Recent developments and prospects of inorganic nanozymes for biomedical applications
Siqi
Zhan
a,
Yan
Fu
a,
Hong Yu
Yang
 *a and
Doo Sung
Lee
*a and
Doo Sung
Lee
 *b
*b
aCollege of Materials Science and Engineering, Jilin University of Chemical Technology, Jilin City 132022, Jilin Province, P. R. China. E-mail: yhyqdu@163.com
bSchool of Chemical Engineering, Sungkyunkwan University, Suwon, Gyeonggi-do 16419, Republic of Korea
First published on 24th October 2025
Abstract
The development of inorganic nanozymes has revolutionized the field of nanotechnology by providing a new class of catalytic materials that exhibit enzyme-like activities. Compared with traditional natural enzymes, nanozymes have broad application prospects in the field of biomedicine due to their higher chemical stability, stronger environmental adaptability, and ability to maintain their activity under extreme conditions. To provide a comprehensive overview of the recent progress made in this field, herein, an overview of inorganic nanozymes for biomedical applications is provided. In this review, the structure, synthesis methods, and catalytic mechanism of inorganic nanozymes are summarized. Subsequently, the latest progress of various inorganic nanozymes for the applications in biomedicine is reviewed, including diagnostic applications, therapeutic applications and drug delivery systems. Then, the recent developments in the modification and multifunctionalization of novel inorganic nanozymes are discussed. Finally, the challenges and prospects of inorganic nanozymes in the field of biomedicine are highlighted and pointed out. We hope that this timely review can further advance this promising field.
1. Introduction
Natural enzymes, as a class of extremely important biological catalysts, have long been known for their exceptional substrate specificity and catalytic efficiency, enabling chemical reactions within organisms to proceed efficiently and specifically under physiological conditions.1,2 However, their practical utility in industrial and biomedical settings is significantly hampered by their inherent limitations. Natural enzymes exhibit poor stability, with their activity readily compromised by environmental fluctuations in temperature, pH, and pressure.3 Furthermore, their susceptibility to other proteolytic degradation presents additional challenges.To overcome the above-mentioned limitations, some alternative enzyme mimics like enzymatic hybrid nanoflowers and nanozymes have been actively explored.4 Nanozymes combine the intrinsic properties of nanomaterials with enzyme-like catalytic functions, which have the ability to convert catalytic substrates because of the presence of specific nanostructures with similar active sites or charge/electron transfer.5 The term “nanozymes” was coined by Scrimin and co-workers in 2004 in their work focusing on the synthesis of triazacyclononane/Zn2+–gold nanoparticles and their catalytic behavior in the transphosphorylation reaction.6 In 2007, the discovery of inherent peroxidase-like activity in inert ferromagnetic nanoparticles led to the in-depth and sustained exploration of nanozymes.7
Compared with natural enzymes, artificial nanozymes have been demonstrated to exhibit enhanced stability, simplified manufacturing, and superior durability.8 More notably, some non-biological processes can also be catalyzed by nanozymes.9 As a type of nanozyme, inorganic nanozymes can achieve higher stability and adjustable enzyme activity than organic nanozymes, making them more suitable for industrial and commercial applications.10–12 Consequently, research efforts dedicated to advancing the design and applications of inorganic nanozymes have surged considerably in recent years (Scheme 1).
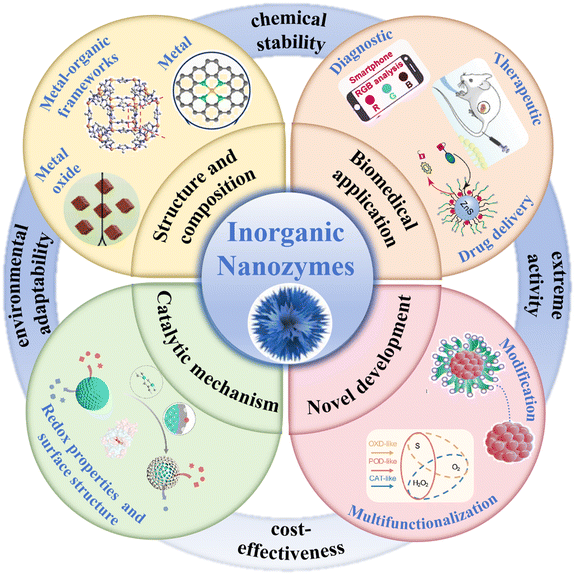 | ||
| Scheme 1 Schematic of the development of inorganic nanozymes and their applications in the biomedical field. The light-yellow region (inner circle) presents the various structures and compositions of inorganic nanozymes (reproduced from ref. 68 with permission from Elsevier, Copyright 2024; ref. 22 with permission from Elsevier, Copyright 2024; and ref. 41 with permission from Elsevier, Copyright 2024). The light-green region (inner circle) shows the catalytic mechanism of inorganic nanozymes (reproduced from ref. 30 with permission from the American Chemical Society, Copyright 2024). The light-pink region (inner circle) presents the multifunctionalization and modification of novel inorganic nanozymes (reproduced from ref. 86 with permission from Wiley, Copyright 2023). The light-orange region (inner circle) shows their diverse biomedical applications (reproduced from ref. 83 with permission from The Royal Society of Chemistry, Copyright 2022; ref. 49 with permission from Elsevier, Copyright 2022; ref. 148 with permission from Journal of the American Chemical Society, Copyright 2022). The surrounding blue region (outer circle) presents the advantages of inorganic nanozymes. | ||
1.1 Characteristics of inorganic nanozymes
Inorganic nanozymes are a class of enzyme mimics predominantly composed of metal or metal oxide nanoparticles or their composites with other materials.13 The advantages of metal nanoparticles and functionalized polyoxometalates are combined in inorganic nanozymes.14 These nanomaterials are engineered to exhibit specific enzyme-like activities and emulate the activity of specific natural enzymes, such as peroxidase-like, oxidase-like, catalase-like, or superoxide dismutase-like activities.15 Crucially, their catalytic performance is primarily governed by their physicochemical properties, such as size, shape, surface characteristics, and nature of the core material.16 | ||
| Fig. 1 (a) Multifunctional nanozyme-enabled assays. Reproduced from ref. 13 with permission from the American Chemical Society, Copyright 2022. (b) Illustration of the effects of four neurotransmitters, namely, serotonin (5-HT), 5-hydroxyindoleacetic acid (5-HIAA), γ-aminobutyric acid, and Ach, on the hippocampus. Reproduced from ref. 15 with permission from the American Chemical Society, Copyright 2024. (c) Antitumor effect in vivo after different treatments. Reproduced from ref. 83 with permission from The Royal Society of Chemistry, Copyright 2022. | ||
1.2 Comparative advantages of inorganic nanozymes
As discussed above, inorganic nanozymes have emerged as promising alternatives to overcome the inherent limitations of natural enzymes.29 Compared to their traditional counterparts, the distinct advantages of inorganic nanozymes are mainly reflected in the their catalytic activity, enhanced stability, and cost-effectiveness/synthetic scalability.30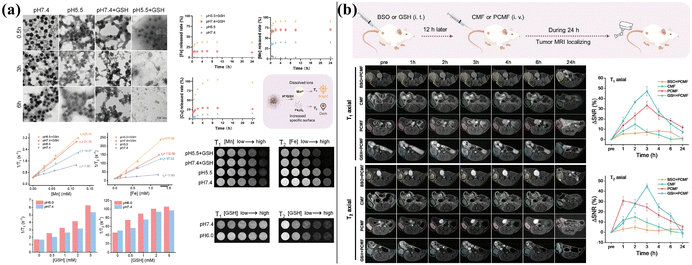 | ||
| Fig. 2 The T1–T2 dual-modal MRI dynamic activation of CMF. (a) TEM images observation: the morphology changes of CMF after different treatments. (b) The release curves of Fe, Mn, and Cu ions from CMF under different conditions. Reproduced from ref. 36 with permission from Wiley, Copyright 2023. | ||
1.3 Applications of inorganic nanozymes
The discovery of inorganic nanozymes has fundamentally challenged the long-held view of inorganic nanomaterials as biologically inert, revealing their intrinsic biological effects and advancing the field of enzyme mimics.44 Combining the high catalytic activity of natural enzymes with exceptional stability and cost-effectiveness, the research field of nanozymes has gradually expanded since they were first reported in 2007, mainly including environmental remediation, industrial catalysis, and biomedicine.45,46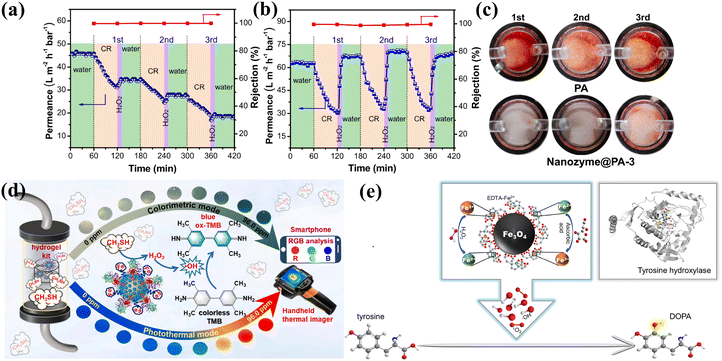 | ||
| Fig. 3 Applications of inorganic nanozymes. Water permeance and CR rejection of PA membrane (a) and nanozyme@PA-3 membrane (b), and digital pictures of the tested membranes (c) in fouling cleaning cycles. Reproduced from ref. 48 with permission from Elsevier, Copyright 2025. (d) Diagram of the AOX-assisted Fe&Cu@CD nanozyme-based agarose portable hydrogel kit for enzymatic cascade catalytic H2O2-self-supplying colorimetric and photothermal signal synergistic amplification to achieve the onsite visual monitoring of the atmospheric CH3SH around a waste pile. Reproduced from ref. 49 with permission from Elsevier, Copyright 2024. (e) Schematic of the reaction process for the hydroxylation of tyrosine to DOPA by the Fe3O4@EDTA–Fe2+ nanozyme. Reproduced from ref. 54 with permission from the American Chemical Society, Copyright 2022. | ||
In summary, inorganic nanozymes have rapidly evolved into a class of multifunctional catalytic materials. Continued research advances and the development of increasingly sophisticated nanozyme systems are poised to unlock novel pathways for their integration in practical applications.
2. Types of inorganic nanozymes
2.1 Structure and composition
To date, inorganic nanozyme systems have been constructed from diverse nanomaterials. Notably, a single nanozyme can exhibit multiple enzyme activities (such as catalase-, peroxidase-, oxidase-like activities) under different conditions.58 Based on their primary composition, inorganic nanozymes can be roughly divided into three main types, including metal-based, metal oxide-based, and metal–organic framework (MOF)-based nanozymes.59Pioneering work in 2007 identified that Fe3O4 nanoparticles possess intrinsic peroxidase-like activity, which was systematically investigated.65 Fe3O4 catalyzes the H2O2-dependent oxidation of chromogenic substrates such as TMB, following a ping-pong catalytic mechanism. Since then, numerous other metal oxide nanostructures have been reported to possess enzyme-like properties. For example, MnO2 exhibits stable dual enzymatic activities in the treatment of tumors. Mn2+ catalytically decomposes endogenous substances (often accompanied by glutamate consumption) to generate O2 and highly toxic ˙OH, thus alleviating tumor hypoxia and killing tumor cells.66 Another typical example was NiFe2O4 nanoparticles, which mimic peroxidase-like activity to detect herbicide residues in the presence of H2O2.67 CeO2 was also taken as an example to show peroxidase-like activity, which is primarily governed by the Ce3+/Ce4+ pairs on its surface. During the glucose detection process in a serum sample, the generation of superoxide radicals (O2˙−) from H2O2 and the oxidation of TMB to ox-TMB can be catalyzed by Mn/CeO2 nanozyme at the same time.68 In addition, oxides such as CO3O4, V2O5 and CuO are also known to possess multiple enzyme-like properties, including catalase-like, and superoxide dismutase-like properties.69,70 Although the detailed mechanisms underlying these catalytic reactions are subjects of ongoing investigation, key factors influencing the efficiency and selectivity of metal oxide-based nanozymes are well-recognized, including the metal oxidation state, nanoparticle size, and specific surface characteristics.
 | ||
| Fig. 4 Structure and composition of inorganic nanozymes. (a) Platinum nanoparticle-regulated V2C MXene nanoplatforms. Reproduced from ref. 135 with permission from Wiley, Copyright 2024. (b) CuS antioxidant nanozymes. Reproduced from ref. 119 with permission from the American Chemical Society, Copyright 2023. (c) Metal oxide nanozymes. Reproduced from ref. 68 with permission from Elsevier, Copyright 2024. | ||
For example, Yin hui Yi et al. synthesized a Pt-NP/Fe-MOF hybrid material, consisting of platinum nanoparticles (Pt NPs) integrated with a metal–organic framework (MIL-88B-NH2, Fe-MOF). The superior peroxidase-mimicking activity of the hybrid material for catalyzing the oxidation of TMB is attributed to the synergistic catalysis effect of Pt-NPs and Fe-MOF. Specifically, electron transfer from Pt to Fe accelerates the Fe2+/Fe3+ redox cycling within the MOF, significantly enhancing the peroxidase-like performance. Notably, the affinity of Pt-NPs/Fe-MOF for the substrate TMB and H2O2 exceeds that of natural horseradish peroxidase.73 MOF nanozymes also demonstrate significant potential in biomedical applications, particularly in modulating oxidative stress and inflammation. Yuan Tian et al. investigated the effect of the MOF-818 nanozyme on antioxidant and anti-inflammatory functions. The role of MOF-818 can be summarized as the following two points: (1) eliminating excessive intracellular ROS and improving cell viability and (2) blocking the recruitment and entry of macrophages and reducing the number of inflammatory macrophages.74 Based on this, an MOF-818-2 with a higher Cu(II) loading was reported. Compared with other copper-based mimics reported previously, CN-MOF-818 was observed to exhibit enhanced oxidase-mimicking activity and stability.75 As research progresses, further exploration into the synthesis, design, and optimization of MOF nanozymes will likely unlock novel opportunities for their use in various industrial, environmental, and medical fields.
2.2 Synthesis methods
Inorganic nanozymes can be synthesized on a large scale via simple and scalable methods, which are more cost-effective than natural enzymes at the same time.76 The most prevalent synthesis strategies include chemical reduction, hydrothermal/solvothermal methods, template-assisted synthesis, co-precipitation, and biomineralization.77 These approaches allow precise control of the nanomaterial size, shape, composition, and structure, which directly influence their catalytic performance. | ||
| Fig. 5 (a) Ni-NPC synthesis, adsorption process, and scheme of colorimetric sensing platform for CMP assay based on Ni-NPC. Reproduced from ref. 80 with permission from the American Chemical Society, Copyright 2024. (b) Schematic of the Fe3O4@Au/Cu-MOF-based ABTS˙+ turn-off colorimetric system for the detection of glutathione. Reproduced from ref. 82 with permission from Elsevier, Copyright 2023. (c) SAzymes with competing multiple enzyme-like activities. Reproduced from ref. 86 with permission from Wiley, Copyright 2023. | ||
Based on the above-mentioned content, the structural characteristics, synthesis methods, advantages and disadvantages in the synthesis process of different inorganic nanozymes are summarized in Table 1. Understanding the fundamental principles governing these diverse synthesis strategies is paramount for rationally designing inorganic nanozymes with superior catalytic properties and enhanced stability. Future research efforts should focus on developing more sustainable, scalable and precisely controllable synthesis methodologies to further advance the catalytic performance and broaden the applicability of inorganic nanozymes.
| Types | Structural characteristics | Synthesis methods | Advantages | Disadvantages |
|---|---|---|---|---|
| Metal based | Saturated atoms on the surface acting as active sites | Chemical reduction | Simple; cost-effective | Aggregation; toxic reducing agents |
| Metal oxide based | Metal ions within the oxide lattice as active sites | Hydrothermal and solvothermal | High-quality; well-defined structures | High energy consumption; long synthesis times |
| Co-precipitation | Uniform size distributions | Lack of control over the morphology | ||
| Metal–organic frameworks (MOF) based | Porous structure provides a larger specific surface area as the site for contact | Biomineralization | Eco-friendly; mild-conditions | Complexity of the biological system; long reaction times |
2.3 Catalytic mechanism
The catalytic mechanism of inorganic nanozymes is different from that of traditional enzymes (Fig. 6). It is primarily driven by the redox properties of metal ions and the surface structure of nanoparticles, modulated by intrinsic physicochemical properties including metal center, surface structure, and nanoparticle size.91,92 The focus of attention is mainly on the redox properties, active sites, and factors affecting their catalytic efficiency. | ||
| Fig. 6 Catalytic mechanism of inorganic nanozymes. (a) LiFePO4 NPs as verification materials and their POD-like activity. Reproduced from ref. 94 with permission from Springer, Copyright 2022. (b) Dye degradation analysis of nanozyme with the test conditions of pH 7.0, 50 °C, 75 mg L−1 Ag/Fe3O4@h-BN, 10 mg L−1 dye and 0.4 M H2O. Reproduced from ref. 97 with permission from Elsevier, Copyright 2023. (c) Schematic of the integrated PRTM-RhNC@rGO composite nanozyme with high selectivity, activity, and stability for the catalytic degradation of uric acid. Reproduced from ref. 98 with permission from the American Chemical Society, Copyright 2024. (d) SOD mimetic activity of Hep-H and CS-H that convert the superoxide anion to hydrogen peroxide. Reproduced from ref. 104 with permission from Elsevier, Copyright 2023. | ||
Inorganic nanozymes frequently exhibit redox-active properties due to the ability of metal ions to exist in multiple oxidation states, which enables the transfer of electrons during catalysis.93 The mechanism of Fe3O4 nanozymes with POD-like activity has been displayed, where the interior Fe2+ and surface Fe2+ simultaneously affect their enzyme activity through electron transfer. The slow oxidation of Fe3O4 simultaneously with the catalytic reaction occurs during the electron transfer process.94
The catalytic activity of inorganic nanozymes is closely linked to the structure of their active sites, which are primarily located on the surface of the nanoparticles.95 The surface area-to-volume ratio plays a critical role in determining the catalytic efficiency, given that a larger surface area provides more active sites for reactions.96 This principle can be exemplified by the Ag/Fe3O4@h-BN nanozyme with enhanced removal efficiency in the detection and removal of As(V) compared to other materials. This is mainly attributed to its large specific surface area and porous characteristics after doping Ag and Fe3O4, which could enhance the diffusion and mass transfer, and expose binding sites on the pore walls.97 Additionally, the specific arrangement of metal ions on the nanoparticle surface influences the reactivity and selectivity of the nanozyme.98
The surface structure of metal oxide nanoparticles also plays a role in modulating their catalytic properties. The presence of surface defects, such as oxygen vacancies and under-coordinated metal ions, can enhance the reactivity of the nanoparticle surface by providing additional active sites for catalysis.99 In MoO3-based organic/inorganic superlattices, rich oxygen vacancies have been introduced into their final structure. Oxygen vacancies can not only provide enhanced adsorption sites for O2 activation, but also promote the transfer of electrons from Mo to O2, generating ˙O−, similar to the function of natural oxidase.100
The size of inorganic nanozymes has a profound effect on their catalytic activity.101 Smaller nanoparticles generally exhibit superior catalytic efficiency due to their increased surface-area-to-volume ratios and available active sites. Researchers have demonstrated that cerium oxide nanoparticles (4.5, 7.8, 23 and 28 nm) exhibit enhanced catalase-like and superoxide dismutase-like activity at smaller size.102 The size effect of Pd–Ir core–shell nanoparticles on peroxidase-like activities were also verified by Zheng Xi et al. In the case of the peroxidase-like activities of individual nanoparticles, larger particle size will result in the enhanced catalytic effects. The area-specific catalytic activity in the size range of 3.3–9.8 nm is similar but inversely proportional after the size reached 13.0 nm. In addition, the detection sensitivities in ELISA of biomarkers increases as the particle size decreases.103 Abhishek Sahu et al. proved that the catalase-like and superoxide dismutase-like activity in porphyrin was associated with its size. Compared to hemin-conjugated chitosan and free hemin, higher enzymatic activities and stability were observed in hemin-conjugated heparin due to its more stable and smaller (<50 nm) self-assembled nanostructure.104 Furthermore, the size of nanoparticles can influence their electronic structure, given that the energy levels of the metal ions may shift with changes in particle size.
The shape of nanoparticles is another factor that can influence their catalytic properties.105 Nanocubes, nanorods, and nanosheets of metal oxides have been found to exhibit different catalytic behaviors due to the variations in the arrangement of atoms on their surface. Yongjian Ai and co-workers in 2021 explored the influence of coordination hierarchical superstructures on peroxidase-like activity and tumor photothermal treatment. Based on this reaction system and metal–ligand cross-linking strategy, a series of structures with 0D nanospheres, 1D nanorods, and 3D hierarchical superstructures (flower-like, octopus-like, and hedgehog-like) morphologies was designed. Among them, the hierarchical superstructures exhibited higher enzyme activity and photothermal effect.106
3. Applications of inorganic nanozymes in the biomedical field
3.1 Diagnostic application
Nanozymes exhibit potential for the low-cost and highly sensitive detection of toxic metal ions, which pose significant threats to human health and the ecosystem.110 Congcong Lou et al. designed a dual-functional sensor based on an NH2-MIL-101(Fe)@Cu/CeO2 hybrid nanozyme to investigate Cu2+ and Hg2+ without interference. In this colorimetric sensor, Hg2+ inhibits the glutathione (GSH)-mediated reduction of oxidized TMB (blue → colorless) by binding surface thiol groups. Cu2+ quenches fluorescence via reaction with surface amino groups.111 Similarly, the detection of Al3+ by a single-atom Ce–N–C nanozyme with phosphatase-like activity has been validated, which is attributed to the specific binding of Al3+ with the O atom in the nanozyme, leading to the formation of Al–O bonds and inhibition of enzyme activity.112
The early diagnosis and prevention of cancer can also be achieved through the detection of proteins using nanoenzymes.113 Urinary extracellular vesicles (uEVs) are considered the most promising biomarkers for the prognosis and diagnosis of bladder cancer, but their ultra-low abundance and huge heterogeneity bring great challenges to their detection technology. Thus, to solve this problem, a platinum nanozyme with choline phosphate-grafted was presented to multiplex profile EV protein markers. Relying on the nonspecific polyvalent interactions between the “CP-inverse” phosphatidylcholine (PC) of EV and CP installed on the nanozymes, the immunoassay of nanozymes can not only be used to judge the residual tumor after bladder cancer surgery, but also used to detect and distinguish bladder cancer donors, cystitis donors and healthy donors.114 In the same situation, diseases of the kidney and prostate can also be diagnosed by detecting acid phosphatase in human serum. Their successful detection is due to the inhibition of acid phosphatase on the blue charge transfer complex generated by nanozyme redox TMB.115
Disease diagnostics can also be achieved through the nanozyme detection of nucleic acids. Mengya He established the detection of respiratory syndrome coronavirus 2 through an iron manganese silicate nanozyme. The POD-like activity was inhibited by amplification-generated pyrophosphate ions.116
 | ||
| Fig. 7 Diagnostic application of inorganic nanozymes. (a) Schematic of periodontitis modeling, H&E staining of tissue sections, and 3D micro-CT reconstructed images of the maxillary molar area. Reproduced from ref. 110 with permission from Elsevier, Copyright 2023. (b) EV profiling for BC detection. Reproduced from ref. 113 with permission from the American Chemical Society, Copyright 2024. | ||
A recent study developed a biosensor comprised of crystalline CuS nanoparticles with appreciable superoxide dismutase-mimicking activity and sensitive colorimetric determination and detection of epinephrine (EP) neurotransmitters. This study achieved a linear detection range of 0–16 μm with a limit of detection at 457 nm.119 Bin Hong and co-workers developed a smartphone-assisted paper sensor based on AuPt nanoparticle-coated enzyme–antibiotic–inorganic nanoflowers. The integrated bio-recognition and dual signal amplification capabilities of this system facilitated the rapid, highly sensitive detection of S. typhimurium in food samples.120 Liming Wang et al. established a rapid light-controlled colorimetric sensing platform based on 5,10,15,20-tetrakis(4-carboxylpheyl) cobalt(II) porphyrin–TiO2 nanozymes to determine H2O2 and amikacin in two linear stages for the first time. The biosensing relies on the change in the peroxidase-like activity of nanozymes under light irradiation to control the color reaction.121 As the research in this field progresses, the further development of nanozyme-based biosensors holds promise for revolutionizing diagnostics and enabling more efficient, affordable, and accessible healthcare solutions worldwide.
3.2 Therapeutic application
In addition to ROS production, the versatility of inorganic nanozymes allows effective synergy with established anti-cancer modalities, overcoming inherent limitations.125 To break the poor electrical conductivity limits of electrotherapy, a novel copper-based inorganic nanozyme has been developed by encapsulating liposomes embedded in potassium chloride nanoclusters. Its anti-tumor mechanism mainly includes the following three parts: (1) the release of K+, Cl− and Cu2+ within the tumor microenvironment can improve the electrical ablation and activate the pyroptosis of cancer cells; (2) Cu2+ catalyzes the Fenton-like reaction, generating highly toxic ˙OH to cause mitochondrial damage; and (3) electrical stimulation can enhance the oxidative stress level of cancer cells by promoting the generation of hypochlorite radicals.126
Near-infrared light-induced photothermal therapy (PTT) is also one of the methods for ablating tumors, but the high-temperature damage to proximal tissues caused by PTT has always been a major concern. Ultra-small gold nanozymes immobilized within a metal organic framework can solve the high-temperature problem. The inhibition of tumor growth by nanozymes is mainly attributed to their glucose oxidase (GOD)-like activity and photothermal/photosensitive properties.127 To address the hypoxia issue in photodynamic therapy (PDT), a PtBi-β-CD-Ce6 nanoplatform was synthesized, which exhibited catalase-like activity to promote the production of O2 through H2O2 conversion.128 To overcome the H2O2 deficiency in the tumor ablation of chemodynamic therapy (CDT), an FePBG@GOX nanomimic was designed. Its GOx component consumes intratumoral glucose to generate H2O2, which is then utilized in Fe2+-catalyzed Fenton reactions to produce ROS, while simultaneously depleting tumor nutrients.129
As research into the safety, targeting strategies, and optimization of inorganic nanozymes continues, these materials have the potential to revolutionize cancer therapy by providing highly effective, targeted treatments with minimized side effects.
 | ||
| Fig. 8 Therapeutic applications of inorganic nanozymes. (a) In vivo biodistribution and biosafety of liposome-encapsulated copper oxide (CuO) embedded with potassium chloride nanoclusters. Reproduced from ref. 126 with permission from The Royal Society of Chemistry, Copyright 2023. (b) In vitro anti-cancer ability of Au-FeSAzyme. Reproduced from ref. 127 with permission from Elsevier, Copyright 2022. (c) Detection of the ability of PtBi-β-CD-Ce6 to produce O2 and 1O2. Reproduced from ref. 128 with permission from the American Chemical Society, Copyright 2022. (d) Confocal images of the nontreated NTHi biofilm and NTHi biofilm treated with the V2O5 NW@DPA@AuNP cascade nanozyme and ciprofloxacin, respectively, and stained with the BacTiter-Glo™ Microbial Cell Viability Assay. Reproduced from ref. 138 with permission from the Royal Society of Chemistry, Copyright 2022. | ||
Combining inorganic nanozymes with different catalytic activities or natural enzymes can enhance their antibacterial efficacy.136,137 To address the challenge of against nontypeable Haemophilus influenzae, a cascade nanozyme was designed. The cascade nanozyme consists of two parts of gold nanoparticles and vanadium pentoxide nanowires connected by dopamine, providing glucose oxidase-like activity and haloperoxidase-mimicking activity, respectively. Compared with vanadium pentoxide nanowires, the higher antibacterial efficacy of cascade nanozymes is due to the introduction and combination of dual catalytic functions, where glucose oxidase-like activity generates H2O2 and haloperoxidase-like activity converts H2O2 to the antiseptic ability.138 Cascade nanozymes can be established not only between artificial enzymes, but also between natural enzymes and artificial enzymes. A satisfactory anti-inflammation effect was exhibited by decorating ceria nanoparticles (CeO2) on the surface of Spirulina platensis via electrostatic interaction in ulcerative colitis. The combination of superoxide dismutase in biological systems and catalase in non-biological systems endows the cascade nanozymes with superior ROS elimination activity.139
Despite their promising antibacterial properties, the clinical application of inorganic nanozymes faces challenges related to their long-term stability, biocompatibility, and potential off-target toxicity.140 The development of novel inorganic nanozymes with improved properties and minimized toxicity will pave the way for new strategies in combating antibiotic-resistant bacterial infections and improving public health outcomes.
3.3 Drug delivery system
Inorganic nanozymes represent a highly promising platform for advanced drug delivery, particularly in oncology and complex disease treatment, owing to their catalytic plasticity enabling controlled therapeutic release and enhanced efficacy (Fig. 9).141,142 One of the key advantages of inorganic nanozymes in drug delivery is their ability to mimic natural enzymes and generate bioactive molecules such as reactive oxygen species (ROS) and catalyzing prodrug activation directly within the target site.143,144 This feature can be used to facilitate the release of therapeutic agents in a controlled and localized manner, thereby overcoming many challenges associated with conventional drug delivery systems, including poor solubility, lack of targeting specificity, and systemic toxicity.145 | ||
| Fig. 9 Drug delivery systems based on inorganic nanozymes. (a) In vitro cancer cell ferroptosis evaluation of Pt-MIL-101. (b) In vivo ferroptosis therapy of 4T1 tumor by Pt-MIL-101. Reproduced from ref. 146 with permission from Elsevier, Copyright 2022. | ||
Inorganic nanozymes can orchestrate therapeutic effects through catalytic cascades. For instance, Pt-engineered MIL-101 nanomedicine with a multi-step catalytic process was demonstrated. During the treatment process, the nanomedicine first catalyzes the consumption of nicotinamide adenine dinucleotide phosphate to produce O2 and H2O2. The generated H2O2 subsequently undergoes Fenton-like reactions with iron ions to produce highly toxic ˙OH, and the regeneration inhibition of antioxidant glutathione synergistically drives the death of tumor cells.146
Nanozyme-mediated prodrug activation is also a pivotal strategy for achieving site-specific drug synthesis and localized therapy.147 As the first prodrug used in the context of enzyme prodrug therapy, phosphate demonstrates strong efficacy and solubility advantages. Mammalian experiments have validated phosphate prodrug conversion mediated by the ceria nanozyme with phosphatase-like activity. The conversion of the phosphate prodrug into the parent chemotherapeutic can be mediated in the presence of ceria and reflected as complete release and high toxicity.148 Similarly, surface-functionalized zinc sulfide nanoparticles loaded with ruthenium-based transition metal complexes (TMCs) can kill tumor cells. The thiol ligands released from zinc sulfide nanoparticles act as nucleophiles, thus accelerating the catalytic oxidation of ruthenium. This process facilitates the uncaging of allyl-protected molecules, thereby activating inert prodrugs into chemotherapy agents.149
In addition, stimuli-responsive nanozyme platforms can also enhance spatiotemporal control. Zheng Nie et al. constructed a pH-sensitive nanoplatform based on iron oxide (Fe3O4) nanospheres with platinum nanozymes for drug delivery. In the acidic tumor microenvironment (pH = 6–6.5), the efficiency of 5-fluorouracil released from the nanoplatform can reach 77.8%. This study provides a superior prospect for simultaneous drug delivery to breast tumors.150 Hybrid systems further expand the functionality, where a doxorubicin delivery system based on poly(lactic acid-co-glycolic acid) grafted-Fe2O3 nanoparticles was reported to inhibit lung adenocarcinoma.151
4. Development of novel inorganic nanozymes
Inorganic nanozymes, capable of mimicking natural enzyme activities, have demonstrated broad utility across biomedicine, diagnostics, and environmental remediation.152 Despite this promise, realizing their full potential necessitates strategic optimization of their catalytic activities, stability, biocompatibility, and specificity for targeted applications. The engineering of advanced inorganic nanozymes has emerged as a transformative frontier in the field of nanotechnology.4.1 Strategic surface modification of inorganic nanozymes
Surface modification is pivotal in augmenting the performance and biocompatibility of inorganic nanozymes (Fig. 10).153 The surface properties of nanoparticles dictate their interactions with biomolecules (e.g., proteins, lipids, and nucleic acids), influencing their biological fate and function.154 Modifying their surface can not only improve their catalytic efficiency but also confer desirable properties, such as hydrophilicity, biocompatibility, and targeting ability. | ||
| Fig. 10 (a) In vivo therapeutic effect of core–shell-structured Cu2O@Mn3Cu3O8 nanozyme. Reproduced from ref. 156 with permission from Wiley, Copyright 2022. (b) US enhancement of MoS2 POD-like activity. Reproduced from ref. 158 with permission from Wiley, Copyright 2023. (c) Multimodal imaging of TMPAs in vivo. (d) Schematic of the establishment of 4T1 tumor xenografts and therapeutic outcome with TMPAs. Reproduced from ref. 159 with permission from Wiley, Copyright 2021. | ||
In the process of modifying their surface to optimize their performance, the assembly of a core–shell structure is one of the efficient means.155 Wei Chen et al. constructed a core–shell-structured Cu2O@Mn3Cu3O8 nanozyme to achieve efficient and safe cuproptosis. Mn3Cu3O8 as the shell can reduce the toxicity of the nuclear structure to normal tissue and show higher enzyme activity and biocompatibility at the same time. The enhanced enzyme-mimicking activity can be attributed to the better band continuity near the Fermi surface. The diverse enzyme activities (CAT-like, POD-like, and GSHOx-like activities) of nanozymes are attributed to the presence of the Mn2+/Mn3+/Mn4+ and Cu+/Cu2+ redox couples.156
The surface charge of nanoparticles also plays an essential role in their interaction with biological systems.157 Modifying their surface charge can enhance their cellular uptake, alter their biodistribution, and influence their stability. Cationic surfaces tend to interact more readily with negatively charged cellular membranes to enhance their internalization, while anionic nanoparticles might show reduced toxicity and better biocompatibility. Environmentally responsive charge switching further enables targeted applications. Polydopamine-modified copper oxide nanozymes change from negatively to positively charged in acidic microenvironments, activating their peroxidase-like activity and targeting function for negatively charged bacteria. Longwei Wang et al. proved that piezoelectricity-produced charges can enhance the POD-like activity of MoS2. The designed BTO/MoS2@CA cascade nanocatalyst was composed by exposing MoS2 nanosheets on the surface of piezoelectric tetragonal barium titanate and modifying them with cinnamaldehyde. The achievement of POD-like activity of MoS2 under ultrasound was first reported, which is mainly attributed to the decrease in the binding energy between MoS2 and H2O2, and the promoted decomposition of H2O2. The charge generated by ultrasonic separation piezoelectric tetragonal barium titanate will promote the combination of MoS2 and H2O2, resulting in the continuous generation of –OH to kill tumors.158
Coating inorganic nanozymes with biocompatible polymers (e.g., polyethylene glycol, polylactic acid, or polyvinyl alcohol) is beneficial to improve their stability in physiological environments and reduce their nonspecific interactions with proteins and cells, which offer advanced programmability.159 Baoxuan Huang et al. developed a pH-responsive supramolecular AuP-PS nanoplatform self-assembled from porphyrin-containing block copolymers and pillar[5]arene-capped AuNPs. The dense grafting of porphyrin-containing amphiphilic block copolymers on the surface of Au-PS is attributed to the reversible host–guest interactions. The formation of supramolecular complexes is conducive to improving the biosafety of AuP-PS. The dissociation of the two different nanoparticles under different pH conditions provides a programmable coordinated treatment scheme for tumors.160 In particular, PEGylation is a widely used strategy to enhance the solubility, circulation time, and biocompatibility of nanoparticles, making them less likely to be cleared by the immune system.
4.2 Multifunctionalization of inorganic nanozymes
Inorganic nanozymes exhibit diverse catalytic mechanisms, include peroxidase-, catalase-, and superoxide dismutase (SOD)-like activities, alongside more complex functionalities, such as esterase and phosphatase activities.161 Beyond singular catalytic activity, recent research focuses on engineering multifunctional nanozymes that integrate multiple enzyme-mimicking properties within a single nanostructure (Fig. 11).162 | ||
| Fig. 11 Multifunctionalization of inorganic nanozymes. (a) Thrombin-“switch-on”-induced multiple levels of targeting. Reproduced from ref. 161 with permission from Wiley, Copyright 2023. (b) In vivo antitumor performance of “three-in-one” nanozyme composite. Reproduced from ref. 163 with permission from Wiley, Copyright 2023. | ||
Their multifunctionality leverages the unique capacity to co-localize distinct catalytic activities, enhancing their synergistic effects and application potential. A bimetallic Au–Pt nanozyme with triple-enzyme-mimicking activities was synthesized by anchoring ultrasmall Au–Pt nanoparticles onto metastable Cu2O supports, followed by encapsulation within a Cu-MOF and surface modification with Pluronic F127 to enhance its overall hydrophilicity and biocompatibility. Nanozymes can achieve the following three-in-one functions simultaneously: (1) mimicking catalase to support O2; (2) mimicking oxidase to produce H2O2; and (3) mimicking peroxidase to generate ˙OH. During the process, the metastable Cu2O NPs acted as redox templates to anchor the Au–Pt nanozyme and as a copper source to realize the controllable growth of the MOF shell for preventing protein adsorption. The continuous catalytic process with multiple enzyme activities not only avoids the need for assembling multiple various natural enzymes, but also enhances the therapeutic effect on tumors, while reducing the side effects.163 Similarly, the Co2+-ZIF-67 metal–organic framework nanoparticles prepared by Yi Wu also exhibited three different activities of catalase-like, oxidase-like, and peroxidase-like activities. The catalase activity of the nanoparticles is reflected in the oxidative decomposition of H2O2. The catalytic oxidation of dopamine to aminochrome and NAD+ represents the oxidase activity. The peroxidase activity is reflected in the generation of polyaniline. Different from the previously reported method, the synthesis of nanozyme-driven imprinted particles is different. This method produced imprinted sites near the active catalytic sites of the nanozyme, thus effectively emulating the function of the active sites of natural enzymes.164 The arginine-rich peptide–Pt nanoparticle cluster (ARP-PtNC) nanozymes incorporate two enzymatic cascade systems. The effective degradation of uric acid and the removal of oxygen species can be achieved simultaneously in the ARP-PtNC nanozymes, where the former is due to the excellent uricase-like activity, and the latter is attributed to the two-cascade catalysis (superoxide dismutase/catalase and uricase/catalase). The prepared nanozymes show broad application prospects in gout and hyperuricemia.
At the same time, next-generation artificial enzyme cascade system5s based on a single nanozyme will continue to attract attention in the future.165 Furthermore, inorganic nanozymes can also maintain their activity under more extreme conditions, which makes them particularly useful for industrial and environmental applications. A three-component RGPT-PdNP@rGO nanozyme with highly selective and sensitive properties was developed by integrating an Arg-rich recognition peptide for the accurate detection of UA. The RGPT-PdNP@rGO nanozyme with improved UA recognition, sensing and degrading performances provide more comprehensive information about multifunctional nanozymes for precise biomarker recognition. This study proves that the peptide-based recognition can effectively and flexibly control the structure-dependent recognition ability of composite nanozymes through simulation of the pocket-like structure of natural enzymes.166
5. Challenges and prospects
Although inorganic nanozymes have shown remarkable promise in biomedicine, there are still numerous scientific and translational barriers. Addressing these challenges is paramount for realizing their clinical potential.(1) Limited catalytic precision in biological systems. How to further improve their catalytic efficiency, selectivity, and stability in complex environments is a key issue in current research. While offering advantages in stability, the catalytic efficiency and substrate selectivity of most inorganic nanozymes still significantly lag behind their natural counterparts in complex physiological environments. Non-specific ROS generation (common in peroxidase-mimics) causes off-target effects during anti-tumor therapy, while interference from biomolecules (e.g. glutathione and albumin) compromises the biosensing accuracy. This gap fundamentally limits their sensitivity in diagnostics and efficacy in therapeutics. The mechanisms underlying substrate recognition and specificity remain poorly understood for inorganic surfaces. Enhancing their activity under physiological pH, ionic strength, and in the presence of biological inhibitors is a persistent challenge.
(2) Synthetic reproducibility and scalability. Achieving batch-to-batch consistency during the synthesis of inorganic nanozymes remains a major barrier to their reliable performance evaluation and clinical translation. Minor variations in parameters (pH, temperature, precursor ratios, and reaction time) profoundly influence their critical physicochemical attributes (size, shape, crystallinity, surface defects, and ligand density), which are directly linked to their catalytic activity and stability. Also, scaling up their synthesis while maintaining precise control over these nanoscale features presents significant engineering challenges.
(3) Biocompatibility and long-term toxicity. Unlike natural enzymes, which are biocompatible and biodegradable, inorganic nanozymes are often composed of metals and metal oxides (e.g. Ag+ ions from silver nanozymes and Ce3+/Ce4+ redox cycling), which can raise concerns regarding their safety and potential toxicity in vivo. Current surface modification strategies (e.g. PEGylation) often sacrifice catalytic sites to improve safety. Consequently, ensuring the biocompatibility and reducing the toxicity of inorganic nanozymes are critical steps in their development for medical applications.
(4) Predictability and complexity of in vivo behavior. Lastly, the understanding of the biological behavior and pharmacokinetics of inorganic nanozymes in vivo is limited. Their performance in vivo is often unpredictable due to various factors such as biological fluid composition, organ distribution, and the presence of various biological barriers. For instance, the rapid adsorption of biomolecules alters their surface properties, potentially masking their catalytic sites, changing their targeting ability, and modulating their immune recognition. Their dynamic nature and composition-dependent impact on catalysis are poorly characterized. Additionally, targeted delivery to specific disease sites is hindered by physiological barriers. Passive accumulation strategies often lack efficiency and specificity. Comprehensive studies on the pharmacokinetics, biodistribution, and long-term safety of inorganic nanozymes are essential.
To overcome these challenges and fully realize the potential of inorganic nanozymes in biomedicine, future research should focus on the following.
(1) Rational design of next-generation nanozymes. Future efforts should prioritize the rational design of inorganic nanozymes with enhanced catalytic specificity and efficiency under physiological conditions. Focus should be on predictably boosting their catalytic efficiency and selectivity for biomedically relevant substrates. This will require integrating computational modeling and artificial intelligence (AI), such as molecular dynamics simulations, machine learning-based prediction of catalytic activity, and AI-driven material selection, to enable atomic-level engineering of active sites and enzyme-mimetic substrate. Special emphasis should be placed on mimicking natural enzyme kinetics and achieving predictable interactions with biomedical substrates.
(2) Advanced synthesis and surface engineering. The scalable and reproducible synthesis of inorganic nanozymes is essential for their clinical translation. Robust, scalable synthetic routes with real-time monitoring need to be developed to ensure batch uniformity. Implementing multifunctional surface coatings and stimuli-responsive polymers will simultaneously enhance their biocompatibility, reduce non-specific protein adsorption, enable active targeting, and provide environmental responsiveness.
(3) Integration with emerging technologies and bioinspired systems. Nanozyme research should increasingly intersect with other advancing fields, such as bioinspired engineering and wearable sensors. Emphasis on “smart” theranostic systems that operate in response to pathological stimuli (e.g., inflammation, hypoxia, or acidosis) will enhance the precision in applications such as targeted drug delivery and immunomodulation.
(4) Comprehensive in vivo evaluation frameworks. Establishing standardized protocols for the rigorous assessment of long-term biocompatibility, immunogenicity, and chronic toxicity in relevant disease models. Advanced imaging techniques need to integrated to spatiotemporally track the biodistribution, in situ catalytic activity, and clearance kinetics of nanozymes. This data is vital for predictive pharmacokinetic/pharmacodynamic modeling. At the same time, there is an urgent need to develop standardized protocols and reference materials for quantifying nanozyme activity across laboratories. Efforts should be directed toward establishing ISO-compliant assays. This will facilitate a direct comparison between studies and support regulatory approval.
(5) Bridging the gap with clinical translation. Research efforts should focus on overcoming the most critical translational barriers identified, such as GMP-compliant manufacturing, establishing robust quality control metrics, and designing clinically relevant efficacy and safety studies.
(6) Overcoming translational and regulatory barriers. A translational road map must be developed in collaboration with regulatory agencies to define a clear path from bench to bedside. Key activities include defining critical quality attributes for inorganic nanozyme-based products, designing pivotal pre-clinical and clinical trials that emphasize safety and clinically meaningful endpoints, and creating open-access databases for material safety and efficacy data.
In conclusion, by fostering collaboration among different fields, inorganic nanozymes can transition from bench-scale innovations to applications that redefine medicine. By addressing these multifaceted challenges through collaborative and standardized approaches, this field has the potential to develop into clinically approved frontier technology that can completely change the methods of diagnosis and treatment. As we unravel the complexities of inorganic nanozyme catalysis, these artificial enzymes may ultimately rival or even surpass the sophistication of nature's own catalysts.
Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analyzed as part of this review.Acknowledgements
The authors gratefully acknowledge the funding of this research offered by the Natural Science Foundation Project of Jilin Province (YDZJ202201ZYTS269, YDZJ202201ZYTS644, YDZJ202501ZYTS428); Science and Technology Innovation Development Program of Jilin City (20240103009); and National Natural Science Foundation of China (NSFC) (21805108). The authors acknowledge the assistance of JLICT Center of Analysis Characterization and Analysis.References
- L. Ma, J. Zheng, N. Zhou, R. Zhang, L. Fang, Y. Yang, X. Gao, C. Chen, X. Yan and K. Fan, Nat. Commun., 2024, 15, 233 Search PubMed.
- L. Zhang, H. Wang and X. Qu, Adv. Mater., 2024, 36, e2211147 Search PubMed.
- X. Liu, H. Xu, H. Peng, L. Wan, D. Di, Z. Qin, L. He, J. Lu, S. Wang and Q. Zhao, Coord. Chem. Rev., 2024, 502, 215610 Search PubMed.
- B. Xu, S. Li, L. Zheng, Y. Liu, A. Han, J. Zhang, Z. Huang, H. Xie, K. Fan, L. Gao and H. Liu, Adv. Mater., 2022, 34, e2107088 Search PubMed.
- F. Wang, Y. Wang, H. Wang, G. Zhao, J. Li and Y. Wang, Microchem. J., 2023, 191, 108817 Search PubMed.
- H. Wei, L. Gao, K. Fan, J. Liu, J. He, X. Qu, S. Dong, E. Wang and X. Yan, Nano Today, 2021, 40, 101269 Search PubMed.
- Q. Liu, A. Zhang, R. Wang, Q. Zhang and D. Cui, Nano-Micro Lett., 2021, 13, 154 Search PubMed.
- J. Razlivina, A. Dmitrenko and V. Vinogradov, J. Phys. Chem. Lett., 2024, 15, 5804–5813 Search PubMed.
- K. A. Abdullah, T. F. Tahir, A. F. Qader, R. A. Omer and K. A. Othman, J. Fluoresc., 2025, 35, 4973–4987 Search PubMed.
- Q. Tian, S. Li, Z. Tang, Z. Zhang, D. Du, X. Zhang, X. Niu and Y. Lin, Adv. Healthcare Mater., 2025, 14, 2401630 Search PubMed.
- A. Baranwal, S. A. Polash, V. K. Aralappanavar, B. K. Behera, V. Bansal and R. Shukla, Nanomaterials, 2024, 14, 244 Search PubMed.
- M. Kumawat, H. Madhyastha, A. Umapathi, M. Singh, N. Revaprasadu and H. K. Daima, Langmuir, 2022, 38, 1877–1887 Search PubMed.
- S. Li, Y. Zhang, Q. Wang, A. Lin and H. Wei, Anal. Chem., 2022, 94, 312–323 Search PubMed.
- X. Wang, X. Zhong, J. Li, Z. Liu and L. Cheng, Chem. Soc. Rev., 2021, 50, 8669–8742 Search PubMed.
- H. Jia, J. Gong, Z. Hu, T. Wen, C. Li, Y. Chen, J. Huang and W. He, Langmuir, 2024, 40, 19739–19750 Search PubMed.
- T. G. Halmagyi, L. Noureen, A. Szerlauth and I. Szilagyi, Dalton Trans., 2024, 53, 14132–14138 Search PubMed.
- W. Yang, X. Yang, L. Zhu, H. Chu, X. Li and W. Xu, Coord. Chem. Rev., 2021, 448, 214170 Search PubMed.
- Z. Liu, R. Wu, Y. Jiang, L. Qi, J. Liu, M. Du, J. Wang, L. Liu, G. Feng and L. Zhang, Colloids Surf., B, 2024, 233, 113644 Search PubMed.
- S. Liang, X. Wu, M. Zong and W. Lou, J. Colloid Interface Sci., 2022, 622, 860–870 Search PubMed.
- M. Perwez, S. Y. Lau, D. Hussain, S. Anboo, M. Arshad and P. Thakur, Colloids Surf., B, 2023, 225, 113241 Search PubMed.
- X. Huang, New J. Chem., 2022, 46, 15273–15291 Search PubMed.
- X. Yuan, J. Xiong, X. Wu, N. Ta, S. Liu, Z. Li and W. Lou, Chem. Eng. J., 2024, 480, 148246 Search PubMed.
- B. Cakiroglu, Mikrochim. Acta, 2024, 191, 268 Search PubMed.
- Z. Ge, Y. Zhao, J. Li, Z. Si, W. Du and H. Su, Nanoscale, 2024, 16, 2608–2620 Search PubMed.
- K. Fan, Y. Lin and V. Bansal, Front. Chem., 2021, 9, 670767 Search PubMed.
- Q. Li, M. Dong, Q. Han, Y. Zhang, D. Yang, D. Wei and Y. Yang, J. Controlled Release, 2024, 365, 905–918 Search PubMed.
- H. Liu, B. Yu, C. Zhou, Z. Deng, H. Wang, X. Zhang and K. Wang, Mater. Today Bio, 2024, 27, 101137 Search PubMed.
- X. Han, B. Li, W. Wang, B. Feng, Q. Tang, Y. Qi, R. Zhao, W. Qiu, S. Zhao, Z. Pan, X. Guo, H. Du, J. Qiu, H. Liu, G. Li and H. Xue, ACS Nano, 2024, 18, 19836–19853 Search PubMed.
- M. Liu, H. Yu, T. Zhao and X. Li, Dalton Trans., 2023, 52, 15203–15215 Search PubMed.
- H. Fan, R. Zhang, K. Fan, L. Gao and X. Yan, ACS Nano, 2024, 18, 2533–2540 Search PubMed.
- J. Mujtaba, J. Liu, K. K. Dey, T. Li, R. Chakraborty, K. Xu, D. Makarov, R. A. Barmin, D. A. Gorin, V. P. Tolstoy, G. Huang, A. A. Solovev and Y. Mei, Adv. Mater., 2021, 33, e2007465 Search PubMed.
- L. Zhang, C. Zhu, R. Huang, Y. Ding, C. Ruan and X. Shen, Front. Chem., 2021, 9, 630969 Search PubMed.
- P. Wang, D. Min, G. Chen, M. Li, L. Tong and Y. Cao, Front. Chem., 2021, 9, 773285 Search PubMed.
- M. Zandieh and J. Liu, Adv. Mater., 2024, 36, e2211041 Search PubMed.
- Y. Gao, X. Gao, H. Hou, Z. Qu, H. Li, B. Du and P. Tang, Sens. Actuators, B, 2024, 404, 135280 Search PubMed.
- W. Xie, Y. Gan, L. Wang, Y. Si, Q. Li, T. Song, P. Wei, Z. Wu and G. Zhang, Small, 2024, 20, e2306446 Search PubMed.
- H. Zhu, P. Liu, L. Xu, X. Li, P. Hu, B. Liu, J. Pan, F. Yang and X. Niu, Biosensors, 2021, 11, 11100382 Search PubMed.
- C. P. Kurup and M. U. Ahmed, Biosensors, 2023, 13, 13040461 Search PubMed.
- X. Li, H. Zhu, P. Liu, M. Wang, J. Pan, F. Qiu, L. Ni and X. Niu, TrAC, Trends Anal. Chem., 2021, 143, 116379 Search PubMed.
- L. Li, D. Wang, L. Ren, T. Wang, X. Tan, F. Cui, T. Li and J. Li, Int. J. Biol. Macromol., 2024, 258, 129098 Search PubMed.
- N. Tao, S. Chen, S. Mahdinloo, Q. Zhang, T. Lan, Q. Saiding, S. Chen, Y. Xiong, W. Tao and J. Ouyang, Nano Today, 2024, 57, 102371 Search PubMed.
- R. Zhang, X. Yan and K. Fan, Acc. Mater. Res., 2021, 2, 534–547 Search PubMed.
- Z. Mohamad and J. Liu, ACS Nano, 2021, 15, 15645–15655 Search PubMed.
- A. Moradi Hasan-Abad, A. Shabankare, A. Atapour, G. A. Hamidi, M. Salami Zavareh and A. Sobhani-Nasab, Front. Pharmacol., 2024, 15, 1339580 Search PubMed.
- X. Ren, D. Chen, Y. Wang, H. Li, Y. Zhang, H. Chen, X. Li and M. Huo, J. Nanobiotechnol., 2022, 20, 92 Search PubMed.
- J. Shen, J. Chen, Y. Qian, X. Wang, D. Wang, H. Pan and Y. Wang, Adv. Mater., 2024, 36, 2313406 Search PubMed.
- A. A. P. Mansur, A. G. Leonel, K. Krambrock and H. S. Mansur, Catal. Today, 2022, 397–399, 129–144 Search PubMed.
- L. Wang, L. Zhao, X. Xiao, X. Wang, J. Cao, H. Ye, H. Li and Y. Zhang, Sep. Purif. Technol., 2025, 359, 130551 Search PubMed.
- X. Gao, H. Chen, H. Qiu, Y. Zhang, J. Cheng and Y. Shen, J. Hazard. Mater., 2024, 469, 133871 Search PubMed.
- Q. Diao, X. Chen, Z. Tang, S. Li, Q. Tian, Z. Bu, H. Liu, J. Liu and X. Niu, Environ. Sci.: Nano, 2024, 11, 766–796 Search PubMed.
- Y. Zhou, Y. Li, Y. Fei, M. Zhang, S. Wang, F. Li and X. Bao, Front. Chem., 2021, 9, 681566 Search PubMed.
- R. Xu, S. Zhang, P. Wang, R. Zhang, P. Lin, Y. Wang, L. Gao, H. Wei, X. Zhang, D. Ling, X. Yan and K. Fan, Coord. Chem. Rev., 2024, 501, 215519 Search PubMed.
- C. Qiao, X. Wang, Y. Gao, Y. Ma, H. Luo, S. Zhang, D. Huo and C. Hou, ACS Appl. Nano Mater., 2024, 7, 9375–9387 Search PubMed.
- Q. Shang, L. Zhang, C. Chen, W. Tang, M. Han, Q. Chen and W. Liu, ACS Appl. Nano Mater., 2022, 5, 2678–2687 Search PubMed.
- C. N. Zhu, X. Chen, Y. Q. Xu, F. Wang, D. Y. Zheng, C. Liu, X. H. Zhang, Y. Yi and D. B. Cheng, ACS Biomater. Sci. Eng., 2024, 10, 7352–7371 Search PubMed.
- C. Wang, H. Wang, B. Xu and H. Liu, View, 2020, 2, 20200045 Search PubMed.
- S. Jimenez-Falcao, J. M. Méndez-Arriaga, V. García-Almodóvar, A. A. García-Valdivia and S. Gómez-Ruiz, Catalysts, 2022, 13, 13010013 Search PubMed.
- A. A. P. Mansur and H. S. Mansur, J. Nanotheranostics, 2025, 6, 4 Search PubMed.
- F. Wei and X. Han, Chin. J. Anal. Chem., 2021, 49, 581–592 Search PubMed.
- Y. Ai, Z. Hu, X. Liang, H. Sun, H. Xin and Q. Liang, Adv. Funct. Mater., 2021, 32, 202110432 Search PubMed.
- L. Tang, S. Wang, J. Hu, L. Meng, J. Zhang, Y. Chang, X. Ma and Y. Guo, Colloids Surf., B, 2025, 245, 114188 Search PubMed.
- B. Gao, Y. Ding, Z. Cai, S. Wu, J. Wang, N. Ling, Q. Ye, M. Chen, Y. Zhang, X. Wei, Y. Ye and Q. Wu, Microchim. Acta, 2024, 191, 438 Search PubMed.
- C. Cheng, H. Wang, J. Zhao, Y. Wang, G. Zhao, Y. Zhang, X. Liu and Y. Wang, Colloids Surf., B, 2024, 235, 113767 Search PubMed.
- S. Maddheshiya and S. Nara, Front. Bioeng. Biotechnol., 2022, 10, 880214 Search PubMed.
- A. Sen, J. Oswalia, S. Yadav, M. Vachher and A. Nigam, Curr. Res. Biotechnol., 2024, 7, 100205 Search PubMed.
- J. Li, X. Cai, Y. Zhang, K. Li, L. Guan, Y. Li, T. Wang and T. Sun, ChemistrySelect, 2022, 7, 202201127 Search PubMed.
- N. A. Omar and H. S. Jabbar, Inorg. Chem. Commun., 2022, 146, 110104 Search PubMed.
- J. Li, S. Wei, N. Wang, M. Yang, X. Xu, J. Tang and Z. Li, Talanta, 2024, 279, 126603 Search PubMed.
- R. Li, C. Yang and Z. Li, New J. Chem., 2025, 49, 4682–4691 Search PubMed.
- J. Huo, J. Hao, J. Mu and Y. Wang, ACS Appl. Bio Mater., 2021, 4, 3443–3452 Search PubMed.
- X. Huang, S. Zhang, Y. Tang, X. Zhang, Y. Bai and H. Pang, Coord. Chem. Rev., 2021, 449, 214216 Search PubMed.
- X. Zhang, S. Lin, S. Liu, X. Tan, Y. Dai and F. Xia, Coord. Chem. Rev., 2021, 429, 213652 Search PubMed.
- Y. Yi, X. Zhou, D. Liao, J. Hou, H. Liu and G. Zhu, Inorg. Chem., 2023, 62, 13929–13936 Search PubMed.
- Y. Tian, Z. Ye, X. Wang, H. Guan, W. Liu, X. Duan, Y. Liu, G. Zeng and H. Liu, Adv. Healthcare Mater., 2024, e2401574 Search PubMed.
- X. Liu, P. An, Y. Han, H. Meng and X. Zhang, Microchem. J., 2024, 201, 110559 Search PubMed.
- A. Ali, M. Ovais, H. Zhou, Y. Rui and C. Chen, Biomaterials, 2021, 275, 120951 Search PubMed.
- M. K. Sarangi, L. D. Patel, G. Rath, S. S. Nanda and D. K. Yi, Chin. Chem. Lett., 2024, 35, 109381 Search PubMed.
- Z. Zhang, Y. Li, Z. Yuan, L. Wu, J. Ma, W. Tan, Y. Sun, G. Zhang and H. Chai, Inorg. Chem. Front., 2025, 12, 400–429 Search PubMed.
- B. Li, D. Xu, Y. Chen, W. Li, H. Liu, A. A. Ansari and R. Lv, ACS Appl. Nano Mater., 2024, 7, 9428–9440 Search PubMed.
- X. Xu, M. Ma, J. Gao, T. Sun, Y. Guo, D. Feng and L. Zhang, Inorg. Chem., 2024, 63, 1225–1235 Search PubMed.
- R. Wang, B. Guan, W. Wang, H. Yan, J. Jin, Y. Yang, L. Li, L. Guo and Y. Zeng, Sens. Actuators, B, 2025, 427, 137178 Search PubMed.
- H. Guan, B. Han, B. Peng, H. Ye, Q. Zhang and D. Wang, Microchem. J., 2023, 194, 109311 Search PubMed.
- G. Zhu, P. Zheng, M. Wang, W. Chenb and C. Li, Inorg. Chem. Front., 2022, 9, 1006–1015 Search PubMed.
- J. Xia, Z. Li, Y. Ding, L. A. Shah, H. Zhao, D. Ye and J. Zhang, Anal. Chem., 2024, 96, 8221–8233 Search PubMed.
- Z. Wang, R. Wang, Z. Geng, X. Luo, J. Jia, S. Pang, X. Fan, M. Bilal and J. Cui, Crit. Rev. Biotechnol., 2024, 44, 674–697 Search PubMed.
- Y. Wu, Y. Tang, W. Xu, R. Su, Y. Qin, L. Jiao, H. Wang, X. Cui, L. Zheng, C. Wang, L. Hu, W. Gu, D. Du, Y. Lin and C. Zhu, Small, 2023, 19, e2302929 Search PubMed.
- L. Jiang, Y. Han, S. Fernández-García, M. Tinoco, Z. Li, P. Nan, J. Sun, J. J. Delgado, H. Pan, G. Blanco, J. Martínez-López, A. B. Hungría, J. J. Calvino and X. Chen, Appl. Surf. Sci., 2023, 610, 155502 Search PubMed.
- L. Tu, S. Chen, Z. Yuan, Y. Xiong, B. Luo, Y. Chen, Z. Hou, S. Ke, N. Lin, C. Li and S. Ye, J. Colloid Interface Sci., 2024, 663, 810–824 Search PubMed.
- T. Chen, Y. Wang, K. Wang, M. Dai, Y. Duan, C. Mao and M. Wan, Mater. Chem. Front., 2024, 8, 3383–3412 Search PubMed.
- X. Zeng, S. Yan and B. Liu, Microporous Mesoporous Mater., 2022, 335, 111826 Search PubMed.
- Q. Ye, E. Yuan, J. Shen, M. Ye, Q. Xu, X. Hu, Y. Shu and H. Pang, Adv. Sci., 2023, 10, e2304149 Search PubMed.
- Y. Huang, J. Ren and X. Qu, Chem. Rev., 2019, 119, 4357–4412 Search PubMed.
- X. Shen, Z. Wang, X. Gao and X. Gao, Adv. Mater., 2024, 36, e2211151 Search PubMed.
- H. Dong, W. Du, J. Dong, R. Che, F. Kong, W. Cheng, M. Ma, N. Gu and Y. Zhang, Nat. Commun., 2022, 13, 5365 Search PubMed.
- Y. Song, C. Huang and Y. Li, Inorg. Chem., 2023, 62, 12697–12707 Search PubMed.
- Y. Wang, X. Zhang, Y. Dai and F. Xia, Chem. Eng. J., 2023, 455, 140811 Search PubMed.
- Z. Xin, K. Duan, Q. Zhuo, Q. He, X. Zhang, C. Zheng, X. Han, T. Han, Z. Fu, X. Xu and X. Zhao, Chem. Eng. J., 2023, 461, 141589 Search PubMed.
- Y. Liu, N. Li, K. Su, J. Du and R. Guo, Inorg. Chem., 2024, 63, 13602–13612 Search PubMed.
- Z. Tong, Y. Gao, H. Yang, W. Wang and Z. Mao, View, 2021, 2, 20200133 Search PubMed.
- T. Hu, Q. Liu, Z. Zhou, W. Zhao, H. Huang, F. Meng, W. Liu, Q. Zhang, L. Gu, R. Liang and C. Tan, Small, 2022, 18, e2200595 Search PubMed.
- J. Sheng, Y. Wu, H. Ding, K. Feng, Y. Shen, Y. Zhang and N. Gu, Adv. Mater., 2024, 36, e2211210 Search PubMed.
- V. Baldim, F. Bedioui, N. Mignet, I. Margaill and J. F. Berret, Nanoscale, 2018, 10, 6971–6980 Search PubMed.
- Z. Xi, W. Gao and X. Xia, ChemBioChem, 2020, 21, 2440–2444 Search PubMed.
- A. Sahu, K. Min, S. H. Jeon, K. Kwon and G. Tae, Carbohydr. Polym., 2023, 316, 121088 Search PubMed.
- S. Singh, Int. J. Biol. Macromol., 2024, 260, 129374 Search PubMed.
- Y. Ai, H. Sun, C. Wang, W. Zheng, Q. Han and Q. Liang, Small, 2022, 18, e2105304 Search PubMed.
- N. K. Dega, A. B. Ganganboina, H. L. Tran, E. P. Kuncoro and R. A. Doong, Talanta, 2022, 237, 122957 Search PubMed.
- Y. Li, IOP Conf. Ser.: Earth Environ. Sci., 2021, 804, 042018 Search PubMed.
- T. Sahare, N. Singh, B. N. Sahoo and A. Joshi, Biosens. Bioelectron.: X, 2024, 21, 100544 Search PubMed.
- B. Shen, L. Yang, H. Xu, Y. Zhang, D. Ming, L. Zhu, Y. Wang and L. Jiang, J. Colloid Interface Sci., 2023, 650, 211–221 Search PubMed.
- C. Lou, F. Yang, L. Zhu, Q. Sun, Y. Yang and J. Guo, Colloids Surf., A, 2023, 677, 132398 Search PubMed.
- G. Song, J. Li, Z. Majid, W. Xu, X. He, Z. Yao, Y. Luo, K. Huang and N. Cheng, Food Chem., 2022, 390, 133127 Search PubMed.
- Q. Li, S. Zhan, X. Yang, Z. Zhang, N. Sun, X. Wang, J. Kang, R. Du, X. Hong, M. Yue, X. Li, Y. Tang, G. Liu, Y. Liu and D. Liu, ACS Nano, 2024, 18, 16113–16125 Search PubMed.
- Z. Wang, Y. Shao, Z. Zhu, J. Wang, X. Gao, J. Xie, Y. Wang, Q. Wu, Y. Shen and Y. Ding, Coord. Chem. Rev., 2023, 495, 215369 Search PubMed.
- Q. Zhang, L. Song and K. Zhang, Mater. Chem. Front., 2023, 7, 44–64 Search PubMed.
- M. He, X. Xu, H. Wang, Q. Wu, L. Zhang, D. Zhou, Y. Tong, X. Su and H. Liu, Small, 2023, 19, e2208167 Search PubMed.
- S. Khan, M. M. N. Babadaei, A. Hasan, Z. Edis, F. Attar, R. Siddique, Q. Bai, M. Sharifi and M. Falahati, J. Adv. Res., 2021, 33, 227–239 Search PubMed.
- Z. Wang, M. Li, H. Bu, D. S. Zia, P. Dai and J. Liu, Mater. Chem. Front., 2023, 7, 3625–3640 Search PubMed.
- S. Ali, S. Sikdar, S. Basak, M. S. Haydar, K. Mallick, M. Mondal, D. Roy, S. Ghosh, S. Sahu, P. Paul and M. N. Roy, Inorg. Chem., 2023, 62, 11291–11303 Search PubMed.
- B. Hong, T. Qin, W. Wang, L. Luo, Y. Li, Y. Ma and J. Wang, Sens. Actuators, B, 2025, 426, 137097 Search PubMed.
- L. Wang, Y. Chu, B. Cao, R. Zhang, Z. Hussain and Q. Liu, Talanta, 2025, 281, 126889 Search PubMed.
- M. Luo, F. Zhao, Y. Wang and J. Bian, J. Transl. Med., 2024, 22, 814 Search PubMed.
- H. Wang, B. Wang, J. Jiang, Y. Wu, A. Song, X. Wang, C. Yao, H. Dai, J. Xu, Y. Zhang, Q. Ma, F. Xu, R. Li and C. Wang, Molecules, 2022, 27, 27238552 Search PubMed.
- A. A. P. Mansur, H. S. Mansur and S. M. Carvalho, Catal. Today, 2022, 388–389, 187–198 Search PubMed.
- W. He, Z. Zhang and X. Sha, Biomaterials, 2021, 277, 121111 Search PubMed.
- X. Qin, J. Xiao, H. Li, H. Huang, H. Jin, Y. Zhang, G. Tian, G. Wang and G. Zhang, Inorg. Chem. Front., 2023, 10, 7176–7186 Search PubMed.
- N. Feng, Q. Li, Q. Bai, S. Xu, J. Shi, B. Liu and J. Guo, J. Colloid Interface Sci., 2022, 618, 68–77 Search PubMed.
- Y. Xie, M. Wang, Q. Sun, D. Wang, S. Luo and C. Li, Inorg. Chem., 2022, 61, 6852–6860 Search PubMed.
- Y. Yao, Z. Wang, Q. Cao, H. Li, S. Ge, J. Liu, P. Sun, Z. Liu, Y. Wu, W. Wang and J. Liu, ACS Appl. Mater. Interfaces, 2022, 14, 17153–17163 Search PubMed.
- L. Xie, H. Wu, Y. Li, L. Shi and Y. Liu, Adv. Healthcare Mater., 2024, e2402659 Search PubMed.
- Y. Zhang, F. Lei, W. Qian, C. Zhang, Q. Wang, C. Liu, H. Ji, Z. Liu and F. Wang, J. Controlled Release, 2024, 373, 929–951 Search PubMed.
- M. Zhang and W. Tong, Biomater. Sci., 2023, 11, 5769–5780 Search PubMed.
- S. Ahmadi, K. Rahimizadeh, A. Shafiee, N. Rabiee and S. Iravani, Process Biochem., 2023, 131, 154–174 Search PubMed.
- B. Unnikrishnan, C. W. Lien, H. Chu and C. Huang, J. Hazard. Mater., 2021, 401, 123397 Search PubMed.
- X. He, Y. Lv, Y. Lin, H. Yu, Y. Zhang, Y. Tong and C. Zhang, Adv. Mater., 2024, 36, e2400366 Search PubMed.
- N. Singh, G. R. Sherin and G. Mugesh, Angew. Chem., Int. Ed., 2023, 62, e202301232 Search PubMed.
- Q. Wang, J. Jiang and L. Gao, Biomed. Mater., 2021, 16, 042002 Search PubMed.
- X. Ma, J. Lang, P. Chen, W. Tang, S. Shindler and R. Yang, Nanoscale, 2023, 15, 1014–1023 Search PubMed.
- Q. Chen, M. Cao, J. Qiao, Q. Li and X. Zhang, Nanoscale Horiz., 2023, 8, 489–498 Search PubMed.
- Z. Sun, J. Yao, J. Wang, R. Huang, X. Liu, F. Li, X. Jiang and W. Chen, Inorg. Chem., 2022, 61, 1169–1177 Search PubMed.
- G. Luo, J. Zhang, Y. Sun, Y. Wang, H. Wang, B. Cheng, Q. Shu and X. Fang, Nano-Micro Lett., 2021, 13, 88 Search PubMed.
- D. Jiang, D. Ni, Z. T. Rosenkrans, P. Huang, X. Yan and W. Cai, Chem. Soc. Rev., 2019, 48, 3683–3704 Search PubMed.
- M. Kumawat, H. Madhyastha, M. Singh, D. Jain and H. K. Daima, Colloids Surf., A, 2022, 650, 129294 Search PubMed.
- M. Kumawat, A. Umapathi, E. Lichtfouse and H. K. Daima, Environ. Chem. Lett., 2021, 19, 3951–3957 Search PubMed.
- H. Li, Z. Liu, P. Zhang and D. Zhang, Drug Delivery, 2024, 31, 2417986 Search PubMed.
- C. Wu, D. Xu, M. Ge, J. Luo, L. Chen, Z. Chen, Y. You, Y. Zhu, H. Lin and J. Shi, Nano Today, 2022, 46, 101574 Search PubMed.
- Y. Wu, W. Chen, C. Wang and D. Xing, Chin. Chem. Lett., 2024, 35, 109096 Search PubMed.
- L. Li, D. Wang, L. Ren, T. Wang, X. Tan, F. Cui, T. Li and J. Li, Int. J. Biol. Macromol., 2024, 258, 129098 Search PubMed.
- X. Zhang, S. Lin, R. Huang, A. Gupta, S. Fedeli, R. Cao-Milan, D. C. Luther, Y. Liu, M. Jiang, G. Li, B. Rondon, H. Wei and V. M. Rotello, J. Am. Chem. Soc., 2022, 144, 12893–12900 Search PubMed.
- Z. Nie, Y. Vahdani, W. C. Cho, S. H. Bloukh, Z. Edis, S. Haghighat, M. Falahati, R. Kheradmandi, L. A. Jaragh-Alhadad and M. Sharifi, Arabian J. Chem., 2022, 15, 103966 Search PubMed.
- M. Cui, H. Zhang, F. Ouyang, Y. Guo, R. Li, S. Duan, T. Xiong, Y. Wang and X. Wang, J. Biomed. Nanotechnol., 2021, 17, 1007–1019 Search PubMed.
- A. Jouyban and R. Amini, Microchem. J., 2021, 164, 105970 Search PubMed.
- G. Wei, S. Liu, Y. Peng and H. Wei, Chin. J. Chem., 2024, 42, 1515–1522 Search PubMed.
- Y. Li and J. Liu, Mater. Horiz., 2021, 8, 336–350 Search PubMed.
- X. Zhu, N. Xu, L. Zhang, D. Wang and P. Zhang, Eur. J. Med. Chem., 2022, 238, 114456 Search PubMed.
- W. Chen, W. Xie, Z. Gao, C. Lin, M. Tan, Y. Zhang and Z. Hou, Adv. Sci., 2023, 10, e2303694 Search PubMed.
- Z. Wang, Y. Shao, Z. Zhu, J. Wang, X. Gao, J. Xie, Y. Wang, Q. Wu, Y. Shen and Y. Ding, Coord. Chem. Rev., 2023, 495, 215369 Search PubMed.
- L. Wang, X. Zhang, Z. You, Z. Yang, M. Guo, J. Guo, H. Liu, X. Zhang, Z. Wang, A. Wang, Y. Lv, J. Zhang, X. Yu, J. Liu and C. Chen, Angew. Chem., Int. Ed., 2023, 62, e202217448 Search PubMed.
- Z. Ma, M. F. Foda, H. Liang, Y. Zhao and H. Han, Adv. Funct. Mater., 2021, 31, 202103765 Search PubMed.
- B. Huang, J. Tian, Z. Cui, S. Weng, W. Wang, X. Jiang and W. Zhang, Chem. Eng. J., 2022, 444, 136164 Search PubMed.
- Z. Wang, Y. Zhao, Y. Hou, G. Tang, R. Zhang, Y. Yang, X. Yan and K. Fan, Adv. Mater., 2024, 36, e2210144 Search PubMed.
- H. Yang, B. Xu, S. Li, Q. Wu, M. Lu, A. Han and H. Liu, Small, 2021, 17, e2007090 Search PubMed.
- Y. Cheng, Y. Xia, Y. Sun, Y. Wang and X. Yin, Adv. Mater., 2024, 36, e2308033 Search PubMed.
- Y. Wu, Y. Qin, M. Muppidathi, R. Carmieli, M. Fadeev, W. Lei, M. Xia and I. Willner, Adv. Funct. Mater., 2023, 34, 202306929 Search PubMed.
- Y. Liu, Y. Qin, Q. Zhang, W. Zou, L. Jin and R. Guo, J. Colloid Interface Sci., 2021, 600, 37–48 Search PubMed.
- Y. Liu, N. Li, H. Zhang, D. Wei and R. Guo, Sens. Actuators, B, 2025, 423, 136784 Search PubMed.
| This journal is © The Royal Society of Chemistry 2025 |




