric charge on the inter-crystalline boundaries and structural defects within the Maxwell–Wagner conduction mechanism framework.71
The local site distortion caused by Bi3+ substitution, together with its stereochemically active lone pair of electrons (6s2), can essentially cause a certain degree of polarization under an applied electric field. The computed spontaneous polarization in the pristine PrFeO3 is almost zero. Further, spontaneous polarization calculations have been carried out utilizing the Berry phase approach for 50% Bi-substituted PrFeO3. The computed value of spontaneous polarization (Ps) comes out to be 0.10 μC cm−2. As a comparative study,72 polarization was calculated at the PBE and PBEsol levels; both yielded the same results. In the Bi-substituted scenario, the electron localization function was also calculated, and a minor charge density shared between the O2− and Bi3+ ions was observed, generating this polar behavior (Fig. 10).
 | ||
| Fig. 10 Electron localization function (ELF) plot for (a) pristine PrFeO3 and (b) 50% Bi-doped PrFeO3. | ||
Encouraged by the theoretical calculation results, P–E loops of Pr1−xBixFeO3 (x = 0.10–0.40) have been measured at RT and are reproduced in Fig. S14 (ESI†). It is known that the present system is a case of non-classical ferroelectrics and expected to exhibit a leaky ferroelectric behavior as these are measured in ceramic pellets.
In the P–E loops of 10 and 20 mol% Bi-substituted PrFeO3 samples, vesica piscis-shaped behavior has been observed. No sharp saturation is noticed in the P–E loops of the samples with x > 0.20 (recorded at a field of 20 kV cm−1). This leaky behavior may be due to the interfacial polarization receiving contributions from the grain boundary effect, oxygen vacancies, and the multivalence of iron. Under these circumstances, the true intrinsic polarization may be masked. To deduce actual ferroelectric behavior in these samples, the C–V loop of the Pr0.60Bi0.40FeO3 sample is measured in the range of −20 V to +20 V at 50 Hz, and the results are presented in Fig. S15(a) (ESI†). The subtle signature of a butterfly-like C–V loop with a pair of peaks (typical for a ceramic possessing ferroelectric characteristics) demonstrates polarization reversal in the sample.15 The butterfly-type shape is the natural result of switching behavior where the maximum occurs near the coercive voltage. To gain further insight into the occurrence of the intrinsic ferroelectric polarization in the sample, the PUND method is employed. The PUND data is collected at RT with a pulse width of 1 ms (Fig. S15(b), ESI†). The measured polarizations during the pulse and in the delay times are designated as P*, 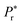 and P∧, P∧r in the primary and secondary pulses, respectively, for both the positive and negative top-up sequences. The total polarization (P*) of 0.778 μC cm−2 includes both switched charge density (QSW) and leakage current, and the second polarization (P∧) of 0.603 μC cm−2 originates mainly from the leakage current. The difference between the remnant polarization from the switching pulse (P*) and the non-switching pulse (P∧) is equivalent to the net switchable polarization (QSW). The true switchable polarization (QSW = P* − P∧) of ∼0.175 μC cm−2 falls in the same range reported earlier in orthoferrite systems.73,74 A saturated P–E loop is obtained for the Pr0.60Bi0.40FeO3 ceramic sample at RT on applying a 20 kV cm−1 field with the maximum Pr (∼0.17 μC cm−2) matching closely to the theoretical value of 0.1 μC cm−2 (Fig. S15(c), ESI†). Hence, the above results reveal that the sample contains some ferroelectric component introduced locally, within the global centrosymmetric structure, by the random distribution of the stereochemically active 6s2 lone pair on Bi3+.13,73,74 Our results demonstrate that including Bi3+ with a stereochemically active lone pair of electrons can be an additional strategy to induce intrinsic and switchable polarization in rare-earth orthoferrites containing magnetic rare-earth ions.
and P∧, P∧r in the primary and secondary pulses, respectively, for both the positive and negative top-up sequences. The total polarization (P*) of 0.778 μC cm−2 includes both switched charge density (QSW) and leakage current, and the second polarization (P∧) of 0.603 μC cm−2 originates mainly from the leakage current. The difference between the remnant polarization from the switching pulse (P*) and the non-switching pulse (P∧) is equivalent to the net switchable polarization (QSW). The true switchable polarization (QSW = P* − P∧) of ∼0.175 μC cm−2 falls in the same range reported earlier in orthoferrite systems.73,74 A saturated P–E loop is obtained for the Pr0.60Bi0.40FeO3 ceramic sample at RT on applying a 20 kV cm−1 field with the maximum Pr (∼0.17 μC cm−2) matching closely to the theoretical value of 0.1 μC cm−2 (Fig. S15(c), ESI†). Hence, the above results reveal that the sample contains some ferroelectric component introduced locally, within the global centrosymmetric structure, by the random distribution of the stereochemically active 6s2 lone pair on Bi3+.13,73,74 Our results demonstrate that including Bi3+ with a stereochemically active lone pair of electrons can be an additional strategy to induce intrinsic and switchable polarization in rare-earth orthoferrites containing magnetic rare-earth ions.
4 Conclusions
PrFeO3 ceramic samples, substituted progressively (up to 50 mol%) with larger-sized Bi3+ (equipped with a stereochemically active lone pair) in place of Pr, have been successfully synthesized by fine-tuning the synthesis conditions. The Fe–O bond lengths elongated with increased Bi content, and the FeO6 octahedra tilt around the pseudocubic [111] direction was retained, possibly due to the repulsions generated by the stereochemically active lone pair on bismuth. Bi's inclusion created local site disorder and promoted the reduction of more Fe3+ to Fe2+, creating higher oxygen vacancies. The randomized distribution of Fe3+, Fe2+, and oxygen vacancies resulting from incremental Bi3+-substitution caused a minor disruption in the antiferromagnetic superexchange interactions, thereby reducing the overall magnetization. At room temperature, a negative exchange bias effect was observed in the Pr0.70Bi0.30FeO3 sample. A spin reorientation transition temperature (TSR) at ∼101 K was observed for the Pr0.70Bi0.30FeO3 sample at lower magnetic fields (below 0.02 T). The room-temperature dielectric constant increased with increasing bismuth content in PrFeO3. The DFT calculations predicted an electric field-induced polarization resulting from the asymmetric charge distribution around Bi3+. The intrinsic and switchable polarization (0.17 μC cm−2) was experimentally verified for the Bi-substituted sample after PUND measurements. The multifunctionality invoked upon Bi3+-incorporated PrFeO3 can be mimicked in other perovskites, forming an effective strategy for exploring untapped functions. The properties exhibited by Bi-substituted PrFeO3 can find practical applicability in magnetically modulated transducers, spintronics, ceramic capacitors, chemical sensors, and optoelectronic devices.Author contributions
Shivangi Rao – methodology, data curation, writing the original draft; Subhajit Sau – methodology, validation, investigation; V. Kanchana – supervision, resources, formal analysis; G. Vaitheeswaran – supervision, resources, formal analysis; Rajamani Nagarajan – supervision, conceptualization, funding acquisition, writing – review and editing.Data availability
The data supporting this study are included within the article and/or the ESI.†Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors acknowledge the financial support by the SERB (Govt. of India) with file no. CRG/2021/000248 and the Institution of Eminence Grant (IoE/2024-25/12/FRP) from University of Delhi to carry out this research work. Shivangi Rao and Subhajit Sau thank the Council Scientific Industrial Research (CSIR) and MHRD Govt. of India for the research fellowship. G. V. would like to thank CMSD and UOH for providing the computational facility and the institute of Eminence, University of Hyderabad (UOH-IOE-RC3-21-675 046), for their financial support. The authors sincerely thank Prof. Arumugum Thamizhavel and Mr Ganesh Jangam of TIFR, Mumbai for their help with the magnetic measurements. We acknowledge National Supercomputing Mission (NSM) for providing computing resources of ‘PARAM Porul’ at NIT Tiruchirappalli, which is implemented by C-DAC and supported by the Ministry of Electronics and Information Technology (MeitY) and Department of Science and Technology (DST), Government of India.Notes and references
- Y. Shimakawa, Perovskite structure compounds, John Wiley & Sons, 2017 Search PubMed.
- R. J. Tilley, Perovskites: Structure-property relationships, John Wiley & Sons, 2016 Search PubMed.
- C. N. R. Rao and B. Raveau, Transition metal oxides: Structure, properties, and synthesis of ceramic oxides, Wiley-VCH, Weinheim, 1998 Search PubMed.
- C. N. R. Rao and J. Gopalakrishnan, New directions in solid state chemistry, Cambridge University Press, 1997 Search PubMed.
- A. K. Jena, A. Kulkarni and T. Miyasaka, Chem. Rev., 2019, 119, 3036–3103 CrossRef CAS.
- K. Y. Tsui, N. Onishi and R. F. Berger, J. Phys. Chem. C, 2016, 120, 23293–23298 CrossRef CAS.
- D. Kan, C. J. Long, C. Steinmetz, S. E. Lofland and I. Takeuchi, J. Mater. Res., 2012, 27, 2691–2704 CrossRef CAS.
- C.-H. Yang, D. Kan, I. Takeuchi, V. Nagarajan and J. Seidel, Phys. Chem. Chem. Phys., 2012, 14, 15953–15962 RSC.
- D. Kan, L. Pálová, V. Anbusathaiah, C. J. Cheng, S. Fujino, V. Nagarajan, K. M. Rabe and I. Takeuchi, Adv. Funct. Mater., 2010, 20, 1108–1115 CrossRef CAS.
- A. Moskvin, Magnetochemistry, 2021, 7, 111 CrossRef CAS.
- R. Saha, A. Sundaresan and C. N. R. Rao, Mater. Horiz., 2014, 1, 20–31 RSC.
- S. Ning, A. Kumar, K. Klyukin, E. Cho, J. H. Kim, T. Su, H.-S. Kim, J. M. LeBeau, B. Yildiz and C. A. Ross, Nat. Commun., 2021, 12, 4298 CrossRef CAS PubMed.
- M. Shang, C. Zhang, T. Zhang, L. Yuan, L. Ge, H. Yuan and S. Feng, Appl. Phys. Lett., 2013, 102, 062903 CrossRef.
- Y. Tokunaga, N. Furukawa, H. Sakai, Y. Taguchi, T.-H. Arima and Y. Tokura, Nat. Mater., 2009, 8, 558–562 CrossRef CAS PubMed.
- J.-H. Lee, Y. K. Jeong, J. H. Park, M.-A. Oak, H. M. Jang, J. Y. Son and J. F. Scott, Phys. Rev. Lett., 2011, 107, 117201 CrossRef.
- C. Zhang, M. Shang, M. Liu, T. Zhang, L. Ge, H. Yuan and S. Feng, J. Alloys Compd., 2016, 665, 152–157 CrossRef CAS.
- Y. Tokunaga, S. Iguchi, T.-h. Arima and Y. Tokura, Phys. Rev. Lett., 2008, 101, 097205 CrossRef CAS.
- B. Rajeswaran, D. Sanyal, M. Chakrabarti, Y. Sundarayya, A. Sundaresan and C. N. R. Rao, Europhys. Lett., 2013, 101, 17001 CrossRef.
- K. Dey, A. Indra, S. Mukherjee, S. Majumdar, J. Strempfer, O. Fabelo, E. Mossou, T. Chatterji and S. Giri, Phys. Rev. B, 2019, 100, 214432 CrossRef CAS.
- N. Aparnadevi, K. Saravana Kumar, M. Manikandan, D. Paul Joseph and C. Venkateswaran, J. Appl. Phys., 2016, 120, 034101 CrossRef.
- H. J. Zhao, W. Ren, Y. Yang, X. M. Chen and L. Bellaiche, J. Phys.: Condens. Matter, 2013, 25, 466002 CrossRef.
- H. J. Zhao, Y. Yang, W. Ren, A.-J. Mao, X. M. Chen and L. Bellaiche, J. Phys.: Condens. Matter, 2014, 26, 472201 CrossRef PubMed.
- E. Martínez-Aguilar, H. Hmŏk, J. Ribas-Ariño, J. M. Siqueiros Beltrones and R. Lozada-Morales, Int. J. Quantum Chem., 2021, 121, e26551 CrossRef.
- O. Rosales-González, F. Sánchez-De Jesús, F. Pedro-García, C. A. Cortés-Escobedo, M. Ramírez-Cardona and A. M. Bolarín-Miró, Materials, 2019, 12, 2054 CrossRef.
- Q. Yao, C. Tian, Z. Lu, J. Wang, H. Zhou and G. Rao, Ceram. Int., 2020, 46, 20472–20476 CrossRef CAS.
- N. Imam, G. Aquilanti, A. Azab and S. E. Ali, J. Mater. Sci.: Mater. Electron., 2021, 32, 3361–3376 CrossRef CAS.
- T. Pikula, J. Dzik, P. Guzdek, M. Kowalczyk, K. Siedliska and E. Jartych, Ceram. Int., 2020, 46, 1804–1809 CrossRef.
- Y. Ma, H. Shen, Y. Fang, H. Geng, Y. Zhao, Y. Li, J. Xu and Y. Ma, Magnetochemistry, 2023, 9, 45 CrossRef CAS.
- K. Bharati, P. R. Tiwari, R. P. Singh, A. Singh, B. C. Yadav, M. P. Singh and S. Kumar, Appl. Nanosci., 2024, 14, 277–289 CrossRef CAS.
- B. H. Toby, J. Appl. Crystallogr., 2001, 34, 210–213 CrossRef CAS.
- P. Hohenberg and W. Kohn, Phys. Rev., 1964, 136, B864 CrossRef.
- W. Kohn and L. J. Sham, Phys. Rev., 1965, 140, A1133 CrossRef.
- P. E. Blöchl, Phys. Rev. B, 1994, 50, 17953 CrossRef.
- G. Kresse and J. Furthmüller, Phys. Rev. B, 1996, 54, 11169 CrossRef CAS.
- G. Kresse and D. Joubert, Phys. Rev. B, 1999, 59, 1758 CrossRef CAS.
- J. P. Perdew, A. Ruzsinszky, G. I. Csonka, O. A. Vydrov, G. E. Scuseria, L. A. Constantin, X. Zhou and K. Burke, Phys. Rev. Lett., 2008, 100, 136406 CrossRef PubMed.
- S. L. Dudarev, G. A. Botton, S. Y. Savrasov, C. J. Humphreys and A. P. Sutton, Phys. Rev. B, 1998, 57, 1505 CrossRef CAS.
- D. Stoeffler and Z. Chaker, J. Magn. Magn. Mater., 2017, 442, 255–264 CrossRef CAS.
- I. Ameri, A. Boularaf, F. Drief, A. Zaoui and S. Kacimi, J. Magn. Magn. Mater., 2021, 537, 168214 CrossRef CAS.
- M. Pugaczowa-Michalska and J. Kaczkowski, J. Mater. Sci., 2015, 50, 6227–6235 CrossRef CAS.
- H. J. Monkhorst and J. D. Pack, Phys. Rev. B, 1976, 13, 5188 CrossRef.
- A. M. Ganose, A. J. Jackson and D. O. Scanlon, J. Open Source Software, 2018, 3, 717 CrossRef.
- R. D. King-Smith and D. Vanderbilt, Phys. Rev. B, 1993, 47, 1651 CrossRef CAS PubMed.
- D. Vanderbilt and R. D. King-Smith, Phys. Rev. B, 1993, 48, 4442 CrossRef CAS PubMed.
- Z. Zhou, L. Guo, H. Yang, Q. Liu and F. Ye, J. Alloys Compd., 2014, 583, 21–31 CrossRef CAS.
- F. A. Mir, M. Ikram and R. Kumar, Vib. Spectrosc., 2011, 55, 307–310 CrossRef CAS.
- E. Traversa, P. Nunziante, L. Sangaletti, B. Allieri, L. E. Depero, H. Aono and Y. Sadaoka, J. Am. Ceram. Soc., 2000, 83, 1087–1092 CrossRef CAS.
- F. Chang, S. Yuan, X. Wang, Y. Cao, B. Lu and S. Cao, Solid State Commun., 2012, 152, 168–172 CrossRef CAS.
- E. Chatla, N. Vankudothu, S. Shravan Kumar Reddy, S. Shanmukharao Smatham, M. Sreenath Reddy, C. Gopal Reddy and P. Yadagiri Reddy, Appl. Phys. A, 2024, 130, 10 CrossRef CAS.
- C.-S. Tu, C.-S. Chen, P.-Y. Chen, Y. L. Hsieh, R. R. Chien, V. H. Schmidt, K.-C. Feng and H.-W. Chang, J. Alloys Compd., 2018, 768, 903–913 CrossRef CAS.
- S. Ward, M. A. Isaacs, G. Gupta, M. Mamlouk and S. S. Pramana, Sustainable Energy Fuels, 2021, 5, 154–165 RSC.
- S. Lütkehoff, M. Neumann and A. Ślebarski, Phys. Rev. B, 1995, 52, 13808 CrossRef.
- G. H. Major, N. Fairley, P. Sherwood, M. R. Linford, J. Terry, V. Fernandez and K. Artyushkova, J. Vac. Sci. Technol., A, 2020, 38, 061203 CrossRef CAS.
- T. J. Frankcombe and Y. Liu, Chem. Mater., 2023, 35, 5468–5474 CrossRef CAS.
- J. Wang, D. N. Mueller and E. J. Crumlin, J. Eur. Ceram. Soc., 2024, 44, 116709 CrossRef CAS.
- B. Jiang, L. Li, Q. Zhang, J. Ma, H. Zhang, K. Yu, Z. Bian, X. Zhang, X. Ma and D. Tang, J. Mater. Chem. A, 2021, 9, 13008–13018 RSC.
- X. Zhang, C. Pei, X. Chang, S. Chen, R. Liu, Z.-J. Zhao, R. Mu and J. Gong, J. Am. Chem. Soc., 2020, 142, 11540–11549 CrossRef CAS PubMed.
- M. Pires, C. Israel, W. Iwamoto, R. Urbano, O. Agüero, I. Torriani, C. Rettori, P. Pagliuso, L. Walmsley and Z. Le, et al. , Phys. Rev. B, 2006, 73, 224404 CrossRef.
- A. Egorysheva, O. Ellert, O. Gajtko, A. Berseneva, Y. V. Maksimov and R. Svetogorov, Ceram. Int., 2016, 42, 13976–13982 CrossRef CAS.
- L. Boudad, M. Taibi, W. Belayachi and M. Abd-Lefdil, J. Solid State Chem., 2022, 309, 122933 CrossRef CAS.
- X.-H. Zhu, X.-B. Xiao, X.-R. Chen and B.-G. Liu, RSC Adv., 2017, 7, 4054–4061 RSC.
- Q. Yang, H. Qin, K. Huang, R. Zhang, L. Yang, Z. Lu, Q. Yao, L. Cheng, C. Huang and Q. Long, et al. , J. Magn. Magn. Mater., 2023, 565, 170274 CrossRef CAS.
- J.-S. Zhou, L. Marshall, Z.-Y. Li, X. Li and J.-M. He, Phys. Rev. B, 2020, 102, 104420 CrossRef CAS.
- D. Triyono, S. Fitria and U. Hanifah, RSC Adv., 2020, 10, 18323–18338 RSC.
- V. L. Mathe, J. Magn. Magn. Mater., 2003, 263, 344–352 CrossRef CAS.
- B. V. Prasad, G. N. Rao, J. Chen and D. S. Babu, Solid State Sci., 2012, 14, 225–228 CrossRef CAS.
- S. Katba, S. Jethva, G. Panchal, R. Choudhary, D. Phase and D. Kuberkar, J. Alloys Compd., 2019, 789, 814–824 CrossRef CAS.
- J. Sheikh, S. A. Acharya and U. P. Deshpande, Mater. Chem. Phys., 2020, 242, 122457 CrossRef CAS.
- D. Sanghvi, H. Boricha, B. Hirpara, S. Solanki, V. Shrimali, A. Joshi, P. Solanki and N. Shah, J. Sol-Gel Sci. Technol., 2020, 93, 666–677 CrossRef CAS.
- R. Shukla, R. Dhaka, S. Dash, S. C. Sahoo, B. Bahera, P. Babu, R. Choudhary and A. K. Patra, et al. , Ceram. Int., 2020, 46, 8730–8744 CrossRef.
- I. Makoed, N. Liedienov, A. Pashchenko, G. Levchenko, D. Tatarchuk, Y. Didenko, A. Amirov, G. Rimski and K. Yanushkevich, J. Alloys Compd., 2020, 842, 155859 CrossRef CAS.
- Y. Zhang, J. Sun, J. P. Perdew and X. Wu, Phys. Rev. B, 2017, 96, 035143 CrossRef.
- A. Panchwanee, V. R. Reddy and A. Gupta, J. Magn. Magn. Mater., 2018, 448, 38–42 CrossRef CAS.
- J. Ramesh, N. Raju, S. S. K. Reddy, M. S. Reddy, C. G. Reddy, P. Y. Reddy, K. R. Reddy and V. R. Reddy, J. Alloys Compd., 2017, 711, 300–304 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Details of synthesis, characterization, refinement results, magnetic configurations of PrFeO3, results of magnetic and ferroelectric measurements. See DOI: https://doi.org/10.1039/d5cp00004a |
| This journal is © the Owner Societies 2025 |






