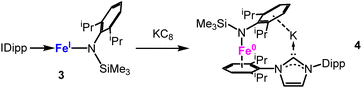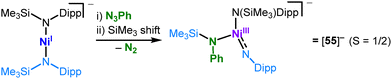Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Linear open-shell 3d-metal silylamides – a versatile tool in coordination chemistry
Alessandra
Casnati
a and
C. Gunnar
Werncke
 *ab
*ab
aDepartment of Chemistry, Philipps-University Marburg, Hans-Meerwein-Str. 4, D-35037 Marburg, Germany. E-mail: gunnar.werncke@chemie.uni-marburg.de
bFaculty of Chemistry, Institute of Inorganic Chemistry and Crystallography, Leipzig University, Johannisallee 29, D-04103 Leipzig, Germany. E-mail: gunnar.werncke@chemie.uni-leipzig.de
First published on 15th July 2025
Abstract
Linear 3d-metal complexes with open-shell metal ions have fascinated coordination chemists for decades. Originally the focus was on metal(II) species, yet the synthetic possibilities to acquire their reduced analogues, together with the introduction of NHCs as stabilizing co-ligands, have expanded the structural knowledge and feasible oxidation states to +I and even 0. Despite a certain wealth of structural data, insights into the reactivity or even catalytic applications of such compounds remained comparably thin for a long time given the intrinsic lability of these coordinatively and electronically unsaturated compounds. In this feature article we review the emerging chemistry and reactivity of silylamide based species, as mostly for these a comprehensive, broader picture of their reactivity patterns has been developed in recent years. Together with related, selected reactivity examples of other linear metal complexes, this review will serve as an entry point for interested molecular organic/inorganic chemists to join the scientific endeavours within this field.
Introduction
Low-coordinate open-shell 3d-metal complexes, more precisely linear complexes, have fascinated inorganic chemists for decades, as reflected in a number of reviews over time.1,2 The interest is founded foremost in acquiring a fundamental understanding of an electronically and coordinatively highly unsaturated metal ion. Furthermore, these molecules may provide general insights into elusive low-coordinate intermediates in catalysis. Seminal research on low-coordinate 3d-metal complexes dates back to the 1960s when Bürger and Wannagat used the hexamethyldisilazanide ligand (–N(SiMe3)2) for the synthesis of the di- or trivalent 3d-transition metal complexes (Mn(N(SiMe3)2)n) (n = 2, 3).3,4 They were unaware at the time that the divalent (M(N(SiMe3)2)2) actually forms dimers (Mn–Co)5,6 or solvent adducts (Cr–Ni)6–12 but can occur as monomeric in solution.10,12–14 Evidence for actual linear open-shell 3d-metal complexes was obtained in the gas phase via electron diffraction studies in 1985 for Mn(CH2tBu)2.15 In the same year Mn(C(SiMe3)3)2 (Fig. 1, left) was structurally characterized by single-crystal X-Ray diffraction analysis.16 In the following decades a variety of two-coordinate 3d-metal(II) complexes bearing different sterically encumbering anionic ligands X− such as terphenyls,17 (thio)phenolates,18 primary arylamides19–21 or secondary (aryl)silylamides (Fig. 1) were reported.21,22 These complexes are often stabilized by intramolecular metal–arene interactions,2 with interligand dispersion forces being a further contributing factor.23Despite the well-developed synthetic access, comprehensive reactivity studies on linear complexes is still lacking. For a long time it allowed only for a fragmented insight into the reactivity of a two-coordinate metal(II) ion. A fundamental problem is associated with maintaining the integrity of the central MX2 fragment. The reaction with substrates is often associated with insertion into the M–X bond, subsequent rearrangements and/or redox state dis/comproportionation, often in an unpredictable manner. A representative example is the reaction of Fe(Aryl)2 with CO (Scheme 1): it leads amongst others to purely organic products and aryl free metal carbonyls.24
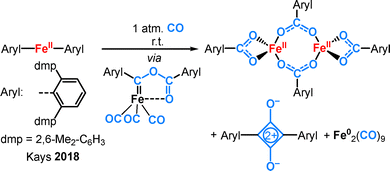 | ||
| Scheme 1 Deconstruction of a linear iron(II) aryl complex by reaction with CO under M–L bond insertion, redox disproportionation and complete ligand rearrangement. | ||
Furthermore, the coordinatively and electronically deficient nature of a two-coordinate 3d-metal(II) ion lacks to a certain degree the electrons/reducibility for many bond activation and cleavage processes. This was overcome by reduction of the metal ion to the monovalent and even zerovalent state, aided by N-heterocyclic carbenes (NHC) as well as cyclic amino alkyl carbenes (cAAC, Fig. 2).25 Here the π-accepting capabilities of carbenes stabilize the low-valent metal center via back donation into the empty p-orbital of the carbene.25,26 The electron-accepting nature of especially cAACs is exemplified for Mn(cAAC)2:27 Here, the metal is not zero-valent but adopts the oxidation state +I with a 4s1d5 configuration. The metal(I) ion couples antiferromagnetically with an electron that resides equally on both cAAC ligands. For Fe and Co, delocalisation of spin density onto the carbene ligands is less pronounced (approx. 0.25e−)28,29 and is mostly absent for Ni due to its stable d10 configuration.30 For cationic complexes [ML2]+ (L = NHC or cAAC; M = Cr, Fe–Ni)28,31 the spin density is predicted to be metal centered due to charge-dependent d-orbital contraction.
Scope
In this perspective we will give an overview of the synthesis of linear metal(II/I) complexes, their electronic properties and emerging reactivity patterns. We will mostly regard compounds containing silylamide ligands (=X), namely those of the type [MX2]0,− and MX(NHC). The focus on silylamide complexes is due to the fact that reports on respective metal complexes as stable platforms for substrate activation are the most prevalent and recent, thus allowing the extraction of predictable knowledge for future endeavours. Suitable studies on related and other complexes will be addressed where appropriate or at the end. By that we aim to inspire interested chemists to further exploit the chemistry of these seemingly simple and intriguing yet synthetically challenging class of molecules.Synthesis
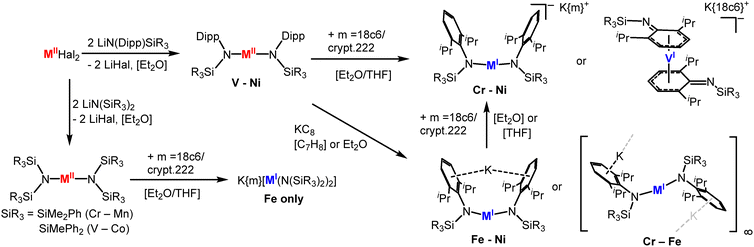
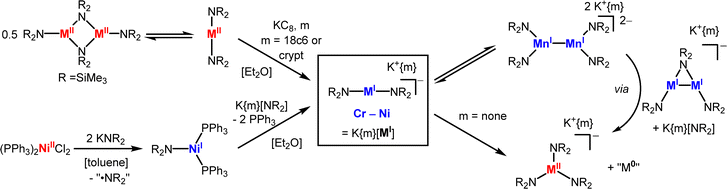
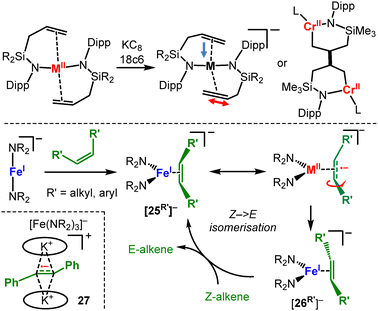
M(NR2)2 type complexes (R = SiMe3) with comparably less bulky disilazanide ligands form dimers in the solid state (Mn–Co) or are isolated as mononuclear solvent adducts (Cr–Cu6–10,45), yet exist in part as monomers in solution (Scheme 3). This behaviour allows for their reduction with KC8 to [M(NR2)2]− (= [MI]− for R = SiMe3) for Cr–Ni in the presence of crypt.222 or 18c6.13,32,46 The masking agents are mandatory as otherwise oxidation state redistribution to metal(II) and metal(0) species is observed:46 it is likely induced by the competition between the 3d-metal(I) and AM cations for the amide ligands. For [Ni(NR2)2]− a different synthetic approach proved synthetically more viable, given the notoriously labile Ni(NR2)2 precursor.4,10 In monovalent [Ni(PPh3)2NR2], which is obtained from reduction and ligand exchange of [Ni(PPh3)2Cl2] with LiNR2, both phosphines can be easily replaced by KNR2 in the presence of 18-crown-6 or crypt.222 (Scheme 3).13 The synthesis of these unencumbered metal(I) complexes can be performed in THF without solvent coordination, which hints to the electron rich nature of the metal(I) ion in these anionic complexes. Furthermore, the anionic charge (partially) compensates the reduced steric encumbrance of the ligands and gives an open metal site for substrate activation. The reduced steric protection leads for [Mn(NR2)2]− to dimerization in the solid state with formation of an unsupported Mn–Mn bond.32 This kind of behaviour is in stark contrast to the amide bridged neutral parent compounds. It can be reasoned by the electron configuration of 3d54s1 for MnI, and is in line with the observations made for the few other Mn(I)-Mn(I) dimers.47 It hints to the decomposition pathway of these less encumbered metal(I) silylamides: dimerization may initiate amide ligand loss to give [M2(NR2)2(μ2-NR2)]− (Scheme 3), which was isolated in case of nickel.13 Subsequent redox disproportionation and ligand distribution then yield in [M(NR2)3]− and amorphous metal M0.32
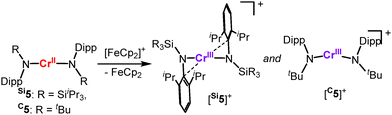
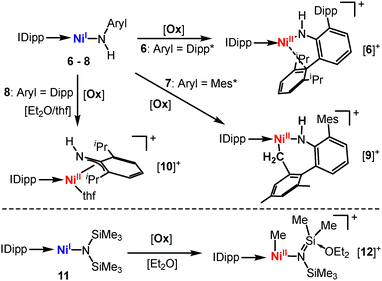
Oxidation of [Cr(N(Dipp)SiiPr3)2], Si5,33 leads to cationic [Si5]+ that displays a shorter metal-amide bond with a persistent strong secondary metal–arene interaction due to its electronic deficiency (Scheme 6). The seemingly very similar [Cr(N(Dipp)tBu)2], C5,57 does not exhibit such secondary interactions – even upon oxidation ([C5]+). Here, it is attributed to a stronger π-donating effect of the alkyl amide ligands. Oxidation of the heteroleptic complexes Ni(IDipp)(NHDipp*), 6 (Dipp* = 2,6-Dipp-C6H3),53 invokes stronger secondary metal–ligand interactions ([6]+, Scheme 7, top), overall illustrating nicely the impact of the complexes’ charge and oxidation state of the metal on its electron deficiency in these linear complexes. Reducing the steric bulk for the nickel complexes Ni(IDipp)(NHAryl) results in a C–H bond activated complex ([9]+, Aryl = Mes* =2,6-Mes-C6H3)53 or for Aryl = Dipp in disruption of the aromaticity of the arylimido ligand and an allyl-like coordination of the ligand ([10]+).52 For the silylamide complex [Ni(IDipp)N(SiMe3)2], 11, oxidation effectuates a methyl-shift from the ligand to the metal ([12]+, Scheme 7, bottom).52
Structural features and electronic as well as magnetic properties of two-coordinate metal complexes
 | ||
| Fig. 3 (A) Exemplary d-orbital splitting scheme of a linear 3d-metal(II) complex with σ-donor ligands. (B) calculated d-orbital splitting of the iron(II) silylmethanide Fe(C{SiMe3}3)2 under consideration of 4s orbital involvement.60 (C) Non-Aufbau electron configuration of a d7-ion calculated for [CoII(C{SiMe2Onaph}3)2].59 (D) Calculated d-orbital splitting of the iron(I) silylmethanide [Fe(C{SiMe3}3)2]−.58 (E) d-Orbital splitting of the metal(I) silylamides [MI(N{SiMe3}3)2]− (M = Cr–Co).58 (F) Magnetic relaxation pathways (depicted for a MJ = ±7/2 system). | ||
General coordination behaviour. As intuitively expected, the general coordination behaviour of linear complexes should be dictated by their coordinative and electronic unsaturation. Indeed, in the neutral and moreover cationic state the complexes behave mostly as Lewis acids. This property is however lost in the anionic state, a difference that can be nicely illustrated for the three metal(I) complexes shown in Scheme 8. The cationic [Fe(IMes)2]+ readily forms Lewis-base adducts with THF ([10]+, Scheme 8A).69 In contrast, the anionic [Fe(NR2)2]− is indifferent towards any regular donor ligands (e.g. phosphines or NHCs).70 Moreover, in the interaction with the ethylene bridged diphosphine 1,2-bis(diphenylphosphine)ethylene the metal in [Fe(NR2)2]− does not bind via the P-donors but the ethylene backbone ([11]−, Scheme 8B).
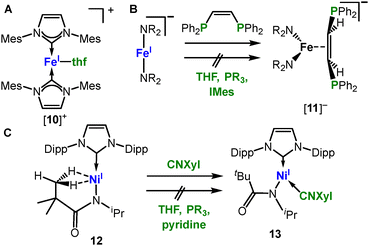 | ||
| Scheme 8 Examples for the general coordination behaviour of linear open-shell metal(I) complexes towards donor ligands. | ||
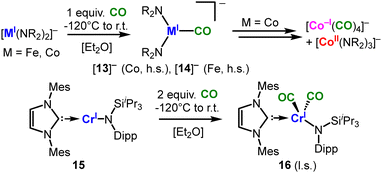
The neutral Ni(I) amidate complex 12 (Scheme 8C) displays bis(σ-C–H)-agostic interactions between the amidate's tert-butyl function and the nickel(I) ion due to its coordinative unsaturation. On the other hand, the complex is inert towards donor-ligands with the exception of isonitriles that are also capable of M → L backbonding (13). This observation emphasizes the role of the overall charge of the complex, irrespective of a low oxidation state of the central metal ion. The second effect of a low-coordinate state is the strong preference for higher spin-states. It was nicely demonstrated in the case of CO: its reaction with [MI]− at low temperatures yields in the isolation of monocarbonyl adducts ([13]− and [14]−, Scheme 9, top).71 Despite considerable M → ligand π-backbonding the high-spin state is maintained. The monocarbonyl complexes are however highly labile and undergo dismutation reactions (e.g. for cobalt it gives [Co(CO)4]− and [Co(NR2)3]−).71 In the case of the neutral chromium(I) complex 15 the coordination of two CO ligands are sufficient to invoke the low-spin state (16, Scheme 9, bottom).
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/b_char_e002.gif) C/C
C/C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/b_char_e001.gif) C/C
C/C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/b_char_e002.gif) N bond activation.
Besides CO, alkenes and alkynes are known to interact strongly with low-valent 3d-metals in organometallic chemistry. The group of Tilley reported first on the heteroleptic iron(I) complex [(IDipp)Fe(N{Dipp}SiMe3)] as a catalyst for cyclotrimerisation of terminal and internal alkynes (Scheme 10).49 Detailed studies of the reaction mechanism and isolation of intermediates gave a bimetallic pathway under carbene dissociation (17).72 At 60 °C oxidative coupling takes place resulting in the dimetalla(II) butadienediyl complex 18 as evidenced by isolation of both reaction intermediates. At this point a third alkyne likely coordinates and subsequently inserts into a metal–carbon bond to generate a dimetalla cycloheptatriene that finally undergoes ring closure to produce the aromatic ring. The metal(I) silylamide fragment is either captured by the previously liberated NHC ligand or re-enters into the catalytic cycle by alkyne coordination. In the case of anionic complexes [MI]− (Cr–Co) the interaction with alkynes leads mostly only to η2-alkyne complexes ([M19R′]−, Scheme 11, top).73 For cobalt alkyne coordination is weak and reversible, yet becomes stronger when going to the lighter metals (Co → Cr). CASSCF calculations on these complex series revealed their primary electronic structure as metal(II) radical anions (Scheme 11, bottom), whereas the textbook dichotomy of such compounds as π-complexes or metallacyclopropenes are a lesser factor. One has to bear in mind that the classic description of π-complexes along the Chatt–Dewar–Duncanson concept was originally adopted for closed-shell/low-spin organometallics. Here, the π-backbonding proceeds from a doubly occupied metal centered orbital with no substantial unpaired spin transfer. The observed deviation for the [MI]− complexes shown herein is likely a result of their high-spin state in conjunction with the anionic charge that facilitates the electron transfer to the π*-orbital of the substrate. Steric factors are also in play as for the more encumbered [FeI(N{Dipp}SiMe3)2]− alkyne coordination is labile.43 A similar bond trichotomy is formulated for side-on bound organo nitrile iron complexes ([20R′]−, Scheme 11, top).74 The radical anionic nature of the bound alkyne rationalizes subsequent reduction to bismetallated alkene dianions in the case of manganese ([21]2−) and chromium.73 Moreover, for manganese a series of C
N bond activation.
Besides CO, alkenes and alkynes are known to interact strongly with low-valent 3d-metals in organometallic chemistry. The group of Tilley reported first on the heteroleptic iron(I) complex [(IDipp)Fe(N{Dipp}SiMe3)] as a catalyst for cyclotrimerisation of terminal and internal alkynes (Scheme 10).49 Detailed studies of the reaction mechanism and isolation of intermediates gave a bimetallic pathway under carbene dissociation (17).72 At 60 °C oxidative coupling takes place resulting in the dimetalla(II) butadienediyl complex 18 as evidenced by isolation of both reaction intermediates. At this point a third alkyne likely coordinates and subsequently inserts into a metal–carbon bond to generate a dimetalla cycloheptatriene that finally undergoes ring closure to produce the aromatic ring. The metal(I) silylamide fragment is either captured by the previously liberated NHC ligand or re-enters into the catalytic cycle by alkyne coordination. In the case of anionic complexes [MI]− (Cr–Co) the interaction with alkynes leads mostly only to η2-alkyne complexes ([M19R′]−, Scheme 11, top).73 For cobalt alkyne coordination is weak and reversible, yet becomes stronger when going to the lighter metals (Co → Cr). CASSCF calculations on these complex series revealed their primary electronic structure as metal(II) radical anions (Scheme 11, bottom), whereas the textbook dichotomy of such compounds as π-complexes or metallacyclopropenes are a lesser factor. One has to bear in mind that the classic description of π-complexes along the Chatt–Dewar–Duncanson concept was originally adopted for closed-shell/low-spin organometallics. Here, the π-backbonding proceeds from a doubly occupied metal centered orbital with no substantial unpaired spin transfer. The observed deviation for the [MI]− complexes shown herein is likely a result of their high-spin state in conjunction with the anionic charge that facilitates the electron transfer to the π*-orbital of the substrate. Steric factors are also in play as for the more encumbered [FeI(N{Dipp}SiMe3)2]− alkyne coordination is labile.43 A similar bond trichotomy is formulated for side-on bound organo nitrile iron complexes ([20R′]−, Scheme 11, top).74 The radical anionic nature of the bound alkyne rationalizes subsequent reduction to bismetallated alkene dianions in the case of manganese ([21]2−) and chromium.73 Moreover, for manganese a series of C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C bond activation processes results in alkyne cyclotrimerisation and the formation of the rare manganese(−I) arene [Mn(η6-C6Ph6)(η2-PhCCPh)].73
C bond activation processes results in alkyne cyclotrimerisation and the formation of the rare manganese(−I) arene [Mn(η6-C6Ph6)(η2-PhCCPh)].73
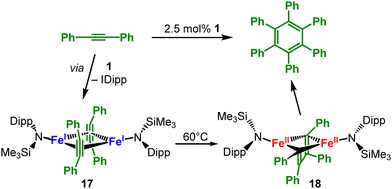 | ||
| Scheme 10 [(IPr)Fe(N{SiMe3}Dipp)], 1, as a precatalyst for cyclotrimerisation of terminal and internal alkynes, and isolable reaction intermediates 17 and 18. | ||
 | ||
| Scheme 11 General reaction of alkynes with linear metal(I) silylamides (top) and electronic resonance structures obtained from CASSCF calculations. | ||
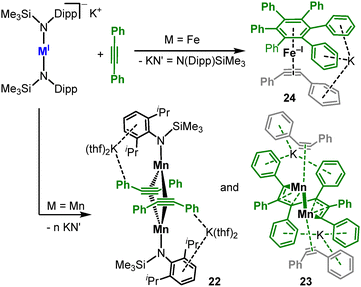
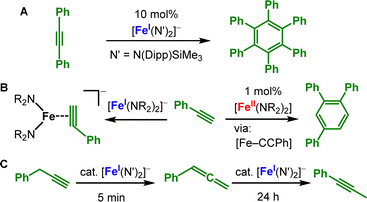
Complex dismutation is more pronounced for the overall neutral, more encumbered [KM(N{Dipp}SiMe3)2] (M = Fe, Mn) where the alkali metal cation is not separated from the complex (Scheme 12).43 [KMn(N{Dipp}SiMe3)2] undergoes redox disproportionation, formation of a dinuclear manganese(0) complex (20) and incomplete alkyne cyclotrimerisation (21). For iron the complete cyclotrimerisation by isolation of the arene iron(−I) complex [K(Fe(C6Ph6)(PhCCPh))], 19, is observed. Together with the intermediates 17 and 18 by Tilley (Scheme 10), 20 and 21 provide snapshots into the cyclotrimerisation steps which seems dinuclear in nature for these low-coordinate systems. For the anionic metal(I) complexes only K{m}[Fe(N{Dipp}SiMe3)2]− proved competent in cyclotrimerization of diphenyl acetylene (Scheme 13A), likely by in situ complex disproportionation. The reaction of terminal alkynes with [Fe(NR2)2]− resulted not in the cyclotrimerisation, but simple side-one coordination.75 Contrastingly, the neutral [Fe(NR2)2] facilitates the selective cyclotrimerisation of terminal alkynes to the 1,2,4-substituted benzene regioisomers (Scheme 13B).76 However, the reaction presumably occurs under ligand exchange via deprotonation of the alkyne by the silylamides, and likely the formation of multinuclear iron alkinyl species. In the special case of terminal benzyl alkynes [Fe(NR2)2]− catalyses its transformation to phenyl allene within minutes (Scheme 13C)75 and to the internal alkyne within 24 hours. For the interaction of alkenes with divalent metals reports are restricted to intramolecular interactions with a C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond as in [M(N{Dipp}Si{Me2}C3H5)2] (Scheme 14, top).37 The interaction is purely electrostatic with marginal C
C double bond as in [M(N{Dipp}Si{Me2}C3H5)2] (Scheme 14, top).37 The interaction is purely electrostatic with marginal C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond elongation. It changes upon reduction as exemplified for cobalt: shortening of the Co–(C
C bond elongation. It changes upon reduction as exemplified for cobalt: shortening of the Co–(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C) distance is observed (Δ = −0.4 Å) with a widening of C
C) distance is observed (Δ = −0.4 Å) with a widening of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C-bond (Δ = 0.03 Å). Akin to the situation for metal alkynes (Scheme 11), the stronger metal–alkene interaction is likely attributed to the situation as predominantly a metal(II) alkene characteristic. It becomes more obvious for chromium where intermolecular C–C-coupling via one of the alkene functions is observed.37 In the reaction of the iron(I) silylamide [Fe(NR2)2]− with external substrates, that bear an internal C
C-bond (Δ = 0.03 Å). Akin to the situation for metal alkynes (Scheme 11), the stronger metal–alkene interaction is likely attributed to the situation as predominantly a metal(II) alkene characteristic. It becomes more obvious for chromium where intermolecular C–C-coupling via one of the alkene functions is observed.37 In the reaction of the iron(I) silylamide [Fe(NR2)2]− with external substrates, that bear an internal C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond, side-on complexes are formed ([25R′]− and [26R′]−, Scheme 14, bottom).75,77 Interestingly, Z-substituted substrates undergo rapid isomerisation to the thermodynamically more stable E-isomer. This observation gives evidence for the metal(I) alkenes as actual iron(II) bound radical anions (Scheme 11, bottom), which was supported by 57Fe Mössbauer spectroscopy analysis.70 The isomerisation can be performed in a catalytic fashion with either aromatic or aliphatic substituted simple alkenes such as 3-hexene. By comparison with the behaviour of the prototypical E-stilbene radical anion,77 whose first isolation was achieved by encapsulation between two K{18c6}+ moieties (27), it was concluded that the isomerisation takes place in the coordination sphere of the metal and not by intermittent decoordination of alkene substrates as “free” radical anions.
C bond, side-on complexes are formed ([25R′]− and [26R′]−, Scheme 14, bottom).75,77 Interestingly, Z-substituted substrates undergo rapid isomerisation to the thermodynamically more stable E-isomer. This observation gives evidence for the metal(I) alkenes as actual iron(II) bound radical anions (Scheme 11, bottom), which was supported by 57Fe Mössbauer spectroscopy analysis.70 The isomerisation can be performed in a catalytic fashion with either aromatic or aliphatic substituted simple alkenes such as 3-hexene. By comparison with the behaviour of the prototypical E-stilbene radical anion,77 whose first isolation was achieved by encapsulation between two K{18c6}+ moieties (27), it was concluded that the isomerisation takes place in the coordination sphere of the metal and not by intermittent decoordination of alkene substrates as “free” radical anions.
Radical anions stabilized by linear complexes. The pronounced reduction characteristics of the anionic metal(I) complexes were further exploited to generate other, electronically more defined metal(II)-bound radical anions. In this context we have reported the formation of metal(II) stabilized bipyridyl radical anions [M28]− (M = Cr–Co) starting from 2,2′-bipyridine (Scheme 15, top).78
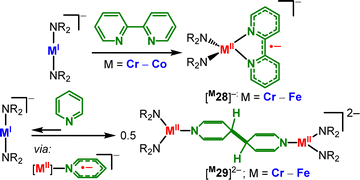 | ||
| Scheme 15 Reaction of 2,2′-bipyridine with (top) and reductive coupling of pyridine by (bottom) linear metal(I) complexes. | ||
The reduction is remarkable in the case of cobalt where the reduction potential of the metal(I) precursor is 1 V less negative than that of free bipyridine (Ered([Co(NR2)2]−) = −1.47 V vs. [Fc/Fc+], E1/2(bipyridine): approx. −2.67). With pyridine, the formation of binuclear metal(II) complexes [M29]2− (M = Cr–Fe, Scheme 15, bottom) occured.79 Here, pyridine underwent reversible reductive C–C coupling forming a bridging 4,4′-dihydrobipyridyl ligand via a transient monomeric metal(II) complex bearing a pyridyl radical anion. Benzophenone was also used in combination with linear anionic complexes of iron and cobalt to produce metal(II) stabilized ketyl radical anions ([M30]−, Scheme 16), supported by Mössbauer spectroscopic and computational analysis. This approach was extended to unprecedented metal bound ketiminyl ([M31]−) and aldiminyl radical anions ([32]−),80 with the reductive coordination being in part found to be reversible. Finally, [FeI]− can act as an Et2O-soluble one-electron reductant in the isolation of the first diarsene radical anion [Mes*As=AsMes*]− ([33]−).81
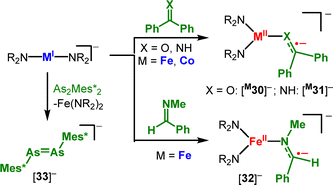 | ||
| Scheme 16 Reduction of diaryl ketones, ketimines, aldimines and a diarsene with [M(NR2)2]− complexes. (Mes* = 2,6-Mes2-C6H3). | ||
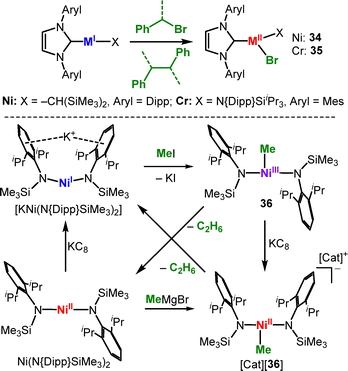
C–Halide bond cleavage. 3d-Metal mediated cross-coupling (e.g. with iron) plays an important role in organic coupling reactions, with recent mechanistic insights having revealed the importance of low-valent, low-coordinate species.54,82 In this context Hillhouse showed for the linear nickel(I) complex (IDipp)Ni(CH{SiMe3}2) radical homocoupling of a benzyl bromide derivative and formation of the trigonal nickel(II) bromide 34 (Scheme 17, top). A similar reactivity was observed in the case of an analogous linear chromium(I) complex to yield the chromium halide 35.56 The nickel(I) complex [KNi(N{Dipp}SiMe3)2] exhibits different reactivity when combined with methyl iodide (Scheme 17, bottom). It yields the rare Ni(III) alkyl complex [Ni(Me)(N{SiMe3}Dipp)2] (36) via a two-electron oxidative addition, which is unusual for open-shell 3d metal ions.3836 decomposes at room temperature over the course of 24 hours to Ni(N{Dipp}SiMe3)2 and ethane, however does not engage in cross-coupling reactions. In contrast Ni(N{SiMe3}Dipp)2 mediates the coupling between aryl chlorides/bromides and Grignard reagents at ambient conditions.83 Mechanistic studies revealed the involvement of radical intermediates as well as the presence of trigonal organo nickel(II/III) intermediates ([36]− and [36]−). [FeI]− reacts with organo halides instantaneously even below −80 °C (Scheme 18),84 which yields stoichiometric mixtures of the organo iron(II) ([37R]−) and iron(II) halide ([37X]−) complexes under involvement of organic radicals. Bond cleavage is also feasible for aromatic and even aliphatic C–F bonds, however at ambient or elevated temperatures and longer reaction times. For an excess of benzyl halides, selective formation of the iron(II) halide ([37X]−) and subsequently the iron(III) dihalide ([38X]−) is observed together with the organic homocoupling products. In a similar fashion aliphatic isonitriles can be cleaved to yield 1
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixtures of alkyl iron(II) and cyanido iron(II) complexes (Scheme 18).74 The capability of linear metal(I) to activate aromatic C–F bonds was further explored for fluoropyridines (Scheme 19).
1 mixtures of alkyl iron(II) and cyanido iron(II) complexes (Scheme 18).74 The capability of linear metal(I) to activate aromatic C–F bonds was further explored for fluoropyridines (Scheme 19).
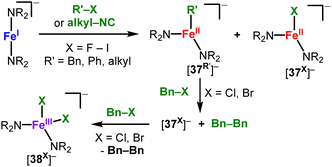 | ||
| Scheme 18 Reaction of [FeI(NR2)2]− (R = SiMe3) with benzyl, phenyl and alkyl halides as well as aliphatic isonitriles. | ||
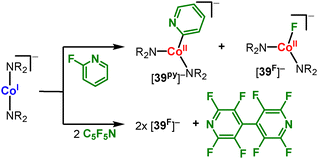
Depending on the degree of fluorination of the pyridine its reaction with [CoI]− yields either a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of cobalt(II) pyridyl ([39py]−) and cobalt(II) fluoride ([39F]−) complexes, or homocoupling to fluorinated 4,4′-bipyridines.79 The selective formation of metal(II) fluorides is observed as well in the case of [MnI]− and [FeI]− using perfluoropyridine.79
1 mixture of cobalt(II) pyridyl ([39py]−) and cobalt(II) fluoride ([39F]−) complexes, or homocoupling to fluorinated 4,4′-bipyridines.79 The selective formation of metal(II) fluorides is observed as well in the case of [MnI]− and [FeI]− using perfluoropyridine.79
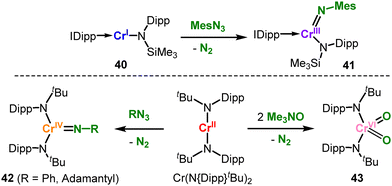
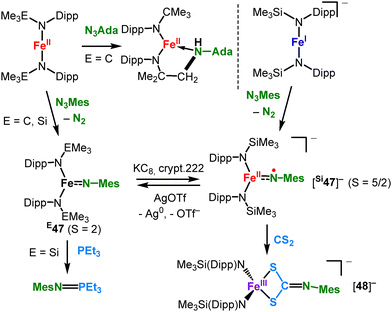
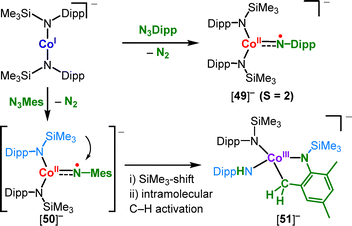
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NDipp] imide unit (Scheme 25) that originated from a SiMe3 shift from a co-ligand, as seen for cobalt ([50]−, Scheme 23).
NDipp] imide unit (Scheme 25) that originated from a SiMe3 shift from a co-ligand, as seen for cobalt ([50]−, Scheme 23).
 | ||
| Scheme 21 Intramolecular C–H amination catalysed by FeII(NR2)2 (top) and detection of a trigonal imido iron complex by irradiation of an iron(II) organo azide adduct (bottom). | ||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
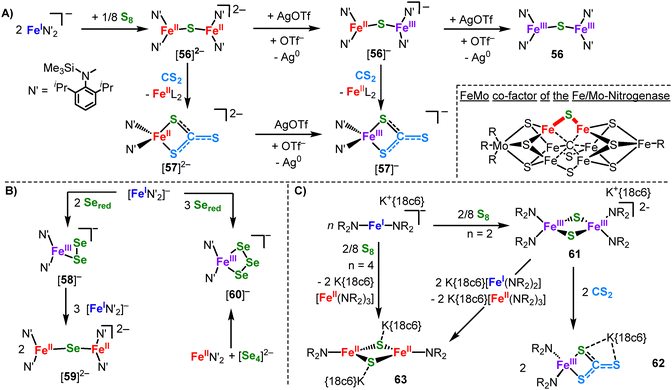
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Se ratio resulted in the mononuclear iron(III) di- (582−) and triselenides (602−). It is contemplated that elemental selenium is initially reduced by [Fe(N{Dipp}SiMe3)2]− to anionic polyselenides. Subsequently, the chalcogenide anions recombine with the now Lewis-acidic iron(II) complexes – with or without further oxidation to iron(III) – to yield monomeric iron polyselenides or dimeric diiron monoselenides depending on the Fe
Se ratio resulted in the mononuclear iron(III) di- (582−) and triselenides (602−). It is contemplated that elemental selenium is initially reduced by [Fe(N{Dipp}SiMe3)2]− to anionic polyselenides. Subsequently, the chalcogenide anions recombine with the now Lewis-acidic iron(II) complexes – with or without further oxidation to iron(III) – to yield monomeric iron polyselenides or dimeric diiron monoselenides depending on the Fe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Se ratio. Accordingly, pre-reduced polychalcogenides can be used like-wise in conjunction with divalent complexes (Scheme 26B).98 If the steric demand of the silylamide ligands of the employed iron(I) complex is lowered, a bis-μ-S bridged complex with an [Fe2S2] diamond core was obtained (61, Scheme 26C).99 It corresponds to the central [Fe2S2] motif of well-known Rieske-type clusters. 61 could be deconstructed using CS2 to yield the mononuclear iron(III) thiocarbonate 62. Reaction of 61 with additional [Fe(NR2)2]− lead to reduction of the iron sulfur complex and was accompanied by decoordination of one silyl amide ligand per iron (63). Two other synthetic super-reduced [Fe2S2]0 clusters were so far known, which however retained the four-coordinate coordination environment due to the use of chelating ligands.10063 thus represents the first example of a super-reduced [Fe2S2]0 complex with only three-coordinate iron. It is important to note that formation of 63 does not occur using the K{crypt.222} salt of [FeI]− or KC8 in the presence of 18-crown-6. It reveals a dual role of the employed [Fe(NR2)2]0,−: it acts first as a 1e−–reductant in the oxidation state +I and then as a strong Lewis acid in the divalent state. The approach of combining a linear metal(II) fragment with unusual polyanions is also applicable for Zintl anions (Scheme 27).101 Reaction of Mn(NR2)2 with K{crypt.222}[TlBi3], which is only soluble in very polar solvents such as ethylene diamine or NH3(l), allowed for the unprecedented reactive dissolution of Zintl ions in regular solvents such as THF. The integrity of the Zintl ion remains in formed [64]2−, which is also rather uncommon. This was emphasized for [TrBi3]2− (Tr = Ga, In), where a [Bi2]2− bridged dinuclear complex ([65]2−) is observed with concomittant101 elimination of the triel metal. Moreover, the reaction with [TrBi3]2− also yielded additional ligand exchange and redox state changes of the Mn(NR2)2 fragment, as shown for the tetranuclear cluster [66]2−. [66]2− is a mixed [MnI2MnII2] complex, thus underscoring the ease of redox state changes in these low-coordinate high-spin metal ions.
Se ratio. Accordingly, pre-reduced polychalcogenides can be used like-wise in conjunction with divalent complexes (Scheme 26B).98 If the steric demand of the silylamide ligands of the employed iron(I) complex is lowered, a bis-μ-S bridged complex with an [Fe2S2] diamond core was obtained (61, Scheme 26C).99 It corresponds to the central [Fe2S2] motif of well-known Rieske-type clusters. 61 could be deconstructed using CS2 to yield the mononuclear iron(III) thiocarbonate 62. Reaction of 61 with additional [Fe(NR2)2]− lead to reduction of the iron sulfur complex and was accompanied by decoordination of one silyl amide ligand per iron (63). Two other synthetic super-reduced [Fe2S2]0 clusters were so far known, which however retained the four-coordinate coordination environment due to the use of chelating ligands.10063 thus represents the first example of a super-reduced [Fe2S2]0 complex with only three-coordinate iron. It is important to note that formation of 63 does not occur using the K{crypt.222} salt of [FeI]− or KC8 in the presence of 18-crown-6. It reveals a dual role of the employed [Fe(NR2)2]0,−: it acts first as a 1e−–reductant in the oxidation state +I and then as a strong Lewis acid in the divalent state. The approach of combining a linear metal(II) fragment with unusual polyanions is also applicable for Zintl anions (Scheme 27).101 Reaction of Mn(NR2)2 with K{crypt.222}[TlBi3], which is only soluble in very polar solvents such as ethylene diamine or NH3(l), allowed for the unprecedented reactive dissolution of Zintl ions in regular solvents such as THF. The integrity of the Zintl ion remains in formed [64]2−, which is also rather uncommon. This was emphasized for [TrBi3]2− (Tr = Ga, In), where a [Bi2]2− bridged dinuclear complex ([65]2−) is observed with concomittant101 elimination of the triel metal. Moreover, the reaction with [TrBi3]2− also yielded additional ligand exchange and redox state changes of the Mn(NR2)2 fragment, as shown for the tetranuclear cluster [66]2−. [66]2− is a mixed [MnI2MnII2] complex, thus underscoring the ease of redox state changes in these low-coordinate high-spin metal ions.
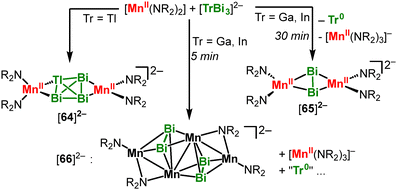 | ||
| Scheme 27 Reaction of Mn(NR2)2 with tetraatomic trielbismuthates [TrBi3]2− (Tr = In, Ga, Ta). K{crypt.222} cations are omitted. | ||
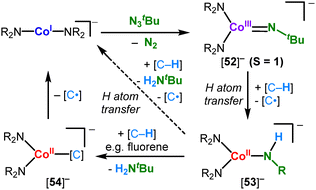
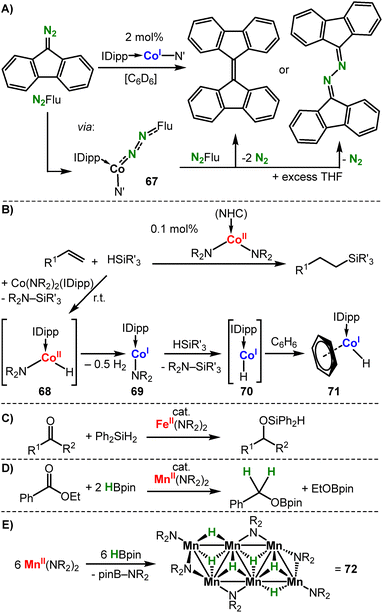
Other reactivities involving linear metal amides. The cobalt(I) complex (IPr)Co(N{SiMe3}Dipp) reacts with diazoalkane to give the respective adduct 67, which is formulated as a cobalt(II) diazoalkane radical anion (Scheme 28A).50
In the presence of an excess of the diazoalkane the catalytic homocoupling to an alkene under N2 liberation is observed in non-polar solvents. The reactivity switches to hydrazine formation when donor solvents are employed. This is attributed to blocking of the metal site by the solvent and by that prevents complete metal-mediated N2 liberation.50 Metal(II) hexamethyldisilazanides have also been used in a number of hydro-elementation reactions using silanes or boranes (Scheme 28B–D). These transformations rely on the loss of complex integrity via amide/hydride exchange and redox-processes: the cobalt(II) amide Co(NR2)2 or their NHC adduct (Scheme 28B) catalyses the hydrosilylation of a variety of terminal alkenes with tertiary silanes.102 Stoichiometric studies on NHC adducts revealed reduction to the linear cobalt(I) amide 69via presumed hydride formation (68) and subsequent reductive H2 liberation. The linear hydride 70, as inferred from the isolable arene complex 71 (or the silyl derivative), are likely the active alkene hydrosilylation agents. The neutral iron and manganese complexes MII(NR2)2 were found to be active in the hydrosilylation or hydroboration of carbonyl compounds at room temperature (Scheme 28C and D).103 Under catalytic conditions MII(NR2)2 serves as a precatalyst and initially forms mono- or polymeric metal hydrides.104,105 The latter was evidenced exemplarily for Mn(NR2)2, that reacted with HAliPr2 to form the hexameric, sheet-like [MnII6H6(NR2)6] (72, Scheme 28E).105
Conclusion
For decades, linear open-shell 3d-metal complexes have been a curiosity and synthetic challenge in coordination chemistry. However, in recent years their potential as single ion magnets, as reaction partners and even as (pre)catalysts, has been developed. Of these, silylamide based complexes have gained prominence, either in the divalent or monovalent state. Divalent metal silylamides can serve most notably as platforms for imido metal species. They exhibit intriguing electronic structures of mixed metal(III) imidyl and metal(II) nitrene characteristics, which were further exploited for catalytic C–H amination catalysis. In this context, the pronounced Lewis acidity of M(NR2)2 helped in the isolation of weakly bound organoazide adducts as the initial species in the catalytic cycle. These adducts can be irradiated in-crystallo under N2 liberation and metal imido formation, which were otherwise too reactive in solution chemistry. Looking also to other linear, mainly aryl-based metal(II) complexes, they served as precatalysts for a variety of transformations. However, the employed compounds are subject to intricate transformation reactions that can involve substrate insertion into the original metal–ligand bonds as well as substantial redox state changes.The chemistry of the monovalent siblings, either anionic and homoleptic or neutral and heteroleptic with NHC ligation, exhibits a larger reactivity variety due to a more electron-rich metal centre. The most salient features of linear metal(I) complexes are a pronounced loss of Lewis acidity despite the coordinatively unsaturated state. Furthermore, the interaction of these compounds with substrates is characterized by one-electron transfer processes. This property is most pronounced for anionic complexes ([M(NR2)2]−) and was exploited for the generation of a number of metal(II) stabilized radical anions. It was exemplified for carbonyl or iminyl based substrates and could also be extended to alkynes and alkenes. Complexes with the latter could undergo radical-anion based bond transformations, such as cis → trans isomerisation. Here, certain limits of linear metal(I) based chemistry with π-accepting ligands also became evident as the interaction with C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C triple bonds can lead to complex dismutation driven by the formation of comparably stable 18 VE organometal complexes. Like for the neutral metal(II) species, anionic and neutral linear metal(I) complexes are suitable platforms for imido metal complexes in higher spin states using organo azides as transfer agents. This allows for catalytic applications, with evidence that the employed metal silylamide fragments remains intact. For similar diazo methane derivatives, they can be catalytically coupled by a linear cobalt(I) complex under N2 extrusion to give either alkenes or hydrazine derivatives.
C triple bonds can lead to complex dismutation driven by the formation of comparably stable 18 VE organometal complexes. Like for the neutral metal(II) species, anionic and neutral linear metal(I) complexes are suitable platforms for imido metal complexes in higher spin states using organo azides as transfer agents. This allows for catalytic applications, with evidence that the employed metal silylamide fragments remains intact. For similar diazo methane derivatives, they can be catalytically coupled by a linear cobalt(I) complex under N2 extrusion to give either alkenes or hydrazine derivatives.
A very different area is the bottom-up construction of small metal/main-group clusters, in which the dual role of metal silylamides is emphasized. Anionic iron(I) silylamides can activate elemental chalcogenes, that likely proceeds via initial single-electron-transfer from the metal to yield chalcogenide (poly)anions. These immediately recombine with the now Lewis acidic iron(II) silylamides to unusual metal sulfide or selenide motifs. Accordingly, neutral metal(II) silylamides can directly be combined with main-group polyanions (e.g. zintl ions). In all cases, further redox state changes can be observed, yielding highly unusual clusters with low-coordinate metal ions.
Outlook
Despite the presented knowledge for linear metal silylamides, it is evident that for this compound class alone a multitude of combinations with already examined substrates are still open for discovery. Possible variations concern for example other main-group element/metal combinations or advancing the field of catalytically relevant imido metal species. Furthermore, the flurry of intriguing organic transfer-agents in recent years, such as diazoolefins (NHC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N2)114 or the carbon atom transfer agent Ph2S
N2)114 or the carbon atom transfer agent Ph2S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N2,115 might pose unchartered territory with regards to metal-main group multiple bonds in higher spin state.
N2,115 might pose unchartered territory with regards to metal-main group multiple bonds in higher spin state.
Looking beyond, the introduction of NHC ligation further opened up the variability in terms of charge and gave access to formally zero- or even subvalent compounds. Here, the non-innocence of alkyl amino carbene ligands is prospective to facilitate multi-electron bond activation processes. These NHC complexes were so far only little examined in a broader fashion, which would be needed to address the integrity and stability of the employed two-coordinate [M(NHC)2]n fragments during bond activation processes. In this context, the NHC based metallates, namely [Fe(cAAC)2(N2)]− or the higher coordinate [Co(IMes)2(N2)]−![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 116 with their labile N2 ligation, are possible candidates. These can serve as M−I synthons, similar to arene metallates such as [Fe(anthracen)2]−,117 yet with more persistent ancillary ligands. The anionic charge might prevent unwanted decomposition or aggregation reactions, enhance the likelihood of higher spin states and provide beneficial crystallisation properties as seen for anionic metal(I) silylamides. In conclusion, the chemistry of linear open shell complexes still leaves a wide open chemical space. Its further exploration is promising for uncovering new and unusual reactivity patterns, which we and others will continue to address.
116 with their labile N2 ligation, are possible candidates. These can serve as M−I synthons, similar to arene metallates such as [Fe(anthracen)2]−,117 yet with more persistent ancillary ligands. The anionic charge might prevent unwanted decomposition or aggregation reactions, enhance the likelihood of higher spin states and provide beneficial crystallisation properties as seen for anionic metal(I) silylamides. In conclusion, the chemistry of linear open shell complexes still leaves a wide open chemical space. Its further exploration is promising for uncovering new and unusual reactivity patterns, which we and others will continue to address.
Author contributions
A. C. and C. G. W. conceptionalised and wrote the manuscript.Conflicts of interest
There are no conflicts to declare.Acknowledgements
C. G. W. acknowledges the Deutsche Forschungsgemeinschaft (grants WE 5627/7-1 and WE 5627/8-1).Notes and references
- (a) M. F. Lappert, Metal amide chemistry, Wiley, Chichester, UK, 2009 Search PubMed; (b) Metal and metalloid amides: syntheses, structures, and physical and chemical properties, Chichester, 1979 Search PubMed; (c) P. P. Power, Comment. Inorg. Chem., 1989, 8, 177–202 CrossRef CAS; (d) P. P. Power, Chemtracts, 1994, 6, 181–194 CAS; (e) D. L. Kays, Dalton Trans., 2011, 40, 769–778 RSC; (f) A. Noor, Coord. Chem. Rev., 2023, 476, 214941 CrossRef CAS; (g) L. J. Taylor and D. L. Kays, Dalton Trans., 2019, 48, 12365–12381 RSC.
- P. P. Power, Chem. Rev., 2012, 112, 3482–3507 CrossRef CAS PubMed.
- H. Bürger and U. Wannagat, Monatsh. Chem., 1963, 94, 1007–1012 CrossRef.
- H. Bürger and U. Wannagat, Monatsh. Chem., 1964, 95, 1099–1102 CrossRef.
- (a) D. C. Bradley, M. B. Hursthouse, K. M. Abdul Malik and R. Möseler, Transition Met. Chem., 1978, 3, 253–254 CrossRef CAS; (b) B. D. Murray and P. P. Power, Inorg. Chem., 1984, 23, 4584–4588 CrossRef CAS; (c) R. A. Andersen, K. Faegri, J. C. Green, A. Haaland, M. F. Lappert, W. P. Leung and K. Rypdal, Inorg. Chem., 1988, 27, 1782–1786 CrossRef CAS.
- M. M. Olmstead, P. P. Power and S. C. Shoner, Inorg. Chem., 1991, 30, 2547–2551 CrossRef CAS.
- A. M. Bryan, G. J. Long, F. Grandjean and P. P. Power, Inorg. Chem., 2013, 52, 12152–12160 CrossRef CAS PubMed.
- D. C. Bradley, M. B. Hursthouse, C. W. Newing and A. J. Welch, Chem. Commun., 1972, 567 RSC.
- D. C. Bradley, M. B. Hursthouse, A. A. Ibrahim, K. Malik, M. Motevalli, R. Möseler, H. Powell, J. D. Runnacles and A. C. Sullivan, Polyhedron, 1990, 9, 2959–2964 CrossRef CAS.
- M. Faust, A. M. Bryan, A. Mansikkamäki, P. Vasko, M. M. Olmstead, H. M. Tuononen, F. Grandjean, G. J. Long and P. P. Power, Angew. Chem., Int. Ed., 2015, 54, 12914–12917 CrossRef CAS PubMed.
- (a) D. L. J. Broere, I. Čorić, A. Brosnahan and P. L. Holland, Inorg. Chem., 2017, 56, 3140–3143 CrossRef CAS PubMed; (b) R. Weller, A. Gonzalez, H. Gottschling, C. von Hänisch and C. G. Werncke, Z. Anorg. Allg. Chem., 2022, 648, e202100338 CrossRef CAS.
- A. M. Borys and E. Hevia, Organometallics, 2021, 40, 442–447 CrossRef CAS.
- A. Reckziegel, B. Battistella, A. Schmidt and C. G. Werncke, Inorg. Chem., 2022, 61, 7794–7803 CrossRef CAS PubMed.
- P. P. Power, Inorganic Syntheses, ed. P. P. Power, John Wiley & Sons, 2018, vol. 37, pp. 1–14 Search PubMed.
- R. A. Andersen, A. Haaland, K. Rypdal and H. V. Volden, J. Chem. Soc., Chem. Commun., 1985, 1807 RSC.
- N. H. Buttrus, C. Eaborn, P. B. Hitchcock, J. D. Smith and A. C. Sullivan, J. Chem. Soc., Chem. Commun., 1985, 1380 RSC.
- (a) W. Seidel, H. Müller and H. Görls, Angew. Chem., Int. Ed. Engl., 1995, 34, 325–327 CrossRef; (b) R. J. Wehmschulte and P. P. Power, Organometallics, 1995, 14, 3264–3267 CrossRef CAS; (c) C. Ni, T. A. Stich, G. J. Long and P. P. Power, Chem. Commun., 2010, 46, 4466 RSC.
- (a) C. Ni and P. P. Power, Chem. Commun., 2009, 5543–5545 RSC; (b) J. J. Ellison, K. Ruhlandt-Senge and P. P. Power, Angew. Chem., Int. Ed. Engl., 1994, 33, 1178–1180 CrossRef; (c) T. Nguyen, A. D. Sutton, M. Brynda, J. C. Fettinger, G. J. Long and P. P. Power, Science, 2005, 310, 844–847 CrossRef CAS PubMed; (d) A. M. Bryan, G. J. Long, F. Grandjean and P. P. Power, Inorg. Chem., 2014, 53, 2692–2698 CrossRef CAS PubMed.
- (a) J. N. Boynton, J.-D. Guo, J. C. Fettinger, C. E. Melton, S. Nagase and P. P. Power, J. Am. Chem. Soc., 2013, 135, 10720–10728 CrossRef CAS PubMed; (b) J. Li, H. Song, C. Cui and J.-P. Cheng, Inorg. Chem., 2008, 47, 3468–3470 CrossRef CAS PubMed; (c) A. M. Bryan, W. A. Merrill, W. M. Reiff, J. C. Fettinger and P. P. Power, Inorg. Chem., 2012, 51, 3366–3373 CrossRef CAS PubMed.
- W. Alexander Merrill, T. A. Stich, M. Brynda, G. J. Yeagle, J. C. Fettinger, R. D. Hont, W. M. Reiff, C. E. Schulz, R. D. Britt and P. P. Power, J. Am. Chem. Soc., 2009, 131, 12693–12702 CrossRef CAS PubMed.
- J. N. Boynton, W. A. Merrill, W. M. Reiff, J. C. Fettinger and P. P. Power, Inorg. Chem., 2012, 51, 3212–3219 CrossRef CAS PubMed.
- (a) H. Chen, R. A. Bartlett, M. M. Olmstead, P. P. Power and S. C. Shoner, J. Am. Chem. Soc., 1990, 112, 1048–1055 CrossRef CAS; (b) R. A. Bartlett and P. P. Power, J. Am. Chem. Soc., 1987, 109, 7563–7564 CrossRef CAS; (c) S. N. Konig, C. Schadle, C. Maichle-Mossmer and R. Anwander, Inorg. Chem., 2014, 53, 4585–4597 CrossRef PubMed; (d) K. Freitag, C. R. Stennett, A. Mansikkamäki, R. A. Fischer and P. P. Power, Inorg. Chem., 2021, 60, 4108–4115 CrossRef CAS PubMed.
- D. J. Liptrot and P. P. Power, Nat. Rev. Chem., 2017, 1, 0004 CrossRef.
- (a) Y. Liu, N. T. Coles, N. Cajiao, L. J. Taylor, E. S. Davies, A. Barbour, P. J. Morgan, K. Butler, B. Pointer-Gleadhill, S. P. Argent, J. McMaster, M. L. Neidig, D. Robinson and D. L. Kays, Chem. Sci., 2024, 15, 9599–9611 RSC; (b) H. R. Sharpe, A. M. Geer, L. J. Taylor, B. M. Gridley, T. J. Blundell, A. J. Blake, E. S. Davies, W. Lewis, J. McMaster, D. Robinson and D. L. Kays, Nat. Commun., 2018, 9, 3757 CrossRef PubMed.
- S. Roy, K. C. Mondal and H. W. Roesky, Acc. Chem. Res., 2016, 49, 357–369 CrossRef CAS PubMed.
- S. Kumar Kushvaha, A. Mishra, H. W. Roesky and K. Chandra Mondal, Chem. – Asian J., 2022, 17, e202101301 CrossRef CAS PubMed.
- P. P. Samuel, K. C. Mondal, H. W. Roesky, M. Hermann, G. Frenking, S. Demeshko, F. Meyer, A. C. Stückl, J. H. Christian, N. S. Dalal, L. Ungur, L. F. Chibotaru, K. Pröpper, A. Meents and B. Dittrich, Angew. Chem., Int. Ed., 2013, 52, 11817–11821 CrossRef CAS PubMed.
- G. Ung, J. Rittle, M. Soleilhavoup, G. Bertrand and J. C. Peters, Angew. Chem., Int. Ed., 2014, 53, 8427–8431 CrossRef CAS PubMed.
- K. C. Mondal, S. Roy, S. De, P. Parameswaran, B. Dittrich, F. Ehret, W. Kaim and H. W. Roesky, Chem. – Eur. J., 2014, 20, 11646–11649 CrossRef CAS PubMed.
- K. C. Mondal, P. P. Samuel, Y. Li, H. W. Roesky, S. Roy, L. Ackermann, N. S. Sidhu, G. M. Sheldrick, E. Carl, S. Demeshko, S. De, P. Parameswaran, L. Ungur, L. F. Chibotaru and D. M. Andrada, Eur. J. Inorg. Chem., 2014, 818–823 CrossRef CAS.
- (a) P. P. Samuel, R. Neufeld, K. Chandra Mondal, H. W. Roesky, R. Herbst-Irmer, D. Stalke, S. Demeshko, F. Meyer, V. C. Rojisha, S. De, P. Parameswaran, A. C. Stückl, W. Kaim, J. H. Christian, J. K. Bindra and N. S. Dalal, Chem. Sci., 2015, 6, 3148–3153 RSC; (b) R. C. Poulten, M. J. Page, A. G. Algarra, J. J. Le Roy, I. López, E. Carter, A. Llobet, S. A. Macgregor, M. F. Mahon, D. M. Murphy, M. Murugesu and M. K. Whittlesey, J. Am. Chem. Soc., 2013, 135, 13640–13643 CrossRef CAS PubMed.
- C. G. Werncke, E. Suturina, P. C. Bunting, L. Vendier, J. R. Long, M. Atanasov, F. Neese, S. Sabo-Etienne and S. Bontemps, Chem. – Eur. J., 2016, 22, 1668–1674 CrossRef CAS PubMed.
- I. C. Cai, M. I. Lipschutz and T. D. Tilley, Chem. Commun., 2014, 50, 13062–13065 RSC.
- J. M. Zadrozny, M. Atanasov, A. M. Bryan, C.-Y. Lin, B. D. Rekken, P. P. Power, F. Neese and J. R. Long, Chem. Sci., 2013, 4, 125–138 RSC.
- C.-Y. Lin, J.-D. Guo, J. C. Fettinger, S. Nagase, F. Grandjean, G. J. Long, N. F. Chilton and P. P. Power, Inorg. Chem., 2013, 52, 13584–13593 Search PubMed.
- (a) C. L. Wagner, L. Tao, J. C. Fettinger, R. D. Britt and P. P. Power, Inorg. Chem., 2019, 58, 8793–8799 CrossRef CAS PubMed; (b) M. I. Lipschutz and T. D. Tilley, Chem. Commun., 2012, 48, 7146–7148 RSC.
- R. Weller, L. Ruppach, A. Shlyaykher, F. Tambornino and C. G. Werncke, Dalton Trans., 2021, 50, 10947–10963 RSC.
- M. I. Lipschutz, X. Yang, R. Chatterjee and T. D. Tilley, J. Am. Chem. Soc., 2013, 135, 15298–15301 CrossRef CAS PubMed.
- I. C. Cai, M. S. Ziegler, P. C. Bunting, A. Nicolay, D. S. Levine, V. Kalendra, P. W. Smith, K. V. Lakshmi and T. D. Tilley, Organometallics, 2019, 38, 1648–1663 CrossRef CAS.
- P.-C. Yang, K.-P. Yu, C.-T. Hsieh, J. Zou, C.-T. Fang, H.-K. Liu, C.-W. Pao, L. Deng, M.-J. Cheng and C.-Y. Lin, Chem. Sci., 2022, 13, 9637–9643 RSC.
- (a) Y. W. Chao, P. A. Wexler and D. E. Wigley, Inorg. Chem., 1989, 28, 3860–3868 CrossRef CAS; (b) D. K. Kennepohl, S. Brooker, G. M. Sheldrick and H. W. Roesky, Chem. Ber., 1991, 124, 2223–2225 CrossRef CAS; (c) R. J. Schwamm, C. M. Fitchett and M. P. Coles, Chem. – Asian J., 2019, 14, 1204–1211 CrossRef CAS PubMed.
- C. L. Wagner, L. Tao, E. J. Thompson, T. A. Stich, J. Guo, J. C. Fettinger, L. A. Berben, R. D. Britt, S. Nagase and P. P. Power, Angew. Chem., Int. Ed., 2016, 55, 10444–10447 CrossRef CAS PubMed.
- R. Weller, I. Müller, C. Duhayon, S. Sabo-Etienne, S. Bontemps and C. G. Werncke, Dalton Trans., 2021, 50, 4890–4903 RSC.
- R. Weller, M. Atanasov, S. Demeshko, T.-Y. Chen, I. Mohelsky, E. Bill, M. Orlita, F. Meyer, F. Neese and C. G. Werncke, Inorg. Chem., 2023, 62, 3153–3161 CrossRef CAS PubMed.
- (a) N. L. Bell, M. Gladkikh, C. Fraser, M. Elsayed, E. Richards and R. D. Turnbull, Angew. Chem., Int. Ed., 2025, e202505408 CAS; (b) A. Eichhöfer, Y. Lan, V. Mereacre, T. Bodenstein and F. Weigend, Inorg. Chem., 2014, 53, 1962–1974 CrossRef PubMed.
- C. G. Werncke, P. C. Bunting, C. Duhayon, J. R. Long, S. Bontemps and S. Sabo-Etienne, Angew. Chem., Int. Ed., 2015, 54, 245–248 CrossRef CAS PubMed.
- (a) D.-Y. Lu, J.-S. K. Yu, T.-S. Kuo, G.-H. Lee, Y. Wang and Y.-C. Tsai, Angew. Chem., Int. Ed., 2011, 50, 7611–7615 CrossRef CAS PubMed; (b) J. Chai, H. Zhu, A. C. Stückl, H. W. Roesky, J. Magull, A. Bencini, A. Caneschi and D. Gatteschi, J. Am. Chem. Soc., 2005, 127, 9201–9206 CrossRef CAS PubMed.
- A. A. Danopoulos, P. Braunstein, K. Y. Monakhov, J. van Leusen, P. Kögerler, M. Clémancey, J.-M. Latour, A. Benayad, M. Tromp, E. Rezabal and G. Frison, Dalton Trans., 2017, 46, 1163–1171 RSC.
- M. I. Lipschutz, T. Chantarojsiri, Y. Dong and T. D. Tilley, J. Am. Chem. Soc., 2015, 137, 6366–6372 CrossRef CAS PubMed.
- Y. Dong, M. I. Lipschutz, R. J. Witzke, J. A. Panetier and T. D. Tilley, ACS Catal., 2021, 11, 11160–11170 CrossRef CAS.
- R. J. Witzke, D. Hait, M. Head-Gordon and T. D. Tilley, Organometallics, 2021, 40, 1758–1764 CrossRef CAS.
- C. A. Laskowski and G. L. Hillhouse, J. Am. Chem. Soc., 2008, 130, 13846–13847 CrossRef CAS PubMed.
- C. A. Laskowski, G. R. Morello, C. T. Saouma, T. R. Cundari and G. L. Hillhouse, Chem. Sci., 2013, 4, 170–174 RSC.
- T. Inatomi, Y. Fukahori, Y. Yamada, R. Ishikawa, S. Kanegawa, Y. Koga and K. Matsubara, Catal. Sci. Technol., 2019, 9, 1784–1793 RSC.
- M. I. Lipschutz and T. D. Tilley, Organometallics, 2014, 33, 5566–5570 CrossRef CAS PubMed.
- A. Gonzalez, T.-Y. Chen, S. Demeshko, F. Meyer and C. G. Werncke, Organometallics, 2023, 42, 1198–1204 CrossRef CAS.
- K.-C. Hsiao, P.-C. Yang, C.-T. Fang, H.-K. Liu and C.-Y. Lin, Chem. – Asian J., 2024, 19, e202300924 CrossRef CAS PubMed.
- J. M. Zadrozny, D. J. Xiao, M. Atanasov, G. J. Long, F. Grandjean, F. Neese and J. R. Long, Nat. Chem., 2013, 5, 577–581 CrossRef CAS PubMed.
- P. C. Bunting, M. Atanasov, E. Damgaard-Møller, M. Perfetti, I. Crassee, M. Orlita, J. Overgaard, J. van Slageren, F. Neese and J. R. Long, Science, 2018, 362, 7319 CrossRef PubMed.
- M. K. Thomsen, A. Nyvang, J. P. S. Walsh, P. C. Bunting, J. R. Long, F. Neese, M. Atanasov, A. Genoni and J. Overgaard, Inorg. Chem., 2019, 58, 3211–3218 CrossRef CAS PubMed.
- M. Atanasov, J. M. Zadrozny, J. R. Long and F. Neese, Chem. Sci., 2013, 4, 139–156 RSC.
- W. M. Reiff, A. M. LaPointe and E. H. Witten, J. Am. Chem. Soc., 2004, 126, 10206–10207 CrossRef CAS PubMed.
- D. Errulat, K. L. M. Harriman, D. A. Gálico, J. S. Ovens, A. Mansikkamäki and M. Murugesu, Inorg. Chem. Front., 2021, 8, 5076–5085 RSC.
- (a) A. M. Bryan, C.-Y. Lin, M. Sorai, Y. Miyazaki, H. M. Hoyt, A. Hablutzel, A. LaPointe, W. M. Reiff, P. P. Power and C. E. Schulz, Inorg. Chem., 2014, 53, 12100–12107 CrossRef CAS PubMed; (b) X.-N. Yao, J.-Z. Du, Y.-Q. Zhang, X.-B. Leng, M.-W. Yang, S.-D. Jiang, Z.-X. Wang, Z.-W. Ouyang, L. Deng, B.-W. Wang and S. Gao, J. Am. Chem. Soc., 2017, 139, 373–380 CrossRef CAS PubMed.
- J. M. Frost, K. L. M. Harriman and M. Murugesu, Chem. Sci., 2016, 7, 2470–2491 RSC.
- J. M. Zadrozny, D. J. Xiao, J. R. Long, M. Atanasov, F. Neese, F. Grandjean and G. J. Long, Inorg. Chem., 2013, 52, 13123–13131 Search PubMed.
- P. P. Samuel, K. C. Mondal, N. Amin Sk, H. W. Roesky, E. Carl, R. Neufeld, D. Stalke, S. Demeshko, F. Meyer, L. Ungur, L. F. Chibotaru, J. Christian, V. Ramachandran, J. van Tol and N. S. Dalal, J. Am. Chem. Soc., 2014, 136, 11964–11971 CrossRef CAS PubMed.
- Y.-S. Meng, Z. Mo, B.-W. Wang, Y.-Q. Zhang, L. Deng and S. Gao, Chem. Sci., 2015, 6, 7156–7162 Search PubMed.
- Z. Ouyang, J. Du, L. Wang, J. L. Kneebone, M. L. Neidig and L. Deng, Inorg. Chem., 2015, 54, 8808–8816 CrossRef CAS PubMed.
- C. G. Werncke and I. Müller, Chem. Commun., 2020, 56, 2268–2271 RSC.
- C. Schneider, L. Guggolz and C. G. Werncke, Dalton Trans., 2021, 51, 179–184 RSC.
- R. J. Witzke, D. Hait, K. Chakarawet, M. Head-Gordon and T. D. Tilley, ACS Catal., 2020, 10, 7800–7807 CrossRef CAS.
- I. Müller, D. Munz and C. G. Werncke, Inorg. Chem., 2020, 59, 9521–9537 CrossRef PubMed.
- C. G. Werncke, I. Müller, K. Weißer and C. Limberg, Inorg. Chem., 2024, 63, 15236–15246 CrossRef CAS PubMed.
- R. Weller, I. Müller and G. Werncke, Eur. J. Inorg. Chem., 2022, e202100955 CrossRef CAS.
- D. Brenna, M. Villa, T. N. Gieshoff, F. Fischer, M. Hapke and A. Jacobi von Wangelin, Angew. Chem., Int. Ed., 2017, 56, 8451–8454 CrossRef CAS PubMed.
- G. Sieg, I. Müller, K. Weißer and G. Werncke, Chem. Sci., 2022, 13, 13872 RSC.
- I. Müller, C. Schneider, C. Pietzonka, F. Kraus and C. G. Werncke, Inorganics, 2019, 7, 117 CrossRef.
- I. Müller and C. G. Werncke, Chem. – Eur. J., 2021, 27, 4932–4938 CrossRef PubMed.
- G. Sieg, Q. Pessemesse, S. Reith, S. Yelin, C. Limberg, D. Munz and C. G. Werncke, Chem. – Eur. J., 2021, 27, 16760–16767 CrossRef CAS PubMed.
- G. Sieg, M. Fischer, F. Dankert, J.-E. Siewert, C. Hering-Junghans and C. G. Werncke, Chem. Commun., 2022, 58, 9786–9789 RSC.
- (a) S. Sandl and A. Jacobi von Wangelin, Angew. Chem., Int. Ed., 2020, 59, 5434–5437 CrossRef CAS PubMed; (b) J. D. Sears, P. G. N. Neate and M. L. Neidig, J. Am. Chem. Soc., 2018, 140, 11872–11883 CrossRef CAS PubMed; (c) C.-Y. Lin and P. P. Power, Chem. Soc. Rev., 2017, 46, 5347–5399 RSC.
- M. I. Lipschutz and T. D. Tilley, Angew. Chem., Int. Ed., 2014, 53, 7290–7294 CrossRef CAS PubMed.
- C. G. Werncke, J. Pfeiffer, I. Müller, L. Vendier, S. Sabo-Etienne and S. Bontemps, Dalton Trans., 2019, 48, 1757–1765 RSC.
- (a) P. Müller and C. Fruit, Chem. Rev., 2003, 103, 2905–2920 CrossRef PubMed; (b) L. Zhang and L. Deng, Chin. Sci. Bull., 2012, 57, 2352–2360 CrossRef CAS; (c) Y. Park, Y. Kim and S. Chang, Chem. Rev., 2017, 117, 9247–9301 CrossRef CAS PubMed; (d) Y. Liu, T. You, H.-X. Wang, Z. Tang, C.-Y. Zhou and C.-M. Che, Chem. Soc. Rev., 2020, 49, 5310–5358 RSC; (e) K. Kawakita, Y. Kakiuchi, H. Tsurugi, K. Mashima, B. F. Parker, J. Arnold and I. A. Tonks, Coord. Chem. Rev., 2020, 407, 213118 Search PubMed.
- (a) P. F. Kuijpers, J. I. van der Vlugt, S. Schneider and B. de Bruin, Chem. – Eur. J., 2017, 23, 13819–13829 CrossRef CAS PubMed; (b) K. Ray, F. Heims and F. F. Pfaff, Eur. J. Inorg. Chem., 2013, 3784–3807 CrossRef CAS; (c) A. Grünwald, S. S. Anjana and D. Munz, Eur. J. Inorg. Chem., 2021, 4147–4166 CrossRef.
- W. Stroek and M. Albrecht, Chem. Sci., 2023, 14, 2849–2859 RSC.
- W. Stroek, M. Keilwerth, L. A. Malaspina, S. Grabowsky, K. Meyer and M. Albrecht, Chem. – Eur. J., 2024, 30, e202303410 CrossRef CAS PubMed.
- A. Gonzalez, S. Demeshko, F. Meyer and C. G. Werncke, Chem. Commun., 2023, 59, 11532–11535 RSC.
- S. Reith, S. Demeshko, B. Battistella, A. Reckziegel, C. Schneider, A. Stoy, C. Lichtenberg, F. Meyer, D. Munz and C. G. Werncke, Chem. Sci., 2022, 13, 7907–7913 RSC.
- A. Reckziegel, M. Kour, B. Battistella, S. Mebs, K. Beuthert, R. Berger and C. G. Werncke, Angew. Chem., Int. Ed., 2021, 60, 15376–15380 CrossRef CAS PubMed.
- A. Reckziegel, C. Pietzonka, F. Kraus and C. G. Werncke, Angew. Chem., Int. Ed., 2020, 59, 8527–8531 CrossRef CAS PubMed.
- A. Reckziegel, B. Battistella and C. G. Werncke, Eur. J. Inorg. Chem., 2022, e202101102 CrossRef CAS.
- C. Schneider, S. Demeshko, F. Meyer and C. G. Werncke, Chem. – Eur. J., 2021, 27, 6348–6353 CrossRef CAS PubMed.
- (a) K. M. Lancaster, M. Roemelt, P. Ettenhuber, Y. Hu, M. W. Ribbe, F. Neese, U. Bergmann and S. DeBeer, Science, 2011, 334, 974–977 CrossRef CAS PubMed; (b) T. Spatzal, M. Aksoyoglu, L. Zhang, S. L. A. Andrade, E. Schleicher, S. Weber, D. C. Rees and O. Einsle, Science, 2011, 334, 940 CrossRef CAS PubMed; (c) O. Einsle, F. A. Tezcan, S. L. A. Andrade, B. Schmid, M. Yoshida, J. B. Howard and D. C. Rees, Science, 2002, 297, 1696–1700 CrossRef CAS PubMed; (d) D. C. Rees, F. Akif Tezcan, C. A. Haynes, M. Y. Walton, S. Andrade, O. Einsle and J. B. Howard, Philos. Trans. R. Soc., A, 2005, 363, 971 CrossRef CAS PubMed.
- (a) D. Sippel, M. Rohde, J. Netzer, C. Trncik, J. Gies, K. Grunau, I. Djurdjevic, L. Decamps, S. L. A. Andrade and O. Einsle, Science, 2018, 359, 1484–1489 CrossRef CAS PubMed; (b) W. Kang, C. C. Lee, A. J. Jasniewski, M. W. Ribbe and Y. Hu, Science, 2020, 368, 1381–1385 CrossRef CAS PubMed.
- (a) M. J. Ryle, H. I. Lee, L. C. Seefeldt and B. M. Hoffman, Biochemistry, 2000, 39, 1114–1119 CrossRef CAS PubMed; (b) M. Kumar, W. P. Lu and S. W. Ragsdale, Biochemistry, 1994, 33, 9769–9777 CrossRef CAS PubMed; (c) L. C. Seefeldt, M. E. Rasche and S. A. Ensign, Biochemistry, 1995, 34, 5382–5389 CrossRef CAS PubMed.
- C. Schneider, S. Ivlev and C. G. Werncke, Eur. J. Inorg. Chem., 2022, e202200706 Search PubMed.
- C. Schneider, S. J. Groß, S. Demeshko, S. Bontemps, F. Meyer and C. G. Werncke, Chem. Commun., 2021, 57, 10751–10754 RSC.
- (a) A. Albers, S. Demeshko, K. Pröpper, S. Dechert, E. Bill and F. Meyer, J. Am. Chem. Soc., 2013, 135, 1704–1707 CrossRef CAS PubMed; (b) S. Yao, F. Meier, N. Lindenmaier, R. Rudolph, B. Blom, M. Adelhardt, J. Sutter, S. Mebs, M. Haumann, K. Meyer, M. Kaupp and M. Driess, Angew. Chem., Int. Ed., 2015, 54, 12506–12510 CrossRef CAS PubMed.
- J. Rienmüller, A. Schmidt, N. J. Yutronkie, R. Clérac, C. G. Werncke, F. Weigend and S. Dehnen, Angew. Chem., Int. Ed., 2022, 61, e202210683 CrossRef PubMed.
- Y. Liu and L. Deng, J. Am. Chem. Soc., 2017, 139, 1798–1801 CrossRef CAS PubMed.
- J. Yang and T. D. Tilley, Angew. Chem., Int. Ed., 2010, 49, 10186–10188 CrossRef CAS PubMed.
- (a) Y. Ohki, Y. Shimizu, R. Araake, M. Tada, W. M. C. Sameera, J.-I. Ito and H. Nishiyama, Angew. Chem., Int. Ed., 2016, 55, 15821–15825 CrossRef CAS PubMed; (b) T. Higaki, K. Tanaka, H. Izu, S. Oishi, K. Kawamoto, M. Tada, W. M. C. Sameera, R. Takahata, T. Teranishi, S. Kikkawa, S. Yamazoe, T. Shiga, M. Nihei, T. Kato, R. E. Cramer, Z. Zhang, K. Meyer and Y. Ohki, J. Am. Chem. Soc., 2025, 147, 3215–3222 CrossRef CAS PubMed; (c) T. N. Gieshoff, U. Chakraborty, M. Villa and A. Jacobi von Wangelin, Angew. Chem., Int. Ed., 2017, 56, 3585–3589 CrossRef CAS PubMed.
- U. Chakraborty, E. Reyes-Rodriguez, S. Demeshko, F. Meyer and A. Jacobi von Wangelin, Angew. Chem., Int. Ed., 2018, 57, 4970–4975 CrossRef CAS PubMed.
- A. J. South, A. M. Geer, L. J. Taylor, H. R. Sharpe, W. Lewis, A. J. Blake and D. L. Kays, Organometallics, 2019, 38, 4115–4120 CrossRef CAS.
- H. R. Sharpe, A. M. Geer, W. Lewis, A. J. Blake and D. L. Kays, Angew. Chem., Int. Ed., 2017, 56, 4845–4848 CrossRef CAS PubMed.
- H. R. Sharpe, A. M. Geer, H. E. L. Williams, T. J. Blundell, W. Lewis, A. J. Blake and D. L. Kays, Chem. Commun., 2017, 53, 937–940 RSC.
- A. M. Geer, J. Navarro, P. Alamán-Valtierra, N. T. Coles, D. L. Kays and C. Tejel, ACS Catal., 2023, 13, 6610–6618 CrossRef CAS PubMed.
- H. R. Sharpe, A. M. Geer, T. J. Blundell, F. R. Hastings, M. W. Fay, G. A. Rance, W. Lewis, A. J. Blake and D. L. Kays, Catal. Sci. Technol., 2018, 8, 229–235 RSC.
- D. S. Belov, L. Mathivathanan, M. J. Beazley, W. B. Martin and K. V. Bukhryakov, Angew. Chem., Int. Ed., 2021, 60, 2934–2938 CrossRef CAS PubMed.
- G. Ung and J. C. Peters, Angew. Chem., Int. Ed., 2015, 54, 532–535 CrossRef CAS PubMed.
- J. Du, W. Chen, Q. Chen, X. Leng, Y.-S. Meng, S. Gao and L. Deng, Organometallics, 2020, 39, 729–739 CrossRef CAS.
- P. Varava, Z. Dong, R. Scopelliti, F. Fadaei-Tirani and K. Severin, Nat. Chem., 2021, 13, 1055–1060 CrossRef CAS PubMed.
- Q. Sun, J.-N. Belting, J. Hauda, D. Tymann, P. W. Antoni, R. Goddard and M. M. Hansmann, Science, 2025, 387, 885–892 CrossRef CAS PubMed.
- Y. Gao, G. Li and L. Deng, J. Am. Chem. Soc., 2018, 140, 2239–2250 CrossRef CAS PubMed.
- (a) W. W. Brennessel, R. E. Jilek and J. E. Ellis, Angew. Chem., Int. Ed., 2007, 46, 6132–6136 CrossRef CAS PubMed; (b) W. W. Brennessel, V. G. Young and J. E. Ellis, Angew. Chem., Int. Ed., 2006, 45, 7268–7271 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2025 |