NaOH/urea aqueous solution facilitates spectroscopic quantitation of lignin in corn stalk†
Kexin
Yan
a,
Nan
Li
a,
Qifan
Tian
a,
Zijian
Tan
ab,
Qingchen
Qin
a,
Jinao
Duan
 a,
Leilei
Zhu
a,
Leilei
Zhu
 *b and
Haifeng
Liu
*b and
Haifeng
Liu
 *a
*a
aJiangsu Collaborative Innovation Centre of Chinese Medicinal Resources Industrialization, School of Pharmacy, Nanjing University of Chinese Medicine, Nanjing 210023, China. E-mail: haifeng.liu@njucm.edu.cn
bKey Laboratory of Engineering Biology for Low-Carbon Manufacturing, Tianjin Institute of Industrial Biotechnology, Chinese Academy of Sciences, Tianjin 300308, China. E-mail: zhu_ll@tib.cas.cn
First published on 1st February 2025
Abstract
A facile spectrometric determination of lignin in corn straw was constructed through dissolving the lignin–carbohydrate complex in aqueous solution at room temperature, where NaOH/urea was induced to prepare a transparent aqueous solution of carbohydrate-linked lignin for quantification at 298 nm without any interference from the carbohydrate.
Lignin is the most abundant source of renewable aromatics in nature,1 and plays a key role in bridging cellulose and hemicellulose of the plant cell wall.2 Besides working as a green raw material3–5 for industrial adhesives, antibacterial gels,6,7 redox electrodes8 and energy storage materials,9 the abundant functional groups such as phenolic hydroxyl groups, methoxy groups and carboxyl groups endow lignin with antibacterial,10 anti-ultraviolet,11 anti-oxidation6 and other biological activities.12 However, as part of lignocellulose (15–35 wt%), lignin combines cellulose and hemicellulose with diverse cross-linking bonds to form a complex three-dimensional network structure, which makes the determination and further high value-added utilization of lignin a real challenge.
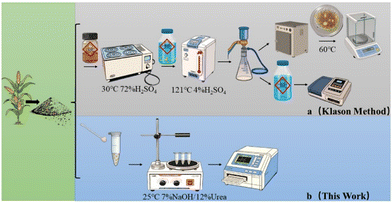
The Klason method (Fig. 1a) is the most used method for directed quantitation of lignin, where the insoluble residue obtained from two-step hydrolysis of concentrated/diluted acid (acid-insoluble lignin, AIL) is quantified by a gravimetric method and the dissolved lignin (acid-soluble lignin, ASL) is quantified by spectrophotometry. Based on the Klason method, two modified methods for quantification of lignin have been widely used in the pulp/paper industry and lignocellulosic biomass refining: the pulp and paper industry technology association (TAPPI) method and the National Renewable Energy Laboratory (NREL) protocol. However, these chemical processes need large amounts of samples, destroy the lignin structure and introduce interfering substances during measurement.13
The spectral methods for lignin detection include ultraviolet (UV) spectroscopy,13,14 infrared spectroscopy,15,16 and nuclear magnetic resonance spectroscopy,17,18 which do not destroy the structure of the lignin sample and only need a small amount of lignin. The principle of lignin quantitation by UV spectroscopy is based on the conjugated system in lignin, which has a strong selective absorption at 205 nm and 280 nm following Lambert–Beer's law. However, UV quantitative analysis needs to be carried out in a transparent solution. Meanwhile, it is normally difficult to prepare transparent solutions of lignocellulosic biomass in conventional solvents. Therefore, it is necessary to develop appropriate dissolving methods for lignocellulose samples for the following spectrometric determination of lignin.
Crop straw is an important source of lignocellulose. According to the statistics of the Food and Agriculture Organization of the United Nations, corn straw has a global annual output of more than one billion tons. Compared with other straws, corn stalk has higher carbon–nitrogen ratio/hardness/calorific value and more cellulose, which makes it more attractive for further utilization.19–27 Currently, besides the Klason method, near-infrared reflectance (NIR) spectroscopy28 and pyrolysis-GC-SIM-MS29 are also used for the determination of lignin in corn stover.
In the study of Cai Jie et al.,30 it was found that the short-staple cotton can be completely dissolved in a pre-cooled aqueous solution of NaOH (7%)/urea (12%) at 3000 rpm within 5 min to get a transparent solution (>60 g L−1). When we followed this method to extract cellulose from corn stover, it was found that part of the lignin in corn stover was also dissolved in this NaOH/urea aqueous solution. Besides the dissolution of cellulose in NaOH-based aqueous solutions,31 3% NaOH aqueous solution is a powerful solvent to dissolve hemicellulose.32 Therefore, NaOH/urea aqueous solution is a potential solvent to dissolve all of the biomass fractions of corn stover (cellulose, hemicellulose and lignin) into a transparent solution for the following spectrometric determination of lignin. A method for spectrometric determination of lignin in corn stover was established from this unexpected discovery (Fig. 1b).
Firstly, the standard lignin (Sigma-Aldrich, 471003), cellulose (Macklin, C804602) and hemicellulose (Macklin, X820567) were dissolved in NaOH (7%)/urea (12%) aqueous solution to prepare a 0.1 mg mL−1 solution, respectively. 200 μL of the solution (0.1 mg mL−1) was transferred to a 96-well microtiter plate for full-wavelength scanning (200–800 nm), which was recorded by a microplate reader (SuperMax 3000FA-PLUS, Shanghai Shanpu Biospectrum Biotechnology Co., Ltd). It was found that the lignin solution (0.1 mg mL−1) has a characteristic absorption at 298 nm (A298 = 1.39); meanwhile, the absorbances at the same wavelength of cellulose solution (A298 = 0.084) and hemicellulose (A298 = 0.089) were quite close to that of NaOH (7%)/urea (12%) solution (background, A298 = 0.080, Fig. 2). Further investigation showed that neither cellulose nor hemicellulose has characteristic absorbance at 298 nm (Fig. S1 and S2, ESI†). Therefore, lignin in NaOH/urea aqueous solution has characteristic absorbance at 298 nm without interference from cellulose and hemicellulose in the same solution.
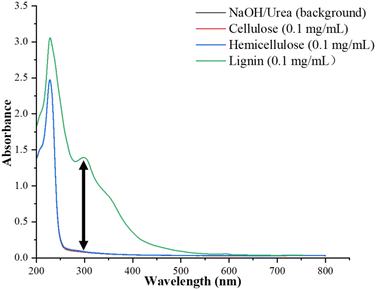 | ||
| Fig. 2 Full-wavelength scanning (200–800 nm) of standard lignin, cellulose and hemicellulose in 7% NaOH/12% urea aqueous solution. | ||
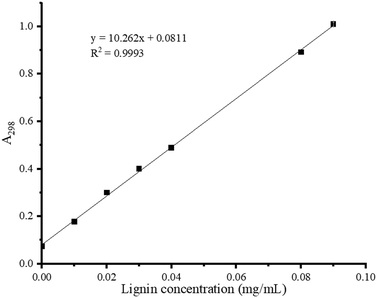
Based on the characteristic absorption at 298 nm, a calibration curve for standard lignin in 7% NaOH/12% urea solution was established with excellent linearity (R2 = 0.9993, Fig. 3).
The corn straw was powdered and passed through a 40-mesh sieve. The powder was mixed with 95% ethanol in a Soxhlet extractor and then refluxed until the extraction solution was colorless. The pretreated powder (2 mg) was mixed with NaOH/urea aqueous solution (4 mL) at 25 °C. After 24 h standing, the mixture was centrifuged at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 3 min, and part of the supernatant (200 μL) was transferred to a 96-well microtiter plate to measure the absorbance at 298 nm by microplate reader (SuperMax 3000FA-PLUS) and then the lignin concentration was calculated according to the calibration curve. All tests were performed at least in triplicate for the following calculation of the mean and standard deviation (Fig. 4).
000 rpm for 3 min, and part of the supernatant (200 μL) was transferred to a 96-well microtiter plate to measure the absorbance at 298 nm by microplate reader (SuperMax 3000FA-PLUS) and then the lignin concentration was calculated according to the calibration curve. All tests were performed at least in triplicate for the following calculation of the mean and standard deviation (Fig. 4).
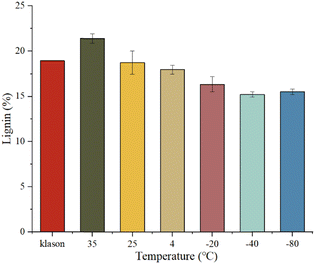
The pretreated corn straw (2 mg) was mixed with NaOH (7%)-urea (12%) aqueous solution (4 mL) in a glass vial, sealed and placed in refrigerators at 4 °C/−20 °C/−40 °C/−80 °C, in an oven (35 °C) or at room temperature (25 °C) for 24 h, and then centrifuged to collect the supernatant for absorbance measurement (200 μL per well) in a microtiter plate at 298 nm. All tests were performed at least in triplicate for the following calculation of the mean and standard deviation (Fig. 5). The measured concentration of lignin was increased with a rise of incubation temperature. The optimal temperature is 25 °C because after incubation at this temperature, the measured concentration (18.77%) is very closed to the concentration from the Klason method (18.94%), which is also consistent with data from the literature.33–35 In the case of completely dissolving pure cellulose (>60 mg mL−1) in NaOH/urea aqueous solution,30 the best ratio among NaOH, urea and water is 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12
12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 81, which is consistent with our test (Fig. S3, ESI†). Meanwhile their aqueous solution has to be precooled to −10 °C; therefore, low incubation temperatures between −80 °C to 4 °C were also tested here. Experimental results have shown that the low incubation temperatures decreased the measured lignin concentration probably because the solubility of the lignin–carbohydrate complex at low temperature is limited. Incubation of the lignin–carbohydrate complex at high temperature can depolymerize the complex and then interfere with the measurement of the lignin concentration; besides, the high incubation temperature (for example, 80 °C) can deconstruct standard lignin and interfere with its characteristic absorbance at 298 nm (Fig. S4d, ESI†).
81, which is consistent with our test (Fig. S3, ESI†). Meanwhile their aqueous solution has to be precooled to −10 °C; therefore, low incubation temperatures between −80 °C to 4 °C were also tested here. Experimental results have shown that the low incubation temperatures decreased the measured lignin concentration probably because the solubility of the lignin–carbohydrate complex at low temperature is limited. Incubation of the lignin–carbohydrate complex at high temperature can depolymerize the complex and then interfere with the measurement of the lignin concentration; besides, the high incubation temperature (for example, 80 °C) can deconstruct standard lignin and interfere with its characteristic absorbance at 298 nm (Fig. S4d, ESI†).
Finally, in order to figure out which kind of lignin can be completely dissolved in NaOH/urea aqueous solution, the neutralization reaction was carried out to precipitate the lignin of corn straw dissolved in NaOH/urea aqueous solution for further 1H–13C HSQC NMR analysis, and the spectrum was recorded on a Quantum-l 400 MHz spectrometer equipped with QOT X/H-F probe at 25 °C in DMSO-d6 (number of scans = 64, spectra width 1H = 13 ppm, center of the proton dimension = 4.7 ppm, spectra width 13C = 165 ppm, center of carbon dimension = 74 ppm, td in the indirect dimension = 128 (Fig. S5, ESI†)).
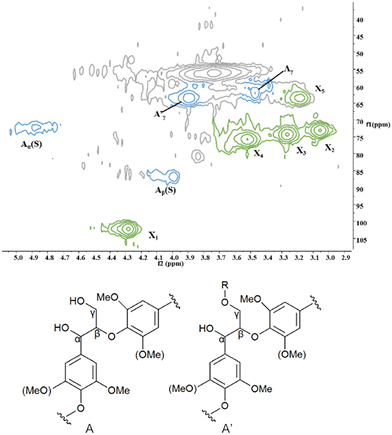
A typical β-O-4 linkage has been found in the side chain region of the 2D-HSQC spectrum of the lignin–carbohydrate complex from corn stover (Fig. 6) because the cross-signals of γ-acetylated β-O-4′ linkages (A′) (δC/δH 63.5/3.9) indicated the unique acylation at the γ-position of β-O-4 linkage. Additionally, compared with Cγ–Hγ correlations (Aγ) (δC/δH 59.9/3.40) and Cβ–Hβ correlation linked to S (Aβ(S)) units in β-O-4′ linkages (δC/δH 87.0/4.00), the signals of CαHα correlations (Aα(S)) in β-O-4′ linkages (δC/δH 72.3/4.87) were relatively dominant, suggesting that β-O-4′ was mainly coupled with S-units. The signals of β-D-xylopyranoside (X), the main lignin-xylan type, were also found at δC/δH 102.2/4.28, 73.1/3.06, 74.2/3.27, 75.7/3.52 and 63.5/3.19 corresponding to their C1–H1, C2–H2, C3–H3, C4–H4 and C5–H5, respectively (Table S1, ESI†) which proved that the major linkage is β-O-4, which is chemically linked to hemicellulose fraction (xylan).36,37
This study developed a facile spectrometric method to determine the concentration of lignin in corn straw based on dissolving carbohydrate linked lignin in NaOH/urea aqueous solution at room temperature for recording the absorbance at 298 nm.
Data availability
The data supporting this article have been included as part of the ESI.†Conflicts of interest
There are no conflicts to declare.References
- Q. Zou, H. He, J. Xie, S. Han, W. Lin, A. K. Mondal and F. Huang, Chem. Eng. J., 2023, 142029 CrossRef CAS.
- Y. Zhang, H. Meng, F. Lyu, X. Fan, P. Liu, X. He, Y. Huang, B. Chen, Y. Yang and J. Wei, Chin. Herb. Med., 2023, 15, 37–44 Search PubMed.
- D. Gan, W. Xing, L. Jiang, J. Fang, C. Zhao, F. Ren, L. Fang, K. Wang and X. Lu, Nat. Commun., 2019, 10, 1487 CrossRef PubMed.
- H. Zhang, K. Xue, X. Xu, X. Wang, B. Wang, C. Shao and R. Sun, Small, 2024, 20, e2305502 CrossRef PubMed.
- T. Liu, Y. Yang, L. Yan, B. Lin, L. Dai, Z. Huang and C. Si, Int. J. Biol. Macromol., 2024, 258, 129132 CrossRef CAS PubMed.
- X. Han, Y. Su, G. Che, Q. Wei, H. Zheng, J. Zhou and Y. Li, ACS Appl. Mater. Interfaces, 2022, 14(44), 50199–50214 CrossRef CAS PubMed.
- Y. Chao, S. Yu, H. Zhang, D. Gong, J. Li, F. Wang, J. Chen, J. Zhu and J. Chen, ACS Appl. Bio Mater., 2023, 6, 1525–1535 CrossRef CAS PubMed.
- X. He, L. Li, S. Yan, H. Fu, F. Zhong, J. Cao, M. Ding, Q. Sun and C. Jia, J. Colloid Interface Sci., 2024, 653, 1455–1463 CrossRef CAS PubMed.
- H. Liu, Y. Guan, L. Yan, Y. Zheng, C. Si and L. Dai, Green chemistry: an international journal and green chemistry resource: GC, 2024 Search PubMed.
- K. Li, W. Zhong, P. Li, J. Ren, K. Jiang and W. Wu, Int. J. Biol. Macromol., 2023, 252, 126281 CrossRef CAS PubMed.
- P. Feng, H. Wang, S. Gan, B. Liao and L. Niu, Small, 2024, e2312085 CrossRef PubMed.
- C. Libretti, L. S. Correa and M. A. Meier, Green Chem., 2024, 4358–4386 RSC.
- H. Zhang, H. Zhao, Y. Yang, H. Ren and H. Zhai, Green Chem., 2022, 24, 2212–2221 RSC.
- F. Lu, C. Wang, M. Chen and F. Y. A. J. Ralph, Green Chem., 2021, 5106–5112 RSC.
- X. Jin, X. Chen, C. Shi, M. Li, Y. Guan, C. Y. Yu, T. Yamada, E. J. Sacks and J. Peng, Bioresour. Technol., 2017, 241, 603–609 CrossRef CAS PubMed.
- Y. Tamaki and G. Mazza, J. Agric. Food Chem., 2011, 59, 504–512 CrossRef CAS PubMed.
- N. Jiang, Y. Pu and A. J. Ragauskas, ChemSusChem, 2010, 3, 1285–1289 CrossRef CAS PubMed.
- L. Fu, S. A. McCallum, J. Miao, C. Hart, G. J. Tudryn, F. Zhang and R. J. Linhardt, Fuel, 2015, 141, 39–45 CrossRef CAS PubMed.
- L. Xiang, H. Sheng, M. Xu, M. Redmile-Gordon, Y. Bian, X. Yang, X. Jiang and F. Wang, Sci. Total Environ., 2019, 663, 29–37 CrossRef CAS PubMed.
- Z. R. Staley, A. M. Schmidt, B. Woodbury, K. M. Eskridge, L. Durso and X. Li, J. Environ. Qual., 2020, 49, 745–753 CrossRef CAS PubMed.
- X. Gao, H. Zhang, J. Zhang, N. Weng and S. Huo, Bioresour. Technol., 2024, 394, 130259 CrossRef CAS PubMed.
- Z. Sun, X. Li and L. Liu, J. Anim. Physiol. Anim. Nutr., 2018, 102, 596–599 CrossRef CAS PubMed.
- J. Zhang, Y. H. Wang, Q. Y. Wei, X. J. Du and Y. S. Qu, Bioresour. Technol., 2018, 249, 342–347 CrossRef CAS PubMed.
- Y. Liu, H. Fan, X. Wang, J. Zhang, W. Li and R. Wang, Carbohydr. Polym., 2022, 276, 118763 CrossRef CAS PubMed.
- L. Yan, N. Liu, Y. Wang, H. Machida and X. Qi, Bioresour. Technol., 2014, 173, 462–466 CrossRef CAS PubMed.
- R. C. Qin, Y. Y. Ma, D. Wang, N. Z. Bao and C. G. Liu, J. Agric. Food Chem., 2023, 71, 1907–1920 CrossRef CAS PubMed.
- X. Lu, F. Li, X. Zhou, J. Hu and P. Liu, Food Chem., 2022, 384, 132622 CrossRef CAS PubMed.
- X. Zhang, Z. Chen, S. Yi and J. Liu, Infrared Phys. Technol., 2023, 104787 CrossRef CAS.
- G. van Erven, R. de Visser, D. W. H. Merkx, W. Strolenberg, P. de Gijsel, H. Gruppen and M. A. Kabel, Anal. Chem., 2017, 10907–10916 CrossRef CAS PubMed.
- J. C. J. Cai, L. Z. L. Zhang, J. Z. J. Zhou, H. L. H. Li, H. C. H. Chen and H. J. H. Jin, Macromol. Rapid Commun., 2004, 1558–1562 CrossRef CAS.
- T. Budtova and P. Navard, Cellulose, 2016, 5–55 CrossRef CAS.
- G. Fu, P. Yue, Y. Hu, N. Li, Z. Shi and F. Peng, Int. J. Polym. Sci., 2018, 1–8 Search PubMed.
- Z. Yang, S. Du, J. Mei, J. Li, E. Liu and L. Han, Trans. Chin. Soc. Agric. Eng., 2019, 280–286 Search PubMed.
- Y. Wen, X. Liu, F. He, Y. Shi, F. Chen, W. Li, Y. Song, L. Li, H. Jiang, L. Zhou and L. Wu, Int. J. Biol. Macromol., 2024, 136140 CrossRef CAS PubMed.
- Y. Chen, H. Fan and F. Meng, J. Sci. Food Agric., 2017, 1592–1598 CrossRef CAS PubMed.
- X. Li, Y. Xia, X. Hu, Q. Liu, W. Li, L. Yan and L. Ma, Ind. Crops Prod., 2023, 116896 CrossRef CAS.
- M. L. M. Lei, H. Z. H. Zhang, H. Z. H. Zheng, Y. L. Y. Li, H. H. H. Huang and R. X. R. Xu, Chin. J. Chem. Eng., 2013, 427–433 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4cc06423b |
| This journal is © The Royal Society of Chemistry 2025 |