Advancing metal–organic frameworks and covalent organic frameworks for photocatalytic CO2 reduction
Jeewon
Bu†
 a,
Woo Seok
Cheon†
a,
Woo Seok
Cheon†
 a,
Hyojung
Kim
a,
Hyojung
Kim
 *b,
Myoung Hwan
Oh
*c and
Ho Won
Jang
*b,
Myoung Hwan
Oh
*c and
Ho Won
Jang
 *ad
*ad
aDepartment of Materials Science and Engineering, Research Institute of Advanced Materials (RIAM), Seoul National University, Seoul, 08826, Republic of Korea. E-mail: hwjang@snu.ac.kr; Web: https://sites.google.com/view/onnl Fax: +82-2-884-1413; Tel: +82-2-880-1720
bDepartment of Semiconductor Systems Engineering, Sejong University, Seoul, 05006, Republic of Korea
cDepartment of Energy Engineering, KENTECH Institute for Environmental and Climate Technology, Korea Institute of Energy Technology, Naju, 58330, Republic of Korea
dAdvanced Institute of Convergence Technology, Seoul National University, Suwon, 16229, Republic of Korea
First published on 5th September 2025
Abstract
Increasing atmospheric carbon dioxide concentration necessitates innovative approaches to transform CO2 into valuable compounds utilizing solar energy. The photocatalytic CO2 reduction reaction (PC CRR) offers a sustainable solution, yet wide band gaps, rapid electron–hole recombination, and poor CO2 adsorption capacity limit traditional metal oxide semiconductors for the PC CRR. This review systematically evaluates recent engineering advances in metal–organic frameworks (MOFs) and covalent organic frameworks (COFs) as efficient photocatalysts for the PC CRR. Their intrinsic porosity, structural versatility, and well-defined modular architectures facilitate enhanced light absorption, efficient charge separation, and improved CO2 activation. Their electronic structures are easily regulated by ligand functionalization, metal substitution/introduction, and incorporating photosensitizers. Once they form a favorable heterojunction with other semiconductors, the interfacial electric field facilitates charge separation and transport, increasing the light utilization efficiency. MOFs can also be used as sacrificial templates to fabricate porous photocatalytic materials with enhanced light harvesting and high concentrations of active sites. After thoroughly discussing strategies for photocatalyst engineering, current challenges such as achieving more valuable hydrocarbons as reaction products are addressed, and future research directions are proposed.
1. Introduction
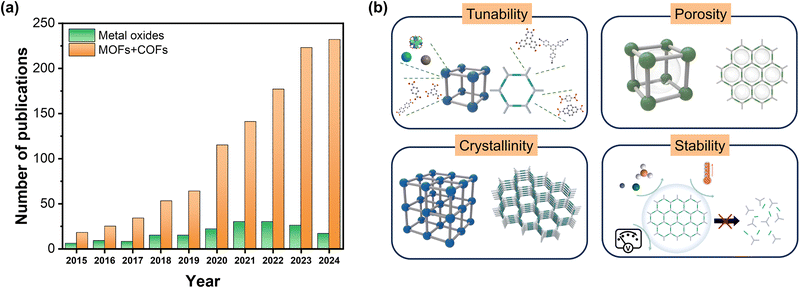
Global warming and climate change are largely caused by the fast-rising concentration of carbon dioxide (CO2) in the atmosphere, which results from increased industrial activity and the ongoing use of fossil fuels.1 Various strategies are being researched to reduce carbon dioxide and achieve carbon neutrality, and among them, converting CO2 into valuable chemical feedstocks and fuels using renewable energies is attracting attention. The photocatalytic CO2 reduction reaction (PC CRR) has been discussed to be a candidate for an eco-friendly and sustainable solution.2,3 The artificial photosynthetic process demonstrates several advantages over other approaches, such as mild reaction conditions, simple and scalable reactors, and direct utilization of limitless and unrestricted solar energy.4 An effective PC CRR requires the development of photocatalytic materials with optimal band gaps for light harvesting, electronic structures for efficient charge separation and transfer, high concentration of active sites for CO2 adsorption and activation.5Initially, many researchers developed metal oxide-based photocatalysts such as TiO2, ZnO, WO3, and Ga2O3 due to their robustness, facile synthesis, and light absorption ability.6–12 However, the large band gaps of metal oxides limit the range of absorbable light to ultraviolet (UV) rays (only ∼5% of solar energy),13 and rich defects within their structure induce rapid charge recombination.14 The lack of design versatility is also a drawback for the PC CRR. Since the CO2 reduction reaction (CRR) is a multi-electron transfer reaction, the reaction pathway is complex and thus requires a catalytic site with well-designed chemical microenvironments to increase the selectivity of a specific product.15,16
Metal–organic frameworks (MOFs) and covalent organic frameworks (COFs) are emerging as innovative materials that are promising alternatives to conventional photocatalysts (Fig. 1a).17 They offer superior properties like high CO2 adsorption capacity, photosensitivity to visible light, high crystallinity and porosity, and structural diversity and tunability (Fig. 1b).5,18 Molecular building blocks and their extensive connections constitute the reticular chemicals, enabling the fabrication of a modularly designed microenvironment.19 MOFs are composed of metal nodes and organic ligands, while COFs are composed of organic building blocks with π-conjugated systems. The structural diversity and tunability allow for modulating the band gap, facilitating charge transfer, and enhancing catalytic activity. For example, changing the linkage between the Pd-complex and the azobenzene in the COF backbone from a simple coordination bond to cyclopalladation can significantly increase the absorption in the near-infrared region.20 Additionally, their high porosity and large surface area maximize the exposure of active sites and mass transport of reactants. Both MOFs and COFs exhibit high crystallinity, which reduces charge recombination and promotes efficient charge separation and transport.21 MOFs can host atomically dispersed active sites that show high selectivity for the CRR over competing side reactions.22 Engineering the coordination of metal nodes and organic linkers modulates the electronic structure to improve light absorption.23–25 COFs possess strong covalent bonding, which provides superior thermal and chemical stability, ensuring the retention of structural integrity over prolonged catalytic cycles.21,26 Moreover, MOFs and COFs can be combined with other materials (such as semiconductors, photosensitizers, single atoms, etc.) or can be precursors of derivatives to improve their electrical conductivity and photocatalytic activity. Integration of the crystalline porous materials with semiconductors endows a favorable heterojunction for photo-generated charge carrier separation and transport. Metal chalcogenide nanoparticles or organic semiconductors such as g-C3N4 have been researched to enhance electron transfer to redox sites and obtain a higher quantum yield.27–30
When the reticular chemicals are utilized as precursors for nanomaterial fabrication, the derivatives maintain some characteristics, such as the porosity and morphology of the parent materials.31 Post-synthetic treatment, including oxidation of MOFs, results in porous structures with a wider absorption range and enhanced chemical stability.32–34
As the interest in the synthesis and photocatalytic application of materials based on reticular chemistry continues to grow, a comprehensive review of such materials is required. It is time for a review that summarizes the recent developments and achievements of MOFs and COFs, focusing on CO2 reduction, which can contribute the most to carbon neutrality among the many reactions that can occur under sunlight. This review aims to provide an in-depth analysis of the photocatalytic properties of MOF and COF-based photocatalysts, systematically summarizing strategies for enhancing their performance. First, principles of the PC CRR and design strategies will be introduced in several aspects such as light harvesting, charge separation and transport and catalytic activity. Afterward, pristine and functionalized MOFs and COFs, their composites and the derivatives will be discussed with selected examples. This work seeks to offer insights into the future development of MOF and COF-based materials and to outline key directions for the design of next-generation photocatalysts for CO2 reduction.
2. Principles and design strategies of photocatalysts for CO2 reduction
The PC CRR is a process that utilizes solar energy to convert CO2 into valuable chemical products, following a principle similar to artificial photosynthesis. In MOF- and COF-based photocatalytic systems, the PC CRR proceeds through three key steps. First, the photocatalyst absorbs solar energy, then electrons excite from the highest occupied molecular orbital (HOMO) to the lowest unoccupied molecular orbital (LUMO).31,35,36 The energy difference between these orbitals plays a crucial role in determining the feasibility of the CRR.5,26,31,36 Second, the photogenerated electrons and holes migrate within the photocatalyst toward the reactive sites. However, during this process, charge recombination can frequently occur, reducing the overall efficiency of the reaction.37 Third, electrons interact with the adsorbed CO2, initiating a reduction process. The type of reduction products depends on the number of electrons transferred, with key products including carbon monoxide (CO), formic acid (HCOOH), formaldehyde (HCHO), methanol (CH3OH), and methane (CH4).35,38Based on the understanding of the principles of the PC CRR, the following properties can be modified in MOF- and COF-based materials to improve their performance: CO2 adsorption and activation, light absorption capacity, charge separation and transport efficiency, and the catalytic activity at redox centers.5,37 These frameworks can function as light-harvesting units, redox-active centers, or host matrices for functionalized units, often fulfilling multiple roles simultaneously. Accordingly, the strategy for performance improvement is closely linked to the specific function for which the material is employed, as illustrated in Fig. 2.
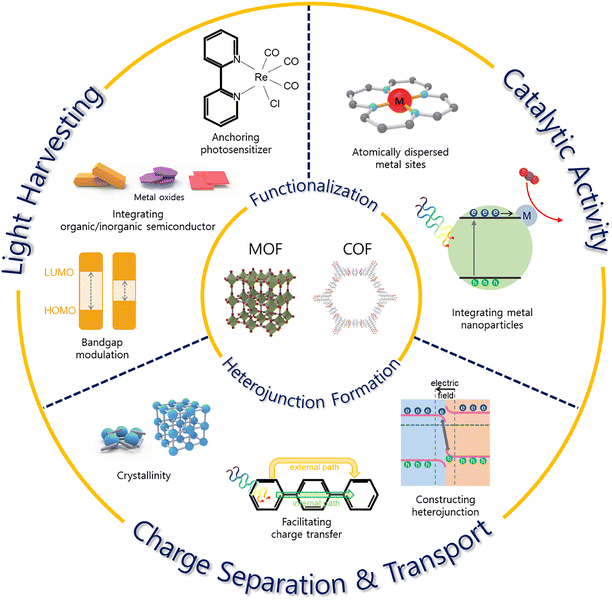 | ||
| Fig. 2 Schematic illustration of strategies for photocatalysts based on MOFs and COFs for CO2 reduction reaction. | ||
The ability to absorb light is one of the most important characteristics that a photocatalyst must possess. Conventional metal oxide semiconductors typically absorb light in the UV spectrum, which limits the efficiency of catalytic reactions using sunlight. Therefore, if the light absorption spectrum of the photocatalyst is extended to the visible light range, solar energy can be utilized more effectively. The width of the band gap is a primary factor that dictates the light absorption range. A narrow band gap allows the absorption of a wide range of light, enhancing the generation and mobility of photoinduced charges; yet, it also elevates the possibility of electron recombination. For effective performance of the CRR, the energy gap between the HOMO and LUMO must be appropriately adjusted.39 The LUMO level must be more negative than the reduction potential of CO2. In contrast, the HOMO level should be more positive than the oxidation potential of the water oxidation reaction or other counter reactions.39,40 MOFs and COFs have structural diversity, allowing various design strategies to tailor their electronic and optical properties. Selecting appropriate metal centers and organic linkers adjusts the electronic properties, and incorporating π-conjugated systems or donor–acceptor motifs enhances light absorption and charge transfer.41 For instance, Fe–O clusters can absorb light and directly excite electrons to enable CO2 reduction.42 Likewise, porphyrin-based linkers exhibit significant light absorbance in the visible spectrum and efficiently generate charge carriers upon light excitation.43–46 Moreover, a high CO2 adsorption capacity can be expected due to the strong interaction between porphyrin-based linkers and CO2. However, porphyrin-based linkers may suffer from photooxidative degradation under aerobic conditions. Modifying ligand and metal nodes can effectively improve light absorption and optimize bandgap alignment.47 Linker functionalization contributes to improving light absorption ability, thereby enabling more efficient utilization of visible light. In particular, the amine group acts as a light-absorbing antenna, increasing the light absorption range into visible light. Also, the amine group functions as an electron donor, enhancing catalytic activity and promoting electron transfer. Fu and coworkers demonstrated this by introducing an amine-functionalized organic ligand (BDC-NH2) into MIL-125(Ti), forming NH2-MIL-125(Ti).48 Before the functionalization, light absorption mainly occurred in the UV spectrum. However, following the incorporation of the amine functional group, absorption within the visible light spectrum became feasible. Light absorption capability can be enhanced by strategically tuning the organic linker and metal cluster, while simultaneously improving CO2 adsorption and charge transfer efficiency. For catalysts exhibiting insufficient light absorption, they can act as photocatalysts by combining with semiconductors (i.e. photosensitizers, metal oxides, etc.). Introducing a photosensitizer into MOFs and COFs can increase light absorption ability and optimize electron transfer. Bandgap engineering strategies will later be exemplified in Section 3.1 for MOFs with functionalized linkers and in Section 4.1 for COFs with extended π-systems.
To achieve effective CO2 reduction, the photoinduced electrons must be efficiently transferred to the catalyst surface, while charge recombination must be minimized during this process. MOFs and COFs have high porosity and crystallinity, which promote charge transfer and suppress charge recombination. In MOFs, discrete metal clusters are periodically connected by organic linkers to construct a three-dimensional network, resulting in short electron hopping distances between adjacent metal nodes.49 This framework architecture facilitates rapid photoinduced electron migration via site-to-site hopping, thereby decreasing electron–hole recombination probability.50 By contrast, COFs possess extended π-conjugated networks within two-dimensional layers, supporting the delocalization of charge carriers throughout the covalently bonded planes.51 Such continuous π-conjugation enables higher in-plane charge mobility than many 3D MOFs and suppresses carrier recombination by minimizing carrier localization. This delocalization reduces carrier localization and can sustain higher photocurrent densities. However, MOFs can offer shorten inter-node electron hopping distances in 3D space, which may benefit isotropic charge transport in bulk powders.
Furthermore, the charge transfer properties of MOFs and COFs are also influenced by their morphology. The regulation of crystallinity and bond polarization can improve carrier mobility.21,52 The crystal size and arrangement of MOFs, as well as the stack arrangement of COFs, control the electron mobility path, which directly impacts the catalytic performance. For example, Song et al. synthesized two MOFs (Ni-MOF and Ni-MOF(H2O)) with different crystal structures using the same linker and metal but with different synthesis methods.53 Ni-MOF(H2O) has a rhombus-shaped plate morphology while the Ni-MOF has a flexible nanosheet morphology. The Ni–C bond length of Ni-MOF(H2O) has been determined to be 1.87 Å, shorter than that of the Ni-MOF of 1.95 Å, enhancing the electronic interaction with CO2 and facilitating effective electron transport. As a result, Ni-MOF(H2O) exhibited a better CO generation rate and selectivity than the Ni-MOF. Additionally, the integration of MOFs and COFs with metal oxides such as TiO2, Cu2O, WO3, etc. to create a type-II or Z-scheme heterojunction results in the spatial separation of electrons and holes, thereby inhibiting recombination and enhancing charge transfer efficiency.54 The presence of an internal electric field within heterostructures further enhances charge carrier separation. A Z-scheme heterojunction consists of two or more semiconductors in which electrons from one component recombine with holes from another. Compared with simple type-II junctions, Z-scheme configurations can better preserve strong redox potentials on both components, albeit often at the cost of increased recombination loss at the mediator interface.55 The choice between these architectures should therefore balance redox driving force and recombination risk in the intended reaction pathway.56 Wang et al. maximized the CO2 photoreduction performance by combining TiO2 and Cu(II) porphyrin-functionalized UiO-66 MOFs to form a MOF-semiconductor heterojunction.57 Photocurrent analysis and electrochemical impedance analysis demonstrated that the heterojunction formation between MOFs and TiO2 was a key factor in promoting electron transfer and enhancing catalytic reactivity. Besides semiconductors, the integration with diverse materials such as quantum dots (QDs), g-C3N4, and single atoms can yield a high-performance catalyst that leverages the benefits of MOFs and COFs while addressing their limitations. In Sections 3.2 and 4.2, MOF/COF-semiconductor heterojunctions demonstrate how band alignment at the interface improves charge separation, or π–π interfacial coupling boosts carrier transfer.
Sufficient active sites are needed for the reactants to be easily accessible, after the electrons have moved to the catalyst surface. The inherently high specific surface area and interconnected pore channels of MOFs and COFs further facilitate the efficient transport of CO2 and H2O (or other electron donors) to the active sites. Using these structural benefits, metal nanoparticles can be introduced into MOFs and COFs to improve catalytic activity. Han and coworkers developed Pt/Au@Pd@MOF-74 by encapsulating Au@Pd within MOF-74, with additional Pt nanoparticle deposition on its surface.58 The CO2 reduction efficiency and product selectivity were markedly enhanced in comparison to the usage of MOF-74 alone. Finally, the introduction of atomically dispersed metal sites (ADMSs) into MOFs and COFs enables strong interactions with CO2 and suppresses undesirable side reactions.59 For instance, Zhong et al. incorporated a single Ni active site into a COF (TpBpy) containing 2,2-bipyridine.60 The COF facilitated CO2 adsorption via its porous architecture, while the Ni active sites established a strong bond with CO2, promoting its conversion into CO. As a result, it generated 4057 μmol g−1 of CO for 5 h with 96% selectivity. Examples in Sections 3.3 and 4.3 show how MOF- and COF-derived structures optimize catalytic sites for stable and efficient CO2 photoreduction.
The efficiency of the CO2 reduction reaction can be maximized by using various strategies to improve catalytic performance by combining the advantages of MOFs and COFs with the properties of other materials. The following section will discuss adjusting appropriate strategies to design MOFs and COFs and their effects on catalytic properties.
3. MOF-based photocatalysts
MOFs have received significant attention as photocatalysts in recent years. The superior photocatalytic performance of MOFs is attributed to their unique physical and chemical properties;25,61–63 (i) MOFs offer numerous combinations of organic ligands and metal nodes. Consequently, energy bandgaps can be modified to improve photocatalytic activity; (ii) high porosity and large specific areas of MOFs provide an abundance of active sites. Moreover, the inherent pore structure promotes CO2 adsorption; (iii) high CO2 adsorption capacity in many MOFs can increase the CO2 concentration around catalytic active sites and thereby enhance reaction efficiency. The selection and modification of organic ligands and metal nodes significantly influence the performance of the photocatalyst. Moreover, MOFs exhibiting limited photoactivity can be integrated with additional components (such as semiconductors, photosensitizers, single atoms, etc.) to create MOF-based composites or transformed into MOF derivatives to enhance the efficiency of photoreactions. Herein, photocatalytic MOFs are categorized into 3 sections and summarized (Table 1).| Photocatalyst | Light source | Efficiency (μmol g−1 h−1) | Selectivity | AQYa (at λ/nm) | TOFb (h−1) | Ref. | |
|---|---|---|---|---|---|---|---|
| a Apparent quantum yield. b Turnover frequency. c The amount of photocatalyst wasn’t specified. | |||||||
| Pristine MOF | BUT-110–50%-Co | 300 W Xe lamp (320–780 nm) | CO: 64 | 89% | N/A | N/A | 46 |
| Ni-MOF(H2O) | 5 W white LED lamp (400–1000 nm) | CO: 9.61 | 95.24% | 1.28% (420) | N/A | 53 | |
| PCN-223 | 300 W Xe lamp (420–800 nm) | HCOO−: 65.2 | N/A | N/A | N/A | 65 | |
| Bilayer nMOF 2 | 300 W Xe lamp | CO: 112.25 | 100% | N/A | N/A | 45 | |
| UiO-66-NH2 (Cu-MOF) | 300 W Xe lamp (λ > 420 nm) | CH3COCH3: 70.9 | 97.1% | N/A | N/A | 66 | |
| Co-OAC | 300 W Xe lamp (λ > 380 nm) | CO: 2325.7 | 99.1% | N/A | 14.4 | 67 | |
| NH2-MIL-125(Ti) (NM111) | 300 Xe lamp (AM 1.5G) | CO: 8.25, CH4: 1.01 | N/A | N/A | N/A | 68 | |
| 0.25MIL-101-F,F | 300 W Xe lamp (λ > 420 nm) | CO: 688 | N/A | N/A | N/A | 69 | |
| Ni-74-Am | 300 W Xe lamp (λ > 400 nm) | CO: 1380 | 94% | N/A | N/A | 70 | |
| MOF composite | CuTCPP ⊂ UiO-66/TiO2 (CTU/0.6TiO2) | Xe lamp (λ > 300 nm) | CO: 31.32 CH4: 0.148 | N/A | N/A | N/A | 71 |
| In-MOF@TP-TA | 300 Xe lamp | CO: 25 CH4: 11.67 | N/A | N/A | N/A | 72 | |
| Co-MOF/Cu2O (xCMC) | 300 W Xe lamp (λ > 420 nm) | CO: 3.38 | 100% | N/A | N/A | 73 | |
| Cu-ZnTCPP/g-C3N4 | 300 W Xe lamp (λ > 360 nm) | CO: 92 CH4: 11.3 | N/A | N/A | N/A | 74 | |
| NM/n-WO3−x | 300 Xe lamp | CO: 12.57 H2O2: 8.41 | N/A | 0.17% (365) | N/A | 75 | |
| Cs3Bi2Br9/Bi-MOF | 300 Xe lamp | CO: 572.24 CH4: 37.5 | 82.6% | N/A | N/A | 76 | |
| MOF-808-PBA-MV | 300 W Xe lamp (λ > 420 nm) | CH4: 460 | 99% | N/A | N/A | 77 | |
| Ru@Cu-HHTP | Xe lamp (λ > 400 nm) | CO: 130 | 92.9% | N/A | N/A | 78 | |
| Ir1/A-aUiO membrane | 300 W Xe lamp (760 > λ > 420 nm) | HCOOH: 3.38 | 99.4% | 15.76% (420) | N/A | 59 | |
| Cu2O@Cu3(BTC)2 | 500 W Xe lamp (λ > 400 nm) | CH4: 0.09125 | N/A | N/A | N/A | 79 | |
| Zr-MBA-Ru/Re-MOF | 300 W Xenon lamp (400–800 nm) | CO: 440 | 99% | 0.11% (450) | N/A | 80 | |
| MOF-808-CuNi | 300 W Xe lamp (760 > λ > 420 nm) | CH4: 158.7 | 97.5% | 2.31% (420) | N/A | 81 | |
| PQ-CDs6.67@Cu-TCA | 300 W Xe lamp (λ > 420 nm) | CH4: 44.43 | 90.22% | 3.35% (700) | N/A | 82 | |
| 50-Bi4O5Br2/Bi-MOF | 300 W Xe lamp | CO: 23.78 CH4: 2.39 | N/A | N/A | N/A | 83 | |
| [DMC@cMOF]-PVK | 300 W Xe lamp (λ > 400 nm) | CO: 133.36 | N/A | N/A | N/A | 84 | |
| MOF derivate | H-Co3O4/In2O3 | 300 W Xe lamp (λ > 420 nm) | CO: 4828 | 80% | 0.59% (450) | N/A | 85 |
| Ni(OH)2-NC-2 | 100 W LED lamp | CO: 144000 | 96.1% | N/A | N/A | 86 | |
| Fe@C | 300 W Xe lamp | CO: 26120 | 99.9% | N/A | N/A | 87 | |
| In2S3–CdIn2S4 | 300 W Xe lamp (λ > 400 nm) | CO: 825 | N/A | N/A | N/A | 88 | |
| ZnIn2S4–In2O3 | 300 W Xe lamp (λ > 400 nm) | CO: 3075 | N/A | N/A | N/A | 89 | |
| ZnO@Co3O4 | 300 W Xe lamp | CH4: 0.99 | N/A | N/A | N/A | 90 | |
| In2O3–C/CdIn2S4 | 300 W Xe lamp (λ > 400 nm) | CO: 2432 | 75.4% | 0.93% (420) | N/A | 91 | |
| NPC-MoS2@Bi4O5Br2 | 250 W high-pressure mercury lamp (λ = 380 nm) | CO: 95.8 CH4: 159.9 | N/A | N/A | N/A | 92 | |
| In2O3@InP60/Cu2O−1 | 300 W Xe lamp | CH4: 38.8 | 91.9% | N/A | N/A | 93 | |
| In2−xS3/Cd1+xIn2−xS4 | 300 W Xe lamp (λ > 400 nm) | CH4: 132.5 CO: 392.1 | N/A | N/A | N/A | 94 | |
| c-In2O3 | 300 W Xe lamp (λ > 320 nm) | CO: 29.19 | 94.47% | N/A | N/A | 95 | |
3.1. Pristine and functionalized MOFs
Research on MOF-based photocatalysts has been active since 2007, when Garcia et al. showed that MOF-5 exhibited the behaviour of a semiconductor, indicating its potential as a photocatalyst.64 MOFs’ remarkable tunability allows them to optimize their catalytic effectiveness while forming various structures based on their composition and synthesis circumstances. The morphology of MOFs significantly influences the photocatalytic performance by affecting light absorption, charge transfer efficiency, and the exposure of active sites. The charge transfer pathways and catalytic properties were optimized by controlling the crystal planes in NH2-MIL-125(Ti).68 The ratio of {001} to {111} planes of NH2-MIL-125(Ti) can be regulated by adjusting the solvent composition ratio and the reaction conditions (Ti source, use of a modulator) during synthesis. The differences in crystal planes significantly influenced the properties of charge transfer and the catalytic active sites of the MOF. The calculated HOMO–LUMO values indicate that the {111} plane of NH2-MIL-125(Ti) exhibits the smallest energy gap, resulting in enhanced photo response (Fig. 3a). In addition, the CO2 adsorption capacity and the exposure concentration of Ti3+ increase, providing a strong reducing power. As a result, the {111} dominant sample showed a CO production rate nine times higher than the {001} dominant sample. Other than the crystal morphology and structure of MOFs, the layered structure can be used. Liang's group enhanced photocatalytic performance by designing a bilayer structure using a 2D MOF.45 The bilayer MOF has a staggered layer structure, and Cl− ions connect the layers and stabilize the structure. The bilayer MOF exhibits low CO2 adsorption energy, allowing smooth charge transfer. The research findings indicate that the performance of CO2 reduction can be significantly enhanced by implementing MOFs’ intrinsic physical and chemical properties through structural optimization. Porphyrin-based MOFs are photosensitive organic compounds commonly used in MOF photocatalysis.96–98 Porphyrins have significant light absorption properties over a wide wavelength range, and when combined with metal nodes, they can provide good electron transfer paths, maximizing catalytic efficiency.96,98 Chen et al. proposed a strategy to enhance the catalytic performance of CO2 reduction by optimizing charge transfer between porphyrin ligands and Zr-O clusters (PCN-233).65 PCN-233 showed light absorption in a broad wavelength range (200–750 nm) due to its porphyrin ligands, and it possessed a narrow band gap of 1.68 eV. By absorbing light and transferring electrons to metal clusters, the ligand-to-metal charge transfer (LMCT) process suppresses electron–hole recombination and the ligands themselves serve as catalytic active sites (Fig. 3b). Therefore, both the ligand and the metal function as catalysts, demonstrating significant catalytic efficiency. The HCOO− production rate of PCN-233 is 65.2 μmol g−1 h−1, showing superior performance compared to the existing Zr/Ti-MOF under identical conditions. LMCT describes an electronic transition in which an electron moves from a ligand-based orbital to a metal-based orbital within a coordination compound, often following light absorption.99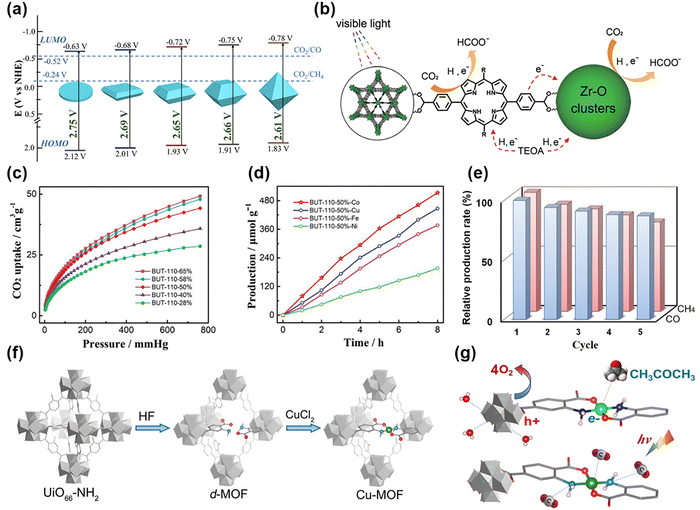 | ||
| Fig. 3 Pristine and functionalized MOFs for photocatalytic CO2 reduction reaction. (a) HOMO–LUMO gap of NH2-MIL-125(Ti) with different exposed facets. Reproduced from ref. 68 with permission from American Chemical Society, copyright 2020. (b) Photocatalytic mechanism proposed for CO2 reduction over PCN-223. Reproduced from ref. 65 with permission from Royal Society of Chemistry, copyright 2022. (c) CO2 adsorption isotherms of BUT-110-X (X is the portion of the porphyrin ligand) samples. (d) BUT-110–50%-M with varied metal species under full-spectrum irradiation. (e) Relative production rate of CO and CH4 obtained from CO2 photoreduction over BUT-110–50%-Co catalyst in five cycles. Reproduced from ref. 46 with permission from Wiley-VCH, copyright 2021. (f) Synthetic procedure for the Cu-MOF. (g) The overall approach to reducing CO2 into CH2COCH3 at CuN2O2 active sites under visible light irradiation. Reproduced from ref. 66 with permission from Wiley-VCH, copyright 2024. | ||
MOFs can be structurally optimized by combining diverse metal nodes and organic ligands, employing various synthetic techniques. However, the performance of MOFs can be further adjusted through post-synthetic modification (PSM) such as ligand substitution, metal substitution, and doping after synthesis.100,101 Kong et al. employed porphyrin ligands to enhance MOFs' chemical stability and catalytic performance.46 To address the low chemical stability of the BUT-109(Zr), the porphyrin ligand was incorporated into a MOF via ligand substitution (NDIDB2− → DCPP2−). As the porphyrin concentration increased, the structurally stronger covalent bond was maintained, and the MOF structure remained stable over pH 1–10. The CO2 uptake capacity improved with higher porphyrin content (Fig. 3c), as the porphyrin ligand provides a strong affinity for CO2 molecules. The researchers performed experiments to enhance catalytic performance by varying the metal center to Ni, Co, Fe, and Cu, along with modifying the ligand of the MOF. Among these, the Co MOF showed the highest catalytic performance and selectivity (Fig. 3d). Additionally, in the recycling test, BUT-110–50%-Co showed no significant variation in CO production during five catalytic cycles (Fig. 3e) and maintained crystallinity and shape even after the catalytic reaction, demonstrating high stability. Xu et al. proposed a strategy to improve the PC CRR by utilizing a fluorinated MOF via ligand functionalization.69 Pristine MIL-101(Fe) has high porosity and excellent CO2 adsorption capacity, but its photocatalytic efficiency is limited due to poor electron transfer. To address this issue, fluorinated ligands (2-fluoroterephthalic acid and 2,5-difluoroterephthalic acid) were introduced to improve the electron transfer properties of MIL-101(Fe). The fluorinated MOF enhances hydrogen bonding with the photosensitizer ([Ru(bpy)3]2+), hence facilitating intermolecular electron transfer and improving the efficiency of the CO2 reduction reaction. The fluorinated MOF showed a CO production rate of 688 μmol g−1 h−1, which is about 3 times higher than that of pristine MIL-101(Fe). This demonstrates that fluorination can effectively enhance the photocatalytic performance of MOF-based catalysts. Researchers also confirmed that the same technique is applicable to additional Fe-based MOFs, such as MIL-53(Fe) and MIL-88(Fe), indicating the potential for improved catalytic performance by ligand engineering of MOFs. The catalytic activity of MOFs differs according to the electronic structure and coordination environment of the metal nodes. Sun et al. substituted Zn in the Zn-based MOF (CFA-1) with Co (Co-OAc, Co-Br, Co-CN), which has different coordination environments.67 The oxidation states and spin states of Co varied among the three MOFs. According to X-ray photoelectron spectroscopy (XPS) and electron paramagnetic resonance (EPR), Co-OAc demonstrated the highest spin state. Co-OAc produced a CO with a high generation rate of 2325.7 μmol g−1 h−1 and a high selectivity of 99.1%, while the high spin state enhanced the charge separation efficiency and CO2 adsorption. DFT calculations indicated that the high spin state inhibited electron–hole recombination and reduced the energy barrier for forming *COOH intermediates. In addition, superior electron mobility and high charge separation efficiency were attributed to low carrier density.
Zhang and co-workers used defect engineering to modify the active sites inside MOFs.66 Cu2+ was incorporated into the defective UiO-66-NH2 (d-MOF), forming NH2-Cu-NH2 ternary catalytic active sites (Fig. 3f). Upon light irradiation, Cu2+ accepts electrons and converts to Cu+, which promotes electron transfer for CO porphyrin ligand reduction. The NH2-Cu-NH2 active sites subsequently adsorb CO2, stabilizing CO and CH3 intermediates and facilitating C–C bond formation (Fig. 3g). Finally, CO2 is selectively converted to acetone (CH3COCH3) via a multi-electron reduction process, demonstrating a superior C3 product formation rate of 70.9 μmol g−1 h−1 compared to UiO-66-NH2 and d-MOF. Defect engineering could also be used to make defects in ligands. Dong et al. induced ligand defects in Ni-MOF-74 to increase the accessibility of metal active sites.70 Ni-74-Am with an increased ligand defect density than Ni-MOF-74 was created by adjusting the synthesis temperature and solvent conditions. This improved the charge transfer properties and exposed more Ni active sites, which raised the catalytic activity. Ni-74-Am demonstrated 3 times higher CO2 reduction performance than Ni-MOF-74, achieving a CO production rate of 1380 μmol g−1 h−1 and a CO selectivity of 94%. In addition, it was confirmed that the electron transfer rate was increased and charge recombination was suppressed in Ni-74-Am through photocurrent analysis. DFT calculation results showed that Ni-74-Am has lower CO2 adsorption energy than the conventional Ni-MOF-74, and the formation of a COOH intermediate is more favorable, so that CO2 reduction proceeds smoothly.
3.2. MOF composites
MOFs have a lot of benefits; however, their charge transportation and light absorption ability might be insufficient. Producing MOF composites is a critical strategy to enhance MOFs' photocatalytic performance. MOF composites offer enhanced light absorption ability, electron mobility, charge separation efficiency, and reaction selectivity when paired with semiconductors, photosensitizers, COFs, QDs, etc.Integrating MOFs with semiconductors has attracted interest as a method to enhance photocatalytic performance, utilizing the significant CO2 adsorption abilities of MOFs with the superior charge transfer properties of semiconductors.5,36 Cu2O is a p-type semiconductor commonly used in photocatalysis. Cu2O provides multiple advantages, including a narrow band gap and an extensive light absorption spectrum.102,103 However, light and water vapor-induced corrosion reduces the catalyst's long-term stability.104 Wu et al. employed a Cu3(BTC)2 MOF as a protective layer to create a core–shell structure protecting Cu2O, hence enhancing the catalyst's durability.79 Whereas Cu2O without a MOF became amorphous due to photo-corrosion after the reaction, the composite preserved its crystalline. Additionally, the MOF coating improved CO2 adsorption by 7 times, and the carboxyl (–COO−) ligand established hydrogen bonds with the intermediate to stabilize it and improved charge separation. Cu2O was also utilized by Dong and colleagues.73 The synergy effect between MOFs and semiconductors can be maximized by forming a p–n heterojunction when Cu2O and Co-MOF (which possess n-type semiconductor properties) are combined. Combining the Co-MOF and Cu2O generates an internal electric field that promotes electron transfer and prevents charge recombination. Upon light exposure, the electrons produced in the conduction band (CB) of Cu2O move to the CB of the Co-MOF and promote the PC CRR (Fig. 4a). In addition, the Co-oxo cluster acts as an electron storage, increasing the catalyst's electron density and CO2 activation. Due to these structural attributes, the CO production rate of the Co-MOF/Cu2O composite was 3.83 μmol g−1 h−1 and it had an almost 100% selectivity. WO3 is a conventional n-type semiconductor that can modulate the Fermi level (Ef) by regulating oxygen vacancy (OV) concentration.105 The introduction of a substantial quantity of oxygen vacancies (OVs) into WO3 (WO3−x) enhances light absorption, accelerates carrier separation, and has strong oxidizing power. However, the exclusive usage of WO3−x has drawbacks, such as rapid charge recombination and low reducing power. Integrating WO3−x with the NH2-MIL-125(Ti) MOF to build an S-scheme heterojunction may resolve the limitations.75 A built-in electric field (BEF) was generated between the two materials, which could be regulated by adjusting the concentration of OVs. The BEF was crucial for accelerating charge transfer and inhibiting charge recombination. As a result of the CRR, the NM/WO3−x composite showed 8 times improved productivity of CO. A S-scheme heterojunction is an interface between two semiconductors engineered such that the system achieves both efficient charge separation and strong overall redox ability. This architecture combines features of type-II and Z-scheme heterojunctions: photogenerated charge carriers with weaker redox potential recombine at the interface, while high-energy electrons and holes accumulate on separate semiconductors, maximizing both charge separation and redox power.56
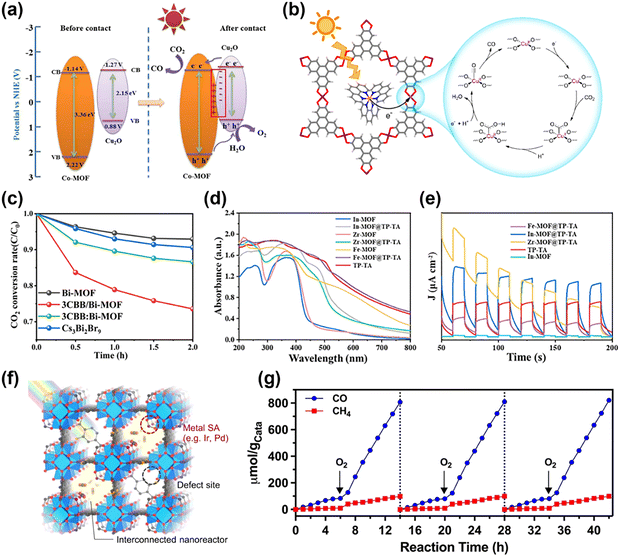 | ||
| Fig. 4 MOF composites for photocatalytic CO2 reduction reaction. (a) Schematic illustration of the electro-transfer process before and after contact with the Co-MOF/Cu2O composite. Reproduced from ref. 73 with permission from Elsevier, copyright 2022. (b) Proposed reaction pathway of the photocatalyst Ru@Cu-HHTP. Reproduced from ref. 78 with permission from American Chemical Society, copyright 2021. (c) CO2 conversion rate of Cs3Bi2Br9, Bi-MOF, 3CBB/Bi-MOF, and 3CBB:Bi-MOF. Reproduced from ref. 76 with permission from American Chemical Society, copyright 2023. (d) UV-vis spectra and (e) transient photocurrent response to compare MOFs, COF, and MOF composites. Reproduced from ref. 72 with permission from Elsevier, copyright 2022. (f) Structure of Ir1/A-aUiO, defect-engineered MOF with single atom. Reproduced from ref. 59 with permission from Springer Nature, copyright 2021. (g) Cyclic experiment of PC CRR on pretreated Cu-ZnTCPP/g-C3N4, the gas atmosphere was switched between anaerobic and aerobic with in each cycle. Reproduced from ref. 74 with permission from Wiley-VCH, copyright 2023. | ||
Immobilizing photosensitizers is another method that has been reported to enhance photocatalytic performance. For MOFs which require photosensitizers separately, the electron transfer efficiency is insufficient and needs a high concentration of photosensitizers. The close interaction of a photosensitizer with MOFs might expand the light absorption spectrum and optimize charge transfer, allowing more efficient utilization of visible light.5,36 Huang et al. used electrostatic attraction to unite a cationic photosensitizer ([Ru(phen)3]2+) to the interior of an anionic MOF (Cu-HHTP) (Fig. 4b).78 UV-vis study results showed increased absorption in the visible-light spectrum compared to the MOF alone. Furthermore, photoluminescence (PL) validated that charge transfer between the photosensitizer and MOF was very efficient. As a result, high catalytic activity was observed even at much lower photosensitizer concentrations. Karmakar's group utilized the pore structure of MOF-808 to optimize the electron transfer path by integrating not only the photosensitizer ([Ru(bpy)3]2+) but also the molecular catalyst ([Re(bpy)(CO)3Cl]) by covalent bonds (Zr-MBA-Ru/Re-MOF).80 MOF-808 has a nanometer (nm)-sized pore structure, allowing the close placement of catalytic active sites and the photosensitizer when both are immobilized within the pores. The composite prevents charge recombination and increases electron transport. In addition, MOF-808 could absorb light only in the UV range (200–320 nm), but the photosensitizer extended the light absorption range to the visible light range of 400–700 nm. As a result, the Zr-MBA-Ru/Re-MOF catalyst maintained high CO production selectivity (99%) and catalytic stability even without a sacrificial electron donor. After that, the same research team introduced 1-pyrenebutyric acid (PBA) and methyl viologen (MV) into the MOF pores to construct a charge transfer complex to optimize the charge transfer pathway and broaden the visible light absorption spectrum.77 Consequently, MOF-808-PBA-MV showed significant light absorption in the 450–700 nm range and featured an electronic structure suitable to the CRR. Due to these modifications, MOF-808-PBA-MV exhibited improved efficiency compared to the original MOF in the selective reduction of CO2 to CH4, achieving 7.3 mmol g−1 of CH4 production over 16 hours while sustaining a selectivity above 99%. Furthermore, photocurrent analysis and in situ diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) studies proved that electron transfer proceeded smoothly and the CO2 reduction intermediate was efficiently stabilized.
Besides semiconductors and photosensitizers, complexation strategies, including diverse materials such as QDs, COFs, single atoms, etc., are under active research. Ma et al. introduced a method to optimize CO2 photoreduction efficiency through the covalent grafting of phenanthraquinone (PQ)-modified carbon quantum dots (CDs) onto a copper-based MOF (Cu-TCA MOF).82 Despite the large porosity and CO2 adsorption capacity of the standard Cu-TCA MOF, its photocatalytic activity was constrained by low conductivity and ineffective electron transport. To improve this, CDs were covalently grafted with Cu active sites to shorten the charge transfer distance, and additionally, dual-active sites and parallel charge transfer pathways were formed through PQ modification. As a result, PQ-CDs6.67@Cu-TCA showed a 2.47 times improvement in the electron consumption rate (393.98 μmol g−1 h−1) compared to the Cu-TCA, and the CH4 selectivity also increased up to 90.22%. Ding et al. proposed an approach for the in situ growth of Cs3Bi2Br9 QDs on the surface of the Bi-based MOF (Bi-MOF) to form a strong interfacial interaction between the MOF and the QDs.76 The bond formed between the MOF and QDs through the shared Bi atoms enhanced the electron transfer pathway and expanded the light absorption spectrum. Electrons were excited by light absorption from the Cs3Bi2Br9, and they moved from the QDs to the MOF. Next, the CRR proceeded at the Bi active sites in the MOF. The Cs3Bi2Br9/Bi-MOF showed enhanced efficiency in reducing CO2 to CO compared to traditional MOFs (Fig. 4c). Yuan et al. integrated a conductive metal–organic framework (cMOF) with perovskite quantum dots (PVK QDs).84 A dinuclear metal catalyst (DMC) was selectively placed within the pores of the MOF, and an ultrathin film structure was developed by immobilizing PVK QDs on the MOF surface to optimize the charge transfer path. The photoinduced electrons produced from PVK QDs were effectively transported to DMC active sites via the cMOF, suppressing charge recombination and resulting in enhanced selectivity and catalytic activity in the CRR. The optimized [DMC@cMOF]-PVK composite recorded a CO production rate of 133.36 μmol g−1 h−1, which was more than 10 times higher than that of the conventional PVK or DMC-PVK catalysts.
Meanwhile, a COF has a unique π-conjugated structure and high charge mobility, and when combined with a MOF, it provides properties that can optimize the electron transfer path of the catalyst.21,26,72 In particular, triazine-based COF (TP-TA) has excellent light absorption properties, and it promotes charge transfer. Wang and coworkers formed a type-II heterojunction by combining a TP-TA COF with three MOFs, respectively.72 The UV-Vis indicated that the MOF–COF composite exhibited an enhanced light absorption range in the visible spectrum of 400–700 nm relative to the traditional MOF (Fig. 4d). Under light irradiation, electrons easily transferred from COF to MOF, and charge separation was promoted, which proved by transient photocurrent response (Fig. 4e). Then, electron–hole recombination was suppressed inside MOF and CRR was conducted at MOF's CB. As a result, the In-MOF@TP-TA composite showed the highest activity in the reaction of reducing CO2 to CO (25.0 μmol g−1 h−1) and CH4 (11.67 μmol g−1 h−1). Combining single atoms with MOFs is also an effective strategy. Individual atoms can act as separate active sites compared to traditional nanoparticle catalysts and have the advantage of sustaining high selectivity by modulating interactions with reaction intermediates. Hao et al. induced metal single atoms (Ir or Pd) in MOFs (Fig. 4f).59 They generated defects in MOFs to allow single atom metals to act as catalytic active sites and fabricated a highly permeable MOF membrane. This structural change significantly improved the light absorption properties of MOFs. Unlike conventional MOFs that mainly react only in the ultraviolet region, the MOF membrane with single atoms could absorb visible light (over 600 nm). In addition, the single atom active sites facilitated electron transfer within the MOF and suppressed unnecessary electron–hole recombination. These modifications resulted in substantial enhancements in CO2 reduction processes. Li et al. incorporated Cu/Ni dual-metal-site pairs (DMSPs) into MOF-808.81 Conventional dual metal catalysts have a fixed active site structure, making it difficult to optimize the interaction with the reaction intermediate. To address this issue, self-adaptive DMSPs were developed by incorporating ethylenediaminetetraacetic acid (EDTA) into the Zr-oxo clusters inside the MOF to generate flexible Cu/Ni active sites. This structural change improved the light absorption performance of the MOF and facilitated the electron transfer at the catalytic active sites. Compared to Cu or Ni single catalysts, the MOF-composite promoted the conversion of CO2 to CH4 more effectively and suppressed the formation of by-products. The creation of a Z-scheme heterojunction by integrating Bi4O5Br2 with a Bi-based MOF (Bi-MOF) also can enhance the CO2 photoreduction efficiency.83 Bi4O5Br2 and Bi-MOF were designed to be self-assembled to simultaneously utilize the high reducing power of Bi4O5Br2 and the excellent CO2 adsorption ability of the Bi-MOF. The optimized 50-Bi4O5Br2/Bi-MOF composite catalyst showed an increased CO production rate (23.78 μmol g−1 h−1) compared to pure Bi4O5Br2 and increased CH4 production rate (5.39 μmol g−1 h−1) compared to Bi-MOF, and also exhibited high catalytic stability and reusability. In addition, the Z-scheme charge transfer mechanism was demonstrated by ESR, UPS, and band structure analysis, and it was confirmed that Bi4O5Br2 acts as an electron reservoir to suppress electron–hole recombination and promote electron transfer.
The CRR via photocatalysis has attracted attention as a fundamental technique for sustainable fuel production; however, in most catalytic systems, the presence of oxygen (O2) is an obstacle to the CRR. Due to the higher reducibility of O2 compared to CO2, the oxygen reduction reaction (ORR) competes with the CRR on the catalyst surface, hence diminishing the overall reduction efficiency.74,106,107 Previous studies have explored strategies to obstruct O2 or to introduce an extra layer that selectively adsorbs CO2 to address this issue. However, these approaches have the disadvantage of requiring separate energy consumption and complex processes.74,106,107 Xie's group proposed a unique approach to activate the catalyst using O2 rather than removing oxygen.74 This photocatalyst, integrating the copper–porphyrin-based MOF (Cu-ZnTCPP) with g-C3N4, can selectively convert CO2 to CO and CH4, despite the presence of O2. The CRR showed a reaction rate five times greater in a 20% O2 environment compared to an O2-free environment (Fig. 4g), and the selectivity for converting CO2 to CO and CH4 was enhanced. The appearance was mostly attributed to the structural modification of the Cu active site. X-ray absorption fine structure (XAFS) and in situ infrared (IR) studies indicated that Cu nodes were hydroxylated when O2 was reduced on the catalyst surface, and these hydroxylated Cu nodes acted as active sites to enhance the CRR activity. Pd(II)-porphyrin-based polymer-coated hollow TiO2 (Pd-HPP-TiO2) also demonstrates a significant advancement in the aerobic PC CRR.108 Unlike typical photocatalysts that suffer from the competing ORR, Pd-HPP-TiO2 maintained high CO2 selectivity and catalytic efficiency even under aerobic conditions. This performance was attributed to the porous Pd(II)-porphyrin polymer (HPP) layer, which selectively adsorbed CO2 while suppressing O2-induced side reactions, thereby enhancing charge separation and electron transfer. As a result, Pd-HPP-TiO2 exhibits a 4.5-fold increase in CH4 production compared to conventional TiO2 under identical aerobic conditions. These studies suggest the possibility of designing a practical photocatalyst that can directly utilize CO2 from air without further purification. Despite this promising performance under aerobic conditions, porphyrinic linkers are known to be susceptible to O2-induced photooxidative degradation and, in some cases, demetallation upon prolonged irradiation.109 Such degradation can compromise long-term catalytic activity and structural integrity. Therefore, future studies should verify the durability of such systems through extended operational testing and post-reaction characterization. Addressing these stability concerns is essential for designing practical photocatalysts capable of directly utilizing CO2 from ambient air without additional purification.
3.3. MOF-derived materials
MOF derived catalysts exhibit excellent performance in a variety of catalytic reactions due to their high surface area, tunable nanostructure, and porous nature. In particular, in the PC CRR, catalysts utilizing MOFs as precursors can maximize performance through effects such as band gap adjusting, increase reaction active sites and charge transfer facilitation.5,31,36 The design strategies of MOF-derived catalysts and recent studies are discussed in this section. Designing MOF-derived catalysts as composite structures is a way to maximize the synergistic effect by combining the advantages of individual components. An In2−xS3/Cd1+xIn2−xS4 heterojunction was developed by Zeng et al. to enhance the performance of wastewater treatment and CO2 reduction simultaneously.94 The OV of In2−xS3 promoted charge transfer, and by combining with Cd1+xIn2−xS4 to construct a Z-scheme photocatalytic system, the CO and CH4 production increased by more than twice that of the conventional In2O3. The crystal structure of In2O3 also could be the key factor to optimize the CO2 photoreduction performance. Wang et al. proposed a strategy to control the crystal structure of In2O3 and optimize the CO2 photoreduction performance through the phase-selective synthesis of MOF-derived catalysts.95 Rhombohedral (rh-In2O3), cubic (c-In2O3), and mixed (rh/c-In2O3) structures can be selectively synthesized by controlling the heat treatment temperature using MOF (MIL-68(In)-NH2) as a precursor. DFT calculation showed that the high charge transfer efficiency and low activation energy barrier affects catalytic activity. Moreover, the presence of surface functional groups (amino groups) of MOFs acts as an important factor inducing the formation of a specific phase during the heat treatment process. As a result of evaluating the CO2 reduction performance according to the phase, c-In2O3 showed the highest CO production rate of 29.19 μmol g−1 h−1 and selectivity of 94.47%.Han and coworkers used a MOF-on-MOF structure grown ZIF-67 on InOF-1 as a precursor and converted it into a hollow heterometal oxide (H-Co3O4/In2O3) through acid treatment and low-temperature oxidation (Fig. 5a).85 The H-Co3O4/In2O3 maximized catalytic performance through the advantages of each component. Co3O4 provided an active site for photocatalytic reactions, while In2O3 enhanced CO2 adsorption and increased electron transport, thereby limiting charge recombination. These researches suggested that MOF-derived multicomponent composites can play an important role in improving catalytic performance. Also, the hollow structure offered more exposed active sites, promoting more efficient catalytic reaction. Compared to traditional single oxide catalysts, the H-Co3O4/In2O3 exhibited enhanced selectivity and greater efficacy in reducing CO2 to CO. The advantage of this hollow structure was also confirmed in the study by Su et al.86 ZIF-8 was used as a precursor to create hollow Ni(OH)2 nanocages (Ni(OH)2-NCs) through ion exchange and corrosion with NiCl2. The nanocage structure promoted light absorption by encouraging multiple internal reflections and increased the catalytic efficiency by exposing more active sites (Fig. 5b). UV-vis analysis results showed that Ni(OH)2-NC had an extended absorption spectrum to the visible light (400–700 nm), which was attributed to the multiple light reflection and scattering effects. Moreover, the presence of a large amount of oxygen vacancies in the catalyst promoted CO2 adsorption and charge transfer, which suppressed electron–hole recombination. Ni(OH)2-NC demonstrated higher catalytic activity than bulk Ni(OH)2, with a high CO production of 1.44 × 105 μmol g−1 h−1 and a selectivity of 96.1%. This activity is among the highest reported for MOF-based systems (Table 1). However, it is important to note that many reported record values are not directly comparable. For instance, higher apparent quantum yield (AQY) and productivity are often achieved under high-intensity monochromatic irradiation or with excess sacrificial agents, which do not reflect practical solar operational environments. It was clearly shown in Su's work that the photocatalytic activity degrades after 3 h as the photosensitizer was deactivated. The choice and concentration of sacrificial reagents can artificially boost product yields, and some sacrificial agents can even serve as unintended carbon sources, leading to overestiation of genuine CO2 reduction activity.110,111
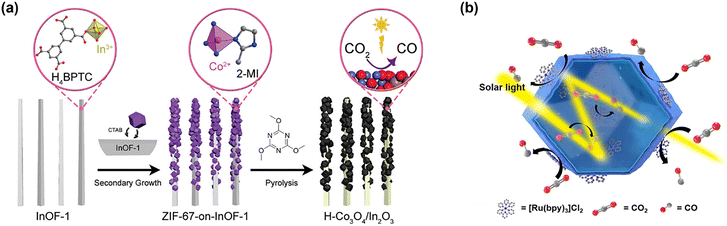 | ||
| Fig. 5 MOF derivatives for photocatalytic CO2 reduction reaction. (a) Stepwise fabrication of MOF-on-MOF-derived hollow bimetallic photocatalyst H-Co3O4/In2O3 for the PC CRR. Reproduced from ref. 85 with permission from Wiley-VCH, copyright 2023. (b) Structure-facilitated multiple light reflection and static charge-transfer processes of MOF-derived photocatalyst Ni(OH)2-NC-2. Reproduced from ref. 86 with permission from American Chemical Society, copyright 2020. | ||
Metal oxides derived from MOFs have the benefit of controllable nanostructures while maintaining high crystallinity. Zhao et al. synthesized an In2O3-C/CdIn2S4 (IOC/CIS) heterojunction by growing CdIn2S4 on In2O3-C, derived from a MOF (In-MIL-68) to optimized the CO2 reduction activity.91 In2O3 has high chemical stability, however it has a wide band gap (2.8 eV) that limits visible light absorption. To overcome this issue, CdIn2S4 was bonded to form a type-II heterojunction structure. As a result, charge recombination was suppressed, and the CRR was accelerated. In particular, carbon doping enhanced charge transfer and increased the CO2 adsorption amount, recording a CO production amount of 2432 μmol g−1 h−1. Sulfide-based catalysts have relatively narrow band gaps and excellent conductivity, and it is easy to form a heterojunction structure for improved charge separation. Sun and colleagues fabricated a p–n heterojunction structure (NPC-MoS2@Bi4O5Br2) by combining Bi4O5Br2 and MoS2 nanorods derived from the Mo-MOF to improve the CO2 reduction performance.92 NPC-MoS2@Bi4O5Br2 had a hierarchically bonded structure of 1D MoS2 nanorods and 2D Bi4O5Br2 nanoflakes, and MoS2 acted as an electron transfer mediator to promote charge transfer. At the optimal composition containing 5% MoS2, CO production of 95.8 μmol g−1 h−1 and CH4 production of 159.9 μmol g−1 h−1 were recorded without additional photosensitizers or cocatalysts, showing a significant improvement in performance compared to the conventional Bi4O5Br2. Phosphide-based catalysts have attracted attention in the CRR due to their high conductivity and excellent charge transfer ability. Xu's team developed In2O3@InP/Cu2O dual Z-scheme photocatalysts to minimize electron–hole recombination.93 While In2O3 has strong oxidizing ability, InP has a narrow band gap (1.35 eV) that increases the visible-light utilization. By depositing p-type Cu2O onto the catalyst, the Z-scheme charge transfer mechanism was induced, which enhanced the conversion of CO2 to CH4 up to 91.9%.
4. COF-based photocatalysts
COFs were discovered approximately a decade after MOFs and have only recently been explored for photocatalytic applications.18 Like MOFs, COFs offer advantages such as tunability, high porosity, large surface area, and high crystallinity. Moreover, when used as photocatalysts, COFs provide additional benefits compared to MOFs.17 Specifically, the strong covalent bonds between monomers endow COFs with superior chemical and thermal stability relative to MOFs. Furthermore, the π-conjugated structure of COFs enhances charge carrier mobility in-plane and stacking directions, thereby improving electrical conductivity. Considering these numerous advantages, this chapter examines the research developments undertaken to advance COFs for CO2 photoreduction. The production rate and selectivity of COF-based photocatalysts are summarized in Table 2.| Photocatalyst | Light source | Efficiency (μmol g−1 h−1) | Selectivity | AQYa (at λ/nm) | TOFb (h−1) | Ref. | |
|---|---|---|---|---|---|---|---|
| a Apparent quantum yield. b Turnover frequency. c The amount of catalyst was not specified. | |||||||
| Pristine COF | Re-f-COF | Xe lamp (λ > 420 nm) | CO: 787.5 | N/A | N/A | N/A | 52 |
| Cu-Bpy-COF | 60 W white LED lamp | CH4: 17.5 in H2O CO: 10.2 in DMF | CH4: ∼100% CO: ∼100% | N/A | N/A | 112 | |
| Co-2,3-DHTA-COF | 300 W Xe lamp (λ > 420 nm) | CO: 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
95.7% | 0.47% (450) | 111.8 | 15 | |
| LaNi-Phen/COF-5 | 300 W xenon lamp (∼100 mW cm−2) | CO: 605.8 | 98.2% | N/A | N/A | 113 | |
| 2N-COF | 300 W xenon lamp (∼100 mW cm−2) | CO: 310 | 100% | 0.36% (420) | N/A | 114 | |
| CoNi−COF-3 | 300 W Xe lamp (λ > 420 nm) | CO: 2567 | 92.2% | 2.95% (450) | N/A | 115 | |
| MCOF-Ti6Cu3 | 300 Xe lamp (AM 1.5 cut-off filter) | HCOOH: 169.8 | N/A | N/A | N/A | 116 | |
| Fe SAS/TrCOF | 300 W Xe lamp (λ > 420 nm) | CO: 980.3 | 96.4% | 3.17% (420) | N/A | 16 | |
| Mo-COF | 300 W Xe lamp (λ > 420 nm) | CO: 6.19 CH4: 1.08 C2H4: 3.57 | CO: 57.10% CH4: 9.96% C2H4: 32.92% | N/A | N/A | 117 | |
| COF-367-CoIII | 300 W Xe lamp (λ > 380 nm) | HCOOH: 93 | 97.1% | N/A | N/A | 118 | |
| CT-COF | 300 W Xe lamp (λ > 420 nm) | CO: 102.7 | 98% | 0.104% (420) | N/A | 41 | |
| Re-Bpy-sp2c-COF | 300 W Xe lamp (λ > 420 nm) | CO: 1400 | 86% | 0.5% (420) | N/A | 21 | |
| COF-367-Co NSs | 300 W Xe lamp (λ > 420 nm) | CO: 10162 | 78% | N/A | N/A | 119 | |
| Ni-TpBpy | 300 W Xe lamp (λ ≥ 420 nm) | CO: 811.4 | 96% | 0.3% (420) | N/A | 60 | |
| TTCOF-Zn | 300 W Xe lamp (800 > λ > 420 nm) | CO: 2.06 | 100% | N/A | N/A | 120 | |
| DQTP COF-Co, | 300 W Xe lamp (λ ≥ 420 nm) | CO: 1020 | 59.4% | N/A | 0.55 | 121 | |
| DQTP COF-Zn | 300 W Xe lamp (λ ≥ 420 nm) | HCOOH: 152.5 | 90% | N/A | 0.08 | 121 | |
| NOP-COP | 300 W Xe lamp (λ ≥ 420 nm) | CH4: 22.5 | 90.2% | 0.32% (420) | N/A | 122 | |
| Re-CTF-py | 300 W Xe lamp (1100 > λ > 200 nm) | CO: 353.05 | N/A | N/A | N/A | 123 | |
| N3-COF | 500 W Xe lamp (800 ≥ λ ≥ 420 nm) | CH3OH: 0.5708 | N/A | N/A | N/A | 47 | |
| Re-COF | 225 W Xe lamp (λ > 420 nm) | CO: ∼750 | 98% | N/A | N/A | 124 | |
| Tpy-COF-Co | 300 W Xe lamp (λ ≥ 420 nm) | CO: 426000 | 55.4% | 2.14% (450) | 524 | 125 | |
| Cu4COF-2 | 300 W Xe lamp (λ > 420 nm) | CO: 23.8 | 94.3 | N/A | N/A | 126 | |
| EPCo-COF-AT | 300 W Xe lamp | CO: 17700 | 97.8 | 0.49% (450) | 54.89 | 127 | |
| Co-THD-COF | 300 W Xe lamp (λ ≥ 420 nm) | CO: 9357 | 95.1 | 9.35% (420) | N/A | 128 | |
| H-COF-Ni | 300 W Xe lamp (λ ≥ 420 nm) | CO: 2847 | 96 | 0.9% (420) | N/A | 129 | |
| Ni-TAPT-COF | 300 W Xe lamp (λ ≥ 420 nm) | CO: 25500 | 98.8 | 1.62% (475) | 31.6 | 130 | |
| CTF | 450 W Xe lamp (λ > 420 nm) | HCOOH: 881300 | N/A | N/A | N/A | 131 | |
| COF composite | GO-COF-366-Co | 300 W Xe lamp (λ > 320 nm) | HCOOH: 1975 in acetonitrile, CO: 6525 in acetonitrile/H2O | CO: 96.1% HCOO−: 94.4% | N/A | N/A | 132 |
| CdS/BiVO4@T-COF | 300 W Xe lamp (720 ≥ λ ≥ 420 nm) | CO: 183.8 H2: 269.8 | CO: 40.5 H2: 59.5 | 1.1% (420) | N/A | 133 | |
| 25% PRGO/TP-COF | 300 W Xe lamp (λ > 400 nm) | CO: 9.762 | N/A | N/A | N/A | 134 | |
| [Emim]BF4@Zn-S-COF | 300 W Xe lamp (λ > 420 nm) | CO: 267.95 | 97.6% | 2.8% (420) | N/A | 135 | |
| MoS2@COF-15 | 60 W white LED lamp | C2H6: 56.2 | 83.8% | 0.03% (450) | N/A | 136 | |
| CdS@COF | Xe lamp (λ > 420 nm) | CO: 507.13 | 72% | 0.21% (420) | N/A | 137 | |
| g-C3N4(NH)/COF | 300 W Xe lamp (λ > 400 nm) | CO: 11.25 | 90.40% | N/A | N/A | 28 | |
| Ru/TpPa-1 | 300 W Xe lamp (800 ≥ λ ≥ 420 nm) | HCOOH: 108.8 | N/A | N/A | N/A | 132 | |
| COF-318-TiO2 | 300 W Xe lamp (380 ≥ λ ≥ 800 nm) | CO: 69.67 | N/A | N/A | N/A | 138 | |
4.1. Pristine and functionalized COFs
COFs with intrinsic light-absorbing capabilities can be directly utilized even in their pristine state. Among these, some COFs exhibit a metal-free composition, which results in lower density and cost-effectiveness. Baeg and co-workers were one of the pioneering research teams that explored COFs for CO2 photoreduction.131 The group synthesized a triazine-based, metal-free, two-dimensional COF via condensation polymerization between cyanuric chloride and perylene diimide. By drop-casting COF onto polyimide substrates, they fabricated a large-area flexible film photocatalyst demonstrating 3.4 times increase in HCOOH production compared to the monomers. The polymeric structure with a band gap of 2.05 eV and an ordered π-electron channel enabled effective PC CRR. However, due to the limited catalytic activity, converting CO2 molecules necessitated the regeneration of nicotinamide adenine dinucleotide (NAD+/NADH). In contrast, COFs capable of converting CO2 and H2O into CH3OH without any sacrificial agent have also been reported.47 Using triformylbenzene (TFB) and triazine, two distinct azine-based COFs were synthesized; both exhibited a higher CH3OH production rate compared to the widely studied g-C3N4. Brunauer–Emmett–Teller (BET) and UV–Vis analysis revealed that the triazine-based COF possessed a larger specific surface area and a smaller band gap than its TFB-based counterpart, resulting in an enhanced visible-light sensitivity. Consequently, the triazine-based COF generated a higher photocurrent. This improvement is further attributed to the electron-deficient nature of triazine, which effectively stabilizes photogenerated electrons and enhances the photocatalytic activity. The extensive focus on triazine in this research area is largely due to its CO2-philic properties. Similarly, to amine groups and imidazole groups, triazine had already been widely exploited for CO2 capture. To achieve more effective band structure tuning and increased conductivity, Liu's group investigated heteroatom doping within the triazine-based framework.122 By condensing hexachlorocyclo-triphosphazene (HCCP) with barbituric acid, they synthesized an N, O, P-containing covalent organic polymer (NOP-COP). Compared to the phosphorus-free counterpart (NO-COP), NOP-COP exhibited an extended lifetime of photoinduced charge carriers. Remarkably, CH4 was produced as the sole carbonaceous product with a selectivity exceeding 90%. Moreover, the CB position of NOP-COP was more negative than that of NO-COP, thereby facilitating a more efficient PC CRR. It was also suggested that the high CO2 affinity of the –P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N groups promoted the adhesion and activation of CO2 molecules. Also, intramolecular charge transport (ICT) in COFs can be enhanced by modifying the monomers. Lei et al. designed a donor–acceptor COF by linking an electron-rich moiety with an electron-deficient moiety.41 This design enabled effective exciton dissociation and band gap modulation, resulting in a CO evolution rate of 102.7 μmol g−1 h−1 without any co-catalyst. DFT calculations further identified the nitrogen atoms within the triazine rings as the active sites. In contrast to the above findings, Wang's group proposed that the triazine ring functions as an electron reservoir while the imine bond serves as the catalytic center.114 They argued that the powder-type samples used in previous studies contributed to discontinuous charge and mass transfer. As a solution, they fabricated a homogeneous COF membrane. To elucidate the roles of the structural units, the authors compared photocurrent density, PL intensity, and PL lifetime. These results demonstrated that increasing the number of triazine groups led to enhanced charge separation efficiency and prolonged lifetimes of photogenerated electrons while the recombination rate decreased (Fig. 6a). DFT calculations indicated that the LUMO is localized near the imine moiety, whereas the HOMO is concentrated on the triazine moiety. According to these findings, the imine moiety acts as an electron acceptor, while the triazine moiety functions as an electron donor and, at the same time, promotes the separation of photogenerated charge carriers. A similar research to Lei et al.'s work was conducted recently by Yu's group.41 In this study, the electron donor (triphenylamine) and acceptor (trinuclear Cu units) were connected via π-conjugated vinylene linkage. Vinylene linkage is superior to imine linkage that are commonly used in COFs. The delocalization reduces the charge recombination rates and charge transfer resistances. The directional transfer of photogenerated electrons from triphenyl amine to redox centers was investigated by using in situ XPS. The well-defined pathway from electron-rich unit to π-conjugation and then to electron deficient unit in the crystalline structure was the key to the improved photocatalytic performance.
N groups promoted the adhesion and activation of CO2 molecules. Also, intramolecular charge transport (ICT) in COFs can be enhanced by modifying the monomers. Lei et al. designed a donor–acceptor COF by linking an electron-rich moiety with an electron-deficient moiety.41 This design enabled effective exciton dissociation and band gap modulation, resulting in a CO evolution rate of 102.7 μmol g−1 h−1 without any co-catalyst. DFT calculations further identified the nitrogen atoms within the triazine rings as the active sites. In contrast to the above findings, Wang's group proposed that the triazine ring functions as an electron reservoir while the imine bond serves as the catalytic center.114 They argued that the powder-type samples used in previous studies contributed to discontinuous charge and mass transfer. As a solution, they fabricated a homogeneous COF membrane. To elucidate the roles of the structural units, the authors compared photocurrent density, PL intensity, and PL lifetime. These results demonstrated that increasing the number of triazine groups led to enhanced charge separation efficiency and prolonged lifetimes of photogenerated electrons while the recombination rate decreased (Fig. 6a). DFT calculations indicated that the LUMO is localized near the imine moiety, whereas the HOMO is concentrated on the triazine moiety. According to these findings, the imine moiety acts as an electron acceptor, while the triazine moiety functions as an electron donor and, at the same time, promotes the separation of photogenerated charge carriers. A similar research to Lei et al.'s work was conducted recently by Yu's group.41 In this study, the electron donor (triphenylamine) and acceptor (trinuclear Cu units) were connected via π-conjugated vinylene linkage. Vinylene linkage is superior to imine linkage that are commonly used in COFs. The delocalization reduces the charge recombination rates and charge transfer resistances. The directional transfer of photogenerated electrons from triphenyl amine to redox centers was investigated by using in situ XPS. The well-defined pathway from electron-rich unit to π-conjugation and then to electron deficient unit in the crystalline structure was the key to the improved photocatalytic performance.
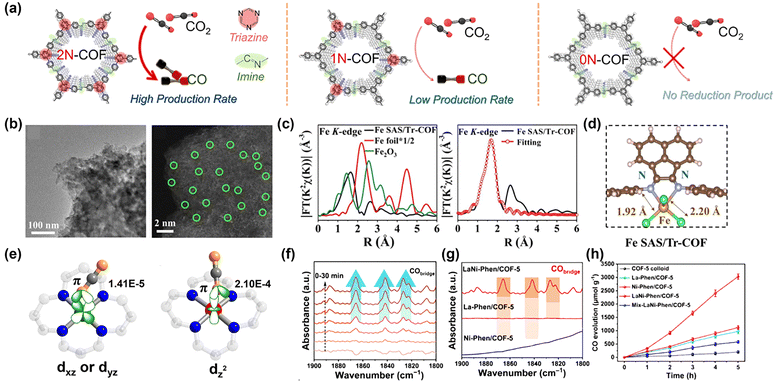 | ||
| Fig. 6 Pristine and functionalized COFs for photocatalytic CO2 reduction reaction. (a) Schematic illustration of 2N-COF, 1N-COF, and 0N-COF membranes. Reproduced from ref. 114 with permission from American Chemical Society, copyright 2023. (b) TEM and aberration-corrected HAADF–STEM image. (c) Fourier-transformed EXAFS spectra and the fitting curves in R space of Fe SAS/Tr-COFs. (d) Schematic model of Fe SAS/Tr-COFs. Reproduced from ref. 16 with permission from American Chemical Society, copyright 2022. (e) CO2-adsorbed at Co sites with different spin states. Reproduced from reference with permission from American Chemical Society, copyright 2020. (f) In situ DRIFTS spectra of adsorbed CO on LaNi-Phen/COF-5. (g) DRIFTS spectra of adsorbed CO at 1800–1900 cm−1 over La-Phen/COF-5, Ni-Phen/COF-5, and LaNi-Phen/COF-5. (h) Time-dependent CO evolution curves under UV-vis light irradiation (λ > 380 nm). Reproduced from ref. 111 with permission from Springer Nature, copyright 2023. | ||
Strategies such as linker functionalization and non-metallic heteroatom doping of COFs have been used to increase light absorption ability and separation efficiency through electronic structure modulation. However, they have not been very effective in increasing their catalytic activity. The nitrogen-rich structure of COFs allows for anchoring the catalytically active site. The Re carbonyl complex has been used as a CO2 reduction photocatalyst since 1983.139 The fac-Re(bpy)(CO)3L (bpy = 2,2′-bipyridine, L = monodentate ligand) undergoes reductive quenching of triplet metal-to-ligand charge transfer (3MLCT) excited state by triethanolamine (TEOA). Subsequently, CO2 capture occurs with loss of L and electron donation by another one-electron reduced species.140 Following the above process, Re or Ru complexes can produce CO with high selectivity and quantum yield. However, due to various disadvantages of homogeneous catalysts, such as non-recoverability, anchoring with COF as support has been investigated. Huang and co-workers synthesized an imine-linked Re-COF using 2,2-bipyridyl-5,5-dialdehyde (BPDA) and 4,4′,4′′-(1,3,5-triazine-2,4,6-triyl) trianiline (TTA), followed by the reaction between the bipyridine ligand and Re(CO)5Cl.124 The Fourier transform infrared (FT-IR) spectroscopy spectrum and X-ray absorption spectroscopy (XAS) analysis confirmed the presence of Re(bpy)(CO)3Cl, and no porosity or crystal structure changed. Due to the partial distribution of the excited ICT state in Re(bpy)(CO)3Cl, photogenerated electrons transfer to the Re complex and the charge recombination is suppressed. The electrons with extended lifetime reduce CO2 to CO2 radical anion at the Re center. The addition of the catalytic active site enhanced the CO selectivity to 98%. However, the coordinated Re complexes suffered a significant mass loss in a solid–liquid system. After reaction in a mixed solution of anhydrous acetonitrile (MeCN) and TEOA, 57.2% of Re was leached out of the photocatalysts, and dissociated from the nitrogen coordination sites.123 Xu et al. employed a solid–gas system instead to prevent Re leaching, and the system showed enhanced cyclic performance.123 When measuring stability in a solid–gas system, the cumulative turnover number (TON) increased with each cycle. Another study utilizing Re(bpy)(CO)3Cl was conducted by Cooper's group.21 Transient absorption spectroscopy showed that the presence of the Re center within the COF increased the lifetime of the charge separated state. A notable result of this study is that crystallinity and porosity have a significant impact on photoreduction performance. The use of 1,4-dioxane resulted in a highly crystalline COF, while in the mixture of 1,2-dichlorobenzene/1-butanol an amorphous analog was made. The amorphous polymer showed much lesser activity than crystalline COF even after being loaded with Re, due to the numerous structural defects that hinder charge transfer. In contrast, the crystalline structure and connected pore channels in the crystalline COF ensure the exposure of active sites for CO2 adsorption and activation. The synthesis of COFs with π-conjugated structures plays a crucial role in their application as photocatalysts. Lu et al. compared the anchoring of other earth-abundant transition metals other than Re to COFs.121 The strong bonding of Co, Ni, or Zn to two layers of appropriately spaced anthraquinone oxygen atoms results in immobilized active sites. The columnar orientation of the COFs makes an ordered π-electron pathway, with electrons efficiently transported to the metal moiety along the pathway, resulting in CO2 reduction. The type of metal greatly affects the activity and selectivity. The Co active site produces mainly CO, while anchored Zn produces mainly HCOOH. According to the two-pathway mechanism proposed by the authors, Co(II), a good π-donor, weakens and breaks the C–O bond of the *COOH intermediate to form CO, while Zn(II), a poor π-donor, forms HCOOH. A total of seven metal elements were anchored to triazine-based COFs by Hou's group.16 Among Fe, Co, Ni, Zn, Cu, Mn, and Ru, Fe had the highest CO generation rate of 980.3 μmol g−1 h−1, 26 times higher than the pristine COF. Atomically dispersed Fe atoms could be identified by high-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) and Fourier-transformed extended X-ray absorption fine structure (EXAFS) (Fig. 6b and c). The bond lengths obtained from the optimized atomic structure model were also in good agreement with the EXAFS fitting results, confirming the existence of Fe–N bonding (Fig. 6d). By utilizing metalloporphyrin, the catalytic active site can be incorporated into the COF structure from the COF synthesis stage. Lan's group has enabled artificial photosynthesis through effective covalent coupling between electron-rich tetrathiafulvalene (TTF) and electron-deficient metalloporphyrin.120 Through effective photoinduced electron transfer (PET), electrons reaching Zn generated CO, while in TTF, photogenerated holes triggered a water oxidation reaction to produce oxygen gas. Around the same time, Jiang's group developed a method for the bulk synthesis of metalloporphyrin and imine-based COF nanosheets.119 By adding an excessive amount of 2,4,6-trimethylbenzaldehyde (TBA) in a solvothermal process hinders the π–π stacking, resulting in 2D COFs instead of bulk material. Compared to the conventional top-down approach of the exfoliation method, they have realized impressive high yield (>55%) and large scale (>100 mg) synthesis. Metalloporphyrins are frequently used due to the facile incorporation of single atomic metal sites with strong bonds and electrical interaction. Lin et al. also utilized Co, Ni, and Cu-containing perflourinated metallophtalocyanine and ellagic acid to demonstrate a photocatalyst with a high CO production rate.127 Moreover, the post-synthetic treatment using an alkaline solution exposed the carboxylic anions and hydroxyl groups on the COF backbone and theoretical calculations suggest that the functional groups reduced the adsorption energy of CO2 molecules. The COF sometimes acts as a host for the reactant and catalytic sites. In Zhong et al.'s work, electrons generated from the photosensitizer are transferred to a COF-supported single Ni site to cause CO2 conversion.60 Condensation of 1,3,5-triformylphloroglucinol and 5,5′-diamino-2,2′-bipyridine generate a 2,2′-bipyridine-based COF. Ni is incorporated by treating the COF in an acetonitrile solution containing Ni(ClO4). Interior cavities of the COF show high CO2 capture ability due to the Lewis acid–base interaction between the CO2 gas molecules and the coordinated Ni single atoms. Thereby the selectivity of CO production remains high at 76% even at low CO2 partial pressure. The reduction of diluted CO2 is important in a way that the concentrating process of CO2 is expensive, and most of the point sources of greenhouse gases contain other gases, too. The effective adsorption and activation of CO2 molecules are thus necessary. By chelating Ni atoms into the pre-designed imine–pyridine positions, the atomically dispersed active sites achieved a high CO selectivity of 98.8% even at 10% diluted CO2 gas.130 The use of 1,3,5-tris(4-aminophenyl) triazine (TAPT) led to a delocalization of the LUMO across the imine-pyridine moiety, making it an efficient electron acceptor. The incorporation of atomically dispersed metals suffered from the problem that the reaction product was limited to CO or syngas. The longer and heavier high-value hydrocarbons are produced, the better the cost-effectiveness of the system. In this regard, Kou and co-workers have done some interesting work.117 They converted CO2 to C2H4 with a high selectivity of about 33% at the MoN2 active site bound to the dipyridyl site. At the Mo site, the CO2 molecule is activated and undergoes a nonlinear conformational change. This lowers the energy barrier required to generate *CO. In addition, the strong adsorption between Mo and CO facilitated the subsequent process to C2H4. This is an impressive result because the more electrons required for conversion, the harder it is for the reaction to occur. The spin state of the metal atom can also change the reaction product selectivity. The cobalt embedded in COF-367 can have a spin ground state of 0 or 1/2 depending on the oxidation number of Co.118 This changes the electron distribution/orientation of the Co 3d orbital. In the case of COF-367-CoII (S = 1/2), 3dxz or 3dyz interacts weakly with the O 2p orbital of CO2, while in COF-367-CoIII (S = 0), 3dz2 strongly overlaps (Fig. 6e). The large difference in adsorption
energy results in different catalytic activities and a much lower energy barrier for the transition states to undergo the HCOOH reaction pathway. Wang's group anchored the Co single atom to the hydroxyl group rather than the bipyridine site on the COF backbone.15 The COF with the Co–O4 site showed a benchmark performance of a CO production rate of 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 μmol g−1 h−1 with the help of Ru-based photosensitizer and sacrificial agent TEOA. The distinct coordination environment of Co(II) accelerated the electron transfer from the photosensitizer to the catalyst. Yue's group was inspired by carbon monoxide dehydrogenases and their coordination environment of metal active centers.128 Thiophene and imine modules were used to anchor Co atoms onto the biomimetic coordination sites. The strong affinity of Co atoms to the CO2 molecules ensured a higher local concentration of CO2 at the catalytic sites. Also, the extended lifetime of the excited state and reduced charge recombination were evidenced by PL analysis. The researchers used actual seawater instead of deionized water to carry out PC CRR experiments. Due to the metal impurities and high concentration of chlorine ions, catalytic reactions in seawater often encounter significant degradation. However, the bioinspired photocatalyst maintained its structural and chemical state after an extended test in the natural seawater. Yang et al. also explored the effect of the microenvironment of metal active centers bonded to COF.129 They found that in the experimented COFs, the affinity with CO2 was much more influential to the photocatalytic activity when comparing the effect of the separation and transfer of charge carriers. The Ni–C bond length in the N-acylhydrazone linkage (3.074 Å) was shorter than the imine-linked counterpart (3.277 Å) according to DFT calculations, indicating the stronger affinity. Also, the stabilization of chelated Ni atoms in the five-membered ring prevented Ni leaching and ensured long-term photostability. Cu single site on COFs can also enhance light absorption, charge separation efficiency, and CO2 adsorption and activation. More importantly, the reaction product, CH4 or CO, depended on the solvent used, between H2O and dimethylformamide (DMF).112 The higher the ratio of H2O to DMF, the lower the CH4 selectivity, while the higher the CO selectivity. Similar results were observed in a cyclic test conducted over approximately 20 hours. The 13CO2 labelling experiment confirmed that both CO and CH4 originated from CO2 gas. It was speculated that the selective products differed due to the difference in proton source and reaction mechanism. In DMF, the subsequent hydrogenation of *CO intermediate had a higher energy barrier (2.81 eV), producing CO without further reaction steps. However, in H2O, the hydrogenation of triethylamine (TEA) required a much lower energy barrier (0.89 eV) for supplying hydrogen to the key reaction intermediate. Thus, *CO further reacted to form *CHO and CH4. One of the most recent work on functionalizing COF is to improve the charge separation by modulating the direction of imine linkage in the COF backbone.52 In a forward direction (f-COF), due to the polarization of the imine bond, the bipyridyl unit acts as an electron acceptor. On the other hand, in the reverse imine case (r-COF), the bipyridyl unit acts as an electron donor. In r-COF, Re(bpy)(CO)3Cl cannot receive electrons to cause a CRR, and charge recombination occurs rapidly. Therefore, the photocatalytic activity is negligible. The orientation of covalent bondings in COFs should be designed according to the same principle so that the separation of photogenerated charge carriers and ICT can occur efficiently. Instead of Cu single atoms, Xu et al. anchored a pre-synthesized tetranuclear Cu cluster on the organic skeleton.126 When compared to the Cu MOFs with coordination driven assembly, the covalent linkage between metallic cluster and monomer displays high-stability characteristics. The long-term photoreduction test was performed for 12 hours without showing any decrease in the CO yield per hour, and the durability was retained for 6 cycles. The topology of the imine-based 2D cluster COFs was regulated, and the qtz topology showed higher CO yield and selectivity over dia topology, which was attributed to the more densely arranged structure. A benchmark CO production rate was achieved by Zhou's group.125 A cobalt-anchored triazine framework reached 426
000 μmol g−1 h−1 with the help of Ru-based photosensitizer and sacrificial agent TEOA. The distinct coordination environment of Co(II) accelerated the electron transfer from the photosensitizer to the catalyst. Yue's group was inspired by carbon monoxide dehydrogenases and their coordination environment of metal active centers.128 Thiophene and imine modules were used to anchor Co atoms onto the biomimetic coordination sites. The strong affinity of Co atoms to the CO2 molecules ensured a higher local concentration of CO2 at the catalytic sites. Also, the extended lifetime of the excited state and reduced charge recombination were evidenced by PL analysis. The researchers used actual seawater instead of deionized water to carry out PC CRR experiments. Due to the metal impurities and high concentration of chlorine ions, catalytic reactions in seawater often encounter significant degradation. However, the bioinspired photocatalyst maintained its structural and chemical state after an extended test in the natural seawater. Yang et al. also explored the effect of the microenvironment of metal active centers bonded to COF.129 They found that in the experimented COFs, the affinity with CO2 was much more influential to the photocatalytic activity when comparing the effect of the separation and transfer of charge carriers. The Ni–C bond length in the N-acylhydrazone linkage (3.074 Å) was shorter than the imine-linked counterpart (3.277 Å) according to DFT calculations, indicating the stronger affinity. Also, the stabilization of chelated Ni atoms in the five-membered ring prevented Ni leaching and ensured long-term photostability. Cu single site on COFs can also enhance light absorption, charge separation efficiency, and CO2 adsorption and activation. More importantly, the reaction product, CH4 or CO, depended on the solvent used, between H2O and dimethylformamide (DMF).112 The higher the ratio of H2O to DMF, the lower the CH4 selectivity, while the higher the CO selectivity. Similar results were observed in a cyclic test conducted over approximately 20 hours. The 13CO2 labelling experiment confirmed that both CO and CH4 originated from CO2 gas. It was speculated that the selective products differed due to the difference in proton source and reaction mechanism. In DMF, the subsequent hydrogenation of *CO intermediate had a higher energy barrier (2.81 eV), producing CO without further reaction steps. However, in H2O, the hydrogenation of triethylamine (TEA) required a much lower energy barrier (0.89 eV) for supplying hydrogen to the key reaction intermediate. Thus, *CO further reacted to form *CHO and CH4. One of the most recent work on functionalizing COF is to improve the charge separation by modulating the direction of imine linkage in the COF backbone.52 In a forward direction (f-COF), due to the polarization of the imine bond, the bipyridyl unit acts as an electron acceptor. On the other hand, in the reverse imine case (r-COF), the bipyridyl unit acts as an electron donor. In r-COF, Re(bpy)(CO)3Cl cannot receive electrons to cause a CRR, and charge recombination occurs rapidly. Therefore, the photocatalytic activity is negligible. The orientation of covalent bondings in COFs should be designed according to the same principle so that the separation of photogenerated charge carriers and ICT can occur efficiently. Instead of Cu single atoms, Xu et al. anchored a pre-synthesized tetranuclear Cu cluster on the organic skeleton.126 When compared to the Cu MOFs with coordination driven assembly, the covalent linkage between metallic cluster and monomer displays high-stability characteristics. The long-term photoreduction test was performed for 12 hours without showing any decrease in the CO yield per hour, and the durability was retained for 6 cycles. The topology of the imine-based 2D cluster COFs was regulated, and the qtz topology showed higher CO yield and selectivity over dia topology, which was attributed to the more densely arranged structure. A benchmark CO production rate was achieved by Zhou's group.125 A cobalt-anchored triazine framework reached 426![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 μmol g−1 h−1 for the first time, surpassing the results of preceding literatures. The high CO yield was attributed to the lowered energy barrier of the formation of key reaction intermediate, *COOH, on the Co-N4Cl2 site. The researchers also showed that the ratio of hydrogen and carbon monoxide in the syngas can be fundamentally regulated by changing the catalyst composition.
000 μmol g−1 h−1 for the first time, surpassing the results of preceding literatures. The high CO yield was attributed to the lowered energy barrier of the formation of key reaction intermediate, *COOH, on the Co-N4Cl2 site. The researchers also showed that the ratio of hydrogen and carbon monoxide in the syngas can be fundamentally regulated by changing the catalyst composition.
As high entropy alloys have received much attention in the field of catalysis, designing active sites using multiple metals can be expected to have a synergistic effect than using only one metal species.141 Lan et al. arranged an oxidative cluster and a reductive cluster to achieve the interaction between two clusters efficiently.116 By letting the CRR occur in the Cu cluster, and the water oxidation reaction occur in the Ti cluster, photoinduced charge separation and charge consumption can occur rapidly. The linkage due to the dynamic covalent bond between the two clusters was very effective for electron transfer. In some cases, two different metal species are both effective in the reductive reaction. Lan's group used Co and Ni to increase the catalytic activity and optimized the light absorption properties by adjusting the carbonyl groups of the building units comprising the COF.115 In the CoNi-COF, the Ni sites enhanced CO2 adsorption and promoted the formation of *COOH, while the Co sites were responsible for electron trapping and electron transfer to CO2. The CO desorption energy was also reduced compared to a single metal, which mitigated the active site screening due to poisoning. The delocalization of the cross-linked framework and the push–pull effect facilitated charge transfer from the photosensitizer. Carbonyl groups enhanced the absorption properties of the COFs in the visible region, and the fully tautomerized COFs with a β-ketoenamine structure facilitated photogenerated charge transfer. In situ analysis monitors surface adsorbates and is necessary to obtain insights into reaction mechanism.142 Zhou and co-workers used in situ DRIFTS analysis to reveal the presence of efficient CO adsorption sites only on the dual-atom catalyst (Fig. 6f).113In situ XAFS analysis revealed that La atoms are optically active centers and act as sites for CO2 activation, with a reduction reaction occurring on Ni (Fig. 6g). Direct observation of the adsorbed/activated CO2 and key reaction intermediates on the LaNi COF surface was also made possible (Fig. 6h).
4.2. COF composites
The modification of COFs is mainly done by adding catalytic active centers or modulating the structural units to change their electronic properties. By creating a composite of COFs and other materials, the charge separation efficiency in the heterojunction can be dramatically increased, and the bandgap difference allows a broader spectrum of light to be utilized. The most common inorganic semiconductor used with COFs is metal oxide. Metal oxide has several disadvantages compared to COFs when used alone, but they can work in synergy when used together. In a study by Zhang et al., 3 different oxides were combined with COF-316/318 to construct a Z-scheme for artificial photosynthesis.138 TiO2, Bi2WO6, and Fe2O3 are all photocatalysts with good performance in water photooxidation and form a Z-scheme system with COF-318 due to the appropriate bandgap position. Therefore, the CO production rate is much higher than pure COF-318 or TiO2. Notably, when the semiconductor was covalently coupled with COF via hydroxyl groups, the electron transfer was facilitated and the performance was superior to the simple physical mixture of these two materials. Many different COFs utilize imine linkages, but the strong polarization of the imine leads to poor delocalization of the π-electron. In a recent study by Wang's group, a semiconductor channel was demonstrated to be a possible solution to this challenge.137 When CdS and COF were formed into a core–shell structure, photogenerated electrons from Zn–porphyrin were transported through the CdS to the CO2 reduction site. The ICT via this new and efficient route resulted in a much more active PC CRR. The authors conducted control experiments in which the Zn–porphyrin, CdS, and Co-bipyridyl units were each removed to confirm the role of individual units. By comparing the bandgap values through UV-vis diffuse reflectance spectra, they found that the bandgap of CdS and COF changed after the heterostructure was created, indicating the existence of interfacial electronic interactions between the two materials. When MoS2 forms a heterojunction with an anthraquinone-based COF, the interfacial e-field facilitates charge separation and transfer (Fig. 7a).136 The hybrid composite, which used amino-modified MoS2 to enhance the electronic interaction, produced hydrocarbon and ethane under visible light irradiation. When CO2 adsorption–desorption measurement was performed, COF showed a high adsorption capacity of 62.7 cm3 g−1, while MoS2 showed negligible adsorption capacity due to its low porosity. MoS2@COF showed similar adsorption behavior to COFs but 31.2 times more ethane production performance. In situ DRIFTS analysis of MoS2@COF confirmed the existence of *COCO intermediate, and a high occurrence of C–C coupling was observed. The photocatalytic syngas production can be maximized when BiVO4 (BVO) is inserted in addition to CdS and COF.133 According to Fan et al., radiative charge recombination is suppressed in the composite and charge transfer resistance is also reduced.133 Based on the band alignment of the components, the CdS/COF composite has a single S-scheme system, and the CdS/COF/BVO composite has a dual S-scheme system. In the single S-scheme, CdS and COF have a weak internal electric field because the Ef difference is not large, so photoinduced electron and hole recombination occurs easily. On the other hand, in the dual S-scheme, severe band bending occurs at the heterojunction at CdS/BVO interface and COF/BVO interface, and strong internal electric fields are generated. Under illumination, the electrons of BVO and holes of CdS and COF at the interface are cancelled out, leaving only charge carriers for photoredox catalysis. CdS, BVO, and COF each play a different catalytic role. CO2 reduction to CO occurs in COF, O2 evolution in BVO, and H2 production in CdS. The photocatalyst was synthesized with the help of a silica template for close interaction of the components, and the distribution of the elements was confirmed by scanning transmission electron microscopy energy-dispersive X-ray spectroscopy (STEM-EDS) mapping (Fig. 7b and c). | ||
| Fig. 7 COF composites for photocatalytic CO2 reduction reaction. (a) Schematic illustrating the charge transfer process and the relative band positions of COF and MoS2. Reproduced from ref. 136 with permission from Elsevier, copyright 2023. (b) Schematic illustration of the synthetic procedure and (c) STEM images and EDS-mapping profiles of CBT. Reproduced from ref. 133 with permission from Elsevier, copyright 2024. (d) Schematic illustration of g-C3N4/COF vdW heterojunction. Reproduced from ref. 28 with permission from Elsevier, copyright 2022. (e) DRUV–vis spectra of TP-COF and (f) 25% PRGO/TP-COF. Reproduced from ref. 134 with permission from Elsevier, copyright 2024. (g) Photocurrent tests and (h) EIS plots of GO-COF-366-Co, COF-366-Co, and GO/COF-366-C. (i) HCOO− yields and (j) CO yields after 8 h of reaction in CH3CN. Reproduced from ref. 143 with permission from Wiley-VCH, copyright 2023. | ||
In addition to inorganic semiconductors, organic semiconductors such as graphitic carbon nitride are also useful for forming a favorable interface with COFs. Wang et al. utilized defective g-C3N4 to form van der Waals heterojunction with a triazine-based COF (Fig. 7d).28 Nitrogen vacancy of the g-C3N4 increases the Ef gap between the two materials, which promotes the recombination of invalid photogenerated charge carriers. Meaningful electrons cause CO2 conversion in g-C3N4. In the temperature-programmed desorption of CO2 (CO2-TPD) analysis, g-C3N4/COF exhibited a stronger CO2 adsorption capacity than g-C3N4. Furthermore, while pristine g-C3N4 showed a sharp decrease in CO2 conversion rate to below 30% during 7 h of irradiation, g-C3N4/COF showed a relatively more stable performance because of accelerated separation and transfer of photoinduced charge carriers. Similarly, photo-reduced graphene oxide (PRGO) and triazine-based COFs have a strong interaction to form a composite photocatalyst.134 According to a study by Liu et al. PRGO and COF are combined through electrostatic self-assembly and have similar carbon skeletons, which leads to π–π stacking.134 After heterojunction formation, electrons are transferred from the CB of PRGO to the valence band (VB) of COF, resulting in a BEF and band bending, which maximizes the separation efficiency of photogenerated charges. The light absorption was greater than the pristine COF (Fig. 7e and f). In the FT-IR spectrum, the peak caused by the vibration of tri-s-triazine is red-shifted, suggesting that the two materials form a composite and have a strong coupling interaction. For more intense cross-linking, covalent anchoring of COF on graphene oxide (GO) has also been studied. According to Gong et al.'s research, the formation of covalent bonds between COF-366-Co and GO shortens the charge transfer distance and reduces charge recombination, thereby increasing the catalytic activity (Fig. 7g–j).143 FT-IR analysis confirmed the bond formed between the amine group of COF-366-Co and the functional group on the GO surface, and the binding energy shift in the XPS Co 2p spectrum demonstrated the strong interaction and charge transfer from COF to GO. They further identified a tendency for the reactant selectivity to change depending on the solvent. In CH3CN, HCOOH was the major product with 94.4% selectivity, whereas in a solution mixed with H2O, CO was generated with 96.1% selectivity.
The co-existence of COFs and metal nanoparticles can also be used to improve visible light harvesting and photocatalytic reduction of CO2. Since Ru, a noble metal, is a good electron trap, it can accept electrons from the LUMO of the COF to improve charge separation.132 However, excessive nanoparticle content may block the COF pores or interfere with light absorption. According to the photocurrent measurement, the best response was obtained when the weight percentage of Ru was 3%. Uniquely, some studies have utilized the abundant pores of COFs to create an interface with ionic liquids. Considering the commercialization and economic viability of CO2 reduction, a high selectivity and conversion rate in dilute CO2 mixed gas rather than pure CO2 is required, and Yang's group realized an enriched CO2 atmosphere through ionic liquid.135 Ionic liquids have been previously used to load porous MOFs to increase their CO2 absorption ability. In this study, CO2 capture and photocatalytic conversion were integrated to create an overall more efficient system. Introducing 1-ethyl-3-methylimidazole tetrafluoroborate ([Emim]BF4) into the Zn-S-COF allows the captured CO2 to be supplied to the Zn active site. Calculations suggested that Emim+ plays a role in supplying proton-electron pairs, which further promotes hydrogenation.
5. Conclusions and outlook
This review summarizes the latest research on MOFs and COFs used in the PC CRR. Compared to conventional photocatalysts such as metal oxides, MOFs and COFs have received much attention due to their advantages, such as high CO2 adsorption capacity and recombination suppression resulting from their intrinsic porosity and high crystallinity. Among these advantages, design flexibility has played a major role in improving the properties and performance of MOFs and COFs. Various strategies such as linker functionalization, immobilizing photosensitizers, and metal node modification have been effective in regulating the electronic structure, including bandgap and increasing light utilization efficiency. Additionally, the formation of heterojunctions with organic or inorganic semiconductors has promoted favorable band alignment and interfacial electric fields to increase the lifetime of photoexcited charge carriers and facilitate their transfer to the redox centers. Embedding atomic metal sites and strengthening interactions between catalytic units have improved CO2 adsorption and activation, stabilizing key intermediates.Although many advances have been made in the light harvesting, charge transfer and catalytic activity of MOF and COF-based photocatalysts, several challenges remain. First, the products generated by the PC CRR are mostly CO or HCOOH. Recent studies have reported the production of C2+ hydrocarbons, such as ethane and ethylene. However, their production rates remain approximately three orders of magnitude lower than the established benchmark for CO production, indicating that further efforts are required to achieve economically and energetically viable multi-carbon products. For example, bimetallic active sites have been studied up to now, but the development of high-entropy active sites could be a breakthrough, where we can expect unprecedented synergistic effects between multiple elements. Secondly, an in-depth understanding of the reaction pathway is required. For hydrocarbon generation, it is necessary to design the microenvironment of the active site based on the understanding of the reaction mechanism rather than developing a photocatalyst by the trial-and-error method. In addition to in situ FT-IR, advanced analysis and prediction techniques such as in situ XAS, in situ TEM, DFT calculations and machine learning should be converged to create active sites capable of C–C coupling and multielectron processes. Also, many MOFs and COFs demonstrate promising performance under simulated light; however, their efficiency under full-spectrum sunlight often remains suboptimal. Bandgap engineering via π-conjugated linkers, donor–acceptor frameworks, and broadband light-harvesting units extends light absorption into the visible and near-infrared regions. Hierarchical pore architectures and photonic crystal-inspired morphologies enhance light trapping and scattering, thereby improving solar-to-chemical conversion efficiency under natural sunlight. Finally, only water should be used as an electron donor for the construction of an environmental-friendly and practical PC CRR system. Although organic sacrificial agents such as TEOA are often used to improve PC CRR performance, it is advisable to avoid using them due to toxicity and byproduct formation issues. Therefore, it is necessary to improve the structural stability in water and develop a photocatalyst with strong water oxidation ability. Structural reinforcement through robust metal–oxygen clusters (e.g., Zr6 and Ti8) and covalent cross-linking of organic linkers can enhance framework stability and resilience. Surface hydrophobic functionalization or core–shell encapsulation can effectively mitigate environmental hydrolysis and photocorrosion, while integrating conductive and chemically stable supports helps maintain efficient electron transport pathways during prolonged operation.
Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work was supported by the Nano & Material Technology Development Program through the National Research Foundation of Korea (NRF) funded by Ministry of Science and ICT(RS-2024-00405016) and the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (MSIT), South Korea (GTL25021-230). The Inter-University Semiconductor Research Center, Institute of Engineering Research, and SOFT Foundry at Seoul National University provided research facilities for this work.Notes and references
- P. N. Pearson and M. R. Palmer, Nature, 2000, 406, 695–699 CrossRef CAS PubMed.
- S. Fang, M. Rahaman, J. Bharti, E. Reisner, M. Robert, G. A. Ozin and Y. H. Hu, Nat. Rev. Methods Primers, 2023, 3, 61 CrossRef CAS.
- Y. Wang, E. Chen and J. Tang, ACS Catal., 2022, 12(12), 7300–7316 CrossRef CAS PubMed.
- Y. Zhang, D. Yao, B. Xia, M. Jaroniec, J. Ran and S. Z. Qiao, ACS Energy Lett., 2022, 7(5), 1611–1617 CrossRef CAS.
- R. Li, W. Zhang and K. Zhou, Adv. Mater., 2018, 30(35), 1705512 CrossRef PubMed.
- Y. X. Pan, Z. Q. Sun, H. P. Cong, Y. L. Men, S. Xin, J. Song and S. H. Yu, Nano Res., 2016, 9, 1689–1700 CrossRef CAS.
- M. Reli, M. Kobielusz, L. Matějová, S. Daniš, W. Macyk, L. Obalová, P. Kuśtrowski, A. Rokicińska and K. Kočí, Appl. Surf. Sci., 2017, 391, 282–287 CrossRef CAS.
- Z. Xiong, Z. Lei, Z. Xu, X. Chen, B. Gong, Y. Zhao, H. Zhao, J. Zhang and C. Zheng, J. CO2 Util., 2017, 18, 53–61 CrossRef CAS.
- Y. Yang, F. Zhan, H. Li, W. Liu and S. Yu, J. Solid State Electrochem., 2017, 21, 2231–2240 CrossRef CAS.
- Y. Ha, J. Kwon, H. An and D. Jung, J. Sens. Sci. Technol., 2022, 31, 343–347 CrossRef.
- K. Ganesh Kumar, P. Balaji Bhargav, N. Ahmed and C. Balaji, Trans. Electr. Electron. Mater., 2021, 22, 717–724 CrossRef.
- N. R. Khalid, S. Ilyas, F. Ali, T. Iqbal, M. Rafique, M. Imran and M. A. Assiri, Electron. Mater. Lett., 2024, 20, 85–94 CrossRef CAS.
- T. Hisatomi, J. Kubota and K. Domen, Chem. Soc. Rev., 2014, 43, 7520 RSC.
- N. Shehzad, M. Tahir, K. Johari, T. Murugesan and M. Hussain, J. CO2 Util., 2018, 26, 98–122 CrossRef CAS.
- Q. Zhang, S. Gao, Y. Guo, H. Wang, J. Wei, X. Su, H. Zhang, Z. Liu and J. Wang, Nat. Commun., 2023, 14, 1147 CrossRef CAS PubMed.
- L. Ran, Z. Li, B. Ran, J. Cao, Y. Zhao, T. Shao, Y. Song, M. K. H. Leung, L. Sun and J. Hou, J. Am. Chem. Soc., 2022, 144, 17097–17109 CrossRef CAS PubMed.
- T. Luo, L. Gilmanova and S. Kaskel, Coord. Chem. Rev., 2023, 490, 215210 CrossRef CAS.
- Z. He, J. Goulas, E. Parker, Y. Sun, X. Dong Zhou and L. Fei, Catal. Today, 2023, 409, 103–118 CrossRef CAS.
- K. Endo, S. Canossa, F. Heck, D. M. Proserpio, M. S. Istek, F. Stemmler, J. van Slageren, S. Hartmann, A. Hartschuh and B. V. Lotsch, Nat. Synth., 2025, 4, 603–613 CrossRef CAS.
- A. Rodríguez-Camargo, M. W. Terban, M. Paetsch, E. A. Rico, D. Graf, R. Hirpara, V. Duppel, I. Moudrakovski, M. Etter, N. Guijarro, C. Ochsenfeld, R. E. Dinnebier, L. Yao and B. V. Lotsch, Nat. Synth., 2025, 4, 710–719 CrossRef.
- Z. Fu, X. Wang, A. M. Gardner, X. Wang, S. Y. Chong, G. Neri, A. J. Cowan, L. Liu, X. Li, A. Vogel, R. Clowes, M. Bilton, L. Chen, R. S. Sprick and A. I. Cooper, Chem. Sci., 2020, 11, 543–550 RSC.
- C. Zhang, Z. Lin, L. Jiao and H.-L. Jiang, Angew. Chem., Int. Ed., 2024, 63, e202414506 CrossRef CAS PubMed.
- Y. Li, H. Xu, S. Ouyang and J. Ye, Phys. Chem. Chem. Phys., 2016, 18, 7563 RSC.
- S. A. Younis, E. E. Kwon, M. Qasim, K. H. Kim, T. Kim, D. Kukkar, X. Dou and I. Ali, Prog. Energy Combust. Sci., 2020, 81, 100870 CrossRef.
- H. Zhang, J. Li, Q. Tan, L. Lu, Z. Wang and G. Wu, Chem. – Eur. J., 2018, 24, 18137–18157 CrossRef CAS PubMed.
- S. Liu, M. Wang, Y. He, Q. Cheng, T. Qian and C. Yan, Coord. Chem. Rev., 2023, 475, 214882 CrossRef CAS.
- S. Wang, J. Lin and X. Wang, Phys. Chem. Chem. Phys., 2014, 16, 14656–14660 RSC.
- J. Wang, Y. Yu, J. Cui, X. Li, Y. Zhang, C. Wang, X. Yu and J. Ye, Appl. Catal., B, 2022, 301, 120814 CrossRef CAS.
- K. K. Khaing, D. Yin, Y. Ouyang, S. Xiao, B. Liu, L. Deng, L. Li, X. Guo, J. Wang, J. Liu and Y. Zhang, Inorg. Chem., 2020, 59, 6942–6952 CrossRef CAS PubMed.
- S. Yan, S. Ouyang, H. Xu, M. Zhao, X. Zhang and J. Ye, J. Mater. Chem. A, 2016, 4, 15126–15133 RSC.
- J. Chen, R. Abazari, K. A. Adegoke, N. W. Maxakato, O. S. Bello, M. Tahir, S. Tasleem, S. Sanati, A. M. Kirillov and Y. Zhou, Coord. Chem. Rev., 2022, 469, 214664 CrossRef CAS.
- L. Hu, H. Yang, S. Wang, J. Gao, H. Hou and W. Yang, J. Mater. Chem. C, 2021, 9, 5343–5348 RSC.
- S. Li, J. Tan, Z. Jiang, J. Wang and Z. Li, Chem. Eng. J., 2020, 384, 123354 CrossRef CAS.
- M. Lan, R. M. Guo, Y. Dou, J. Zhou, A. Zhou and J. R. Li, Nano Energy, 2017, 33, 238–246 CrossRef CAS.
- Q. Su, Q. Guo, H. Wang, M. Liu and C. Zuo, Carbon Resour. Convers., 2024, 7(1), 100211 CAS.
- D. Li, M. Kassymova, X. Cai, S. Q. Zang and H. L. Jiang, Coord. Chem. Rev., 2020, 412, 213262 CrossRef CAS.
- Y. He, L. Yin, N. Yuan and G. Zhang, Chem. Eng. J., 2024, 481, 148754 CrossRef CAS.
- G. H. Han, J. Bang, G. Park, S. Choe, Y. J. Jang, H. W. Jang, S. Y. Kim and S. H. Ahn, Small, 2023, 19, 2205765 CrossRef CAS PubMed.
- L. Zhang, J. Ran, S. Z. Qiao and M. Jaroniec, Chem. Soc. Rev., 2019, 48, 5184 RSC.
- W. Huang, W. Luo and Y. Li, Mater. Today, 2020, 40, 160–172 CrossRef CAS.
- K. Lei, D. Wang, L. Ye, M. Kou, Y. Deng, Z. Ma, L. Wang and Y. Kong, ChemSusChem, 2020, 13, 1725–1729 CrossRef CAS PubMed.
- D. Wang, R. Huang, W. Liu, D. Sun and Z. Li, ACS Catal., 2014, 4, 4254–4260 CrossRef CAS.
- H. Q. Xu, J. Hu, D. Wang, Z. Li, Q. Zhang, Y. Luo, S. H. Yu and H. L. Jiang, J. Am. Chem. Soc., 2015, 137, 13440–13443 CrossRef CAS PubMed.
- S. Ali Akbar Razavi and A. Morsali, Coord. Chem. Rev., 2019, 399, 213023 CrossRef CAS.
- J. Liang, H. Yu, J. Shi, B. Li, L. Wu and M. Wang, Adv. Mater., 2023, 35, 2209814 CrossRef CAS PubMed.
- X. J. Kong, T. He, J. Zhou, C. Zhao, T. C. Li, X. Q. Wu, K. Wang and J. R. Li, Small, 2021, 17, 2005357 CrossRef CAS PubMed.
- Y. Fu, X. Zhu, L. Huang, X. Zhang, F. Zhang and W. Zhu, Appl. Catal., B, 2018, 239, 46–51 CrossRef CAS.
- Y. Fu, D. Sun, Y. Chen, R. Huang, Z. Ding, X. Fu and Z. Li, Angew. Chem., Int. Ed., 2012, 51, 3364–3367 CrossRef CAS PubMed.
- S. Lin, P. M. Usov and A. J. Morris, Chem. Commun., 2018, 54, 6965–6974 RSC.
- T. Zhang and W. Lin, Chem. Soc. Rev., 2014, 43, 5982 RSC.
- K. Merkel, J. Greiner and F. Ortmann, Sci. Rep., 2023, 13, 1685 CrossRef CAS PubMed.
- D. H. Streater, E. R. Kennehan, D. Wang, C. Fiankor, L. Chen, C. Yang, B. Li, D. Liu, F. Ibrahim, I. Hermans, K. L. Kohlstedt, L. Luo, J. Zhang and J. Huang, J. Am. Chem. Soc., 2024, 146, 4489–4499 CrossRef CAS PubMed.
- K. Song, S. Liang, X. Zhong, M. Wang, X. Mo, X. Lei and Z. Lin, Appl. Catal., B, 2022, 309, 121232 CrossRef CAS.
- J. Lin, W. Tian, H. Zhang, X. Duan, H. Sun and S. Wang, Energy Fuels, 2021, 35, 7–24 CrossRef CAS.
- L. Schumacher and R. Marschall, Top. Curr. Chem., 2022, 380, 53 CrossRef CAS PubMed.
- H. Sudrajat, H. Sudrajat, H. Sudrajat and M. Nobatova, RSC Appl. Interfaces, 2025, 2, 599–619 RSC.
- Q. Wang, J. Lian, Y. Bai, J. Hui, J. Zhong, J. Li, N. An, J. Yu and F. Wang, Mater. Sci. Semicond. Process., 2015, 40, 418–423 CrossRef CAS.
- Y. Han, H. Xu, Y. Su, Z. Liang Xu, K. Wang and W. Wang, J. Catal., 2019, 370, 70–78 CrossRef CAS.
- Y. C. Hao, L. W. Chen, J. Li, Y. Guo, X. Su, M. Shu, Q. Zhang, W. Y. Gao, S. Li, Z. L. Yu, L. Gu, X. Feng, A. X. Yin, R. Si, Y. W. Zhang, B. Wang and C. H. Yan, Nat. Commun., 2021, 12, 2682 CrossRef CAS PubMed.
- W. Zhong, R. Sa, L. Li, Y. He, L. Li, J. Bi, Z. Zhuang, Y. Yu and Z. Zou, J. Am. Chem. Soc., 2019, 141, 7615–7621 CrossRef CAS PubMed.
- M. Eddaoudi, H. Li and O. M. Yaghi, J. Am. Chem. Soc., 2000, 122, 1391–1397 CrossRef CAS.
- A. Dhakshinamoorthy, Z. Li and H. Garcia, Chem. Soc. Rev., 2018, 47, 8134–8172 RSC.
- J. Lee, O. K. Farha, J. Roberts, K. A. Scheidt, S. T. Nguyen and J. T. Hupp, Chem. Soc. Rev., 2009, 38, 1450–1459 RSC.
- M. Alvaro, E. Carbonell, B. Ferrer, F. X. Llabrés, I. Xamena and H. Garcia, Chem. – Eur. J., 2007, 13, 5106–5112 CrossRef CAS PubMed.
- D. Chen, Z. Guo, B. Li and H. Xing, New J. Chem., 2022, 46, 16297–16302 RSC.
- M. Zhang, D. Zhang, X. Jing, B. Xu and C. Duan, Angew. Chem., Int. Ed., 2024, 63, e202402755 CrossRef CAS PubMed.
- K. Sun, Y. Huang, Q. Wang, W. Zhao, X. Zheng, J. Jiang and H. L. Jiang, J. Am. Chem. Soc., 2024, 146, 3241–3249 CrossRef CAS PubMed.
- X. M. Cheng, X. Y. Dao, S. Q. Wang, J. Zhao and W. Y. Sun, ACS Catal., 2021, 11, 650–658 CrossRef CAS.
- W. Xu, G. R. Zhang, J. Wang, H. Yu, W. Zhang, L. L. Shen and D. Mei, Adv. Funct. Mater., 2023, 34(9), 2312691 CrossRef.
- Y. L. Dong, Y. Jiang, S. Ni, G. W. Guan, S. T. Zheng, Q. Guan, L. M. Pei and Q. Y. Yang, Small, 2023, 20(23), 2308005 CrossRef PubMed.
- L. Wang, P. Jin, S. Duan, H. She, J. Huang and Q. Wang, Sci. Bull., 2019, 64, 926–933 CrossRef CAS PubMed.
- L. Wang, J. Mao, G. Huang, Y. Zhang, J. Huang, H. She, C. Liu, H. Liu and Q. Wang, Chem. Eng. J., 2022, 446, 137011 CrossRef CAS.
- W. W. Dong, J. Jia, Y. Wang, J. R. An, O. Y. Yang, X. J. Gao, Y. L. Liu, J. Zhao and D. S. Li, Chem. Eng. J., 2022, 438, 135622 CrossRef CAS.
- S. Xie, C. Deng, Q. Huang, C. Zhang, C. Chen, J. Zhao and H. Sheng, Angew. Chem., Int. Ed., 2023, 62, e202216717 CrossRef CAS PubMed.
- H. Jiang, L. Wang, X. Yu, L. Sun, J. Li, J. Yang and Q. Liu, Chem. Eng. J., 2023, 466, 143129 CrossRef CAS.
- D. Lan, D. Yongping, B. Fenghua, C. Gonglai, Z. Shuwei, Y. Xiaoxue, L. Huiqin and W. Xiaojing, Inorg. Chem., 2023, 62, 2289–2303 CrossRef PubMed.
- S. Karmakar, S. Barman, F. A. Rahimi, D. Rambabu, S. Nath and T. K. Maji, Nat. Commun., 2023, 14, 4508 CrossRef CAS PubMed.
- N. Y. Huang, H. He, S. Liu, H. L. Zhu, Y. J. Li, J. Xu, J. R. Huang, X. Wang, P. Q. Liao and X. M. Chen, J. Am. Chem. Soc., 2021, 143, 17424–17430 CrossRef CAS PubMed.
- H. Wu, X. Y. Kong, X. Wen, S. P. Chai, E. C. Lovell, J. Tang and Y. H. Ng, Angew. Chem., Int. Ed., 2021, 60, 8455–8459 CrossRef CAS PubMed.
- S. Karmakar, S. Barman, F. A. Rahimi and T. K. Maji, Energy Environ. Sci., 2021, 14, 2429–2440 RSC.
- J. Li, H. Huang, W. Xue, K. Sun, X. Song, C. Wu, L. Nie, Y. Li, C. Liu, Y. Pan, H. L. Jiang, D. Mei and C. Zhong, Nat. Catal., 2021, 4, 719–729 CrossRef CAS.
- H. Ma, T. Wang, Y. Xu, W. Shi, R. Ma, Z. Xia, Q. Yang, G. Xie and S. Chen, Appl. Catal., B, 2024, 349, 123857 CrossRef CAS.
- D. Liu, J. Hua, W. Zhang, K. Wei, S. Song, Q. Wang, Z. Song, H. Han, C. Ma and S. Feng, Colloids Surf., A, 2024, 695, 134101 CrossRef CAS.
- K. Yuan, K. Tao, T. Song, Y. Zhang, T. Zhang, F. Wang, S. Duan, Z. Chen, L. Li, X. Zhang, D. Zhong, Z. Tang, T. B. Lu and W. Hu, J. Am. Chem. Soc., 2024, 146, 6893–6904 CrossRef CAS PubMed.
- C. Han, X. Zhang, S. Huang, Y. Hu, Z. Yang, T. T. Li, Q. Li and J. Qian, Adv. Sci., 2023, 10, 2300797 CrossRef CAS PubMed.
- Y. Su, Z. Song, W. Zhu, Q. Mu, X. Yuan, Y. Lian, H. Cheng, Z. Deng, M. Chen, W. Yin and Y. Peng, ACS Catal., 2021, 11, 345–354 CrossRef CAS.
- H. Zhang, T. Wang, J. Wang, H. Liu, T. D. Dao, M. Li, G. Liu, X. Meng, K. Chang, L. Shi, T. Nagao and J. Ye, Adv. Mater., 2016, 28, 3703–3710 CrossRef CAS PubMed.
- S. Wang, B. Y. Guan, Y. Lu and X. W. Lou, J. Am. Chem. Soc., 2017, 139, 17305–17308 CrossRef CAS PubMed.
- S. Wang, B. Y. Guan and X. W. D. Lou, J. Am. Chem. Soc., 2018, 140, 5037–5040 CrossRef CAS PubMed.
- T. Wang, L. Shi, J. Tang, V. Malgras, S. Asahina, G. Liu, H. Zhang, X. Meng, K. Chang, J. He, O. Terasaki, Y. Yamauchi and J. Ye, Nanoscale, 2016, 8, 6712–6720 RSC.
- S. Zhao, K. Li, J. Wu, J. Zhang, X. Li, X. Guo and C. Song, ACS Appl. Mater. Interfaces, 2022, 14, 20375–20384 CrossRef CAS PubMed.
- J. Sun, X. Li, J. Li, M. Mu and X. Yin, Colloids Surf., A, 2022, 653, 129940 CrossRef CAS.
- Y. Wang, J. Xu, J. Wan, J. Wang and L. Wang, J. Colloid Interface Sci., 2022, 616, 532–538 CrossRef CAS PubMed.
- Y. Xiao, Q. Zhang, L. Yang, Q. Zhang, X. Fu, L. Cheng and Q. Zeng, Sep. Purif. Technol., 2023, 311, 123172 CrossRef CAS.
- Y. Wang, W. Qian, G. Zhou, S. Zhang, X. Zhu, L. Li, X. Zhu, X. Wang, X. Han and J. Yi, Chem. Eng. J., 2024, 501, 157513 CrossRef CAS.
- X. H. Chen, Y. S. Zhang, W. Bin Li, X. W. Guan, J. W. Ye, L. Chen, H. P. Wang, J. Bai, Z. W. Mo and X. M. Chen, Inorg. Chem. Front., 2022, 9, 2328–2335 RSC.
- G. Zhou, B. Wang and R. Cao, J. Am. Chem. Soc., 2020, 142, 14848–14853 CrossRef CAS PubMed.
- Z. Liang, H. Y. Wang, H. Zheng, W. Zhang and R. Cao, Chem. Soc. Rev., 2021, 50, 2540–2581 RSC.
- A. M. May and J. L. Dempsey, Chem. Sci., 2024, 15, 6661–6678 RSC.
- S. Mandal, S. Natarajan, P. Mani and A. Pankajakshan, Adv. Funct. Mater., 2021, 31, 2006291 CrossRef CAS.
- W. S. Cheon, J. Bu, S. Jung, J. Y. Yang, S. Choi, J. Kim, J. H. Baek, S. Park, M. K. Lee, S. E. Jun, S. H. Park, H. Park, S. A. Lee, S. H. Cho, M. Shokouhimehr, M. Senna and H. W. Jang, Chem. Eng. J., 2024, 489, 151004 CrossRef CAS.
- E. Kecsenovity, B. Endrödi, P. S. Tóth, Y. Zou, R. A. W. Dryfe, K. Rajeshwar and C. Janáky, J. Am. Chem. Soc., 2017, 139, 6682–6692 CrossRef CAS PubMed.
- M. Schreier, J. Luo, P. Gao, T. Moehl, M. T. Mayer and M. Grätzel, J. Am. Chem. Soc., 2016, 138, 1938–1946 CrossRef CAS PubMed.
- C. Y. Toe, Z. Zheng, H. Wu, J. Scott, R. Amal and Y. H. Ng, Angew. Chem., 2018, 130, 13801–13805 CrossRef.
- M. Duan, C. Hu, H. Li, Y. Chen, R. Chen, W. Gong, Z. Lu, N. Zhang, R. Long, L. Song and Y. Xiong, JACS Au, 2022, 2, 1160–1168 CrossRef CAS PubMed.
- P. Li, X. Lu, Z. Wu, Y. Wu, R. Malpass-Evans, N. B. McKeown, X. Sun and H. Wang, Angew. Chem., Int. Ed., 2020, 59, 10918–10923 CrossRef CAS PubMed.
- X. Lu, Z. Jiang, X. Yuan, Y. Wu, R. Malpass-Evans, Y. Zhong, Y. Liang, N. B. McKeown and H. Wang, Sci. Bull., 2019, 64, 1890–1895 CrossRef CAS PubMed.
- Y. Ma, X. Yi, S. Wang, T. Li, B. Tan, C. Chen, T. Majima, E. R. Waclawik, H. Zhu and J. Wang, Nat. Commun., 2022, 13, 1400 CrossRef CAS PubMed.
- Y. Shu, X. Liu, M. Zhang, B. Liu and Z. Wang, Appl. Catal., B, 2024, 346, 123746 CrossRef CAS.
- R. Das, S. Chakraborty and S. C. Peter, ACS Energy Lett., 2021, 6(9), 3270–3274 CrossRef CAS.
- G. Liao, G. Ding, B. Yang and C. Li, Precis. Chem., 2024, 2(2), 49–56 CrossRef CAS PubMed.
- Y. Zhang, L. Cao, G. Bai and X. Lan, Small, 2023, 19, 2300035 CrossRef CAS PubMed.
- M. Zhou, Z. Wang, A. Mei, Z. Yang, W. Chen, S. Ou, S. Wang, K. Chen, P. Reiss, K. Qi, J. Ma and Y. Liu, Nat. Commun., 2023, 14, 2473 CrossRef CAS PubMed.
- S. Gao, Q. Zhang, X. Su, X. Wu, X. G. Zhang, Y. Guo, Z. Li, J. Wei, H. Wang, S. Zhang and J. Wang, J. Am. Chem. Soc., 2023, 145, 9520–9529 CrossRef CAS PubMed.
- J. Wang, W. Zhu, F. Meng, G. Bai, Q. Zhang and X. Lan, ACS Catal., 2023, 13, 4316–4329 CrossRef CAS.
- J. Zhou, J. Li, L. Kan, L. Zhang, Q. Huang, Y. Yan, Y. Chen, J. Liu, S. L. Li and Y. Q. Lan, Nat. Commun., 2022, 13, 4681 CrossRef CAS PubMed.
- M. Kou, W. Liu, Y. Wang, J. Huang, Y. Chen, Y. Zhou, Y. Chen, M. Ma, K. Lei, H. Xie, P. K. Wong and L. Ye, Appl. Catal., B, 2021, 291, 120146 CrossRef CAS.
- Y. N. Gong, W. Zhong, Y. Li, Y. Qiu, L. Zheng, J. Jiang and H. L. Jiang, J. Am. Chem. Soc., 2020, 142, 16723–16731 CrossRef CAS PubMed.
- W. Liu, X. Li, C. Wang, H. Pan, W. Liu, K. Wang, Q. Zeng, R. Wang and J. Jiang, J. Am. Chem. Soc., 2019, 141, 17431–17440 CrossRef CAS PubMed.
- M. Lu, J. Liu, Q. Li, M. Zhang, M. Liu, J. Wang, D. Yuan and Y. Lan, Angew. Chem., 2019, 131, 12522–12527 CrossRef.
- M. Lu, Q. Li, J. Liu, F. M. Zhang, L. Zhang, J. L. Wang, Z. H. Kang and Y. Q. Lan, Appl. Catal., B, 2019, 254, 624–633 CrossRef CAS.
- S. Guo, H. Zhang, Y. Chen, Z. Liu, B. Yu, Y. Zhao, Z. Yang, B. Han and Z. Liu, ACS Catal., 2018, 8, 4576–4581 CrossRef CAS.
- R. Xu, X. S. Wang, H. Zhao, H. Lin, Y. B. Huang and R. Cao, Catal. Sci. Technol., 2018, 8, 2224–2230 RSC.
- S. Yang, W. Hu, X. Zhang, P. He, B. Pattengale, C. Liu, M. Cendejas, I. Hermans, X. Zhang, J. Zhang and J. Huang, J. Am. Chem. Soc., 2018, 140, 14614–14618 CrossRef CAS PubMed.
- P. Fu, C. Chen, C. Wu, B. Meng, Q. Yue, T. Chen, W. Yin, X. Chi, X. Yu, R. Li, Y. Wang, Y. Zhang, W. Luo, X. Liu, Y. Han, J. Wang, S. Xi and Y. Zhou, Angew. Chem., Int. Ed., 2025, 64, e202415202 CrossRef CAS PubMed.
- Y. Xu, J. P. Dong, L. Wang, R. L. Geng, R. Wang, Y. N. Si, S. Q. Zang and T. C. W. Mak, Angew. Chem., Int. Ed., 2025, 64, e202501391 CrossRef CAS PubMed.
- W. Lin, F. Lin, J. Lin, Z. Xiao, D. Yuan and Y. Wang, J. Am. Chem. Soc., 2024, 146, 16229–16236 CrossRef CAS PubMed.
- Z. X. Pan, S. Yang, X. Chen, J. X. Luo, R. Z. Zhang, P. Yang, Q. Xu and J. Y. Yue, Chem. Eng. J., 2024, 493, 152798 CrossRef CAS.
- S. Yang, R. Sa, H. Zhong, H. Lv, D. Yuan and R. Wang, Adv. Funct. Mater., 2022, 32, 2110694 CrossRef CAS.
- Y. Zhang, Y. Liu, H. Li, G. Bai and X. Lan, Chem. Eng. J., 2024, 489, 151479 CrossRef CAS.
- R. K. Yadav, A. Kumar, N. J. Park, K. J. Kong and J. O. Baeg, J. Mater. Chem. A, 2016, 4, 9413–9418 RSC.
- K. Guo, X. Zhu, L. Peng, Y. Fu, R. Ma, X. Lu, F. Zhang, W. Zhu and M. Fan, Chem. Eng. J., 2021, 405, 127011 CrossRef CAS.
- Z. Fan, Y. Cai, Z. Yang, X. Zhang, R. Shao, S. Zhong, L. Zhao, D. Liu and S. Bai, Appl. Catal., B, 2024, 351, 123979 CrossRef CAS.
- Y. Liu, Y. Wang, J. Shang, J. Peng and T. Zhu, Appl. Catal., B, 2024, 350, 123937 CrossRef CAS.
- Y. Yang, H. Y. Zhang, Y. Wang, L. H. Shao, L. Fang, H. Dong, M. Lu, L. Z. Dong, Y. Q. Lan and F. M. Zhang, Adv. Mater., 2023, 35, 2304170 CrossRef CAS PubMed.
- X. Yang, X. Lan, Y. Zhang, H. Li and G. Bai, Appl. Catal., B, 2023, 325, 122393 CrossRef CAS.
- L. Zou, R. Sa, H. Zhong, H. Lv, X. Wang and R. Wang, ACS Catal., 2022, 12, 3550–3557 CrossRef CAS.
- M. Zhang, M. Lu, Z. Lang, J. Liu, M. Liu, J. Chang, L. Li, L. Shang, M. Wang, S. Li and Y. Lan, Angew. Chem., 2020, 132, 6562–6568 CrossRef.
- J. C. Soc and C. Commun, J. Chem. Soc., Chem. Commun., 1983, 536–538 Search PubMed.
- H. Takeda, K. Koike, H. Inoue and O. Ishitani, J. Am. Chem. Soc., 2008, 130, 2023–2031 CrossRef CAS PubMed.
- S. A. Lee, J. Bu, J. Lee and H. W. Jang, Small Sci., 2023, 3, 2200109 CrossRef CAS PubMed.
- W. S. Cheon, S. G. Ji, J. Kim, S. Choi, J. W. Yang, S. E. Jun, C. Kim, J. Bu, S. Park, T. H. Lee, J. Wang, J. Y. Kim, S. A. Lee, J. Y. Kim and H. W. Jang, EES Catal., 2025, 3, 140–151 RSC.
- Y. N. Gong, J. H. Mei, W. J. Shi, J. W. Liu, D. C. Zhong and T. B. Lu, Angew. Chem., Int. Ed., 2024, 63, e202318735 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2025 |