Open Access Article
Open Access ArticleLess is more? Probing the template amount required for solid-state olefin [2 + 2] photocycloaddition in a ball mill
Mario
Pajić
 ,
Senada
Muratović
,
Senada
Muratović
 and
Marina
Juribašić Kulcsár
and
Marina
Juribašić Kulcsár
 *
*
Ruđer Bošković Institute, Bijenička cesta 54, HR-10000 Zagreb, Croatia. E-mail: marina.juribasic@irb.hr
First published on 15th October 2025
Abstract
The first template-assisted olefin [2 + 2] photocycloaddition performed in a ball mill is reported. Using catalytic amounts of template resorcinol (as low as 10 mol%) enables rapid, quantitative and stereoselective dimerisation of trans-1,2-bis(4-pyridyl)ethene. In situ Raman monitoring of the photomechanochemical reactions reveals sigmoidal reaction profiles for olefin consumption, which were analyzed using the Finke–Watzky and JMAK kinetic models.
One of the most extensively studied reactions involving reactive supramolecular assemblies is the template-assisted olefin photodimerization.1,2 This long-known3–6 [2 + 2] cycloaddition typically requires a pre-formed co-crystal of the olefin with a template, where the molecules are pre-aligned in the Schmidt orientation.7 Conventional methodologies rely on irradiating these photoactive co-crystals, often with periodic pauses to manually mix the sample.8–13 This human intervention limits scalability and reproducibility of the reaction.3–5,9,10
Resorcinol (res) derivatives are efficient templates for olefin photodimerisation.3–13 Until now, res-assisted cycloaddition of olefins has been carried out using manual10,11 or vortex12,13 mixing. The lowest reported template loading to achieve near-quantitative yield was 10 mol%, but it required 80 hours of irradiation with intermittent mixing.10 More recently, 50 mol% of a res derivative enabled dimerisation in just 30 minutes in the vortex.13 Despite these advances, real-time monitoring of the solid-state reaction remains challenging.
Ball milling14 has emerged as a sustainable alternative to conventional solution-based protocols15,16 and has recently extended into the area of photochemistry.17 Since the initial breakthroughs in 2017,18,19 solid-state photochemical transformations under ball-milling conditions have become an exciting field of research.20–24 Ball milling offers advantages such as reproducibility, solvent-free conditions, and the ability to monitor reactions in real time without interrupting milling by solid-state Raman spectroscopy.14–16,25
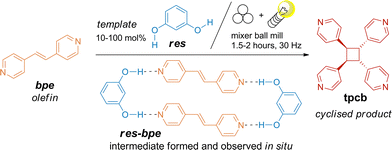
In this study, we report the first example of a template-assisted olefin [2 + 2] photocycloaddition performed in a ball mill. Using as little as 10 mol% of res as a supramolecular catalyst, trans-1,2-bis(4-pyridyl)ethene (bpe) was quantitatively and stereoselectively converted to rctt-1,2,3,4-tetrakis(4-pyridyl)cyclobutane (tpcb) within 2 hours, without any need for manual workup (Scheme 1). The reported solid-state approach is fast and simpler than previously reported methods.10–13 The reactions were monitored in situ using Raman spectroscopy. Conversion profiles for bpe in reactions with 10–50 mol% of res showed sigmoidal kinetics dependent on the template amount, with 25 mol% res yielding the fastest transformation. Kinetic analysis using the Finke–Watzky (FW) and the Johnson–Mehl–Avrami–Kolmogorov (JMAK) models26 revealed how template loading influences the reaction dynamics.
The olefin bpe and the template res (10–100 mol% relative to bpe) were milled under simultaneous irradiation for 3 hours. Quantitative photodimerisation was confirmed by powder X-ray diffraction (PXRD) and 1H NMR analysis of the crude reaction mixtures. Reactions with catalytic amounts of res (10–50 mol%) proceeded faster than that with the equimolar res/bpe mixture, with 25 mol% res giving the fastest conversion. PXRD showed that the formed cyclobutane product tpcb was either unbound (CSD code: PYRBTA27) or part of a res-tpcb assembly (CSD code: ABELAU8), with the ratio of these two depending on the res loading. At 100 mol% res, all product existed as res-tpcb (Fig. 1a) while at lower loadings, all res was bound in res-tpcb, and the excess tpcb remained unbound. Isolation of pure tpcb after the catalytic reactions asks for a solvent-based work-up28 or sublimation.29 Similarly, milling res and bpe in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 molar ratio, yielded only a 1
2 molar ratio, yielded only a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 co-crystal of res and bpe (res-bpe, CSD code: ABEKUN8) and the unreacted bpe according to PXRD of the crude reaction mixture. When res-tpcb was milled with bpe in a 1
1 co-crystal of res and bpe (res-bpe, CSD code: ABEKUN8) and the unreacted bpe according to PXRD of the crude reaction mixture. When res-tpcb was milled with bpe in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 molar ratio, only res-bpe and tpcb were detected by PXRD, showing the higher stability of res-bpe over res-tpcb.
2 molar ratio, only res-bpe and tpcb were detected by PXRD, showing the higher stability of res-bpe over res-tpcb.
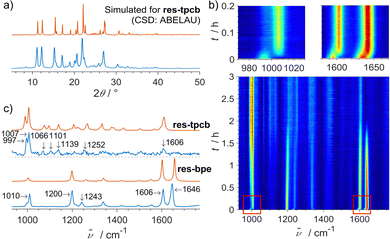 | ||
Fig. 1 Photo-mechanochemical reaction of bpe and res in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio: (a) experimental (blue) and simulated (red) PXRD patterns of the obtained product res-tpcb, (b) 2D plot of the in situ Raman monitoring with insets showing fast formation of res-bpe, and (c) experimental (blue) and calculated (red) Raman spectra of res-bpe and res-tpcb. Raman experiments were performed following the previously described method25 using a portable Raman system with the PD-LD (now Necsel) BlueBox laser source (785 nm excitation wavelength), equipped with the B&W-Tek fibre optic Raman BAC102 probe and coupled to the OceanOptics Maya2000Pro spectrometer. Raman spectra were collected automatically for 8 seconds every 16 seconds, with a laser power of 400 mW and a probe distance of approx. 0.5 cm from the jar with a focus placed 1 mm inside the jar. For the reaction duration of 3 hours, 675 Raman spectra were collected. 1 molar ratio: (a) experimental (blue) and simulated (red) PXRD patterns of the obtained product res-tpcb, (b) 2D plot of the in situ Raman monitoring with insets showing fast formation of res-bpe, and (c) experimental (blue) and calculated (red) Raman spectra of res-bpe and res-tpcb. Raman experiments were performed following the previously described method25 using a portable Raman system with the PD-LD (now Necsel) BlueBox laser source (785 nm excitation wavelength), equipped with the B&W-Tek fibre optic Raman BAC102 probe and coupled to the OceanOptics Maya2000Pro spectrometer. Raman spectra were collected automatically for 8 seconds every 16 seconds, with a laser power of 400 mW and a probe distance of approx. 0.5 cm from the jar with a focus placed 1 mm inside the jar. For the reaction duration of 3 hours, 675 Raman spectra were collected. | ||
In situ Raman monitoring during milling revealed two distinct steps (Fig. 1b). First, the co-crystal res-bpe forms within five minutes, evidenced by a blue shift in key Raman bands of bpe at 1599 and 1636 cm−1 to 1606 and 1646 cm−1. This intermediate was previously shown to form readily under mild conditions.9 Similar was observed for the mechanochemical formation of the H-bonded assembly of phenylboronic acid and 4,4′-bipyridine.30 The second, slower step involves the photodimerisation of bpe within the co-crystal, as seen by the gradual loss of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C-related Raman bands at 1646 and 1200 cm−1 and the appearance of new bands at 997, 1066, 1101 and 1139 cm−1 corresponding to the cyclobutane product (Fig. 1c).
C-related Raman bands at 1646 and 1200 cm−1 and the appearance of new bands at 997, 1066, 1101 and 1139 cm−1 corresponding to the cyclobutane product (Fig. 1c).
Calculations were used to check the feasibility of formation of various H-bonded assemblies and assign the Raman spectra. Results support the experimental findings and are in line with the available report.10 Both applied functionals identify the res-bpe assembly as the most stable H-bonded assembly (Fig. 2). H-bonding between tpcb and res to form res-tpcb is also favourable, explaining why res is observed exclusively as res-tpcb after the reaction under catalytic conditions. The res-bpe assembly is significantly more stable than res-tpcb suggesting that, in presence of the unbound bpe, res-bpe can regenerate from res-tpcb, enabling catalytic turnover.
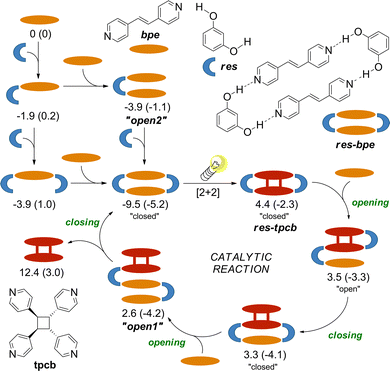 | ||
| Fig. 2 Possible steps leading to the res-catalysed photodimerisation of bpe. Calculations were performed in the gas phase at B3LYP-D3(bj)/def2tzvp and ωB97X-D/def2tzvp (in parentheses) levels of theory using the programme package Gaussian16.31 The reported free energies are in kcal mol−1 relative to reactants at 298.15 K and 1 atm without corrections. More details are listed in the SI. | ||
The catalytic reaction likely involves a series of bpe/tpcb exchanges accompanied by changes in the H-bonding of res. Among the possible pathways, the formation of the most stable supramolecular assembly, the “closed” assembly res-bpe, should be involved in the preferred pathway. As outlined in Fig. 2, the plausible catalytic cycle begins with photoreaction of res-bpe to form res-tpcb. Subsequent binding of bpe to res-tpcb yields the “open” intermediate (res)2(bpe)(tpcb), which undergoes transition first to the more stable “closed” (res)2(bpe)(tpcb) and then to “open2” upon binding another bpe, and finally closes again to regenerate res-bpe, releasing tpcb and completing the cycle.
While the route similar to the simplified pathway shown in Fig. 2 likely prevails, alternative routes may also contribute depending on the reaction conditions and the solid-state environment, particularly at low res loadings. These include direct photocyclisation in “open” assemblies such as “open1” or “open2”, or tpcb-to-bpe exchange in “open1” prior to cyclisation. In these intermediates, bpe is positioned similarly as in “open2” and, as suggested by previous studies,32–35 may still engage in the template-assisted reaction. We note that ex situ monitoring of the reactions by PXRD and NMR methods showed no unidentified species in crude mixtures preventing further analysis.
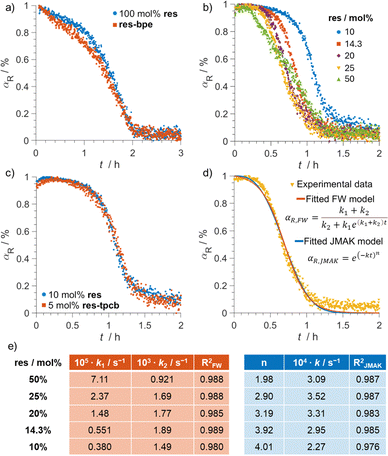
In reactions with equimolar mixtures of res and bpe, or with pre-formed co-crystals res-bpe, the reaction starts quickly due to the (almost) immediate presence of the reactive intermediate. However, some of this intermediate may be trapped inside particles, inaccessible to light.36,37 This might explain the partially sigmoidal reaction profile of bpe determined from Raman changes in the 1620–1660 cm−1 region (Fig. 3a). During the first hour, the reaction appears light-limited, resembling first-order behaviour. In the second hour, the reaction accelerates as mechanical action exposes more reaction sites by fracturing particles and continuously generating new reactive surfaces. This can be tentatively connected to grinding as well as to accumulation of stress, strain and defects introduced by formation of the photoproduct.3,36 These observations align with an earlier report showing that macro-sized res-bpe crystals tend to crack under irradiation, whereas smaller micro- or nanometre-sized crystals undergo single-crystal-to-single-crystal (SCSC) transformations.38
Ex situ monitoring of repeated irradiation and manual mixing with catalytic res-loadings showed an almost linear progression of bpe photodimerisation,10 while the data for the 50 mol% template reaction under vortex conditions was best described by the A2 kinetic model.12In situ Raman monitoring of our reactions with 10–50 mol% res performed in a ball mill revealed characteristic sigmoidal profiles for bpe (Fig. 3b). These curves show an initial induction phase with minimal product formation, which becomes significantly longer as the template loading decreases. This is followed by a rapid conversion phase and then a plateau, indicating reaction completion.
The observed sigmoidal kinetics suggests an autocatalytic component, possibly due to the product itself accelerating the reaction. To test this, a control reaction was conducted using 5 mol% of res-tpcb. The obtained kinetic profile is highly similar to that of the reaction using 10 mol% of res (Fig. 3c). This confirmed that the exchange of bound molecules on the template is fast, and that the induction period is changed more likely due to nucleation and growth processes rather than molecular exchange.
The reaction occurs at supramolecular sites formed by the template. At low res loadings, the limited number of active sites slows the reaction. At high loadings, some res-bpe assemblies may become inaccessible to light, which may also hinder the conversion. An optimal balance between these effects likely reflects in the fastest reaction using 25 mol% of res.
To better understand the solid-state dynamics, the kinetic data for the reactions using catalytic amounts of res was fitted using two common models: the Finke–Watzky (FW) and the Johnson–Mehl–Avrami–Kolmogorov (JMAK) models,26 both previously applied to mechanochemical transformations.39
FW modelling (data in red, Fig. 3e) revealed that nucleation (characterized by k1), which dominates in the initial period, slows with lower res loading, likely due to fewer reaction sites. In contrast, the growth phase (characterized by k2) was faster at lower template concentrations (from 50 to 14.3 mol%), possibly because the limited res is more efficiently used at the particle surface. The reaction may benefit from enhanced local concentration of bpe, rapid product-template exchange, and mechanical effects such as cracking and fragmentation that expose new reactive surfaces.
JMAK analysis complemented the FW findings (data in blue, Fig. 3e). At 50 mol% res, the Avrami exponent n is 2.0, suggesting a 1D process possibly related to growth of the product and/or erosion of product from bpe grain surfaces. At 10 mol% res, the Avrami exponent increases to 4.0, indicative of random nucleation and 3D growth. This shift suggests that under dilute conditions, the reaction proceeds uniformly across the powder, exploiting random nucleation and unhindered growth, with milling aiding in continuous exposure of fresh surfaces.12
The sigmoidal profiles reported here are not due to thermal runaway or “snowballing”40 effects often seen in highly exothermic or paste-forming reactions. The powders remained free-flowing during milling, and the initial temperature increase was attributed to LED illumination and mechanical agitation, not the reaction itself. No temperature rise was observed during the production phase of the reaction.
In conclusion, this study demonstrates that precise control of the template loading is key to optimizing the kinetics of solid-state olefin photodimerization in a ball mill. Using ball milling and just 10 mol% of the resorcinol template enables clean and stereoselective cyclobutane formation without metal catalysts or solvents that is significantly faster than in the only previously reported work with template loading below 50 mol%.10 The described fully automated method, with real-time Raman monitoring, is the first to combine low loadings of the template (catalyst) with rapid, quantitative conversion and kinetic analysis. Together, these advances provide a sustainable and efficient solution to long-standing challenges in the solid-state photochemical dimerization of olefins.
Financial support was provided by Croatian Science Foundation (IP-2019-04-9951, IP-2020-02-1419 and DOK-2020-01-7515). We thank Dr Ivica Cvrtila for helpful discussions. Calculations were performed on the Supek supercomputer at SRCE, Zagreb.
Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.Part of the metadata that supports the findings of this study are available in the supporting information (SI) of this article. Supplementary information: detailed experimental procedures, in situ Raman monitoring plots, kinetic analysis, spectroscopic characterization data for the obtained compounds, and DFT results. See DOI: https://doi.org/10.1039/d5cc04696c.
Notes and references
- F. Toda, K. Tanaka and A. Sekikawa, J. Chem. Soc., Chem. Commun., 1987, 279–280 RSC.
- S. Poplata, A. Tröster, Y.-Q. Zou and T. Bach, Chem. Rev., 2016, 116, 9748–9815 CrossRef PubMed.
- S. P. Yelgaonkar and L. R. MacGillivray in Supramolecular Catalysis, New Directions and Developments, ed. P. W. N. M. van Leeuwen and M. Raynal, WILEY-VCH, Weinheim, 2022, pp. 401–411 Search PubMed.
- L. R. MacGillivray, G. S. Papaefstathiou, T. Friščić, T. D. Hamilton, D.-K. Bučar, Q. Chu, D. B. Varshney and I. G. Georgiev, Acc. Chem. Res., 2008, 41, 280–291 CrossRef.
- M.-M. Gan, J.-G. Yu, Y.-Y. Wang and Y.-F. Han, Cryst. Growth Des., 2018, 18, 553–565 CrossRef.
- V. Ramamurthy and J. Sivaguru, Chem. Rev., 2016, 116, 9914–9993 CrossRef.
- G. M. J. Schmidt, Pure Appl. Chem., 1971, 27, 647–678 CrossRef.
- L. R. MacGillivray, J. L. Reid and J. A. Ripmeester, J. Am. Chem. Soc., 2000, 122, 7817–7818 CrossRef.
- M. B. J. Atkinson, D.-K. Bučar, A. N. Sokolov, T. Friščić, C. N. Robinson, M. Y. Bilal, N. G. Sinada, A. Chevannes and L. R. MacGillivray, Chem. Commun., 2008, 5713–5715 RSC.
- A. N. Sokolov, D.-K. Bučar, J. Baltrusaitis, S. X. Gu and L. R. MacGillivray, Angew. Chem., Int. Ed., 2010, 49, 4273–4277 CrossRef PubMed.
- J. Holdaway, E. Bosch, D. K. Unruh and R. H. Groeneman, Cryst. Growth Des., 2024, 24, 6101–6104 CrossRef.
- J. Stojaković, B. S. Farris and L. R. MacGillivray, Chem. Commun., 2012, 48, 7958–7960 RSC.
- A. A. Colmanet, D. K. Unruh and R. H. Groeneman, RSC Mechanochem., 2025, 2, 631–635 RSC.
- J. F. Reynes, F. Leon and F. García, ACS Org. Inorg. Au, 2024, 4, 432–470 CrossRef PubMed.
- K. J. Ardila-Fierro and J. G. Hernández, ChemSusChem, 2021, 14, 2145–2162 CrossRef.
- J. Alić, M. Schlegel, F. Emmerling and T. Stolar, Angew. Chem., Int. Ed., 2024, 63, e202414745 CrossRef.
- F. Mele, A. M. Constantin, A. Porcheddu, R. Maggi, G. Maestri, N. Della Ca and L. Capaldo, Beilstein J. Org. Chem., 2025, 21, 458–472 CrossRef.
- J. G. Hernández, Beilstein J. Org. Chem., 2017, 13, 1463–1469 CrossRef.
- V. Štrukil and I. Sajko, Chem. Commun., 2017, 53, 9101–9104 RSC.
- D. M. Baier, C. Spula, S. Fanenstich, S. Grätz and L. Borchardt, Angew. Chem., Int. Ed., 2023, 62, e202218719 CrossRef PubMed.
- S. Biswas, S. Banerjee, M. A. Shlain, A. A. Bardin, R. V. Ulijn, B. L. Nannenga, A. M. Rappe and A. B. Braunschweig, Faraday Discuss., 2023, 241, 266–277 RSC.
- F. Millward and E. Zysman-Colman, Angew. Chem., Int. Ed., 2024, 63, e202316169 CrossRef.
- X. Xin, J. Geng, D. Zhang, H. T. Ang, H. Wang, Y. Cheng, Y. Liu, R. W. Toh, J. Wu and H. Wang, Nat. Synth., 2024, 4, 177–187 CrossRef.
- I. Cvrtila, V. Štrukil, M. Alešković, I. Kulcsár, T. Mrla, E. Colacino and I. Halasz, Chem. Methods, 2025, 5, e202400089 CrossRef.
- S. Lukin, K. Užarević and I. Halasz, Nat. Protoc., 2021, 16, 3492–3521 CrossRef PubMed.
- M. A. Watzky and R. G. Finke, J. Am. Chem. Soc., 1997, 119, 10382–10400 CrossRef.
- J. Vansant, S. Toppet, G. Smets, J. P. Declercq, G. Germain and M. Van Meerssche, J. Org. Chem., 1980, 45, 1565–1573 CrossRef.
- T. Friščić, T. D. Hamilton, G. S. Papaefstathiou and L. R. MacGillivray, J. Chem. Educ., 2005, 82, 1679–1681 CrossRef.
- B. Zhu, J.-R. Wang, Q. Zhang and X. Mei, Cryst. Eng. Commun., 2016, 18, 6327–6330 RSC.
- M. Pajić and M. Juribašić Kulcsár, Chem. – Eur. J., 2024, 30, e202400190 CrossRef.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery Jr., J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman and D. J. Fox, Gaussian16, Revision C.01, Gaussian Inc., Wallingford CT, 2016 Search PubMed.
- L. R. MacGillivray, J. L. Reid, J. A. Ripmeester and G. S. Papaefstathiou, Ind. Eng. Chem. Res., 2002, 41, 4494–4497 CrossRef.
- D.-K. Bučar, A. Sen, S. V. S. Mariappan and L. R. MacGillivray, Chem. Commun., 2012, 48, 1790 RSC.
- K. M. Hutchins, J. C. Sumrak and L. R. MacGillivray, Org. Lett., 2014, 16, 1052–1055 CrossRef.
- C. Li, D. C. Swenson and L. R. MacGillivray, Chem. – Eur. J., 2022, 28, e202200978 CrossRef PubMed.
- K. Morimoto, D. Kitagawa, C. J. Bardeen and S. Kobatake, Chem. – Eur. J., 2023, 29, e202203291 CrossRef PubMed.
- W. Li, T. J. Gately, D. Kitagawa, R. O. Al-Kaysi and C. J. Bardeen, J. Am. Chem. Soc., 2024, 146, 32757–32765 CrossRef PubMed.
- D.-K. Bučar and L. R. MacGillivray, J. Am. Chem. Soc., 2007, 129, 32–33 CrossRef.
- R. J. Allenbaugh, T. M. Ariagno and J. Selby, RSC Mechanochem., 2025, 2, 30–36 RSC.
- B. P. Hutchings, D. E. Crawford, L. Gao, P. Hu and S. L. James, Angew. Chem., Int. Ed., 2017, 56, 15252–15256 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2025 |