Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Phthalimide: a potential warhead for switchable and bioorthogonal conjugation
Kosuke
Chiba
 ab,
Takumi
Yoshida
a,
Kana
Okada
a,
Minoru
Ishikawa†
ab,
Takumi
Yoshida
a,
Kana
Okada
a,
Minoru
Ishikawa†
 b,
Yuichi
Hashimoto
b,
Satoshi
Obika
b,
Yuichi
Hashimoto
b,
Satoshi
Obika
 *a and
Takao
Yamaguchi
*a and
Takao
Yamaguchi
 *ab
*ab
aGraduate School of Pharmaceutical Sciences, The University of Osaka, 1-6 Yamadaoka, Suita, Osaka 565-0871, Japan. E-mail: obika@phs.osaka-u.ac.jp; yamaguchi-ta@phs.osaka-u.ac.jp
bInstitute of Molecular and Cellular Biosciences, The University of Tokyo, 1-1-1 Yayoi, Bunkyo-ku, Tokyo 113–0032, Japan
First published on 6th August 2025
Abstract
4-Azidophthalimide, previously reported as a photoaffinity labeling (PAL) tag, was repurposed as a post-conjugation warhead. Using the phthalimide–amine reaction, cyanine amines were successfully conjugated to target proteins after PAL and later replaced with other amines, demonstrating its “switchable” nature. These findings highlight phthalimide's unique characteristics as a novel chemical tool for bioorthogonal conjugations.
Elucidating both the on-target and off-target proteins of bioactive small molecules is essential for understanding their mechanisms of action and potential side effects. To address this challenge, various affinity-based target identification (ID) methods have been developed.1,2 Among these, affinity labeling stands out as a particularly powerful technique for identifying target proteins,3,4 as it allows protein labeling under conditions that closely resemble the native biological environment. This approach involves introducing an electrophilic or photo-reactive group into bioactive small molecules, enabling covalent bond formation with nearby proteins. The labeled proteins can then be visualized using fluorophores or enriched via biotin for pull-down and subsequent analysis. First introduced in the 1960s, affinity labeling has undergone substantial refinement over the decades.5–7 In particular, photoaffinity labeling (PAL) has gained widespread attention due to its broad reactivity with amino acid residues and its spatiotemporal precision enabled by light activation.8 To minimize interference with the bioactivity of small molecules, compact PAL tags have been designed, typically consisting of a small photo-reactive group (e.g., diazirine, arylazide) and a biorthogonal handle (e.g., azide, alkyne) for subsequent click reactions with fluorophores or biotin.9–11
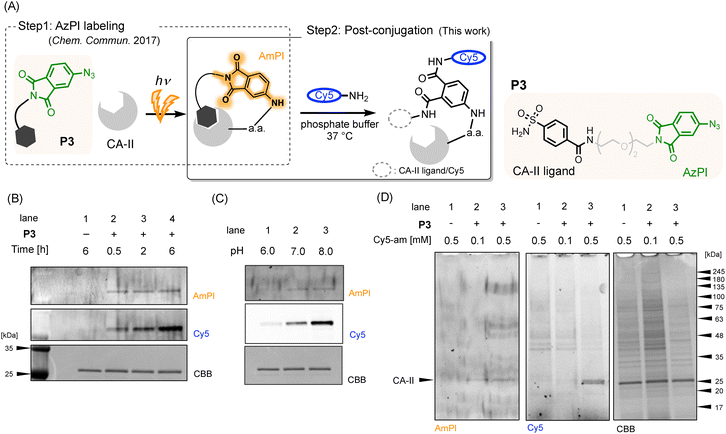
In our previous work, we introduced 4-azidophthalimide (AzPI) as a compact PAL tag (Fig. 1A).12,13 Upon low-energy UV irradiation (365 nm), AzPI generates a nitrene intermediate that covalently binds to target proteins, yielding 4-aminophthalimide (AmPI), a fluorescent group that enables protein detection. However, its fluorescence quantum yield is lower than that of commonly used fluorophores (e.g., cyanine and rhodamine dyes), resulting in a low signal-to-noise (S/N) ratio and limited detection of low-abundance proteins.
 | ||
| Fig. 1 Post-conjugation strategy based on the phthalimide structure of AzPI. (A) Photo-affinity labeling (PAL) utilizing the AzPI unit [ref. 12]. (B) Post-conjugation via amine substitution at the phthalimide moiety. | ||
To overcome this limitation, we propose repurposing the phthalimide scaffold of AzPI as a post-conjugation warhead following PAL (Fig. 1B). Phthalimide is a well-established electrophilic moiety known for its reactivity with amines—particularly in the Gabriel synthesis—and has been employed in various chemical transformations.14–16 Upon reaction with an amine, phthalimide can yield either an N-substituted product (e.g., compound A in Fig. 2) or a ring-opened structure (e.g., compound B in Fig. 2). Notably, this reaction proceeds under ambient conditions, with the N-substituted product formed via a ring-closure mechanism following the addition of a single amine.16 We hypothesize that the phthalimide moiety in AmPI can function as a post-conjugation warhead under physiological conditions. Moreover, by leveraging the ring-opening and closure cycle, a switchable post-conjugation strategy may be achieved using different amines.
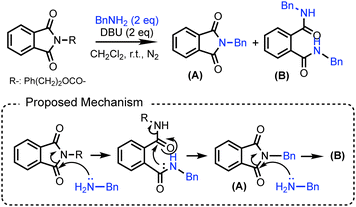 | ||
| Fig. 2 Amine substitution on the phthalimide moiety and its proposed mechanism [ref. 16]. | ||
Here we report the reactivity of the phthalimide moiety with amines under aqueous conditions and its application for post-conjugation following AzPI-mediated PAL. Using a cyanine-based amine, we demonstrated successful post-conjugation through the AzPI scaffold. Intriguingly, a switchable post-conjugation strategy was also achieved by introducing two distinct cyanine (Cy5 and Cy7) amines. These findings highlight the potential of AzPI for target ID and establish the phthalimide warhead as a versatile chemical tool for switchable and biorthogonal conjugation.
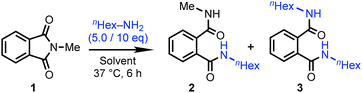
First, we evaluated whether the phthalimide-amine reaction could occur under aqueous conditions (Table 1). To verify this, N-methylphthalimide (1) was selected as a model compound. Initially, compound 1 was dissolved in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) mixture of THF and water and stirred with an excess amount of n-hexylamine at 37 °C (entry 1). Under these conditions, the reaction proceeded efficiently, reaching nearly 90% conversion after 6 h. To assess the reactivity under more physiologically relevant conditions, the reaction was carried out in phosphate buffer (pH 8.0) by suspending compound 1 (entry 2). This condition also facilitated the reaction, affording products 2 and 3 with a total conversion of approximately 60%. Notably, the major product was the di-substituted product 3, indicating that the reaction proceeded through a ring-closure step following the initial n-hexylamine addition, as illustrated in Fig. 2. This reaction showed time-dependent progression from mono- to di-substitution (Table S1, entries 1 and 2, SI), and also proceeded with 4-amino-N-methylphthalimide, resembling the AzPI PAL product (entry 3). These findings support the suitability of phthalimide as a reactive warhead for amine post-conjugation and demonstrate its potential for a switchable conjugation strategy under aqueous, biocompatible conditions.
1 (v/v) mixture of THF and water and stirred with an excess amount of n-hexylamine at 37 °C (entry 1). Under these conditions, the reaction proceeded efficiently, reaching nearly 90% conversion after 6 h. To assess the reactivity under more physiologically relevant conditions, the reaction was carried out in phosphate buffer (pH 8.0) by suspending compound 1 (entry 2). This condition also facilitated the reaction, affording products 2 and 3 with a total conversion of approximately 60%. Notably, the major product was the di-substituted product 3, indicating that the reaction proceeded through a ring-closure step following the initial n-hexylamine addition, as illustrated in Fig. 2. This reaction showed time-dependent progression from mono- to di-substitution (Table S1, entries 1 and 2, SI), and also proceeded with 4-amino-N-methylphthalimide, resembling the AzPI PAL product (entry 3). These findings support the suitability of phthalimide as a reactive warhead for amine post-conjugation and demonstrate its potential for a switchable conjugation strategy under aqueous, biocompatible conditions.
| Entry | Solvent | Yield [%] | Total conversion yield [%] | ||
|---|---|---|---|---|---|
| 1 | 2 | 3 | |||
| a Conditions: 1.2 mmol of 1, 12 mmol (10 equiv.) of n-hexylamine, and 12 mL of solvent. b Conditions: 0.67 mmol of 1, 3.3 mmol (5.0 equiv.) of n-hexylamine, and 67 mL of buffer. | |||||
| 1a | THF/H2O = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
— | 17 | 72 | 89 |
| 2b | Phosphate buffer (pH 8.0) | — | Trace | 58 | 58 |
Next, we applied the phthalimide-amine reaction to AzPI-based PAL. In our previous study, we demonstrated the selective labeling and detection of carbonic anhydrase II (CA-II) using the AzPI probe P3, which contains a CA-II ligand, benzene sulfonamide.12,13 Labeling was successfully achieved both in a mixture of CA-II and HEK293 cell lysate, as well as in living bovine red blood cells. To assess post-conjugation, P3 was incubated with CA-II in phosphate buffer and irradiated with UV light (365 nm), thereby activating the AzPI moiety and inducing covalent bond formation with CA-II (Fig. 3A, step1). The resulting AmPI-bound CA-II was then incubated with cyanine5 amine (Cy5-amine; see Fig. S1 for the structure, SI) to enable post-conjugation via the phthalimide moiety (Fig. 3A, step2). Conjugation was analyzed by SDS-PAGE followed by fluorescence detection. Fluorescence bands on the gel were visualized using a fluorescence detector capable of distinguishing AmPI-originated fluorescence (λex: 455–485 nm, λem: 508–557 nm) and Cy5-originated fluorescence (λex: 610–660 nm, λem: 672–720 nm).
We initially tested the conjugation at pH 8.0, guided by the favourable reactivity of N-methylphthalimide under this condition (Table 1 and Fig. 3B). As a result, Cy5-originated fluorescence was clearly observed at the CA-II band, with the fluorescence intensity increasing over the incubation time with Cy5-amine (lanes 2–4), confirming successful conjugation. No fluorescence—neither from AmPI nor from Cy5—was observed in the absence of P3 (lane 1), demonstrating that AzPI-mediated CA-II labeling (step 1 in Fig. 3A) is essential for subsequent Cy5-amine conjugation (step 2). These results strongly support that Cy5-amine is covalently attached to CA-II through the phthalimide moiety of AmPI. Furthermore, a clear pH-dependence was observed between pH 6.0 and 8.0 (Fig. 3C), consistent with the proposed mechanism in which basic conditions favour nucleophilic attack by amines. Interestingly, AzPI-mediated PAL was less efficient at pH 6.0 (lane 1), as indicated by the lower AmPI-originated fluorescence, although the cause of this reduced labeling efficiency remains unclear.
We further evaluated whether this post-conjugation method could be applied in a biologically relevant environment by adding HEK293 cell lysate. As shown in Fig. 3D, a strong Cy5 fluorescent band was detected at approximately 25 kDa in the presence of 0.5 mM P3 (lane 3), and its position correlated with that of the AmPI fluorescent band. As previously reported, P3 selectively labels CA-II in a ligand-directed manner,12 supporting that Cy5-amine selectively conjugated to CA-II even in the presence of other cellular proteins. Notably, the AmPI fluorescence exhibited a low S/N ratio, with significantly weaker intensity compared to Cy5 fluorescence (Fig. 3D, lane 3). These results further demonstrate that this post-conjugation strategy effectively compensates for the low fluorescence quantum yield of AmPI. We also observed several weak Cy5 fluorescent bands around 40 kDa even in the absence of P3, suggesting that some proteins underwent nonspecific labeling by Cy5-amine. We hypothesize that this may result from the reaction of Cy5-amine with electrophilic succinimide intermediates, which are formed via the deamidation of asparagine or glutamine residues in proteins.17 Phthalimide's reactivity with endogenous amines, particularly lysine residues, is also noteworthy. We quantified Cy5 fluorescence and CA-II dimer formation based on CBB staining (Fig. S3, SI). While Cy5 intensity increased over time with Cy5-amine incubation, dimer levels remained unchanged, suggesting minimal lysine involvement under these conditions.
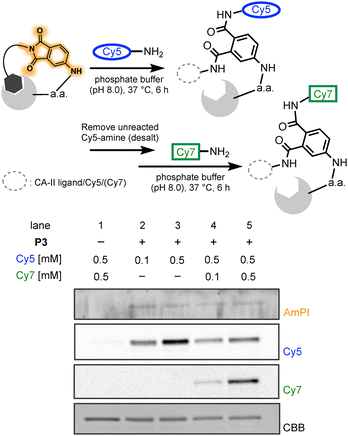
Finally, encouraged by the reaction described in Table 1—where the bis(hexylamine)-conjugated product 3 was likely formed by the phthalimide's ring-closing and opening cycle—we explored the concept of “switchable” post-conjugation using the phthalimide warhead. As illustrated in Fig. 4. Cy5-amine was first conjugated to the phthalimide moiety of AmPI after PAL. After removing unreacted Cy5-amine using a desalting column, the sample was incubated with Cy7-amine (see Fig. S1 for the structure, SI), enabling a switchable conjugation step. Since Cy7 and Cy5 possess distinct fluorescent properties, this allowed us to track the substitution process. As expected, Cy7-derived fluorescence was observed only in the presence of AmPI, with signal intensity increasing in a concentration-dependent manner (Fig. 4, lanes 1, 4, and 5). Moreover, the Cy5 fluorescence intensity decreased upon Cy7-amine addition, indicating that Cy5 was displaced by Cy7 (Fig. 4, lanes 3 and 5). These results demonstrated that the phthalimide warhead can serve as a switchable conjugation handle under biologically relevant conditions, consistent with the mechanism proposed in Fig. 1B.
In this study, we propose a novel post-conjugation method by repurposing the phthalimide structure of AzPI. Cy5-amine was successfully introduced via phthalimide-amine post-conjugation, enabling target ID with a high S/N ratio compared to AmPI fluorescence, thereby enhancing the utility of AzPI-based PAL. Furthermore, we demonstrated that Cy7-amine could be conjugated to target protein even after initial Cy5-amine labeling, likely via a ring-opening and closure mechanism of the phthalimide scaffold. These findings reveal that the phthalimide moiety functions as a switchable post-conjugation unit. Previous pioneering studies have explored post-conjugation strategies using disulfide bonds or hydrazone/oxime exchange reactions.18,19 In particular, disulfide bonds are known to exhibit switchable behavior via redox interconversion.20 Our study presents a new approach to post-conjugation for chemical biology applications, utilizing AzPI not only as a novel warhead but also as a scaffold with in situ switchable (or replaceable) reactivity. This approach could enable more definitive target ID by initially conjugating a biotin-amine for affinity-based enrichment, followed by switching to a cyanin-amine for fluorescence-based detection. Notably, this concept extends beyond AzPI and may be broadly applicable to other bioactive molecules by incorporating the phthalimide moiety into bioactive molecules such as antibodies, peptides, and oligonucleotides.
We recognize two limitations of the current study. First, the in vitro phthalimide–amine reaction proceeded more slowly than the model reaction described in Table 1, likely due to the lower effective concentration of the phthalimide moiety in the protein-conjugated state. Structural optimization of AzPI may improve its reactivity. Second, this concept was validated using only a single, well-characterized ligand-target pair (benzene sulfonamide and CA-II). To broaden its applicability, we aim to extend this approach to bioactive molecules with unknown targets. Despite these limitations, we believe that AzPI-based PAL holds strong promise as a versatile platform for post-labeling functionalization and target identification in biological systems.
This work was supported in part by the Japan Society for the Promotion of Science (JSPS) KAKENHI (grant numbers: 26810091, 16K17930, and 20K05748), by the Japan Agency for Medical Research and Development (AMED) (grant numbers: JP19am0401003, JP21ae0121022, JP21ae0121023, and JP21ae0121024), by Institute for Open and Transdisciplinary Research Initiatives (OTRI) (The fifth wave program), and by Hoansha Foundation.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the SI.Supplementary figures, experimental procedures, and 1H and 13C NMR spectral data. See DOI: https://doi.org/10.1039/d5cc03171k
Notes and references
- M. Schenone, V. Dančík, B. K. Wagner and P. A. Clemons, Nat. Chem. Biol., 2013, 9, 232–240 CrossRef CAS PubMed.
- K. Shiraiwa, R. Cheng, H. Nonaka, T. Tamura and I. Hamachi, Cell Chem. Biol., 2020, 27, 970–985 CrossRef CAS PubMed.
- J. Ha, H. Park, J. Park and S. B. Park, Cell Chem. Biol., 2021, 28, 394–423 CrossRef CAS PubMed.
- M. Chen, M. Asanuma, M. Takahashi, Y. Shichino, M. Mito, K. Fujiwara, H. Saito, S. N. Floor, N. T. Ingolia, M. Sodeoka, K. Dodo, T. Ito and S. Iwasaki, Cell Chem. Biol., 2021, 28, 475–486.e8 CrossRef CAS PubMed.
- L. Wofsy, H. Metzger and S. J. Singer, Biochemistry, 1962, 1, 1031–1039 CrossRef CAS PubMed.
- S. Siriwongsup, A. M. Schmoker, S. B. Ficarro, J. A. Marto and J. Kim, Chemistry, 2024, 10, 1306–1315 CrossRef CAS PubMed.
- Y. Kawamata, K. A. Ryu, G. N. Hermann, A. Sandahl, J. C. Vantourout, A. K. Olow, L.-T. A. Adams, E. Rivera-Chao, L. R. Roberts, S. Gnaim, M. Nassir, R. C. Oslund, O. O. Fadeyi and P. S. Baran, Nat. Chem., 2023, 15, 1267–1275 CrossRef CAS PubMed.
- J. L. Babulic, F. V. De León González and C. J. Capicciotti, Curr. Opin. Chem. Biol., 2024, 80, 102456 CrossRef CAS PubMed.
- Z. Li, P. Hao, L. Li, C. Y. J. Tan, X. Cheng, G. Y. J. Chen, S. K. Sze, H.-M. Shen and S. Q. Yao, Angew. Chem., Int. Ed., 2013, 52, 8551–8556 CrossRef CAS PubMed.
- A. V. West, Y. Amako and C. M. Woo, J. Am. Chem. Soc., 2022, 144, 21174–21183 CrossRef CAS PubMed.
- A. C. S. Page, S. O. Scholz, K. N. Keenan, J. N. Spradlin, B. P. Belcher, S. M. Brittain, J. A. Tallarico, J. M. McKenna, M. Schirle, D. K. Nomura and F. D. Toste, Chem. Sci., 2022, 13, 3851–3856 RSC.
- K. Chiba, M. Asanuma, M. Ishikawa, Y. Hashimoto, K. Dodo, M. Sodeoka and T. Yamaguchi, Chem. Commun., 2017, 53, 8751–8754 RSC.
- K. Chiba, Y. Hashimoto and T. Yamaguchi, Chem. Pharm. Bull., 2017, 65, 994–996 CrossRef CAS PubMed.
- M. S. Gibson and R. W. Bradshaw, Angew. Chem., Int. Ed. Engl., 1968, 7, 919–930 CrossRef CAS.
- L. Becerra-Figueroa, A. Ojeda-Porras and D. Gamba-Sánchez, J. Org. Chem., 2014, 79, 4544–4552 CrossRef CAS PubMed.
- M. Chanmiya Sheikh, S. Takagi, A. Ogasawara, M. Ohira, R. Miyatake, H. Abe, T. Yoshimura and H. Morita, Tetrahedron, 2010, 66, 2132–2140 CrossRef.
- N. E. Robinson and A. B. Robinson, Proc. Natl. Acad. Sci. U. S. A., 2001, 98, 12409–12413 CrossRef CAS PubMed.
- I. Hamachi, T. Nagase and S. Shinkai, J. Am. Chem. Soc., 2000, 122, 12065–12066 CrossRef CAS.
- Y. Takaoka, H. Tsutsumi, N. Kasagi, E. Nakata and I. Hamachi, J. Am. Chem. Soc., 2006, 128, 3273–3280 CrossRef CAS PubMed.
- S. L. Kuan, T. Wang and T. Weil, Chem. – Eur. J., 2016, 22, 17112–17129 CrossRef CAS PubMed.
Footnote |
| † Current Address: Graduate School of Life Sciences, Tohoku University, 2-1-1 Katahira, Aoba-ku, Sendai, Miyagi 980-8577, Japan. |
| This journal is © The Royal Society of Chemistry 2025 |