Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Recent advances in dopant–matrix afterglow systems: high-performance organic afterglow materials and the critical role of organic matrices in materials fabrication
Yue
Zhang†
,
Zhe
Mo†
,
Zi
Ye†
,
Qianqian
Yan
,
Xixi
Piao
,
Jialiang
Jiang
,
Biao
Xu
and
Kaka
Zhang
 *
*
State Key Laboratory of Organometallic Chemistry and Shanghai Hongkong Joint Laboratory in Chemical Synthesis, Key Laboratory of Synthetic and Self-Assembly Chemistry for Organic Functional Molecules, Ningbo Zhongke Creation Center of New Materials, Shanghai Institute of Organic Chemistry, University of Chinese Academy of Sciences, Chinese Academy of Sciences, 345 Lingling Road, Shanghai 200032, People's Republic of China. E-mail: zhangkaka@sioc.ac.cn
First published on 6th May 2025
Abstract
Organic afterglow materials have garnered significant attention due to their long-lived excited states, demonstrating promising applications across diverse fields. Over the past few decades, these materials have experienced rapid development, particularly dopant–matrix systems. This review focuses on the progress made in dopant–matrix organic afterglow materials over the past three years, emphasizing two key aspects: high-performance organic afterglow materials and the critical role of organic matrices in materials fabrication. In the first section, we summarize strategies for enhancing afterglow performance through molecular design, focusing on representative luminescent systems such as benzophenone derivatives, polycyclic aromatic hydrocarbons, and difluoroboron β-diketonate compounds. The second section explores the pivotal functions of organic matrices, including protecting triplet excited states, facilitating intersystem crossing, sensitizing triplet states, and promoting charge separation, which collectively contribute to novel functionalities of afterglow materials. Beyond the molecular design of luminophores, the selection of organic matrices is equally crucial for achieving high-performance afterglow materials and expanding their functionality. This review provides a comprehensive compilation of chemical structures for various organic matrices, serving as a valuable reference for researchers. Given the intricate photophysical processes in organic afterglow systems, we also present experimental methods that support or refute specific mechanisms, providing critical insights for future studies. Overall, dopant–matrix organic afterglow materials represent a highly promising class of luminescent materials. We anticipate their large-scale adoption and high-value applications in real-world scenarios in the near future.
1. Introduction
Luminescent materials play a vital role in applications such as displays, lighting, and analytical detection. Among these, room-temperature phosphorescent (RTP) materials and organic afterglow materials, with emission lifetimes exceeding milliseconds, have demonstrated exceptional performance in oxygen sensing, bioimaging, and anti-counterfeiting, witnessing rapid advancements over the past few decades.1–18 Based on the number of components required for materials fabrication, organic RTP materials can be categorized into single-component, two-component, and multi-component systems. Two-component and multi-component materials are typically prepared using doping methods, with dopant concentrations often below a few weight percent and organic matrices comprising over 90% of the total composition. Consequently, these systems are commonly referred to as dopant–matrix afterglow materials. Five years ago, single-component afterglow materials dominated the reported examples.19–23 However, in the past five years, research into dopant-matrix afterglow materials has expanded significantly, with many research groups shifting their focus from single-component systems to dopant–matrix systems.24–26 This transition has greatly enriched the diversity of dopant–matrix materials and led to the development of various functionalities, as documented in recent literature. Several comprehensive reviews have summarized the progress in dopant–matrix afterglow materials.24–26 Additionally, reviews on polymer-based RTP materials,27–29 supramolecular assembly systems,4,30–32 stimuli-responsive RTP materials,33–35 aqueous-phase RTP materials,36–38 multicolor RTP systems,39,40 functional afterglow materials,41–44 and biomass-based RTP materials45 have also briefly addressed advances in dopant–matrix afterglow systems. However, the examples covered in these reviews predominantly date back two to three years or earlier. Given the substantial progress in dopant–matrix afterglow materials in the past few years, we believe it is timely and essential to systematically organize and summarize these recent findings, thereby contributing to the further advancement of this field.The dopant–matrix system presents several advantages in materials preparation, mechanistic studies, and functional applications (Fig. 1). Its components can be flexibly tailored by selecting or synthesizing various dopants and organic matrices, allowing researchers to obtain diverse samples with minimal synthetic effort, saving both time and costs. Moreover, the system facilitates systematic mechanistic investigations by enabling the modification of specific variables while keeping others constant, providing critical insights into the rational design of high-performance afterglow materials. Additionally, if dopants or organic matrices possess desirable properties—such as high-performance polymers or analytical detection capabilities—the resulting dopant–matrix materials can inherit these functionalities, enhancing their application potential. Furthermore, interactions between dopants and matrices that respond to external physical, chemical, or biological stimuli can result in tunable photophysical properties, enabling the development of intelligent sensing RTP materials with dynamic and responsive capabilities. These features collectively underscore the versatility and value of dopant–matrix systems in advancing room-temperature phosphorescent materials.
Owing to these advantages, the dopant–matrix system has, in recent years, yielded materials with extended luminescence lifetimes, enhanced efficiencies, or unique photophysical properties. Significant progress has been made in the rational design of luminophores and the selection of organic matrices, with some dopant–matrix systems already successfully developed into functional afterglow materials. This review aims to summarize the advancements in dopant–matrix afterglow systems over the past three years. Given the existence of excellent recent reviews on supramolecular afterglow systems,4,30–32 this work excludes discussions on supramolecular host–guest systems. This review focuses on two major aspects. The first is high-performance organic afterglow materials, introducing several representative luminophore systems, including benzophenone derivatives, coronene, triphenylene, pyrene, and difluoroboron β-diketonates. By emphasizing these systems, we aim to provide concrete examples of systematic strategies for controlling the excited-state properties of organic luminophores, with the hope of distilling universally applicable methodologies extendable to other luminescent systems. We also discuss emerging materials, such as TADF-type afterglow materials, organic long persistent luminescence (OLPL) systems, and narrowband afterglow emitters. The second aspect is the critical role of organic matrices in materials fabrication. Organic matrices typically provide crystalline or glassy rigid environments that suppress molecular motion of triplet excited states, thereby reducing nonradiative decay (knr). Crystalline and some polar polymer matrices can inhibit oxygen diffusion, preventing quenching of triplet states by oxygen (kq). Additionally, the triplet energy levels (T1) of organic matrices can act as intermediaries to facilitate intersystem crossing (ISC) in luminophores. Certain matrices can reduce the singlet–triplet energy gap and enhance ISC or reverse ISC (RISC) via dipole–dipole interactions with singlet excited states of luminophores. Matrices that efficiently form triplet excited states upon excitation can sensitize dopants through energy transfer. In organic long persistent luminescence systems, the HOMO–LUMO alignment between matrices and dopants enables photo-induced intermolecular charge transfer and separation, with subsequent slow charge recombination leading to afterglow emissions lasting minutes to hours. In dopant–matrix OLPL systems based on two-photon ionization, organic luminophores absorb two photons to ionize, forming radical cations, while organic matrices capture free electrons as electron traps. Slow charge recombination in these systems generates OLPL emissions. This review integrates these recent findings to provide insights into the rational design and applications of dopant–matrix afterglow systems, fostering future advancements in this rapidly evolving field.
In dopant–matrix afterglow systems, the design of luminophores is essential for achieving high-performance materials, while the choice of organic matrices significantly influences afterglow properties, making their selection equally critical. This review not only highlights advancements in this field but also incorporates guidelines for selecting suitable organic matrices in relevant examples. Additionally, we provide a list of commonly used organic matrices, complete with their chemical structures and CAS numbers, to facilitate further research by potential investigators. Given the complexity of photophysical processes in organic afterglow systems, elucidating afterglow mechanisms constitutes a crucial aspect of research. To this end, this review includes experimental approaches designed to exclude certain mechanisms and validate others, offering a comprehensive framework for understanding the underlying principles of various dopant–matrix afterglow materials.
As the dopant–matrix organic afterglow systems are undergoing rapid development, this review cannot comprehensively cover all advancements in this field over the past three years. Instead, we focus on two key aspects: high-performance afterglow materials and the critical role of organic matrices. The specific content is outlined as follows.
2. High-performance organic afterglow materials
2.1 Benzophenone-containing luminescent dopants
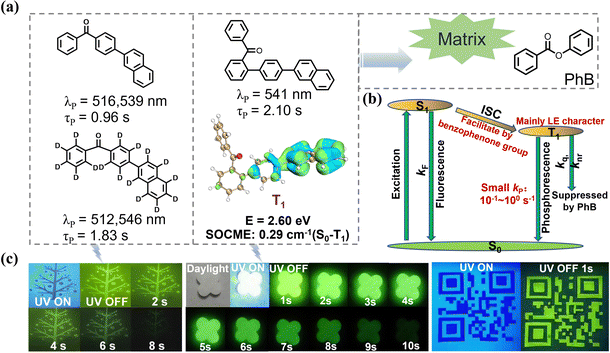
Benzophenone is a classic organic phosphorescent molecule, with its S1 state exhibiting n–π* character, resulting in a relatively small fluorescence emission rate constant. The T2 excited state of benzophenone is very close in energy to the S1 state and exhibits π–π* character.2,20 According to the energy gap rule and the El-Sayed rule, the intersystem crossing (ISC) rate constant from S1 to T2 is significantly enhanced, resulting in an ISC efficiency approaching 100%. The resulting T2 excited state rapidly undergoes internal conversion to the T1 state, which has n–π* character, and the phosphorescence emission rate constant in the T1 state is on the order of 103 s−1.2 In organic systems, this large phosphorescence rate constant aids in harvesting T1 excited state energy; however, it also implies that even when nonradiative decay and oxygen quenching are sufficiently suppressed, the phosphorescence lifetime is limited to the millisecond range. This presents a challenge for the development of long persistent afterglow materials.To enhance the phosphorescence lifetime of the benzophenone system, one feasible approach is to introduce substituents, such as aromatic groups with significant π–π* transition properties, into the benzophenone framework. This modification facilitates the mixing of 3π–π* and 3n–π* states, which weakens the n–π* character of the T1 excited state and reduces the phosphorescence emission rate constant as reported by Tang and Shuai.46,47 In a recent study, researchers introduced a series of common aromatic substituents at the para position of benzophenone.48 The resulting luminescent molecules showed no room-temperature afterglow in their single-component powder or crystal state. However, when doped into a phenyl benzoate matrix, the dopant–matrix material exhibited phosphorescence lifetimes of several hundred milliseconds. Phenyl benzoate effectively protected the T1 excited state under ambient conditions. Notably, the naphthalene-substituted benzophenone molecule in phenyl benzoate exhibited a phosphorescence lifetime approaching one second (Fig. 2). After deuteration, where C–D vibrations are significantly weaker than C–H vibrations, the knr of the T1 excited state is further suppressed, resulting in a room-temperature phosphorescence lifetime of 1.8 seconds.48 In TD-DFT calculations, the researchers found that the T1 excited state of naphthalene exhibits typical localized excitation characteristics and a lower T1 energy level. Therefore, the naphthalene group significantly contributes to the formation of the T1 excited state in the naphthalene-substituted benzophenone molecule, imparting typical localized excitation features. This results in a reduction of the phosphorescence emission rate constant by about three orders of magnitude, which explains the second-level phosphorescence lifetime of the system.
In further studies, the phenyl-naphthalene group was covalently attached to different substitution positions on the benzophenone group.49 Among the isomers obtained, the ortho-substituted molecule, when doped into phenyl benzoate, exhibited a remarkable room-temperature phosphorescence lifetime of 2 seconds (Fig. 2). TD-DFT calculations revealed that the T1 excited state of the ortho-substituted molecule is predominantly composed of the phenyl-naphthalene functional group. Additionally, the ortho-substitution-induced steric hindrance significantly increased the dihedral angle between the benzophenone backbone and the phenyl-naphthalene substituent. As a result, the n–π* transition character of the T1 excited state in the ortho-substituted molecule was further weakened, leading to a reduction in the phosphorescence emission rate constant and thereby extending the phosphorescence lifetime of the system. In a very recent study conducted by Tang and coworkers,46b near-infrared phosphorescent materials have been obtained by balancing the n–π* and π–π* transitions in benzophenone derivative systems; the benzophenone matrix was used to enhance the RTP performance of the near-infrared materials.
In the benzophenone and its derivative systems, the presence of n–π* transitions generally results in a relatively high spin–orbit coupling matrix element (SOCME), leading to efficient intersystem crossing. In some cases, the steady-state and delayed fluorescence spectra of dopant–matrix materials overlap, both exhibiting phosphorescence emission bands. To confirm that the long afterglow originates from the T1 excited state of the luminescent dopant, it is common to measure the delayed spectra of the dopant–matrix material at 77 K as well as the delayed spectra of the dopant at 77 K. If the two spectra show similar peak shapes and peak positions to the room-temperature delayed spectrum of the dopant–matrix material, it can be concluded that the room-temperature long afterglow originates from the T1 excited state of the luminescent dopant.
2.2 Polycyclic aromatic hydrocarbon luminescent dopants
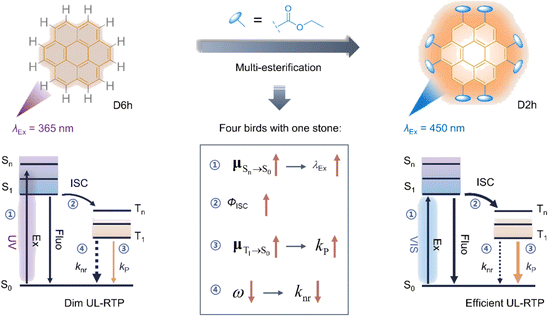
Among polycyclic aromatic hydrocarbons, coronene is a particularly unique luminescent molecule due to its D6h symmetry. The symmetry of its S1 excited state is forbidden for fluorescence, resulting in a fluorescence emission rate constant of 106–107 s−1.50–52 In rigid environments, its fluorescence lifetime can extend to several hundred nanoseconds. The T1–S0 transition of coronene is both spin-forbidden and symmetry-forbidden, with a phosphorescence emission rate constant in the 10−2 s−1 range, meaning that under conditions where non-radiative decay and oxygen quenching are effectively suppressed, the phosphorescence lifetime can reach tens of seconds.50–52 PMMA (polymethyl methacrylate) can serve as the matrix for coronene, as its rigid glassy state helps suppress non-radiative decay of the T1 state. Under anoxic conditions, the quenching rate constant for the T1 state can also be reduced, allowing for phosphorescence lifetimes exceeding ten seconds at room temperature. Coronene and its derivatives have been doped into materials such as β-estradiol, 4-methoxybenzophenone, polyvinyl alcohol, polycarbonate, polyvinylpyridine, ABS (acrylonitrile–butadiene–styrene), and even SBS (styrene–butadiene–styrene), resulting in long afterglow emissions.53 These coronene-based long-lived systems are used as afterglow donors, and when suitable fluorescent acceptors are incorporated, the system can achieve long-wavelength afterglow emission through excited state energy transfer. Coronene and its derivatives typically exhibit yellow-green phosphorescence, and to ensure efficient energy transfer, the fluorescent acceptor is often selected for good absorption in the yellow-green region. The efficiency of the energy transfer can be estimated by the shortening of the afterglow donor's phosphorescence lifetime.Recently, researchers have synthesized several ester-substituted coronene derivatives through a multi-step synthesis route (Fig. 3).54 These coronene derivatives exhibit enhanced solubility or dispersibility in organic solvents and polymer matrices. Additionally, the introduction of ester groups can enhance the intersystem crossing (ISC) process, altering the symmetry of the coronene system. This change leads to a red shift in absorption and an increase in luminescence quantum yield. The esterification process does not affect the rigidity or large planar aromatic structure of coronene, allowing the derivatives to retain their second-level organic room-temperature phosphorescence properties. This multi-esterification strategy has successfully led to high-performance, visible-light-excited, ultra-long organic room-temperature phosphorescent materials in pure organic amorphous materials.
Some applications of organic afterglow materials are in aqueous or physiological systems, making the preparation of water-dispersible organic afterglow materials an important research topic. The typical approach is to first select a system with excellent afterglow properties in the solid state and then process the solid afterglow material into a water-dispersible afterglow system using mechanical grinding, strong ultrasound, or nanoprecipitation methods.36–38 However, after these treatments, the rigid environment that originally protected the T1 excited state is often disrupted, leading to a significant reduction in room-temperature afterglow lifetime and efficiency. Leveraging the flexibility of the dopant–matrix system, researchers have proposed a method to prepare long-lifetime afterglow materials through emulsion polymerization.55 In this process, coronene or deuterated coronene is dissolved in a monomer, and after deoxygenation, emulsion polymerization is carried out. Once the monomer polymerizes into a polymer, the coronene molecules dissolved in it become encapsulated in the polymer latex particles. Using methyl methacrylate (MMA) for emulsion polymerization, the resulting PMMA latex particles provide a glassy environment that effectively suppresses non-radiative decay of the T1 state at room temperature. Moreover, as the emulsion polymerization occurs in an oxygen-free environment, the resulting water-dispersible solution exhibits a phosphorescence emission with a lifetime of up to 10 seconds at room temperature. This method avoids the disruption of the rigid environment commonly seen in traditional methods, effectively protecting the T1 excited state. Additionally, the latex particle size is controllable, ensuring long-term dispersion in water without precipitation.
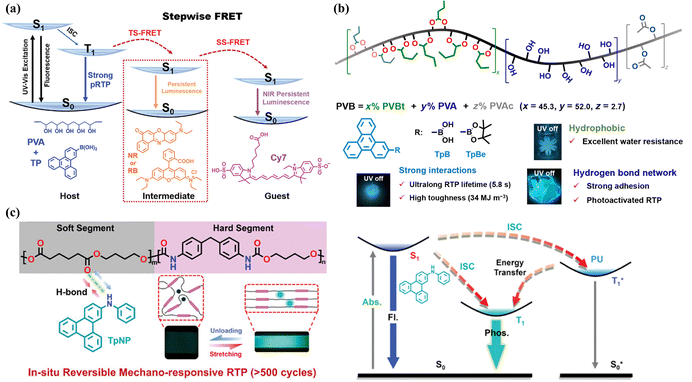
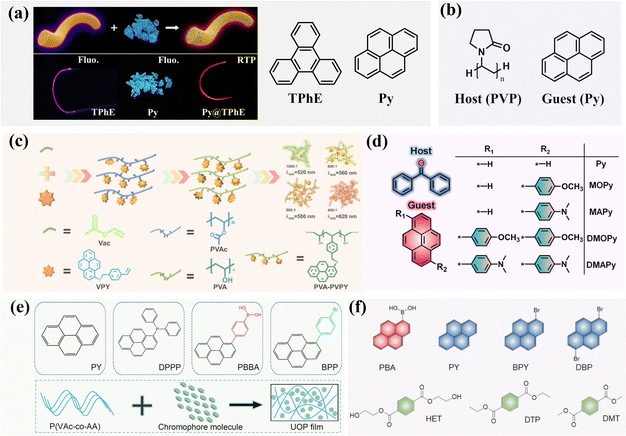
Similar to the coronene system, the T1–S0 transition in the triphenylene system is spin-forbidden and symmetry-forbidden. Therefore, when the triphenylene system is well protected in the T1 state, it can exhibit second-level phosphorescence lifetimes. When triphenylene boronic acid (triphenylene-2-ylboronic acid) is doped into a polyvinyl alcohol (PVA) matrix (Fig. 4a), the resulting dopant–matrix sample displays a room-temperature phosphorescence lifetime of 3.29 seconds.56 PVA is a transparent polymer that provides a rigid environment for the luminescent molecules, and it has a strong oxygen barrier capability. In a further study, Nile red and Cyanine 7 were doped into the triphenylene boronic acid–PVA system, achieving a two-step excitation energy transfer process that resulted in near-infrared (NIR) room-temperature afterglow materials, with an emission at 810 nm and a lifetime of 0.2 seconds.56 Additionally, when triphenylene boronic acid was doped into a polyvinyl butyral (PVB) resin matrix (Fig. 4b), the researchers observed a room-temperature phosphorescence lifetime of 5.82 seconds, and this system could function as a binder for anti-counterfeiting labels.57 Interestingly, when triphenylene-based aromatic secondary amines were doped into the polyurethane (PU) elastomer (Fig. 4c), the resulting dopant–matrix material exhibited mechano-responsive properties, with its phosphorescence brightness, quantum yield, and phosphorescence lifetime correlating well with the applied stress.58
Pyrene is also an excellent building block for constructing room-temperature phosphorescent materials. Dopant–matrix systems containing pyrene are characterized by a rich variety of afterglow colors, which can be tuned by controlling molecular aggregation and selecting excitation wavelengths, allowing for different afterglow colors, including red and even near-infrared phosphorescence.59–65Fig. 5 lists the chemical structures of these pyrene-containing luminescent molecules. Pyrene derivatives with polar functional groups or hydrogen-bonding donor/acceptor properties disperse well in polar matrices like polyvinyl alcohol (PVA), and the degree of molecular aggregation can be controlled by the doping concentration.59 Hydrophobic pyrene derivatives can be dispersed in matrices like benzophenone (Fig. 5d),60 and the resulting dopant–matrix afterglow materials can be ultrasonically processed using surfactants to achieve dispersion in water. Based on this approach, researchers have successfully developed near-infrared room-temperature phosphorescent materials dispersed in water, enabling deep-tissue biological imaging with minimal background interference.
2.3 Difluoroboron β-diketonate luminescent dopants
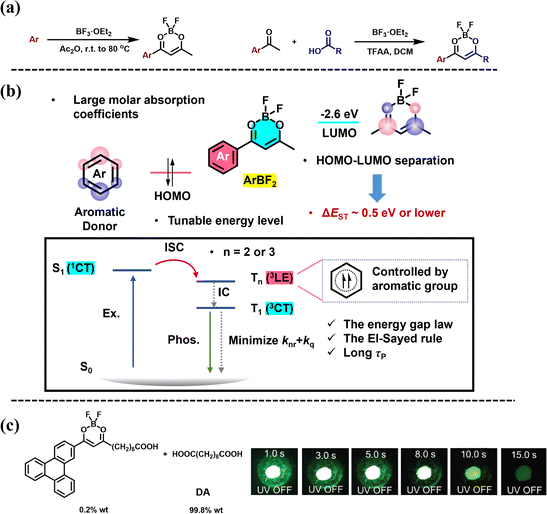
Difluoroboron β-diketonate (BF2bdk) molecules are an interesting class of luminescent compounds, typically characterized by excited-state properties involving intramolecular charge transfer transitions.19,66–71 These molecules often exhibit high molar absorption coefficients and high photoluminescence quantum yields. Fraser et al. were the first to report the room-temperature phosphorescence properties of difluoroboron β-diketonate–polymer systems.19 In their work, the difluoroboron β-diketonate group was covalently bonded to the polymer, creating a single-component system that required multiple steps for materials synthesis. In recent years, there has been increasing research on small-molecule difluoroboron β-diketonate–matrix binary systems.66–71 This progress can be attributed to the flexibility of the dopant–matrix approach, as well as the cascade reaction developed by Zhang et al. for the one-step synthesis of difluoroboron β-diketonate small molecules (Fig. 6a).67–69 Essentially, a typical chemist can synthesize a difluoroboron β-diketonate molecule in one day using the cascade reaction and subsequently incorporate it into various commercial small-molecule matrices. This enables the preparation of multiple dopant–matrix materials and the observation of their afterglow properties within a day. This approach has significantly advanced the preparation of high-performance difluoroboron β-diketonate–matrix materials and the study of their photophysical mechanisms.After synthesizing approximately 300 difluoroboron β-diketonate molecules and screening hundreds of organic matrices, researchers have obtained several high-performance organic room-temperature afterglow materials. In some high-performance afterglow systems, the structure of the luminescent molecule is fixed, while the properties of the organic matrix are adjusted. It was found that the organic matrix not only protects the T1 excited state of difluoroboron β-diketonate but also promotes the formation of the T1 excited state through dipole effects (as discussed later).70 Through the analysis and summarization of data on the afterglow systems of difluoroboron β-diketonate molecules, researchers have revealed their role in binary afterglow systems (Fig. 6b).69,71 First, difluoroboron β-diketonate molecules have large molar absorption coefficients, which give them strong light absorption capabilities. Secondly, these molecules feature intramolecular charge transfer transitions, with a HOMO–LUMO separation and a ΔEST of about 0.5 eV or less, which facilitates intersystem crossing.71 The LUMO of difluoroboron β-diketonate molecules is primarily determined by the dioxaborine functional group, with an energy level of around −2.6 eV, while the HOMO is mainly determined by the aromatic group. Common aromatic groups such as anisole, biphenyl, fluorenyl, and naphthyl have HOMO levels ranging from −6.4 eV to −5.8 eV. As a result, the S1 energy level of the difluoroboron β-diketonate molecule is around 3.0 eV, which coincides with the 3LE (LE represents localized excitation) energy levels of the aromatic groups. The Tn energy level formed by the aromatic group's 3LE in the difluoroboron β-diketonate molecule is close to S1, with a significant difference in the type of excited state.69 According to the energy gap rule and the El-Sayed rule, the intersystem crossing rate from S1 to Tn is significantly enhanced, promoting the formation of the T1 state. Based on this understanding, researchers can select the appropriate aromatic group based on its HOMO and 3LE energy levels to connect with the difluoroboron β-diketonate functional group. In the triphenylene-containing BF2bdk molecule system, they achieved the brightest room-temperature BF2bdk afterglow material to date, which continues to emit a white afterglow for up to 5 seconds after the excitation source is turned off (Fig. 6c).69
2.4 TADF-type organic afterglow materials
In the dopant–matrix systems of difluoroboron β-diketonates and small-molecule matrices, researchers have designed difluoroboron β-diketonate molecules and utilized the organic matrix to modulate the excited-state properties of these molecules. This has led to the development of a new class of thermally activated delayed fluorescence (TADF) materials with room-temperature afterglow lifetimes of up to 300 milliseconds and afterglow quantum yields as high as 60–70% (Fig. 7).72 It is important to note that neither of the two components exhibits room-temperature afterglow properties when used individually. By deuterating the luminescent molecules, molecular vibrations are reduced, which suppresses non-radiative decay of the excited state. In deuterated TADF-type afterglow material systems, the afterglow efficiency is further enhanced.73 TADF-type organic afterglow systems are characterized by moderate reverse intersystem crossing rate constants (kRISC, 100–102 s−1), which significantly improve the utilization of triplet excited states while maintaining the room-temperature afterglow properties of organic materials. This provides a new pathway for constructing efficient organic room-temperature afterglow materials. The moderate RISC rate constant is a distinguishing feature of these systems, in contrast to the much higher RISC rate constants (103–106 s−1) typically used in OLED-based TADF molecules.74 In this system with a moderate RISC rate constant, other processes that deplete the triplet excited state must be suppressed. Crystalline organic matrices are ideal for protecting the triplet excited state, resulting in room-temperature long afterglow materials. In TADF-type afterglow systems, the reverse intersystem crossing rate is much larger than the phosphorescence rate at room temperature, showing TADF signals in the delayed emission spectra without significant RTP signals. If kRISC and kP are similar at room temperature, both TADF and RTP signals are prominent in the delayed spectra, indicating a TADF/RTP dual-emission afterglow material.75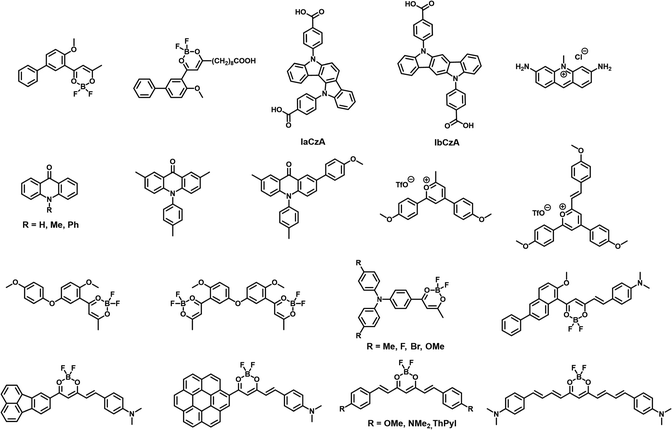 | ||
| Fig. 7 Molecular structure of selected TADF-type afterglow dopants and molecules with TADF/RTP dual emission behaviors. | ||
In the literature, the majority of TADF molecules exhibit emission lifetimes ranging from microseconds to milliseconds, and even the emission behavior with lifetimes in the tens of milliseconds has often been used to rule out the TADF mechanism.76 To address this discrepancy, researchers have further strengthened the characterization methods for TADF-type afterglow materials. Specifically, several approaches77 have been employed to investigate the triplet-state behavior of TADF materials: temperature-dependent delay emission experiments, which study the behavior of the triplet excited state, show that as the temperature increases, reverse intersystem crossing and delayed fluorescence are enhanced; TD-DFT theoretical calculations, which are used to study the singlet and triplet excited state energy levels, the nature of the excited states, and the intersystem crossing channels, providing insight into the mechanisms involved; oxygen-dependence experiments, which help explore the properties of the triplet excited state, as the presence of oxygen can quench triplet states and influence afterglow behavior; the linear dependence of delayed fluorescence intensity on excitation light intensity, which supports the TADF afterglow mechanism, as it demonstrates the expected relationship for a TADF process; temperature-dependent experiments, where the change in emission lifetimes with temperature, combined with the calculation of phosphorescence rate constants and reverse intersystem crossing rate constants, allows for the fitting of singlet–triplet splitting energies; and low-temperature studies, where when materials are excited under ultraviolet light at liquid nitrogen temperatures and then the excitation is turned off, materials with only triplet excited states are obtained. These materials emit green afterglow, and when placed in hot water, the afterglow color shifts to blue. The blue emission originates from the singlet excited state, providing a direct demonstration of the reverse intersystem crossing from triplet to singlet states as the temperature increases.73 Additionally, in the short-wavelength region of the emission spectrum, where only the singlet excited state emission is present and there is no interference from triplet emission, lifetime measurements still show emission lifetimes exceeding 100 ms, further confirming the presence of TADF-type organic afterglow materials.77 These comprehensive experiments offer a more robust validation of the TADF-type afterglow mechanism.
Regarding the design of TADF-type organic afterglow materials, the kRISC of luminescent dopants at room temperature or higher should fall in the range of 100–102 s−1, while this kRISC should be much larger than kP by around one order of magnitude or higher. Since such systems with moderate/modest kRISC and small kP of triplet excited states can be readily quenched, a protective environment provided by the organic matrix to reduce knr and kq is required for TADF-type afterglow materials design.
Because kRISC decreases with lower temperatures, while kP remains temperature-insensitive, the afterglow colors or spectral properties of TADF afterglow materials or TADF/RTP dual-emission materials are sensitive to temperature changes. When IaCzA and IbCzA (Fig. 7) are doped into PVA, the delayed spectra reveal that the resulting material exhibits both TADF and RTP dual-emission at room temperature.75a The TADF emission band is in the 400–470 nm range, while the RTP emission band spans 480–600 nm. As the temperature increases, the proportion of TADF in the delayed spectra increases, the afterglow color changes significantly, and the afterglow lifetime decreases substantially. This material can thus be used as a temperature-sensitive anti-counterfeiting material. A very recent study showed that a coronene system in a melamine formaldehyde polymer matrix exhibited TADF/RTP dual emission and showed afterglow color change by temperature control. This system has a TADF lifetime of up to around 1 second above 100 °C.75b
In the field of organic afterglow, the vast majority of reported materials require UV light excitation, sometimes even higher energy UVB light. In contrast, in commercially available fluorescent dyes, there are many visible-light-excitable fluorescent molecules to choose from. Through screening, researchers discovered that doping commercial acriflavine into a PVA matrix resulted in a thin film exhibiting TADF afterglow properties.78 Due to the absorption capacity of acriflavine in the visible light region, the TADF afterglow film can be excited by white light, making it suitable for use as an afterglow anti-counterfeiting label. Furthermore, researchers found that the TADF-type organic afterglow system can also be extended to pyrylium salts,79 acridone,80 and other compounds (Fig. 7).
To address the issue of low efficiency in long-wavelength room-temperature afterglow materials, researchers have applied charge transfer technology and introduced a phenyl linkage design along with the strong electron-donating triphenylamine functional group (Fig. 7).81 The resulting difluoroboron β-diketonate, when doped into a 4-methoxybenzophenone matrix, exhibits yellow TADF-type afterglow with a quantum yield of 42.8% and an afterglow lifetime of 375 ms. Further enhancing the electron-donating ability of the triphenylamine functional group led to the development of a TADF-type afterglow material with an emission wavelength of 600 nm. By designing molecules with different electron-donating functional groups and increasing the conjugation of the donor groups, the doped difluoroboron β-diketonate molecules in the 4-methoxybenzophenone matrix resulted in an orange-red TADF afterglow material with a quantum efficiency of 56.3% and an afterglow lifetime of 313 ms.82 Difluoroboron β-diketonate materials containing fluorene functional groups exhibit room-temperature phosphorescence, while extending the conjugation length of the molecular structure causes spectral redshift and TADF afterglow with a fluorescence lifetime of 100 ms and a red TADF afterglow efficiency of 13%.83 The introduction of the hemicurcuminoid structure into the boron difluoride system results in red TADF room-temperature afterglow materials.84 Additionally, by using a curcuminoid-based BF2bdk molecule and increasing the conjugation of the donor functional groups, near-infrared emitting TADF afterglow materials with an emission wavelength of 723 nm can be achieved.85
2.5 Organic long persistent luminescent systems
Organic long persistent luminescence (OLPL) materials are characterized by their ability to emit light for extended periods, ranging from minutes to hours or even longer.86–101 Early studies showed that a two-component system with N,N,N′,N′-tetramethylbenzidine (TMB) doped into a PMMA matrix could exhibit hour-long OLPL afterglow at 20 K through a two-photon ionization mechanism.86 When TMB was doped into a PPT (2,8-bis(diphenylphosphoryl)dibenzo[b,d] thiophene) acceptor in a two-component system, the material demonstrated OLPL at room temperature in an inert atmosphere.87 Upon single-photon or normal excitation light, charge separation occurred, resulting in the formation of TMB cation free radicals and PPT anion free radicals. The subsequent slow charge recombination led to the emission of light with power law decay, lasting for hours. Further studies reported that CzPhAP molecules (a TADF molecule) doped into PPT, TPBi (2,2′,2′′-(1,3,5-benzinetriyl)-tris(1-phenyl-1-H-benzimidazole)), or PMMA, when irradiated with high-power light, could absorb two photons and undergo two-photon ionization.88 The released electrons were captured by the surrounding matrix, forming charge-separated states. Later, CzPhAP molecules harvested energy from the recombination of charges, resulting in both singlet and triplet state emissions and exhibiting OLPL. These studies, along with other reported OLPL research studies, indicate that the charge separation and slow charge recombination processes are essential for the long-lasting photoluminescence observed in organic materials.The performance of OLPL materials is closely related to the various processes involved in their photophysical mechanisms. These processes include the degree of charge separation, where high-power excitation light sources are often needed to irradiate the sample for extended periods in two-photon ionization systems, promoting charge separation.88 In donor–acceptor OLPL systems, careful selection of components can ensure that the excited state energy levels of the molecular charge transfer are lower than the T1 levels of both the donor and acceptor, helping achieve a good degree of charge separation.89,90 Another important factor is the stabilization or protection of radical intermediate species. In two- or three-component OLPL systems, at least two or three radical intermediates are involved, which are highly susceptible to deactivation. If these intermediates deactivate before charge recombination occurs, the OLPL properties will significantly deteriorate. Conversely, if the stability of the radical intermediates is higher, this is crucial for high-performance OLPL materials. It has been reported that inert atmospheres and encapsulation can prevent quenching by oxygen and water vapor, protecting the reactive species in the intermediate state.87 Crystalline matrices have also been shown to effectively protect these intermediates, enabling the development of high-performance OLPL materials, including those dispersed in water.91,92 The kinetics of charge recombination also plays a key role. The addition of electron traps in n-type systems and hole traps in p-type systems can control the kinetics of charge recombination, significantly extending the OLPL duration.87,93,94 Temperature and external fields have also been reported to influence the dynamics of charge recombination.95,96 Finally, the photoluminescence quantum yield (PLQY) of luminescent molecules is crucial. Charge recombination produces singlet and triplet excitons, and the ability of luminescent molecules to harvest these states is key. The stronger the ability to harvest singlet and triplet excitons, the higher the brightness and longer the duration of the OLPL.88,96 Therefore, enhancing the PLQY of the luminescent molecules is a key factor in improving the performance of OLPL materials.
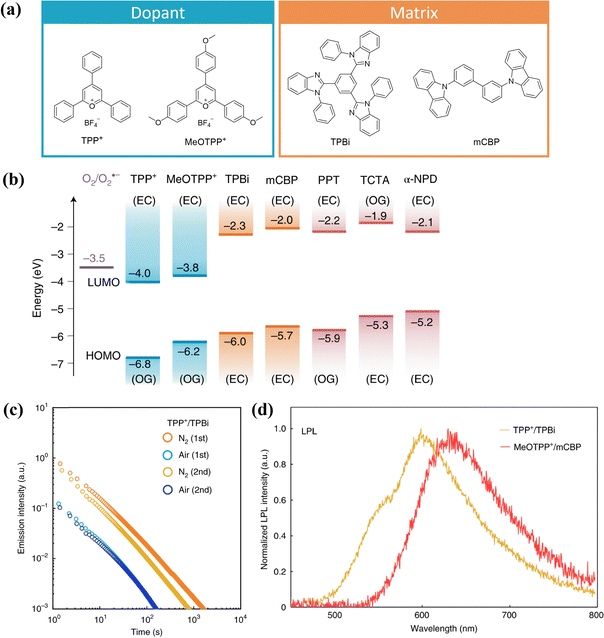
Currently, most OLPL systems reported in the literature are n-type systems, which are highly sensitive to oxygen. For example, the OLPL duration of the TMB-PPT system is on the order of hours in an inert atmosphere, but it significantly decreases when exposed to air.87 To address this issue, researchers have developed p-type OLPL systems, utilizing commercial compounds such as pyrylium salts and TPBi or 3,3′-di(9H-carbazol-9-yl)biphenyl (mCBP) as matrices (Fig. 8).93 The resulting dopant–matrix OLPL materials exhibit excellent OLPL properties under ambient conditions (Fig. 8c). Moreover, the emission wavelengths of these materials can extend into the red or near-infrared regions, making them promising for various practical applications (Fig. 8d).
In OLPL systems, the addition of fluorescent components can act as traps to prolong the OLPL duration. These components also serve as energy acceptors for charge recombination, enhancing the afterglow brightness and tuning the afterglow color.94 In some systems, incorporating multi-resonant TADF dopants allows the absorption of visible light to sensitize donor–acceptor OLPL systems, giving them visible light excitation properties.97 Some polymer-based systems, through doping and melt-processing, can also achieve OLPL materials with durations on the order of hours. Researchers synthesized a carbazole derivative (CBBU) doped into a polyethylene terephthalate (PET) matrix, yielding a transparent polymer film with an afterglow lifetime of up to 11 hours.98 By attaching electron donor and acceptor functional groups to the main chain of the polymer molecules, they achieved uniform dispersion of the donor and acceptor, solving the issue of aggregation and phase separation typically found in p-type OLPL systems.99 Specifically, using free radical polymerization, a copolymer was prepared with a small amount of electron-accepting 4-(4-(acryloyloxy)phenyl)-2,6-bis(4-methoxyphenyl)-pyrylium tetrafluoroborate (ATPP) monomer and a large amount of electron-donating 2-(9H-carbazol-9-yl)ethyl methacrylate (EMCz) monomer. Thin films were fabricated via a deposition method, and compared to simple molecular-doped films; the polymer films showed uniformity and excellent phase stability. This strategy led to higher-performance near-infrared LPL emission, which remained stable even under air exposure and at higher temperatures. The near-infrared emission property suggests potential applications in biological imaging.99
The methods for preparing OLPL materials mainly rely on melt casting, which requires high temperatures, and sometimes solution casting, which is time-consuming and involves large amounts of organic solvents. Mild and solvent-free methods for OLPL materials remain rarely reported. In a recent study, researchers used pyrylium salts as photocatalysts to induce the polymerization of vinyl carbazole, resulting in pyrylium salt-polyvinyl carbazole (PVK) materials that exhibited OLPL properties.100 This preparation method does not require organic solvents or heating to temperatures of 200–300 °C, and the photopolymerization reaction occurs rapidly, with the material forming just a few seconds after light exposure. Furthermore, by selecting various compounds with HOMO energy levels higher than that of PVK (such as N,N,N′,N′-tetrakis(4-methylphenyl)-benzidine), these compounds were used as hole traps and incorporated into the pyrylium salt–PVK system, leading to an improvement in the OLPL performance.
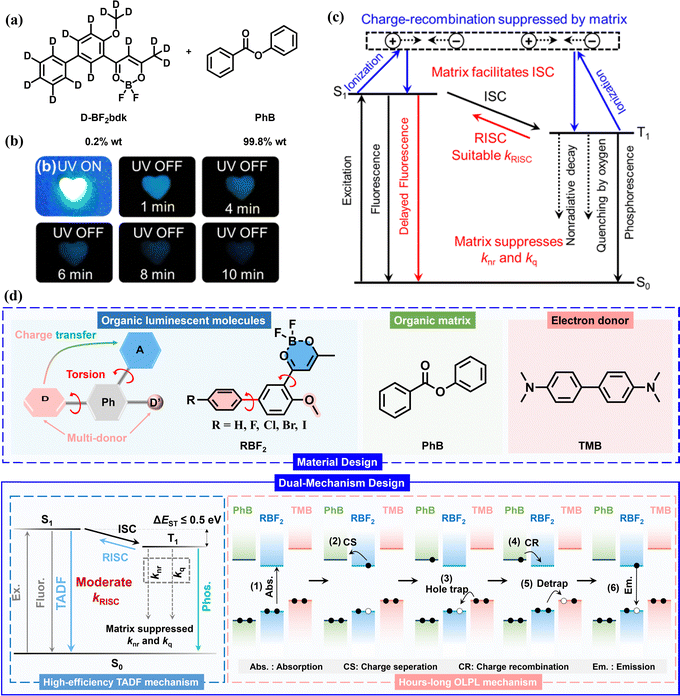
In the difluoroboron β-diketonate–phenyl benzoate system, in addition to the TADF-type organic afterglow, under strong excitation conditions, an organic long persistent photoluminescence phenomenon was observed (Fig. 9).73 The emission originates from the singlet excited state, and the double logarithmic decay plot is linear, conforming to a power law decay, indicating a charge separation–slow recombination OLPL mechanism. In this system, upon excitation, difluoroboron β-diketonate molecules absorb two photons consecutively, resulting in ionization and the formation of free radical cations, with free electrons being captured by the matrix (Fig. 9c). In the solid-state medium, electrons and holes slowly recombine, producing long-lasting emission lasting for tens of minutes. When the TMB dopant was further added to the difluoroboron β-diketonate–phenyl benzoate system, the resulting three-component material exhibited significant OLPL properties under normal or weak excitation light sources (Fig. 9d).101 In the three-component system, after excitation, difluoroboron β-diketonate molecules form long-lived excited-state species. Their LUMO electrons have a certain probability of entering the LUMO orbitals of phenyl benzoate, and the HOMO electrons of TMB enter the HOMO orbitals of difluoroboron β-diketonate, promoting charge separation and the formation of phenyl benzoate radical anions and TMB radical cations. Charge recombination occurs on difluoroboron β-diketonate molecules, utilizing the TADF properties of difluoroboron β-diketonate to simultaneously harvest singlet and triplet excitons generated during recombination (Fig. 9d). This three-component system possesses both TADF-type afterglow and OLPL long-lasting luminescence mechanisms, with the two mechanisms coexisting in the same material, achieving the difficult task of efficiently preparing long-lifetime materials in this field.
In research on OLPL systems, some specific mechanisms are still not fully understood. The well-known and effective electron acceptors are PPT and TPBi. In recent work, a dopant–matrix material with TPBi as the matrix and TADF luminescent molecules as dopants was prepared, and the resulting material exhibited an OLPL lifetime of up to 24 hours (Fig. 10).96 The researchers proposed that the trap depth in the system is determined by the energy difference between the LUMO levels of the dopant and the matrix. By adjusting the composition, the trap depth can be controlled, thus regulating the properties of the OLPL material. Additionally, some ester-based crystalline small molecules or polymers can also serve as electron acceptors. The electron donors are typically aromatic amines, some of which can also act as hole traps to enhance OLPL performance. In two-photon ionization or other mechanisms, TADF luminescent molecules are necessary. Studies have demonstrated the importance of long-lifetime luminescent molecules in these mechanisms. Firstly, in two-photon ionization mechanisms, long-lifetime luminescent molecules have a higher efficiency in absorbing two photons consecutively. Secondly, in other mechanisms, the charge separation process involves electron transfer, and long-lifetime luminescent molecules are essential as the starting point or bridge for electron transfer, leading to higher charge separation efficiency. Lastly, TADF luminescent molecules can simultaneously harvest singlet and triplet excitons generated during charge recombination, resulting in more efficient energy utilization.
2.6 Organic narrowband afterglow systems
Organic RTP and afterglow materials with narrowband emission hold great potential for applications such as multiplexed bioimaging, where they reduce spectral interference among luminescent probes, and high-density information encryption, addressing the challenge of spectral congestion. Nevertheless, current research predominantly features RTP and afterglow systems with wide emission spectra, often exhibiting full-width at half-maximum (FWHM) exceeding 50 nm, and in some cases approaching 100 nm.BODIPYs are a class of narrowband fluorescence molecules with high absorption coefficients and PLQYs, and they are commercially available. The absorption and emission wavelengths of BODIPY molecules can be freely selected over a broad range. Researchers have incorporated BODIPY into organic room-temperature phosphorescent materials that are spectrally matched (Fig. 11a).102 Through excited state energy transfer, they have achieved narrowband afterglow materials with a FWHM of 23 nm. The spectrum and lifetime of the system can be tuned by adjusting the BODIPY and the RTP donor. In another study, researchers selected benzophenone as a room-temperature phosphorescent donor and doped it with narrowband emitting molecules (Fig. 11b), resulting in a narrowband phosphorescent material with a FWHM of 30 nm at room temperature.103 Furthermore, in polymer-based phosphorescent systems (Fig. 11c), the introduction of narrowband emitting molecules also enables the preparation of narrowband afterglow materials through excited-state energy transfer, with a lifetime of 1.64 s and a FWHM of 38 nm.104
The narrowband afterglow systems mentioned above are based on excited state energy transfer, while intrinsic narrowband organic afterglow is challenging to achieve. Analyzing the excited state properties of luminescent molecules, it is evident that the broadening of emission spectra in reported RTP/afterglow systems is mainly caused by two factors: (1) structural relaxation of excited states, particularly in luminescent systems with intramolecular charge transfer characteristics and (2) vibronic coupling between excited states and ground states. Recent studies on organic light-emitting diodes (OLEDs) have shown that the multiple resonance effect in organic systems can result in localization of the HOMO and the LUMO on different atoms, suppressing vibronic coupling between excited states and ground states, thus yielding narrowband emission.105 Based on the multiple resonance effect, a variety of multi-resonant TADF emitters were synthesized to display narrowband emission and high color purity, suitable for high-quality, energy-saving OLED devices. These multi-resonant TADF emitters exhibit a reverse intersystem crossing rate (kRISC) of 103–106 s−1, which is necessary to prevent efficiency roll-off in OLED devices at high luminance. However, in the context of afterglow emitters, a large kRISC of 103–106 s−1 corresponds to a short TADF lifetime (10−6 to 10−3 s), which would be detrimental to afterglow materials designed for emission lifetimes longer than 100 ms. Therefore, a moderate or modest kRISC (100 to 101 s−1) in multi-resonant systems is proposed to be important to achieving narrowband multi-resonant TADF-type organic afterglow materials with an emission lifetime greater than 100 ms. The multiple resonance effect of acridone can weaken vibronic coupling, and the rigidity of its molecular skeleton can suppress excited-state relaxation. Based on these two structural characteristics, researchers have doped acridone into a phenyl benzoate matrix to obtain a dopant–matrix TADF room-temperature afterglow material with a FWHM of less than 40 nm (Fig. 11d).80,106 Additionally, using emulsion polymerization and the dopant–matrix strategy, narrowband afterglow nanoparticles that are stably dispersed in water can also be achieved. Recently, researchers have synthesized a series of indolo[3,2,1-kl]phenothiazine derivatives by molecular cyclization to suppress excited-state relaxation (Fig. 11e) and doped them into suitable polymer matrices, resulting in a dopant–matrix narrowband afterglow material with a FWHM of 36 nm.107
3. Critical role of organic matrices in materials fabrication
In addition to the design of luminescent molecules, the selection of organic matrices is crucial for the development of high-performance and functional afterglow materials. Fig. 12 and 13 illustrate the chemical structures and CAS numbers of commonly used organic matrices.The basic role of organic matrices is to disperse luminescent molecular dopants. Several methods are commonly employed for this purpose. First, the matrix and luminescent molecules are dissolved together, and after the solvent evaporates, the dopant–matrix material is obtained. Second, the melt-casting method involves heating the matrix to its melting point, allowing it to dissolve or disperse the luminescent molecules more effectively. After cooling, the dopant–matrix material is formed. Third, the grinding method involves mechanically blending the matrix and luminescent molecules, allowing the latter to disperse within the matrix. During grinding, low-boiling solvents may be added to facilitate better dispersion of the luminescent molecules. Lastly, in situ formation involves using a liquid precursor of the organic matrix, which can dissolve the luminescent molecules. Through polymerization or other conversion processes, the matrix solidifies, encapsulating the luminescent molecules within.
Organic matrices are typically either crystalline or amorphous small molecules or polymers, and their rigid environments help suppress the intramolecular motion of dopant's triplet excited state, thereby reducing non-radiative deactivation at room temperature (Fig. 14a). For amorphous small molecules, such as PMMA, oxygen molecules are allowed to diffuse relatively freely within their structures. As a result, when these afterglow materials are prepared in an inert gas environment, they will immediately exhibit afterglow upon excitation. However, when prepared under ambient conditions, the materials typically require UV or visible light irradiation for a period of time to deplete the oxygen molecules, which are consumed by the triplet excited states formed, thus activating the materials to display their afterglow properties. In contrast, crystalline small-molecule matrices and certain polar polymers, such as PVA, effectively inhibit oxygen diffusion, preventing contact between the dopant's triplet excited states and oxygen molecules, thereby suppressing oxygen quenching. As a result, afterglow samples produced with these matrices either do not require activation or only need a brief activation (a few seconds) before exhibiting afterglow.
In RTP and TADF afterglow systems, the organic matrices typically possess structures with relatively low conjugation, which results in high T1 energy levels. This is essential because, upon excitation of dopant–matrix samples, the T1 excited state of the luminescent molecules may undergo energy transfer to the T1 state of the matrix, leading to afterglow quenching. Studies have reported that if the T1 energy level of the organic matrix is sufficiently high, it can prevent this energy transfer, thus contributing to the development of high-performance organic afterglow materials.108–111
In addition to the aforementioned scenario, the T1 energy level of the organic matrix plays a crucial role in afterglow performance through other mechanisms. In some luminescent molecular systems where the energy gap between the S1 and T1 states is large, researchers have proposed that introducing an organic matrix with a T1 energy level located between the S1 and T1 of the luminescent molecule can facilitate matrix-mediated intersystem crossing, thereby enabling the creation of dopant–matrix afterglow materials (Fig. 14b).112 It has been observed that by adjusting the T1 energy level of the organic matrix, the afterglow lifetime of the dopant–matrix system can be controlled, leading to the development of lifetime-encoded afterglow anti-counterfeiting materials.113 A very recent study further discussed the role of the matrix's T1 state in the afterglow system.114
In luminescent molecular systems of intramolecular charge transfer (ICT) character, researchers have found that upon excitation, the S1 state of the luminescent molecule exhibits a significant dipole moment. By selecting an organic matrix with a pronounced ground-state dipole moment, a dipole–dipole interaction between the S1 state of the luminescent molecule and the organic matrix can be established. This interaction effectively lowers the S1 energy level while having minimal impact on the T1 energy level, thereby reducing the ΔEST (Fig. 14c).67,70,72 This facilitates both intersystem crossing (ISC) and reverse intersystem crossing (RISC), which are advantageous for the preparation of high-performance afterglow materials. According to estimates, a 0.1 eV reduction in ΔEST can increase the ISC/RISC rate by approximately 100 times.115 This dipole effect on enhancing ISC has also been supported by the literature.116 In practice, researchers have accumulated experience in selecting matrices for ICT-based afterglow systems. When the T1 energy level of the luminescent molecule is high, matrices with a high T1 energy level, such as phenyl benzoate, are preferred, as they promote ISC while preventing energy transfer from the luminescent molecule's T1 state to the matrix's T1 state, which could lead to afterglow quenching. On the other hand, if the T1 energy level of the luminescent molecule is lower, matrices like benzophenone or 4-methoxybenzophenone are more suitable, as these matrices have a stronger dipole moment than phenyl benzoate enhancing the ISC/RISC process. Moreover, they are effective at dispersing ICT luminescent molecules, enabling higher doping concentrations and resulting in dopant–matrix materials with higher afterglow brightness. In systems where benzophenone and its derivatives are used as matrices, excited state energy transfer from the matrix to the dopant may lead to afterglow. To test whether there is energy transfer, experimentally, a longer excitation wavelength is chosen to selectively excite luminescent dopants, avoiding the absorption region of the matrix. If there is still significant afterglow by long-wavelength excitation, one can confirm that the luminescent molecules' own ISC and phosphorescence decay processes are sufficient to produce afterglow, without relying on energy transfer from the matrix to the luminescent molecules.
Excited state energy transfer is an effective method for preparing dopant–matrix afterglow materials (Fig. 14d).117–120 By selecting an efficient ISC organic matrix in combination with an organic luminescent dopant that effectively accepts T1 energy from the matrix—particularly those with small kP values—the resulting dopant–matrix materials exhibit efficient and long-lived afterglow properties.117 Additionally, excited state energy transfer serves as a strategy for preparing long-wavelength afterglow materials.118 By selecting spectrally matched long afterglow donor materials and long-wavelength luminescent acceptors, doping the luminescent acceptors into the long afterglow donor materials enables the fabrication of afterglow materials in the red to near-infrared regions, which are typically difficult to achieve using direct methods.56,120
In OLPL systems based on the donor–acceptor mechanism, the organic matrix not only plays a role in matching the HOMO/LUMO levels with the dopants but can also function as an electron trap or hole trap. In fact, there are numerous donor–acceptor pairs, and what kind of specific pair that leads to effective performance is still under investigation. Currently, it is known that PPT and TPBi are promising matrices,87 and some long-lived OLPL materials have been successfully developed using these matrices. Furthermore, if certain components can form relatively stable radical intermediates during charge separation, this significantly impacts the performance of OLPL materials. For instance, the TMB component can form a stable TMB radical cation.87 Some ester-based small molecule or polymer matrices,86,98,101 when combined with suitable luminescent molecules, can also yield OLPL materials, possibly through the two-photon ionization mechanism or other undefined mechanisms. However, the exact role of the organic matrix in these OLPL systems is not yet fully understood. Further systematic research is required to elucidate the matrix's contribution in OLPL systems, which is crucial for the development of high-performance materials.
The organic matrix has a significant influence on afterglow properties. In certain systems, the change in the type of organic matrix can result in alterations in the color of room-temperature phosphorescence.121–123 In a difluoroboron β-diketonate system containing a triphenyl group, researchers observed different phosphorescent colors depending on the matrix used.121 According to Kasha's exciton model, the T1 excited state in organic systems exhibits minimal dipole moment in its transition to the ground state, making the T1 energy level (NOT phosphorescence intensity) insensitive to the microenvironment. Once the molecular structure is determined, the T1 energy level is essentially fixed. The triphenyl group-containing difluoroboron β-diketonate molecule, which is linked by multiple single bonds, exhibits flexibility and adopts different conformations in various matrices. These conformational changes lead to variations in its T1 energy level, resulting in different phosphorescent colors in different polymer matrices, making it a potential probe for label-free polymer phase separation in afterglow applications.121
Small structural changes in the organic matrix can lead to significant alterations in the afterglow mechanism of dopant–matrix materials.122,123 In a recent study, researchers doped a luminescent molecule containing triphenylamine and triphenyl groups into a benzophenone matrix, achieving dopant–matrix afterglow materials with the RTP afterglow mechanism.123 Interestingly, when a fluorinated benzophenone matrix was used, intermolecular charge transfer occurred between the dopant and the matrix, leading to the formation of a TADF-type organic afterglow material.
The responsiveness of organic afterglow materials to external mechanical stimuli has been reported in the literature, primarily in single-component systems.33–35 Recent studies have demonstrated the response of afterglow in dopant–matrix materials to ultrasound.124 Afterglow materials based on fluorinated benzophenone, prepared via the melt-drop emulsification method, were found to form dispersions in water. Researchers discovered that after subjecting these dispersions to ultrasonic treatment, the afterglow properties were significantly weakened. This was attributed to the ultrasound disrupting the fluorinated benzophenone microcrystals, which in turn compromised the protection of the dopant's T1 state. This ultrasound-responsive behavior was also extended to long-wavelength TADF afterglow materials in water-dispersible systems, highlighting their potential as smart, responsive afterglow materials.124
When the organic matrix is a polymer, the resulting polymer-based room-temperature afterglow material exhibits excellent processing, dispersion, and mechanical properties.27–29 Conventional methods for preparing polymer-based afterglow materials include solution casting and melt molding. Solution casting requires a large amount of solvent and slow evaporation, while melt molding necessitates high temperatures that may damage certain luminescent molecules. In contrast, some researchers have opted for solvent-free, mild photo-initiated bulk polymerization.125–127 In this method, a small amount of phosphorescent molecules is dissolved in the liquid monomer, and after exposure to light, the material is cured to form polymer-based room-temperature afterglow materials with excellent processability and flexibility.125 The residual monomer is less than 0.2% after polymerization, and the presence of photoinitiator residues does not affect the afterglow performance.
In previous reports, polymers with a glass transition temperature (Tg) higher than room temperature have often been used as matrices because they can suppress molecular motion. Researchers developed flexible RTP materials by doping 4-biphenylboronic acid into polyvinylidene fluoride (PVDF) with a Tg of −27.1 °C, achieving for the first time RTP emission with a long lifetime of 1275.7 ms in a low-Tg polymer (Fig. 15a).128 The RTP performance can be further tuned by adjusting the crystallinity and various polymorphs (α, β, and γ phases). The low Tg of the PVDF matrix allows segmental motion of the polymer under microwave radiation. Upon exposure to 2.45 GHz microwave radiation, the crystallinity of the polymer decreases, and the fraction of the α phase increases, resulting in an RTP material with microwave-stimulated responsiveness (Fig. 15b).
 | ||
| Fig. 15 (a) Illustration of BPBA in PVDF. (b) The crystalline modifications and phase transformation of BPBA/PVDF films with crystalline regions and different polymorph microenvironments. | ||
Through emulsion polymerization and the dopant–matrix strategy, a small amount of luminescent molecules is dissolved in monomer droplets, and after polymerization, the monomers are transformed into a glassy polymer matrix.129–131 This rigid microenvironment protects the T1 state, resulting in afterglow emulsions with lifetimes comparable to bulk materials. The emulsions are stable, with high solid content, uniform size, and adjustable properties. By copolymerizing in emulsions and performing specific ligand surface modification, afterglow microspheres were obtained with the capability to specifically recognize proteins in aqueous dispersion.129 Utilizing the stability of luminescent molecules of all aryl structures, afterglow emulsions were developed, which are resistant to acids and bases and are biocompatible, demonstrating functionality for microenvironmental oxygen detection.130 TADF-type afterglow emulsions with dual responses to temperature and oxygen were also constructed.131 In these emulsion systems, as long as the luminescent molecules are in a nanometer-scale glassy microenvironment, knr can be effectively suppressed. This concept challenges the conventional belief that afterglow can only be observed in bulk, powder, or film samples. Extending this idea, it can be envisioned that in certain multicomponent polymer systems, if one component can protect the T1 state of the organic luminescent molecules and induce afterglow, the selection of other components becomes relatively flexible, which is beneficial for the development of functional afterglow materials. Researchers have blended phosphorescent polymers into a polydimethylsiloxane (PDMS) matrix to create a range of room-temperature phosphorescent elastomers (Fig. 16).132 After repeated stretching of the elastomer, it was found that its phosphorescence intensity and lifetime only decreased slightly, indicating excellent resistance to mechanical deformation. This property promotes the practical application of RTP materials in wearable optoelectronic devices, flexible displays, and advanced anti-counterfeiting technologies.
4. Concluding remarks and outlook
This review summarizes the recent advancements in the field of dopant–matrix organic phosphorescence over the past three years, from the perspective of high-performance materials and the role of the matrix. With the increasing number of research publications and the deepening of studies, strategies for designing luminescent molecules have gradually been mastered. From the n–π* systems represented by benzophenone to the π–π* systems of polycyclic aromatic hydrocarbons, and then to the intramolecular charge transfer systems exemplified by difluoroboron β-diketonate compounds, various methods for achieving high-performance afterglow materials have been identified. Research over the years has also increasingly highlighted the crucial role organic matrices play in the manipulation of excited states, such as protecting the T1 state, promoting ISC/RISC, and forming donor–acceptor OLPL systems. The dopant–matrix system consists of many slow processes, and because organic matrices can suppress knr and kq in the T1 state of luminescent molecules to very low values, some traditionally overlooked slow processes in conventional photophysics may emerge or even dominate in afterglow systems at room temperature. This could lead to unprecedented photophysical phenomena, which are of significant importance for the development of photophysics and photochemistry, as well as for the development and application of new photofunctional materials. Given that inorganic afterglow materials have already found large-scale applications, organic afterglow materials outperform their inorganic counterparts in flexibility, processability, sustainability, and transparency. Therefore, the successful enhancement of organic materials’ performance and large-scale production will represent a key milestone in the real-world applications of organic afterglow materials. Achieving these future prospects will require intense effort from researchers and close collaboration with industry, but it is believed that they will be realized in the near future.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Conflicts of interest
The authors declare no competing financial interest.Acknowledgements
We thank the financial support from the National Natural Science Foundation of China (22475228 and 22175194), the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB0610000), Hundred Talents Program from the Shanghai Institute of Organic Chemistry (Y121078), Pioneer Hundred Talents Program of the Chinese Academy of Sciences (E320021), and Ningbo Natural Science Foundation (2023J243).References
- V. W.-W. Yam, V. K.-M. Au and S. Y.-L. Leung, Light-Emitting Self-Assembled Materials Based on d8 and d10 Transition Metal Complexes, Chem. Rev., 2015, 115, 7589–7728 CrossRef CAS PubMed.
- W. Zhao, Z. He and B. Z. Tang, Room-temperature phosphorescence from organic aggregates, Nat. Rev. Mater., 2020, 5, 869–885 CrossRef CAS.
- N. Gan, H. Shi, Z. An and W. Huang, Recent Advances in Polymer-Based Metal-Free Room-Temperature Phosphorescent Materials, Adv. Funct. Mater., 2018, 28, 1802657 CrossRef.
- X. Ma, J. Wang and H. Tian, Assembling-Induced Emission: An Efficient Approach for Amorphous Metal-Free Organic Emitting Materials with Room-Temperature Phosphorescence, Acc. Chem. Res., 2019, 52, 738–748 CrossRef CAS PubMed.
- S. Hirata, Recent Advances in Materials with Room-Temperature Phosphorescence: Photophysics for Triplet Exciton Stabilization, Adv. Opt. Mater., 2017, 5, 1700116 CrossRef.
- A. Forni, E. Lucenti, C. Botta and E. Cariati, Metal free room temperature phosphorescence from molecular self-interactions in the solid state, J. Mater. Chem. C, 2018, 6, 4603–4626 RSC.
- Kenry, C. Chen and B. Liu, Enhancing the performance of pure organic room-temperature phosphorescent luminophores, Nat. Commun., 2019, 10, 2111 CrossRef CAS PubMed.
- M. Singh, K. Liu, S. Qu, H. Ma, H. Shi, Z. An and W. Huang, Recent Advances of Cocrystals with Room Temperature Phosphorescence, Adv. Opt. Mater., 2021, 9, 2002197 CrossRef CAS.
- T. Zhang, X. Ma, H. Wu, L. Zhu, Y. Zhao and H. Tian, Molecular Engineering for Metal-Free Amorphous Materials with Room-Temperature Phosphorescence, Angew. Chem., Int. Ed., 2020, 59, 11206–11216 CrossRef CAS PubMed.
- W. Jia, Q. Wang, H. Shi, Z. An and W. Huang, Manipulating the Ultralong Organic Phosphorescence of Small Molecular Crystals, Chem. – Eur. J., 2020, 26, 4437–4448 CrossRef CAS PubMed.
- S. Hirata, Molecular physics of persistent room temperature phosphorescence and long-lived triplet excitons, Appl. Phys. Rev., 2022, 9, 011304 CAS.
- H. Gao and X. Ma, Recent progress on pure organic room temperature phosphorescent polymers, Aggregate, 2021, 2, e38 CrossRef CAS.
- Q. Li and Z. Li, Molecular Packing: Another Key Point for the Performance of Organic and Polymeric Optoelectronic Materials, Acc. Chem. Res., 2020, 53, 962–973 CrossRef CAS PubMed.
- X. Yang, G. I. N. Waterhouse, S. Lu and J. Yu, Recent advances in the design of afterglow materials: mechanisms, structural regulation strategies and applications, Chem. Soc. Rev., 2023, 52, 8005–8058 RSC.
- S. Cai, X. Yao, H. Ma, H. Shi and Z. An, Manipulating intermolecular interactions for ultralong organic phosphorescence, Aggregate, 2023, 4, e320 CrossRef CAS.
- Y. Zhu, X. Wang and M. Wu, Intriguing Room Temperature Phosphorescence in Crystalline Porous Organic Frameworks, Adv. Funct. Mater., 2023, 33, 2308096 CrossRef CAS.
- L. Ma and X. Ma, Recent advances in room-temperature phosphorescent materials by manipulating intermolecular interactions, Sci. China: Chem., 2023, 66, 304–314 CrossRef CAS.
- T. Yang, Y. Li, Z. Zhao and W. Z. Yuan, Clustering-triggered phosphorescence of nonconventional luminophores, Sci. China: Chem., 2023, 66, 367–387 CrossRef CAS.
- G. Zhang, G. M. Palmer, M. W. Dewhirst and C. L. Fraser, A dual-emissive-materials design concept enables tumour hypoxia imaging, Nat. Mater., 2009, 8, 747–751 CrossRef CAS PubMed.
- W. Z. Yuan, X. Y. Shen, H. Zhao, J. W. Y. Lam, L. Tang, P. Lu, C. Wang, Y. Liu, Z. Wang, Q. Zheng, J. Z. Sun, Y. Ma and B. Z. Tang, Crystallization-Induced Phosphorescence of Pure Organic Luminogens at Room Temperature, J. Phys. Chem. C, 2010, 114, 6090–6099 CrossRef CAS.
- Z. Yang, Z. Mao, X. Zhang, D. Ou, Y. Mu, Y. Zhang, C. Zhao, S. Liu, Z. Chi, J. Xu, Y. Wu, P. Lu, A. Lien and M. R. Bryce, Intermolecular Electronic Coupling of Organic Units for Efficient Persistent Room-Temperature Phosphorescence, Angew. Chem., Int. Ed., 2016, 55, 2181–2185 CrossRef CAS PubMed.
- Z. Yang, C. Xu, W. Li, Z. Mao, X. Ge, Q. Huang, H. Deng, J. Zhao, F. L. Gu, Y. Zhang and Z. Chi, Boosting the Quantum Efficiency of Ultralong Organic Phosphorescence up to 52% via Intramolecular Halogen Bonding, Angew. Chem., Int. Ed., 2020, 59, 17451–17455 CrossRef CAS PubMed.
- X.-F. Wang, H. Xiao, P.-Z. Chen, Q.-Z. Yang, B. Chen, C.-H. Tung, Y.-Z. Chen and L.-Z. Wu, Pure Organic Room Temperature Phosphorescence from Excited Dimers in Self-Assembled Nanoparticles under Visible and Near-Infrared Irradiation in Water, J. Am. Chem. Soc., 2019, 141, 5045–5050 CrossRef CAS PubMed.
- S. Guo, W. Dai, X. Chen, Y. Lei, J. Shi, B. Tong, Z. Cai and Y. Dong, Recent Progress in Pure Organic Room Temperature Phosphorescence of Small Molecular Host–Guest Systems, ACS Mater. Lett., 2021, 3, 379–397 CrossRef CAS.
- X. Yan, H. Peng, Y. Xiang, J. Wang, L. Yu, Y. Tao, H. Li, W. Huang and R. Chen, Recent Advances on Host–Guest Material Systems toward Organic Room Temperature Phosphorescence, Small, 2022, 18, 2104073 CrossRef CAS PubMed.
- J. Li, G. Wang, X. Chen, X. Li, M. Wu, S. Yuan, Y. Zou, X. Wang and K. Zhang, Manipulation of Triplet Excited States in Two-Component Systems for High-Performance Organic Afterglow Materials, Chem. – Eur. J., 2022, 28, e202200852 CrossRef CAS PubMed.
- J. Guo, C. Yang and Y. Zhao, Long-Lived Organic Room-Temperature Phosphorescence from Amorphous Polymer Systems, Acc. Chem. Res., 2022, 55, 1160–1170 CrossRef CAS PubMed.
- X. Dou, X. Wang, X. Xie, J. Zhang, Y. Li and B. Tang, Advances in Polymer-Based Organic Room-Temperature Phosphorescence Materials, Adv. Funct. Mater., 2024, 34, 2314069 CrossRef CAS.
- B. Ding, X. Ma and H. Tian, Recent Advances of Pure Organic Room Temperature Phosphorescence Based on Functional Polymers, Acc. Mater. Res., 2023, 4, 827–838 CrossRef CAS.
- H. Nie, Z. Wei, X.-L. Ni and Y. Liu, Assembly and Applications of Macrocyclic-Confinement-Derived Supramolecular Organic Luminescent Emissions from Cucurbiturils, Chem. Rev., 2022, 122, 9032–9077 CrossRef CAS.
- X.-Y. Dai, M. Huo and Y. Liu, Phosphorescence resonance energy transfer from purely organic supramolecular assembly, Nat. Rev. Chem., 2023, 7, 854–874 CrossRef CAS.
- H. Zheng, Z. Zhang, S. Cai, Z. An and W. Huang, Enhancing Purely Organic Room Temperature Phosphorescence via Supramolecular Self-Assembly, Adv. Mater., 2024, 36, 2311922 CrossRef CAS.
- G. Ye, Y. Yang, W. Yuan, J. Gu, S. Li, Q. Li and Z. Li, Multistimuli-Responsive Room-Temperature Phosphorescence: Adjustable Electrostatic Interactions and the Corresponding Accurate Molecular Packing, ACS Mater. Lett., 2024, 6, 4639–4648 CrossRef CAS.
- Q. Zhou, C. Yang and Y. Zhao, Dynamic organic room-temperature phosphorescent systems, Chem, 2023, 9, 2446–2480 CAS.
- Y. Lei, W. Dai, G. Li, Y. Zhang, X. Huang, Z. Cai and Y. Dong, Stimulus-Responsive Organic Phosphorescence Materials Based on Small Molecular Host–Guest Doped Systems, J. Phys. Chem. Lett., 2023, 14, 1794–1807 CrossRef CAS PubMed.
- H. Sun and L. Zhu, Achieving purely organic room temperature phosphorescence in aqueous solution, Aggregate, 2023, 4, e253 CrossRef CAS.
- Y. Zhang, H. Li, M. Yang, W. Dai, J. Shi, B. Tong, Z. Cai, Z. Wang, Y. Dong and X. Yu, Organic room-temperature phosphorescence materials for bioimaging, Chem. Commun., 2023, 59, 5329–5342 RSC.
- Y. Fan, Q. Li and Z. Li, Afterglow bio-applications by utilizing triplet excited states of organic materials, Sci. China: Chem., 2023, 66, 2930–2940 CrossRef CAS.
- N. Gan, X. Zou, Y. Zhang, L. Gu and Z. An, Recent advances in multicolor organic room-temperature phosphorescence, Appl. Phys. Rev., 2023, 10, 021313 CAS.
- Y. Shen, Z. An, H. Liu, B. Yang and Y. Zhang, Excitation-Dependent Multicolour Luminescence of Organic Materials: Internal Mechanism and Potential Applications, Angew. Chem., Int. Ed., 2023, 62, e202214483 CrossRef CAS PubMed.
- Z. Zhou, X. Xie, Z. Sun, X. Wang, Z. An and W. Huang, Recent advances in metal-free phosphorescent materials for organic light-emitting diodes, J. Mater. Chem. C, 2023, 11, 3143–3161 RSC.
- X. Xu and B. Yan, Recent advances in room temperature phosphorescence materials: design strategies, internal mechanisms and intelligent optical applications, Phys. Chem. Chem. Phys., 2023, 25, 1457–1475 RSC.
- S. Datta and J. Xu, Recent Advances in Organic Molecular-to-Supramolecular Self-Assembled Room-Temperature Phosphorescent Materials for Biomedical Applications, ACS Appl. Bio Mater., 2023, 6, 4572–4585 CrossRef CAS PubMed.
- X. Wang, S. Ma, B. Zhao and J. Deng, Frontiers in Circularly Polarized Phosphorescent Materials, Adv. Funct. Mater., 2023, 33, 2214364 CrossRef CAS.
- X. Luo, B. Tian, Y. Zhai, H. Guo, S. Liu, J. Li, S. Li, T. D. James and Z. Chen, Room-temperature phosphorescent materials derived from natural resources, Nat. Rev. Chem., 2023, 7, 800–812 CrossRef PubMed.
- (a) W. Zhao, Z. He, J. W. Y. Lam, Q. Peng, H. Ma, Z. Shuai, G. Bai, J. Hao and B. Z. Tang, Rational Molecular Design for Achieving Persistent and Efficient Pure Organic Room-Temperature Phosphorescence, Chem, 2016, 1, 592–602 CrossRef CAS; (b) Z. Deng, J. Zhang, J. Zhou, W. Shen, Y. Zuo, J. Wang, S. Yang, J. Liu, Y. Chen, C.-C. Chen, G. Jia, P. Alam, J. W.-Y. Lam and B. Z. Tang, Dynamic Transition between Monomer and Excimer Phosphorescence in Organic Near-Infrared Phosphorescent Crystals, Adv. Mater., 2024, 36, 2311384 CrossRef CAS PubMed.
- H. Ma, Q. Peng, Z. An, W. Huang and Z. Shuai, Efficient and Long-Lived Room-Temperature Organic Phosphorescence: Theoretical Descriptors for Molecular Designs, J. Am. Chem. Soc., 2019, 141, 1010–1015 CrossRef CAS PubMed.
- Y. Su, M. Wu, G. Wang, J. Li, X. Chen, X. Li, G. Wang and K. Zhang, Benzophenone-containing phosphors with an unprecedented long lifetime of 1.8 s under ambient conditions, Chem. Commun., 2023, 59, 1525–1528 RSC.
- Y. Su, X. Du, B. Fu, G. Wang, X. Piao, G. Wang and K. Zhang, Disrupting n–π* Transition of Benzophenone Derivatives’ T1 States to Achieve Ultralong-Lived Room-Temperature Phosphorescence, ACS Mater. Lett., 2024, 6, 1042–1049 CrossRef CAS.
- J. L. Kropp and W. R. Dawson, Radiationless deactivation of triplet coronene in plastics, J. Phys. Chem., 1967, 71, 4499–4506 CrossRef CAS.
- S. Hirata and M. Vacha, Large Reverse Saturable Absorption at the Sunlight Power Level Using the Ultralong Lifetime of Triplet Excitons, J. Phys. Chem. Lett., 2017, 8, 3683–3689 CrossRef CAS PubMed.
- X. Zhai, Z. Mo, Q. Yan, Y. Zhao, B. Wang and K. Zhang, Anti-Smudge Organic Afterglow Panels, Adv. Funct. Mater., 2024, 2405637 CrossRef CAS.
- M. Wu, X. Wang, Y. Pan, J. Li, X. Li, Y. Sun, Y. Zou, H. Zhang and K. Zhang, Two-Component Design Strategy: Achieving Intense Organic Afterglow and Diverse Functions in Coronene-Matrix Systems, J. Phys. Chem. C, 2021, 125, 26986–26998 CrossRef CAS.
- J. Yu, Z. Sun, H. Ma, C. Wang, W. Huang, Z. He, W. Wu, H. Hu, W. Zhao and W. Zhu, Efficient Visible-Light-Activated Ultra-Long Room-Temperature Phosphorescence Triggered by Multi-Esterification, Angew. Chem., Int. Ed., 2023, 62, e202316647 CrossRef CAS PubMed.
- X. Zhai, Y. Zeng, X. Deng, Q. Lou, A. Cao, L. Ji, Q. Yan, B. Wang and K. Zhang, Visible-light-excitable aqueous afterglow exhibiting long emission wavelength and ultralong afterglow lifetime of 7.64 s, Chem. Commun., 2023, 59, 10500–10503 RSC.
- F. Lin, H. Wang, Y. Cao, R. Yu, G. Liang, H. Huang, Y. Mu, Z. Yang and Z. Chi, Stepwise Energy Transfer: Near-Infrared Persistent Luminescence from Doped Polymeric Systems, Adv. Mater., 2022, 34, 2108333 CrossRef CAS PubMed.
- Y. Miao, F. Lin, D. Guo, J. Chen, K. Zhang, T. Wu, H. Huang, Z. Chi and Z. Yang, Stable and ultralong room-temperature phosphorescent copolymers with excellent adhesion, resistance, and toughness, Sci. Adv., 2024, 10, eadk3354 CrossRef CAS PubMed.
- J. Chen, F. Lin, D. Guo, T. Tang, Y. Miao, Y. Wu, W. Zhai, H. Huang, Z. Chi, Y. Chen and Z. Yang, In Situ Reversible and Robust Mechano-Responsive Ultralong Phosphorescence of Polyurethane Elastomer, Adv. Mater., 2024, 2409642 CrossRef CAS PubMed.
- Y. Su, Y. Zhang, Z. Wang, W. Gao, P. Jia, D. Zhang, C. Yang, Y. Li and Y. Zhao, Excitation-Dependent Long-Life Luminescent Polymeric Systems under Ambient Conditions, Angew. Chem., Int. Ed., 2020, 59, 9967–9971 CrossRef CAS PubMed.
- F. Xiao, H. Gao, Y. Lei, W. Dai, M. Liu, X. Zheng, Z. Cai, X. Huang, H. Wu and D. Ding, Guest-host doped strategy for constructing ultralong-lifetime near-infrared organic phosphorescence materials for bioimaging, Nat. Commun., 2022, 13, 186 CrossRef CAS PubMed.
- Y. Xia, C. Zhu, F. Cao, Y. Shen, M. Ouyang and Y. Zhang, Host–Guest Doping in Flexible Organic Crystals for Room-Temperature Phosphorescence, Angew. Chem., Int. Ed., 2023, 62, e202217547 CrossRef CAS PubMed.
- G. Yang, S. Hao, X. Deng, X. Song, B. Sun, W. J. Hyun, M.-D. Li and L. Dang, Efficient intersystem crossing and tunable ultralong organic room-temperature phosphorescence via doping polyvinylpyrrolidone with polyaromatic hydrocarbons, Nat. Commun., 2024, 15, 4674 CrossRef CAS PubMed.
- J. Huang, L. Qu, L. Gao, X. Wang, Q. Chen, Y. Wang, Y. Zhu, C. Li, Y. Li and C. Yang, Multicolor Room-Temperature Phosphorescence Achieved by Intrinsic Polymers Containing Solely One Phosphor Unit, Macromolecules, 2024, 57, 5018–5027 CrossRef CAS.
- Z. Wang, L. Gao, Y. Zheng, Y. Zhu, Y. Zhang, X. Zheng, C. Wang, Y. Li, Y. Zhao and C. Yang, Four-in-One Stimulus-Responsive Long-Lived Luminescent Systems Based on Pyrene-Doped Amorphous Polymers, Angew. Chem., Int. Ed., 2022, 61, e202203254 CrossRef CAS PubMed.
- J. Shi, Y. Zhou, W. Wang, Z. Yang, P. Zhang and G. Liang, Scalable fabrication of red Room-Temperature phosphorescent materials through molecular doping strategy for versatile applications, Chem. Eng. J., 2024, 492, 152419 CrossRef CAS.
- P.-Z. Chen, L.-Y. Niu, Y.-Z. Chen and Q.-Z. Yang, Difluoroboron β-diketonate dyes: Spectroscopic properties and applications, Coord. Chem. Rev., 2017, 350, 196–216 CrossRef CAS.
- B. Zhou, G. Wang, X. Wang, W. Guo, J. Li and K. Zhang, Highly efficient room-temperature organic afterglow achieved by collaboration of luminescent dimeric TADF dopants and rigid matrices, J. Mater. Chem. C, 2021, 9, 3939–3947 RSC.
- Y. Sun, G. Wang, X. Li, B. Zhou and K. Zhang, Achieving High Afterglow Brightness in Organic Dopant-Matrix Systems, Adv. Opt. Mater., 2021, 9, 2100353 CrossRef CAS.
- J. Li, W. Xia, J. Li, G. Wang, X. Wang, Z. Mo, X. Chen and K. Zhang, Developing Bright Afterglow Materials via Manipulation of Higher Triplet Excited States and Relay Synthesis in Difluoroboron β-Diketonate Systems, Adv. Opt. Mater., 2024, 12, 2302311 CrossRef CAS.
- Y. Sun, J. Liu, J. Li, X. Li, X. Wang, G. Wang and K. Zhang, Manipulation of Triplet Excited States for Long-Lived and Efficient Organic Afterglow, Adv. Opt. Mater., 2022, 10, 2101909 CrossRef CAS.
- G. Wang, X. Chen, J. Liu, S. Ding and K. Zhang, Advanced charge transfer technology for highly efficient and long-lived TADF-type organic afterglow with near-infrared light-excitable property, Sci. China: Chem., 2023, 66, 1120–1131 CrossRef CAS.
- X. Wang, Y. Sun, G. Wang, J. Li, X. Li and K. Zhang, TADF-Type Organic Afterglow, Angew. Chem., Int. Ed., 2021, 60, 17138–17147 CrossRef CAS PubMed.
- X. Wang, J. Li, Y. Zeng, X. Chen, M. Wu, G. Wang, X. Li, B. Wang and K. Zhang, Merging thermally activated delayed fluorescence and two-photon ionization mechanisms for highly efficient and ultralong-lived organic afterglow, Chem. Eng. J., 2023, 460, 141916 CrossRef CAS.
- H. Uoyama, K. Goushi, K. Shizu, H. Nomura and C. Adachi, Highly efficient organic light-emitting diodes from delayed fluorescence, Nature, 2012, 492, 234–238 CrossRef CAS PubMed.
- (a) Y. Yang, Y. Liang, Y. Zheng, J. Li, S. Wu, H. Zhang, T. Huang, S. Luo, C. Liu, G. Shi, F. Sun, Z. Chi and B. Xu, Efficient and Color-Tunable Dual-Mode Afterglow from Large-Area and Flexible Polymer-Based Transparent Films for Anti-Counterfeiting and Information Encryption, Angew. Chem., Int. Ed., 2022, 61, e202201820 CrossRef CAS PubMed; (b) Y. Liang, P. Hu, H. Zhuang, Q. Yang, H. Wei, R. Chen, J. Yu, C. Liu, Y. Wang, S. Luo, G. Shi, Z. Chi and B. Xu, Enabling Highly Robust Full-Color Ultralong Room-Temperature Phosphorescence and Stable White Organic Afterglow from Polycyclic Aromatic Hydrocarbons, Angew. Chem., Int. Ed., 2024, 63, e202318516 CrossRef CAS PubMed.
- T. Wang, J. De, S. Wu, A. K. Gupta and E. Zysman-Colman, Thermally Activated and Aggregation-Regulated Excitonic Coupling Enable Emissive High-Lying Triplet Excitons**, Angew. Chem., Int. Ed., 2022, 61, e202206681 CrossRef CAS PubMed.
- W. Xia, J. Li, G. Wang, Q. Yan, J. Huang, X. Chen, B. Fu, Z. Mo and K. Zhang, Visible-Light-Excitable Sky-Blue TADF-Type Organic Afterglow, J. Phys. Chem. C, 2023, 127, 19805–19813 CrossRef CAS.
- J. Wang, Y. Yang, K. Li, L. Zhang and Z. Li, Purely Organic Fluorescence Afterglow: Visible-Light-Excitation, Inherent Mechanism, Tunable Color, and Practical Applications with Very Low Cost, Angew. Chem., Int. Ed., 2023, 62, e202304020 CrossRef CAS PubMed.
- G. Wang, J. Li, X. Li, X. Wang, Y. Sun, J. Liu and K. Zhang, Two-component design strategy: TADF-Type organic afterglow for time-gated chemodosimeters, Chem. Eng. J., 2022, 431, 134197 CrossRef CAS.
- G. Wang, S. Ding, J. Li, Z. Ye, W. Xia, X. Chen and K. Zhang, A narrow-band deep-blue MRTADF-type organic afterglow emitter, Chem. Commun., 2023, 59, 12302–12305 RSC.
- Y. Pan, J. Li, X. Wang, Y. Sun, J. Li, B. Wang and K. Zhang, Highly Efficient TADF-Type Organic Afterglow of Long Emission Wavelengths, Adv. Funct. Mater., 2022, 32, 2110207 CrossRef CAS.
- J. Zhang, J. Li, X. Li, S. Yuan, Y. Sun, Y. Zou, Y. Pan and K. Zhang, Boosting organic afterglow efficiency via triplet–triplet annihilation and thermally-activated delayed fluorescence, J. Mater. Chem. C, 2022, 10, 4795–4804 RSC.
- Y. Xu, S. Yuan, G. Wang, J. Li, X. Wang, X. Li, S. Ding, X. Chen and K. Zhang, Manipulation of Organic Afterglow in Fluoranthene-Containing Dopant-Matrix Systems: From Conventional Room-Temperature Phosphorescence to Efficient Red TADF-Type Organic Afterglow, Chem. – Eur. J., 2023, 29, e202203670 CrossRef CAS PubMed.
- M. Wu, J. Li, J. Huang, X. Wang, G. Wang, X. Chen, X. Li, X. Chen, S. Ding, H. Zhang and K. Zhang, The unexpected mechanism of transformation from conventional room-temperature phosphorescence to TADF-type organic afterglow triggered by simple chemical modification, J. Mater. Chem. C, 2023, 11, 2291–2301 RSC.
- Z. Ye, J. Li, X. Li, G. Wang and K. Zhang, Near-infrared TADF-type organic afterglow materials, Org. Electron., 2024, 134, 107128 CrossRef CAS.
- H. Ohkita, W. Sakai, A. Tsuchida and M. Yamamoto, Charge Recombination Luminescence via the Photoionization of a Dopant Chromophore in Polymer Solids, Macromolecules, 1997, 30, 5376–5383 CrossRef CAS.
- R. Kabe and C. Adachi, Organic long persistent luminescence, Nature, 2017, 550, 384–387 CrossRef CAS PubMed.
- W. Li, Z. Li, C. Si, M. Y. Wong, K. Jinnai, A. K. Gupta, R. Kabe, C. Adachi, W. Huang, E. Zysman-Colman and I. D. W. Samuel, Organic Long-Persistent Luminescence from a Thermally Activated Delayed Fluorescence Compound, Adv. Mater., 2020, 32, 2003911 CrossRef CAS PubMed.
- Z. Lin, R. Kabe, K. Wang and C. Adachi, Influence of energy gap between charge-transfer and locally excited states on organic long persistence luminescence, Nat. Commun., 2020, 11, 191 CrossRef CAS PubMed.
- N. Nishimura, Z. Lin, K. Jinnai, R. Kabe and C. Adachi, Many Exciplex Systems Exhibit Organic Long-Persistent Luminescence, Adv. Funct. Mater., 2020, 30, 2000795 CrossRef CAS.
- P. Alam, N. L. C. Leung, J. Liu, T. S. Cheung, X. Zhang, Z. He, R. T. K. Kwok, J. W. Y. Lam, H. H. Y. Sung, I. D. Williams, C. C. S. Chan, K. S. Wong, Q. Peng and B. Z. Tang, Two Are Better Than One: A Design Principle for Ultralong-Persistent Luminescence of Pure Organics, Adv. Mater., 2020, 32, 2001026 CrossRef CAS PubMed.
- Y. Wang, H. Gao, J. Yang, M. Fang, D. Ding, B. Z. Tang and Z. Li, High Performance of Simple Organic Phosphorescence Host–Guest Materials and their Application in Time-Resolved Bioimaging, Adv. Mater., 2021, 33, 2007811 CrossRef CAS PubMed.
- K. Jinnai, R. Kabe, Z. Lin and C. Adachi, Organic long-persistent luminescence stimulated by visible light in p-type systems based on organic photoredox catalyst dopants, Nat. Mater., 2022, 21, 338–344 CrossRef CAS PubMed.
- K. Jinnai, R. Kabe and C. Adachi, Wide-Range Tuning and Enhancement of Organic Long-Persistent Luminescence Using Emitter Dopants, Adv. Mater., 2018, 30, 1800365 CrossRef PubMed.
- M. Sakurai, R. Kabe, M. Fuki, Z. Lin, K. Jinnai, Y. Kobori, C. Adachi and T. Tachikawa, Organic photostimulated luminescence associated with persistent spin-correlated radical pairs, Commun. Mater., 2021, 2, 74 CrossRef CAS.
- C. Lin, Z. Wu, H. Ma, J. Liu, S. You, A. Lv, W. Ye, J. Xu, H. Shi, B. Zha, W. Huang, Z. An, Y. Zhuang and R.-J. Xie, Charge trapping for controllable persistent luminescence in organics, Nat. Photonics, 2024, 18, 350–356 CrossRef CAS.
- P. Jin, X. Wei, B. Yin, L. Xu, Y. Guo and C. Zhang, Stepwise Charge/Energy Transfer in MR-TADF Molecule-Doped Exciplex for Ultralong Persistent Luminescence Activated with Visible Light, Adv. Mater., 2024, 36, 2400158 CrossRef CAS PubMed.
- Y. Zhou, P. Zhang, Z. Liu, W. Yan, H. Gao, G. Liang and W. Qin, Sunlight-Activated Hour-Long Afterglow from Transparent and Flexible Polymers, Adv. Mater., 2024, 36, 2312439 CrossRef CAS PubMed.
- Z. Lin, M. Li, R. Yoshioka, R. Oyama and R. Kabe, Oxygen-Tolerant Near-Infrared Organic Long-Persistent Luminescent Copolymers**, Angew. Chem., Int. Ed., 2024, 63, e202314500 CrossRef CAS PubMed.
- G. Wang, X. Chen, X. Li, Y. Zeng and K. Zhang, Mechanism landscape in pyrylium induced organic afterglow systems, Chem. Sci., 2023, 14, 8180–8186 RSC.
- G. Wang, X. Chen, Y. Zeng, X. Li, X. Wang and K. Zhang, Dual-Mechanism Design Strategy for High-Efficiency and Long-Lived Organic Afterglow Materials, J. Am. Chem. Soc., 2024, 146, 24871–24883 CrossRef CAS PubMed.
- X. Zou, N. Gan, M. Dong, W. Huo, A. Lv, X. Yao, C. Yin, Z. Wang, Y. Zhang, H. Chen, H. Ma, L. Gu, Z. An and W. Huang, Narrowband Organic Afterglow via Phosphorescence Förster Resonance Energy Transfer for Multifunctional Applications, Adv. Mater., 2023, 35, 2210489 CrossRef CAS PubMed.
- X. Yao, Y. Li, H. Shi, Z. Yu, B. Wu, Z. Zhou, C. Zhou, X. Zheng, M. Tang, X. Wang, H. Ma, Z. Meng, W. Huang and Z. An, Narrowband room temperature phosphorescence of closed-loop molecules through the multiple resonance effect, Nat. Commun., 2024, 15, 4520 CrossRef CAS PubMed.
- X. Zhang, M. Zeng, Y. Zhang, C. Zhang, Z. Gao, F. He, X. Xue, H. Li, P. Li, G. Xie, H. Li, X. Zhang, N. Guo, H. Cheng, A. Luo, W. Zhao, Y. Zhang, Y. Tao, R. Chen and W. Huang, Multicolor hyperafterglow from isolated fluorescence chromophores, Nat. Commun., 2023, 14, 475 CrossRef CAS PubMed.
- Y. Kondo, K. Yoshiura, S. Kitera, H. Nishi, S. Oda, H. Gotoh, Y. Sasada, M. Yanai and T. Hatakeyama, Narrowband deep-blue organic light-emitting diode featuring an organoboron-based emitter, Nat. Photonics, 2019, 13, 678–682 CrossRef CAS.
- G. Wang, S. Ding, J. Li, X. Li, W. Xia, X. Chen, H. Yao, Z. Ye and K. Zhang, Intrinsic Narrowband Organic Afterglow, Chem. Mater., 2024, 36, 3000–3012 CrossRef CAS.
- S. Kong, H. Wang, J. Liao, Y. Xiao, T. Yu and W. Huang, Intrinsic Narrowband Blue Phosphorescent Materials and Their Applications in 3D Printed Self-monitoring Microfluidic Chips, Adv. Mater., 2024, 36, 2412468 CrossRef CAS PubMed.
- S. Hirata, K. Totani, J. Zhang, T. Yamashita, H. Kaji, S. R. Marder, T. Watanabe and C. Adachi, Efficient Persistent Room Temperature Phosphorescence in Organic Amorphous Materials under Ambient Conditions, Adv. Funct. Mater., 2013, 23, 3386–3397 CrossRef CAS.
- N. Notsuka, R. Kabe, K. Goushi and C. Adachi, Confinement of Long-Lived Triplet Excitons in Organic Semiconducting Host–Guest Systems, Adv. Funct. Mater., 2017, 27, 1703902 CrossRef.
- J. Li, X. Wang, Y. Pan, Y. Sun, G. Wang and K. Zhang, Unexpected long room-temperature phosphorescence lifetimes of up to 1.0 s observed in iodinated molecular systems, Chem. Commun., 2021, 57, 8794–8797 RSC.
- J. Li, S. Hao, M. Li, Y. Chen, H. Li, S. Wu, S. Yang, L. Dang, S. Su and M. Li, Triplet Energy Gap-Regulated Room Temperature Phosphorescence in Host–Guest Doped Systems, Angew. Chem., Int. Ed., 2024, e202417426 Search PubMed.
- Y. Lei, W. Dai, J. Guan, S. Guo, F. Ren, Y. Zhou, J. Shi, B. Tong, Z. Cai, J. Zheng and Y. Dong, Wide-Range Color-Tunable Organic Phosphorescence Materials for Printable and Writable Security Inks, Angew. Chem., Int. Ed., 2020, 59, 16054–16060 CrossRef CAS PubMed.
- Z. Xie, X. Zhang, H. Wang, C. Huang, H. Sun, M. Dong, L. Ji, Z. An, T. Yu and W. Huang, Wide-range lifetime-tunable and responsive ultralong organic phosphorescent multi-host/guest system, Nat. Commun., 2021, 12, 3522 CrossRef CAS PubMed.
- Z. Deng, F. Kong, Z. Deng, J. Zhou, S. Yang, S. He, J. Zhang, Y. Zuo, J. Wang, X. Chen, R. T. K. Kwok, G. Jia, P. C. Y. Chow, D. L. Phillips, P. Alam, J. W. Y. Lam and B. Zhong Tang, Visualizing Triplet Energy Transfer in Organic Near-Infrared Phosphorescent Host-Guest Materials, Angew. Chem., Int. Ed., 2024, e202412182 CAS.
- P. K. Samanta, D. Kim, V. Coropceanu and J.-L. Brédas, Up-Conversion Intersystem Crossing Rates in Organic Emitters for Thermally Activated Delayed Fluorescence: Impact of the Nature of Singlet vs. Triplet Excited States, J. Am. Chem. Soc., 2017, 139, 4042–4051 CrossRef CAS PubMed.
- A. J. Gillett, A. Pershin, R. Pandya, S. Feldmann, A. J. Sneyd, A. M. Alvertis, E. W. Evans, T. H. Thomas, L.-S. Cui, B. H. Drummond, G. D. Scholes, Y. Olivier, A. Rao, R. H. Friend and D. Beljonne, Dielectric control of reverse intersystem crossing in thermally activated delayed fluorescence emitters, Nat. Mater., 2022, 21, 1150–1157 CrossRef CAS PubMed.
- S. Xu, W. Wang, H. Li, J. Zhang, R. Chen, S. Wang, C. Zheng, G. Xing, C. Song and W. Huang, Design of highly efficient deep-blue organic afterglow through guest sensitization and matrices rigidification, Nat. Commun., 2020, 11, 4802 CrossRef CAS PubMed.
- S. Kuila and S. J. George, Phosphorescence Energy Transfer: Ambient Afterglow Fluorescence from Water-Processable and Purely Organic Dyes via Delayed Sensitization, Angew. Chem., Int. Ed., 2020, 59, 9393–9397 CrossRef CAS PubMed.
- J.-X. Wang, H. Zhang, L.-Y. Niu, X. Zhu, Y.-F. Kang, R. Boulatov and Q.-Z. Yang, Organic Composite Crystal with Persistent Room-Temperature Luminescence Above 650 nm by Combining Triplet–Triplet Energy Transfer with Thermally Activated Delayed Fluorescence, CCS Chem., 2020, 2, 1391–1398 CrossRef CAS.
- Q. Dang, Y. Jiang, J. Wang, J. Wang, Q. Zhang, M. Zhang, S. Luo, Y. Xie, K. Pu, Q. Li and Z. Li, Room-Temperature Phosphorescence Resonance Energy Transfer for Construction of Near-Infrared Afterglow Imaging Agents, Adv. Mater., 2020, 32, 2006752 CrossRef PubMed.
- S. Ding, X. Wang, G. Wang, M. Wu, J. Li, X. Zhao, H. Li, S. Ren and K. Zhang, A Twisted Phosphor: Breaking T1 Energy Conservation in Dopant-Matrix Organic Phosphorescence Systems, Adv. Opt. Mater., 2023, 11, 2202540 CrossRef CAS.
- K. Chen, Y. Jiang, Y. Zhu, Y. Lei, W. Dai, M. Liu, Z. Cai, H. Wu, X. Huang and Y. Dong, Host to regulate the T1–S1 and T1–S0 processes of guest excitons in doped systems to control the TADF and RTP emissions, J. Mater. Chem. C, 2022, 10, 11607–11613 RSC.
- Z. Mo, G. Wang, J. Li, Q. Yan and K. Zhang, Dopant-Matrix Afterglow Systems: Manipulation of Room-Temperature Phosphorescence/Thermally Activated Delayed Fluorescence Afterglow Mechanism via Mismatch/Match of Intermolecular Charge Transfer between Dopants and Matrices, J. Phys. Chem. Lett., 2023, 14, 11142–11151 CrossRef CAS PubMed.
- X. Deng, J. Huang, J. Li, G. Wang and K. Zhang, Sonication-Responsive Organic Afterglow Emulsions, Adv. Funct. Mater., 2023, 33, 2214960 CrossRef CAS.
- X. Chen, G. Wang, M. Wu, J. Liu, Z. Liu, X. Wang, Y. Zou and K. Zhang, Merging photoinitiated bulk polymerization and the dopant-matrix design strategy for polymer-based organic afterglow materials, Polym. Chem., 2022, 13, 4641–4649 RSC.
- Y. Cao, D. Wang, Y. Zhang, G. Li, C. Gao, W. Li, X. Chen, X. Chen, P. Sun, Y. Dong, Z. Cai and Z. He, Multi-Functional Integration of Phosphor, Initiator, and Crosslinker for the Photo-Polymerization of Flexible Phosphorescent Polymer Gels, Angew. Chem., Int. Ed., 2024, 63, e202401331 CrossRef CAS PubMed.
- Y. Su, G. Wang, B. Fu, X. Piao and K. Zhang, A biomimetic phosphor that can build a rigid microenvironment for its long-lived afterglow in aqueous medium, Commun. Chem., 2024, 7, 270 CrossRef CAS PubMed.
- Y. Zhang, W. Zhang, J. Xia, C. Xiong, G. Li, X. Li, P. Sun, J. Shi, B. Tong, Z. Cai and Y. Dong, Microwave-Responsive Flexible Room-Temperature Phosphorescence Materials Based on Poly(vinylidene fluoride) Polymer, Angew. Chem., Int. Ed., 2023, 62, e202314273 CrossRef CAS PubMed.
- J. Liu, Y. Sun, G. Wang, X. Chen, J. Li, X. Wang, Y. Zou, B. Wang and K. Zhang, Organic Afterglow Emulsions Exhibiting 2.4 s Phosphorescence Lifetimes and Specific Protein Binding Property, Adv. Opt. Mater., 2022, 10, 2201502 CrossRef CAS.
- J. Huang, X. Deng, J. Li, G. Wang, X. Li, H. Yao, C. Lei and K. Zhang, Developing robust organic afterglow emulsion for dissolved oxygen sensing, Chem. Eng. J., 2023, 474, 145809 CrossRef CAS.
- H. Yao, Y. Zhang, G. Wang, J. Li, J. Huang, X. Wang, Z. Zhang, L. Chen, Z. Pan and K. Zhang, TADF-Type Organic Afterglow Nanoparticles with Temperature and Oxygen Dual-Responsive Property for Bimodal Sensing, ACS Appl. Nano Mater., 2023, 6, 15138–15146 CrossRef CAS.
- J. Wei, M. Zhu, T. Du, J. Li, P. Dai, C. Liu, J. Duan, S. Liu, X. Zhou, S. Zhang, L. Guo, H. Wang, Y. Ma, W. Huang and Q. Zhao, Full-color persistent room temperature phosphorescent elastomers with robust optical properties, Nat. Commun., 2023, 14, 4839 CrossRef CAS PubMed.
Footnote |
| † Equal contribution. |
| This journal is © the Owner Societies 2025 |