Integrating β-ketoenamine linkages into covalent organic frameworks toward efficient overall photocatalytic hydrogen peroxide production†
Chang
Shu‡
,
Peixuan
Xie‡
,
Xiaoju
Yang
,
Xuan
Yang
,
Hui
Gao
,
Bien
Tan
 * and
Xiaoyan
Wang
* and
Xiaoyan
Wang
 *
*
Key Laboratory of Material Chemistry for Energy Conversion and Storage, Ministry of Education, Hubei Key Laboratory of Material Chemistry and Service Failure, School of Chemistry and Chemical Engineering, Huazhong University of Science and Technology, Wuhan, Hubei 430074, China. E-mail: xiaoyan_wang@hust.edu.cn
First published on 17th August 2024
Abstract
Covalent organic frameworks (COFs) are promising photocatalysts for hydrogen peroxide (H2O2) production from water and oxygen. However, the H2O2 generation from a dual-channel pathway, the oxygen reduction reaction (ORR), and especially the water oxidation reaction (WOR), is still challenging for COF photocatalysts. Here, a series of COFs were constructed by introducing different amounts of hydroxy groups into the COF skeleton to adjust the redox potentials, exciton dissociation and active center. Overall, the β-ketoenamine-linked COF, Tz–THBZ, exhibited a more positive oxidation potential and lower exciton binding energy, leading to efficient photo-excited electron and hole separation ability, and shows an overall photocatalytic H2O2 production yielding a rate of 4688 μmol h−1 g−1 in pure water. Density functional theory calculations further demonstrate the keto-formed benzene and triazine rings as the photooxidation and reduction centers.
Introduction
Hydrogen peroxide (H2O2) ranks among the top 100 most important chemicals globally with a demand exceeding 5 million tons annually,1 and shows extensive applications in industries such as textile, pulp and paper bleaching.2 Nowadays, the dominance of the anthraquinone oxidation method accounts for over 95% of industrial H2O2 production, while its high energy consumption and generation of hazardous by-products limit its environmental sustainability.3 The emergence of photocatalytic H2O2 production is a promising technology that offers a sustainable approach to convert solar energy to chemical energy.4 The photocatalytic production of H2O2 typically involves two key half-reactions: the oxygen reduction reaction (ORR) and the water oxidation reaction (WOR).5 Currently, most established photocatalysts produce H2O2 through the ORR pathway, often relying on sacrificial reagents (such as ethanol or benzyl alcohol) to quench photogenerated holes to achieve significant production efficiency.6–8 To promote solar energy utilization efficiency, optimizing the ORR pathway and adequately leveraging the WOR pathway to directly generate H2O2 from H2O are crucial. However, challenges arise due to the high oxidation potential in thermodynamic terms (e.g., the conversion of H2O to H2O2 requires 1.78 V vs. NHE at pH = 0) and the presence of competitive reactions such as hydroxyl radical or oxygen generation, limiting the overall performance of the WOR pathway.9,10 Furthermore, the microenvironment of the reaction sites plays a critical role in which the hydrophilic environment can facilitate the water adsorption that results in the acceleration of the WOR process.11 Therefore, developing dual-channel H2O2 production that can synergistically coordinate the ORR and WOR pathways to achieve maximum solar energy and 100% atomic utilization remains a significant challenge in advancing photocatalytic H2O2 production.Organic photocatalysts, such as graphitic carbon nitride (g-C3N4),12 conjugated microporous polymers (CMPs),13 covalent triazine frameworks (CTFs),14 and conjugated linear polymers (CLPs),15 have emerged as promising candidates for photocatalytic H2O2 production. These polymers show large surface areas, tunable electronic and optical properties, and functional diversity, offering significant potential in this field.16 Among them, covalent organic frameworks (COFs) have emerged as promising candidates for H2O2 photosynthesis due to their strong covalent bonds, crystalline structures, and ordered pore arrangements,7,9,17 which allow COFs to provide highly accessible active centers with reactants. The structural versatility also allows COFs to tune the photoelectronic properties at the molecular level. Various strategies have been employed to boost photocatalytic H2O2 production with COFs. Van Der Voort and co-workers constructed diarylamine units into COFs to promote strong reduction properties to efficiently reduce oxygen to H2O2;7 Han's group introduced oxygen functional groups as a strong electron/proton extractor in COFs to enhance the photocatalytic H2O2 production;18 Thomas and co-workers using a three-component Doebner reaction synthesized multicomponent COFs with acid–base bifunctionality for efficient photocatalytic H2O2 evolution;19 Xu and co-workers introduced acetylene or diacetylene moieties into CTFs to enhance photocatalytic H2O2 production.14 Despite these advancements, only a limited number of studies exhibited a successful H2O2 generation using COF-based photocatalysts through both the WOR and ORR pathways. For instance, the work by Tang and co-workers has reported thiophene-containing COFs by adjusting the N-heterocycle modules to achieve dual ORR and WOR performance.20 Challenges including insufficient electron–hole separation and limited oxidizing ability often hinder the exciton dissociation and photoexcited hole utilization, resulting in poor WOR ability. Moreover, the availability of catalytic sites is also important, necessitating stronger interactions with reactants (e.g. O2 and H2O).
In this study, a series of COFs, namely Tz–HBZ, Tz–DHBZ, and Tz–THBZ, were synthesized by incorporating varying numbers of hydroxy groups. The presence of electron-donating hydroxy groups allows the formation of a β-ketoenamine structure for the modulation of oxidation ability and the planar structure in the COFs, facilitating efficient charge transfer and creating a viable WOR pathway for enhanced H2O2 photosynthesis.21,22 Among the three COFs, Tz–THBZ, featuring irreversible β-ketoenamine linkages, exhibited the highest H2O2 production yield of 4688 μmol h−1 g−1 under visible light (λ > 420 nm) irradiation compared to Tz–HBZ and Tz–DHBZ. Additionally, Tz–THBZ achieved a solar-to-chemical conversion (SCC) efficiency of 0.36% and demonstrated excellent stability over six cycles (equivalent to 60 hours) with minimal changes in its chemical structure. Control experiments confirmed that all three COFs produced H2O2 through both the ORR and WOR dual-channel pathways, underscoring the strategic utilization of the WOR route to enhance the photocatalytic efficiency of H2O2 production.
Results and discussion
Synthesis and structural characterization
Three ordered COFs, Tz–HBZ, Tz–DHBZ and Tz–THBZ, were constructed under solvothermal conditions via the Schiff-base condensation reaction. As illustrated in Fig. 1, the 4,4′,4′′-(1,3,5-triazine-2,4,6-triyl)trianiline (Tz) as an amine monomer was reacted with different aldehyde precursors, 2-hydroxy-1,3,5-benzenetricarboxaldehyde (HBZ), 2,4-dihydroxy-1,3,5-benzenetricarboxaldehyde (DHBZ) and 1,3,5-triformylphloroglucinol (THBZ), which contain varying numbers of hydroxy groups (Fig. S1–S3†). Tz–THBZ exhibited an irreversible keto–enol tautomerization towards the keto form, while Tz–HBZ and Tz–DHBZ displayed tautomerization reversibility. All the COFs exhibited a similar 2D layer structure with hexagonal topology, displaying long-range ordered characteristics. The crystal structures of these COFs were analyzed using powder X-ray diffraction (PXRD). The most intense peak at 6.1° was attributed to the (100) planes, while additional weak reflections and a broad reflection around 9.9°, 11.8°, and 25.4° corresponded to the (110), (200), and (001) planes, respectively (Fig. 1a, b and c). By comparing the experimental PXRD with calculated patterns, it can be confirmed that the structural arrangement of the three COFs matched well with the simulated eclipsed (AA) stacked models through Pawley refinements (Fig. S5†).Solid-state 13C CP-MAS NMR spectroscopy and Fourier transform infrared (FT-IR) spectroscopy were carried out to investigate the chemical structure of the synthesized COFs. As depicted in Fig. 2a, all three COFs exhibited peaks around 170 ppm and 140–106 ppm which were attributed to the carbon atoms in the triazine and aromatic rings, respectively.14,23 Tz–THBZ displayed a prominent peak at approximately 183 ppm, corresponding to the carbonyl carbon (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) group and indicating the prevalence of the keto form.24,25 In contrast, the absence of a peak around 180 ppm in Tz–HBZ, along with the appearance of a peak at 157 ppm for the imine bond (C
O) group and indicating the prevalence of the keto form.24,25 In contrast, the absence of a peak around 180 ppm in Tz–HBZ, along with the appearance of a peak at 157 ppm for the imine bond (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N),25,26 suggested the presence of the enol form. Owing to the reversible enol-to-keto tautomerization, Tz–DHBZ exhibited both signals at around 183 ppm and 157 ppm, related to the C
N),25,26 suggested the presence of the enol form. Owing to the reversible enol-to-keto tautomerization, Tz–DHBZ exhibited both signals at around 183 ppm and 157 ppm, related to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and C
O and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N group.27 Furthermore, all three COFs exhibited characteristic peaks in the FT-IR spectra (Fig. 2b and S6†), notably at 1628 cm−1 and 1580–1509 cm−1, corresponding to the stretching vibration of the C
N group.27 Furthermore, all three COFs exhibited characteristic peaks in the FT-IR spectra (Fig. 2b and S6†), notably at 1628 cm−1 and 1580–1509 cm−1, corresponding to the stretching vibration of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N and C
N and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds.28 Additionally, an increased peak at 1287 cm−1 in Tz–DHBZ and Tz–THBZ was attributed to the stretching vibration of the C–N bond.22
C bonds.28 Additionally, an increased peak at 1287 cm−1 in Tz–DHBZ and Tz–THBZ was attributed to the stretching vibration of the C–N bond.22
The permanent porosity of the synthesized COFs was confirmed through nitrogen adsorption–desorption experiments. The apparent Brunauer–Emmett–Teller (BET) surface areas of the three COFs were calculated to be 1334.6, 973.5 and 1032.7 m2 g−1 for Tz–HBZ, Tz–DHBZ and Tz–THBZ, respectively (Fig. 2c). The pore size distribution analysis using the nonlocal density functional theory (NLDFT) and cylindrical pore model revealed that the pore size of all the COFs was predominantly centered around 1.2 nm and also showed some other distributions which may attributed to the stacking faults in the COF structure (Fig. S7†). Scanning electron microscopy (SEM) and high-resolution transmission electron microscopy (HRTEM) images depicted nanorod and nanoparticle structures for all three COFs (Fig. S8 and S9†) and HRTEM images also observed clear lattice fringes for all COFs (Fig. S9†). The water contact angles decreased from 57.4° to 35.5° with the increase of hydroxy groups from Tz–HBZ to Tz–THBZ, indicating that Tz–THBZ shows stronger interactions with water (Fig. 2d). Thermal stability for all COFs was determined using thermogravimetric analysis (TGA) which showed that Tz–THBZ exhibited exceptional stability up to 400 °C under a nitrogen atmosphere (Fig. S10†). Tz–THBZ maintained its crystallinity even after immersion in a 1 M H2O2 solution for 48 hours in contrast to Tz–HBZ and Tz–DHBZ (Fig. S11†). The more stable chemical structure belongs to the more hydrogen bonding interactions within the molecule, which is highly desired for photocatalytic H2O2 production.
Photophysical and photochemical property study
The ultraviolet-visible diffuse reflection spectroscopy (UV/vis DRS) spectra revealed strong absorption of all the COFs in the ultraviolet and visible regions (Fig. 3a). The optical band gaps (Eg) were calculated to be 1.85 eV for Tz–HBZ, 1.96 eV for Tz–DHBZ, and 2.25 eV for Tz–THBZ using Tauc plots (Fig. S12†). Mott–Schottky plots showed positive slopes for all COFs, indicating n-type semiconductor behavior (Fig. S13†). The conduction band (CB) potentials were estimated to be −0.46, −0.41, and −0.39 V (vs. NHE, pH = 7) for Tz–HBZ, Tz–DHBZ, and Tz–THBZ, respectively, based on the Nernst equation. Combining the band gaps and CB potentials, the valence band (VB) potentials were also calculated to be 1.39, 1.55, and 1.85 V (vs. NHE, pH = 7) using the equation (EVB − ECB = Eg). The band positions, as shown in Fig. 3c, indicated that these COFs are thermodynamically feasible for overall photocatalytic H2O2 production via both the ORR and WOR pathways. In particular, Tz–THBZ showed much more positive potential, implying a stronger oxidizing ability. Density functional theory (DFT) simulations also demonstrated that Tz–THBZ exhibited a more extensive distribution of the highest occupied molecular orbitals (HOMO) compared to the other two COFs (Fig. S14†). In particular, Tz–THBZ showed a more positive VB potential than the others, indicating a favorable HOMO energy level beneficial for photoinduced oxidation reactions.22Based on the feasible thermodynamic properties of these COFs for redox reactions, an in-depth analysis of the photochemical properties was conducted to gain a comprehensive understanding of the photocatalysis process. In Fig. S15,† all COFs exhibited significantly enhanced light response compared to the dark conditions, while Tz–THBZ demonstrated superior photocurrent response capability to Tz–HBZ and Tz–DHBZ. Electrochemical impedance spectroscopy (EIS) curves (Fig. S16†) further illustrated that Tz–THBZ displayed a reduced arc radius, indicative of the smallest charge transfer resistance within the system. The time-resolved photoluminescence (TR-PL) spectra revealed that Tz–THBZ boasted a fluorescence lifetime of 1.13 ns, surpassing the 0.93 ns values for both Tz–HBZ and 1.02 for Tz–DHBZ (Fig. 3b). This longer fluorescence lifetime suggests that Tz–THBZ possesses an extended carrier diffusion time and superior charge transfer capability. To delve deeper into the separation behaviour of photoexcited electron–hole pairs, temperature-dependent photoluminescence (TD-PL) spectra were employed to estimate the exciton binding energy for these COFs. As depicted in Fig. 3d–f (insets), the PL intensity of all three COFs decreased monotonically as the temperature rose from 80 K to 298 K. The exciton binding energy (Eb) was then estimated by combining PL intensity and temperature using the Arrhenius equation.29 By utilizing the Arrhenius equation, the Eb was determined to be 57.8 meV for Tz–HBZ, 59.0 meV for Tz–DHBZ, and notably lower at 25.6 meV for Tz–THBZ. The lower Eb value for Tz–THBZ suggests that it is more favourable for exciton dissociation and charge delocalization during the photocatalytic process,30 aligning with its superior photochemical behaviour discussed previously.
Photocatalytic H2O2 production performances
The photocatalytic H2O2 production experiments were initially conducted to assess the performance of the three COF-based catalysts under visible light (λ > 420 nm) irradiation in an O2-saturated aqueous solution without using any sacrificial agents. As illustrated in Fig. 4a, all three COFs exhibited steady H2O2 production as the reaction time extended to 5 hours. All the COFs showed a much higher performance than their ingredients and physical mixtures, indicating the necessity of the COF structure (Fig. S17†). The H2O2 production yields were measured to be 1880 μmol h−1 g−1 for Tz–HBZ, 1250 μmol h−1 g−1 for Tz–DHBZ, and 2580 μmol h−1 g−1 for Tz–THBZ. The higher performance of Tz–THBZ can be attributed to its enhanced hydrophilicity, stronger oxidation potential and more favourable exciton dissociation. Furthermore, by optimizing the dosage to prevent catalyst aggregation and ensuring efficient light utilization, the performance of Tz–THBZ was further enhanced to reach 4688 μmol h−1 g−1 (Fig. S18†).31 The reproducibility of the photocatalytic performance of Tz–THBZ was confirmed by conducting photocatalysis experiments using three different batches of the material under the same conditions. The results revealed that all three batches of the as-prepared Tz–THBZ exhibited similar photocatalytic performance (Fig. S19†). The wavelength-dependent external quantum efficiency (EQE) of Tz–THBZ was found to align closely with the UV/vis DRS spectra, confirming that the reaction was initiated by the absorption of incident photons (Fig. 4b). Tz–THBZ achieved the highest EQE of 5.5% at 420 nm. Furthermore, the solar-to-chemical efficiency of Tz–THBZ, evaluated under simulated sunlight (AM 1.5 G, 100 mW cm−2), was calculated to be 0.36%, significantly surpassing the solar-to-biomass efficiency of plants (∼0.1%).32The photo-stability of a photocatalyst is a crucial parameter for its potential applications (Fig. 4c). The photocatalytic performance of Tz–THBZ demonstrated excellent stability over 6 cycles with a total reaction time of 60 hours. Only a slight decrease was observed in the last cycle, likely attributed to inevitable catalyst losses during the recycling process (Fig. S20†).33 The stability of Tz–THBZ surpassed that of most reported organic materials (Table S1†). In contrast to Tz–HBZ and Tz–DHBZ, where aromatic signals were detected in the 1H-NMR spectra of the photocatalytic solution after 50 hours of reaction (Fig. S21 and S22†), Tz–THBZ showed negligible changes, at least at the NMR level, in its chemical structure for the recycled catalyst (Fig. S21 and S22†). Following the reaction, it is important to prevent the generated H2O2 from decomposing in a light-driven process.34 As depicted in Fig. 4d, even in the absence of any catalyst, the H2O2 concentration gradually decreased after 5 hours of irradiation. Notably, Tz–HBZ and Tz–DHBZ were found to facilitate this decomposition process further, which might be the promoted H2O2 adsorption of the imine groups in the COF skeleton.35 In contrast, the presence of β-ketoenamine-linked Tz–THBZ inhibited the H2O2 decomposition, indicating its superior oxidizing ability to utilize holes for H2O2 generation through the WOR rather than decomposition.
Photocatalytic mechanism
The photocatalytic reactions were further investigated under different gas atmospheres to understand the performance of the COF-based catalysts. In Fig. 5a, it is observed that the performance of all COFs improved as the gas atmosphere changed from nitrogen to air to oxygen, indicating that the photocatalytic H2O2 production reaction was primarily driven by oxygen-involved reactions. In the nitrogen atmosphere, Tz–HBZ, Tz–DHBZ, and Tz–THBZ show the H2O2 yields of 390, 470, and 940 μmol h−1 g−1, respectively (Fig. S23†). This production showed a linear increase over 5 hours of irradiation, highlighting the ability of these COFs to drive the WOR through H2O to H2O2 conversion. These data suggest that all COFs possess dual-channel photocatalytic H2O2 production capabilities through WOR and ORR pathways.Further investigations were conducted to elucidate the photocatalytic pathway. Under dark conditions, no H2O2 was detected. The introduction of quenching agents such as potassium bromate (KBrO3), isopropanol (IPA), and p-benzoquinone (p-BQ) revealed insights into the involvement of electrons, holes, and 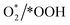 intermediates during the photocatalytic reaction. In a nitrogen atmosphere, the presence of methanol (hole quenching agent) inhibited performance compared to the blank condition (pure water). The presence of KBrO3 (1 mM) can quench electrons that increase H2O2 production yields for all COFs, which demonstrates the occurrence of the WOR (Fig. 5b and S24†). Moreover, under an oxygen atmosphere, the introduction of IPA as a hole-quenching agent significantly boosted performance, indicating the dominance of the ORR. In contrast, the presence of p-BQ inhibited H2O2 production yields, underscoring the crucial role of ˙O2− and the two-step single-electron ORR pathway (Fig. 5c). Electron paramagnetic resonance (EPR) measurements were conducted using 5,5-dimethyl-1-pyrroline N-oxide (DMPO) as a spin-trap agent to detect radicals generated during the photocatalytic reaction. In Fig. S25,† a comparison between the dark conditions and after-lighting revealed the presence of typical six-line characteristic peaks corresponding to DMPO–˙O2− for all COFs.36 This observation provides further evidence supporting the involvement of the indirect 2e− ORR pathway in the photocatalytic process. Tz–THBZ exhibited a stronger EPR signal compared to the other COFs, indicating its superior ability to generate active ˙O2− radicals.
intermediates during the photocatalytic reaction. In a nitrogen atmosphere, the presence of methanol (hole quenching agent) inhibited performance compared to the blank condition (pure water). The presence of KBrO3 (1 mM) can quench electrons that increase H2O2 production yields for all COFs, which demonstrates the occurrence of the WOR (Fig. 5b and S24†). Moreover, under an oxygen atmosphere, the introduction of IPA as a hole-quenching agent significantly boosted performance, indicating the dominance of the ORR. In contrast, the presence of p-BQ inhibited H2O2 production yields, underscoring the crucial role of ˙O2− and the two-step single-electron ORR pathway (Fig. 5c). Electron paramagnetic resonance (EPR) measurements were conducted using 5,5-dimethyl-1-pyrroline N-oxide (DMPO) as a spin-trap agent to detect radicals generated during the photocatalytic reaction. In Fig. S25,† a comparison between the dark conditions and after-lighting revealed the presence of typical six-line characteristic peaks corresponding to DMPO–˙O2− for all COFs.36 This observation provides further evidence supporting the involvement of the indirect 2e− ORR pathway in the photocatalytic process. Tz–THBZ exhibited a stronger EPR signal compared to the other COFs, indicating its superior ability to generate active ˙O2− radicals.
In situ attenuated total reflection surface-enhanced infrared absorption spectroscopy (ATR-SEIRAS) was employed to gain insights into the intermediates formed during the photocatalytic process of Tz–THBZ under an O2 atmosphere. In Fig. 5d, it is observed that once Tz–THBZ reaches equilibrium with water in the dark, the peak at around 949 cm−1 corresponding to O–O bonding shows a continuous increase upon light irradiation. Additionally, vibrational peaks at approximately 1100 cm−1 and 1283 cm−1 gradually increase, indicating the presence of ˙O2− and endoperoxide intermediates, supporting the two-step single-electron ORR pathway.23 Furthermore, a negative peak around 3190 cm−1 is observed during the ongoing photocatalytic process, suggesting the consumption of adsorbed water and indicating water involvement in the WOR (Fig. 5e).33
The electrostatic surface potential (ESP) map and DFT calculations also provide crucial insights into the charge distribution and adsorption energy within Tz–THBZ. As shown in Fig. 5f, the keto-formed Tz–THBZ, resulting from the irreversible keto-enamine form, exhibits a distinct charge distribution compared to the enol-formed structure. The positive charge, denoted by the blue region, is predominantly localized on the triazine unit, establishing it as a reduction center within the structure. Conversely, the negative charge, depicted by the red region, is specifically situated on the carbonyl group of the keto-Tp unit, suggesting its role as an oxidation center. Furthermore, DFT calculations have elucidated the adsorption energy between the redox center and reactants in the enol/keto forms of Tz–THBZ. In the enol-formed structure, the O2 tended to interact with the –OH group of the enol-Tp unit (ΔEO2 = −3.47 kJ mol−1) (Fig. S27a†), while H2O preferred binding to the oxygen and nitrogen atom of the –OH and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bonds (ΔEH2O = −13.7 kJ mol−1) (Fig. S27b†). Conversely, in the keto-formed structure, the O2 and H2O favoured interaction with the oxygen atom of the keto-Tp unit (ΔEO2 = −6.61 kJ mol−1) (Fig. 5g) and the carbon and nitrogen atom of the triazine unit (ΔEH2O = −24.91 kJ mol−1) (Fig. 5h). As a result, the β-keto-enamine linked structure demonstrates a more favourable interaction with reactants (e.g. O2 and H2O), exhibiting higher adsorption energies compared to the enol-formed structure.
N bonds (ΔEH2O = −13.7 kJ mol−1) (Fig. S27b†). Conversely, in the keto-formed structure, the O2 and H2O favoured interaction with the oxygen atom of the keto-Tp unit (ΔEO2 = −6.61 kJ mol−1) (Fig. 5g) and the carbon and nitrogen atom of the triazine unit (ΔEH2O = −24.91 kJ mol−1) (Fig. 5h). As a result, the β-keto-enamine linked structure demonstrates a more favourable interaction with reactants (e.g. O2 and H2O), exhibiting higher adsorption energies compared to the enol-formed structure.
DFT simulations also demonstrated that the HOMO were mainly concentrated on the hydroxy group containing-unit, while the LUMO were concentrated on the Tz unit, which was consistent with their ESP results. Moreover, Tz–THBZ showed a more separated HOMO and LUMO distribution, indicating an efficient separation of light-induced electrons and holes.31 Based on the above results, a probable mechanism for photocatalytic H2O2 production is proposed. When exposed to visible light, the COF absorbs photons, leading to the generation of electron–hole pairs. These charge carriers then migrate to the surface of the photocatalyst. The photogenerated electrons engage in a reaction with the adsorbed oxygen on the COF surface, ultimately producing H2O2 through an indirect sequential two-step single-electron ORR pathway. Meanwhile, the holes are involved in the WOR process through a direct 2e− reaction.
Conclusions
In summary, a series of COF photocatalysts were synthesized by incorporating varying amounts of hydroxy groups to enable visible light-induced hydrogen peroxide production. Both experimental and theoretical analyses suggest that Tz–THBZ, featuring a β-ketoenamine linkage, exhibits a higher HOMO energy and positively charged potentials, enhancing its WOR capability. Time-resolved PL and temperature-dependent PL studies demonstrate that Tz–THBZ displays prolonged charge carrier lifetimes and improved exciton dissociation, thereby boosting photocatalysis performance. Control experiments confirm that the β-ketoenamine-linked COF, Tz–THBZ, can generate H2O2 through both the ORR and WOR pathways, achieving a peak photocatalytic activity of 4688 μmol h−1 g−1 in pure water under visible light irradiation. DFT calculations further reveal that the triazine unit acts as the photoreduction center, while the keto-formed benzene ring serves as the oxidation site. This study underscores how the linkages within COFs impact the HOMO energy, excited-state properties, and electronic structure, significantly influencing oxidation capabilities and active centers in the photocatalytic process.Data availability
The data supporting this article have been included as part of the ESI.†Author contributions
Xiaoyan Wang and Bien Tan conceived the study and supervised this work. Chang Shu and Peixuan Xie performed the experiments and analysis and contributed equally to this work. Xiaoju Yang and Xuan Yang performed and conceived the ATR-SEIRAS analysis. Hui Gao carried out PXRD analyses. All authors interpreted the data and contributed to the preparation of the manuscript.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (Grant No. 52203259; Grant No. 21975086; Grant. No. 22204054); Natural Science Foundation of Hubei Province (Grant No. 2022CFB720); Fundamental Research Funds for the Central Universities (Grant No. 2024JYCXJJ041).Notes and references
- R. L. Myers, The 100 Most Important Chemical Compounds: a Reference Guide, Bloomsbury Publishing USA, 2007 Search PubMed.
- Y. Wu, J. Wu, F. Yang, C. Tang and Q. Huang, Polymers, 2019, 11, 776 CrossRef PubMed.
- G. Goor, in Catalytic Oxidations with Hydrogen Peroxide as Oxidant, ed. G. Strukul, Springer Netherlands, Dordrecht, 1992, pp. 13–43 Search PubMed.
- H. Hou, X. Zeng and X. Zhang, Angew. Chem., Int. Ed., 2020, 59, 17356–17376 CrossRef CAS PubMed.
- S. Qu, H. Wu and Y. H. Ng, Adv. Energy Mater., 2023, 13, 2301047 CrossRef CAS.
- H. Wang, C. Yang, F. Chen, G. Zheng and Q. Han, Angew. Chem., Int. Ed., 2022, 61, e202202328 CrossRef CAS PubMed.
- C. Krishnaraj, H. Sekhar Jena, L. Bourda, A. Laemont, P. Pachfule, J. Roeser, C. V. Chandran, S. Borgmans, S. M. J. Rogge, K. Leus, C. V. Stevens, J. A. Martens, V. Van Speybroeck, E. Breynaert, A. Thomas and P. Van Der Voort, J. Am. Chem. Soc., 2020, 142, 20107–20116 CrossRef CAS PubMed.
- L. Chen, C. Chen, Z. Yang, S. Li, C. Chu and B. Chen, Adv. Funct. Mater., 2021, 31, 2105731 CrossRef CAS.
- J. Sun, J. Chakraborty, M. Deng, A. Laemont, X. Feng, Y.-Y. Liu and P. Van Der Voort, J. Mater. Chem. A, 2023, 11, 21516–21540 RSC.
- S. Wu and X. Quan, ACS ES&T Eng., 2022, 2, 1068–1079 Search PubMed.
- W. Yu, C. Hu, L. Bai, N. Tian, Y. Zhang and H. Huang, Nano Energy, 2022, 104, 107906 CrossRef CAS.
- Y. Shiraishi, S. Kanazawa, Y. Kofuji, H. Sakamoto, S. Ichikawa, S. Tanaka and T. Hirai, Angew. Chem., Int. Ed., 2014, 53, 13454–13459 CrossRef CAS PubMed.
- Y.-X. Ye, J. Pan, Y. Shen, M. Shen, H. Yan, J. He, X. Yang, F. Zhu, J. Xu, J. He and G. Ouyang, Proc. Natl. Acad. Sci. U.S.A., 2021, 118, e2115666118 CrossRef CAS PubMed.
- L. Chen, L. Wang, Y. Wan, Y. Zhang, Z. Qi, X. Wu and H. Xu, Adv. Mater., 2020, 32, 1904433 CrossRef CAS PubMed.
- L. Liu, M.-Y. Gao, H. Yang, X. Wang, X. Li and A. I. Cooper, J. Am. Chem. Soc., 2021, 143, 19287–19293 CrossRef CAS PubMed.
- X. Xu, Y. Sui, W. Chen, W. Huang, X. Li, Y. Li, D. Liu, S. Gao, W. Wu, C. Pan, H. Zhong, H.-R. Wen and M. Wen, Appl. Catal., B, 2024, 341, 123271 CrossRef CAS.
- Z. Yong and T. Ma, Angew. Chem., Int. Ed., 2023, 62, e202308980 CrossRef CAS PubMed.
- L. Li, X. Lv, Y. Xue, H. Shao, G. Zheng and Q. Han, Angew. Chem., Int. Ed., 2024, 63, e202320218 CrossRef CAS PubMed.
- P. Das, G. Chakraborty, J. Roeser, S. Vogl, J. Rabeah and A. Thomas, J. Am. Chem. Soc., 2023, 145, 2975–2984 CrossRef CAS PubMed.
- J.-Y. Yue, L.-P. Song, Y.-F. Fan, Z.-X. Pan, P. Yang, Y. Ma, Q. Xu and B. Tang, Angew. Chem., Int. Ed., 2023, 62, e202309624 CrossRef CAS PubMed.
- L.-J. Wang, P.-Y. Dong, G. Zhang and F.-M. Zhang, Energy Fuels, 2023, 37, 6323–6347 CrossRef CAS.
- H. Wang, C. Qian, J. Liu, Y. Zeng, D. Wang, W. Zhou, L. Gu, H. Wu, G. Liu and Y. Zhao, J. Am. Chem. Soc., 2020, 142, 4862–4871 CrossRef CAS PubMed.
- D. Chen, W. Chen, Y. Wu, L. Wang, X. Wu, H. Xu and L. Chen, Angew. Chem., Int. Ed., 2023, 62, e202217479 CrossRef CAS PubMed.
- W.-R. Cui, C.-R. Zhang, R.-H. Xu, X.-R. Chen, R.-H. Yan, W. Jiang, R.-P. Liang and J.-D. Qiu, Small, 2021, 17, 2006882 CrossRef CAS PubMed.
- S. Haldar, D. Chakraborty, B. Roy, G. Banappanavar, K. Rinku, D. Mullangi, P. Hazra, D. Kabra and R. Vaidhyanathan, J. Am. Chem. Soc., 2018, 140, 13367–13374 CrossRef CAS PubMed.
- L. Zhang, S.-L. Wang, G.-H. Zhang, N. Shen, H. Chen, G. Tao, G.-H. Tao, F. Yong, J. Fu, Q.-H. Zhu and L. He, Cell Rep. Phys. Sci., 2022, 3, 101114 CrossRef CAS.
- M. Traxler, S. Gisbertz, P. Pachfule, J. Schmidt, J. Roeser, S. Reischauer, J. Rabeah, B. Pieber and A. Thomas, Angew. Chem., Int. Ed., 2022, 61, e202117738 CrossRef CAS PubMed.
- S. Chandra, D. Roy Chowdhury, M. Addicoat, T. Heine, A. Paul and R. Banerjee, Chem. Mater., 2017, 29, 2074–2080 CrossRef CAS.
- X. Li, Y. Wu, S. Zhang, B. Cai, Y. Gu, J. Song and H. Zeng, Adv. Funct. Mater., 2016, 26, 2435–2445 CrossRef CAS.
- G. Fu, D. Yang, S. Xu, S. Li, Y. Zhao, H. Yang, D. Wu, P. S. Petkov, Z.-A. Lan, X. Wang and T. Zhang, J. Am. Chem. Soc., 2024, 146, 1318–1325 CrossRef CAS PubMed.
- C. Shu, C. Han, X. Yang, C. Zhang, Y. Chen, S. Ren, F. Wang, F. Huang and J.-X. Jiang, Adv. Mater., 2021, 33, 2008498 CrossRef CAS PubMed.
- X.-G. Zhu, S. P. Long and D. R. Ort, Curr. Opin. Biotechnol., 2008, 19, 153–159 CrossRef CAS PubMed.
- C. Shu, X. Yang, L. Liu, X. Hu, R. Sun, X. Yang, A. I. Cooper, B. Tan and X. Wang, Angew. Chem., Int. Ed., 2024, 63, e202403926 CrossRef CAS PubMed.
- Y. Nosaka and A. Y. Nosaka, Chem. Rev., 2017, 117, 11302–11336 CrossRef CAS PubMed.
- J. Sun, H. Sekhar Jena, C. Krishnaraj, K. Singh Rawat, S. Abednatanzi, J. Chakraborty, A. Laemont, W. Liu, H. Chen, Y.-Y. Liu, K. Leus, H. Vrielinck, V. Van Speybroeck and P. Van Der Voort, Angew. Chem., 2023, 135, e202216719 CrossRef.
- G. R. Buettner, Free Radical Biol. Med., 1987, 3, 259–303 CrossRef CAS PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4ta03950e |
| ‡ These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2024 |





