Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
A multicomponent approach for the stereoselective synthesis of atropisomeric N–N peptide analogues
Natalie J.
Roper
 a,
George M.
Hardy
a,
George M.
Hardy
 a,
Jack M.
Wootton
a,
Jack M.
Wootton
 a,
Paul G.
Waddell
a,
Paul G.
Waddell
 a,
James
Wilson
b and
Roly J.
Armstrong
a,
James
Wilson
b and
Roly J.
Armstrong
 *a
*a
aChemistry – School of Natural and Environmental Sciences, Newcastle University, Newcastle Upon Tyne, NE1 7RU, UK. E-mail: roly.armstrong@newcastle.ac.uk
bChemical Development, Pharmaceutical Technology and Development, AstraZeneca, Macclesfield Campus, Macclesfield, SK10 2NA, UK
First published on 13th October 2025
Abstract
Peptide analogues featuring both central and N–N axial chirality were efficiently synthesised using a 4-component Ugi reaction. This method demonstrates a broad substrate scope, affording the target compounds with excellent stereoselectivity (up to >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 d.r.). Thermal atropisomerization studies revealed the identity of the substituents significantly influences the configurational stability and thermodynamic preferences of the atropisomeric products.
5 d.r.). Thermal atropisomerization studies revealed the identity of the substituents significantly influences the configurational stability and thermodynamic preferences of the atropisomeric products.
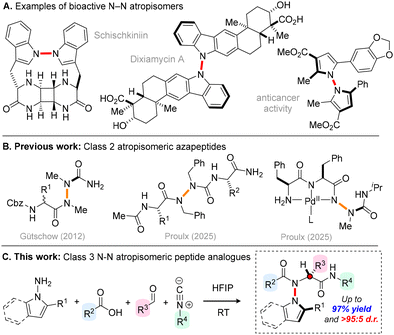
Atropisomers are valuable chiral molecules whose stereogenicity arises via restricted rotation about a single bond. To date, most studies in this area have explored systems whose chirality is derived from restricted C–C or C–N bond rotation,1 but recently atropisomeric compounds featuring a stereogenic N–N axis have gained prominence as valuable bioactive scaffolds (Scheme 1A).2 For example, the N–N atropisomeric natural product Schischkiniin exhibits activity against Caco-2 colon cancer cell lines,3 and the indolosesquiterpene alkaloid dixiamycin A is active against several strains of bacteria.4 N–N atropisomerism is also a key structural element in non-natural bioactive small molecules, for example bispyrroles reported by Zhang, Shi and co-workers, displaying promising activity against QGP-1 cancer cells.5 The emerging utility of these scaffolds has inspired the development of various powerful strategies for the synthesis of N–N atropisomers, including N-functionalization, de novo ring formation, C–H functionalization and desymmetrization approaches.6
Despite these advances, the integration of N–N atropisomerism into peptidomimetics (molecules designed to mimic the structure and function of peptides) remains markedly underexplored. A pioneering contribution in this area was reported by Gütschow and co-workers, who showed that bis-methylated azadipeptides could be prepared via N-acylation (Scheme 1B).7 These scaffolds were shown to display class 2 atropisomerism, with free energy barriers for enantiomerization (ΔG‡) up to 117 kJ mol−1. Substituted α-amino acids (R1 ≠ H) could be introduced, leading to the formation of diastereoisomeric mixtures of products. Very recently, Proulx and co-workers explored the late-stage alkylation of more complex azapeptides, affording class 1–2 atropisomeric dialkylated azapeptides (ΔG‡ = 63–100 kJ mol−1).8 Proulx and co-workers have also reported evidence of atropisomerism within metallo-azapeptides, where co-ordination of the hydrazide nitrogen to a palladium centre imparts restricted N–N rotation.9 Despite these important advances, the synthesis of N–N atropisomeric peptide frameworks remains significantly underdeveloped. In particular, no general methods are available to access configurationally robust class 3 atropisomeric N–N atropisomeric peptide analogues, or to achieve kinetic control over their relative stereochemistry.10 With these challenges in mind, based upon our recent work on C–N atropisomeric scaffolds, we envisioned that it might be possible to prepare N–N atropisomeric peptide analogues via a four-component Ugi reaction between sterically encumbered N-aminated heterocycles, aldehydes, carboxylic acids and isocyanides (Scheme 1C).10a Based upon previous reports, we believed that these products would display stable atropisomerism and the proposed methodology could provide an efficient and modular approach for their diastereocontrolled synthesis.6 Herein, we report the successful realization of this goal, with a library of 22 N–N atropisomeric peptide analogues prepared in up to 97% yield and >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 d.r. Kinetic epimerization studies reveal these scaffolds to be configurationally robust class 3 atropisomers, with ΔG‡ = 132–144 kJ mol−1, with the substitution pattern playing a key role in dictating the rate and position of equilibrium in the atropisomerization process.
5 d.r. Kinetic epimerization studies reveal these scaffolds to be configurationally robust class 3 atropisomers, with ΔG‡ = 132–144 kJ mol−1, with the substitution pattern playing a key role in dictating the rate and position of equilibrium in the atropisomerization process.
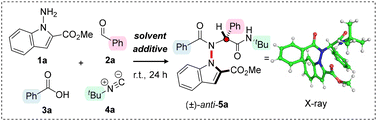
We commenced our study by exploring a model multicomponent reaction between methyl 1-amino-1H-indole-2-carboxylate (1a), benzaldehyde (2a), benzoic acid (3a), and tert-butyl isocyanide (4a) in 2,2,2-trifluoroethanol (TFE) at room temperature for 24 hours. Pleasingly, the intended Ugi product 5a was obtained in a modest, but promising 17% yield, and analysis of the crude reaction mixture confirmed that the reaction proceeds with excellent diastereocontrol (>95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 d.r.) (Table 1, entry 1). The relative configuration of the product was unambiguously confirmed using single crystal X-ray analysis, revealing the ester group of the indole and the α-phenyl substituent to have an anti-arrangement. Encouraged by this result, we focused on optimising the reaction conditions. Upon replacing the solvent with 1,1,1,3,3,3-hexafluoroisopropanol (HFIP), a substantial increase in yield to 69% was observed (Table 1, entry 2). Furthermore, employing an excess of the isocyanide (2.0 equivalents) led to a further improvement, with the desired product obtained in 79% yield and >95
5 d.r.) (Table 1, entry 1). The relative configuration of the product was unambiguously confirmed using single crystal X-ray analysis, revealing the ester group of the indole and the α-phenyl substituent to have an anti-arrangement. Encouraged by this result, we focused on optimising the reaction conditions. Upon replacing the solvent with 1,1,1,3,3,3-hexafluoroisopropanol (HFIP), a substantial increase in yield to 69% was observed (Table 1, entry 2). Furthermore, employing an excess of the isocyanide (2.0 equivalents) led to a further improvement, with the desired product obtained in 79% yield and >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 d.r. (Table 1, entry 3). We then explored whether addition of a desiccant might improve the efficiency of the transformation by promoting imine formation. While 4 Å molecular sieves produced inferior results, both 5 Å and 3 Å molecular sieves led to slightly increased yields of 80% and 82%, respectively (Table 1, entries 4–6). Finally, we turned our attention back to reagent stoichiometry. Limiting the amount of aldehyde 2a in the reaction resulted in a slightly decreased yield (Table 1, entry 7). In contrast, employing aminated indole 1a as the limiting reagent led to a significant increase in conversion, enabling the isolation of the product 5a by column chromatography in 89% yield as a single diastereoisomer (Table 1, entry 8). The selectivity for the anti-diastereoisomer was confirmed via a thermal epimerization experiment, which enabled unambiguous characterization of syn-5a (vide infra).
5 d.r. (Table 1, entry 3). We then explored whether addition of a desiccant might improve the efficiency of the transformation by promoting imine formation. While 4 Å molecular sieves produced inferior results, both 5 Å and 3 Å molecular sieves led to slightly increased yields of 80% and 82%, respectively (Table 1, entries 4–6). Finally, we turned our attention back to reagent stoichiometry. Limiting the amount of aldehyde 2a in the reaction resulted in a slightly decreased yield (Table 1, entry 7). In contrast, employing aminated indole 1a as the limiting reagent led to a significant increase in conversion, enabling the isolation of the product 5a by column chromatography in 89% yield as a single diastereoisomer (Table 1, entry 8). The selectivity for the anti-diastereoisomer was confirmed via a thermal epimerization experiment, which enabled unambiguous characterization of syn-5a (vide infra).
| Entry | Solvent (0.5 M) |
1a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2a 2a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3a 3a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4a 4a |
Additive | Yield anti-5a (%)a | d.r. anti-5ab |
|---|---|---|---|---|---|
| a Yields determined by quantitative 1H NMR analysis vs. (CH3)2SO2 as an internal standard; values in parentheses indicate the yield of isolated product. b Diastereoselectivity determined by 1H NMR analysis of the crude reaction mixture. | |||||
| 1 | TFE | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
— | 17 | >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
| 2 | HFIP | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
— | 69 | >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
| 3 | HFIP | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
— | 79 | >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
| 4 | HFIP | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
4 Å MS | 76 | >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
| 5 | HFIP | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
5 Å MS | 80 | >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
| 6 | HFIP | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
3 Å MS | 82 | >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
| 7 | HFIP | 1.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.2 1.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
3 Å MS | 81 | >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
| 8 | HFIP | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.2 1.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.2 1.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
3 Å MS | 87 (89) | >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 5 |
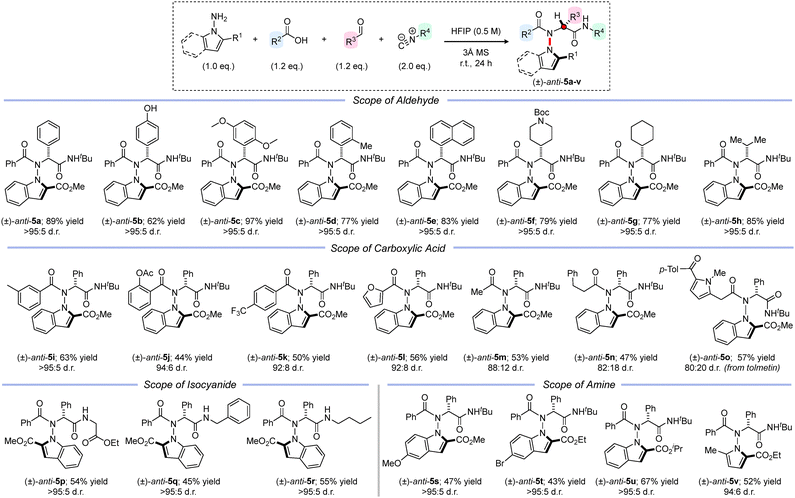
With optimal conditions in hand, we next investigated the generality of the reaction (Scheme 2). A range of aromatic aldehydes, including examples bearing unprotected phenols (anti-5b) and sterically demanding groups (anti-5c–5e), afforded the corresponding products in high yields and excellent diastereoselectivity (>95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 d.r.). Encouragingly, the methodology also proved effective with aliphatic aldehydes, with products anti-5f–5h isolated in similarly high yields and stereoselectivity. We next explored the scope of the carboxylic acid partner. Electron-rich benzoic acid analogues were well tolerated, furnishing products anti-5i and anti-5j in reasonable yields and high diastereomeric ratios (>95
5 d.r.). Encouragingly, the methodology also proved effective with aliphatic aldehydes, with products anti-5f–5h isolated in similarly high yields and stereoselectivity. We next explored the scope of the carboxylic acid partner. Electron-rich benzoic acid analogues were well tolerated, furnishing products anti-5i and anti-5j in reasonable yields and high diastereomeric ratios (>95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 and 94
5 and 94![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 d.r. respectively). Electron-deficient and heteroaromatic carboxylic acids were also effective substrates, delivering atropisomeric products anti-5k–5l with good stereoselectivity (92
6 d.r. respectively). Electron-deficient and heteroaromatic carboxylic acids were also effective substrates, delivering atropisomeric products anti-5k–5l with good stereoselectivity (92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 d.r.). Aliphatic carboxylic acids also underwent the desired multicomponent reaction, enabling access to peptide analogues anti-5m–5o in good yields albeit with slightly diminished diastereoselectivities (80
8 d.r.). Aliphatic carboxylic acids also underwent the desired multicomponent reaction, enabling access to peptide analogues anti-5m–5o in good yields albeit with slightly diminished diastereoselectivities (80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20–88
20–88![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 d.r.). Collectively, these results demonstrate the steric and electronic properties of the acid component significantly influence the stereochemical outcome of the reaction. A selection of commercially available isocyanides were examined, which all reacted smoothly under the reaction conditions, delivering compounds anti-5p–5r with essentially complete diastereocontrol (>95
12 d.r.). Collectively, these results demonstrate the steric and electronic properties of the acid component significantly influence the stereochemical outcome of the reaction. A selection of commercially available isocyanides were examined, which all reacted smoothly under the reaction conditions, delivering compounds anti-5p–5r with essentially complete diastereocontrol (>95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 d.r.). Finally, we explored the scope of the heterocyclic locking group. Aminated indoles bearing different backbone substituents reacted efficiently, affording the corresponding products anti-5s–5t in moderate to good yields and excellent diastereoselectivity (>95
5 d.r.). Finally, we explored the scope of the heterocyclic locking group. Aminated indoles bearing different backbone substituents reacted efficiently, affording the corresponding products anti-5s–5t in moderate to good yields and excellent diastereoselectivity (>95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 d.r.). The ester substituent could also be varied with an isopropyl analogue anti-5u obtained in 67% yield and >95
5 d.r.). The ester substituent could also be varied with an isopropyl analogue anti-5u obtained in 67% yield and >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 d.r. Finally, we found that 2,5-disubstituted aminated pyrroles could also be successfully employed in the reaction, yielding atropisomeric peptide anti-5v with similarly high stereoselectivity (94
5 d.r. Finally, we found that 2,5-disubstituted aminated pyrroles could also be successfully employed in the reaction, yielding atropisomeric peptide anti-5v with similarly high stereoselectivity (94![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 d.r.).
6 d.r.).
To assess the configurational stability of our N–N atropisomeric peptide analogues, we explored thermal epimerization about the stereogenic axis.11 Model substrate anti-5a was heated to 120 °C in DMSO-d6 for several days, with aliquots removed at regular intervals and analysed by 1H NMR spectroscopy (Scheme 3, gray dataset). Under these conditions, we were pleased to observe smooth epimerization to syn-5a which was formed in 88![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 d.r. with equilibrium achieved after approximately 60 hours. Note that the structure and relative stereochemistry of syn-5a were unambiguously confirmed by single crystal X-ray analysis. From the kinetic profile shown in Scheme 3, values for ΔG‡ of 132 kJ mol−1 and 139 kJ mol−1 for the respective forward and backward isomerization processes of anti-5a were calculated (see SI for details), confirming class 3 atropisomerism. Furthermore, based upon the final equilibrium ratio of 88
12 d.r. with equilibrium achieved after approximately 60 hours. Note that the structure and relative stereochemistry of syn-5a were unambiguously confirmed by single crystal X-ray analysis. From the kinetic profile shown in Scheme 3, values for ΔG‡ of 132 kJ mol−1 and 139 kJ mol−1 for the respective forward and backward isomerization processes of anti-5a were calculated (see SI for details), confirming class 3 atropisomerism. Furthermore, based upon the final equilibrium ratio of 88![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 d.r. we calculated a standard Gibbs free energy difference (ΔG°) between the diastereomers of 5a of −6.4 kJ mol−1, indicating a clear thermodynamic preference for the syn-isomer, which is in line with our previous work on structurally related C–N atropisomeric peptide analogues.10a
12 d.r. we calculated a standard Gibbs free energy difference (ΔG°) between the diastereomers of 5a of −6.4 kJ mol−1, indicating a clear thermodynamic preference for the syn-isomer, which is in line with our previous work on structurally related C–N atropisomeric peptide analogues.10a
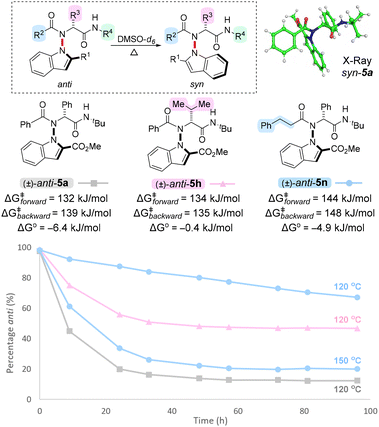 | ||
| Scheme 3 Thermal isomerization of N–N atropisomeric peptide analogues. Aliquots were removed at regular time intervals with d.r. determined by quantitative 1H NMR analysis (see SI for details). | ||
To explore how the steric and electronic properties of different substituents may influence the stability of these N–N atropisomeric peptide analogues, an analogous epimerization study was performed with anti-5h, which bears an isopropyl group α- to the carbonyl of the secondary amide (Scheme 3, pink dataset). In this case, isomerization proceeded at a similar rate (ΔGforward‡ = 134 kJ mol−1; ΔGbackward‡ = 135 kJ mol−1), but intriguingly an almost equimolar ratio of anti- and syn-5h was obtained at equilibrium (ΔG° = −0.4 kJ mol−1). This result indicates that replacing the α-substituent with an aliphatic group preserves class 3 atropisomerism, but effectively eliminates the thermodynamic preference for the syn-diastereoisomer. Finally, we investigated the isomerization of anti-5n, which bears an aliphatic acyl substituent (Scheme 3, blue dataset). Interestingly, in this case epimerization proceeded very slowly when heated to 120 °C, indicating significantly increased configurational stability. Therefore, an additional thermal isomerization experiment was conducted at 150 °C, under which conditions the system reached an equilibrium ratio of 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 (ΔG° = −4.9 kJ mol−1), which is similar to that of anti-5a. Based upon this data we calculated values of ΔG‡ of 144 kJ mol−1 and 148 kJ mol−1 for the respective forward and backward isomerization processes, confirming very high configurational stability.
20 (ΔG° = −4.9 kJ mol−1), which is similar to that of anti-5a. Based upon this data we calculated values of ΔG‡ of 144 kJ mol−1 and 148 kJ mol−1 for the respective forward and backward isomerization processes, confirming very high configurational stability.
Taken together, these results may imply that the α-substituent predominantly dictates thermodynamic stability, whereas the acyl substituent impacts the kinetics of epimerization. The substantially slower rate of isomerization for examples bearing an aliphatic acyl substituent is in line with our previous work on C–N atropisomeric amides, which was attributed to a racemization mechanism involving correlated bond rotation.12 We tentatively conclude that a similar mechanism involving geared rotation of the N–N and N–CO bonds may also be operative in N–N atropisomeric amides.
In summary, we have developed an efficient multicomponent approach for the synthesis of novel N–N atropoisomeric peptide analogues which proceeds in excellent yields (up to 97%) with very high levels of kinetic selectivity in favour of the anti-configured diastereomer (up to >95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 d.r.). Thermal isomerization studies reveal that these molecules are configurationally stable class 3 atropisomers (ΔG‡ ≥ 132 kJ mol−1). The thermodynamics and kinetics of the epimerization process appear to be substantially influenced by the α- and acyl substituents respectively, and further work is underway in our laboratory to explore the generality of these observations and to assess their implications regarding the mechanism of N–N atropisomerization.
5 d.r.). Thermal isomerization studies reveal that these molecules are configurationally stable class 3 atropisomers (ΔG‡ ≥ 132 kJ mol−1). The thermodynamics and kinetics of the epimerization process appear to be substantially influenced by the α- and acyl substituents respectively, and further work is underway in our laboratory to explore the generality of these observations and to assess their implications regarding the mechanism of N–N atropisomerization.
We gratefully acknowledge financial support from EPSRC (EP/S022791/1, UKRI124 and EP/W524700/1) and Newcastle University. N. J. R. thanks the Bill and Milica Beck PhD Endowment Fund. G. M. H. gratefully acknowledges AstraZeneca for studentship support. We are grateful to Dr Corinne Wills and Dr Casey Dixon (Newcastle University) for assistance with NMR experiments.
Conflicts of interest
There are no conflicts to declare.Data availability
The data underlying this study are available in the published article and in the supplementary information (SI). Supplementary information: detailed experimental procedures and characterization data for new compounds. See DOI: https://doi.org/10.1039/d5cc05091j.CCDC 2476444 and 2476445 contain the supplementary crystallographic data for this paper.13a,b
Notes and references
- For selected review articles, see: (a) T. A. Schmidt, V. Hutskalova and C. Sparr, Nat. Rev. Chem., 2024, 8, 497–517 CrossRef PubMed; (b) S.-H. Xiang, W.-Y. Ding, Y.-B. Wang and B. Tan, Nat. Catal., 2024, 7, 483–498 CrossRef CAS; (c) J. S. Sweet and P. C. Knipe, Synthesis, 2022, 2119–2132 CAS; (d) Y.-J. Wu, G. Liao and B.-F. Shi, Green Synth. Catal., 2022, 3, 117–136 Search PubMed; (e) P. Rodríguez-Salamanca, R. Fernández, V. Hornillos and J. M. Lassaletta, Chem. – Eur. J., 2022, 28, e202104442 CrossRef PubMed; (f) A. D. G. Campbell and R. J. Armstrong, Synthesis, 2023, 2427–2438 CAS.
- Z. Wang, L. Meng, X. Liu, L. Zhang, Z. Yu and G. Wu, Eur. J. Med. Chem., 2022, 243, 114700 CrossRef CAS PubMed.
- M. Shoeb, S. Celik, M. Jaspars, Y. Kumarasamy, S. M. MacManus, L. Nahar, P. K. Thoo-Lin and S. D. Sarker, Tetrahedron, 2005, 61, 9001–9006 CrossRef CAS.
- Q. Zhang, A. Mándi, S. Li, Y. Chen, W. Zhang, X. Tian, H. Zhang, H. Li, W. Zhang, S. Zhang, J. Ju, T. Kurtán and C. Zhang, Eur. J. Org. Chem., 2012, 5256–5262 CrossRef CAS.
- K.-W. Chen, Z.-H. Chen, S. Yang, S.-F. Wu, Y.-C. Zhang and F. Shi, Angew. Chem., Int. Ed., 2022, 61, e202116829 CrossRef CAS PubMed.
- For selected review articles, see: (a) G. Centonze, C. Portolani, P. Righi and G. Bencivenni, Angew. Chem., Int. Ed., 2023, 62, e202303966 CrossRef PubMed; (b) G.-J. Mei, W. L. Koay, C.-Y. Guan and Y. Lu, Chem, 2022, 8, 1855–1893 CrossRef CAS; (c) H.-H. Zhang and F. Shi, Acc. Chem. Res., 2022, 55, 2562–2580 CrossRef CAS PubMed; (d) J. Feng, C.-J. Lu and R.-R. Liu, Acc. Chem. Res., 2023, 56, 2537–2554 CrossRef CAS PubMed; (e) J. Feng and R.-R. Liu, Chem. – Eur. J., 2024, 30, e202303165 CrossRef CAS PubMed.
- (a) P. A. Ottersbach, G. Schnakenburg and M. Gütschow, Chem. Commun., 2012, 48, 5772–5774 RSC; (b) P. A. Ottersbach, G. Schnakenburg and M. Gütschow, Tetrahedron Lett., 2015, 56, 4889–4891 CrossRef CAS.
- M. E. Helton, C. S. Howard, K. Prieto Bruno, K. D. Cartrette, M. O. Bowles, W. A. Hearne and C. Proulx, J. Org. Chem., 2025, 90, 9549–9558 CrossRef CAS PubMed.
- M. O. Bowles, E. L. Willis, M. D. Trombley, C.-H. Chen and C. Proulx, J. Am. Chem. Soc., 2025, 147, 1404–1410 CrossRef CAS PubMed.
- For C–N and C–C atropisomeric peptidomimetics, see: (a) N. J. Roper, A. D. G. Campbell, P. G. Waddell, A. K. Brown, K. Ermanis and R. J. Armstrong, Chem. Sci., 2024, 15, 16743–16751 Search PubMed; (b) B. Paul, G. L. Butterfoss, M. G. Boswell, P. D. Renfrew, F. G. Yeung, N. H. Shah, C. Wolf, R. Bonneau and K. Kirshenbaum, J. Am. Chem. Soc., 2011, 133, 10910–10919 CrossRef CAS PubMed; (c) B. Paul, G. L. Butterfoss, M. G. Boswell, M. L. Huang, R. Bonneau, C. Wolf and K. Kirshenbaum, Org. Lett., 2012, 14, 926–929 CrossRef CAS PubMed; (d) Y.-J. Wu, P.-P. Xie, G. Zhou, Q.-J. Yao, X. Hong and B.-F. Shi, Chem. Sci., 2021, 12, 9391–9397 Search PubMed; (e) Z.-S. Jia, Y.-J. Wu, Q.-J. Yao, X.-T. Xu, K. Zhang and B.-F. Shi, Org. Lett., 2022, 24, 304–308 CrossRef CAS PubMed; (f) I. J. Knudson, T. L. Dover, D. A. Dilworth, C. Paloutzian, O. Paz, J. Lin, H. Cho, T. Gonen, A. Schepartz and S. J. Miller, Nat. Chem., 2025 DOI:10.1038/s41557-025-01935-4; (g) T.-Y. Jiang, Y.-T. Ke, Y.-J. Wu, Q.-J. Yao and B.-F. Shi, Chem. Commun., 2023, 59, 13518–13521 RSC; (h) M. C. Morejón, A. Laub, B. Westermann, D. G. Rivera and L. A. Wessjohann, Org. Lett., 2016, 18, 4096–4099 CrossRef PubMed; (i) P. W. Glunz, L. Mueller, D. L. Cheney, V. Ladziata, Y. Zou, N. R. Wurtz, A. Wei, P. C. Wong, R. R. Wexler and E. S. Priestley, J. Med. Chem., 2016, 59, 4007–4018 CrossRef CAS PubMed; (j) N. K. Mothahalli Raju, B. Paul, L. TN, S. Bodduna and N. K. Kandukuri, J. Org. Chem., 2025, 90, 4796–4807 CrossRef CAS PubMed.
- J.-P. Heeb, J. Clayden, M. D. Smith and R. J. Armstrong, Nat. Protoc., 2023, 18, 2745–2771 CrossRef CAS PubMed.
- A. D. G. Campbell, N. J. Roper, P. G. Waddell, C. Wills, C. M. Dixon, R. M. Denton, K. Ermanis and R. J. Armstrong, Chem. Commun., 2024, 60, 3818–3821 RSC.
- (a) CCDC 2476444: Experimental Crystal Structure Determination, 2025 DOI:10.5517/ccdc.csd.cc2p3y92; (b) CCDC 2476445: Experimental Crystal Structure Determination, 2025 DOI:10.5517/ccdc.csd.cc2p3yb3.
| This journal is © The Royal Society of Chemistry 2025 |