Unveiling the synthesis mechanisms of (Bi,Sb,Sn)–Te and observation of the n-type to p-type semiconducting transition in Sb–Bi2Te3 nanostructured chalcogenides derived via an aqueous-based reflux method†
Received
6th June 2024
, Accepted 17th March 2025
First published on 11th April 2025
Abstract
A systematic study of a surfactant-assisted, aqueous-based, low-temperature chemical method for the synthesis of (Bi,Sb,Sn)–Te nanostructures was carried out. A detailed understanding of the chemistry involved in the reaction mechanism and decomposition of ethylenediaminetetraacetic acid was proposed for different precursors of Bi, Sb and Sn, and the possibilities for the aqueous-based, low-temperature synthesis of phase pure Bi2Te3, SnTe, and Sb2Te3 was demonstrated. The reaction mechanism revealed that the aqueous-based reflux reaction is suitable for the synthesis of Bi2Te3, unsuitable for the synthesis of SnTe, and suitable for the partial formation of Sb2Te3. Hot-pressed Sb-doped Bi2Te3 samples exhibited an n- to p-type semiconducting transition with variations in temperature and Sb dopant concentration. A significant improvement in Seebeck coefficient and power factor values of 215.12 μV K−1 and 7.1 μW m−1 K−2, respectively, was observed at 465 K for the 5% Sb-doped Bi2Te3 nanostructures. This work focuses primarily on the reaction mechanism of Bi-, Sb- and Sn-based tellurides in an aqueous medium, providing further possibilities in the field of chalcogenide nanostructures for thermoelectric applications.
1. Introduction
Increased energy demand since the last decade has boosted the interest in highly efficient materials for power generation and cooling applications. Thermoelectric (TE) materials are a class of materials that can reversibly convert heat energy into electrical energy. By analyzing the TE principles, the TE performance of materials can be efficiently optimized. A material with a high TE performance should have a good electrical conductivity (σ), high Seebeck coefficient (S) for electronic transport, appropriate carrier concentration and low thermal conductivity (κ) for maintaining the temperature difference. Tuning the carrier concentration and κ through various methods is an efficient way to adjust the TE properties of materials. A dimensionless quantity called the figure of merit (ZT) is a measure of the TE efficiency of any material, which is generally expressed using the following relation:1
Specifically, the focus is on high-performance TE devices comprising n- and p-type components derived from high ZT materials. A series connection is maintained electrically and a parallel connection is maintained thermally on these TE devices or modules. From the above relation, it is clear that a good TE material should possess a “phonon-glass electron-crystal” (PGEC) behavior, resulting in a large S.2 Interestingly, the researchers aim to win the conflict among the interdependent parameters S, σ and κ, and generally, it is an extremely tedious job to satisfy these criteria in any TE material possessing simple crystal structures. With these conflicting properties, any attempt to improve any of the S, σ or κ parameters may significantly deteriorate other properties and thereby the overall TE performance. By considering this key issue, it is interesting to observe that there is a possibility of getting a prominently increased value of power factor (S2σ) when the carrier concentration is nearly 1019, which is exhibited by the materials in the region where there is a crossover among metals and semiconductors.2
The most studied and interesting TE materials in the chalcogenide category are Bi2Te3, Sb2Te3 and SnTe, which have a wide temperature range for thermoelectric applications. Reports on the aqueous-based synthesis of chalcogenide nanostructures are limited.1,2 Due to the inherent transport properties, a hexagonal crystal structure is preferred for the thermoelectric applications of Bi2Te3 and Sb2Te3.1
Bi2Te3 and Sb2Te3 are narrow band gap semiconductors with homologous layered crystal structures having a rhombohedral unit cell with an R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m space group, possessing good σ and low κ and are used for power generation and in cooling devices.3–5Fig. 1(a) shows the layered crystal structure of M2Te3 (M = Bi and Sb) and is described by a hexagonal unit cell, as shown in Fig. 1. The unit cell is composed of five covalently bonded monatomic sheets along the c-axis in the sequence –Te(1)–Bi–Te(2)–Bi–Te(1)–Te(1)–Bi–Te(2)–Bi–Te(1)–. Bi2Te3 is reported to be an n-type semiconductor, while Sb2Te3 is a p-type semiconductor. However, the presence of excess Te may change their type from n-type to p-type semiconductors. Sb2Te3 and its doped derivatives have been widely studied due to their excellent ZT in the temperature range of 300–500 K.6–8 The self-generation of anti-site defects creates a large number of holes in bulk Sb2Te3 owing to a low S.6 As a consequence, the ZT values are limited to about 1 over the entire temperature range, which corresponds to low efficiencies of the TE device, limiting wider applications. Venkatasubramaniam et al.9 proposed a specially constructed Bi2Te3/Sb2Te3 superlattice with a ZT of 2.4 at room temperature. Recently, Yang et al.10 synthesized Sb2Te3 nanostructures and achieved a ZT of 0.37 at 473 K by employing a surfactant-assisted reflux method and utilizing cold compaction, followed by annealing. Several chemical methods have been employed to prepare nanostructures with different shapes. However, a single phase of Sb2Te3 could not be easily achieved due to the spontaneous occupation of partial Sb atoms at the Te lattice sites.11 Wang et al.12 synthesized hexagonal nanoplates of Sb2Te3 using a solvothermal approach. However, they showed a low ZT due to the Te impurity, which reduced the σ of the Sb2Te3 matrix. Hence, the majority of research work has been focused on the preparation of nanostructures with various techniques, while studies on the consolidation of Bi2Te3, Sb2Te3 and Bi2−xSbxTe3 nanocrystals and their thermoelectric properties are relatively rare.
m space group, possessing good σ and low κ and are used for power generation and in cooling devices.3–5Fig. 1(a) shows the layered crystal structure of M2Te3 (M = Bi and Sb) and is described by a hexagonal unit cell, as shown in Fig. 1. The unit cell is composed of five covalently bonded monatomic sheets along the c-axis in the sequence –Te(1)–Bi–Te(2)–Bi–Te(1)–Te(1)–Bi–Te(2)–Bi–Te(1)–. Bi2Te3 is reported to be an n-type semiconductor, while Sb2Te3 is a p-type semiconductor. However, the presence of excess Te may change their type from n-type to p-type semiconductors. Sb2Te3 and its doped derivatives have been widely studied due to their excellent ZT in the temperature range of 300–500 K.6–8 The self-generation of anti-site defects creates a large number of holes in bulk Sb2Te3 owing to a low S.6 As a consequence, the ZT values are limited to about 1 over the entire temperature range, which corresponds to low efficiencies of the TE device, limiting wider applications. Venkatasubramaniam et al.9 proposed a specially constructed Bi2Te3/Sb2Te3 superlattice with a ZT of 2.4 at room temperature. Recently, Yang et al.10 synthesized Sb2Te3 nanostructures and achieved a ZT of 0.37 at 473 K by employing a surfactant-assisted reflux method and utilizing cold compaction, followed by annealing. Several chemical methods have been employed to prepare nanostructures with different shapes. However, a single phase of Sb2Te3 could not be easily achieved due to the spontaneous occupation of partial Sb atoms at the Te lattice sites.11 Wang et al.12 synthesized hexagonal nanoplates of Sb2Te3 using a solvothermal approach. However, they showed a low ZT due to the Te impurity, which reduced the σ of the Sb2Te3 matrix. Hence, the majority of research work has been focused on the preparation of nanostructures with various techniques, while studies on the consolidation of Bi2Te3, Sb2Te3 and Bi2−xSbxTe3 nanocrystals and their thermoelectric properties are relatively rare.
 |
| | Fig. 1 Crystal structure of (a) Bi2Te3/Sb2Te3 and (b) SnTe. | |
SnTe is another narrow band gap semiconductor with a direct band gap of 0.18 eV. SnTe normally forms a p-type semiconductor due to Sn vacancies, and at low temperatures, it becomes a superconductor. SnTe exhibits a very low ZT ∼0.40 at 900 K13 due to its intrinsically high carrier concentration, large ΔEL-Σ of 0.3 eV and high lattice thermal conductivity.14,15 SnTe exists in 3 crystallographic phases. At low temperatures, where the hole concentration is less than 1.5 × 1020 cm−3, SnTe exists in a rhombohedral structure and is known as α-SnTe. At room temperature and atmospheric pressure, SnTe crystallizes in the rock salt structure16 and SnTe is composed of a face-centered cubic lattice of Te atoms with Sn atoms filling all the octahedral voids and is known as β-SnTe. The β-SnTe transforms to γ-SnTe at 18 kbar pressure, with an orthorhombic structure and Pnma space group. The β-SnTe (referred to as SnTe) with cubic structure and Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m space group finds application as TE materials. The crystal structure of SnTe is shown in Fig. 1(b).
m space group finds application as TE materials. The crystal structure of SnTe is shown in Fig. 1(b).
SnTe has received limited attention as a TE material due to its inability to control its very high carrier concentration, which is of the order of 1021 cm−3 at 300 K, resulting in a low S and high κe.17 A large concentration of intrinsic Sn vacancies is responsible for the high p-type carrier concentration in SnTe.18 In SnTe, the energy difference between the light hole valence band (L band) and the heavy hole valence band (Σ band) is 0.3 eV.17,19–23 The large energy separation between the light and heavy hole valence bands restricts the contribution of heavy hole mass to the Seebeck coefficient. Recently, alloying SnTe with other metal tellurides, such as AgSbTe2, MgTe, SrSe, and CdSe, was found to improve its TE performance.24–28 A remarkable enhancement in the ZT was achieved due to the formation of resonance levels in the valence band through In doping in SnTe synthesized by a high energy ball milling and spark plasma sintering.29 Alloying of SnTe with SnSe, which has a high band gap of 0.9 eV, helps reduce the κ value and also results in an improved S due to the convergence of the two valence bands of SnTe. Moreover, the κ value of SnTe can be further decreased by the solid solution alloying with SnSe. Alloying Cd or Hg in SnTe also results in an enhancement in S due to the decreased energy separation between the light and heavy hole valence band.30,31 In the present investigation, we have synthesized the desired crystal structures of different chalcogenide materials such as Bi2Te3, Sb2Te3 and SnTe nanostructures using an aqueous-based low-temperature reflux method and investigated the feasibility of phase formation in an aqueous medium. The aqueous-based reflux method using NaBH4 could successfully lead to the complete reduction of Te without the formation of TeO2, which is highly desirable for the formation of phase pure compounds. Hence, we attempted to synthesize different chalcogenide materials to achieve a good control over the morphology. Further, the TE properties of Sb-doped Bi2Te3 nanostructures are also studied in the present work.
2. Experimental section
The Bi2Te3, Sb2Te3 and SnTe nanostructures were prepared using BiCl3 (99.9%, metals basis, supplied by Alfa Aesar), Sb powder (99.999%, metals basis, supplied by Alfa Aesar), SbCl3 (99.997%, metals basis, supplied by Alfa Aesar), SnCl2 (99.99%, metals basis, supplied by Sigma Aldrich) and Te powder (99.99%, metals basis, supplied by Alfa Aesar) as metal ion precursors and ethylenediaminetetraacetic acid (EDTA) as the surfactant. Deionised water was used as the reaction medium, sodium borohydride (NaBH4) as the reducing agent and the reaction was carried out at the boiling point of the medium. The detailed synthesis procedure is reported in our earlier studies.1 After being stirred for 10 min, the solution was refluxed, which constitutes the primary stage of the reaction, where BiCl3, SbCl3 and SnCl2 could ionize into the corresponding metal ions and chloride ions. EDTA is essential to cap the bismuth ions and to control the morphology over different reaction times. The reaction was carried out using a 4.4 M NaBH4 and reaction time of 24 h to get stable and phase pure nanostructures based on our previous studies. In order to get the nanostructured Bi2Te3, a pH of 10 was maintained during the synthesis (using NaOH). As soon as the reaction was completed, the synthesized products were collected, centrifuged, washed with acetone, ethanol, and deionized water several times, and then dried under a vacuum environment at 373 K for 6 h. The further removal of EDTA in all the samples was ensured by heat treating at temperatures of 473, 533 and 623 K, respectively, for 3 h under inert conditions with intermediate grinding. The samples are labelled as BT, ST-1, ST-2 and TT for Bi2Te3, Sb2Te3 (using Sb powder), Sb2Te3 (using SbCl3) and SnTe, respectively. The 1%, 3% and 5% Sb-doped Bi2Te3 samples are labelled as BS1T, BS3T and BS5T, respectively. The hot pressing of BS1T, BS3T and BS5T samples was carried out at 723 K and 60 Mpa, which are labelled as BS1T-HP, BS3T-HP and BS3T-HP, respectively. Their structures and phase purities were studied by powder X-ray diffraction (XRD) at room temperature using PANalytical X’Pert Pro diffractometer using Cu-Kα radiation and an X-ray wavelength of 1.5406 Å with a step size of 0.0167°. The particle size, morphology and crystallinity are characterized by high-resolution transmission electron microscopy and HR-TEM (FEI Tecnai F20, operated at 200 kV). To investigate the electrical properties, including electrical resistivity (ρ = 1/σ), S and the corresponding thermoelectric power factor S2σ, a rectangular block (dimensions 3 mm × 3 mm × 15 mm) was taken from the hot-pressed pellet and measured in the ZEM-3 (ULVAC-RIKO) system under helium atmosphere from room temperature to 573 K.
3. Results and discussion
3.1. Structural analysis of Bi2Te3, Sb2Te3 and SnTe samples
Fig. 2 shows the XRD patterns of all the studied samples. A phase pure Bi2Te3 is obtained for BT with a hexagonal crystal structure and without any impurity peaks of Bi or Te. The BT sample matches well with the Bi2Te3 hexagonal phase with ICSD No: 20289. The observed peaks for ST1 correspond to the diffraction from the hexagonal Sb2Te3 material having space group R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m with the JCPDS number 15-0874.32 In addition, minor impurity peaks of Sb are noticed in ST1. It is observed that ST2 contains both Sb2Te3 peaks and several impurity peaks of intermediate phases such as Te and Sb2O3. A similar observation was also made for the TT sample, where impurity peaks such as SnTe3O8 and Te are present. Hence, the XRD results suggest that the formation of phase pure compounds of chalcogenides through an aqueous-based chemical method is feasible only for selected cases, especially for BT.
m with the JCPDS number 15-0874.32 In addition, minor impurity peaks of Sb are noticed in ST1. It is observed that ST2 contains both Sb2Te3 peaks and several impurity peaks of intermediate phases such as Te and Sb2O3. A similar observation was also made for the TT sample, where impurity peaks such as SnTe3O8 and Te are present. Hence, the XRD results suggest that the formation of phase pure compounds of chalcogenides through an aqueous-based chemical method is feasible only for selected cases, especially for BT.
 |
| | Fig. 2 XRD patterns of BT, ST1, ST2 and TT nanostructures. | |
3.2. Mechanism of the thermal decomposition of EDTA
Ligands such as EDTA act as a capping agent for tailoring the crystal growth and may also provide the possibility of breaking the crystalline nature, which may lead to different morphologies and wider applications.33–35 EDTA can coordinate with several inorganic ions to form multinuclear complexes. It is well-reported that such structures can also act as a template in the self-assembly process.36–38 It is expected that an oriented attachment may occur with the template effect of EDTA by adding appropriate ligands to the reaction mixtures. Also, EDTA added as an anion surfactant in the solution could connect with Bi3+ ions to form large molecular groups, which facilitates Bi2Te3 nuclei to grow along the surfaces of EDTA agglomerates. When the EDTA selectively binds to one of the facets, the growth rate in either a- or b-axis directions kinetically slows down, with a relative increase in the c-axis direction. The movements of these fine hexagonal crystals together with molecular groups in the solution, make it possible for the particles to connect with each other by suspended bonds according to the definite epitaxy in the c-axis direction and consequently form long nanorods or nanosheets. These rods and sheets are arranged through the template action of the EDTA additive, which is a favourable mechanism as per the recent reports where expensive techniques such as solvothermal/hydrothermal methods and high reaction temperature were used to develop the desired materials.39–41 Hence, the EDTA-assisted synthesis using EDTA as a surfactant gives good control over the morphology and helps to prevent oxidation of the synthesized nanostructures.
However, EDTA residues in the final product can detrimentally affect its structural integrity, crystallinity and thermoelectric performance. To obtain the nanostructured Bi2Te3, a pH of 10 must be maintained during the synthesis (using NaOH) and finally, the precipitated product is to be washed with acetone, ethanol and deionized water several times. The residual EDTA can dissolve well in acetone when the pH is above 8; hence, from the slurry of the aqueous-based Bi2Te3 final product, EDTA and other unreacted residues can be removed by treating it initially with acetone till a clear solution is obtained. Again, upon heat treatment, the EDTA primarily decomposes to N-(2-hydroxyethyl) iminodiacetic acid and iminodiacetic acid at 473 K. At higher temperatures, i.e., at 533 K, these primary products can yield ethylene glycol as the final product. Ethylene glycol controls the morphology of our nanostructures even at a sintering temperature of 623 K. Hence, EDTA is removed partially by washing with different solvents and partially by converting it to ethylene glycol at a higher temperature. The temperature-dependent decomposition of EDTA in the aqueous synthesis of chalcogenide nanostructures is explained as follows: At 533 K, EDTA decomposes into N-(2-hydroxyethy1)iminodiacetic acid and iminodiacetic acid, as shown in eqn (1). Further hydrolysis of N-(2-hydroxyethy1)iminodiacetic acid results in the formation of ethylene glycol and iminodiacetic acid (eqn (2)).42
| | 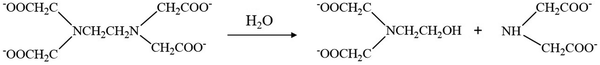 | (1) |
| | 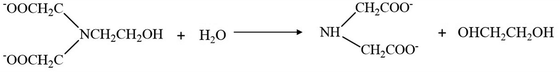 | (2) |
A portion of iminodiacetic acid decomposes to iminodiacetic anhydride with the evolution of water at 512 K (eqn (3)).
| | 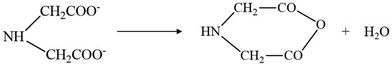 | (3) |
At around 593 K, iminodiacetic anhydride boils off. Beyond 593 K, iminodiacetic acid decomposes into water, carbon dioxide, carbon monoxide, acetonitrile and hydrogen gas as by-products, as shown in eqn (4). Acetonitrile boils at 354 K and ethylene glycol vaporises at 470 K.
| | 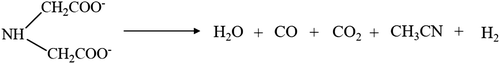 | (4) |
Deng et al. synthesized Bi2Te3 nanorods using EDTA as the surfactant.43 Srashti et al. synthesized Bi2Te3 nanostructures by a refluxing method using EDTA as a surfactant, KOH and NaBH4 and showed that the growth and morphology of Bi2Te3 depends upon the surfactant, concentration of KOH and reaction timings.44 Yang et al. showed that EDTA-Na2 could be used as a complexing agent in the synthesis of nanocrystalline Sb2Te3 and achieved a power factor (PF) of 2.04 μW cm−1 K2 at 140 °C and σ remained in the range of 5–10 × 103 S m−1.45 Dharmaiah et al. synthesized Sb2Te3 nanoplates using EDTA and achieved a high S of 181 μV K−1, high σ of 763 Ω−1 cm−1 and low κ of 1.15 W m−1 K−1.46
3.3. Mechanism for the formation of secondary phases in Sb2Te3 and SnTe samples
To explore further the unstable phases of ST2 and TT, a detailed understanding of the chemistry of these materials is necessary. Let us consider the chemical reactions that could possibly occur in each case. Fig. 3 represents the chemical compounds that can generate Bi3+, Sb3+ and Sn2+/Sn4+ in an aqueous medium to combine with Te2− to form the corresponding chalcogenide materials. The existence of Te2− ions in the aqueous medium is validated with the chemical equations as represented in eqn (5)–(7).| | | 3Te + 6OH− → 2Te2− + TeO32− + 3H2O | (5) |
| | | 2TeO32− + NaBH4 → Te2− + Te + NaBO3 + H2O + 2OH− | (6) |
| | | 5Te + 10OH− + NaBH4 → 5Te2− + NaBO3 + 7H2O | (7) |
 |
| | Fig. 3 Possible chemical compounds generating cations of (a) Bi, (b) and (c) Sb and (d) Sn to form the corresponding chalcogenide nanostructures. | |
While using BiCl3 in the formation of Bi2Te3, it is obvious that Bi(OH)2Cl, which is stable at room temperature, will generate Bi3+ ions in the aqueous medium, whereas Bi4O5Cl2 and BiOCl will not take part in the chemical reaction,47 as represented in eqn (8)–(12).48
| | 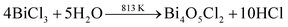 | (8) |
| | 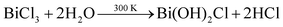 | (9) |
| |  | (10) |
| | 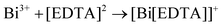 | (11) |
| |  | (12) |
Thus, the formation of stable BT nanostructures is possible using BiCl3. While using SbCl3 as the starting material, the following reactions could occur as mentioned below in eqn (13)–(17)
| | 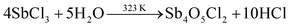 | (13) |
| |  | (14) |
| |  | (15) |
| |  | (16) |
| |  | (17) |
It is well understood from eqn (13) and (15) that the formation of Sb4O5Cl2 and SbOCl is unavoidable, and only the partial synthesis of Sb2Te3 is possible. An easy way to obtain phase pure Sb2Te3 in an aqueous medium is through Sb metallic powder where Sb(OH)3 can contribute Sb3+, as shown in eqn (18).
| |  | (18) |
A small trace of metallic Sb could be present here; still, this method is valid for eqn (16) and (17) to develop Sb2Te3 nanomaterials in an aqueous medium, which corroborates with the XRD results of ST1.
Interestingly, it is observed that the Sn-based reactions are not recommended in aqueous medium as Sn2+ can easily undergo oxidation to form Sn4+, as represented in eqn (19)–(21).
| |  | (19) |
| |  | (20) |
| |  | (21) |
It could be seen that SnCl2 reacts with water to form an insoluble basic salt Sn(OH)Cl. This basic salt could further combine with Te ions present in the solution to form a secondary phase SbTe3O8 in addition to SnTe. Thus, the aqueous-based synthesis using EDTA is not suitable for the synthesis of SnTe. Other methods, such as the one described in the recent report,49 must be adopted to synthesize phase pure SnTe nanopowders.
In this regard, it could be presumed that there are limitations to the formation of Sb2Te3 and SnTe in aqueous-based reflux synthesis. However, the partial formation of Sb2Te3 using EDTA prompted us to synthesize Sb-doped Bi2Te3 and to investigate the n-to-p-type behaviour with Sb doping. Hence, we have attempted to synthesize Sb-doped Bi2Te3. Small amounts of Sb, such as 1%, 3% and 5%, were doped into the Bi site using the reflux method. The use of Sb powder may not bring satisfactory results, and hence, SbCl3 was used as the precursor for the synthesis of Sb-doped Bi2Te3 as SbCl3 will be suitable for replacing Bi ions to form the doped compound.
3.4. Structural analysis of Sb-doped Bi2Te3
Fig. 4 shows the XRD patterns of the Sb-doped BT samples prepared for 1%, 3% and 5% concentrations by keeping the molarity of NaBH4 at 4.4 M. It could be seen that BS1T exists in a single phase at lower Sb concentrations and crystallizes into a hexagonal Bi2Te3 structure. However, BS3T and BS5T contain small amounts of secondary phases of Sb and Sb2O3. Hence, it could be seen that for higher concentrations of Sb, the samples do not become phase pure. Hence, the reflux method alone is not sufficient to produce phase-pure samples of Sb-doped Bi2Te3.
 |
| | Fig. 4 XRD patterns of cold-pressed Sb-doped BT nanostructures. | |
Hence, as a second step, these samples were hot pressed at a pressure of 50 MPa and a temperature of 723 K and the XRD patterns were obtained for the Sb-doped samples, as shown in Fig. 5(a). Upon hot pressing, all the Sb-doped BT samples crystallized into a combination of hexagonal Bi2Te3 and BiTe phases. The Rietveld refinement of all the samples is shown in Fig. 5(b)–(d). No peaks corresponding to Sb or Sb2O3 were indexed in the XRD patterns. The structural parameters determined from Rietveld refinement are listed in Table 1. A low value of χ2 is observed, which justifies the quality and goodness of the refinement.
 |
| | Fig. 5 (a) XRD patterns of hot-pressed Sb-doped BT nanostructures. Refined XRD patterns of (b) BS1T-HP, (c) BS3T-HP and (d) BS5T-HP. | |
Table 1 Refined XRD parameters of BS1T-HP, BS3T-HP and BS5T-HP
| BTA structure |
BS1T-HP |
BS3T-HP |
BS5T-HP |
| Phase |
Bi2Te3 + BiTe |
Bi2Te3 + BiTe |
Bi2Te3 + BiTe |
| Crystal structure |
Hexagonal + rhombohedral |
Hexagonal + rhombohedral |
Hexagonal + rhombohedral |
| Space group |
R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m + P m + P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1 m1 |
R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m + P m + P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1 m1 |
R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m + P m + P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1 m1 |
|
|
| Lattice parameters |
|
a (Å) |
4.38(3) |
4.34(8) |
4.37(3) |
4.34(8) |
4.36(3) |
4.35(8) |
|
b (Å) |
4.38(3) |
4.34(8) |
4.37(3) |
4.34(8) |
4.36(3) |
4.35(8) |
|
c (Å) |
30.08(5) |
24.80(9) |
30.08(5) |
24.80(9) |
30.08(5) |
24.80(9) |
|
γ (deg) |
120 |
120 |
120 |
120 |
120 |
120 |
| Volume (Å)3 |
504.99(6) |
406.50(1) |
504.99(6) |
406.50(1) |
504.99(6) |
406.50(1) |
|
|
| Residual parameters |
|
R
p
|
3.40 |
3.34 |
3.27 |
| wRp |
2.29 |
2.13 |
2.09 |
|
χ
2
|
1.89 |
1.68 |
1.54 |
It is seen from the refinement data that there is a decrease in unit cell volume with an increase in Sb concentration. This is attributed to the small size of Sb3+ ions substituting Bi3+ ions in the lattice. Also, upon hot pressing, there could be a redistribution of ions in the lattice, and the insoluble compounds could have vaporised, leading to the formation of phase-pure Bi2Te3 without any impurity peaks.
3.5. Morphological analysis of hot-pressed Sb-doped Bi2Te3
Fig. 6(a) and (b) show the TEM images of BS1T-HP. The TEM images of BS1T-HP indicate the formation of nanosheets. The formation of nanosheets in BS1T-HP could be attributed to the template action of EDTA, as a result of which an oriented attachment occurs along a preferred direction, resulting in the formation of nanosheets. The (006) plane and the SAED patterns shown in Fig. 6(c) and (d) correspond to the lattice planes of Bi2Te3.
 |
| | Fig. 6 (a) and (b) TEM images showing the formation of BS1T-HP nanosheets. (c) (006) plane corresponding to Bi2Te3 and (d) SAED patterns of BS1T-HP. | |
3.6. Mechanism for the formation of hot-pressed Sb-doped Bi2Te3
The detailed reaction mechanism showing the formation of Bi2Te3 nanostructures was reported in our earlier studies.48 The mechanism for the formation of Bi2−xSbxTe3 nanostructures using EDTA is demonstrated in Fig. 7. Here, the secondary phases of Sb and Sb2O3 present in the as-prepared BS3T and BS5T disappear upon hot pressing. This could be due to the additional energy provided during hot pressing, which results in the phase formation of Bi2−xSbxTe3 nanostructures. The TEM images indicate the formation of sheet-like structures of BS1T-HP. It is well known that EDTA is a multidentate ligand with polyfunctional groups, and it could effectively serve as a bridging ligand to form multinuclear complexes with metal ions above some critical concentration. Then, chains of crystalline seeds would form in the nucleation process, especially with reaction time, which finally yields sheet/rod-like structures of the desired compound. The present approach using the reflux technique could favour the reaction mechanism through nucleation and growth processes. The crystalline Bi2−xSbxTe3 structures could form in the reflux reaction process through a homogeneous nucleation process if the reaction time is a varying parameter along with EDTA concentration. It is well known that the bismuth tellurium-based materials have a highly anisotropic structure, which favours the growth, primarily confined to a particular direction and the crystalline seeds of the material tend to grow into the rod/sheet shape under the influence of EDTA, which would be a soft template, and induce the formation of 1D/2D structures. Hence, EDTA could promote preferential directional growth under the template effect, where the formation of inorganic nanoparticles in liquid media is associated with monomer growth. Previous reports suggest that the nucleus is characterized by various shapes and facets with different surface energies and grows by bonding with other monomers in the solution. Crystal surface energy and facet attachment highly influence nanoparticle growth and shape formation. If any capping agent is present in the solution, it could bind to specific facets of the nucleus to coat it with a monolayer, which precisely occurs between Bi and EDTA in the present technique. These attached surfactants could lower the total surface energy when Bi2−xSbxTe3 nanocrystals are formed, specifically by blocking high-energy facets and exposing low-energy facets.
 |
| | Fig. 7 Schematic showing the formation of EDTA-assisted (a) ST1, (b) 3% and 5% Bi2−xSbxTe3 and (c) hot-pressed Bi2−xSbxTe3 nanostructures. | |
3.7. Seebeck coefficient, electrical resistivity and power factor of hot-pressed Sb-doped Bi2Te3
Fig. 8 shows the temperature dependence of S in the temperature range of 280–480 K. A negative value of S is obtained for BS1T at temperatures below 425 K, revealing an n-type semiconducting behaviour indicating that electrons are the majority carriers up to 425 K. As the temperature increases to 425 K, a clear transition from a negative to a positive value of S is evident, which marks a crossover from the n-type to the p-type. As the Sb doping concentration increases to 5%, the sample becomes completely p-type, which is evident from the obtained positive values of S from Fig. 8. Hence, an n-type to p-type semiconducting behaviour has been achieved with Sb doping in Bi2Te3. However, due to the presence of mixed phases in BS1T-HP, the value of S is quite low, reaching a maximum value of −54.78 μV K−1 at 306 K and 11.2 μV K−1 at 448 K. In the case of BS5T-HP, a maximum value of 215.12 μV K−1 is achieved at 465 K, which is remarkable.
 |
| | Fig. 8 Temperature dependence of Seebeck coefficient (S) of BS1T-HP and BS5T-HP samples. | |
To understand the electrical transport behaviour of the synthesized Sb-doped BT nanostructures, a detailed resistivity analysis was performed on the BS1T-HP and BS5T-HP samples in the temperature range of 280–480 K. Fig. 9 presents the temperature-dependent ρ of these samples, and reveals the semiconducting behaviour for BS1T-HP and BS5T-HP. BS1T-HP exhibits a ρ value of 0.00347 Ω m and BS5T-HP exhibits 0.01848 Ω m at 300 K, which is much higher than the values obtained for the BT samples reported recently.48 Due to this, a considerable decrease in electrical conductivity can happen, which could ultimately lower the PF.
 |
| | Fig. 9 Temperature dependence of resistivity (ρ) of BS1T-HP and BS5T-HP. | |
The temperature variation of PF, which is calculated as S2σ, is plotted and shown in Fig. 10. The PF value of the BS1T-HP sample decreases with temperature, giving a maximum of 0.89 μW m−1 K−2 at 306 K and this low value of PF is arising due to the decreased S and increased ρ value compared to the Bi2Te3 samples discussed in the previous section. With the increase in Sb concentration in BS5T-HP, the PF follows an inverse trend where it increases with an increase in temperature, reaching a maximum value of 7.1 μW m−1 K−2 at 465 K. The decreased value of PF in BS1T-HP could be attributed to the presence of multiphases of Bi2Te3 and BiTe present in the system. It could also be seen that there is a crossover from n-type (negative Seebeck) to p-type (positive Seebeck) in BS1T-HP at 440 K, as shown in Fig. 8. For BS5T-HP, the material becomes completely p-type and Brahmi et al. reported that this transition could be due to the movement of the Fermi level (EF) away from the conduction band and towards the valence band caused by the increased contribution from acceptors by changing the temperature.50 This result could provide a new pathway towards finding the appropriate reaction mechanism and doping scheme for tailoring the TE properties for novel technological applications.
 |
| | Fig. 10 Temperature dependence of PF of BS1T-HP and BS5T-HP. | |
4. Conclusion
In summary, the Sb-doped Bi2Te3 nanostructures were synthesised successfully by a simple, low-cost and low-temperature reflux method using deionized water as the solvent. Structural and morphological changes were obtained by varying the reaction time in 75 mmol EDTA, which is confirmed by XRD and TEM analyses. The reaction mechanism for Bi2Te3, Sb2Te3 and SnTe was investigated in detail. Mechanism for the decomposition of EDTA and the concentration of which could facilitate the formation of chalcogenide nanostructures is explained. EDTA behaves not only as a capping agent but also as a soft template for lowering the surface energy, which could be the reason for the formation of sheet-like structures in Sb-doped Bi2Te3. An n-type semiconducting behaviour is witnessed for the 1% Sb-doped Bi2Te3 at temperatures below 440 K, and above this, a clear transition of semiconducting behaviour from the n-type to p-type is observed. Upon further increase in Sb concentration to 5%, the sample becomes completely p-type. Thus, it is understood that the hot-pressed Sb-doped Bi2Te3 shows an n-type to p-type semiconducting behaviour as a function of both temperature and Sb dopant concentration. However, the 5% Sb-doped Bi2Te3 exhibits a power factor value of 7.1 μW m−1 K−2 at 465 K. These results indicate that Bi and Sb-based chalcogenide nanostructures could be synthesized successfully at low temperatures using a simple aqueous-based reflux technique, where appropriate reaction pathways could help in the formation of phase pure compounds, enhancing the overall thermoelectric performance of nanostructured chalcogenide materials.
Data availability
The data supporting this article have been included as part of the ESI.†
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
The authors would like to acknowledge the financial support received from the Council of Scientific and Industrial Research (CSIR), Govt. of India. V. R. A. is thankful to the Academy of Scientific and Innovative Research and CSIR for granting the Fellowship. The authors would also like to thank the Department of Science and Technology sponsored project number SPF/2023/00018 for partially supporting this work. M Vasundhara acknowledges the support offered by the Department of K&IM (IICT/Pubs./2024/117).
References
- V. R. Akshay, M. V. Suneesh and M. Vasundhara, Tailoring Thermoelectric Properties through Structure and Morphology in Chemically Synthesised n-type Bismuth Telluride Nanostructures, Inorg. Chem., 2017, 56, 6264 CrossRef CAS PubMed.
- L. Yang, Z.-G. Chen, M. Hong, G. Han and J. Zou, ACS Appl. Mater. Interfaces, 2015, 7, 23694–23699 CrossRef CAS PubMed.
- M. N. Touzelhaev, P. Zhou, R. Venkatasubramanian and K. B. Goodson, Thermal characterization of Bi2Te3/Sb2Te3 superlattices, J. Appl. Phys., 2001, 90, 763 CrossRef.
- Y. Kim, A. DiVenere, G. K. L. Wong, J. B. Kelterson, S. Cho and S. J. R. Meyer, Structural and thermoelectric transport properties of Sb2Te3 thin films grown by molecular beam epitaxy, J. Appl. Phys., 2002, 91, 715 CrossRef CAS.
- D. D. Frari, S. Diliberto, N. Stein and J. M. Lecuire, Comparative study of the electrochemical preparation of Bi2Te3, Sb2Te3, and (BixSb1−x)2Te3 films, Thin Solid Films, 2005, 483, 40 Search PubMed.
- S. Schulz, S. Heimann, J. Friedrich, M. Engenhorst, G. Schierning and W. Assenmacher, Synthesis of hexagonal Sb2Te3 nanoplates by thermal decomposition of the single-source precursor (Et2Sb)2Te, Chem. Mater., 2012, 24, 2228 CrossRef CAS.
- P. Dharmaiah, H. S. Kim, C. H. Lee and S. J. Hong, Influence of powder size on thermoelectric properties of p-type 25%Bi2Te3-75%Sb2Te3 alloys fabricated using gas-atomization and spark plasma sintering, J. Alloys Compd., 2016, 686, 1 CrossRef CAS.
- J. Horak, C. Drasar, R. Novotny, S. Karamazov and P. Lostak, Non-stoichiometry of the crystal lattice of antimony telluride, Phys. Status Solidi A, 1995, 149, 549 CrossRef CAS.
- R. Venkatasubramanian, E. Siivola, T. Colpitts and B. O. Quinn, Thin-film thermoelectric devices with high room-temperature figures of merit, Nature, 2011, 413, 597 CrossRef PubMed.
- H. Q. Yang, L. Miao and C. Y. Liu,
et al., A facile surfactant-assisted reflux method for the synthesis of single-crystalline Sb2Te3 nanostructures with enhanced thermoelectric performance, ACS Appl. Mater. Interfaces, 2015, 7, 14263 CrossRef CAS PubMed.
- J. Horak, Z. Stary and J. Klikorka, Relations between structure, bonding, and nature of point defects in layered crystals of tetradymite structure, Phys. Status Solidi B, 1988, 147, 501 CrossRef CAS.
- W. Z. Wang, B. Poudel, J. Yang, D. Z. Wang and Z. F. Ren, High-yield synthesis of single-crystalline antimony telluride hexagonal nanoplates using a solvothermal approach, J. Am. Chem. Soc., 2005, 127, 13792 CrossRef CAS PubMed.
- M. Zhou, Z. M. Gibbs, H. Wang, Y. M. Han, C. N. Xin, L. F. Li and G. J. Snyder, Optimization of thermoelectric efficiency in SnTe: the case for the light band, Phys. Chem. Chem. Phys., 2014, 16, 20741 RSC.
- J. He, J. T. Xu, G. Q. Liu, H. Z. Shao, X. J. Tan, Z. Liu, J. Xu, H. Jiang and J. Jiang, Enhanced thermopower in rock-salt SnTe–CdTe from band convergence, RSC Adv., 2016, 6, 32189 RSC.
- X. J. Tan, H. Z. Shao, J. He, G. Q. Liu, J. T. Xu, J. Jiang and H. C. Jiang, Band engineering and improved thermoelectric performance in M-doped SnTe (M = Mg, Mn, Cd, and Hg), Phys. Chem. Chem. Phys., 2016, 18, 7141 RSC.
- V. Vedeneev, S. Krivoruchko and E. Sabo, Tin Telluride Based Thermoelectrical Alloys, Semiconductors, 1998, 32, 241 CrossRef.
- R. F. Brebrick and A. J. Strauss, Anomalous Thermoelectric Power as Evidence for Two-Valence Bands in SnTe, Phys. Rev., 1963, 131, 104 CrossRef.
- R. F. Brebrick, Deviations from stoichiometry and electrical properties in SnTe, J. Phys. Chem. Solids, 1963, 24, 27 CrossRef CAS.
- L. M. Rogers, Br. Valence band structure of SnTe, J. Appl. Phys., 1968, 1, 845 Search PubMed.
- B. A. Efimova, V. I. Kaidanov, B. Y. Moizhes and I. A. Chernik, Band model of SnTe, Phys. Solid State, 1966, 7, 2032 Search PubMed.
- W. Li, Y. Wu, S. Lin, Z. Chen, J. Li, X. Zhang, L. Zheng and Y. Pei, Advances in Environment-Friendly SnTeThermoelectrics, ACS Energy Lett., 2017, 2(10), 2349 CrossRef CAS.
- A. Banik, U. S. Shenoy, S. Anand, U. V. Waghmare and K. Biswas, Mg Alloying in SnTe Facilitates Valence Band Convergence and Optimizes Thermoelectric Properties, Chem. Mater., 2015, 27(2), 581 CrossRef CAS.
- A. Banik and K. Biswas, Lead-free thermoelectrics: promising thermoelectric performance in p-type SnTe1−xSex system, J. Mater. Chem. A, 2014, 2, 9620 RSC.
- M. K. Han, J. Androulakis, S. J. Kim and M. G. Kanatzidis, Lead-Free Thermoelectrics: High Figure of Merit in p-type AgSnmSbTem + 2, Adv. Energy Mater., 2012, 2, 157 CrossRef CAS.
- Y. Chen, M. D. Nielsen, Y.-B. Gao, T. J. Zhu, X. Zhao and J. P. Heremans, SnTe– AgSbTe2 Thermoelectric Alloys, Adv. Energy Mater., 2012, 2, 58 CrossRef CAS.
- L. D. Zhao, H. J. Wu, S. Q. Hao, C. I. Wu, X. Y. Zhou, K. Biswas, J. Q. He, T. P. Hogan, C. Uher, C. Wolverton, V. P. Dravid and M. G. Kanatzidis, All-scale hierarchical thermoelectrics: MgTe in PbTe facilitates valence band convergence and suppresses bipolar thermal transport for high performance, Energy Environ. Sci., 2013, 6, 3346 RSC.
- H. Wang, Z. M. Gibbs, Y. Takagiwa and G. J. Snyder, Tuning bands of PbSe for better thermoelectric efficiency, Energy Environ. Sci., 2014, 7, 804 RSC.
- B. Gao, J. Tang, F. Meng and W. Li, Band manipulation for high thermoelectric performance in SnTe through heavy CdSe alloying, J. Materiomics, 2019, 5(1), 111 CrossRef.
- Q. Zhang, B. Liao, Y. Lan, K. Lukas, W. Liu, K. Esfarjani, C. Opeil, D. Broido, G. Chen and Z. Ren, High thermoelectric performance by resonant dopant indium in nanostructured SnTe, Proc. Natl. Acad. Sci. U. S. A., 2013, 110, 13261 CrossRef CAS PubMed.
- G. Tan, L. D. Zhao, F. Shi, J. W. Doak, S.-H. Lo, H. Sun, C. Wolverton, V. P. Dravid, C. Uher and M. G. Kanatzidis, High thermoelectric performance of p-type SnTe via a synergistic band engineering and nanostructuring approach, J. Am. Chem. Soc., 2014, 136, 7006 CrossRef CAS PubMed.
- G. Tan, F. Shi, J. W. Doak, H. Sun, L. D. Zhao, P. Wang, C. Uher, C. Wolverton, V. P. Dravid and M. G. Kanatzidis, Extraordinary role of Hg in enhancing the thermoelectric performance of p-type SnTe, Energy Environ. Sci., 2015, 8, 267 RSC.
- U. Nithiyanantham, R. E. Sivasankara, M. Fevzi Ozaydin, H. Liang, A. Rathishkumar and S. Kundu, Low temperature, shape-selective formation of Sb2Te3 nanomaterials and their thermoelectric applications, RSC Adv., 2015, 5, 89621 Search PubMed.
- Y. D. Yin, Y. Lu, Y. G. Sun and Y. N. Xia, Silver nanowires can be directly coated with amorphous silica to generate well controlled coaxial nanocables of silver/silica, Nano Lett., 2002, 2, 427 CrossRef CAS.
- X. Peng, L. Manna and W. Yang,
et al., Shape control of CdSe nanocrystals, Nature, 2000, 404(6773), 59 CrossRef CAS PubMed.
- L. Manna, E. C. Scher and A. P. Alivisatos, Synthesis of soluble and processable rod-, arrow-, teardrop-, and tetrapod-shaped CdSe nanocrystals, J. Am. Chem. Soc., 2000, 122(51), 12700 CrossRef CAS.
- Y. C. Ha, H. J. Sohn, G. J. Jeong, C. K. Lee and K. I. Rhee, Electrowinning of Tellurium from Alkaline Leach Liquor of Cemented Te, J. Appl. Electrochem., 2000, 30, 315 CrossRef CAS.
- R. S. Mane, B. R. Sankapal and C. D. Lokhande, Studies on Chemically Deposited Nanocrystalline Bi2S3 Thin Films, Mater. Res. Bull., 2000, 35, 587 CrossRef CAS.
- S. P. Summers, K. A. Abbound, S. R. Farrah and G. J. Palenik, Syntheses and Structures of Bismuth (III) Complexes with Nitrilotriacetic Acid, Ethylenediaminetetraacetic Acid, and Diethylenetriaminepentaacetic Acid, Inorg. Chem., 1994, 33, 88 CrossRef CAS.
- R. J. Mehta, Y. Zhang, C. Karthik, B. Singh, R. W. Siegel, T. Borca-Tasciuc and G. Ramanath, A new class of doped nanobulk high-figure-of-merit thermoelectrics by scalable bottom-up assembly, Nat. Mater., 2012, 11, 233 CrossRef CAS PubMed.
- J. S. Son, M. K. Choi, M.-K. Han, K. Park, J.-Y. Kim, S. J. Lim, M. Oh, Y. Kuk, C. Park, S.-J. Kim and T. Hyeon, n-Type Nanostructured thermoelectric materials prepared from chemically synthesized ultrathin Bi2Te3 nanoplates, Nano Lett., 2012, 12, 640 CrossRef CAS PubMed.
- P. Kumar, P. Srivastava, J. Singh, R. Belwal, M. K. Pandey, K. S. Hui, K. N. Hui and K. Singh, Morphological Evolution and Structural Characterization of Bismuth Telluride (Bi2Te3) Nanostructures, J. Phys. D: Appl. Phys, 2013, 46, 285301 CrossRef.
- A. E. Martell, R. J. Motekaitis, A. R. Fried, J. S. Wilson and D. T. Macmillian, Thermal decomposition of EDTA, NTA and Nitrilotrimethylenephosphonic acid in aqueous solution, Can. J. Chem., 1975, 53, 3471 CrossRef CAS.
- Y. Deng, C.-W. Nan and L. Guo, A novel approach to Bi2Te3 nanorods by controlling oriented attachment, Chem. Phys. Lett., 2004, 383, 572 CrossRef CAS.
- S. Gupta, S. Neeleshwar, V. Kumar and Y. Y. Chen, Synthesis of bismuth telluride nanostructures by refluxing method, Adv. Mater. Lett., 2012, 3(1), 50 CrossRef CAS.
- H. Q. Yang, L. Miao, C. Y. Liu, C. Li, S. Honda, Y. Iwamoto, R. Huang and S. Tanemura, A Facile Surfactant-Assisted Reflux Method for the Synthesis of. Single-Crystalline Sb2Te3 Nanostructures with Enhanced Thermoelectric Performance, ACS Appl. Mater. Interfaces, 2015, 7(26), 14263 CrossRef CAS PubMed.
- P. Dharmaiah and S.-J. Hong, Hydrothermal method for the synthesis of Sb2Te3 and Bi0.5Sb1.5Te3 nanoplates and their thermoelectric properties, Int. J. Appl. Ceram. Technol., 2017, 15(1), 132 CrossRef.
- H. Kodama, S. Horiuchi and A. Watanabe, The hydrothermal synthesis of bismuth oxide chlorides, J. Solid State Chem., 1988, 75, 279 CrossRef CAS.
- V. R. Akshay, B. Arun, M. V. Suneesh and M. Vasundhara, Surfactant Induced Structural Phase Transitions and Enhanced Room Temperature Thermoelectric Performance in n-Type Bi2Te3 Nanostructures Synthesized via Chemical Route, ACS Applies, Nanomaterials, 2018, 1(7), 3286 Search PubMed.
- G. Han, R. Zhang, S. R. Popuri, H. F. Greer, M. J. Reece, J.-W. G. Bos, W. Zhou, A. R. Knox and D. H. Gregory, Large-Scale Surfactant-Free Synthesis of p-Type SnTe Nanoparticles for Thermoelectric Applications, Materials, 2017, 10, 233 CrossRef PubMed.
- H. Brahmi, R. Neupane, L. Xie, S. Singh, M. Yarali, G. Katwal, S. Chen, M. Paulose, O. K. Varghese and A. Mavrokefalos, Observation of a low temperature n–p transition in individual titania nanotubes, Nanoscale, 2018, 10, 3863 RSC.
|
| This journal is © the Owner Societies 2025 |
Click here to see how this site uses Cookies. View our privacy policy here. ![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m space group, possessing good σ and low κ and are used for power generation and in cooling devices.3–5Fig. 1(a) shows the layered crystal structure of M2Te3 (M = Bi and Sb) and is described by a hexagonal unit cell, as shown in Fig. 1. The unit cell is composed of five covalently bonded monatomic sheets along the c-axis in the sequence –Te(1)–Bi–Te(2)–Bi–Te(1)–Te(1)–Bi–Te(2)–Bi–Te(1)–. Bi2Te3 is reported to be an n-type semiconductor, while Sb2Te3 is a p-type semiconductor. However, the presence of excess Te may change their type from n-type to p-type semiconductors. Sb2Te3 and its doped derivatives have been widely studied due to their excellent ZT in the temperature range of 300–500 K.6–8 The self-generation of anti-site defects creates a large number of holes in bulk Sb2Te3 owing to a low S.6 As a consequence, the ZT values are limited to about 1 over the entire temperature range, which corresponds to low efficiencies of the TE device, limiting wider applications. Venkatasubramaniam et al.9 proposed a specially constructed Bi2Te3/Sb2Te3 superlattice with a ZT of 2.4 at room temperature. Recently, Yang et al.10 synthesized Sb2Te3 nanostructures and achieved a ZT of 0.37 at 473 K by employing a surfactant-assisted reflux method and utilizing cold compaction, followed by annealing. Several chemical methods have been employed to prepare nanostructures with different shapes. However, a single phase of Sb2Te3 could not be easily achieved due to the spontaneous occupation of partial Sb atoms at the Te lattice sites.11 Wang et al.12 synthesized hexagonal nanoplates of Sb2Te3 using a solvothermal approach. However, they showed a low ZT due to the Te impurity, which reduced the σ of the Sb2Te3 matrix. Hence, the majority of research work has been focused on the preparation of nanostructures with various techniques, while studies on the consolidation of Bi2Te3, Sb2Te3 and Bi2−xSbxTe3 nanocrystals and their thermoelectric properties are relatively rare.
m space group, possessing good σ and low κ and are used for power generation and in cooling devices.3–5Fig. 1(a) shows the layered crystal structure of M2Te3 (M = Bi and Sb) and is described by a hexagonal unit cell, as shown in Fig. 1. The unit cell is composed of five covalently bonded monatomic sheets along the c-axis in the sequence –Te(1)–Bi–Te(2)–Bi–Te(1)–Te(1)–Bi–Te(2)–Bi–Te(1)–. Bi2Te3 is reported to be an n-type semiconductor, while Sb2Te3 is a p-type semiconductor. However, the presence of excess Te may change their type from n-type to p-type semiconductors. Sb2Te3 and its doped derivatives have been widely studied due to their excellent ZT in the temperature range of 300–500 K.6–8 The self-generation of anti-site defects creates a large number of holes in bulk Sb2Te3 owing to a low S.6 As a consequence, the ZT values are limited to about 1 over the entire temperature range, which corresponds to low efficiencies of the TE device, limiting wider applications. Venkatasubramaniam et al.9 proposed a specially constructed Bi2Te3/Sb2Te3 superlattice with a ZT of 2.4 at room temperature. Recently, Yang et al.10 synthesized Sb2Te3 nanostructures and achieved a ZT of 0.37 at 473 K by employing a surfactant-assisted reflux method and utilizing cold compaction, followed by annealing. Several chemical methods have been employed to prepare nanostructures with different shapes. However, a single phase of Sb2Te3 could not be easily achieved due to the spontaneous occupation of partial Sb atoms at the Te lattice sites.11 Wang et al.12 synthesized hexagonal nanoplates of Sb2Te3 using a solvothermal approach. However, they showed a low ZT due to the Te impurity, which reduced the σ of the Sb2Te3 matrix. Hence, the majority of research work has been focused on the preparation of nanostructures with various techniques, while studies on the consolidation of Bi2Te3, Sb2Te3 and Bi2−xSbxTe3 nanocrystals and their thermoelectric properties are relatively rare.![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m space group finds application as TE materials. The crystal structure of SnTe is shown in Fig. 1(b).
m space group finds application as TE materials. The crystal structure of SnTe is shown in Fig. 1(b).![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m with the JCPDS number 15-0874.32 In addition, minor impurity peaks of Sb are noticed in ST1. It is observed that ST2 contains both Sb2Te3 peaks and several impurity peaks of intermediate phases such as Te and Sb2O3. A similar observation was also made for the TT sample, where impurity peaks such as SnTe3O8 and Te are present. Hence, the XRD results suggest that the formation of phase pure compounds of chalcogenides through an aqueous-based chemical method is feasible only for selected cases, especially for BT.
m with the JCPDS number 15-0874.32 In addition, minor impurity peaks of Sb are noticed in ST1. It is observed that ST2 contains both Sb2Te3 peaks and several impurity peaks of intermediate phases such as Te and Sb2O3. A similar observation was also made for the TT sample, where impurity peaks such as SnTe3O8 and Te are present. Hence, the XRD results suggest that the formation of phase pure compounds of chalcogenides through an aqueous-based chemical method is feasible only for selected cases, especially for BT.



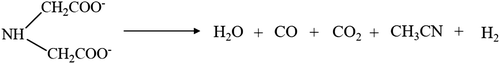

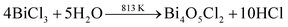
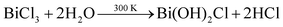

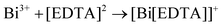

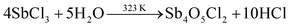









![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m + P
m + P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1
m1![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m + P
m + P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1
m1![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m + P
m + P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1
m1








