Electrochemically promoted diamidation of alkenes to dihydroimidazole skeleton†
Guo-Ao
Wang
,
Kai-Wei
Chen
,
Yu-Da
Huang
,
Yu-Yuan
Zhang
,
Xiu-Jin
Meng
*,
Fei-Hu
Cui
*,
Ying-Ming
Pan
 and
Hai-Tao
Tang
and
Hai-Tao
Tang
 *
*
State Key Laboratory for Chemistry and Molecular Engineering of Medicinal Resources, Key Laboratory for Chemistry and Molecular Engineering of Medicinal Resources (Ministry of Education of China), School of Chemistry and Pharmaceutical Sciences of Guangxi Normal University, Guilin 541004, People's Republic of China. E-mail: xiujinmeng@mailbox.gxnu.edu.cn; cuifeihuhao@163.com; httang@gxnu.edu.cn
First published on 17th October 2024
Abstract
The dihydroimidazole framework is a valuable heterocyclic compound widely found in natural products, ligands, molecular catalysts, pesticides, etc. The construction of a dihydroimidazole framework from alkenes is an attractive synthesis approach. Herein, we report a simple and environmentally friendly electrocatalytic diamination reaction of alkenes. The reaction involves constructing a dihydroimidazole framework from readily available alkenes, CH3CN, and H2O. CH3CN serves as a solvent and a nitrogen source, whereas H2O provides an oxygen source without the need for catalysts and oxidants. The synthesized 3,4-dihydroimidazole compound can be further hydrolyzed into vicinal diamines. As far as we know, the synthesis of dihydroimidazole frameworks through electrochemical Ritter-type diamination of alkenes has not been reported yet.
Introduction
The 1,2-diamine motif is widely present in natural products, pharmaceutical compounds, and molecular catalysts (Fig. 1).1 Difunctionalization of alkenes2 with amination reagents, which is one of the most straightforward and practical strategies3 for assembling scaffolds, has achieved remarkable progress4 in the past few decades. However, these reported methods often require the utilization of transition metal reagents and even stoichiometric amounts of osmium or cobalt.4a,5 Moreover, the use of stoichiometric amounts of chemical oxidants (e.g., iodine(III) reagents or organic peroxides)4d,6 or azide reagents4b,c,7 raises costs and environmental and safety issues, whereas the use of strong oxidizers may produce environmentally hazardous by-products and pose an explosion hazard when used with azide sources, particularly for large-scale applications.8 In addition, they are often limited in substrate scope and sometimes require special amination reagents (e.g., diaziridinone and its analogs4e,9 or azido-iodine compounds).4c Therefore, developing highly efficient, mild, and green synthetic strategies is still desirable, and an additional substrate combination for the 1,2-diamine motif should be found to address the aforementioned drawbacks.Electrochemistry offers an environmentally friendly, gentle, and efficient alternative to traditional chemical methods of redox conversion.10 In the alternative method, common organic starting materials can lose or gain electrons at the electrode surface without the need for an external redox agent; thus, highly reactive intermediates are readily produced. Electrochemistry allows the precise external control of chemoselectivity and flux of reactive intermediates by regulating the applied current or potential. Compared with the energy efficiency in traditional chemical methods, that in the alternative method is maximized because of the latter's precise external control. Recently, electrochemical anodic oxidation has shown great potential in the construction of a 1,2-diamine motif.
For instance, Xu and his colleagues11 reported an electrochemical reaction of synthesizing 1,2-diamine compounds with diastereoselectivity using aryl alkenes and sulfonamides mediated by tris (2,4-dibromophenyl) amine. Lin's group7b has also made remarkable progress in the electrocatalytic diazidation of alkenes (Scheme 1). Despite the remarkable progress made above, issues such as limited types of nitrogen sources and 1,2-diamine structures may exist. Dihydroimidazole frameworks are a class of important nitrogen-containing heterocyclic compounds12 with the structural characteristics of two nitrogen atoms and two sp3 C–H bonds, which can be easily synthesized by 1,2-diamination of alkenes in theory. However, electrochemical synthesis of dihydroimidazole has not been reported because of the lack of well-matched nitrogen sources, alkenes, and electrochemical conditions.
Herein, we report a simple and environmentally friendly electrocatalytic diamination reaction of alkenes. The reaction involves constructing a dihydroimidazole framework from readily available alkenes, CH3CN, and H2O. CH3CN serves as a solvent and a nitrogen source,13 whereas H2O provides an oxygen source without the need for catalysts and oxidants. The synthesized 3,4-dihydroimidazole compound can be further hydrolyzed into vicinal diamines. As far as we know, the synthesis of dihydroimidazole frameworks through electrochemical Ritter-type diamination of alkenes has not been reported yet.
Results and discussion
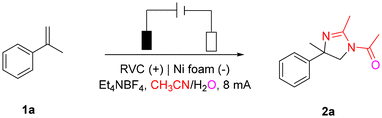
We used 2-phenyl-1-propene (1a) as the model substrate to establish the optimal conditions for this reaction by optimizing the reaction conditions. Under the constant current condition of 8 mA, the reaction proceeded smoothly in a three-necked flask with Et4NBF4 as the electrolyte, reticulated vitreous carbon (RVC) electrode as the anode, and Ni foam electrode as the cathode. The required product 2a (Table 1, entry 1) was obtained with an 82% yield. The yield decreased when the cathode was replaced with platinum or nickel sheets (Table 1, entries 2 and 3), whereas the yield of 2a was 59% when the cathode and anode were replaced with platinum sheets (Table 1, entry 4). However, the yield of 2a was only 35% when the cathode and anode were replaced with carbon rods (Table 1, entry 5). Increasing or decreasing the current would decrease the reaction activity (Table 1, entries 6 and 7). Only trace amounts of product 2a were detected when Et4NCl, Et4NBr, and Et4NI were used as electrolytes (Table 1, entry 8). The reaction could not proceed without current (Table 1, entry 9). In summary, the optimal conditions established are as follows: RVC as anode, foam nickel electrode as cathode, Et4NBF4 (4 equiv.) as electrolyte, extra dry CH3CN and H2O as solvent (extra dry CH3CN = 6 mL; H2O = 5 μL), and reaction at room temperature with a constant current of 8 mA for 3–6 h.| Entry | Variation from standard conditions | Yieldb,c (%) |
|---|---|---|
| a Standard conditions: RVC (100 PPI, 1 cm × 1 cm × 1.2 cm) anode, Ni foam (1 cm × 2 cm × 0.5 cm) cathode, extra dry CH3CN and H2O as solvent (extra dry CH3CN = 6 mL, H2O = 5 μL), undivided cell, constant current = 8 mA, 1a (0.2 mmol, 1 equiv.), Et4NBF4 (0.8 mmol, 4 equiv.), and room temperature. b Isolated yields. c Unless otherwise specified, extra dry CH3CN with a water content of ≤10 ppm was used. | ||
| 1 | None | 82 |
| 2 | RVC (+)|Pt (−) instead of RVC (+)|Ni foam (−) | 68 |
| 3 | RVC (+)|Ni (−) instead of RVC (+)|Ni foam (−) | 61 |
| 4 | Pt (+)|Pt (−) instead of RVC (+)|Ni foam (−) | 59 |
| 5 | C (+)|C (−) instead of RVC (+)|Ni foam (−) | 35 |
| 6 | 10 mA instead of 8 mA | 64 |
| 7 | 6 mA instead of 8 mA | 62 |
| 8 | Et4NCl, Et4NBr, and Et4NI instead of Et4NBF4 | Trace |
| 9 | No electricity | 0 |
Under optimal conditions, the substrate range was explored. The results are shown in Schemes 2 and 3. First, the model substrate 2-phenyl-1-propene (1a) presented the target product in 82% yield, and it could obtain the target product with 76% (2b), 71% (2c), 73% (2d), 72% (2e), and 61% (2f) yields when the benzene ring was para-carried with groups such as fluorine, chlorine, bromine, trifluoromethyl, and nitro groups. When halogen groups such as fluorine and bromine groups exist in the meta position of the aromatic ring, the target product can be obtained with yields of 83% (2g) and 66% (2h), respectively. When a methyl group exists in the meta position of the aromatic ring, the target product can be obtained with a yield of 55% (2j). However, when the CH3CN was replaced with butyronitrile, the target product (2k) could be obtained in 61% yield. When halogen groups such as chlorine are found in the adjacent position of the aromatic ring, the target product can be obtained with a yield of 65% (2i). When the R1 group is replaced with ethyl, propyl, or phenyl, the yield of the target product gradually decreases as the steric hindrance increases. The target product can be obtained with yields of 74% (2l), 66% (2m), and 61% (2n). In addition, electrochemical methods can be used to modify natural products, such as L-menthol, 1-adamantane methanol, and 1-cyclododecanol, affording the corresponding derivatives (2o–2q) in 40%–55% yields.
When we replace 2-phenyl-1-propene (1a) with styrene (3a), the target product can still be obtained with a 75% (4a) yield. On the benzene ring of styrene, a series of different substitutions can be successfully carried out for electrochemical 1,2-diamination reactions, including fluorine, chlorine, bromine, trifluoromethyl, and methyl. Alkenes with 2,6-disubstituted benzene rings are also suitable substrates (4i). We also confirmed the structure of (4j) through X-ray diffraction analysis. In addition, the electrochemical reaction of 1,2-diamination is compatible with chloromethyl groups (4e). After switching to styrene, the yield is generally slightly lower than that of alkenes with substituents at the α-position. The possible reason is that when substituents exist at the α-position, stabilizing carbocations is easy, resulting in a high electrochemical reaction activity for 1,2-diamination. This finding is consistent with our speculated mechanism.
We conducted a gram-scale conversion of the electrocatalytic diamination reaction of alkenes under constant current conditions of 80 mA to demonstrate further the synthetic application of this electrochemical method. The reaction of alkene 1a with CH3CN and H2O was completed at a scale of grams, and the target product 2a (0.71 g) was obtained with a 41% yield (Scheme 4a). In addition, 1,2-diamine compounds are important synthetic units, such as drug intermediates, catalysts, and ligands. Therefore, synthesizing 1,2-diamine compounds from readily available alkenes is important. We dissolved 0.2 mmol 2a in potassium hydroxide, methanol, and ethylene glycol, and it was heated under reflux. After the reaction was completed, the diamine compound 2aaa was obtained with a 67% yield. In addition, 4e can be used to modify BOC Glycine, providing the corresponding derivative with a yield of 45% (4ee).
We conducted several control experiments to gain a deep understanding of the reaction mechanism (Scheme 5). When 5 equiv. BHT and TEMPO are added to the reaction, only trace amounts of product 2a are detected, indicating that the reaction may involve the free radical pathway (Scheme 5a). We conducted an H218O labeling experiment to verify the source of oxygen in product 2a. When H218O (5 μL) is added to the reaction, the results show that the 18O labeled product 2aa can be detected by HRMS analysis (Scheme 5b and Fig. S2†). The results indicate that H2O may provide oxygen atoms for the formation of the target product 2a. Finally, we conducted radical clock experiments using compounds 5a and 6a. Products 5aa and 6aa can be detected by HRMS analysis, indicating that the reaction is undergoing a free radical pathway (see ESI† for additional details).
We also conducted cyclic voltammetry analysis on compound 1a. The test results show that an irreversible oxidation peak is observed at 2.45 V (relative to Ag/AgCl) in the cyclic voltammogram of 1a, indicating that 1a may be oxidized at the anode (RVC).
On the basis of the above experimental results and previous literature reports,14 we proposed a possible mechanism (Scheme 6). 2-Phenyl-1-propene 1a is oxidized at the anode (RVC) to form alkene radical cation I, which is captured by the CH3CN to form intermediate II. Then, intermediate II loses an electron to intermediate III. Intermediate III is captured by a molecular CH3CN to form intermediate IV, which then forms a carbocation intermediate. After hydrolysis, it forms amide product VIII, which is then cyclized to obtain the target product 2a. This process is essentially a Ritter reaction,15 with the difference being the methods and synthetic precursors for generating carbocations.
Conclusions
We have developed a simple and environmentally friendly electrochemically driven 1,2-diamination reaction of alkenes, which is an effective method for constructing 1,2-diamine framework compounds from readily available alkenes, CH3CN, and H2O. The reaction does not require additional catalysts and oxidants, and the raw materials are readily available. We hope that this protocol can provide a complementary way for dihydroimidazole compounds and be widely applied in modern synthetic chemistry and pharmaceutical research.Data availability
The data supporting this article have been included as part of the ESI.†Conflicts of interest
There are no conflicts to declare.Acknowledgements
We thank the Guangxi Natural Science Foundation of China (2021GXNSFFA220005), National Natural Science Foundation of China (22061003, 22301048), the Natural Science Foundation of Guangxi Normal University (No. 2021JC004) for financial support.References
- (a) D. Lucet, T. L. Gall and C. Mioskowski, The Chemistry of Vicinal Diamines, Angew. Chem., Int. Ed., 1998, 37, 2580–2627 CrossRef CAS; (b) S. R. S. S. Kotti, C. Timmons and G. Li, Vicinal Diamino Functionalities as Privileged Structural Elements in Bio-logically Active Compounds and Exploitation of their Synthetic Chemistry, Chem. Biol. Drug Des., 2006, 67, 101–114 CrossRef CAS PubMed; (c) F. Cardona and A. Goti, Metal-Catalysed 1,2-Diamination Reactions, Nat. Chem., 2009, 1, 269–275 CrossRef CAS PubMed.
- (a) A. Corma, Introduction: Chemicals from Coal, Alkynes, and Biofuels, Chem. Rev., 2014, 114, 1545–1546 CrossRef CAS PubMed; (b) M. Crespo-Quesada, F. Cárdenas-Lizana, A.-L. Dessimoz and L. Kiwi-Minsker, Modern Trends in Catalyst and Process Design for Alkyne Hydrogenations, ACS Catal., 2012, 2, 1773–1786 CrossRef CAS; (c) M. Eggersdorfer, D. Laudert, U. Létinois, T. McClymont, J. Medlock, T. Netscher and W. Bonrath, One Hundred Years of Vitamins—a Success Story of the Natural Sciences, Angew. Chem., Int. Ed., 2012, 51, 12960–12990 CrossRef CAS PubMed; (d) R. Bai, G. He, L. Li, T. Zhang, J. Li, X. Wang, X. Wang, Y. Zou, D. Mei, A. Corma and J. Yu, Encapsulation of Palladium Carbide Subnanometric Species in Zeolite Boosts Highly Selective Semihydrogenation of Alkynes, Angew. Chem., Int. Ed., 2023, 62, e202313101 CrossRef CAS PubMed; (e) B. Dong, J. Shen and L.-G. Xie, Recent Developments on 1,2-Difunctionalization and Hydrofunctionalization of Unactivated Alkenes and Alkynes Involving C–S Bond Formation, Org. Chem. Front., 2023, 10, 1322–1345 RSC; (f) K. Yang, J. Liu, D. Fu, L. Niu, S.-J. Li and Y. Lan, A Theoretical Study of Selective Radical Relay and Coupling Reactions for Alkene Difunctionalization, Org. Chem. Front., 2023, 10, 4336–4341 RSC.
- (a) R. Fu, M. Xu, Y. Wang, X. Wu and X. Bao, Organo-Photocatalytic Anti-Markovnikov Hydroamidation of Alkenes with Sulfonyl Azides: A Combined Experimental and Computational Study, Angew. Chem., Int. Ed., 2024, 63, e202406069 CrossRef CAS PubMed; (b) L. Song, N. Fu, B. G. Ernst, W. H. Lee, M. O. Frederick, R. A. DiStasio and S. Lin, Dual Electrocatalysis Enables Enantioselective Hydrocyanation of Conjugated Alkenes, Nat. Chem., 2020, 12, 747–754 CrossRef CAS PubMed.
- (a) P. N. Becker, M. A. White and R. G. Bergman, A New Method for 1,2-Diamination of Alkenes Using Cyclopentadienylnitrosylcobalt Dimer/NO/LiAlH4, J. Am. Chem. Soc., 1980, 102, 5676–5677 CrossRef CAS; (b) B. Zhang and A. Studer, Copper-Catalyzed Intermolecular Aminoazidation of Alkenes, Org. Lett., 2014, 16, 1790–1793 CrossRef CAS PubMed; (c) G. Fumagalli, P. T. G. Rabet, S. Boyd and M. F. Greaney, Three-Component Azidation of Styrene-Type Double Bonds: Light-Switchable Behavior of a Copper Photoredox Catalyst, Angew. Chem., Int. Ed., 2015, 54, 11481–11484 CrossRef CAS PubMed; (d) S.-J. Shen, C.-L. Zhu, D.-F. Lu and H. Xu, Iron-Catalyzed Direct Olefin Diazidation via Peroxyester Activation Promoted by Nitrogen-Based Ligands, ACS Catal., 2018, 8, 4473–4482 CrossRef CAS PubMed; (e) Y. Wen, B. Zhao and Y. Shi, Cu(I)-Catalyzed Diamination of Disubstituted Terminal Olefins: An Approach to Potent NK1 Antagonist, Org. Lett., 2009, 11, 2365–2368 CrossRef CAS PubMed.
- A. O. Chong, K. Oshima and K. B. Sharpless, Synthesis of Dioxobis(tert-alkylimido)osmium(VIII) and Oxotris(tert-alkylimido)osmium(VIII) Complexes. Stereospecific Vicinal Diamination of Olefins, J. Am. Chem. Soc., 1977, 99, 3420–3426 CrossRef CAS.
- (a) D. E. Olson, J. Y. Su, D. A. Roberts and J. D. Bois, Vicinal Diamination of Alkenes under Rh-Catalysis, J. Am. Chem. Soc., 2014, 136, 13506–13509 CrossRef CAS PubMed; (b) K. Muñiz, L. Barreiro, R. M. Romero and C. Martínez, Catalytic Asymmetric Diamination of Styrenes, J. Am. Chem. Soc., 2017, 139, 4354–4357 CrossRef PubMed; (c) Y.-A. Yuan, D.-F. Lu, Y.-R. Chen and H. Xu, Iron-Catalyzed Direct Diazidation for a Broad Range of Olefins, Angew. Chem., Int. Ed., 2016, 55, 534–538 CrossRef CAS PubMed.
- (a) W. E. Fristad, T. A. Brandvold, J. R. Peterson and S. R. Thompson, Conversion of Alkenes to 1,2-Diazides and 1,2-Diamines, J. Org. Chem., 1985, 50, 3647–3649 CrossRef CAS; (b) N. Fu, G. S. Sauer, A. Saha, A. Loo and S. Lin, Metal-Catalyzed Electrochemical Diazidation of Alkenes, Science, 2017, 357, 575–579 CrossRef CAS PubMed; (c) N. Fu, G. S. Sauer and S. Lin, A General, Electrocatalytic Approach to the Synthesis of Vicinal Diamines, Nat. Protoc., 2018, 13, 1725–1743 CrossRef CAS PubMed; (d) J. C. Siu, J. B. Parry and S. Lin, Aminoxyl-Catalyzed Electrochemical Diazidation of Alkenes Mediated by a Metastable Charge-Transfer Complex, J. Am. Chem. Soc., 2019, 141, 2825–2831 CrossRef CAS PubMed.
- (a) S. Caron, R. W. Dugger, S. G. Ruggeri, J. A. Ragan and D. H. Brown, Ripin. Large-Scale Oxidations in the Pharmaceutical Industry, Chem. Rev., 2006, 106, 2943–2989 CrossRef CAS PubMed; (b) H.-T. Zhu, L. Arosio, R. Villa, M. Nebuloni and H. Xu, Process Safety Assessment of the Iron-Catalyzed Direct Olefin Diazidation for the Expedient Synthesis of Vicinal Primary Diamines, Org. Process Res. Dev., 2017, 21, 2068–2072 CrossRef CAS PubMed.
- (a) Y. Zhu, R. G. Cornwall, H. Du, B. Zhao and Y. Shi, Catalytic Diamination of Olefins via N–N Bond Activation, Acc. Chem. Res., 2014, 47, 3665–3678 CrossRef CAS PubMed; (b) B. Zhao, W. Yuan, H. Du and Y. Shi, Cu(I)-Catalyzed Intermolecular Diamination of Activated Terminal Olefins, Org. Lett., 2007, 9, 4943–4945 CrossRef CAS PubMed.
- (a) Z. Zhang, X.-J. Meng, F.-H. Cui, H.-T. Tang, Y.-C. Wang, G.-B. Huang and Y.-M. Pan, Electrochemically Promoted Three-Component Reaction to N-Sulfonyl Amidines, Org. Lett., 2024, 26, 193–197 CrossRef CAS PubMed; (b) Z.-L. Lei, T.-C. Liu, F.-H. Cui, Y.-M. Pan, S.-H. Li and H.-T. Tang, Electrochemical Promoted Three-Component Trifluoromethylation/Spirocyclization Reaction of N-Arylsulfonylacrylamides to 4-Azaspiro[4.5]Decanes, Org. Lett., 2024, 25, 6001–6005 CrossRef PubMed; (c) C. Yin, S. Tang, J. Mei, X. Hu and H. Zhang, Electrochemical Synthesis and Transformation of Organoboron Compounds, Org. Chem. Front., 2023, 10, 3361–3377 RSC; (d) Q. Wan, Z. Zhang, Z.-W. Hou and L. Wang, Recent Advances in the Electrochemical Generation of 1,3-Dicarbonyl Radicals from C–H Bonds, Org. Chem. Front., 2023, 10, 2830–2848 RSC; (e) H.-T. Tang, Y.-Z. Pan and Y.-M. Pan, Research Progress in Electrochemical/Photochemical Utilization of Methanol as a C1 Source, Green Chem., 2023, 25, 8313–8327 RSC; (f) X.-Q. Zhou, H.-T. Tang, F.-H. Cui, Y. Liang, S.-H. Li and Y.-M. Pan, Electrocatalytic Three-Component Reactions: Synthesis of Tellurium-Containing Oxazolidinone for Anticancer Agents, Green Chem., 2023, 25, 5024–5029 RSC; (g) T.-K. Xiong, Q. Xia, X.-Q. Zhou, S.-H. Li, F.-H. Cui, H.-T. Tang, Y.-M. Pan and Y. Liang, Electrochemically Mediated fixation of CO2: Synthesis of Functionalized Oxazolidine-2,4-Diones by Three-Component Reactions, Adv. Synth. Catal., 2023, 365, 2183–2187 CrossRef CAS; (h) X.-Y. Wang, Y.-F. Zhong, Z.-Y. Mo, S.-H. Wu, Y.-L. Xu, H.-T. Tang and Y.-M. Pan, Synthesis of Seleno Oxindoles via Electrochemical Cyclization of N-arylacrylamides with Diorganyl Diselenides, Adv. Synth. Catal., 2021, 363, 208–214 CrossRef CAS.
- C.-Y. Cai, X.-M. Shu and H.-C. Xu, Practical and Stereoselective Electrocatalytic 1,2-Diamination of Alkenes, Nat. Commun., 2019, 10, 4953 CrossRef CAS PubMed.
- (a) L. T. Vassilev, B. T. Vu, B. Graves, D. Carvajal, F. Podlaski, Z. Filipovic, N. Kong, U. Kammlott, C. Lukacs, C. Klein, N. Fotouhi and E. A. Liu, In Vivo Activation of the p53 Pathway by Small-Molecule Antagonists of MDM2, Science, 2004, 303, 844–848 CrossRef CAS PubMed; (b) K. Murai, S. Fukushima, A. Nakamura, M. Shimura and H. Fujioka, C3-Symmetric Chiral Trisimidazoline: The Role of a Third Imidazoline and its Application to the Nitro Michael Reaction and the α-Amination of β-Ketoesters, Tetrahedron, 2011, 67, 4862–4868 CrossRef CAS; (c) C. Dardonville and I. Rozas, Imidazoline Binding Sites and their Ligands: An Overview of the Different Chemical Structures, Med. Res. Rev., 2004, 24, 639–661 CrossRef CAS PubMed.
- (a) X.-Y. Su and P.-Q. Huang, Tf2O-Promoted Morgan–Walls Reaction: From a Flexible Approach to Functionalized Phenanthridines and Quinazolines to the Short and Divergent Total Syntheses of Alkaloids, Synthesis, 2023, 55, 877–891 CrossRef CAS; (b) B. H. Hoff, Acetonitrile as a Building Block and Reactant, Synthesis, 2018, 50, 2824–2852 CrossRef CAS.
- (a) P. Xiong, H. Long, J. Song, Y. Wang, J.-F. Li and H.-C. Xu, Electrochemically Enabled Carbohydroxylation of Alkenes with H2O and Organotrifluoroborates, J. Am. Chem. Soc., 2018, 140, 16387–16391 CrossRef CAS PubMed; (b) Y. Yu, Y. Yuan and K.-Y. Ye, Electrochemical Synthesis of Vicinal Azidoacetamides, Chem. Commun., 2023, 59, 422–425 RSC.
- (a) J. J. Ritter and P. P. Minieri, A New Reaction of Nitriles. I. Amides from Alkenes and Mononitriles, J. Am. Chem. Soc., 1948, 70, 4045–4048 CrossRef CAS PubMed; (b) J. J. Ritter and J. Kalish, A New Reaction of Nitriles. II.Synthesis of t-Carbinamines, J. Am. Chem. Soc., 1948, 70, 4048–4050 CrossRef CAS PubMed; (c) A. Guérinot, S. Reymond and J. Cossy, Ritter Reaction: Recent Catalytic Developments, Eur. J. Org. Chem., 2012, 19–28 CrossRef; (d) D. Jiang, T. He, L. Ma and Z. Wang, Recent Developments in Ritter Reaction, RSC Adv., 2014, 4, 64936–64946 RSC; (e) M.-E. Chen, X.-W. Chen, Y.-H. Hu, R. Ye, J.-W. Lv, B. Li and F.-M. Zhang, Recent Advances of Ritter Reaction and its Synthetic Applications, Org. Chem. Front., 2021, 8, 4623–4664 RSC; (f) L. Bao, B.-B. Zhang, Z.-X. Wang and X.-Y. Chen, Photocatalytic Dehydrations for the Ritter Reaction, Org. Chem. Front., 2023, 10, 1375–1379 RSC; (g) G. M. Ziarani, F. S. Hasankiadeh and F. Mohajer, Recent Applications of Ritter Reactions in Organic Syntheses, ChemistrySelect, 2020, 5, 14349–14379 CrossRef; (h) J. Y. Becker, L. R. Byrd, L. L. Miller and Y.-H. So, Remote Anodic Substitution of Ketones, J. Am. Chem. Soc., 1975, 97, 853–856 CrossRef CAS; (i) Y. Xu, Q. Li, R. Ye, B. Xu and X. Zhou, Electrochemical Oxidative C–H Amination through a Ritter-Type Reaction, J. Org. Chem., 2023, 88, 9518–9522 CrossRef CAS PubMed; (j) T. Shen and T. H. Lambert, C-H Amination via Electrophotocatalytic Ritter-Type Reaction, J. Am. Chem. Soc., 2021, 143, 8597–8602 CrossRef CAS PubMed; (k) R. Bishop, Comprehensive Organic Synthesis, ed. P. Knochel and G. A. Molander, 2014, pp. 239–295 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. CCDC 2368464. For ESI and crystallographic data in CIF or other electronic format see DOI: https://doi.org/10.1039/d4qo01445f |
| This journal is © the Partner Organisations 2024 |