Controllable growth of two dimensional stereoscopic PtTe2 nanosheets for efficient electrocatalytic hydrogen evolution
Jiatian
Fu†
a,
You
Peng†
b,
Liang
Zhou
c,
Jialong
Wang
a,
Yahuan
Huan
a,
Tong
Zhou
b,
Haoxuan
Ding
a,
Rongming
Wang
 c and
Yanfeng
Zhang
c and
Yanfeng
Zhang
 *ab
*ab
aSchool of Materials Science and Engineering, Peking University, Beijing 100871, P. R. China. E-mail: yanfengzhang@pku.edu.cn
bAcademy for Advanced Interdisciplinary Studies, School of Materials Science and Engineering, Peking University, Beijing 100871, P. R. China
cBeijing Advanced Innovation Center for Materials Genome Engineering, Beijing Key Laboratory for Magneto-Photoelectrical Composite and Interface Science, School of Mathematics and Physics, University of Science and Technology Beijing, Beijing 100083, P. R. China. E-mail: rmwang@ustb.edu.cn
First published on 21st October 2025
Abstract
Among the transition metal dichalcogenide family, two-dimensional (2D) PtTe2 has emerged as a promising electrocatalyst for the hydrogen evolution reaction (HER), due to its high edge catalytic activity, excellent conductivity and chemical stability, etc. However, the active sites of 2D PtTe2 synthesized on conventional planar insulating substrates are limited to the edges, and the traditional synthetic method is not compatible with low-cost batch production. Herein, we report the large-area uniform growth of stereoscopic PtTe2 nanosheets on highly conductive, carbon cloth substrates featuring abundant active sites and tunable thicknesses (from ∼3.0 nm to bulk state), via a facile chemical vapor deposition route. We find that the stereoscopic structure of PtTe2 on the carbon fiber surface can facilitate efficient electron transport from the conductive carbon cloth to the active edge sites of the PtTe2 nanosheets along the in-plane direction. And the catalytic activity can be significantly improved by increasing the average nanosheet thickness from ∼3.0 to 7.5 nm, leading to an ultra-low overpotential of ∼38.8 mV at the current density of 10 mA cm−2 and a rather low Tafel slope of ∼59.2 mV dec−1. Hereby, this work is expected to deepen our understanding of the thickness-dependent catalytic activity of 2D layered materials, and stimulate further structure design and synthesis explorations of 2D catalysts on 3D templates for highly efficient water splitting related applications.
Among the huge transition metal dichalcogenide (TMDC) family, two-dimensional (2D) PtTe2 has drawn tremendous attention, due to its unique properties (e.g., type-II Dirac fermion,1 excellent conductivity2 and robust air stability3), and various applications in electrocatalysis,4 terahertz emission, mid-infrared photodetection,3etc. As electrocatalysts in the hydrogen evolution reaction (HER), 2D PtTe2 nanosheets exhibit high edge catalytic activities, due to the ultra-low hydrogen adsorption Gibbs free energy (∼0.09 eV by theoretical calculations).5 Intriguingly, distinguished from common TMDCs (e.g., MoS26 and ReS2) with fewer d-electrons, the d orbitals of group-10 metal atoms were nearly fully occupied, and the pz orbitals of interlayer chalcogen atoms of related TMDCs were highly hybridized. This resulted in strong interlayer interactions, and strong thickness-dependent electronic structures/properties.7 The catalytic activity of group-10 metal related TMDCs should hereby correlate with their thicknesses and edge sites, since thicker flakes expose more catalytically active sites and exhibit tunable band gaps (even to zero). For example, PtSe2 displays an exceptional bandgap transition (from ∼1.28 to 0.23, and 0 eV) from monolayer to trilayer and bulk state,8 enhanced conductivity from bilayer (∼4 × 102 S m−1) to twenty layers (∼2 × 105 S m−1),9 and relatively high catalytic activities.9
Similarly, 2D PtTe2 also displayed a semiconducting-to-metallic transition (bandgap ∼0.5–0 eV) from monolayer to bilayer.10 It crossed from a 2D metal to a three-dimensional (3D) type-II Dirac semimetal with increasing thicknesses from 2–3 to 4–6 layers, for molecular beam epitaxy (MBE) derived PtTe2 films.11 Besides affecting the electronic structure, the thickness of PtTe2 also determines the loading amount of Pt, which makes identifying an optimal PtTe2 nanosheet thickness crucial for maximizing the HER activity while reducing the noble metal usage. In addition, theoretical predictions revealed that the catalytically active sites of intrinsic 2D PtTe2 for the HER were situated on the edges, while its basal plane was inert.12 So far, PtTe2 islands,10 nanosheets2 and thin films3 have been achieved on 2D planar substrates (e.g., 6H-SiC(0001), mica and SiO2/Si) by MBE, CVD or thermal-assisted conversion routes.13
Compared with 2D planar nanosheets, catalysts with stereoscopic structures (e.g., CoS2 microwires or nanowire arrays and vertically-oriented MoS2, TaS2,14 NbS215 and PtSe216 nanosheets) can expose more edge sites, facilitate the depinning and releasing of H2 bubbles from the catalyst surfaces, and improve the overall HER performance. In addition, the stereoscopic structures can enable efficient electron transport from conductive substrates to the active edge sites of TMDCs along the in-plane direction, which can avoid the interlayer potential barrier effect.14 Motivated by these results, we have designed a facile atmospheric-pressure CVD (APCVD) route to achieve the direct synthesis of stereoscopic PtTe2 nanosheets with tunable thicknesses on low-cost, scalable, highly conductive carbon cloth (CC) substrates. Carbon fibers with highly curved surfaces were selected to induce high density nucleation, and the stereoscopic growth of 2D PtTe2 nanosheets. A metal-precursor pre-coating strategy was developed to guarantee the uniform, controllable supply of metal precursor, thus enabling the uniform growth of stereoscopic PtTe2 nanosheets, in line with the recent batch production of 2D materials. The achieved 3D hybrids were selected as perfect candidates to explore the thickness-dependent electrocatalytic properties in the HER. Overall, this work is envisioned to propel spatial structural design and performance improvement in PtTe2-based catalysts.
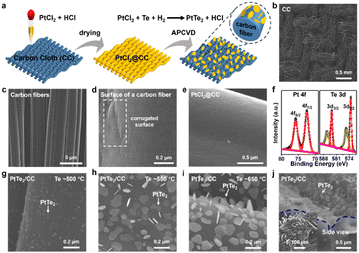
The APCVD growth of 3D vertically-oriented 1T-TaS214 and 1T-PtSe216 nanosheets was realized by our group, by using nanoporous gold (NPG) as a substrate (Fig. S1, Fig. S2, SI). Herein, commercial carbon cloth (CC) is selected as the substrate for the growth of stereoscopic PtTe2 nanosheets (Fig. 1a), since commercial CC is woven from carbon fibers (Fig. 1b), showing good conductivity, excellent thermal/chemical stability and low-cost, etc. Moreover, the 3D curved surfaces (Fig. 1c) can provide large surface areas, enabling stereoscopic nucleation and growth of PtTe2 nanosheets (Fig. 1c and d). The uniform feeding of the metal precursor is also a key step, which was realized by dripping well-optimized PtCl2/HCl solution onto the CC substrate followed by a drying process (Fig. 1e).
A facile APCVD route was then executed, by using Te powder as the precursor (Fig. S3, SI). After APCVD growth (Te precursor at ∼650 °C), the chemical composition of the as-grown samples was first determined by X-ray photoelectron spectroscopy (XPS) (Fig. 1f). The Pt 4f7/2 and 4f5/2 peaks occur at binding energies of ∼72.5 and ∼75.8 eV, while the Te 3d5/2 and 3d3/2 peaks appear at ∼573.2 and ∼583.6 eV, respectively. These results agree well with the reported XPS spectra of mechanically exfoliated 1T-PtTe2 flakes, confirming the successful synthesis of 1T-PtTe2. Notably, the other two peaks at ∼575.6 and 586.0 eV are assigned to Te–O bonds (Fig. S4, SI), possibly due to the formation of TeO2 and its sub-oxide form (TeO2−x).3
The volatilization temperature of the Te precursor was found to be an essential parameter for modulating the growth morphology of PtTe2. The PtCl2@CC with the PtCl2 loading mass of ∼0.8 mg cm−2 was first selected, under the same growth temperature of ∼700 °C for ∼15 min, but with different volatilization temperatures of Te. By gradually increasing the temperature of Te from ∼500 °C, to ∼550 °C, to ∼600 °C, sparsely distributed PtTe2 nanosheets (∼30 nm in edge size) (Fig. 1g), high density irregular shaped flakes (∼100 nm) (Fig. 1h), and regular shaped flakes (truncated triangles or hexagons) were obtained on the carbon fibers (Fig. S5, SI), respectively. At ∼650 °C, the PtTe2 nanosheets showed increased thickness to ∼150 nm, and uniform truncated triangular or hexagonal shapes (Fig. 1i). In addition, the contrasts of the PtTe2 nanosheets gradually deepened with increasing Te temperature, indicating the increased nanosheet thicknesses, as similarly reported for the growth of few-layer WS2 flakes.
According to the representative side-view SEM image, the PtTe2 nanosheets were vertically or inclinedly grown on the highly curved surfaces of the carbon fibers, showing stereoscopic structures (Fig. 1j) (Fig. S6, SI). This growth behaviour is similar to the CVD growth of HfS2 on SiO2/Si17 or VSe2 on sapphire. The growth of PtTe2 should follow the dangling-bond-assisted self-seeding growth mechanism. Altogether, under the uniform supply of metal precursor, an optimal Te precursor temperature (i.e., ∼650 °C) is crucial for achieving large-area uniform, stereoscopic PtTe2 nanosheets with regular shapes on carbon fibers.
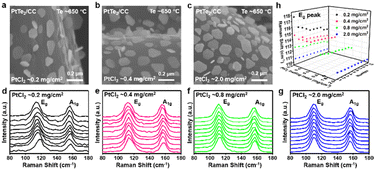
At the optimal Te temperature (∼650 °C), the morphologies of PtTe2 are investigated with varying loading mass of PtCl2 precursor (from ∼0.2 to 2.0 mg cm−2). Notably, the PtTe2 nanosheets present hardly changed flake sizes (Fig. 2a–c, ∼150 nm), showing the unique stereoscopic configurations with truncated triangular or hexagonal shapes. Notably, similar nucleation densities (∼35 nanosheets μm−2) and large-scale uniform morphologies can be achieved (Fig. S6–S9, SI), possibly due to the uniform supply of metal precursor via the designed metal precursor pre-coating strategy. As reported previously, the in-plane (Eg) and out-of-plane (A1g) modes of 2D PtTe2 redshift with increasing thicknesses.2 Herein, a series of Raman spectra were collected to extract the Eg peak positions (Fig. 2d-h) and average thickness of the PtTe2 flakes. With increasing PtCl2 loading amounts from ∼0.2 to 0.4, 0.8, and 2.0 mg cm−2, the Eg peaks redshift from ∼117.7–114.2 cm−1 to 114.6–112.9, 112.6–111.6, and 110.5–110.2 cm−1, corresponding to average thicknesses of ∼3.0, 5.0, 7.5, and ≥12 nm, respectively (Fig. 2d–g).
In summary, by adjusting the PtCl2 precursor amount, thickness-controlled growth of large-area uniform, stereoscopic PtTe2 nanosheets can be achieved on CC substrates. When directly used as electrocatalysts in the HER, electron transport from the carbon fibers to the active edge sites can be realized through in-plane rather than from out-of-plane directions (needs to overcome the interlayer potential barrier). This enables 2D stereoscopic PtTe2 nanosheets as perfect platforms in electrocatalytic related explorations.
High-resolution transmission electron microscopy (HR-TEM) and energy-dispersive X-ray spectroscopy (EDS) characterizations were then performed to examine the crystal quality and chemical composition of the CVD-derived PtTe2 nanosheets. As shown in Fig. 3a, PtTe2 nanosheets are distinguished in the low-magnification TEM image, presenting truncated triangular or hexagonal shapes. The stacked form arises from aggregation of nanosheets from the transfer process, due to the van der Waals interlayer interaction. The selected-area electron diffraction (SAED) pattern (Fig. 3b) presents a single set of diffraction spots with six-fold symmetry, suggesting the hexagonal crystal structure. Moreover, the HR-TEM image of the PtTe2 nanosheets (Fig. 3c, d and Fig. S10 in SI) demonstrates a perfect lattice with a spacing of ∼0.201 nm, consistent with the lattice spacing of the (110) plane of the 1T-PtTe2 flakes.2 Additional EDS mapping images confirm the large area uniform element ratio of Pt/Te ∼1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.94 (Fig. 3e–h). Overall, the CVD-derived stereoscopic PtTe2 nanosheets possess a 1T-phase structure and high crystal quality.
1.94 (Fig. 3e–h). Overall, the CVD-derived stereoscopic PtTe2 nanosheets possess a 1T-phase structure and high crystal quality.
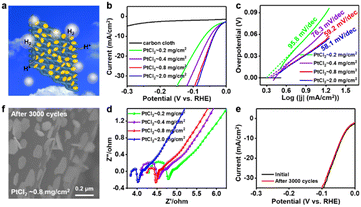
Furthermore, the as-grown stereoscopic PtTe2 nanosheets on CC substrates with different thicknesses are directly used for the electrocatalytic HER (schematic image in Fig. 4a). The polarization curves of the stereoscopic PtTe2 nanosheets are displayed in Fig. 4b, with average thicknesses varying from ∼3.0 nm (PtCl2 ∼0.2 mg cm−2), 5.0 nm (PtCl2 ∼0.4 mg cm−2), and 7.5 nm (PtCl2 ∼0.8 mg cm−2) to the bulk state (≥12 nm) (PtCl2 ∼2.0 mg cm−2). Notably, the overpotentials at the current density of 10 mA cm−2 (η10) significantly decrease from ∼65.6 to 48.5 to 38.8 mV (from ∼3.0 nm to 5.0 nm to 7.5 nm), and the Tafel slopes (extracted from the linear portions of the polarization curves) decrease accordingly from ∼95.8 to 76.3 to 59.2 mV dec−1 (Fig. 4c). The exchange current density (J0) (calculated by extrapolating the Tafel plot) (Fig. 4c) increases from ∼2.0 to 2.17 to 2.29 mA cm−2, possibly due to the increased active sites for the HER with increasing thicknesses. As a result, the catalytic activity of the stereoscopic PtTe2 nanosheets can be enhanced with increasing the PtTe2 thicknesses in a suitable range. Further electrochemical impedance spectroscopy (EIS) measurements were also performed to show the electrode kinetics (Fig. 4d), and the charge transfer resistances (Rct) vary from ∼0.46 to 0.38 to 0.18 Ω, accordingly. This indicates the faster electron transfer of the stereoscopic PtTe2 on CC during the HER process.
However, with increasing the thickness from ∼7.5 nm to bulk (≥12 nm), the η10, Tafel slope, J0 and Rct values change a little from ∼38.8 to 37.5 mV, from ∼59.2 to 58.1 mV dec−1, from ∼2.29 to 2.32 mA cm−2, and from ∼0.18 to 0.16 Ω (Fig. 4b–d), respectively. This nearly stable catalytic property is possibly due to the limited exposure of active edge sites in thicker nanosheets (>7.5 nm). Additionally, the long-term stability of the stereoscopic PtTe2 nanosheets is further evaluated by cyclic voltammetry (CV), which shows a negligible difference before and after 3000 cycles (Fig. 4e). And the PtTe2 nanosheets maintain the initial morphology after the HER tests (Fig. 4f). A comparison of the HER performances of various TMDC-based catalysts is also performed (Table S1, SI). The stereoscopic 2D PtTe2 nanosheets (∼7.5 nm thick) exhibit superior catalytic properties to many reported 2D TMDC-related catalysts, and comparable to noble-metal-based electrocatalysts. As a result, the developed 2D stereoscopic PtTe2 nanosheets on CC substrate systems can present relatively high electrocatalytic activity, low noble metal usage, excellent durability, facile production process, etc.
In summary, we have developed a facile APCVD route for the controllable growth of large-area uniform, thickness tunable stereoscopic PtTe2 nanosheets on CC substrates. The thickness-dependent catalytic activity is unveiled to find ultrathin nanosheets with relatively high catalytic properties, while possessing low noble metal use amount. Notably, nanometer-thick, stereoscopic 3D PtTe2 nanosheets can exhibit superior catalytic properties to many 2D TMDC catalysts, and comparable properties to noble-metal-based catalysts, showing the advantages of relatively high electrocatalytic activity, low noble metal usage, excellent durability, and facile production process, etc. Briefly, this catalytic system can serve as a practical model for examining the thickness-dependent catalytic activity, the internal mechanism and the property modulation of group-10 metal-related TMDCs. This work should promote the direct synthesis and application exploration of related catalysts in various energy related fields.
This work was financially supported by the National Natural Science Foundation of China (No. 92477204, T2188101, and 52021006) and Xianyang Science and Technology Bureau (Project L2024-ZDKJ-ZDCGZH-0009 and Project L2024-ZDYF-ZDYF-GY-0013).
Conflicts of interest
There are no conflicts to declare.Data availability
Data supporting this article are available in the supplementary information (SI). Supplementary information: details of experimental information, supporting SEM images, TEM images and electrochemical measurements. See DOI: https://doi.org/10.1039/d5cc05037e.Notes and references
- M. Yan, H. Huang, K. Zhang, E. Wang, W. Yao, K. Deng, G. Wan, H. Zhang, M. Arita, H. Yang, Z. Sun, H. Yao, Y. Wu, S. Fan, W. Duan and S. Zhou, Nat. Commun., 2017, 8, 257 CrossRef PubMed.
- H. Ma, P. Chen, B. Li, J. Li, R. Ai, Z. Zhang, G. Sun, K. Yao, Z. Lin, B. Zhao, R. Wu, X. Tang, X. Duan and X. Duan, Nano Lett., 2018, 18, 3523–3529 CrossRef PubMed.
- L. Zeng, D. Wu, J. Jie, X. Ren, X. Hu, S. P. Lau, Y. Chai and Y. H. Tsang, Adv. Mater., 2020, 32, 2004412 CrossRef.
- W. Zhao, C. Cui, Y. Xu, Q. Liu, Y. Zhang, Z. Zhang, S. Lu, Z. Rong, X. Li, Y. Fang and W. Huang, Adv. Mater., 2023, 35, 2301593 CrossRef CAS PubMed.
- J. Chen, M. Qin, S. Ma, R. Fan, X. Zheng, S. Mao, C. Chen and Y. Wang, Appl. Catal., B, 2021, 299, 120640 CrossRef CAS.
- Y. Wang, W. Zhai, Y. Ren, Q. Zhang, Y. Yao, S. Li, Q. Yang, X. Zhou, Z. Li, B. Chi, J. Liang, Z. He, L. Gu and H. Zhang, Adv. Mater., 2023, 2307269 Search PubMed.
- Y. Zhao, J. Qiao, P. Yu, Z. Hu, Z. Lin, S. P. Lau, Z. Liu, W. Ji and Y. Chai, Adv. Mater., 2016, 28, 2399–2407 CrossRef CAS PubMed.
- J. Shi, Q. Ji, Z. Liu and Y. Zhang, Adv. Energy Mater., 2016, 6, 1600459 CrossRef.
- D. Hu, T. Zhao, X. Ping, H. Zheng, L. Xing, X. Liu, J. Zheng, L. Sun, L. Gu, C. Tao, D. Wang and L. Jiao, Angew. Chem., Int. Ed., 2019, 58, 6977–6981 CrossRef.
- J. Li, S. Kolekar, M. Ghorbani-Asl, T. Lehnert, J. Biskupek, U. Kaiser, A. V. Krasheninnikov and M. Batzill, ACS Nano, 2021, 15, 13249–13259 CrossRef.
- K. Deng, M. Yan, C.-P. Yu, J. Li, X. Zhou, K. Zhang, Y. Zhao, K. Miyamoto, T. Okuda, W. Duan, Y. Wu, X. Zhong and S. Zhou, Sci. Bull., 2019, 64, 1044–1048 CrossRef PubMed.
- X. Xu, X. Wang, S. Yu, C. Wang, G. Liu, H. Li, J. Yang, J. Li, T. Sun, X. Hai, L. Li, X. Liu, Y. Zhang, W. Zhang, Q. Zhang, K. Wang, N. Xu, Y. Ma, F. Ming, P. Cui, J. Lu, Z. Zhang and X. Xiao, ACS Nano, 2024, 18, 32635–32649 CrossRef.
- F. Liu, Y. Hu, Z. Qu, X. Ma, Z. Li, R. Zhu, Y. Yan, B. Wen, Q. Ma, M. Liu, S. Zhao, Z. Fan, J. Zeng, M. Liu, Z. Jin and Z. Lin, Proc. Natl. Acad. Sci. U. S. A., 2023, 120 (26), e2303262120 Search PubMed.
- Y. Huan, J. Shi, X. Zou, Y. Gong, Z. Zhang, M. Li, L. Zhao, R. Xu, S. Jiang, X. Zhou, M. Hong, C. Xie, H. Li, X. Lang, Q. Zhang, L. Gu, X. Yan and Y. Zhang, Adv. Mater., 2018, 30, 1705916 CrossRef.
- Y. Peng, L. Zhu, C. Li, J. Hu, Y. Lu, J. Fu, F. Cui, X. Wang, A. Cao, Q. Ji, Y. Huan and Y. Zhang, Adv. Energy Mater., 2024, 14, 2302510 CrossRef.
- K. Chen, Y. Huan, W. Quan, L. Zhu, J. Fu, J. Hu, F. Cui, F. Zhou, X. Wang, M. Li and Y. Zhang, ACS Energy Lett., 2022, 7, 3675–3684 CrossRef.
- B. Zheng, Y. Chen, Z. Wang, F. Qi, Z. Huang, X. Hao, P. Li, W. Zhang and Y. Li, 2D Mater., 2016, 3, 035024 CrossRef.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2025 |