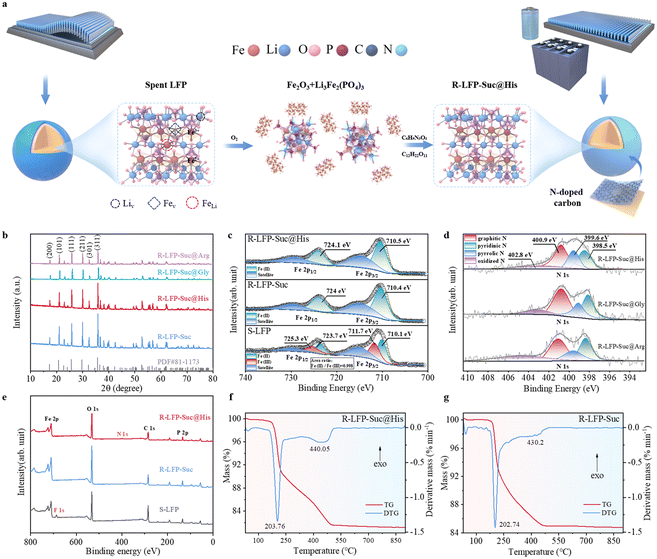
High performance of regenerated LiFePO4 from spent cathodes via an in situ coating and heteroatom-doping strategy using amino acids†
Junwei
Wang
ab,
Shuaijing
Ji
ab,
Qigao
Han
b,
Fengqian
Wang
ab,
Wuxin
Sha
b,
Danpeng
Cheng
b,
Weixin
Zhang
b,
Shun
Tang
 *b,
Yuan-Cheng
Cao
*b,
Yuan-Cheng
Cao
 b and
Shijie
Cheng
b
b and
Shijie
Cheng
b
aSchool of Materials Science and Engineering, Huazhong University of Science and Technology, Wuhan 430074, China
bState Key Laboratory of Advanced Electromagnetic Engineering and Technology, School of Electrical and Electronic Engineering, Huazhong University of Science and Technology, Wuhan 430074, China. E-mail: shuntang@hust.edu.cn
First published on 15th May 2024
Abstract
In recent years, recycling of numerous spent lithium-ion battery cathode materials has received increasing attention in order to protect the environment as well as to conserve resources, and the recovery of spent LiFePO4 (LFP) by direct regeneration has been widely studied. A considerable body of literature has delved into the failure mechanism of LFP. The mechanism is characterized by an irreversible phase change, which is primarily attributed to the sluggish diffusion of lithium ions (Li+) during cycling. Additionally, the migration of iron (Fe) ions to occupy Li+ sites further impedes Li+ diffusion. Consequently, the electrochemical performance of directly regenerated LFP is diminished by the phenomenon of Li defects. Here, a method of direct regeneration of LFP based on a doping strategy using environmentally friendly and economically efficient natural biomass amino acids has been developed, which inhibits Fe ion migration and improves the diffusion kinetics of Li+ and electrons by constructing a nitrogen-doped carbon coating. The regenerated LFP cathode exhibits excellent cycling stability and rate performance (98.7% capacity retention over 100 cycles at 1C current density and a high capacity retention of 87.9% after 500 cycles at 1C).
Introduction
Lithium-ion batteries (LIBs) have been widely applied in electric vehicles and energy storage power plants, with olivine lithium iron phosphate batteries (LiFePO4 or LFP) holding a significant share in the overall lithium-ion battery market due to their high safety, excellent stability, long cycle life, and cost-effectiveness. A large number of spent LFP (S-LFP) batteries are inevitably produced, so effective and harmless recycling technologies need to be developed to cope with the upcoming wave of retired batteries.1–3 Pyrometallurgical and hydrometallurgical methods are commonly employed in the industry for battery recycling. In pyrometallurgy, spent batteries are subjected to high temperature to separate metal resources from the slag. However, pyrometallurgy has been proved to suffer from high energy consumption and low recovery efficiency, and may produce harmful gases. On the other hand, the hydrometallurgical method involves the utilization of substantial quantities of acid and alkali reagents to dissolve the spent battery cathode. Nevertheless, the intricate reprocessing procedures associated with hydrometallurgy lead significantly to environmental degradation due to the substantial generation of waste water and gas.4–7Numerous studies have shown that the degraded performance of S-LFP batteries is attributed to the absence of Li+ during the cycling process, leading to the oxidation of Fe2+ to Fe3+ and inducing the migration of Fe ions to generate Fe–Li anti-site defects (FeLi).8,9 However, the lattice and morphology are maintained in used LFP batteries, offering the possibility of direct regeneration and recycling of cathode materials (Fig. 1a). At present, the direct regeneration methods to recover S-LFP include the solid-state calcination method and hydrothermal method. Xu et al.10 utilized LiOH as the lithium source and citric acid as the reducing agent to directly repair S-LFP through a low-temperature hydrothermal and reannealing process. Jing et al. regenerated and synthesized LFP by a one-step hydrothermal method using N2H4–H2O as the reducing agent.11 The hydrothermal method has been considered to attain higher capacity and better cycling stability of regenerated LFP,4,12 but the potential for industrialization is limited by the high-temperature and high-pressure environment. Solid-state calcination is considered to be more facile compared to industrialized production. Chen et al. directly recovered S-LFP by heat treating S-LFP at different temperatures;13 Li et al. mixed Li2CO3 and S-LFP and the mixture were calcined to achieve direct regeneration of cathode materials.14 However, the performance of direct regeneration of S-LFP by solid-state calcination is not satisfactory, and a technique to obtain regenerated LFP with excellent electrochemical performance is explored.
In recent years, the application of conductive carbon materials such as nitrogen-doped carbons (NCs) to improve the electrochemical performance of LFP has been widely explored,15–18 but research on direct regeneration of S-LFP remains notably limited. More importantly, the selection of suitable nitrogen-containing precursors has been identified as a critical factor because of the low nitrogen content in the final product caused by complete evaporation, sublimation or decomposition of most nitrogen-containing compounds during the high-temperature process.19 Amino acids (AAs), as natural biomass, are one of the lowest cost and most abundant renewable resources, and AAs are more environmentally friendly compared to other macromolecular biomass resources because of natural and harmless degradation. In addition, the products maintain a high nitrogen content and the N element could be doped very homogeneously into the final product even when the AAs are carbonized at higher temperatures.20–22 In this paper, we propose a cost-effective, environmentally friendly and efficient method to directly regenerate LFP. A nitrogen-doped carbon coating was constructed on the surface of regenerated LFP by adding histidine (His), and the introduction of N atoms could raise the d-band center of Fe atoms12 and induce the electrons in the 2p orbitals of O atoms to tend to populate the Fe–O bonds, strengthening the Fe–O bonds, which resulted in better cycling stability of the regenerated LFP (R-LFP-Suc@His). Furthermore, the versatility and innovative superiority of this approach based on carbon coating with AAs are demonstrated by evaluating the electrochemical performance of regenerated LFP using sucrose and different AAs (regenerated LFP materials with glycine (Gly), arginine (Arg), and sucrose (Suc) were named R-LFP-Suc@Gly, R-LFP-Suc@Arg, and R-LFP-Suc, respectively). Compared with conventional recycling methods, our proposed method provides an alternative for industrial recycling of S-LFP cathode materials.
Results and discussion
The elimination of Li vacancies and FeLi has proven critical for the successful regeneration of S-LFP. Conventional direct regeneration methods involve the provision of a lithium source and a reducing environment to facilitate the direct regeneration of S-LFP.23,24 However, discrepancies in the extent of failure, crystal structure, and carbon content of different S-LFP samples have hindered the widespread application of traditional direct regeneration methods. Therefore, the development of a universal method capable of directly recycling various types of S-LFPs is highly desirable. In our LFP regeneration technique, the lattice of S-LFP was initially disrupted and the crystal structure of LFP was subsequently reconstructed via a redox process. Specifically, in an atmosphere of oxygen at high temperature, S-LFP was oxidized and transformed into a mixture of the phosphate and the oxide of iron(III). Simultaneously, conductive carbon and residual polyvinylidene fluoride (PVDF) on the surface of the S-LFP particles were entirely removed.25 Subsequently, the crystal structure of LFP was remodeled via a reduction reaction induced by sucrose and amino acids, while R-LFP particles were encapsulated by a uniform nanoscale nitrogen-doped carbon layer (Fig. 1a).The elemental content of different LFP powders was determined using inductively coupled plasma-optical emission spectrometry (ICP-OES), and the results are shown in Table S1.† The excessive Li+ content in the S-LFP indicates that the lithium salt from the used cathode has not been removed. An apparent decrease in lithium concentration is discerned in S-LFP after it is cleaned with dimethyl carbonate (DMC). In contrast, the recovery of elemental components from the regenerated LFP has been perceived, indicating the effectiveness of our regeneration route. The X-ray diffraction (XRD) pattern of S-LFP is shown in Fig. S1.† The presence of the FePO4 (FP) phase is observed at 30.3°, which is attributed to the absence of Li+ during the cycling process. After oxygen calcination, S-LFP is converted to Li3Fe2(PO4)3 and Fe2O3 (ref. 26 and 27) (Fig. S2†). The phase of FP disappears from the XRD patterns of the four R-LFPs (Fig. 1b), signifying the restoration of the crystal structure in the S-LFP.
The prominent peaks at 711.7 eV and 725.3 eV in the S-LFP, corresponding to Fe 2p3/2 and Fe 2p1/2, respectively, were revealed by the Fe 2p X-ray photoelectron spectroscopy (XPS) data (Fig. 1c), suggesting the presence of Fe3+ and the FP phase in the S-LFP.10,12 In the F 1s spectra (Fig. S3a†), substances including LiF (685.3 eV) and LiFP6 (687.3 eV) were detected in the S-LFP powder,28,29 suggesting the presence of lithium salt in the cathode powder, consistent with the results obtained from ICP testing. Another peak at 687.9 eV represents PVDF,30 which may have been generated during the calcination and separation of S-LFP powder. In addition, the presence of LiOH was observed in the Li 1s spectra (Fig. S3b†), which may have been produced by the contact of S-LFP with air. LiFP6, conductive carbon, and PVDF, collectively referred to as “cathode black mass” (CBM), exert a significant influence on the quality of cathode active materials, leading to the degradation of electrode material performance.31 Consequently, the elimination of CBM emerges as a pivotal technical challenge. This CBM can be effectively removed by our method, and the disappearance of the characteristic peak of the F 1s spectra of S-LFP after oxygen calcination showed that PVDF and LiPF6 were successfully removed (Fig. S3a†). For R-LFP-Suc@His and R-LFP-Suc, the main peaks of Fe 2p1/2 (at 710.5 eV and 710.4 eV) and Fe 2p3/2 (at 724.1 eV and 724 eV) were observed (Fig. 1c) at lower binding energies (compared to S-LFP), respectively, which are attributed to Fe2+, suggesting the successful regeneration of S-LFP.32 Furthermore, various nitrogen functional groups in the nitrogen-doped carbon layer were present in the N 1s spectra of R-LFP-Suc@His (Fig. 1d), including graphitic, pyridinic, pyrrolic, and oxidized nitrogen, which could be discerned at binding energies of 400.9, 398.5, 399.6, and 402.8 eV, respectively.33–35 Hence, nitrogen doping in R-LFP-His was achieved successfully, with graphitic nitrogen being the predominant species, followed by pyridinic nitrogen and pyrrolic nitrogen, thereby enhancing electronic conductivity.36
Thermogravimetric analysis combined with differential scanning calorimetry (TG-DSC) was carried out to study the pyrolysis process of the precursor of R-LFP-Suc@His and the precursor of R-LFP-Suc in a flowing Ar gas atmosphere heated from 30 °C to 900 °C at a heating rate of 5 °C min−1, respectively (Fig. 1f and g). Taking the precursor of R-LFP-Suc@His as an example, the first stage of decomposition occurs roughly around 203.76 °C, which is likely attributed to the decomposition of sucrose. Finally, around 440.05 °C, the transformation of Li3Fe2(PO4)3 and Fe2O3 to the LFP phase is initiated. The reduction process may be
| 3C + 4Li3Fe2(PO4)3 + 2Fe2O3 → 12LiFePO4 + 3CO2 |
The thermodynamics of the LFP crystal structure remodeling process is not particularly affected by the incorporation of amino acids. At temperatures exceeding 800 °C, negligible weight loss is observed, signifying the completion of the process. Therefore, around 800 °C emerges as an optimal temperature for the formation of a pure olivine-type LFP crystal. In addition, the carbon content (Fig. S2†) of R-LFP-Suc@His and R-LFP-Suc was further scrutinized using a carbon sulfur analyzer, revealing values of 4.44% and 2.97%, respectively.
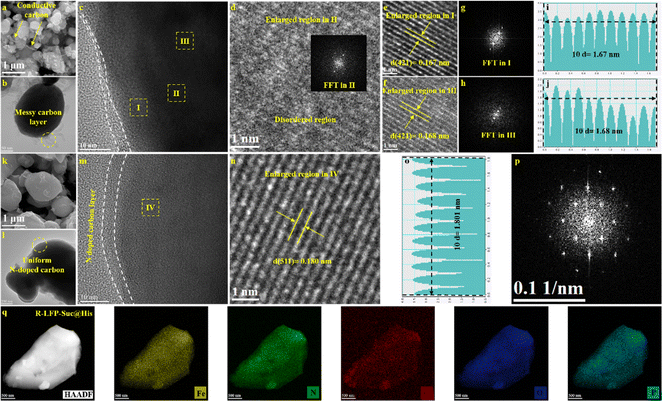
In order to analyze the morphology of S-LFP and regenerated LFP samples, scanning electron microscopy (SEM) was initially used for testing. As shown in Fig. 2a, the S-LFP particles are irregularly shaped, and a large amount of amorphous conductive carbon exists between the particles. After oxygen sintering, a large number of Fe2O3 nanodots are formed on the surface of the particles (Fig. S5†), signaling the structural evolution and phase separation of S-LFP.27 In addition, transmission electron microscopy (TEM) and high-resolution transmission electron microscopy (HRTEM) were utilized to further reveal the microstructural disparities between S-LFP and regenerated LFP. As shown in Fig. 2b and c, a disordered and inhomogeneous carbon layer is observed on the surface of S-LFP particles, which might be caused by electrolyte erosion during the cycling process and mechanical action during recycling. In addition, different regions were selected to investigate the microstructure of S-LFP. In Fig. 2d, the enlarged region II represents a disordered structure, indicating the degradation of the S-LFP crystal structure. The enlarged regions I and III (Fig. 2e and f) and their corresponding fast Fourier transform (FFT) images (Fig. 2g and h) reveal lattice stripe spacings of 0.167 nm and 0.168 nm, respectively, which are attributed to the (421) plane of LFP. For R-LFP-Suc@His (Fig. 2k–m), the particle shape is more regular, and a nanoscale uniform N-doped carbon layer is distinctly evident on the surface of the LFP particles. From the HRTEM image of Fig. 2n, aligned lattice stripes with a crystal spacing of 0.180 nm, corresponding to the (511) crystal plane of LFP, can be clearly recognized, which proves that the crystal structure has been effectively repaired. In addition, the corresponding FFT of the HRTEM image of R-LFP-Suc@His shows regular diffraction spots (Fig. 2p), further indicating the formation of a pure crystalline phase of LFP.37 Furthermore, a uniform distribution of Fe, P, O, C, and N within the R-LFP-Suc@His particles is verified in the TEM mapping (Fig. 2q). The successful and uniform nitrogen doping throughout the bulk structure is confirmed by this observation, aligning with the previously discussed XPS results. For R-LFP-Suc, distinctly aligned lattice stripes are also exhibited by the regenerated LFP particles (Fig. S5c†). The selected region I and region II (Fig. S6d and S6e†) crystallographic spacings are 0.207 nm and 0.205 nm, respectively, which corresponded to the (321) plane of LFP.
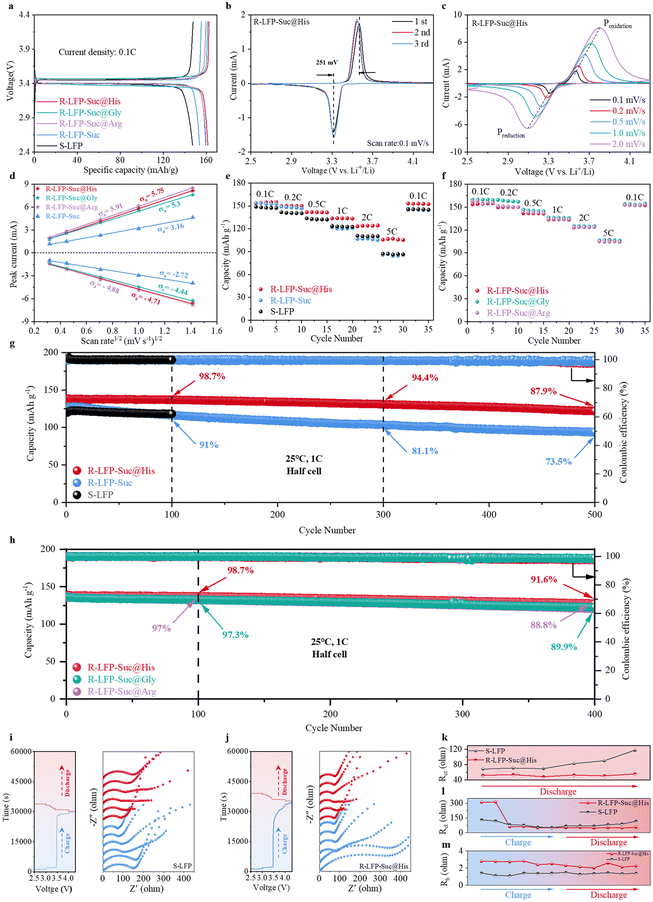
The electrochemical performance of LFP samples was evaluated using half cells in the voltage range of 2.5–4.3 V. The charge–discharge curves of the samples at 0.1C are shown in Fig. 3a. The specific capacities of discharge of R-LFP-Suc@His, R-LFP-Suc@Gly, R-LFP-Suc@Arg, and R-LFP-Suc are 161.5, 159.9, 159.4, and 153.7 mA h g−1, respectively, higher than that of S-LFP (147.3 mA h g−1). In order to test the cyclic reversibility of LFP, cyclic voltammetry (CV) was performed on the LFP at a scan rate of 0.1 mV s−1 within a voltage range of 2.5–4.3 V (Fig. 1b and S7†). The polarization voltage of R-LFP-Suc@His (251 mV) is significantly lower than that of S-LFP (286 mV), indicating superior cyclic reversibility for R-LFP-Suc@His. To elucidate the enhanced Li+ diffusion coefficient in LFP regenerated with the addition of AAs, CV tests were conducted at different scan rates (0.1 mV s−1, 0.2 mV s−1, 0.5 mV s−1, 1 mV s−1, and 2 mV s−1). The oxidation peak (Poxidation) and reduction peak (Preduction) can be observed during the charging and discharging process, respectively, where Poxidation represents the Fe2+ oxidation process and Preduction represents the Fe3+ reduction process (Fig. 3c). According to the relationship between the scan rate (ν0.5) and the peak current (Ip), the Li+ diffusion coefficient of LFP could be obtained according to the Randles–Sevcik equation:38–41
| Ip = (2.65 × 105) n1.5SDLi0.5CLiν0.5 | (1) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 Hz, employing an amplitude of 0.01 VRMS. Throughout the charging and discharging cycles, the duration was maintained at 36
000 Hz, employing an amplitude of 0.01 VRMS. Throughout the charging and discharging cycles, the duration was maintained at 36![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 s, with an interval time of 0.1 s. The equivalent circuit used to fit the experimental EIS data is shown in Fig. S12.† The fitting results are shown in Tables S3 and S4.† The Nyquist plots at high and intermediate frequencies belong to the interfacial impedance (RSEI) and charge transfer impedance (Rct), respectively. The Rct of R-LFP-Suc@His decreases with the increase of the voltage (Fig. 3j), which is attributed to the changes of the electronic and ionic conductivity of the LFP delithiation.42,43 During the discharge process, Rct experiences a marginal increase with the decreasing voltage, ultimately reaching a stable state. The resistance trend of S-LFP (Fig. 3i) changes in the same way as that of R-LFP-Suc@His. However, the superior electrochemical performance of R-LFP-Suc@His is established due to the significantly smaller Rct of R-LFP-Suc@His compared to S-LFP during the discharge process (Fig. 3k).
000 s, with an interval time of 0.1 s. The equivalent circuit used to fit the experimental EIS data is shown in Fig. S12.† The fitting results are shown in Tables S3 and S4.† The Nyquist plots at high and intermediate frequencies belong to the interfacial impedance (RSEI) and charge transfer impedance (Rct), respectively. The Rct of R-LFP-Suc@His decreases with the increase of the voltage (Fig. 3j), which is attributed to the changes of the electronic and ionic conductivity of the LFP delithiation.42,43 During the discharge process, Rct experiences a marginal increase with the decreasing voltage, ultimately reaching a stable state. The resistance trend of S-LFP (Fig. 3i) changes in the same way as that of R-LFP-Suc@His. However, the superior electrochemical performance of R-LFP-Suc@His is established due to the significantly smaller Rct of R-LFP-Suc@His compared to S-LFP during the discharge process (Fig. 3k).
The rate performance of the LFP samples is illustrated in Fig. 3e. R-LFP-Suc@His delivers discharge specific capacities of 124.3 mA h g−1 and 107 mA h g−1 at 2C and 5C, respectively, remarkably surpassing the rate performance of S-LFP, which achieves specific capacities of only 110.9 mA h g−1 and 87.3 mA h g−1 at 2C and 5C. While R-LFP-Suc recovered higher levels at low current density, the capacities at 2C and 5C are only 107 mA h g−1 and 87.2 mA h g−1. Long-term cycling at 1C was performed for LFP between 2.5 V and 4.3 V versus Li+/Li (Fig. 3g). The capacity of R-LFP-Suc@His is 138.8 mA h g−1 at 1C current density, with only a slight capacity decay after 100 cycles and 98.7% capacity retention. R-LFP-Suc@His still provides a capacity of 122 mA h g−1 after 500 cycles, with a high retention rate of 87.9%. In contrast, the capacity of R-LFP-Suc decays very rapidly, with only 73.5% capacity retention after 500 cycles. In addition, the electrochemical performance of LFP regenerated with the addition of two other AAs (Gly and Arg) is shown in Fig. 3h. Remarkably, R-LFP-Suc@Gly and R-LFP-Suc@Arg also show excellent cycling stability, with capacity retention of 89.9% and 88.8%, respectively, after 400 cycles at 1C current density. Thus, a competitive technology for future direct regeneration was established. Moreover, the optimum conditions for regenerating S-LFP were explored, and the results (Fig. S13 and S14†) show that the optimum dosage of Suc was 20 wt% (corresponding to LFP) and the calcination temperature was 800 °C.
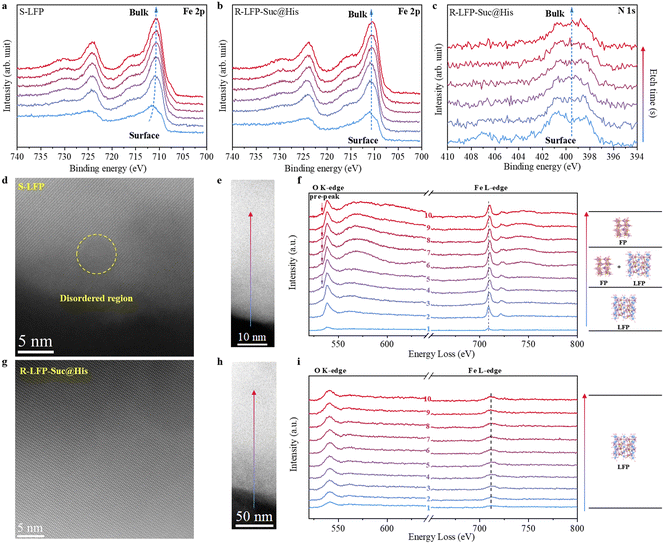
To comprehensively examine the valence states of elements in the cathode powder from the surface to the bulk, in-depth XPS analysis was performed. For S-LFP, the primary peaks of Fe 2p3/2 and Fe 2p1/2 are observed at approximately 711.9 eV and 725.5 eV, respectively (Fig. 4a), signifying the presence of Fe3+. As the etching time increases, the peaks shift towards lower binding energy, indicating a modification in the coordination environment surrounding the Fe atoms from the surface to the bulk. Non-uniform phase distribution within S-LFP is strongly suggested by this phenomenon of Fe characteristic peak shift. In contrast, for R-LFP-Suc@His, the peaks at 710.4 eV and 724 eV corresponding to Fe2+ in the Fe 2p3/2 and Fe 2p1/2 orbitals (Fig. 4b) are not shifted from the surface to the bulk, implying the absence of the FP phase within the R-LFP-Suc@His structure.11,44 In addition, the intensity of the characteristic peaks is hardly changed and the characteristic peaks are hardly shifted from the surface to the bulk in the in-depth XPS of N 1s (Fig. 4c), indicating homogeneous nitrogen doping in the nitrogen-doped carbon layer and a consistent type of doped nitrogen.
High-angle annular dark field (HAADF) scanning transmission electron microscopy (STEM) images were utilized to further understand the failure mechanism of LFP at the atomic level. Electron energy loss spectroscopy (EELS) was utilized to probe the valence states of O and Fe in a single particle from the surface to the bulk. For S-LFP (Fig. 4d), despite a certain degree of crystallinity being exhibited and a small amount of carbon coating layer retained on the surface, disordered regions are still observed on the particle surface of S-LFP. Taking a representative S-LFP particle as an example, the EELS results are shown below. The Fe L-edge spectra from the surface (point 1) to the bulk (point 10) are gradually shifted toward a higher energy loss, indicating the presence of Fe3+ in the particle bulk (Fig. 4f).45 The intermediate region is primarily combined with mixed phases of FP and LFP. In addition, the O K-edge pre-peak appears gradually from the surface to the bulk, also indicating the existence of FP inside the particles.46 The O K-edge is generated by the excitation of the 1s orbital into the unoccupied O 2p orbitals,47 and the O pre-peak is characteristic of phosphates containing Fe3+. An ordered crystallographic structure of R-LFP-Suc@His particles is depicted in Fig. 4g. The Fe L-edge spectra exhibit consistent features across the entire depth, and the disappearance of the O pre-peak (Fig. 4i) suggests the exclusive presence of Fe2+ in the regenerated LFP particles. The successful regeneration of the S-LFP is thereby confirmed, in harmony with the results obtained from previous in-depth XPS.
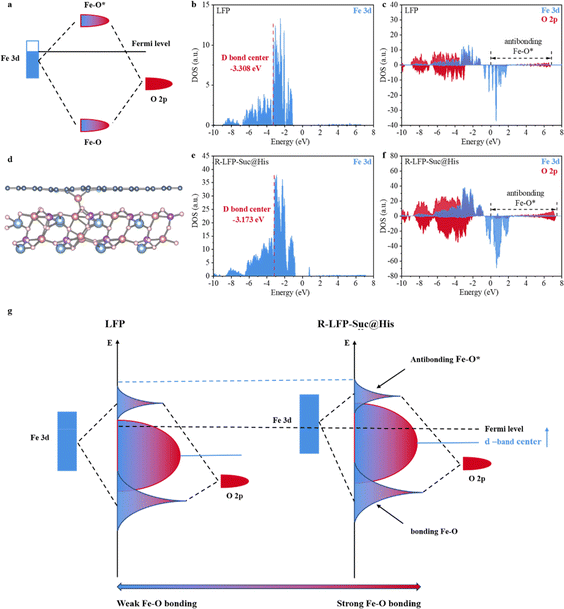
Density Functional Theory (DFT) calculations were employed to elucidate the reasons for the improved cycling performance of R-LFP-Suc@His. The bonding forms of Fe 3d and O 2p are shown in Fig. 5a. The two orbitals interact to re-form the Fe–O bonding orbitals with lower energy states and the Fe–O* antibonding orbitals with higher energy states. The models of LFP with a nitrogen-doped carbon layer and the original LFP are illustrated in Fig. 5d and S15,† respectively. The d-band center of R-LFP-Suc@His is positioned at −3.173 eV, as indicated by the partial density of states (pDOS) in Fig. 5e, implying the successful elevation of the d-band center of Fe through the introduction of elemental N. The d-band center theory has been used to describe bonding interactions between transition-metal atoms and neighboring atoms.48–50 Specifically, the increase of the d-band center leads to a more difficult occupation of the antibonding orbitals, which results in a stronger bonding between the atoms.51 pDOS of Fe 3d and O 2p (Fig. 5e and f) further proves the above theory. The antibonding Fe–O* orbitals of R-LFP-Suc@His are farther away from the Fermi energy level as compared to the LFP, and the structural model is shown in Fig. 5g. The Fe–O bonding was strengthened and the cycling performance of the R-LFP-Suc@His was significantly improved because of the tendency of electrons from the O 2p orbitals to populate the Fe–O bonding orbitals.12 The theoretical computational predictions are effectively supported by our experimental results. Therefore, AAs play a very important role in the regeneration of S-LFP.
Conclusion
In summary, direct regeneration of S-LFP was achieved through a redox process, and green natural biomass amino acids were utilized to generate a coating of nanoscale nitrogen-doped carbon layer on the surface of the regenerated LFP while removing the original disordered carbon layer. The superiority of AAs was demonstrated by various structural characterizations and electrochemical performance tests. The regenerated LFP under optimized conditions exhibited attractive potential for application as a lithium-ion battery cathode. Our direct regeneration method is considered to be economically efficient and environmentally friendly, offering a promising approach for the recycling of spent lithium-ion batteries in the future.Experimental section
Materials and spent LiFePO4 cell cathode materials harvesting
Chemical reagents including histidine (C6H9N3O2), glycine (C2H5NO2) and arginine (C6H14N4O2) were purchased from Aladdin (Shanghai, China). Sucrose (C12H22O11) was purchased from Maclean (Shanghai, China). Acetone (C3H6O) and N-methyl-pyrrolidone (NMP) were purchased from Sinopharm Chemical Reagent Co., Ltd (Shanghai, China). Polyvinylidene fluoride (PVDF) was purchased from Guangdong Canrd New Energy Technology Co. Ltd. The spent LFP cathode sheets utilized in the experiments were acquired from a local company. The cathode sheets were initially calcined under an Ar gas atmosphere at 450 °C for 1 hour. Then the cathode powder was separated from the aluminum foil by vibration. The collected spent LFP cathode powder was stored in a glovebox.LiFePO4 regeneration (with carbon coating generated from sucrose)
The collected spent LFP powder was initially calcined at 700 °C under an oxygen atmosphere to obtain a red powder. The above-obtained red powder (5 g) and sucrose (0.861 g) were added to 30 mL of acetone and the mixture was ball-milled in a planetary mill with a certain rotation speed for 10 h. After drying, the precursor was calcined under an Ar atmosphere at 800 °C for 5 h with a heating rate of 5 °C min−1 to obtain R-LFP-Suc.LiFePO4 regeneration (with a nitrogen-doped carbon coating generated from sucrose and additives of AAs)
The collected spent LFP powder was initially calcined at 700 °C under an oxygen atmosphere to obtain a red powder. The red powder (5 g), sucrose (0.861 g) and histidine (0.162 g) were added to 30 mL of acetone. The mixture was then ball-milled in a planetary mill with a certain rotation speed for 10 h. After drying, the precursor was calcined at 800 °C under an Ar atmosphere for 5 h, with a heating rate of 5 °C min−1 to obtain R-LFP-Suc@His. R-LFP-Suc@Gly was prepared by adding the red powder (5 g), sucrose (0.861 g), and glycine (0.234 g) to the acetone, followed by the same regeneration procedure as for R-LFP-Suc@His. R-LFP-Suc@Arg was prepared by adding the red powder (5 g), sucrose (0.861 g), and arginine (0.136 g) to the acetone, followed by the same regeneration procedure as for R-LFP-Suc@His.Materials characterization
The crystal structure was determined by X-ray diffraction (XRD; Bruker D8 Advance, Germany) with Cu Kα radiation in the 2θ range 10–80°. The elemental composition of the samples was determined by inductively coupled plasma-optical emission spectrometry (ICP-OES; Agilent 730, USA). The cathode powder was analyzed by X-ray photoelectron spectroscopy (XPS; Thermo Scientific ESCALAB Xi+, USA). A carbon sulfur analyzer (C/S analyzer; Leco CS844, USA) was used to measure the carbon content of LFP. The decomposition of LFP precursors was analyzed using a simultaneous thermal analyzer (TG-DSC; PerkinElmer STA 6000, USA) with a heating rate of 5 °C min−1 from 30 °C to 900 °C under an Ar atmosphere. The morphology and microstructure of the cathode powder were observed by scanning electron microscopy (SEM; TESCAN MIRA LMS, Czech Republic) and high-resolution transmission electron microscopy (HRTEM; FEI Talos F200x, USA).Electrochemical characterization
To evaluate the electrochemical performance of LFP samples, different LFP powders were homogeneously mixed with polyvinylidene fluoride (PVDF) and Super P in NMP at a mass ratio of 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and the mixed slurry was coated on aluminum foil and dried under vacuum at 120 °C for 12 h. The positive electrode sheets were cut into small slices with a mass loading of about 3 mg cm−2. The coin cell (2025) was assembled with Li metal discs as counter electrodes, 4102EC as the electrolyte, and Celgard 2500 film as the separator. Constant current measurements were performed on a LAND cell test system (Wuhan Land, CT2001A, China) with a voltage range of 2.5–4.3 V. CV and electrochemical impedance spectroscopy (EIS) tests were performed using an electrochemical workstation (Multi Autolab M204, Netherlands).
1, and the mixed slurry was coated on aluminum foil and dried under vacuum at 120 °C for 12 h. The positive electrode sheets were cut into small slices with a mass loading of about 3 mg cm−2. The coin cell (2025) was assembled with Li metal discs as counter electrodes, 4102EC as the electrolyte, and Celgard 2500 film as the separator. Constant current measurements were performed on a LAND cell test system (Wuhan Land, CT2001A, China) with a voltage range of 2.5–4.3 V. CV and electrochemical impedance spectroscopy (EIS) tests were performed using an electrochemical workstation (Multi Autolab M204, Netherlands).
Data availability
All data that support the findings of this study are included in the article and the ESI† file. All raw data generated during the current study are available from the corresponding authors upon request.Author contributions
W. J. is first author. T. S. and W. J. conceived the project and methodology. W. J. contributed to the experiment design, data analysis, and writing the original draft with the assistance of J. S., H. Q., W. F., S. W., and C. D. T. S. revised the manuscript. All authors critically reviewed the manuscript and approved the final manuscript.Conflicts of interest
The authors declare no competing interests.Acknowledgements
This project was supported by the National Natural Science Foundation of China (NSFC; No. 52107224 and 52077096) and National Key R&D Program of China (2022YFB2404300).References
- G. Harper, R. Sommerville, E. Kendrick, L. Driscoll, P. Slater, R. Stolkin, A. Walton, P. Christensen, O. Heidrich, S. Lambert, A. Abbott, K. Ryder, L. Gaines and P. Anderson, Nature, 2019, 575, 75–86 CrossRef CAS PubMed.
- J. Li and Z.-F. Ma, Chem, 2019, 5, 3–6 CAS.
- J. Deng, C. Bae, A. Denlinger and T. Miller, Joule, 2020, 4, 511–515 CrossRef.
- M. Chen, X. Ma, B. Chen, R. Arsenault, P. Karlson, N. Simon and Y. Wang, Joule, 2019, 3, 2622–2646 CrossRef CAS.
- E. Fan, L. Li, Z. Wang, J. Lin, Y. Huang, Y. Yao, R. Chen and F. Wu, Chem. Rev., 2020, 120, 7020–7063 CrossRef CAS PubMed.
- J. J. Roy, S. Rarotra, V. Krikstolaityte, K. W. Zhuoran, Y. D. I. Cindy, X. Y. Tan, M. Carboni, D. Meyer, Q. Y. Yan and M. Srinivasan, Adv. Mater., 2022, 34, 2103346 CrossRef CAS.
- J. Neumann, M. Petranikova, M. Meeus, J. D. Gamarra, R. Younesi, M. Winter and S. Nowak, Adv. Energy Mater., 2022, 12, 2102917 CrossRef CAS.
- M. S. Islam, D. J. Driscoll, C. A. J. Fisher and P. R. Slater, Chem. Mater., 2005, 17, 5085–5092 CrossRef CAS.
- A. K. Padhi, K. S. Nanjundaswamy, C. Masquelier, S. Okada and J. B. Goodenough, J. Electrochem. Soc., 2019, 144, 1609–1613 CrossRef.
- P. Xu, Q. Dai, H. Gao, H. Liu, M. Zhang, M. Li, Y. Chen, K. An, Y. S. Meng, P. Liu, Y. Li, J. S. Spangenberger, L. Gaines, J. Lu and Z. Chen, Joule, 2020, 4, 2609–2626 CrossRef CAS.
- Q. Jing, J. Zhang, Y. Liu, W. Zhang, Y. Chen and C. Wang, ACS Sustain. Chem. Eng., 2020, 8, 17622–17628 CrossRef CAS.
- K. Jia, J. Ma, J. Wang, Z. Liang, G. Ji, Z. Piao, R. Gao, Y. Zhu, Z. Zhuang, G. Zhou and H. M. Cheng, Adv. Mater., 2022, 35, 2208034 CrossRef PubMed.
- J. Chen, Q. Li, J. Song, D. Song, L. Zhang and X. Shi, Green Chem., 2016, 18, 2500–2506 RSC.
- X. Li, J. Zhang, D. Song, J. Song and L. Zhang, J. Power Sources, 2017, 345, 78–84 CrossRef CAS.
- J.-P. Jegal, K.-C. Kim, M. S. Kim and K.-B. Kim, J. Mater. Chem. A, 2014, 2, 9594–9599 RSC.
- Y. Liu, J. Gu, J. Zhang, F. Yu, L. Dong, N. Nie and W. Li, J. Power Sources, 2016, 304, 42–50 CrossRef CAS.
- Y. Zhang, Y. D. Huang, X. C. Wang, Y. Guo, D. Z. Jia and X. C. Tang, J. Power Sources, 2016, 305, 122–127 CrossRef CAS.
- B. Wang, W. Al Abdulla, D. Wang and X. S. Zhao, Energy Environ. Sci., 2015, 8, 869–875 RSC.
- S. Zhang, S. Tsuzuki, K. Ueno, K. Dokko and M. Watanabe, Angew. Chem., Int. Ed., 2014, 54, 1302–1306 CrossRef PubMed.
- X. Gao, Z. Chen, Y. Yao, M. Zhou, Y. Liu, J. Wang, W. D. Wu, X. D. Chen, Z. Wu and D. Zhao, Adv. Funct. Mater., 2016, 26, 6649–6661 CrossRef CAS.
- M. Kurashvili, I. Polishchuk, A. Lang, S. Strohmair, A. F. Richter, S. Rieger, T. Debnath, B. Pokroy and J. Feldmann, Adv. Funct. Mater., 2022, 32, 2202121 CrossRef CAS.
- T. Wang, L. Wang, D. Wu, W. Xia, H. Zhao and D. Jia, J. Mater. Chem. A, 2014, 2, 8352–8361 RSC.
- K. K. Jena, A. AlFantazi and A. T. Mayyas, Energy Fuels, 2021, 35, 18257–18284 CrossRef CAS.
- H. Gao, D. Tran and Z. Chen, Curr. Opin. Electrochem., 2022, 31, 100875 CrossRef CAS.
- C. Yang, J.-l. Zhang, Q.-k. Jing, Y.-b. Liu, Y.-q. Chen and C.-y. Wang, Int. J. Miner., Metall. Mater., 2021, 28, 1478–1487 CrossRef CAS.
- Y. Zou, J. Cao, H. Li, W. Wu, Y. Liang and J. Zhang, Ind. Chem. Mater., 2023, 1, 254–261 RSC.
- Z. Zeng, P. Xu, J. Li, C. Yi, W. Zhao, W. Sun, X. Ji, Y. Yang and P. Ge, Adv. Funct. Mater., 2023, 2308671, DOI:10.1002/adfm.202308671.
- W. Yu, Z. Yu, Y. Cui and Z. Bao, ACS Energy Lett., 2022, 7, 3270–3275 CrossRef CAS.
- S. T. Oyakhire, H. Gong, Y. Cui, Z. Bao and S. F. Bent, ACS Energy Lett., 2022, 7, 2540–2546 CrossRef CAS.
- S. Q. Jiang, Q. Gao, X. G. Li, C. Z. Deng, S. N. Akanyange, J. Qiu and X. N. Zhu, Sep. Purif. Technol., 2023, 324, 124601 CrossRef CAS.
- V. Gupta, X. L. Yu, H. P. Gao, C. Brooks, W. K. Li and Z. Chen, Adv. Energy Mater., 2023, 13, 2203093 CrossRef CAS.
- M. Chen, W. Hua, J. Xiao, D. Cortie, W. Chen, E. Wang, Z. Hu, Q. Gu, X. Wang, S. Indris, S. L. Chou and S. X. Dou, Nat. Commun., 2019, 10, 1480 CrossRef PubMed.
- Y. F. Deng, Y. Xie, K. X. Zou and X. L. Ji, J. Mater. Chem. A, 2016, 4, 1144–1173 RSC.
- K. Jurewicz, K. Babeł, A. Źiółkowski and H. Wachowska, Electrochim. Acta, 2003, 48, 1491–1498 CrossRef CAS.
- H. Jeong, H. J. Kim, Y. J. Lee, J. Y. Hwang, O.-K. Park, J.-H. Wee, C.-M. Yang, B.-C. Ku and J. K. Lee, Mater. Lett., 2015, 145, 273–278 CrossRef CAS.
- J. Zhang, Y. Yan, X. Wang, Y. Cui, Z. Zhang, S. Wang, Z. Xie, P. Yan and W. Chen, Nat. Commun., 2023, 14, 3701 CrossRef CAS PubMed.
- X. L. Wu, Y. G. Guo, J. Su, J. W. Xiong, Y. L. Zhang and L. J. Wan, Adv. Energy Mater., 2013, 3, 1155–1160 CrossRef CAS.
- K. Jia, J. Wang, J. Ma, Z. Liang, Z. Zhuang, G. Ji, R. Gao, Z. Piao, C. Li, G. Zhou and H.-M. Cheng, Nano Lett., 2022, 22, 8372–8380 CrossRef CAS PubMed.
- J. Zhang, J. Zhang, X. Ou, C. Wang, C. Peng and B. Zhang, ACS Appl. Mater. Interfaces, 2019, 11, 15507–15516 CrossRef CAS.
- R. Wang, X. Chen, Z. Y. Huang, J. L. Yang, F. S. Liu, M. H. Chu, T. C. Liu, C. Q. Wang, W. M. Zhu, S. K. Li, S. N. Li, J. X. Zheng, J. Chen, L. H. He, L. Jin, F. Pan and Y. G. Xiao, Nat. Commun., 2021, 12, 3085 CrossRef CAS PubMed.
- G. Yao, X. Zhang, Y. Yan, J. Zhang, K. Song, J. Shi, L. Mi, J. Zheng, X. Feng and W. Chen, J. Energy Chem., 2020, 50, 387–394 CrossRef.
- Y. Zhang, Y. Katayama, R. Tatara, L. Giordano, Y. Yu, D. Fraggedakis, J. G. Sun, F. Maglia, R. Jung, M. Z. Bazant and Y. Shao-Horn, Energy Environ. Sci., 2020, 13, 183–199 RSC.
- S. Oswald, D. Pritzl, M. Wetjen and H. A. Gasteiger, J. Electrochem. Soc., 2020, 167, 100511 CrossRef CAS.
- G. J. Ji, J. X. Wang, Z. Liang, K. Jia, J. Ma, Z. F. Zhuang, G. M. Zhou and H. M. Cheng, Nat. Commun., 2023, 14, 584 CrossRef CAS PubMed.
- L. Laffont, C. Delacourt, P. Gibot, M. Y. Wu, P. Kooyman, C. Masquelier and J. M. Tarascon, Chem. Mater., 2006, 18, 5520–5529 CrossRef CAS.
- M. K. Kinyanjui, P. Axmann, M. Wohlfahrt-Mehrens, P. Moreau, F. Boucher and U. Kaiser, J. Phys.: Condens. Matter, 2010, 22, 275501 CrossRef CAS PubMed.
- U. Boesenberg, F. Meirer, Y. Liu, A. K. Shukla, R. Dell'Anna, T. Tyliszczak, G. Chen, J. C. Andrews, T. J. Richardson, R. Kostecki and J. Cabana, Chem. Mater., 2013, 25, 1664–1672 CrossRef CAS PubMed.
- J. K. Nørskov, F. Abild-Pedersen, F. Studt and T. Bligaard, Proc. Natl. Acad. Sci. U. S. A., 2011, 108, 937–943 CrossRef.
- B. Hammer and J. K. Norskov, Nature, 1995, 376, 238–240 CrossRef CAS.
- S. Bhattacharjee, U. V. Waghmare and S.-C. Lee, Sci. Rep., 2016, 6, 35916 CrossRef CAS PubMed.
- Z. Wang, J. Huang, L. Wang, Y. Liu, W. Liu, S. Zhao and Z. Q. Liu, Angew. Chem., Int. Ed., 2022, 61, 134 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4ta01098a |
| This journal is © The Royal Society of Chemistry 2024 |