Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Vinyl benzotriazole to indole: an iodine-mediated denitrogenative transannulation approach for the synthesis of indoles
Deepika
Thakur†
a,
Shivam A.
Meena†
a,
Manvi
Sharma
a,
Abhijit
Nandy
b,
Kirti
Arora
a,
Shibdas
Banerjee
*b and
Akhilesh K.
Verma
 *a
*a
aDepartment of Chemistry, University of Delhi, Delhi-110007, India. E-mail: averma@acbr.du.ac.in
bDepartment of Chemistry, IISER Tirupati, Tirupati-517507, India
First published on 18th November 2025
Abstract
A novel, convenient, and molecular iodine-mediated approach for the synthesis of 3-sulfonylated indoles from vinyl benzotriazoles is described. The reaction proceeds via tosylation, denitrogenative ring opening, and cyclization, featuring the cleavage of C–N and N–N bonds and the concomitant formation of C–S, N–H, and C–C bonds. This operationally simple protocol utilizes environmentally benign iodine for ring-opening reaction and circumvents the need for the protection of benzotriazoles with strong electron-withdrawing groups, metal catalysts, elevated temperatures, and extended reaction times. The mechanism is well supported by online real-time mass monitoring analysis and control experiments.
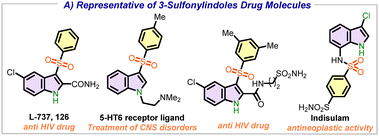
The synthesis of indole derivatives has long been a fundamental focus driven by their extensive applications in the field of drug design, agrochemicals, and alkaloids.1–3 The 3-sulfonyl indole derivatives L-737, 126 serve as potent HIV-1 non-nucleoside reverse transcriptase inhibitors (NNRTIs), whereas 5-HT6 is utilized in the treatment of CNS disorders and Alzheimer's disease (Fig. 1).4–6 Indisulam displays inhibitory effects on carbonic anhydrases and cytosolic malate dehydrogenase.7 Given their significance, several strategies have been devised for the construction of sulfonylated indoles over the years.8
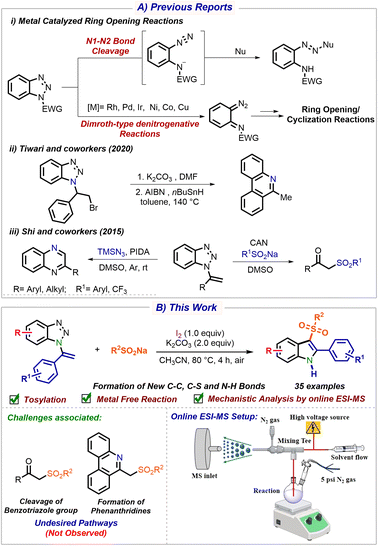
Benzotriazoles have been extensively used as synthetic auxiliaries in organic reactions.9 However, in the last few decades, benzotriazole has emerged as a versatile synthon to access heterocyclic compounds via ring-opening reactions.9,10 These reactions include Dimroth-type cyclizations and Graebe-Ullmann-type reactions or pyrolysis at 600 °C (Scheme 1A(i)).11 Despite their efficiencies, these reactions are limited by poor selectivity, competing intramolecular reactions, and the formation of multiple side products.12 In addition, the flash vacuum pyrolysis protocol requires extremely high temperatures (500–1000 °C) under continuous vacuum and low pressure (10−2 Torr), resulting in significant energy and resource consumption.13a–c
Notably, the literature reveals a limited number of examples addressing the synthesis of heterocycles primarily from vinyl benzotriazole. Tiwari and co-workers have proficiently employed the benzotriazole ring cleavage approach for the two-step synthesis of phenanthridine from 1-(2-bromo-1-phenylethyl)-1H-benzotriazole (Scheme 1A(ii)).14 Remarkably, Shi's group reported that vinyl benzotriazole reacts with TMSN3 to form quinoxaline, whereas in the presence of CAN, it yields α-substituted ketones through benzotriazole elimination without ring opening (Scheme 1A(iii)).15 Inspired by previous works and our ongoing investigation on benzotriazole chemistry and indole synthesis,16,17 we hypothesized that the sulfonyl radical could trigger benzotriazole ring opening with subsequent cyclization, giving divergent outcomes. We herein report an unprecedented iodine-mediated ring-opening/cyclization of vinyl benzotriazole, enabling the efficient synthesis of 3-(arylsulfonyl)-1H-indoles (Scheme 1B).
We started our investigation with 1-(1-phenylvinyl)-1H-benzo[d][1,2,3]triazole 1a and sodium 4-methylbenzene sulfinate 2a as the model substrates (Table 1). When the reaction of 1a and 2a was carried out in the presence of I2 and DBU with MeOH as a solvent, no product 3a was observed (Table 1, entry 1). When the reaction was conducted in K3PO4 and Cs2CO3, product 3a was observed in 23 and 68% yield (entries 2 and 3). Gratifyingly, the use of K2CO3 as a base significantly improved the yield of 3a to 72% (entry 4). CH3CN proved to be the optimal solvent, delivering product 3a in 82% yield (entry 5). Decreasing the loading of 2a and I2 led to 64 and 52% yield of product 3a (entry 6). Notably, decreasing or extending the reaction duration did not enhance the reaction yield (entry 7). Performing the reaction at 25 and 60 °C led to 30–65% yield of product 3a (entries 8 and 9). Use of other iodine sources, such as NIS and PIDA, was found to be inferior for the reaction (entry 10). No product 3a was obtained in the absence of I2, confirming its essential role in radical generation (entry 11). Performing the reaction under an inert atmosphere (N2 or Ar) afforded product 3a in comparable yields, indicating that the atmosphere has little effect on the reaction outcome (entry 12).
| Entry | Reagent | Solvent | Base | Temp/time (°C h−1) | Yieldb (%) |
|---|---|---|---|---|---|
| a Reactions were performed using 0.23 mmol of 1a, 1.2 equiv. of 2a, 1.0 equiv. of reagent, and 2.0 equiv. of base in 3.0 mL solvent. b Isolated yields. c 1.0 equiv. of 2a was used. d Using 0.5 equiv. of I2. e 12 h. f PIDA was used instead of NIS. g N2. h Ar. | |||||
| 1 | I2 | MeOH | DBU | 80/4 | 00 |
| 2 | I2 | MeOH | K3PO4 | 80/4 | 23 |
| 3 | I2 | MeOH | Cs2CO3 | 80/4 | 68 |
| 4 | I2 | MeOH | K2CO3 | 80/4 | 72 |
| 5 | I2 | CH3CN | K2CO3 | 80/4 | 82 |
| 6 | I2 | CH3CN | K2CO3 | 80/4 | 64c,52d |
| 7 | I2 | CH3CN | K2CO3 | 80/2 | 55, 81e |
| 8 | I2 | CH3CN | K2CO3 | 25/6 | 30 |
| 9 | I2 | CH3CN | K2CO3 | 60/6 | 46, 65e |
| 10 | NIS | CH3CN | K2CO3 | 80/6 | 16, 0f |
| 11 | — | CH3CN | K2CO3 | 80/6 | n.r. |
| 12 | I2 | CH3CN | K2CO3 | 80/4 | 80g, 77h |
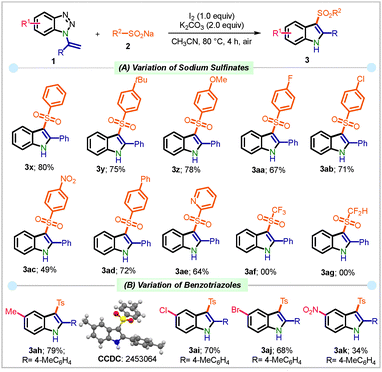
Having established the optimal conditions, the scope of this protocol was investigated using a variety of 1-(1-arylvinyl)-1H-benzo[d][1,2,3]triazoles 1 (Scheme 2). The reactions of vinyltriazoles 1 bearing methyl (1b), ethyl (1c), n-butyl (1d), t-butyl (1e), and n-pentyl (1f) groups at the p-position of the phenyl ring gave the products 3b–3f in 72–80% yields. When a scale-up reaction was performed using 4.52 mmol of 1a, product 3a was obtained in 70% yield. Likewise, p-methoxy and p-ethoxyvinyltriazole (1g–h) furnished the products 3g and 3h in 83 and 81% yield. Substrates 1i–1k bearing halogens at the p-position of the phenyl group provided 3i–3k in 72–75% yield. Notably, electron-withdrawing groups such as –CF3, –NO2, and –CO2Me gave the corresponding products 3l–n in 62–70% yield.
The 1,1′-biphenyl and naphthyl-tethered benzotriazole 1o–q were amenable in this transformation, delivering 3o–q in 73–79% yield. Similar results were obtained with meta-substituted substrates having methyl (1r) and fluoro (1s) groups, which led to the product 3r–s in 69–74% yield. The developed protocol furnished di-substituted product 3t bearing a 2,5-dimethoxy group, and 3u bearing a 3-thienyl group in 71 and 79% yield, respectively. However, substrates 1v–w bearing aliphatic substituents at the R1 position failed to afford the desired products 3v and 3w.
We next extended the developed protocol to a range of sodium sulfinate salts 2 (R2) (Scheme 3A). Arylsulfinates bearing –H, –tBu, and –OMe were found suitable, providing products 3x–z in 75–80% yields. Electron-withdrawing groups such as fluoro, chloro, and nitro at the p-position of the sulfinate derivatives furnished 3aa–ac in 49–71% yield. Furthermore, sodium [1,1′-biphenyl]-4-sulfinate 2h and heterocyclic sodium pyridine-3-sulfinate 2i led to product 3ad–ae in good yields. However, the aliphatic sulfinate salt 2j–k failed to deliver the desired product 3af–ag under the standard reactions conditions. Next, the substrate scope for different benzotriazoles (R1) was evaluated. Introduction of different groups at the 5-position of vinyl benzotriazole (1v) led to 3ah–ak in 34–79% yield (Scheme 3B).
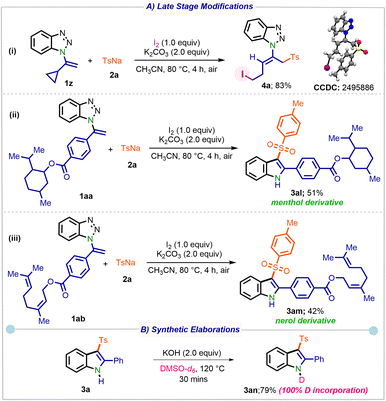
Interestingly, when 1z was subjected to the reaction conditions, ring opening occurred preferentially at the cyclopropyl moiety rather than the benzotriazole, furnishing the sole E-iodo substituted product 4a in 83% yield (Scheme 4A(i)). The applicability of the developed protocol was further showcased through late-stage modifications. Sulfonylated analogues 3al and 3am, derived from menthol and nerol, were obtained in 51 and 42% yield, respectively. When 3a was treated with KOH in DMSO-d6, product 3an was obtained in 79% yield with 100% deuterium incorporation on the nitrogen centre (Scheme 4B).
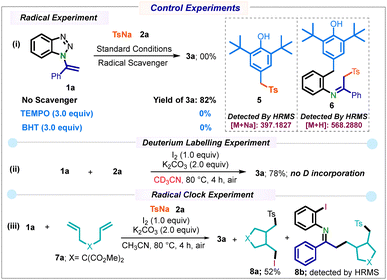
To rationalize the mechanistic pathway, several control experiments were conducted (Scheme 5). The addition of radical scavengers such as TEMPO and BHT to the reaction mixture effectively inhibited the product formation, proving that the transformation initiates via radical reaction (Scheme 5(i)). Also, BHT adduct 5 was detected by HRMS, confirming the in situ generation of tosyl radicals. Meanwhile, the detection of another BHT adduct 6 supports the formation of radical species B. A deuterium-labelling experiment in CD3CN showed no D-incorporation (Scheme 5(ii)), suggesting that the solvent does not serve as a proton source. A radical clock experiment with substrate 7a afforded product 8a in 52% yield along with the ring-opened product 8b (detected by HRMS), supporting a radical ring-opening mechanism.
To further support the mechanism, online ESI-MS analysis for real-time detection of reactive intermediates was conducted (Scheme 6A). Online ESI-MS analysis plots confirm the formation of tosyl iodide 2A, which decreases with time, showing the consumption of intermediates (for more details, see the SI, Fig. S4). The tosyl radical 2B is also detected by online ESI-MS analysis, and BHT trapping confirms that the reaction initiates via a radical pathway. The online ESI-MS analysis also confirms the formation of iodo-substituted indoline species D, which converts into product 3b. Based on these findings from control experiments, online ESI-MS analysis, and previous reports,15,16,18 a plausible mechanism is proposed in Scheme 6B. The reaction begins with the formation of tosyl iodide 2A from sodium sulfinate 2a and I2. The thermal homolytic cleavage of tosyl iodide 2A results in the formation of tosyl radical 2B (detected by online ESI-MS and BHT) and an iodine radical. The tosyl radical attacks vinyl benzotriazole 1b, which generates species A. Then, species A undergoes a ring-opening reaction by the release of N2 gas, generating species B. This species further undergoes rapid tautomerisation to generate C. The radical at the phenyl ring attacks the alkene, which in turn attacks the iodine radical, generating 2-iodo-2-(p-tolyl)-3-tosylindoline D (captured by real-time mass monitoring), which finally, by the elimination of HI, led to 3-tosyl indole 3b.
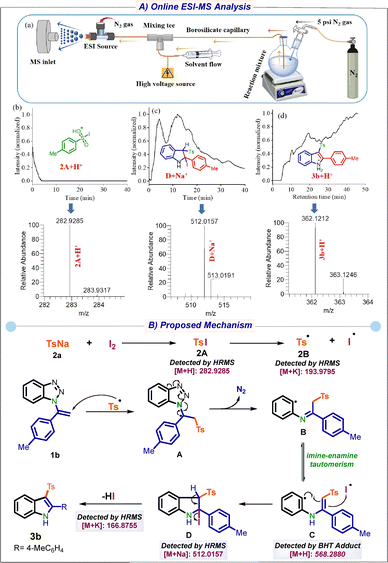 | ||
| Scheme 6 (A) Custom-built online ESI-MS setup and real-time mass monitoring of the reaction. (B) Proposed reaction mechanism. | ||
In conclusion, we have developed a metal-free iodine-mediated ring opening of vinyl benzotriazoles for the synthesis of 3-sulfonyl indoles bearing a free N–H group. This operationally simple method features high functional group tolerance, which renders the late-stage functionalization of relevant molecules such as menthol and nerol. The success of this reaction is attributed to a radical pathway, supported by mechanistic investigations including real-time mass monitoring studies, which involves tosyl iodide-mediated benzotriazole ring opening, N2 extrusion, and subsequent cyclization.
This work was supported by SERB (CRG/2022/008887). D. T. and S. A. M. thank CSIR for SRF fellowships and acknowledge the University of Delhi and USIC for instrumentation support.
Conflicts of interest
There are no conflicts to declare.Data availability
The data underlying this study including 1H-NMR, 13C-NMR, SC-XRD and HRMS of all the synthesized compounds are available in the published article and its supplementary information (SI). See DOI: https://doi.org/10.1039/d5cc05419b.CCDC 2453343, 2453064 and 2495886 contain the supplementary crystallographic data for this paper.19a–c
Notes and references
- (a) T. V. Sravanthi and S. L. Manju, Eur. J. Med. Chem., 2016, 124, 74 Search PubMed; (b) N. K. Kaushik, N. Kaushik, P. Attri, N. Kumar, C. H. Kim, A. K. Verma and E. H. Choi, Molecules, 2013, 18, 6620 CrossRef CAS PubMed.
- P. Sun, Y. Huang, S. Chen, X. Ma, Z. Yang and J. Wu, Chin. Chem. Lett., 2024, 35, 109005 CrossRef CAS.
- A. J. Kochanowska-Karamyan and M. T. Hamann, Chem. Rev., 2010, 110, 4489 CrossRef CAS PubMed.
- F. Zhao, W. Hagui, M. Ballarín-Marión, C. Ollivier, V. Mouriès-Mansuy and L. Fensterbank, ACS Org. Inorg. Au, 2022, 3, 19 CrossRef.
- F. Piscitelli, A. Coluccia, A. Brancale, G. La Regina, A. Sansone, C. Giordano, A. Samuele, R. Cirilli, F. La Torre, A. Lavecchia, E. Novellino and R. Silvestri, J. Med. Chem., 2009, 52, 1922 CrossRef CAS PubMed.
- (a) R. Bernotas, S. Lenicek, S. Antane, G. M. Zhang, D. Smith, J. Coupet, B. Harrison and L. E. Schechter, Bioorg. Med. Chem. Lett., 2004, 14, 5499 CrossRef CAS PubMed; (b) R. C. Bernotas, S. Antane, R. Shenoy, V.-D. Le, B. L. Harrison, A. J. Robichaud, G. M. Zhang, D. Smith and L. E. Schechter, Bioorg. Med. Chem. Lett., 2010, 20, 1657 CrossRef CAS PubMed.
- C. T. Supuran, Expert Opin. Investig. Drugs, 2003, 12, 283 CrossRef CAS PubMed.
- (a) J. K. Qiu, W. J. Hao, D. C. Wang, P. Wei, J. Sun, B. Jiang and S. J. Tu, Chem. Commun., 2014, 50, 14782 RSC; (b) D. Xia, L. Jiang, Y. Li and J. Kong, Org. Biomol. Chem., 2024, 22, 5511 RSC; (c) N. Chakraborty, A. Dahiya, A. Rakshit, A. Modi and B. K. Patel, Org. Biomol. Chem., 2021, 19, 6847 RSC.
- (a) A. R. Katritzky, X. Lan and J. N. Lam, J. Org. Chem., 1991, 56, 4397 CrossRef CAS; (b) A. R. Katritzky and S. Rachwal, Chem. Rev., 2010, 110, 1564 CrossRef CAS PubMed.
- For Selected Reviews: (a) M. Akter, K. Rupa and P. Anbarasan, Chem. Rev., 2022, 122, 13108 CrossRef CAS PubMed; (b) Y. Wang, Z. Wang and Y. Tang, Chem. Rec., 2020, 20, 693 CrossRef CAS PubMed.
- (a) O. Dimroth, Justus Liebigs Ann. Chem., 1909, 364, 183–226 CrossRef CAS; (b) S. J. Barker and R. C. Storr, J. Chem. Soc., Perkin Trans. 1, 1990, 485 RSC; (c) P. Vera-Luque, R. Alajarín, J. Alvarez-Builla and J. J. Vaquero, Org. Lett., 2006, 8, 415 CrossRef CAS PubMed.
- I. Nakamura, T. Nemoto, N. Shiraiwa and M. Terada, Org. Lett., 2009, 11, 1055 CrossRef CAS PubMed.
- (a) M. Teders, L. Pitzer, S. Buss and F. Glorius, ACS Catal., 2017, 7, 4053 CrossRef CAS; (b) V. Helan, A. V. Gulevich and V. Gevorgyan, Chem. Sci., 2015, 6, 1928 RSC; (c) S. W. Kwok, L. Zhang, N. P. Grimster and V. V. Fokin, Angew. Chem., Int. Ed., 2014, 53, 3452 CrossRef CAS PubMed.
- (a) M. S. Yadav, A. S. Singh, A. K. Agrahari, N. Mishra and V. K. Tiwari, ACS Omega, 2019, 4, 6681 CrossRef CAS PubMed; (b) M. Singh, P. Bose, A. S. Singh and V. K. Tiwari, Tetrahedron, 2020, 76, 131078 CrossRef CAS.
- Y. Su, J. L. Petersen, T. L. Gregg and X. Shi, Org. Lett., 2015, 17, 1208 CrossRef CAS PubMed.
- J. Li, S. Wang, J. Zhao and P. Li, Org. Lett., 2022, 24, 5977 CrossRef CAS PubMed.
- (a) M. Sharma, D. Thakur and A. K. Verma, Chem. Commun., 2024, 60, 12840 RSC; (b) A. K. Verma, T. Kesharwani, J. Singh and R. C. Larock, Angew. Chem., Int. Ed., 2009, 48, 1158 CrossRef; (c) A. K. Verma, J. Singh and R. C. Larock, Tetrahedron, 2009, 65, 8434 CrossRef CAS; (d) D. Thakur, S. A. Meena, M. Sharma and A. K. Verma, Chem. Commun., 2025, 61, 5313 RSC; (e) S. A. Meena, D. Thakur, R. Ranjan and A. K. Verma, Chem. Commun., 2024, 60, 9230 RSC.
- (a) H. Long, K. Xu, S. Chen, J. Lin, D. Wu, B. Wu, X. Tian and L. Ackermann, Org. Lett., 2019, 21, 3053 CrossRef CAS PubMed; (b) A. K. Sahoo, A. Dahiya, B. Das, A. Behera and B. K. Patel, J. Org. Chem., 2021, 86, 11968 CrossRef CAS PubMed.
- (a) CCDC 2453343: Experimental Crystal Structure Determination, 2025 DOI:10.5517/ccdc.csd.cc2nbx31; (b) CCDC 2453064: Experimental Crystal Structure Determination, 2025 DOI:10.5517/ccdc.csd.cc2nbm3r; (c) CCDC 2495886: Experimental Crystal Structure Determination, 2025 DOI:10.5517/ccdc.csd.cc2ps5g4.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2025 |


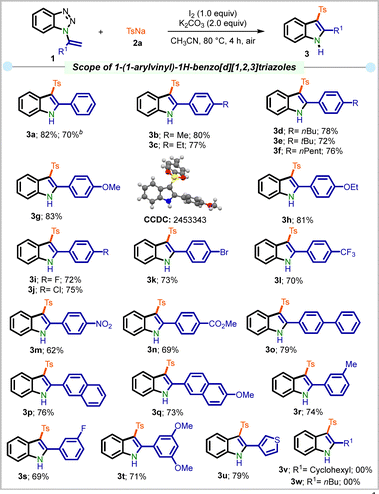
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Gram scale synthesis using 4.52 mmol of 1a.
Gram scale synthesis using 4.52 mmol of 1a.