Synthesis of fluorescent 5-heteroarylpyrimidine-containing oligonucleotides via post-synthetic trifluoromethyl conversion†
Abstract
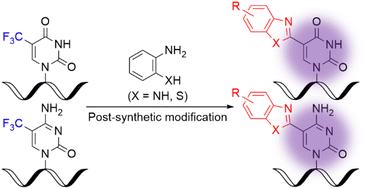
Post-synthetic conversion of the trifluoromethyl group to a heteroaryl group at the C5 position of the pyrimidine base in DNA oligonucleotides was achieved. Specifically, the oligonucleotides containing 5-trifluoromethylpyrimidine bases were treated with o-phenylenediamines and o-aminothiophenols as nucleophiles to afford the corresponding 5-(benzimidazol-2-yl)- and 5-(benzothiazol-2-yl)-pyrimidine-modified bases. Furthermore, evaluation of the fluorescence properties of the obtained oligonucleotides revealed that among them the oligonucleotide containing 5-(5-methylbenzimidazol-2-yl)cytosine exhibited the highest fluorescence intensity. These results indicated that post-synthetic trifluoromethyl conversion, which is practical and operationally simple, is a powerful tool for exploring functional oligonucleotides.


 Please wait while we load your content...
Please wait while we load your content...