Direct X-ray detectors made of zero-dimensional hybrid metal halide perovskite single crystals†
Guangya
Zheng
a,
Haodi
Wu
 *abd,
Zhiwu
Dong
a,
Tong
Jin
a,
Jincong
Pang
a,
Yujue
Liu
a,
Zhiping
Zheng
*abd,
Zhiwu
Dong
a,
Tong
Jin
a,
Jincong
Pang
a,
Yujue
Liu
a,
Zhiping
Zheng
 a,
Guangda
Niu
a,
Guangda
Niu
 acd,
Ling
Xu
acd,
Ling
Xu
 ad and
Jiang
Tang
ad and
Jiang
Tang
 *acd
*acd
aWuhan National Laboratory for Optoelectronics and School of Optical and Electronic Information, Huazhong University of Science and Technology, Wuhan 430074, China. E-mail: whd18501@163.com; xuling@mail.hust.edu.cn
bResearch Center for Medical Artificial Intelligence, Shenzhen Institute of Advanced Technology, Chinese Academy of Sciences, Shenzhen, Guangdong 518055, China
cOptics Valley Laboratory, Wuhan 430074, China
dEzhou Industrial Technology Research Institute, Huazhong University of Science and Technology, Ezhou, 436000, P. R. China. E-mail: jtang@mail.hust.edu.cn
First published on 30th March 2024
Abstract
Zero-dimensional (0D) cadmium(Cd)-based A2BX4-type hybrid metal halide perovskites incorporating large sterically hindered cations define a new type of structure, which shows good performance in photoelectric detection because of their unique charge transmission. Herein, we report the synthesis of novel 0D (C19H18P)2CdCl4 single crystals (SCs) via a solution natural-volatilization method. Bulk (C19H18P)2CdCl4 SCs have sizes of 6 × 5 × 3 mm3. The as-synthesized SCs of (C19H18P)2CdCl4 exhibit a direct bandgap of 4.19 eV, possess a high resistivity of 3.73 × 1011 Ω cm, and demonstrate a low stable dark current. The crystal showcases an observed mobility-lifetime (μτ) product on a scale of 10−4 cm2 V−1. Furthermore, an X-ray photoconductor was successfully fabricated based on the SC of (C19H18P)2CdCl4, which displays an outstanding sensitivity of ∼143.6 μC Gyair−1 cm−2 when biased at 1000 V (333 V mm−1), a response current with exceptional stability, and a stable baseline with the lowest dark current drift of 7.34 × 10−4 pA cm−1 s−1 V−1. This work introduces a groundbreaking approach using single-crystal metal halide perovskites, opening up new possibilities for exploring optoelectronic applications, which holds the potential to unveil a novel generation of materials for X-ray detection and imaging.
1. Introduction
X-ray detectors find extensive use in medical imaging, security scrutiny, and nondestructive inspection.1–9 Semiconductor-based direct X-ray detectors are acknowledged as the upcoming technology for X-ray detection, mainly due to their high sensitivity, high spatial resolution, and simple device configuration.3,5,10–18 The common semiconductors employed for X-ray detection include α-Se (amorphous selenium), silicon, and CdTe (polycrystalline cadmium telluride).14–16 These materials face challenges such as low carrier μτ product, low X-ray absorption coefficient, and high fabrication cost, which limit their application in X-ray detectors.Recently, metal halide perovskites have shown great promise for X-ray detection applications because of their exceptional ability to attenuate X-rays, high μτ products, and the convenience of solution-based fabrication methods.6,19–22 Huang et al. found that X-ray detectors made of CH3NH3PbBr3 SCs demonstrate a remarkable sensitivity of 21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 μC Gyair−1 cm−2 and an impressive detection limit as low as 38 nGyair s−1.1 Gao et al. achieved synergistic strain engineering in 3D perovskites and significantly enhanced the stability of X-ray detectors.20 The structural stability of 0D analogues is significantly higher than that of 3D perovskite SCs, with reduced ionic migration, enhanced self-trapping, and anisotropic charge transport.23–25 Wu et al. used first-principles calculations to reveal that the ion migration of the perovskite SCs is ranked in the order of 0D MA3Bi2I9 < 2D (PEA)2PbI4 < 3D MAPbI3, which well explained the huge difference in dark current drift values of the 0D MA3Bi2I9 SC (5.0 × 10−10 nA cm−1 s−1 V−1), 2D (PEA)2PbI4 SC (1.9 × 10−8 nA cm−1 s−1 V−1) and 3D MAPbI3 SC (2.0 × 10−3 nA cm−1 s−1 V−1).26 In view of this, we hypothesized that the disconnection of inorganic octahedral entities within hybrid perovskite structures could effectively disrupt ion migration pathways. The 0D structure, characterized by the disconnection of inorganic octahedral (or some other configuration) units in all directions, facilitates this objective. Additionally, 0D compounds display electronic structures that are more localized, resulting in a decrease in the concentration of dark carriers when compared to their 3D or 2D equivalents.27,28 By reducing ion migration suppression and decreasing dark carrier concentration, we can achieve the desired X-ray detector.
000 μC Gyair−1 cm−2 and an impressive detection limit as low as 38 nGyair s−1.1 Gao et al. achieved synergistic strain engineering in 3D perovskites and significantly enhanced the stability of X-ray detectors.20 The structural stability of 0D analogues is significantly higher than that of 3D perovskite SCs, with reduced ionic migration, enhanced self-trapping, and anisotropic charge transport.23–25 Wu et al. used first-principles calculations to reveal that the ion migration of the perovskite SCs is ranked in the order of 0D MA3Bi2I9 < 2D (PEA)2PbI4 < 3D MAPbI3, which well explained the huge difference in dark current drift values of the 0D MA3Bi2I9 SC (5.0 × 10−10 nA cm−1 s−1 V−1), 2D (PEA)2PbI4 SC (1.9 × 10−8 nA cm−1 s−1 V−1) and 3D MAPbI3 SC (2.0 × 10−3 nA cm−1 s−1 V−1).26 In view of this, we hypothesized that the disconnection of inorganic octahedral entities within hybrid perovskite structures could effectively disrupt ion migration pathways. The 0D structure, characterized by the disconnection of inorganic octahedral (or some other configuration) units in all directions, facilitates this objective. Additionally, 0D compounds display electronic structures that are more localized, resulting in a decrease in the concentration of dark carriers when compared to their 3D or 2D equivalents.27,28 By reducing ion migration suppression and decreasing dark carrier concentration, we can achieve the desired X-ray detector.
Although lead halide perovskites have made considerable advancements, their practical application remains constrained due to the utilization of hazardous lead and the well-known characteristics of ion migration. Hence, it is imperative to research a new type of metal halide perovskite for further X-ray-detection applications.2,8,15,26 Similar to the hybrid lead halide perovskites, cadmium halide perovskites have a tendency to form anionic structures with low dimensions based on either a basic tetrahedron [CdX4] or octahedron [CdX6].29–33 However, the majority of research studies have primarily focused on the regulation of structures by utilizing organic cations’ directional functions to achieve a wide range of structural types. These include 0D [CdX4]2−, 0D [CdX6]4−, 1D [CdX3]−, 1D [Cd3X8]2−, 1D [Cd4X12]4− ribbons and 2D [Cd3Cl10]4− layers.31,34–37 In contrast to the extensive focus on structural characterizations, there is relatively limited research emphasis on high-energy ray detection, particularly in the area of X-ray detection. The inadequacy prompts us to explore a proficient synthetic approach for fine-tuning the X-ray detection characteristics of 0D hybrid Cd-based perovskites by carefully selecting appropriate organic components as cationic templates.
In this work, a novel 0D Cd-based perovskite was designed by introducing large steric hindrance organic cations (methyltriphenylphosphonium, C19H18P+) into cadmium chloride frameworks, and SCs with sizes of 6 × 5 × 3 mm3 were obtained. The crystal structure, optical characteristics, and electrical behavior of the as-grown (C19H18P)2CdCl4 SC were explored. Furthermore, the successful fabrication of an X-ray photoconductor was achieved using the SCs of (C19H18P)2CdCl4, demonstrating a remarkable ability to directly convert X-ray radiation into electrical signals with a high sensitivity of ∼143.6 μC Gyair−1 cm−2 under a bias of 1000 V (333 V mm−1). Moreover, it exhibits an ultrastable response current and maintains a stable baseline with a minimal dark current drift at a rate as low as 7.34 × 10−4 pA cm−1 s−1 V−1. This study opens up a novel avenue for the utilization of new hybrid halides, particularly in the field of high-energy radiation detectors.
2. Results and discussion
First, we employed a simple solution method to synthesize (C19H18P)2CdCl4 SCs. Specifically, the (C19H18P)2CdCl4 SC was successfully synthesized by a slow volatilization method, where the crystal growth was meticulously controlled at a constant temperature of 40 °C via a slow evaporation process. To prevent extensive nucleation and ensure optimal crystalline quality, it was imperative to maintain a steady and gradual growth of crystals in the evaporating dish. Our largest SC, measuring 6 × 5 × 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mm3 in size, was achieved following a few days of growth in the dish (Fig. 1(a)). The flawless crystals underwent a gentle cleaning and drying process within an N2 glovebox for an extended period, followed by a 2 h annealing treatment at 100 °C to ensure thorough elimination of moisture and alleviation of lattice strain.10Fig. 1(a) depicts the photograph of a (C19H18P)2CdCl4 SC with a well-defined shape. The scanning electron microscopic (SEM) image taken from a top-view perspective reveals a uniformly smooth surface devoid of any discernible grain boundaries (Fig. S1a, ESI†), indicating a flawless standard of the SC, providing additional evidence for the significant benefits offered by the crystal growth technique. The corresponding energy-dispersive spectrum (EDS) analysis reveals a molar ratio of 2.13/0.96/3.85 for P, Cd, and Cl (Fig. S1b, ESI†), which aligns with the stoichiometric ratio of P, Cd, and Cl in (C19H18P)2CdCl4. Additionally, the consistency of the (C19H18P)2CdCl4 SCs is illustrated by the mapping measurements, as depicted in Fig. S2 (ESI†). The SEM analysis with EDS mapping reveals the homogeneous dispersion of the probed elements across the crystal surface, indicating a pristine surface free from any form of contamination.
mm3 in size, was achieved following a few days of growth in the dish (Fig. 1(a)). The flawless crystals underwent a gentle cleaning and drying process within an N2 glovebox for an extended period, followed by a 2 h annealing treatment at 100 °C to ensure thorough elimination of moisture and alleviation of lattice strain.10Fig. 1(a) depicts the photograph of a (C19H18P)2CdCl4 SC with a well-defined shape. The scanning electron microscopic (SEM) image taken from a top-view perspective reveals a uniformly smooth surface devoid of any discernible grain boundaries (Fig. S1a, ESI†), indicating a flawless standard of the SC, providing additional evidence for the significant benefits offered by the crystal growth technique. The corresponding energy-dispersive spectrum (EDS) analysis reveals a molar ratio of 2.13/0.96/3.85 for P, Cd, and Cl (Fig. S1b, ESI†), which aligns with the stoichiometric ratio of P, Cd, and Cl in (C19H18P)2CdCl4. Additionally, the consistency of the (C19H18P)2CdCl4 SCs is illustrated by the mapping measurements, as depicted in Fig. S2 (ESI†). The SEM analysis with EDS mapping reveals the homogeneous dispersion of the probed elements across the crystal surface, indicating a pristine surface free from any form of contamination.
A powder X-ray diffraction (XRD) pattern of the crystal obtained, depicted in black in Fig. 1(b), exhibits a strong resemblance to the simulated outcome (depicted in red), thereby validating its composition as (C19H18P)2CdCl4. To gain insights into the spatial organization of inorganic anion and organic cationic units within the crystal, single-crystal X-ray diffraction was performed. The compound (C19H18P)2CdCl4 forms crystals in the P213 space group, with a unit cell of dimensions characterized by the following parameters: a = 15.49955(3) Å, b = 15.49955(3) Å, and c = 15.49955(3) Å, and α = 90°, β = 90°, and γ = 90° (see Table S1 in the ESI†). The crystal structure of the compound is illustrated in Fig. 1(c), featuring a typical hybrid metal halide architecture. It consists of an infinite isolated tetrahedron formed by CdCl4 units, confirming the (C19H18P)2CdCl4 classification as a 0D perovskite. (C19H18P)2CdCl4 comprises an organic cation [C19H18P]+ and an inorganic structural unit [CdCl4]2− (Fig. 1(d)). In the context of an organic layer, the cations [C19H18P]+ exhibit a globular configuration similar to that of the TMCM+ cation.38 The more detailed crystallographic data are summed up in Tables S1–S7 (ESI†). It is worth noting that the bond angles of Cl1–Cd–Cl2 and Cl1–Cd–Cl11 are 109.14° and 109.80° respectively (Table S5, ESI†), and the bond angles are close to the perfect tetrahedron (109.5°).
X-ray photoelectron spectroscopy (XPS) was performed to investigate the chemical makeup and molecular arrangement of (C19H18P)2CdCl4, as depicted in Fig. 2(a) and (b). The Cl 2p spectrum exhibited the presence of a single chlorine atom in its chemical surroundings, which was accurately represented by a distinct pair of peaks (with approximately 1.6 eV spin–orbit splitting between Cl 2p3/2 and Cl 2p1/2). The doublet observed at 198.0 and 199.5 eV in the binding energy spectrum could be ascribed to the presence of Cl–Cd interactions. The Cd 3d spectrum exhibited a pair of prominent peaks at energy levels of 405.3 and 412.0 eV, in line with the Cd–Cl bonds, specifically representing the contributions from 3d5/2 and 3d3/2 orbitals.
 | ||
| Fig. 2 X-ray photoelectron spectra of (a) Cl 2p and (b) Cd 3d. (c) Calculation of the optical bandgap by the Tauc method according to the absorbed data in Fig. S3 (ESI†). (d) Electronic band structure of (C19H18P)2CdCl4 calculated through the Heyd–Scuseria–Ernzerhof (HSE) functional. (e) Total and partial densities of states of (C19H18P)2CdCl4. (f) PL spectra of the (C19H18P)2CdCl4 SC. | ||
The optical bandgap of the (C19H18P)2CdCl4 crystal was determined by UV-vis absorption spectroscopy, where the absorption edge was observed at approximately 296 nm (Fig. S3, ESI†). By employing the Tauc plot (Fig. 2(c)), we identified the direct bandgap as 4.31 eV. This significant bandgap effectively eliminated the possibility of utilizing it for photovoltaic purposes; however, its interaction with high-energy radiation such as X-rays does not impede the occupation of vacant electron states.39
According to the results of theoretical calculations, it can be observed from Fig. 2(d) that (C19H18P)2CdCl4 exhibits a direct bandgap of 4.91 eV, where the valence band maximum (VBM) and conduction band minimum (CBM) are both located at the Γ point. This estimation appears to be higher than the experimental value of 4.31 eV, which is within reasonable limits. It is worth noting that the density of states plots indicate that the VBM primarily originates from Cl 3p orbitals, with a minor contribution from organic and Cd 4d orbitals. For another, the CBM can be attributed mainly to organic sources (Fig. 2(e)). As illustrated in Fig. S4 (ESI†), the onset of absorption for (C19H18P)2CdCl4 occurs at approximately 4 eV, falling within the ultraviolet spectrum.
In order to further understand the optoelectronic characteristics of (C19H18P)2CdCl4, photoluminescence (PL) analysis was conducted using an excitation wavelength of 260 nm at room temperature. As shown in Fig. 2(f), a faint and slender emission peak at 338 nm was detected, potentially attributed to robust electron–photon interaction in comparison with an unlike broad band also found in previously reported Cd-based organic–inorganic perovskites.36,40–43 The fluorescence decay curve of (C19H18P)2CdCl4, measured at 338 nm, can be accurately described by a mono-exponential function yielding a lifetime value of 3.8 ns (Fig. S5, ESI†), which is close to that of most of the hybrid lead perovskites such as (C4H9NH3)2PbCl4 (2.59–9.68 ns), (2meptH2)PbBr4 (2.23 ns), α-(DMEN)PbBr4 (1.39 ns), (BZA)2PbCl4 (3.96 ns), and (4amp)PbBr4 (2.5 ns).44–48
In addition, we conducted ultraviolet photoelectron spectroscopy (UPS) on a polycrystalline sample to analyze the energy distribution of (C19H18P)2CdCl4. The valence band value relative to the work function can be determined by utilizing the tangent of the cutoff edge (Fig. S6a, ESI†). The valence band maximum (VBM) and conduction band minimum (CBM) energies of (C19H18P)2CdCl4 are observed at −6.35 and −2.16 eV, respectively, suggesting that the semiconductor exhibits P-type conductivity (Fig. S6b, ESI†).
It is worth mentioning that the (C19H18P)2CdCl4 SC has good stability. As shown in Fig. 3(a), we conducted XRD analysis on the finely ground powders that were kept UNDER ambient conditions for a period of three months to assess their stability. It was observed that (C19H18P)2CdCl4 did not undergo any phase transition or decomposition when exposed to air for this duration, indicating its excellent resistance to atmospheric factors. As depicted in Fig. 3(b), the thermogravimetric analyses reveal that (C19H18P)2CdCl4 exhibits a temperature of 376 °C at which it experiences a 5% reduction in mass. This finding suggests that the crystal structure of (C19H18P)2CdCl4 remains thermally stable even under typical operating conditions for devices. In addition, there is no obvious weight loss until 321 °C. These stability results demonstrate that (C19H18P)2CdCl4 exhibits better phase stability compared with other organic–inorganic hybrid materials.41,49,50
Why is the stability of this type of 0D material good? We think it has something to do with his structure. Its 0D structure is the CdCl4 tetrahedron structure and the periodic distribution of cations at A-site intervals, as shown in Fig. 1(c). In this way, electrons are transferred in the form of jumping between the tetrahedral structure and the A-site cation, and the direction of transmission is only one, which will lead to weak ion migration and small polarization built-in electric field. In addition, its band gap is about 4.19 eV, which also increases the material's high bias voltage resistance, so the stability of both the material and the device is enhanced. This feature also makes it show good performance in X-ray detection.
The potential of utilizing the hybrid metal halide SC (C19H18P)2CdCl4 for direct X-ray detection was assessed. Typically, effective direct X-ray detection necessitates significant absorption of X-ray photons by the active layers. To precisely ascertain the photon absorption capacity of (C19H18P)2CdCl4 for X-rays, we extracted the absorption spectra data from NIST's XCOM application, comparing it with traditional semiconductors across various photon energies.51 Furthermore, it is widely acknowledged that a higher atomic number and greater density have advantageous effects on X-ray attenuation as per the equation α ∝ ρZ4/E3, where α is the absorption coefficient, ρ denotes the mass density, Z signifies the atomic number, and E stands for the radiation.52
The calculated average atomic number of (C19H18P)2CdCl4 at 50 keV, which corresponds to the peak intensity of the X-ray source, is 15.3. This value exceeds that of Si (14.0). The software “AutoZeff” was utilized to determine the effective atomic number.53 As shown in Fig. 3(c), we acquired the absorption coefficient for (C19H18P)2CdCl4, CdTe, and Si across an extensive range of photon energies from 0.001 to 100 MeV based on data from the database containing cross-section information for photons. Irrespective of the resonant absorption occurring at the K, L, and M edges, it can be observed that the absorption coefficient of (C19H18P)2CdCl4 is greater than that of Si across all energy ranges. This observation aligns following the trend exhibited by average atomic numbers.
Fig. 3(d) illustrates the energy-dependent attenuation coefficient, covering a broad spectrum spanning from 0.001 to 100 meV, as per the photon cross-section database provided by the National Institute of Standards and Technology.51 The (C19H18P)2CdCl4 compound exhibits comparable attenuation efficiencies to other substances such as CdTe and Si. A 13 mm crystal of (C19H18P)2CdCl4 can absorb approximately 95% of 50 keV X-ray photons, which is slightly lower than that of CdTe but considerably higher than that of Si with an equivalent thickness (approximately 73%). The increased X-ray attenuation in the (C19H18P)2CdCl4 crystal semiconductor can be attributed to the higher Z value of Cd.
The lateral structure of Au/(C19H18P)2CdCl4/Au was utilized to measure photoconductivity, which was based on the 0D (C19H18P)2CdCl4 SC. The I–V curves exhibit linear and symmetrical characteristics, suggesting a strong ohmic connection between the Au electrode and the SC (Fig. 4(a)). Upon exposure to X-ray photoexcitation at a dose rate of 580 μGy s−1, the photocurrent demonstrates a significant increase from 13 pA (dark) to 402 pA (light), resulting in an impressive photocurrent on/off ratio of ∼31. These findings highlight its potential as a highly efficient material for X-ray detection. As illustrated in Fig. S7 (ESI†), the resistivity of (C19H18P)2CdCl4 was calculated to be 3.73 × 1011 Ω cm, which is at least three orders significantly greater than that of CH3NH3PbBr3 as well as various other perovskite materials containing lead (107–108 Ω cm).1,54
The high capacity for attenuating X-rays ensures effective absorption of a significant number of X-ray photons despite the thinness of the material, thereby facilitating the retrieval of charge carriers.32 Inspired by the encouraging photoconductive properties and X-ray with a dose rate of 410![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) μGyair
μGyair![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) s−1, and measurements taken absorption capability, we proceeded to evaluate the detection efficiency of an X-ray source using a vertical-type (C19H18P)2CdCl4 SC detector. Charge collection plays a crucial part in the detection of X-rays, and it is determined by factors such as carrier mobility (μ), carrier lifetime (τ), and trap density. The crucial parameter for evaluating the properties of carriers (such as mobility and drift length) in X-ray detection is μτ, which indicates a semiconductor's ability to extract charge from deep within. For high is preferable to have a high μτ value and low trap density. In this work, we obtained the μτ products by analyzing the photoconductivity data with a customized version of the Hecht equation:10
s−1, and measurements taken absorption capability, we proceeded to evaluate the detection efficiency of an X-ray source using a vertical-type (C19H18P)2CdCl4 SC detector. Charge collection plays a crucial part in the detection of X-rays, and it is determined by factors such as carrier mobility (μ), carrier lifetime (τ), and trap density. The crucial parameter for evaluating the properties of carriers (such as mobility and drift length) in X-ray detection is μτ, which indicates a semiconductor's ability to extract charge from deep within. For high is preferable to have a high μτ value and low trap density. In this work, we obtained the μτ products by analyzing the photoconductivity data with a customized version of the Hecht equation:10
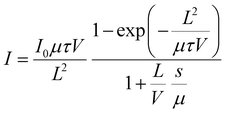 | (1) |
Due to its excellent X-ray absorption, high μτ product, and thermal stability, (C19H18P)2CdCl4 shows great potential for X-ray detection. Here, we employed a vertical photoconductive detector structure and fabricated (C19H18P)2CdCl4 SC (6 × 5 × 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mm3) X-ray detectors with the device configuration of Au/(C19H18P)2CdCl4/Au. The operational principle of our designed direct Au/(C19H18P)2CdCl4/Au detection device when exposed to X-ray radiation can be characterized as a process involving the conversion of light signals into electrical ones.4,15 The X-ray photons are absorbed by the active material, leading to the generation of high-energy electrons. These high-energy electrons have a tendency to dissipate their energy through the creation of additional electron–hole pairs. The energy required for the creation of an electron–hole pair, as determined by the empirical model W = 2Eg + 1.43, was measured to be 10.05 eV in this study, where Eg represents the bandgap of (C19H18P)2CdCl4.56
mm3) X-ray detectors with the device configuration of Au/(C19H18P)2CdCl4/Au. The operational principle of our designed direct Au/(C19H18P)2CdCl4/Au detection device when exposed to X-ray radiation can be characterized as a process involving the conversion of light signals into electrical ones.4,15 The X-ray photons are absorbed by the active material, leading to the generation of high-energy electrons. These high-energy electrons have a tendency to dissipate their energy through the creation of additional electron–hole pairs. The energy required for the creation of an electron–hole pair, as determined by the empirical model W = 2Eg + 1.43, was measured to be 10.05 eV in this study, where Eg represents the bandgap of (C19H18P)2CdCl4.56
For our device based on photoconductivity, the amplification of photocurrent is achieved by capturing photoexcited electrons in shallow defects. During their capture, the photoexcited holes can repeatedly move between the electrodes.57 The X-rays were produced by an X-ray tube (NEWTON SCIENTIFIC Model M237) with a gold target, operating at an accelerating voltage of 50 kV and a power output of 10 W. The photocurrent of the SC device in Fig. 4(c) can be adjusted from 195 to 901 pA by rising the dose rate of X-ray, while maintaining a stable dark current around 200 pA, up to a bias voltage of 1000 V (results at other bias voltages are presented in Fig. S8, ESI†). The rise and fall time of the detector reaches 0.637 s and 0.431 s, respectively (Fig. S9, ESI†). Compared to the crystal structure of 3D, 2D or 1D perovskites, 0D (C19H18P)2CdCl4 hybrid metal halide perovskites consist of isolated metal halide [CdCl4]2− tetrahedral units. This arrangement leads to more localized electronic structures, which is consistent with the previously calculated electronic band structure (Fig. 2(d)), resulting in higher activation energy. Consequently, ion migration is effectively suppressed and the operational stability of X-ray detectors under high working bias is enhanced. Moreover, the dark current density was estimated to be approximately 20 nA cm−2, obtained through dividing the current value with the active area of Au electrodes (approximately 0.01 cm2), which is comparable to the values of Cs3Bi2I9 (18 nA cm−2) and much lower than those of CsPbBr3 (170 nA cm−2) SCs.58,59
The X-ray tube current is varied from 30 to 150 μA in order to modify the X-ray dose rates, which range between 124 and 621 μGyair s−1. Fig. 4(d) illustrates the correlation between the dose rates and X-ray response current densities under various biased voltages. The relationship between the generated current density and X-ray dose rate shows a nearly linear trend for different applied voltages. Fig. 4(e) presents the X-ray detection sensitivities calculated at different applied voltages.
Based on the results of the nearly linear fit, it can be estimated that the Cd-based single-crystal device has an X-ray detection sensitivity of ∼ 62.3 μC Gyair−1 cm−2 when biased at 200 V (with a field strength of 66.67 V mm−1), and ∼ 143.6 μC Gyair−1 cm−2 when biased at 1000 V (with a field strength of 333 V mm−1). These values are similar to those reported for bismuth-based and Cd-based perovskite TMCM-CdCl3 devices (128.9 ± 4.64 μC Gyair−1 cm−2 when biased at a field strength of 10 V mm−1),49 as well as double perovskite Cs2AgBiBr6 X-ray detectors (∼105 μC Gyair−1 cm−2 when biased at a field strength of 25 V mm−1).11 Given the operational efficiency of the amorphous selenium X-ray detector commonly employed in commercial applications (20 μC Gyair−1 cm−2 at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 V mm−1), our X-ray detector's relatively favorable outcomes highlight the potential application of hybrid Cd-based metal halide perovskites in X-ray detection.17 In addition, the photocurrent response of a (C19H18P)2CdCl4 SC X-ray detector to ambient air radiation was observed when biased at a field strength of 333
000 V mm−1), our X-ray detector's relatively favorable outcomes highlight the potential application of hybrid Cd-based metal halide perovskites in X-ray detection.17 In addition, the photocurrent response of a (C19H18P)2CdCl4 SC X-ray detector to ambient air radiation was observed when biased at a field strength of 333![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) V
V![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mm−1 (see Fig. 4(f)). It is evident that upon activation of the X-ray, there is a rapid increase in the photocurrent up to its maximum value of 629 pA. After the radiation is deactivated, the current quickly decreases to a low value of 9
mm−1 (see Fig. 4(f)). It is evident that upon activation of the X-ray, there is a rapid increase in the photocurrent up to its maximum value of 629 pA. After the radiation is deactivated, the current quickly decreases to a low value of 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) pA known as the dark current. Additionally, no noticeable degradation in photocurrent is observed when subjected to air testing for three cycles. The dark current remained stable throughout the measurements, and there was no noticeable drift in current observed vertically. The stable outputs of current suggest that the ion migration were found to be negligible in 0D (C19H18P)2CdCl4 SCs. The remarkable stability of our device aligns with the excellent air storage stability observed in (C19H18P)2CdCl4 SCs, as shown in Fig. 3(a).
pA known as the dark current. Additionally, no noticeable degradation in photocurrent is observed when subjected to air testing for three cycles. The dark current remained stable throughout the measurements, and there was no noticeable drift in current observed vertically. The stable outputs of current suggest that the ion migration were found to be negligible in 0D (C19H18P)2CdCl4 SCs. The remarkable stability of our device aligns with the excellent air storage stability observed in (C19H18P)2CdCl4 SCs, as shown in Fig. 3(a).
We also examined the baseline drift observed in the manufactured detectors. A bias voltage of 1000 V was sustained for a duration of 120 seconds, during which the baseline current exhibited greater stability in the (C19H18P)2CdCl4 device than in the CsPbBr3 device (Fig. S10a, ESI†).10 We determined the present deviation D between the 10th and 110th s by employing the following equation:
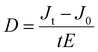 | (2) |
3. Conclusions
In summary, we have successfully fabricated a novel Cd-based hybrid metal halide perovskite (C19H18P)2CdCl4 large crystal and demonstrated its potential for X-ray detection. This crystal reaching dimensions of up to 6 × 5 × 3 mm3 can be acquired from aqueous solutions under ambient conditions, which proves advantageous for the utilization of this material in optoelectronic applications. The crystal exhibited a direct band gap of 4.19 eV, indicating its P-type semiconductor characteristics, and the product of carrier lifetime and mobility achieved a value on the order of 10−4 cm2 V−1. Due to the high product of carrier lifetime and mobility, we can create an exceptionally effective X-ray detector based on crystals, which exhibits a sensitivity of ∼143.6 μC Gyair−1 cm−2. Moreover, it demonstrates an exceptionally stable response current and maintains a steady baseline with the lowest drift in dark current at 7.34 × 10−4 pA cm−1 s−1 V−1. This work showcases the promising capabilities of molecular perovskites in detecting X-rays with high efficiency, thanks to their exceptional charge transport properties. Additionally, it opens up a novel avenue for investigating functional materials in optoelectronics research. Moreover, our findings provide an opportunity to explore reliable and enduring X-ray detectors.4. Experimental section
4.1 Materials
4.2 Synthesis of (C19H18P)2CdCl4 single crystals
A mixture of deionized water (55 mL) and HCl (5 mL) was stirred at room temperature overnight, followed by the addition of a methyltriphenylphosphonium chloride solution (10 mmol, 3.13 g). Subsequently, a solution of CdCl2 (5 mmol, 0.92 g) was added. Here, hydrochloric acid was used to create an acidic setting. The solution was then allowed to naturally evaporate at room temperature. After several days, colorless, transparent SCs were grown from the solution in the form of parallel epipedic shapes.
4.3 Device fabrication with (C19H18P)2CdCl4 single crystals
Prior to the fabrication of the device, the crystal was subjected to multiple rounds of tape lifting in order to eliminate any contaminants and imperfections on its surface. To fabricate vertically aligned photodetectors, square-shaped gold (Au) electrodes measuring 1 mm on each side and with a thickness of 80 nm were thermally evaporated onto the upper and lower surfaces of (C19H18P)2CdCl4 crystals using an already prepared shadow mask. The separation between the two Au electrodes measured 3 mm.4.4 Material property characterization of (C19H18P)2CdCl4
4.5 DFT calculations
DFT calculations were performed using the Vienna Ab initio Simulation Package (VASP) 6.1 software employing the projection-augmented wave (PAW) approach.60 The energy cutoff for plane waves was chosen as 400 eV. The structural optimization utilized the Heyd–Scuseria–Ernzerhof (HSE) hybrid functional, with a mixing parameter of 25%.61,62Γ-centered k-meshes were employed to sample the Brillouin zones, with a k-spacing of 0.2 Å−1. The crystal structures underwent complete relaxation until the total force on each atom reached a value lower than 0.03 eV Å−1.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (62134003, 62074068, 62074066, 62204092, 12050005), Major State Basic Research Development Program of China (2021YFB3201000), China Postdoctoral Science Foundation (2023T160242, 2022M710054), Fund for the Natural Science Foundation of Hubei Province (2021CFA036, 2020CFA034), the Shenzhen Basic Research Program (JCYJ20200109115212546).References
- H. Wei, Y. Fang, P. Mulligan, W. Chuirazzi, H. H. Fang, C. Wang, B. R. Ecker, Y. Gao, M. A. Loi, L. Cao and J. Huang, Nat. Photonics, 2016, 10, 333–339 CrossRef CAS.
- W. Wei, Y. Zhang, Q. Xu, H. Wei, Y. Fang, Q. Wang, Y. Deng, T. Li, A. Gruverman, L. Cao and J. Huang, Nat. Photonics, 2017, 11, 315–321 CrossRef CAS.
- J. A. Rowlands, Nature, 2017, 550, 47–48 CrossRef CAS PubMed.
- Y. C. Kim, K. H. Kim, D. Y. Son, D. N. Jeong, J. Y. Seo, Y. S. Choi, I. T. Han, S. Y. Lee and N. G. Park, Nature, 2017, 550, 87–91 CrossRef CAS PubMed.
- D. M. Panneerselvam and M. Z. Kabir, J. Mater. Sci.: Mater. Electron., 2017, 28, 7083–7090 CrossRef CAS.
- H. M. Thirimanne, K. D. G. I. Jayawardena, A. J. Parnell, R. M. I. Bandara, A. Karalasingam, S. Pani, J. E. Huerdler, D. G. Lidzey, S. F. Tedde, A. Nisbet, C. A. Mills and S. R. P. Silva, Nat. Commun., 2018, 9, 2926 CrossRef CAS PubMed.
- M. Tegze and G. Faigel, Nature, 1996, 380, 49–51 CrossRef CAS.
- J. M. Boone and A. E. Chavez, Med. Phys., 1996, 23, 1997–2005 CrossRef CAS PubMed.
- M. Shanmugam, S. V. S. Murty, Y. B. Acharya, S. K. Goyal, A. R. Patel, B. Shah, A. K. Hait, A. Patinge and D. Subrahmanyam, Adv. Space Res., 2014, 54, 1974–1984 CrossRef CAS.
- R. Zhuang, X. Wang, W. Ma, Y. Wu, X. Chen, L. Tang, H. Zhu, J. Liu, L. Wu, W. Zhou, X. Liu and Y. Yang, Nat. Photonics, 2019, 13, 602–608 CrossRef CAS.
- W. Pan, H. Wu, J. Luo, Z. Deng, C. Ge, C. Chen, X. Jiang, W. J. Yin, G. Niu, L. Zhu, L. Yin, Y. Zhou, Q. Xie, X. Ke, M. Sui and J. Tang, Nat. Photonics, 2017, 11, 726–732 CrossRef CAS.
- C. Ji, S. Wang, Y. Wang, H. Chen, L. Li, Z. Sun, Y. Sui, S. Wang and J. Luo, Adv. Funct. Mater., 2020, 30, 1905529 CrossRef CAS.
- Y. Wang, X. Liu, X. Li, F. Zhai, S. Yan, N. Liu, Z. Chai, Y. Xu, X. Ouyang and S. Wang, J. Am. Chem. Soc., 2019, 141, 8030–8034 CrossRef CAS PubMed.
- W. Zhao and J. A. Rowlands, Med. Phys., 1995, 22, 1595–1604 CrossRef CAS PubMed.
- J. P. Moy, Nucl. Instrum. Methods Phys. Res., Sect. A, 2000, 442, 26–37 CrossRef CAS.
- E. Saucedo, V. Corregidor, L. Fornaro, A. Cuña and E. Dieguez, Thin Solid Films, 2005, 471, 304–309 CrossRef CAS.
- L. Fornaro, A. Cuna, A. Noguera, M. Perez and L. Mussio, IEEE Trans. Nucl. Sci., 2004, 51, 2461–2465 CAS.
- R. A. Street, S. E. Ready, K. Van Schuylenbergh, J. Ho, J. B. Boyce, P. Nylen, K. Shah, L. Melekhov and H. Hermon, J. Appl. Phys., 2002, 91, 3345–3355 CrossRef CAS.
- S. Yakunin, M. Sytnyk, D. Kriegner, S. Shrestha, M. Richter, G. J. Matt, H. Azimi, C. J. Brabec, J. Stangl, M. V. Kovalenko and W. Heiss, Nat. Photonics, 2015, 9, 444–449 CrossRef CAS PubMed.
- J. Jiang, M. Xiong, K. Fan, C. Bao, D. Xin, Z. Pan, L. Fei, H. Huang, L. Zhou, K. Yao, X. Zheng, L. Shen and F. Gao, Nat. Photonics, 2022, 16, 575–581 CrossRef CAS.
- C. C. Stoumpos and M. G. Kanatzidis, Adv. Mater., 2016, 28, 5778–5793 CrossRef CAS PubMed.
- S. Shrestha, R. Fischer, G. J. Matt, P. Feldner, T. Michel, A. Osvet, I. Levchuk, B. Merle, S. Golkar, H. Chen, S. F. Tedde, O. Schmidt, R. Hock, M. Rührig, M. Göken, W. Heiss, G. Anton and C. J. Brabec, Nat. Photonics, 2017, 11, 436–440 CrossRef CAS.
- Z. Shuang, H. Zhou, D. Wu, X. Zhang, B. Xiao, G. Ma, J. Zhang and H. Wang, Chem. Eng. J., 2022, 433, 134544 CrossRef CAS.
- D. Wu, Y. Xu, H. Zhou, X. Feng, J. Zhang, X. Pan, Z. Gao, R. Wang, G. Ma, L. Tao, H. Wang, J. Duan, H. Wan, J. Zhang, L. Shen, H. Wang and T. Zhai, InfoMat, 2022, 4, e12320 CrossRef CAS.
- Z. Gao, H. Zhou, K. Dong, C. Wang, J. Wei, Z. Li, J. Li, Y. Liu, J. Zhao and G. Fang, Nano-Micro Lett., 2022, 14, 215 CrossRef CAS PubMed.
- Y. Liu, Z. Xu, Z. Yang, Y. Zhang, J. Cui, Y. He, H. Ye, K. Zhao, H. Sun, R. Lu, M. Liu, M. G. Kanatzidis and S. Liu, Matter, 2020, 3, 180–196 CrossRef.
- V. Morad, Y. Shynkarenko, S. Yakunin, A. Brumberg, R. D. Schaller and M. V. Kovalenko, J. Am. Chem. Soc., 2019, 141, 9764–9768 CrossRef CAS PubMed.
- S. Yakunin, B. M. Benin, Y. Shynkarenko, O. Nazarenko, M. I. Bodnarchuk, D. N. Dirin, C. Hofer, S. Cattaneo and M. V. Kovalenko, Nat. Mater., 2019, 18, 846–852 CrossRef CAS PubMed.
- A. Yangui, S. Pillet, E. Bendeif, A. Lusson, S. Triki, Y. Abid and K. Boukheddaden, ACS Photonics, 2018, 5, 1599–1611 CrossRef CAS.
- H. Ye, Y. Zhang, D. Fu and R. Xiong, Angew. Chem., Int. Ed., 2014, 126, 11424–11429 CrossRef.
- A. Thorn and R. D. Willett, Cryst. Growth Des., 2005, 5, 673–679 CrossRef CAS.
- A. Thorn, R. D. Willett and B. Twamley, Cryst. Growth Des., 2006, 6, 1134–1142 CrossRef CAS.
- H. Jia, Z. Shi, Q. Yang, J. Yu and J. Xu, Dalton Trans., 2014, 43, 5806–5814 RSC.
- W. Liao, H. Ye, Y. Zhang and R. Xiong, Dalton Trans., 2015, 44, 10614 RSC.
- W. Liao, G. Mei, H. Ye, Y. Mei and Y. Zhang, Inorg. Chem., 2014, 53, 8913–8918 CrossRef CAS PubMed.
- W. Liao, H. Ye, D. Fu, P. Li, L. Chen and Y. Zhang, Inorg. Chem., 2014, 53, 11146–11151 CrossRef CAS PubMed.
- S. Wang, L. Li, Z. Sun, C. Ji, S. Liu, Z. Liu, Z. Wu, S. Zhao, M. Hong and J. Luo, J. Mater. Chem. C, 2017, 5, 4731–4735 RSC.
- L. Xu, J. Gao, X. Chen, X. Hua, D. Wu and W. Liao, Eur. J. Inorg. Chem., 2019, 800–807 CrossRef CAS.
- H. Li, X. Song, C. Ma, Z. Xu, N. Bu, T. Yang, Q. Cui, L. Gao, Z. Yang, F. Gao, G. Zhao, Z. Chen, Z. Ding, K. Zhao and S. Liu, J. Energy Chem., 2022, 64, 209–213 CrossRef CAS.
- K. M. McCall, C. C. Stoumpos, S. S. Kostina, M. G. Kanatzidis and B. W. Wessels, Chem. Mater., 2017, 29, 4129–4145 CrossRef CAS.
- Z. Qi, Y. Chen, Y. Guo, X. Yang, F. Zhang, G. Zhou and X. Zhang, J. Mater. Chem. C, 2021, 9, 88–94 RSC.
- S. Chen, H. Lin, L. Jun, P. Jing, D. Feng, C. Guang, L. Dong and L. Wu, Chem. – Asian J., 2020, 15, 3050–3058 CrossRef PubMed.
- Y. Wang, C. Ji, X. Liu, S. Han, J. Zhang, Z. Sun, A. Khan and J. Luo, Inorg. Chem. Front., 2018, 5, 2450–2455 RSC.
- G. Zhou, S. Guo, J. Zhao, M. Molokeev, Q. Liu, J. Zhang and Z. Xia, Sci. China Mater., 2019, 62, 1013–1022 CrossRef CAS.
- S. Wang, Y. Yao, J. Kong, S. Zhao, Z. Sun, Z. Wu, L. Li and J. Luo, Chem. Commun., 2018, 54, 4053–4056 RSC.
- L. Mao, Y. Wu, C. C. Stoumpos, B. Traore, C. Katan, J. Even, M. R. Wasielewski and M. G. Kanatzidis, J. Am. Chem. Soc., 2017, 139, 11956–11963 CrossRef CAS PubMed.
- M. H. Jung, Inorg. Chem., 2019, 58, 6748–6757 CrossRef CAS PubMed.
- W. He, Y. Yang, C. Li, W. P. D. Wong, F. Cimpoesu, A. M. Toader, Z. Wu, X. Wu, Z. Lin, Q. H. Xu, K. Leng, A. Stroppa and K. P. Loh, J. Am. Chem. Soc., 2023, 145, 14044–14051 CrossRef CAS PubMed.
- C. Ma, F. Chen, X. Song, M. Chen, L. Gao, P. Wang, J. Wen, Z. Yang, Y. Tang, K. Zhao and S. Liu, Adv. Funct. Mater., 2021, 31, 2100691 CrossRef CAS.
- C. Sun, Q. Zhong, X. Zhang, P. Xiao, Y. Cheng, Y. Gao, G. Liu and X. Lei, Inorg. Chem., 2021, 60, 18879–18888 CrossRef CAS PubMed.
- M. J. Berger, XCOM Photon Cross Sections Database: NIST Standard Reference Database, 2013, 8 Search PubMed.
- M. Xia, J. Yuan, G. Niu, X. Du, L. Yin, W. Pan, J. Luo, Z. Li, H. Zhao, K. Xue, X. Miao and J. Tang, Adv. Funct. Mater., 2020, 30, 1910648 CrossRef CAS.
- M. L. Taylor, R. L. Smith, F. Dossing and R. D. Franich, Med. Phys., 2012, 39, 1769–1778 CrossRef CAS PubMed.
- D. Shi, V. Adinolfi, R. Comin, M. Yuan, E. Alarousu, A. Buin, Y. Chen, S. Hoogland, A. Rothenberger, K. Katsiev, Y. Losovyj, X. Zhang, P. A. Dowben, O. F. Mohammed, E. H. Sargent and O. M. Bakr, Science, 2015, 347, 519–522 CrossRef CAS.
- K. Kim, S. Kim, J. Hong, J. Lee, T. Hong, A. E. Bolotnikov, G. S. Camarda and R. B. James, J. Appl. Phys., 2015, 117, 145702 CrossRef.
- R. Devanathan, L. R. Corrales, F. Gao and W. J. Weber, Nucl. Instrum. Methods Phys. Res., Sect. A, 2006, 565, 637–649 CrossRef CAS.
- L. Yao, G. Niu, L. Yin, X. Du, Y. Lin, X. Den, J. Zhang and J. Tang, J. Mater. Chem. C, 2020, 8, 1239–1243 RSC.
- Y. Zhang, Y. Liu, Z. Xu, H. Ye, Z. Yang, J. You, M. Liu, Y. He, M. G. Kanatzidis and S. Liu, Nat. Commun., 2020, 11, 2304 CrossRef CAS PubMed.
- H. Zhang, F. Wang, Y. Lu, Q. Sun, Y. Xu, B. Zhang, W. Jie and M. G. Kanatzidis, J. Mater. Chem. C, 2020, 8, 1248–1256 RSC.
- G. Kresse and J. Furthmüller, Phys. Rev. B: Condens. Matter Mater. Phys., 1996, 54, 11169 CrossRef CAS PubMed.
- J. Heyd, G. E. Scuseria and M. Ernzerhof, J. Chem. Phys., 2003, 118, 8207 CrossRef CAS.
- J. Heyd, G. E. Scuseria and M. Ernzerhof, J. Chem. Phys., 2006, 124, 219906 CrossRef.
Footnote |
| † Electronic supplementary information (ESI) available. CCDC 2332882. For ESI and crystallographic data in CIF or other electronic format see DOI: https://doi.org/10.1039/d4tc00594e |
| This journal is © The Royal Society of Chemistry 2024 |



