Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Investigation of the role of filtration in suppressing laser-induced nucleation of glycine
Thomas
Kendall†
ab,
Divia
Hobson
a,
Nadeem
Javid
 c,
Iain S.
Burns
c,
Iain S.
Burns
 a and
Jan
Sefcik
a and
Jan
Sefcik
 *ab
*ab
aDepartment of Chemical and Process Engineering, University of Strathclyde, 75 Montrose Street, Glasgow, G1 1XJ, UK. E-mail: jan.sefcik@strath.ac.uk
bCMAC, Technology and Innovation Centre, University of Strathclyde, 99 George Street, Glasgow, G1 1RD, UK
cSchool of Chemistry and Biosciences, University of Bradford, Richmond Road, Bradford, BD7 1DP, UK
First published on 8th October 2025
Abstract
Laser-induced non-photochemical nucleation is an intriguing phenomenon, and various mechanisms have been proposed to be responsible. It was previously reported that filtration using syringe nanofilters suppresses laser-induced nucleation of glycine in aqueous solutions, but it is not clear why. In this work we have investigated possible causes of this effect, namely the presence of mesoscale clusters, which may be relevant for an isotropic polarisability mechanism, and the effects of potential impurities which could facilitate a cavitation mechanism. Using syringe filtration with filters of various sizes, we found no direct relationship between the mesoscale cluster population or hydrodynamic radius and the likelihood of laser-induced nucleation of glycine. Furthermore, nanofiltration of deionised water prior to solution preparation was equally effective in suppressing laser-induced nucleation as equivalent filtration of the glycine solutions. This shows that the effect of filtration is not due to removal of impurities originating from glycine itself. Various sources and purification procedures for the water used to prepare glycine solutions were also investigated. Whilst using nanofiltered water or tap water decreased nucleation likelihood, the use of HPLC-grade, distilled, refluxed or degassed water increased nucleation likelihood compared to deionised water. Surprisingly, when either the distillate or the residue fraction from water distillation were used, each of them showed increased nucleation probability. These results taken together show that water treatment is an important determinant of the likelihood of laser-induced nucleation of glycine. We hypothesise that dissolved air is responsible for these observations, whereby syringe filtration leads to supersaturation by air under applied pressure while other treatments lead to degassing. The physical mechanism for this effect is not yet clear and while impurities originating in glycine may facilitate cavitation upon laser irradiation, we suggest that dissolved gas has a key role to play in laser-induced nucleation of glycine, perhaps through cavitation assisted formation of nanobubbles which assist with glycine nucleation.
1. Introduction
In the process of crystallisation there are several ways in which nucleation can be initiated in supersaturated solutions using external forces under isothermal conditions, including shear, agitation, mechanical shock, ultrasound or laser irradiation.1–5 Non-photochemical laser-induced nucleation (NPLIN) was demonstrated in the 1990s by Garetz et al.6 Various mechanisms responsible for this phenomenon have been proposed, including the optical Kerr effect,7 isotropic polarisability8 and cavitation.9However, there is currently no theory which incorporates all relevant observations that have been made around NPLIN.5 The initial explanation was based on the optical Kerr effect, where the electric field of the laser pulses causes the alignment of induced dipoles in solute molecules depending on the polarisation of the incident light.7 It was also reported that this polarisation switching results in selectively forming α and γ glycine polymorphs for circular and linear polarisation, respectively, at certain supersaturations.10,11 However, later experiments did not confirm selective formation of glycine polymorphs with incident light polarisation12 and NPLIN was also demonstrated in experiments involving ionic compounds such as potassium chloride,13 where no dipole can be induced and so the Kerr effect would not be operable. Furthermore, simulation work resulted in the conclusion that the field strength required to account for the observed reduction in nucleation time would need be far greater than is present in experiments.14
In the proposed isotropic polarisability mechanism, molecular arrangement caused by the laser pulses lowers the solution energy state, reducing the required entropic contribution to the crystallisation.8 For this mechanism to be effective, there is a prerequisite for mesoscale solute clusters to be present in supersaturated solutions.15 Such mesoscale clusters have been indeed found in both undersaturated and supersaturated solutions using Brownian microscopy, dynamic light scattering and small angle X-ray scattering and were proposed to play an important role in spontaneous non-classical nucleation mechanisms from solutions.15,16
Cavitation can be caused by laser pulses, where localised heating causes formation of cavitation bubbles17 resulting in crystals nucleating due to highly localised pressure and concentration gradients upon the bubble formation and collapse. Visual observations of nucleation linked to laser-induced cavitation were made using a microscope where cavitation was induced by doping solutions with a substance strongly absorbing at the laser light wavelength used.9 Solid impurities suspended in solutions, such as metallic nanoparticles, have also been suggested to facilitate laser-induced cavitation.18,19 Cavitation can also be caused by ultrasound or mechanical shock and the effects of various means of inducing nucleation on the supersaturation dependence of glycine polymorphism suggest a possible common cause related to cavitation.3
Accordingly, there are currently two main explanations for what causes laser-induced nucleation: isotropic polarisability effects on mesoscale clusters and cavitation induced by impurities present in solutions (solid or dissolved). Previously we have demonstrated that nanofiltration with polyethersulfone 0.2 μm syringe filters suppressed NPLIN in aqueous glycine solution.20 Furthermore, it has been demonstrated that these clusters can be removed through similar nanofiltration15 suggesting a possible link between the two. On the other hand, nanofiltration can also potentially remove some suspended microscopic impurities, which could promote cavitation. We therefore undertook to test whether the presence of mesoscale clusters or filterable impurities in glycine aqueous solutions has a direct impact on the likelihood of laser-induced nucleation. We then set out to investigate how filtration and other solvent purification procedures influence the likelihood of laser-induced nucleation. This was done by systematically testing the effect various filter pore sizes have on mesoscale clusters followed by laser irradiation experiments to investigate induction times for glycine nucleation. From these observations we then proceeded to test alternative solvent purification procedures to assess whether solvent impurities may play role in laser-induced nucleation of glycine.
2. Experimental methods
Solutions were made using electrophoresis-grade glycine (Sigma Aldrich, ≥99% purity) without further purification and deionised water, obtained from an in-house dispenser (Millipore, resistivity > 1 MΩ cm). Solutions of 375 g of glycine in 1 kg of water (i.e. relative supersaturation of 1.47 at 298 K for α polymorph and 1.59 for γ21) were prepared in an incubator at 60 °C with the aid of a magnetic stirrer. Small HPLC vials (Scientific Glass Laboratories, part number T101/V1, diameter 11 mm, volume 2 ml) with screw caps were cleaned with deionised water and pre-heated in the incubator at 60 °C along with syringes and sterile polyethersulfone (PES) syringe filters. Approximately 1.2 ml of solution was either hot-filled or hot-filtered into the HPLC vials at 60 °C, with the non-filtered case serving as a control. Filtration was carried out with polyethersulfone (PES) filters of various pore-sizes ranging from 0.2 μm to 5 μm. For each filter used, the first 10 ml of liquid passing through the filter was discarded to mitigate against potential contamination from any trace residue on the filter. The solutions in vials were then gradually cooled to 25 °C during 3–4 hours and held at that temperature for at least 20 hours. Samples which crystallised within this time were removed before proceeding further. Thus, the solution preparation and hot-filtration methodology are in keeping with those followed previously,20 whereas in the present work a range of filter pore-sizes is used to investigate the mechanism of the effect.Samples to be irradiated were moved from the incubator to a temperature-controlled water bath at 25 °C, and then individually transferred to be exposed to laser pulses for a duration of 1 minute in a Peltier-controlled sample cell. The laser pulses were generated from a Nd:YAG laser (Continuum Surelite II-10) with a wavelength of 1064 nm (pulse duration: 6 ns; repetition rate: 10 Hz; linear polarisation). The beam was condensed by telescope and then passed through a 1-mm aperture to select the central portion of the beam, resulting in a fluence of approximately 3.75 J cm−2 with a peak power density of 0.47 GW cm−2. It is noted that when the laser pulse passes through the vial, the spatial profile is altered since it acts as a cylindrical lens.
All samples were placed on racks inside a temperature-controlled incubator and monitored with digital cameras at a uniform distance from the sample racks. Images of all the vials were automatically recorded every 2 minutes for four days after irradiation. Induction times were obtained from the analysis of image files, the details of which are given in our previous publication.20 The induction time is defined here as the interval between laser irradiation of a sample and the detection of crystals at constant temperature (25 °C). Detection of crystals was based on the time when the apparent size of a crystal in an image reached 1 mm. Strictly speaking, the quantity measured therefore represents a ‘detection time’, since it incorporates the time for a crystal to grow large enough to be observed.
The induction times for the large data set obtained for each set of experimental conditions were plotted as a cumulative probability distribution. For N individual samples in repeated experiments, the probability P(t) that an induction time is between zero and time t is defined as:
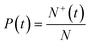 | (1) |
Measurements of dynamic light scattering (DLS) were carried out to investigate the potential presence of sub-micrometre-scale structures that may be removed by nanofiltration, and which may mediate laser-induced nucleation. Samples tested by DLS were made in the same way as irradiated samples and can be considered representative of them. Measurements were performed using an ALV/CGS-3 instrument with scattering angle, θ = 90°, and laser wavelength λ = 632.8 nm. Hydrodynamic radii were calculated from autocorrelation functions using the cumulant method and the Stokes–Einstein equation based on software native to the instrument.
Since the PES filters of any pore size appeared to remove some ‘initiator’ of laser-induced nucleation, the effect of the relative abundance of these hypothetical initiators on nucleation efficiency of laser-induced nucleation was investigated by mixing equal amounts of filtered and non-filtered solution of equal supersaturation, effectively halving the concentration of initiators. At 60 °C, 40 ml of supersaturated aqueous glycine solution (prepared as described above) was hot syringe filtered with a sterile 0.2 μm or 1.2 μm PES filter into 40 ml of unfiltered solution then magnetically stirred for 10 hours. Samples of this 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 mixed solution for irradiation were then hot-filled into vials.
50 mixed solution for irradiation were then hot-filled into vials.
In a separate experiment, a batch of deionised water was passed through a sterile 0.2 μm PES filter at room temperature prior to its use in preparing glycine solutions. The filtered deionised water was then used to make supersaturated glycine solution as described above. This solution was hot-filled into vials without further filtration. The purpose here was to determine whether the agent that seems to promote laser-induced nucleation may originate from the water used to prepare the solutions, rather than from the glycine.
To further investigate the effect of the source and purity of the water used to prepare solutions on the outcome of laser-induced nucleation, experiments were performed with tap water and with HPLC-grade water (in addition to the base case of deionised water described above). Supersaturated aqueous glycine solutions were prepared, in the way already outlined, with water straight from the mains supply (without filtration or other treatment) and with HPLC-grade water (Sigma Aldrich). These solvents and the resulting solutions were not filtered.
Further experiments were carried out to investigate the effect of processing steps used to treat the deionised water prior to solution preparation. Distillation of deionised water was carried out using brand-new bench-top distillation glassware (Scientific Glass Laboratories Ltd) in a sealed assembly. The glassware was cleaned before experimental runs by rinsing with deionised water only. The distillation was continued until the volume of liquid in the boiling flask had reduced by a factor of two (in a first experiment) and three (in a second experiment). In each case, both the distillate and the residue in the boiling flask were used to prepare glycine solutions. Another test was done in which the same distillation apparatus was used to boil and reflux deionised water for 4 hours without collecting any condensate as a top product. These experiments were carried out to isolate whether the effect of distillation in laser-induced nucleation of the resulting, supersaturated glycine solutions was related to the partitioning of some ‘initiator’ between the residue and the condensate, or to some other mechanism. Finally, degassing was done by placing a bottle of deionised water in an ultrasound bath at 323 K on the degas mode for 5 hours, after which it was used to prepare supersaturated glycine solution within an hour. The purpose here was to test the possibility that other observed effects on laser-induced nucleation outcomes may be related to the abundance of dissolved air in irradiated solutions. The conditions for all laser-induced nucleation experiments, and the total number of samples irradiated in each case, are shown in Table 1.
| Experiment number | Filtration | Water source | Water processing steps prior to solution preparation | Number of samples irradiated |
|---|---|---|---|---|
| 1 | — | Deionised | — | 101 |
| 2 | 0.2 μm PES filtration of solution | Deionised | — | 67 |
| 3 | 1.2 μm PES filtration of solution | Deionised | — | 94 |
| 4 | 5 μm PES filtration of solution | Deionised | — | 134 |
| 5 | 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 mix of 0.2 μm PES filtered solution and non-filtered solution 50 mix of 0.2 μm PES filtered solution and non-filtered solution |
Deionised | — | 111 |
| 6 | 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 mix of 1.2 μm PES filtered solution and non-filtered solution 50 mix of 1.2 μm PES filtered solution and non-filtered solution |
Deionised | — | 102 |
| 7 | 0.2 μm PES filtration of water prior to solution preparation (no further filtration) | Deionised | — | 119 |
| 8 | — | HPLC-grade water | — | 167 |
| 9 | — | Mains tap water | — | 111 |
| 10 | — | Deionised | Residue water from distillation (volume reduction by factor of 3) | 40 |
| 11 | — | Deionised | Condensate water from distillation (volume reduction by factor of 3) | 40 |
| 12 | — | Deionised | Residue water from distillation (volume reduction by factor of 2) | 40 |
| 13 | — | Deionised | Condensate water from distillation (volume reduction by factor of 2) | 40 |
| 14 | — | Deionised | Water refluxed in distillation apparatus | 80 |
| 15 | — | Deionised | Ultrasonic degassing | 110 |
3. Results and discussion
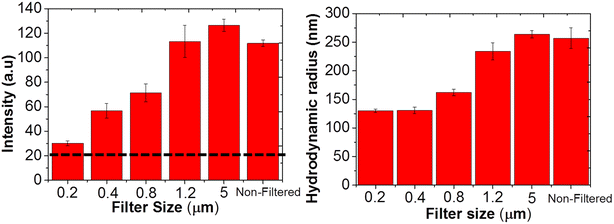
We started by systematically testing the effect of filter pore size on the presence of mesoscale clusters in glycine solutions and the resulting likelihood of laser-induced nucleation. In Fig. 1 we show the results of DLS measurements of aqueous glycine solutions (T = 25 °C; supersaturation = 1.5) filtered with PES syringe filters with a range of pore sizes as well as the non-filtered case. The corresponding DLS data in the form of normalised autocorrelation functions are shown in SI, Fig. S1, while Fig. 1 presents the average scattering intensity and the hydrodynamic radius of mesoscale clusters determined from DLS data.22 It can be seen that solutions filtered with 1.2 μm or 5 μm PES differ little from the unfiltered solution. In contrast, marked reductions both in hydrodynamic radius and scattering intensity are observed for filters of 0.8 μm pore-diameter and below, with these submicron filters being increasingly effective in removing mesoscale clusters with decreasing pore size. For example, when solutions were passed through the 0.2 μm PES filter, we observed a significant decrease in hydrodynamic radius of clusters by about half from 250 to 130 nm and the average scattering intensity was reduced by about 4 times, close to that of pure deionised water.The DLS results are in keeping with our previous observations15,16,23 of liquid-like mesoscale clusters, or ‘nanodroplets’, in non-filtered glycine solutions with submicron hydrodynamic diameters, which depend on solution concentration and can be significantly reduced by syringe filtration using certain nanofiltration membranes. Quantitative interpretation of average scattering intensity is not straightforward since scattering intensity is strongly size and composition dependent but it can be clearly seen from Fig. 1 that for solutions filtered by 0.2 and 0.8 μm PES filters there is a significant reduction in mesoscale cluster number and/or size compared to unfiltered solutions. We conclude that there is a critical pore size in the range 0.8 to 1.2 μm, above which the mesoscale clusters are unaffected by the filtration. If these clusters were to play a pivotal role in mediating laser-induced nucleation, it seems natural to hypothesise that their reduction or removal may explain the role of nanofiltration in suppressing laser-induced nucleation.
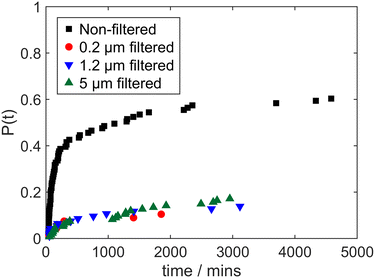
Cumulative probability distributions of induction times in irradiated glycine solutions filtered with 0.2 μm, 1.2 μm and 5 μm PES filters, and without filtration, are shown in Fig. 2. In keeping with previous research,20 there is initially a sharp rise in P(t), which is attributed to laser-induced nucleation, and this is followed by a much shallower increase related to spontaneous nucleation. The magnitude of the initial increase is therefore indicative of the probability of laser-induced nucleation (for example, a probability of about 0.4 for the non-filtered case in Fig. 2); this feature will be used in interpreting our results throughout the paper.
It is striking that there is a pronounced reduction in the probability of laser-induced nucleation for all of the filtered solutions, regardless of the filter pore size. This is in agreement with our previous observations20 that the 0.2 μm PES filters strongly suppress laser-induced nucleation, although not eliminating it completely. Nevertheless, we now also observe that when solutions are filtered through 1.2 μm or 5 μm PES filter, and then irradiated, there still is a suppression of laser-induced nucleation, similar to the effect observed with 0.2 μm filters. This is despite the fact that 1.2 μm and 5 μm PES filters exceed the threshold size to cause a reduction in scattering intensity and hydrodynamic radius of mesoscale clusters. These unexpected results are inconsistent with a potential explanation for the suppression of laser-induced nucleation by filtration due to removal or reduction of mesoscale clusters. Any role these clusters may play in laser-induced nucleation would therefore seem not to be directly dependent of their abundance and size. This leads us to search for some other effect of filtration that may account for these observations. We note that effect of PES syringe filter size on laser-induced nucleation in aqueous urea solutions was recently investigated and it was found that the impact of the filter size on nucleation likelihood was more gradual, from suppressing nucleation with 0.45 μm filters to no significant reduction in nucleation with 2 μm filters in this system,24 however there were differences in experimental protocols used and there may also be variation in the prevailing mechanism between these systems.
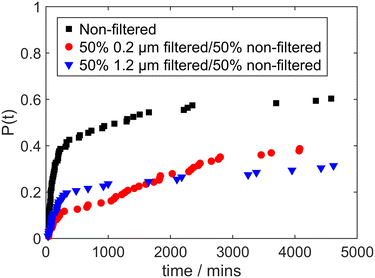
The results of experiments performed with 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 mixtures of filtered and non-filtered solution are shown in Fig. 3, with the results for non-filtered solutions shown again for comparison. Similar outcomes were observed for solutions filtered with 0.2 μm and 1.2 μm filters, whereby the probability of laser-induced nucleation for 50
50 mixtures of filtered and non-filtered solution are shown in Fig. 3, with the results for non-filtered solutions shown again for comparison. Similar outcomes were observed for solutions filtered with 0.2 μm and 1.2 μm filters, whereby the probability of laser-induced nucleation for 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 mixtures is attenuated by approximately 50%. This suggests that the nucleation probability seems to be proportional to the concentration of some species within the solutions. There are two possibilities: (i) this species enhances nucleation and its concentration is reduced by filtration; (ii) this species hinders nucleation and its concentration is increased by filtration. This leads to the question as to the nature and origin of this hypothetical species.
50 mixtures is attenuated by approximately 50%. This suggests that the nucleation probability seems to be proportional to the concentration of some species within the solutions. There are two possibilities: (i) this species enhances nucleation and its concentration is reduced by filtration; (ii) this species hinders nucleation and its concentration is increased by filtration. This leads to the question as to the nature and origin of this hypothetical species.
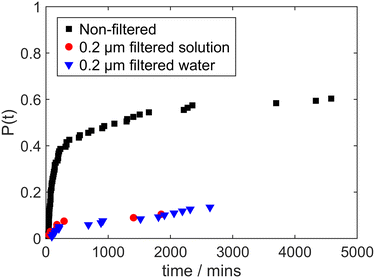
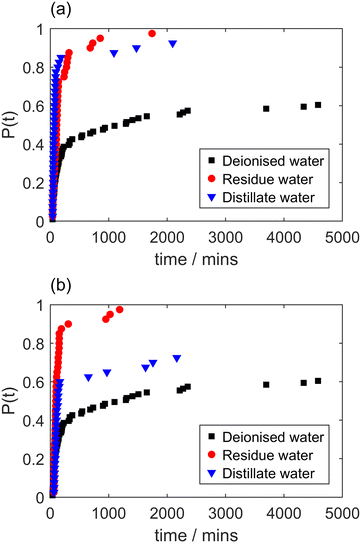
Fig. 4 shows cumulative probability distributions of induction times for laser-induced nucleation of solutions prepared with deionised water that has been filtered (0.2 μm PES) prior to solution preparation (the resulting solutions were not subject to further filtration). Data for non-filtered solutions and solutions filtered with 0.2 μm PES are shown for comparison. It is striking that a similar suppression of laser-induced nucleation of glycine is observed for solutions prepared with filtered water as for filtered solutions. This seems to provide compelling evidence for the hypothetical species affecting laser-induced nucleation being present in the water solvent, rather than in the glycine powder used to prepare solutions. This leads us to consider the effect of preparing solutions with water from other sources and with water that has been subject to various other forms of treatment (without filtration).
The results shown in Fig. 5 compare cumulative probability distribution of induction times after irradiation of solutions made with mains tap water and HPLC-grade water alongside the reference case of deionised water (all without any filtration). Whilst it may have been expected that any component responsible for promoting laser-induced nucleation might be abundant in tap water and largely absent in HPLC-grade water, the data show the reverse trend. The probability of laser-induced nucleation is highest for solutions prepared with HPLC-grade water and lowest for solutions prepared with tap water. This suggests that any component enhancing laser-induced nucleation would be most abundant in HPLC-grade water and least abundant in tap water, with an intermediate concentration in deionised water. Vice versa, any component hindering laser-induced nucleation would be most abundant in tap water and least abundant in HPLC-grade water, with deionised water being intermediate. To investigate these surprising implications, we used distillation to treat the water used to prepare glycine solutions.
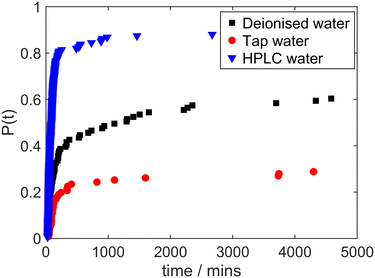 | ||
| Fig. 5 Cumulative distribution of induction times in irradiated glycine solutions made with deionised water (black squares), tap water (red circles), and HPLC-grade water (blue triangles). | ||
The results for laser-induced nucleation of glycine from solutions prepared with deionised water that had been subject to distillation are shown in Fig. 6. The results cover solutions prepared from the water collected as condensed distillate as well as the residue in the boiling flask. Data are presented for cases where the distillation was stopped after the volume of fluid in the boiling flask reduced by a factor of 2 and by a factor of 3, respectively. Strikingly again, we can see that solutions made with either the residue or the distillate each show a clear increase in laser-induced nucleation probability compared to solutions made with untreated deionised water, despite some variation between the two distillation runs. This increase in nucleation probability for solutions prepared either with the distillate or with the residue is quite unexpected. If a hypothetical component hindering laser-induced nucleation was non-volatile, one may expect to see an increase in laser-induced nucleation probability for solutions prepared from the distillate and a decrease for the residue. Likewise, one may expect the reverse for a component hindering laser-induced nucleation that was volatile but condensable. If a hypothetical component was responsible for promoting laser-induced nucleation, distillation would appear to have increased its concentration in both the distillate and the residue, which would be difficult to reconcile. We note that additional experiments (data not presented here) were performed to determine the solid form of glycine crystals formed for laser-irradiated solutions prepared with deionised water, HPLC-grade water, residue water and distillate water (all in the absence of filtration) via Fourier transform infrared (FTIR) spectroscopy.25 The crystals formed were 30–40% γ-polymorph and 60–70% α-polymorph, thus not showing significant variation.
The results of laser-induced nucleation experiments with glycine solutions prepared with deionised water that had been refluxed in the distillation apparatus without collection of any condensed distillate are shown in Fig. 7. Again, this treatment of the deionised water prior to solution preparation is associated with a marked increase in laser-induced nucleation probability, despite the fact that there was no separation of the water into two liquid fractions. These results provide evidence further against a hypothesis of an impurity in solution that promotes laser-induced nucleation but was removed by nanofiltration. We must therefore consider other possible effects of heating and condensing the water. One such effect is the possibility of liberating dissolved gas.
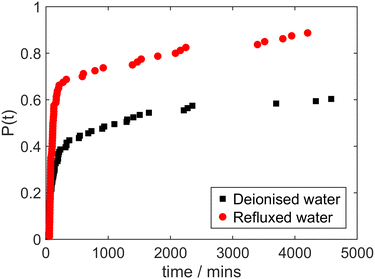 | ||
| Fig. 7 Cumulative distribution of induction times in irradiated glycine solutions made with deionised water (black squares) and water that had been refluxed for 3 hours (red circles). | ||
In Fig. 8 we show the results of experiments performed with solutions prepared with water that had been subject to ultrasonic degassing. There is a clear enhancement of the laser-induced nucleation probability for the solutions prepared with degassed water. This provides further important evidence to complete the results of the sequence of our experiments as a whole.
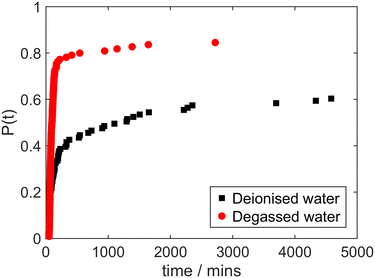 | ||
| Fig. 8 Cumulative distribution of induction times in irradiated glycine solutions made with deionised water (black squares), and degassed deionised water (red circles). | ||
The effect of ultrasonic degassing on increasing the probability of laser-induced nucleation suggests dissolved gas as a way of reconciling all results reported here. This is most readily extended to distillation and refluxing. Boiling is expected to encourage liberation of dissolved air (O2 and N2) from solution, whereas dissolution of gas in the collected condensate would be a much slower process. Regarding sources of water, tap water (at a lower supply temperature and higher pressure) can be expected to have a high concentration of dissolved gas. On the other hand, although the manufacturing process is not fully disclosed by the supplier, HPLC grade water has undergone processing steps, typically including distillation,26 which could therefore plausibly reduce the dissolved gas concentration prior to packaging in a sealed container. Therefore, the laser-induced nucleation outcomes for solutions prepared with the different water sources can be interpreted in the context of increasing dissolved gas concentration resulting in suppressing laser-induced nucleation of glycine.
Returning to nanofiltration of water and of glycine solutions, this had been done by manually applying a significant force to a syringe plunger. Pressure generated in a syringe containing only roughly 1 ml of liquid has previously been estimated to reach as high as 25 bar,27 which would clearly promote increased dissolution of gas which is entrapped in the syringe barrel or inside the filter. The large interfacial area of the liquid in the pores of PES filtration membranes could also aid faster gas dissolution in liquids pushed through the syringe filter.
While it is possible that impurities originating from glycine or glassware used for water treatment such as degassing or distillation may facilitate laser-induced cavitation, it seems likely that dissolved gas has a key role to play in laser-induced nucleation of glycine. The physical mechanism for this effect is not clear, but we speculate that formation of nanobubbles, perhaps assisted by thermal cavitation, may be involved in laser-induced nucleation of glycine. Nanobubbles have been reported to influence crystal nucleation and growth in various systems.28 It was also reported that thermal cavitation can potentially generate nanobubbles and the authors speculated that persistent gas bubbles can then aid nucleation in the context of laser-induced nucleation.18,29 On the other hand, it was reported that neither degassing nor the bubbling of nitrogen through supersaturated potassium sulphate solutions had any effect on laser-induced crystallisation in that system.30 Laser-induced formation of nanobubbles in water was investigated and it was reported that the presence of physical impurities was required.31 The effect of dissolved oxygen in deionised water on nanobubble generation using ultrasound was investigated and it was found that as the dissolved oxygen concentration increased, relatively large bubbles were generated, but nanobubble concentration decreased, and supersaturated conditions had negligible presence of nanobubbles.32 It was also suggested that high dissolved gas concentration can promote nucleation of gas bubbles while hindering cavitation in the process of mechanical shock-induced cavitation.33 Recent simulation and experimental studies of thermal cavitation used the Rayleigh–Plesset equation to predict temperature and concentration profiles at cavitation bubble interfaces,17,34 although the effect of dissolved gas on the pressure threshold for cavitation and nanobubble formation has not yet been investigated using this approach. These recent results taken together provide tantalising hints regarding a potential role of nanobubbles in the mechanism of laser-induced nucleation in glycine aqueous solutions.
4. Conclusions
In this work we have investigated causes of laser-induced nucleation in glycine aqueous solutions. We set out to test whether the presence of mesoscale clusters in glycine aqueous solutions has a direct impact on the likelihood of laser-induced nucleation. We also tested various solvent purification procedures in order to investigate the effects of potential impurities on laser-induced nucleation. Our results show that filtration with PES syringe filters resulted in a strong suppression of laser-induced nucleation regardless of the filter pore size between 0.2 and 5 μm. However, mesoscale clusters were only removed from glycine solutions with filter pore sizes below 1 μm, while for larger filter sizes there was no significant difference between filtered and unfiltered solutions in terms of mean cluster size and scattering intensity. This surprising result indicates that there is no direct relationship between the mesoscale cluster population and the likelihood of laser-induced nucleation in glycine solutions. It also raises a question about the applicability of the isotropic polarisability argument for the laser-induced nucleation of glycine.The effect of potential non-native impurities in glycine aqueous solutions was further investigated by using various sources and purification procedures for the water used to prepare glycine solutions. Using nanofiltered and tap water reduced laser-induced nucleation probability, while on the other hand, using HPLC grade, refluxed, distilled or degassed water increased nucleation probability. Unexpectedly, when the distillate and residue fractions from water distillation were tested, each of them showed increased laser-induced nucleation probability.
These results taken together demonstrate that water treatment is an important determinant of the likelihood of laser-induced nucleation. We hypothesise that dissolved air is responsible for these observations, as heating of water during refluxing or distillation results in decreasing concentration of dissolved gas, while syringe filtration subjects water to significantly elevated pressure leading to increasing concentration of dissolved gas. While impurities originating from glycine or glassware may facilitate laser-induced cavitation, it seems likely that dissolved gas has a key role to play in laser-induced nucleation of glycine. The physical mechanism for this effect is not yet clear and should be subject of future investigations. We speculate that formation of nanobubbles, perhaps assisted by thermal cavitation, may be involved in facilitating laser-induced nucleation of glycine.
Conflicts of interest
The authors declare that there are no conflicts of interest.Data availability
Supplementary information: showing normalised DlS autocorrelation functions for glycine solutions filtered with PES syringe filters of various pore sizes. See DOI: https://doi.org/10.1039/D5CE00516G.The dataset reported in this article is available online: https://doi.org/10.15129/3c3e27b8-eb7f-4cbb-8d81-4715e1e98438.
Acknowledgements
We would like to acknowledge funding for T. K. and J. S. from the EPSRC Future Manufacturing Research Hub in Continuous Manufacturing and Advanced Crystallisation (Grant ref: EP/P006965/1). I. S. B. acknowledges support from the EPSRC (Grant ref: EP/P001661/1).References
- C. Forsyth, P. A. Mulheran, C. Forsyth, M. D. Haw, I. S. Burns and J. Sefcik, Influence of Controlled Fluid Shear on Nucleation Rates in Glycine Aqueous Solutions, Cryst. Growth Des., 2015, 15(1), 94–102, DOI:10.1021/cg5008878.
- N. E. B. Briggs, U. Schacht, V. Raval, T. McGlone, J. Sefcik and A. J. Florence, Seeded Crystallization of beta-L-Glutamic Acid in a Continuous Oscillatory Baffled Crystallizer, Org. Process Res. Dev., 2015, 19(12), 1903–1911, DOI:10.1021/acs.oprd.5b00206.
- Y. Liu, M. H. van den Berg and A. J. Alexander, Supersaturation dependence of glycine polymorphism using laser-induced nucleation, sonocrystallization and nucleation by mechanical shock, Phys. Chem. Chem. Phys., 2017, 19(29), 19386–19392, 10.1039/c7cp03146g.
- M. H. Zamanipoor and R. L. Mancera, The emerging application of ultrasound in lactose crystallisation, Trends Food Sci. Technol., 2014, 38(1), 47–59, DOI:10.1016/j.tifs.2014.04.005 , Review.
- V. Korede, N. Nagalingam, F. M. Penha, N. van der Linden, J. T. Padding, R. Hartkamp and H. B. Eral, A Review of Laser-Induced Crystallization from Solution, Cryst. Growth Des., 2023, 23(5), 3873–3916, DOI:10.1021/acs.cgd.2c01526.
- B. A. Garetz, J. E. Aber, N. L. Goddard, R. G. Young, A. Myerson and S. Nonphotochemical, Polarization-Dependent, Laser-Induced Nucleation in Supersaturated Aqueous Urea Solutions, Phys. Rev. Lett., 1996, 77(16), 3475–3476, DOI:10.1103/PhysRevLett.77.3475.
- J. Matic, X. Y. Sun, B. A. Garetz and A. S. Myerson, Intensity, wavelength, and polarization dependence of nonphotochemical laser-induced nucleation in supersaturated aqueous urea solutions, Cryst. Growth Des., 2005, 5(4), 1565–1567, DOI:10.1021/cg050041c.
- M. R. Ward and A. J. Alexander, Nonphotochemical Laser-Induced Nucleation of Potassium Halides: Effects of Wavelength and Temperature, Cryst. Growth Des., 2012, 12(9), 4554–4561, DOI:10.1021/cg300750c.
- A. Soare, R. Dijkink, M. R. Pascual, C. Sun, P. W. Cains, D. Lohse, A. I. Stankiewicz and H. J. M. Kramer, Crystal Nucleation by Laser-Induced Cavitation, Cryst. Growth Des., 2011, 11(6), 2311–2316, DOI:10.1021/cg2000014.
- X. Y. Sun, B. A. Garetz and A. S. Myerson, Supersaturation and polarization dependence off polymorph control in the nonphotochemical laser-induced nucleation (NPLIN) of aqueous glycine solutions, Cryst. Growth Des., 2006, 6(3), 684–689, DOI:10.1021/cg050460+.
- B. A. Garetz, J. Matic and A. S. Myerson, Polarization switching of crystal structure in the nonphotochemical light-induced nucleation of supersaturated aqueous glycine solutions, Phys. Rev. Lett., 2002, 89, 175501, DOI:10.1103/PhysRevLett.89.175501.
- Y. Liu, M. R. Ward and A. J. Alexander, Polarization independence of laser- induced nucleation in supersaturated aqueous urea solutions, Phys. Chem. Chem. Phys., 2017, 19(5), 3464–3467, 10.1039/c6cp07997k.
- M. R. Ward, I. Ballingall, M. L. Costen, K. G. McKendrick and A. J. Alexander, Nanosecond pulse width dependence of nonphotochemical laser-induced nucleation of potassium chloride, Chem. Phys. Lett., 2009, 481(1–3), 25–28, DOI:10.1016/j.cplett.2009.09.049.
- B. C. Knott, M. F. Doherty and B. Peters, A simulation test of the optical Kerr mechanism for laser-induced nucleation, J. Chem. Phys., 2011, 134(15), 154501, DOI:10.1063/1.3574010.
- A. Jawor-Baczynska, J. Sefcik and B. D. Moore, 250 nm Glycine-Rich Nanodroplets Are Formed on Dissolution of Glycine Crystals But Are Too Small To Provide Productive Nucleation Sites, Cryst. Growth Des., 2013, 13(2), 470–478, DOI:10.1021/cg300150u.
- A. Jawor-Baczynska, B. D. Moore, H. S. Lee, A. V. McCormick and J. Sefcik, Population and size distribution of solute-rich mesospecies within mesostructured aqueous amino acid solutions, Faraday Discuss., 2013, 167, 425–440, 10.1039/c3fd00066d.
- N. Nagalingam, A. Raghunathan, V. Korede, C. Poelma, C. S. Smith, R. Hartkamp, J. T. Padding and H. B. Eral, Laser-Induced Cavitation for Controlling Crystallization from Solution, Phys. Rev. Lett., 2023, 131, 124001, DOI:10.1103/PhysRevLett.131.124001.
- E. R. Barber, M. R. Ward and A. J. Alexander, The role of cavitation and gas bubbles in the non-photochemical laser-induced nucleation of sodium acetate, CrystEngComm, 2024, 26(27), 3634–3642, 10.1039/d4ce00487f.
- M. Briard, C. Brandel and V. Dupray, Strong Enhancement of Nucleation Efficiency of Aqueous Ethylenediamine Sulfate Solutions by Nonphotochemical Laser-Induced Nucleation: Investigations on the Role of Solid Impurities in the Mechanism, Cryst. Growth Des., 2023, 23(10), 7169–7178, DOI:10.1021/acs.cgd.3c00588.
- N. Javid, T. Kendall, I. S. Burns and J. Sefcik, Filtration Suppresses Laser-Induced Nucleation of Glycine in Aqueous Solutions, Cryst. Growth Des., 2016, 16(8), 4196–4202, DOI:10.1021/acs.cgd.6b00046.
- A. Manson, J. Sefcik and L. Lue, Temperature Dependence of Solubility Predicted from Thermodynamic Data Measured at a Single Temperature: Application to α, β, and γ-Glycine, Cryst. Growth Des., 2022, 22(3), 1691–1706, DOI:10.1021/acs.cgd.1c01217.
- J. Stetefeld, S. A. McKenna and T. R. Patel, Dynamic light scattering: a practical guide and applications in biomedical sciences, Biophys. Rev., 2016, 8(4), 409–427, DOI:10.1007/s12551-016-0218-6.
- G. Zimbitas, A. Jawor-Baczynska, M. J. Vesga, N. Javid, B. D. Moore, J. Parkinson and J. Sefcik, Investigation of molecular and mesoscale clusters in undersaturated glycine aqueous solutions, Colloids Surf., A, 2019, 579, 123633, DOI:10.1016/j.colsurfa.2019.123633.
- S. Li, X. Xie, Q. Qiu and Y. Liu, Laser-induced nucleation of urea through the control of Insoluble Impurity, Sci. Rep., 2024, 14(1), 25777, DOI:10.1038/s41598-024-77557-6.
- T. Kendall, Investigating Mechanisms of Laser Induced Nucleation, PhD thesis, University of Strathclyde, Glasgow, 2017 Search PubMed.
- Japanese Standards Association: General rules for high performance liquid chromatography (JIS K 0124:2011), Tokyo, 2011 Search PubMed.
- W. A. Hayward, L. J. Haseler, L. G. Kettwich, A. A. Michael, W. Sibbitt Jr. and A. D. Bankhurst, Pressure generated by syringes: implications for hydrodissection and injection of dense connective tissue lesions, Scand. J. Rheumatol., 2011, 40(5), 379–382, DOI:10.3109/03009742.2011.560892.
- I. D. Tegladza, G. H. Lin, C. Liu and X. H. Gu, Control of crystal nucleation, size and morphology using micro−/nanobubbles as green additives - a review, Sep. Purif. Technol., 2023, 311, 123232, DOI:10.1016/j.seppur.2023.123232.
- E. R. Barber and A. J. Alexander, High-speed imaging of non-photochemical laser-induced nucleation in aqueous cesium chloride, Phys. Chem. Chem. Phys., 2025, 27(12), 6288–6298, 10.1039/d4cp04147j.
- M. Briard, C. Brandel, S. Morin-Grognet, G. Coquerel and V. Dupray, Potassium Sulfate: A New Candidate to Explore Non-Photochemical Laser-Induced Nucleation Mechanisms, Crystals, 2021, 11(12), 1571, DOI:10.3390/cryst11121571.
- M. Jelencic, U. Orthaber, J. Mur, J. Petelin and R. Petkovsek, Evidence of laser-induced nanobubble formation mechanism in water, Ultrason. Sonochem., 2023, 99, 106537, DOI:10.1016/j.ultsonch.2023.106537.
- J. I. Lee, B. S. Yim and J. M. Kim, Effect of dissolved-gas concentration on bulk nanobubbles generation using ultrasonication, Sci. Rep., 2020, 10, 18816, DOI:10.1038/s41598-020-75818-8.
- Y. C. Sun and A. J. Alexander, Mechanical shock-induced nucleation in solution: Is cavitation necessary?, J. Cryst. Growth, 2022, 594, 126786, DOI:10.1016/j.jcrysgro.2022.126786.
- N. Hidman, G. Sardina, D. Maggiolo, H. Ström and S. Sasic, Numerical Frameworks for Laser-Induced Cavitation: Is Interface Supersaturation a Plausible Primary Nucleation Mechanism?, Cryst. Growth Des., 2020, 20(11), 7276–7290, DOI:10.1021/acs.cgd.0c00942.
Footnote |
| † Present address: Technobis, Pyrietstraat 2, 1812 SC Alkmaar, The Netherlands. |
| This journal is © The Royal Society of Chemistry 2025 |