Non-noble metal heterogeneous catalysts for hydrogen-driven deoxydehydration of vicinal diol compounds†
Jianxing
Gan
 a,
Yoshinao
Nakagawa
a,
Yoshinao
Nakagawa
 *ab,
Mizuho
Yabushita
*ab,
Mizuho
Yabushita
 ab and
Keiichi
Tomishige
ab and
Keiichi
Tomishige
 *abc
*abc
aDepartment of Applied Chemistry, School of Engineering, Tohoku University, 6-6-07 Aoba, Aramaki, Aoba-ku, Sendai, 980-8579, Japan. E-mail: yoshinao@erec.che.tohoku.ac.jp; tomishige@tohoku.ac.jp; Tel: +81-22-752-2223 Tel: +81-22-795-7214
bResearch Center for Rare Metal and Green Innovation, Tohoku University, 468-1 Aoba, Aramaki, Aoba-ku, Sendai, 980-0845, Japan
cAdvanced Institute for Materials Research (WPI-AIMR), Tohoku University, 2-1-1 Katahira, Aoba-ku, Sendai, 980-8577, Japan
First published on 10th June 2024
Abstract
Noble-metal-free heterogeneous catalysts for deoxydehydration (DODH) using H2 as a reductant were developed. Among various transition metals examined as additives to modify the MoOx/TiO2 catalyst, Cu showed good conversion and selectivity in the transformation of 1,4-anhydroerythritol (1,4-AHERY) to 2,5-dihydrofuran (2,5-DHF). The performance of the MoOx–Cu/TiO2 catalysts was comparable to those modified with either Ag or Au instead of Cu. Upon the combination of two Mo precursors, i.e. (NH4)6Mo7O24 and Na2MoO4, the selectivity of the catalyst (MoOx–Cu–Na/TiO2) was further enhanced to achieve 81% yield of 2,5-DHF. This catalyst also exhibited broad substrate scope including cyclic and linear alkyl vicinal diols and tartaric ester. Furthermore, MoOx–Cu–Na/TiO2 was reusable at least three times after its calcination as regeneration. The reaction was almost zero-order with respect to the H2 pressure and 1,4-AHERY concentration, suggesting that the release of the alkene is the rate-determining step. The comprehensive characterization using STEM, XRD and XAFS provided insights into the surface structure of the catalyst, revealing that H2 is activated over Cu particles and subsequently transferred to Mo cluster species on the TiO2 surface via the spillover effect to proceed with the DODH reaction.
Introduction
The depletion of fossil resources, coupled with rising mining costs, requires the establishment of an alternative means of supplying chemicals from sustainable feedstock such as biomass.1–3 Although a variety of processes for biomass conversion into chemicals have been established thus far,4–8 most processes are still under development. For example, olefinic compounds are essential platform chemicals because they can be derivatized into various products. Olefinic compounds have been manufactured on an industrial scale from petroleum via cracking reactions, while their production from biomass is difficult due to the high oxygen content in biomass-derived compounds. The development of catalytic systems capable of converting oxygen-rich biomass-derived compounds into alkenes and unsaturated oxygenates is, therefore, considered as the hot topic in the field of biorefinery.9–11The deoxydehydration (DODH) reaction converts vicinal OH groups to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond and is promising as a selective approach to converting biomass to olefinic compounds.12–14 The catalysts for DODH are mainly homogeneous Re complexes such as methyltrioxorhenium.15–17 DODH with such Re catalysts proceeds via the two-electron redox cycle of the high-valent metal center (ReVII and ReV). The catalytic DODH systems developed earlier used expensive and environmentally unfriendly reductants such as triphenylphosphine (PPh3),18,19 sodium sulfite (Na2SO3)20 and secondary alcohols.21–23 Molecular hydrogen (H2) is an inexpensive and green reductant in industry and generally used in the reductive conversion of biomass into chemicals and fuels such as hydrogenated vegetable oil.24–28 However, the use of H2 in homogeneous catalysts is difficult because H2 is hardly activated by homogeneous catalysts, making H2-driven DODH systems uncommon.29,30
C bond and is promising as a selective approach to converting biomass to olefinic compounds.12–14 The catalysts for DODH are mainly homogeneous Re complexes such as methyltrioxorhenium.15–17 DODH with such Re catalysts proceeds via the two-electron redox cycle of the high-valent metal center (ReVII and ReV). The catalytic DODH systems developed earlier used expensive and environmentally unfriendly reductants such as triphenylphosphine (PPh3),18,19 sodium sulfite (Na2SO3)20 and secondary alcohols.21–23 Molecular hydrogen (H2) is an inexpensive and green reductant in industry and generally used in the reductive conversion of biomass into chemicals and fuels such as hydrogenated vegetable oil.24–28 However, the use of H2 in homogeneous catalysts is difficult because H2 is hardly activated by homogeneous catalysts, making H2-driven DODH systems uncommon.29,30
Heterogeneous Re catalysts for DODH have been recently developed, and the combination of ReOx/CeO2 and a metal promoter is effective for H2-driven DODH reactions. In this system, a metal promoter activates H2 to help the reduction step of the Re center in the catalytic DODH cycle. Some catalysts give saturated di-deoxygenated products instead of alkenes because such a metal promotor can also catalyze the hydrogenation of alkene products (DODH + hydrogenation; DODH + HG). Our group developed ReOx–Pd/CeO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 31,32 and ReOx–Au/CeO2
31,32 and ReOx–Au/CeO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 33,34 catalysts as the first examples for DODH + HG and DODH, respectively. However, the high cost of Re, Pd and Au limits their industrial applicability. Very recently, our group has also developed the ReOx–Ag/CeO2 catalyst35 and the ReOx/CeO2 + Ni/CeO2 catalyst,36 both of which do not require expensive additive metals of Au or Pd. However, these state-of-the-art catalysts are still expensive because of the requirement of Re as active species.
33,34 catalysts as the first examples for DODH + HG and DODH, respectively. However, the high cost of Re, Pd and Au limits their industrial applicability. Very recently, our group has also developed the ReOx–Ag/CeO2 catalyst35 and the ReOx/CeO2 + Ni/CeO2 catalyst,36 both of which do not require expensive additive metals of Au or Pd. However, these state-of-the-art catalysts are still expensive because of the requirement of Re as active species.
Owing to their potential to replace the costly Re species in the conventional DODH catalysts, Mo-based catalysts have been extensively investigated for DODH reactions, e.g., dioxo-Mo(VI) complexes, molybdate salts and supported Mo catalysts.16 The first developed Mo-based catalysts are dioxo-Mo(VI) complexes (the first paper was published in 2013![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 37). The Mo centers in the complex catalysts are typically coordinated with a bulky ligand to stabilize the Mo complex.38–44 Non-green reductants such as PPh3 and metallic Al were used in these studies. The reported yields by the Mo complex catalysts were not high in comparison with the systems using homogeneous Re catalysts with the same reductants. Molybdate salts like ammonium heptamolybdate (AHM), a typical commercially-available Mo compound, are also effective in DODH reactions. In 2014, only slightly later than the first report for the Mo complex catalysts for DODH, the use of AHM as a DODH catalyst was reported.45 This report used diol as both a substrate and reductant. Yet, the product yield was not high (45% yield of 1-hexene from 1,2-hexanediol). Later studies also used AHM as the DODH catalyst in combination with various reductants such as 2-propanol46 and Na2SO3,47 and good yields from simple aliphatic diols were achieved with relatively inexpensive 2-propanol as the reductant (77% yield of 1-hexene). To enhance the reusability and stability, supported Mo catalysts were then investigated. In 2017, Palkovits et al. reported the first supported Mo-based DODH catalyst, MoOx/TiO2,48,49 which achieved 55% yield of 2,5-dihydrofuran (2,5-DHF) from 1,4-anhydroerythritol (1,4-AHERY) using 3-octanol as a reducing agent. This catalyst was reusable at least 5 times. Meanwhile, the use of H2 as a reductant with Mo-based DODH catalysts was not reported until our recent paper. In 2022, our group found the combination of MoOx–Au/TiO2 and H2 to achieve 77% yield of 2,5-DHF from 1,4-AHERY.50 The additive metal of Au played a crucial role in the activation of H2, in a similar manner to the ReOx–Au/CeO2 catalyst. Almost at the same time, Pagán-Torres et al. reported the MoOx–Pd/TiO2 catalyst for the DODH + HG reaction using H2 as a reductant.51 Although high product yields were not pursued in this report, very high selectivity (>98%) to tetrahydrofuran (THF) from 1,4-AHERY was reported at the 1,4-AHERY conversion of 29%.
37). The Mo centers in the complex catalysts are typically coordinated with a bulky ligand to stabilize the Mo complex.38–44 Non-green reductants such as PPh3 and metallic Al were used in these studies. The reported yields by the Mo complex catalysts were not high in comparison with the systems using homogeneous Re catalysts with the same reductants. Molybdate salts like ammonium heptamolybdate (AHM), a typical commercially-available Mo compound, are also effective in DODH reactions. In 2014, only slightly later than the first report for the Mo complex catalysts for DODH, the use of AHM as a DODH catalyst was reported.45 This report used diol as both a substrate and reductant. Yet, the product yield was not high (45% yield of 1-hexene from 1,2-hexanediol). Later studies also used AHM as the DODH catalyst in combination with various reductants such as 2-propanol46 and Na2SO3,47 and good yields from simple aliphatic diols were achieved with relatively inexpensive 2-propanol as the reductant (77% yield of 1-hexene). To enhance the reusability and stability, supported Mo catalysts were then investigated. In 2017, Palkovits et al. reported the first supported Mo-based DODH catalyst, MoOx/TiO2,48,49 which achieved 55% yield of 2,5-dihydrofuran (2,5-DHF) from 1,4-anhydroerythritol (1,4-AHERY) using 3-octanol as a reducing agent. This catalyst was reusable at least 5 times. Meanwhile, the use of H2 as a reductant with Mo-based DODH catalysts was not reported until our recent paper. In 2022, our group found the combination of MoOx–Au/TiO2 and H2 to achieve 77% yield of 2,5-DHF from 1,4-AHERY.50 The additive metal of Au played a crucial role in the activation of H2, in a similar manner to the ReOx–Au/CeO2 catalyst. Almost at the same time, Pagán-Torres et al. reported the MoOx–Pd/TiO2 catalyst for the DODH + HG reaction using H2 as a reductant.51 Although high product yields were not pursued in this report, very high selectivity (>98%) to tetrahydrofuran (THF) from 1,4-AHERY was reported at the 1,4-AHERY conversion of 29%.
Although Mo-based DODH catalysts typically necessitate higher reaction temperatures than Re-based catalysts,52,53 their abundance makes them more industrially attractive. Nonetheless, the widespread application of MoOx–M/support (M = Au or Pd) catalysts still remains a challenge due to the high cost of noble additive metals. The role of Au and Pd is to activate H2. In contrast, some non-noble metals, particularly first-row transition metals such as Fe, Ni, Co and Cu, are more accessible and commonly employed for hydrogenation of various organic compounds.54–57 Ni and Cu are also generally used in methanol synthesis via CO hydrogenation.58,59 In DODH reactions, Ni has been demonstrated to be a good co-catalyst for the activation of H2 in the ReOx/CeO2 + Ni/CeO2 system mentioned above.36 Such effectiveness posits the potential of non-noble first-row transition metals as alternatives to Au to reduce catalyst costs. In this research, therefore, to decrease the cost of catalysts, the development of DODH catalysts consisting of only non-noble metals was targeted.
Experimental
Catalyst preparation
MoOx–M/TiO2 catalysts (M = Fe, Co, Ni, Cu, Ag or Pd) were prepared by the sequential impregnation (imp) method. First, 2 g of TiO2 (anatase; Catalysis Society of Japan, JRC-TIO-7; BET surface area 270 m2 g−1) was added to a beaker placed on a heating plate. Then, an aqueous solution of each precursor (nitrate of M, whose amount corresponded to 2 wt% M (Fe, Co, Ni and Cu), 1 wt% Ag or 0.3 wt% Pd) was added to TiO2 until the surface of TiO2 was saturated (i.e., incipient wetness method). The mixture was stirred well with a glass bar at 363 K until being completely dried. The process was repeated until all the precursor solution was consumed. The solid was dried at 383 K overnight in a drying oven and calcined at 773 K for 3 h with a heating rate of 10 K min−1 to give M/TiO2. Next, MoOx was loaded on M/TiO2. An aqueous solution of 0.0184 g Mo precursor ((NH4)6Mo7O24·4H2O (AHM); 10 mg Mo) was added to 1 g of M/TiO2 with the same procedure as the impregnation of M. The catalysts (MoOx–M/TiO2) were used in the activity test after being dried at 383 K overnight in a drying oven without calcination. For the MoOx–Cu–Na/TiO2 catalysts, a mixed solution of AHM and Na2MoO4·2H2O (typical amounts: 0.0166 g AHM and 0.0025 g Na2MoO4·2H2O; Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Na molar ratio = 100
Na molar ratio = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15) was used for the loading of Mo species onto M/TiO2. The loading amount of Mo was 1 wt% for both MoOx–M/TiO2 and MoOx–Cu–Na/TiO2 catalysts unless otherwise noted. The typical loading amount of Cu was 2 wt%. Catalysts with other loading amounts were also prepared by changing the precursor amount(s).
15) was used for the loading of Mo species onto M/TiO2. The loading amount of Mo was 1 wt% for both MoOx–M/TiO2 and MoOx–Cu–Na/TiO2 catalysts unless otherwise noted. The typical loading amount of Cu was 2 wt%. Catalysts with other loading amounts were also prepared by changing the precursor amount(s).
For preparing Au/TiO2 and MoOx–Au/TiO2, Au species was loaded with the deposition–precipitation (dp) method.50 The TiO2 powder was dispersed in a HAuCl4·4H2O aqueous solution (Au content corresponded to 0.3 wt% of TiO2) at 353 K. The pH of the solution was adjusted to 8 by the gradual addition of 0.1 M NH3 aq. with constant stirring for 4 h. The suspension was filtered, and the recovered solid was washed several times with distilled water (2 L) at room temperature. After drying overnight at 383 K, Au/TiO2 was calcined in air at 673 K for 4 h, for which the heating rate was controlled to be 1 K min−1. The loading of Mo onto Au/TiO2 was carried out in the same way as that for the other MoOx–M/TiO2 catalysts (vide supra).
The control catalysts, ReOx–dpAu/CeO2 (Re 1 wt%, Au/Re = 0.3), ReOx–Ag/CeO2 (Re 1 wt%, Ag/Re = 0.3) and ReOx–Pd/CeO2 (Re 2 wt%, Pd/Re = 0.25), were prepared by following the same procedure in our previous reports.32,33,35
Activity test
A 190 mL stainless–steel autoclave with a glass inner vessel was used as the reactor for activity tests. For the typical reaction, 0.15 g catalyst, 0.5 g substrate, 0.1 g internal standard (n-dodecane), 4.0 g solvent (1,2-dimethoxyethane (1,2-DME)) and a stirrer bar were added into the glass vessel. The glass vessel was set into the autoclave, and then, 1 MPa H2 was used to purge the autoclave 3 times, followed by the pressurization with 5.1 MPa (8 MPa at 463 K) H2. A heater was used to control the reaction temperature, and the inner temperature was monitored via a thermocouple. The time when the temperature reached the target one was defined as 0 h. After the reaction, the autoclave was rapidly cooled down to room temperature in a water bath. The gas phase was collected in a gas bag for the runs where gaseous products were generated. Afterwards, the inside of the autoclave was washed with 15 g of the washing solvent (the same liquid as the reaction solvent was employed). The resulting liquid was collected, combined and analyzed. The liquid and gaseous samples were analyzed by GC-FID with a TC-WAX column. For product identification, GC-MS was used in conjunction with commercially available authentic samples. Any undefined peaks in the GC charts were labeled as “others” in this study. In most conditions, the reaction results consistently showed a carbon balance of 100 ± 10%.The conversion, selectivity, carbon balance and yield were calculated by using the following equations (eqn (1)–(4)), where n and Nc represent the mole of each compound and the number of carbon atom in each compound, respectively.
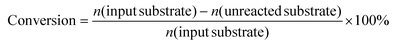 | (1) |
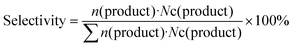 | (2) |
 | (3) |
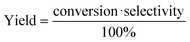 | (4) |
For the reuse experiments, the liquid phase and catalyst were separated via centrifugation at 6000 rpm for 10 min after the reaction. Afterwards, the catalyst was washed three times with 1,2-DME. The catalyst was then dried in a drying oven at the temperature of 383 K for 3 h. Optionally, the dried catalyst was further calcined at 573 K for 3 h with a heating rate of 10 K min−1. After drying (+calcination), the catalyst was used for the next round of activity test.
Catalyst characterization
Thermogravimetry-differential thermal analysis (TG-DTA) was carried out with a Rigaku Thermo Plus EVO-II instrument. About 10 mg of sample was set on a Pt pan and heated in an air flow (30 mL min−1) with a heating rate of 10 K min−1 from room temperature to 1173 K.X-ray diffraction (XRD) patterns were acquired using a Rigaku MiniFlex600 diffractometer. Cu Kα radiation (λ = 0.154 nm) was employed as the X-ray source. Samples were measured at a scanning rate of 10° min−1 along the 2θ axis from 3° to 90°.
Analysis of scanning-transmission electron microscopy (STEM) was conducted with an FEI Titan3 60–300 electron microscope equipped with a Super-X detector (Thermo Fisher Scientific) for energy-dispersive X-ray spectroscopy (EDX). Samples were dispersed in ethanol and ultrasonicated. After transferring the suspension onto a Cu or Pt grid, the grid was air-dried at ambient temperature until the complete evaporation of ethanol.
Temperature-programmed reduction with H2 (H2-TPR) was carried out with a fixed-bed flow-type reactor equipped with a thermal conductivity detector (TCD). The sample amount was 50 mg. Argon was used to dilute H2 gas (5%), and the total flow rate of gas was 30 mL min−1. The reactor was heated from room temperature to 1273 K with a heating rate of 10 K min−1.
X-ray absorption spectroscopy (XAS) was conducted at the BL14B2 beamline of SPring-8 (proposal number: 2022B1746). The catalyst samples after the reaction were collected, dried and sealed in a plastic bag in a glove bag filled in N2 to avoid the undesired sample exposure to air. Cu K-edge XAS data were acquired in a transmission mode. Mo K-edge XAS data were obtained in either a fluorescence or transmission mode. All the measurements were conducted at room temperature. The Demeter software package60 was used to manage the XAS data. Athena software was used to subtract a linear function from the pre-edge region and normalize the data on the basis of the edge jump. The k3-weighted χ(k) data were Fourier-transformed after the Hanning window function was applied. The Artemis software was used to fit the EXAFS oscillations to the Fourier-transformed data in R-space.
Results and discussion
Catalyst development
A series of additive metals (M) in MoOx–M/TiO2 catalysts were tested in the DODH reaction of 1,4-anhydroerythritol (1,4-AHERY) as a model substrate, and the results are summarized in Table 1. Note that 1,4-AHERY is a typical biomass-derived platform molecule that is used for DODH reactions due to its high reactivity.61 The primary DODH product was 2,5-dihydrofuran (2,5-DHF), and typical byproducts were THF (hydrogenated product of 2,5-DHF), 2,3-dihydrofuran (2,3-DHF; isomerized product of 2,5-DHF) and 1,4-butanediol (1,4-BuD; overreaction product of THF and dihydrofurans).62 The formation of 1,4-BuD from 2,5-DHF resulted from the consecutive reaction consisting of isomerization to 2,3-DHF and hydration to 2-hydroxytetrahydrofuran and hydrogenation (Fig. S1†), as reported previously;62 in this subsequent reaction, the isomerization step is the key, and the later steps proceed relatively easily. Carbon-supported ReOx catalysts were reported to be effective for the isomerization of 2,5-DHF and even complete conversion to 1,4-BuD.63| Entry | M | Conv. [%] | Selectivity [%] | Carbon balance [%] | ||||
|---|---|---|---|---|---|---|---|---|
| 2,5-DHF | THF | 2,3-DHF | 1,4-BuD | Others | ||||
| DHF = dihydrofuran; BuD = butanediol.a Reaction conditions: 1,4-AHERY (0.5 g), 1,2-DME (4 g), n-dodecane (internal standard) (0.1 g), catalyst (Mo (1 wt%), M (2 wt%)) (0.15 g), H2 (8 MPa) (initial pressure (5.1 MPa) at r.t.), 463 K, 4 h.b TiO2 (P25) support, Au (0.3 wt%), Mo (1 wt%).c TiO2 (P25) support, Pd (0.3 wt%), Mo (1 wt%).d 24 h reaction.e MoOx/TiO2, without M added. | ||||||||
| 1b | Au | 35.0 | 99.9 | 0.0 | 0.0 | 0.0 | 0.1 | 100.6 |
| 2c | Pd | 43.0 | 0.0 | 99.9 | 0.0 | 0.0 | 0.1 | 96.5 |
| 3 | Ag | 56.7 | 97.4 | 0.1 | 0.3 | 0.4 | 1.8 | 97.0 |
| 4 | Cu | 62.4 | 97.8 | 0.1 | 0.5 | 0.6 | 0.9 | 98.3 |
| 5 | Fe | 8.0 | 67.4 | 1.1 | 0.5 | 4.1 | 27.0 | 98.2 |
| 6 | Co | 4.1 | 84.4 | 2.0 | 0.6 | 5.0 | 8.1 | 92.2 |
| 7 | Ni | 9.3 | 75.1 | 2.1 | 1.9 | 8.0 | 12.9 | 100.6 |
| 8d | Cu | 86.6 | 69.4 | 15.0 | 1.0 | 3.2 | 11.4 | 96.5 |
| 9e | None | 4.1 | 81.3 | 4.7 | 1.9 | 1.6 | 10.5 | 103.5 |
Among the various additive metals (M) in the MoOx–M/TiO2 catalysts tested at 463 K for 4 h, Ag and Cu showed comparably high DODH activity (entries 3 and 4). The yield of DODH (or DODH + HG) product by these catalysts was higher than those by the MoOx–dpAu/TiO2 (P25) and MoOx–impPd/TiO2 (P25) (Au or Pd = 0.3 wt%, Mo = 1 wt%) catalysts, both of which were reported in our previous work (entries 1 and 2),50 under the current reaction conditions. The higher yields provided here are attributable to the difference of TiO2 supports (P25 for the current study vs. anatase for previous studies) in addition to the class of M. Given that Cu is more abundant and cheaper than Ag, we focused more on the Cu-based catalysts in the following investigation.
The extension of the reaction time with the MoOx–Cu/TiO2 catalyst enabled us to achieve high 1,4-AHERY conversion, while the yield of 2,5-DHF did not increase over 65% because of the formation of THF, 1,4-BuD and other unidentified compounds (“others”) as by-products (entry 8 of Table 1 and Table S1†). The decrease of reaction temperature to 413 K suppressed the formation of “others”, but the formation of 1,4-BuD instead became prominent at the high 1,4-AHERY conversion (Table S1†). The high yield of 2,5-DHF was thus not obtained over the MoOx–Cu/TiO2 catalysts.
DODH reaction itself does not require acidic or basic catalysts, while acidic species rather induces undesired side reactions. Based on this insight, a part of Mo precursor ((NH4)6Mo7O24; AHM) was replaced with a basic one (Na2MoO4) in the catalyst preparation in order to decrease the acidity of catalysts (the prepared catalysts are hereafter represented by MoOx–Cu–Na/TiO2). The selectivity to 2,5-DHF was successfully increased upon the partial replacement of acidic precursor (AHM) with the basic one (Na2MoO4) (Table S2†). However, too much Na rather decreased both the selectivity to 2,5-DHF and the activity of MoOx–Cu–Na/TiO2 catalysts. When only Na2MoO4 was used as the Mo precursor (Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Na = 100
Na = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200), almost no 2,5-DHF was obtained in the reaction for 16 h at 463 K. The activity of MoOx–Cu–Na/TiO2 catalysts was not changed much at the Na/Mo molar ratio of less than 1, while that was slightly decreased by the further increase of Na amount (Tables S2 and S3†). The highest selectivity to 2,5-DHF at 463 K was obtained at the Mo
200), almost no 2,5-DHF was obtained in the reaction for 16 h at 463 K. The activity of MoOx–Cu–Na/TiO2 catalysts was not changed much at the Na/Mo molar ratio of less than 1, while that was slightly decreased by the further increase of Na amount (Tables S2 and S3†). The highest selectivity to 2,5-DHF at 463 K was obtained at the Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Na ratio of 100
Na ratio of 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15, which was selected in the following study.
15, which was selected in the following study.
The time course of the DODH reaction of 1,4-AHERY at 463 K over the MoOx–Cu–Na/TiO2 (2 wt% Cu, 1 wt% Mo, Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Na = 100
Na = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15) is shown in Fig. 1 and Table S4.† The high 1,4-AHERY conversion was obtained even at 0 h (just after the inner temperature reached 463 K), indicating the high activity of the MoOx–Cu–Na/TiO2 catalyst. The selectivity to 2,5-DHF was almost 100% at the short reaction time and then slowly decreased until 24 h when the conversion of 1,4-AHERY reached about 90%. During the initial 24 h, the main by-product was THF, which was probably formed via the hydrogenation of 2,5-DHF. After 24 h, the formation of 1,4-BuD started, and the formation of THF was also accelerated. The highest yield of 2,5-DHF (81%) was observed at 24 h. This yield was higher than that obtained over MoOx–dpAu/TiO2 (1 wt% Mo, 0.3 wt% Au, 77% yield) and those with homogeneous Mo catalysts (maximum 75% yield with the AHM catalyst and 2-propanol reductant)50 but lower than that over ReOx–dpAu/CeO2 (1 wt% Re, 0.3 wt% Au, 91% yield)34 (Table S1†). The turnover number (TON) based on Mo atoms in the MoOx–Cu–Na/TiO2 catalyst reached 270, the value of which was also higher than those obtained with reported Mo-based DODH catalysts.
15) is shown in Fig. 1 and Table S4.† The high 1,4-AHERY conversion was obtained even at 0 h (just after the inner temperature reached 463 K), indicating the high activity of the MoOx–Cu–Na/TiO2 catalyst. The selectivity to 2,5-DHF was almost 100% at the short reaction time and then slowly decreased until 24 h when the conversion of 1,4-AHERY reached about 90%. During the initial 24 h, the main by-product was THF, which was probably formed via the hydrogenation of 2,5-DHF. After 24 h, the formation of 1,4-BuD started, and the formation of THF was also accelerated. The highest yield of 2,5-DHF (81%) was observed at 24 h. This yield was higher than that obtained over MoOx–dpAu/TiO2 (1 wt% Mo, 0.3 wt% Au, 77% yield) and those with homogeneous Mo catalysts (maximum 75% yield with the AHM catalyst and 2-propanol reductant)50 but lower than that over ReOx–dpAu/CeO2 (1 wt% Re, 0.3 wt% Au, 91% yield)34 (Table S1†). The turnover number (TON) based on Mo atoms in the MoOx–Cu–Na/TiO2 catalyst reached 270, the value of which was also higher than those obtained with reported Mo-based DODH catalysts.
 | ||
Fig. 1 Time course of DODH of 1,4-AHERY over the MoOx–Cu–Na/TiO2 catalyst. Reaction conditions: 1,4-AHERY (0.5 g), 1,2-DME (4 g), n-dodecane (internal standard) (0.1 g), MoOx–Cu–Na/TiO2 (2 wt% Cu, 1 wt% Mo, Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Na = 100 Na = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15) (0.15 g), H2 (8 MPa) (initial pressure 5.1 MPa at r.t.), 463 K. The details are listed in Table S4.† 15) (0.15 g), H2 (8 MPa) (initial pressure 5.1 MPa at r.t.), 463 K. The details are listed in Table S4.† | ||
Next, the effect of the loading amounts of Mo and Cu in MoOx–Cu–Na/TiO2 was investigated (Fig. 2, 3 and S2, Tables S6 and S7†). In the dependence of activity on the Mo loading amount (Fig. 2), the volcano-type plot with steep slopes on both sides was observed. The highest activity was provided at 1 wt% of Mo loading, while the selectivity to 2,5-DHF (DODH product) was high in the Mo-loading range from 0.1 wt% to 2 wt%. Thus, 1 wt% Mo was selected as the optimum Mo loading amount for the MoOx–Cu–Na/TiO2 catalysts. For the previously developed catalyst MoOx–dpAu/TiO2,50 the excess loading of Mo did not decrease the activity. The difference between MoOx–Cu–Na/TiO2 and MoOx–dpAu/TiO2 can be explained by the low reducibility of Cu species in MoOx–Cu–Na/TiO2 with high Mo loading. At a higher Mo loading amount, the reduction temperature of Cu got significantly increased (Fig. S8†). The details will be discussed in the characterization section (vide infra).
 | ||
Fig. 2 Dependence of the DODH performance of MoOx–Cu–Na/TiO2 catalysts on the Mo loading amount. Reaction conditions: 1,4-AHERY (0.5 g); 1.2-DME (4 g), n-dodecane (internal standard) (0.1 g), MoOx–Cu/TiO2 (Cu 2 wt%, Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Na = 100 Na = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15) (0.15 g), H2 (8 MPa) (initial pressure (5.1 MPa) at r.t.), 463 K, 4 h. The details are listed in Table S6.† 15) (0.15 g), H2 (8 MPa) (initial pressure (5.1 MPa) at r.t.), 463 K, 4 h. The details are listed in Table S6.† | ||
 | ||
Fig. 3 Dependence of DODH performance of MoOx–Cu–Na/TiO2 catalysts on Cu loading amount. Reaction conditions: 1,4-AHERY 0.5 g; 1.2-DME 4 g, n-dodecane (internal standard) 0.1 g, MoOx–Cu/TiO2 (Mo 1 wt%, Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Na = 100 Na = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15) 0.15 g, H2 8 MPa (initial pressure 5.1 MPa at r.t.), 463 K, 4 h. The details are listed in Table S7.† 15) 0.15 g, H2 8 MPa (initial pressure 5.1 MPa at r.t.), 463 K, 4 h. The details are listed in Table S7.† | ||
For the effect of Cu amount (Fig. 3), with the increase of Cu loading amount, the conversion and selectivity to 2,5-DHF were increased until 2 wt%. Over 2 wt%, the conversion and selectivity to 2,5-DHF got decreased due to the formation of by-products, mainly 1,4-BuD. For the Au-promoted catalyst MoOx–dpAu/TiO2,50 the activity was reported to be almost independent on the Au loading amount in the range of 0.1–0.6 wt%, and the selectivity to 2,5-DHF was also almost unchanged. The little dependence of the catalytic performance on the amount of promoter was also reported for ReOx/CeO2 catalysts (promoter: Pd, Ag and Ni).32,35,36 The necessity of a larger amount of Cu compared to the other promoters can be explained by the larger particle size of Cu, which results in the lower number of Cu particles; the details of structural features will be discussed later. The reason for the formation of 1,4-BuD over the MoOx–Cu–Na/TiO2 catalysts with high Cu loadings (>5 wt%) was assumed to be the activity of Cu species for the conversion of 2,5-DHF. In fact, the formation of 1,4-BuD from 2,5-DHF was confirmed in the control reactions using 2,5-DHF as a substrate in the presence of either Cu/TiO2 with a high Cu loading or CuO as a catalyst (Table S8†). After the reaction of 2,5-DHF operated under pressurized H2, CuO was totally reduced to Cu metal, which was visibly obvious by the color of the spent catalyst and also confirmed by XRD (Fig. S2†). Cu metal phase was also found in Cu/TiO2 (5 wt%) and Cu/TiO2 (10 wt%) by XRD after their catalytic use for the reduction of 2,5-DHF. With the increase in the Cu loading amount, both the particle size and number of Cu particles increased, triggering the Cu-catalyzed formation of 1,4-BuD. Thus, 2 wt% was chosen to be the appropriate loading amount of Cu in MoOx–Cu–Na/TiO2.
Applicability of the MoOx–Cu–Na/TiO2 catalyst
The applicability of the MoOx–Cu–Na/TiO2 catalyst to other diols was examined (Table 2). As the compounds consisting of linear carbon backbone with terminal vicinal diol, 1,2-butanediol was converted to 1-butene in 32% yield (at 43% conversion; entry 1), and 1,2-pentanediol was transformed into 1-pentene in 52% yield (at 58% conversion; entry 2). The substrate with a longer carbon chain gave better selectivity. The main by-product was n-alkane generated via the hydrogenation of the target olefin. The similar yield of 1-pentene from 1,2-pentanediol was reported over MoOx–dpAu/TiO2 under almost the same conditions,50 while the reaction time was much shorter with the MoOx–Cu–Na/TiO2 catalyst (4 h for MoOx–Cu–Na/TiO2vs. 24 h for MoOx–dpAu/TiO2). This observed superiority of MoOx–Cu–Na/TiO2 is reflected by the higher promotion ability of Cu (with enough amount) than Au. Meanwhile, the selectivity to alkane (DODH + HG product) over MoOx–Au/TiO2 was <1%, which was lower than 9% given by MoOx–Cu–Na/TiO2. The progress of the reaction by lowering the substrate amount and prolonging the reaction time over MoOx–Cu–Na/TiO2 gave the intensive formation of the alkane product (entry 3).| Entry | Substrate (amount [mmol]) | Reaction time [h] | Conv. [%] | Product (selectivity [%]) | Carbon balance [%] |
|---|---|---|---|---|---|
a Reaction conditions: substrate (0.5 g), 1,2-DME (4 g), n-dodecane (internal standard) (0.1 g), MoOx–Cu–Na/TiO2 catalyst (1.0 wt% Mo, 2.0 wt% Cu, Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Na = 100 Na = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15) (0.15 g), H2 (8 MPa) (initial pressure (5.1 MPa) at r.t.), 463 K.
b Substrate (0.25 g).
c Styrene glycol (0.25 g), catalyst (0.3 g), hydroquinone (0.05 g) as a polymerization inhibitor, n-dodecane (internal standard) (0.1 g), H2 (4 MPa) (initial pressure (2.9 MPa) at r.t.), 413 K. 15) (0.15 g), H2 (8 MPa) (initial pressure (5.1 MPa) at r.t.), 463 K.
b Substrate (0.25 g).
c Styrene glycol (0.25 g), catalyst (0.3 g), hydroquinone (0.05 g) as a polymerization inhibitor, n-dodecane (internal standard) (0.1 g), H2 (4 MPa) (initial pressure (2.9 MPa) at r.t.), 413 K.
|
|||||
| 1 | 1,2-Butanediol (5.55) | 4 | 43.2 | 1-Butene (74.0), n-butane (26.0), others (<0.1) | 99.0 |
|
|||||
| 2 | 1,2-Pentanediol (4.80) | 4 | 58.3 | 1-Pentene (90.4), n-pentane (9.1), others (0.5) | 99.1 |
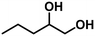
|
|||||
| 3b | 1,2-Pentanediol (2.40) | 8 | 84.8 | 1-Pentene (54.7), n-pentane (38.9), others (6.4) | 99.4 |
| 4b | cis-1,2-Cyclohexanediol (2.15) | 4 | 75.2 | Cyclohexene (96.0), cyclohexane (0.0), trans-isomer (1.5), others (2.5) | 94.5 |
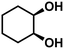
|
|||||
| 5b | cis-1,2-Cyclohexanediol (2.15) | 16 | 93.3 | Cyclohexene (87.8), cyclohexane (5.3), trans-isomer (0.8), others (6.0) | 100.7 |
| 6b | trans-1,2-Cyclohexanediol (2.15) | 4 | 9.1 | Cyclohexene (57.7), cyclohexane (0.0), cis-isomer (28.3), others (14.0) | 98.5 |
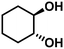
|
|||||
| 7c | Styrene glycol (1.81) | 1 | 89.3 | Styrene (42.7), ethylbenzene (53.6), phenethyl alcohol (3.6), others (<0.1) | 84.0 |
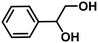
|
|||||
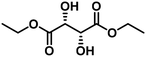
| 8 | Diethyl L-tartrate (2.42) | 4 | 62.6 | Diethyl fumarate (69.3), diethyl succinate (21.4), diethyl maleate (1.4), others (7.9) | 96.5 |
|
|||||
| 9b | Diethyl L-tartrate (1.21) | 8 | >99.9 | Diethyl fumarate (4.0), diethyl succinate (67.7), diethyl maleate (0), others (28.3) | 92.2 |
For cyclic diols, cis-1,2-cyclohexanediol was converted to cyclohexene in 82% yield (entry 5), which was higher than the value obtained over the MoOx–dpAu/TiO2 catalyst (66%).50 The higher yield of cyclohexene by MoOx–Cu–Na/TiO2 originates from the low reactivity of cyclohexene for hydrogenation and also low activity of MoOx–Cu–Na/TiO2 for dehydration. The MoOx–Au/TiO2 catalyst proceeded with the formation of cyclohexanol and cyclohexanone (7.6% selectivity to these compounds at 73% conversion of cis-1,2-cyclohexanediol), while the formation of these monooxygenates was negligible over MoOx–Cu–Na/TiO2. The removal of acid sites by the addition of Na in MoOx–Cu–Na/TiO2 could be responsible for suppressing the formation of monooxygenates. The reactivity of trans-1,2-cyclohexanediol was much lower than that of the cis-isomer (entry 6), whose behavior is quite common in DODH systems.32,34,50 The formation of the cis-isomer was found in the reaction of trans-1,2-cyclohexanediol. Cyclohexene was produced probably via the isomerization to cis-1,2-cyclohexanediol and subsequent DODH.
Styrene glycol showed significantly different reactivity from the other substrates (entry 7, Tables 2 and S8†). Under the standard reaction conditions (463 K, substrate/catalyst = 0.5 g/0.15 g), the styrene glycol was completely converted even during the heating to 463 K (i.e., reaction time = 0 h), and the main product was ethylbenzene (DODH + HG product). However, the carbon balance was only 54%. A possible reason for this poor carbon balance is the formation of polystyrene because styrene is known to readily undergo the polymerization at high temperature.64 The addition of a typical polymerization inhibitor, hydroquinone,65 slightly improved the carbon balance by ca. 12% (Table S9†). However, by TG-DTA (Fig. S3†), the weight loss of the solid after the reaction without hydroquinone was only 5.0 wt% (corresponding to 1.6% of the charged substrate), which was much less than the loss of carbon balance (46% loss of carbon balance as polystyrene (corresponding to 53 wt% weight loss in TG)). Soluble styrene oligomers were assumed to be generated; such species have a wide range of molecular weight due to different numbers of monomeric units involved, and thus, were unable to be detected by GC-FID. Therefore, the milder reaction conditions (lower reaction temperature and H2 pressure) were applied in order to suppress the formation of ethylbenzene and polystyrene. Under the conditions of 363 K and 4 MPa H2, the carbon balance was successfully kept at >90% with the conversion level of ca. 80% (16 h); however, the main product was still ethylbenzene. Also, when 0.5 wt% Cu catalyst was employed (Table S10†), the conversion was significantly low. Compared with the standard 2.0 wt% Cu catalyst, the difference in the conversion level was much larger than that in the reaction of 1,4-AHERY (Fig. 3, Table S7†). The significant decrease in the conversion with the 0.5 wt% Cu catalyst was probably due to the blockage of Cu sites by polystyrene; that is, even if the formation amount of polystyrene was limited, the small number of Cu sites underwent the coverage by polystyrene easily. To increase the yield of styrene over MoOx–Cu–Na/TiO2, the moderate reaction temperature (413 K) and short reaction time were used to suppress the hydrogenation reaction of the produced styrene, and hydroquinone was added as a polymerization inhibitor. As a result, styrene was obtained in 38% yield (entry 7, Table 2). This value is similar to the highest styrene yield in DODH of styrene glycol with H2 ever reported with the ReOx–C catalyst (39%).66
Other heterogeneous DODH or DODH + HG catalysts were also tested in the reaction of styrene glycol (Table S10†). MoOx–Au/TiO2, MoOx–Ag/TiO2, ReOx–Au/CeO2 and ReOx–Ag/CeO2 catalysts, all of which are effective for DODH (not DODH + HG), showed good selectivity to styrene; however, the conversion level (<15%) was much lower than that over MoOx–Cu–Na/TiO2. On the other hand, ReOx–Pd/CeO2, which is an effective DODH + HG catalyst, completely converted styrene glycol to ethylbenzene. Ideally, the activities of ReOx–M/CeO2 catalysts are similar because the rate-determining step of DODH reaction is the extrusion of alkene from the reduced Re diolate species regardless of M species. The high conversion over ReOx–Pd/CeO2 means that the intrinsic reactivity of styrene glycol is very high. The C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond in a styrene molecule is conjugated with the phenyl group, while such conjugation is absent in styrene glycol. The stabilization by conjugation may lead to the high reactivity of styrene glycol toward DODH. The lower conversions obtained over ReOx–Au/CeO2 and ReOx–Ag/CeO2 than that over ReOx–Pd/CeO2 could be due to the blockage of active sites on the former two catalysts by polystyrene and also the inhibition of polystyrene formation on the latter Pd-modified catalyst by rapid hydrogenation of the produced styrene. Similarly, for the Mo-based catalysts, the Au- and Ag-modified catalysts were significantly deactivated because of the formation of polystyrene.
C bond in a styrene molecule is conjugated with the phenyl group, while such conjugation is absent in styrene glycol. The stabilization by conjugation may lead to the high reactivity of styrene glycol toward DODH. The lower conversions obtained over ReOx–Au/CeO2 and ReOx–Ag/CeO2 than that over ReOx–Pd/CeO2 could be due to the blockage of active sites on the former two catalysts by polystyrene and also the inhibition of polystyrene formation on the latter Pd-modified catalyst by rapid hydrogenation of the produced styrene. Similarly, for the Mo-based catalysts, the Au- and Ag-modified catalysts were significantly deactivated because of the formation of polystyrene.
For the reaction of diethyl L-tartrate (entries 8 and 9, Table 2), the converted substrate amount was comparable to those of 1,2-alkanediols. The hydrogenation of the DODH product (diethyl fumarate) to diethyl succinate proceeded readily, and the obtained yield of diethyl fumarate was at most 43%. The increase of conversion by decreasing the substrate amount and prolonging the reaction time rather decreased the yield of diethyl fumarate because of its subsequent hydrogenation. The production of succinic acid from tartaric acid in good yield has been reported with MoOx–M/TiO2 catalysts (M: noble metal).67 Effective catalytic systems for the DODH of tartaric acid or tartrates to the olefinic products with H2 as a reductant have not yet been reported. Our previously reported Re/CeO2-based catalysts were also tested for the reaction of diethyl L-tartrate (Table S11†); however, the conversion was much lower than that of polyols (the considerable conversion required much higher temperature such as 463 K), and diethyl fumarate or diethyl succinate was hardly formed. The TG-DTA results of ReOx–dpAu/CeO2 and ReOx–Pd/CeO2 catalysts after the reaction of diethyl L-tartrate at 463 K are shown in Fig. S4.† The exothermic weight losses of 14.9% and 13.8% were observed at around 473 K for ReOx–dpAu/CeO2 and ReOx–Pd/CeO2, respectively, which were about 16 equivalents of the substrate molecule to Re and corresponded to about 8% conversion. These results suggest that the low carbon balance was due to the intensive adsorption of organic molecules onto the catalyst surfaces as well as formation of products that cannot be detected by GC. Considering that there are several Re-based catalysts that are effective for DODH of sugar acids and their esters,68–72 the CeO2 support seems to be inappropriate for the substrates containing carboxyl group(s) or carboxylate. MoOx/TiO2-based catalysts are more suitable to this type of substrate.
Additionally, the performance of the MoOx–Cu–Na/TiO2 catalyst in alcohol solvents (2-propanol and ethanol) was investigated with 1,4-AHERY as a substrate, because alcohol solvents have higher dissolution ability for highly functionalized biomass-related molecules. As shown in Table S12,† the catalyst exhibited similarly high activity in these alcohol solvents to the case of 1,2-dimethoxyethane (1,2-DME), yet the selectivity to 2,5-DHF became slightly lower. These results demonstrated the applicability of the MoOx–Cu–Na/TiO2 catalyst to not only ether solvents but also alcohol solvents, positing the great potential of this catalyst in the conversion of more functionalized biomass-derived molecules.
Reusability of the MoOx–Cu–Na/TiO2 catalyst
The reusability of MoOx–Cu–Na/TiO2 was evaluated in the DODH reaction of 1,4-AHERY. The activity and selectivity to 2,5-DHF (DODH product) from 1,4-AHERY showed no significant differences in four-times reuse, where the catalysts were dried and calcined at 573 K for 3 h before each run (Fig. 4(A)). However, the activity (conversion of 1,4-AHERY) was significantly decreased when the catalysts were only dried after each run (Fig. 4(B)). The good stability of regenerated (calcined before reuse run) catalyst and low stability without regeneration were also reported for the MoOx–dpAu/TiO2 catalyst;50 however, the MoOx–dpAu/TiO2 catalyst showed a slight decrease in the selectivity to 2,5-DHF and a gradual increase in the selectivity to 1,4-BuD. In this context, the MoOx–Cu–Na/TiO2 catalyst was concluded as a more stable catalyst for the DODH reaction of 1,4-AHERY, compared to MoOx–dpAu/TiO2.The TG-DTA profiles for the spent MoOx–Cu–Na/TiO2 catalysts and as-prepared one are shown in Fig. S5.† The as-prepared catalyst (A) only showed a small loss of weight (0.4%) at around 513 K, which is mainly attributed to the combustion of ammonium ions derived from the Mo precursor (AHM). The spent catalysts (B–E) showed a significant exothermic signal with a loss of weight at around 490 K, which is assignable to the combustion of surface-deposited organic species. The combusted amount in the catalyst after the 1st use was 4.0 wt%, which corresponds to about 4 equivalents of 1,4-AHERY to Mo. A larger amount of organic compounds than Mo diolate was thus adsorbed or deposited. The combusted amount was gradually increased to 5.5 wt% upon the repeated use. The increase of adsorbed or deposited carbonaceous species could be the reason for the gradual deactivation of the MoOx–Cu–Na/TiO2 catalyst. Thus, the good reusability of calcined catalysts results from the removal of such organic species. For MoOx–dpAu/TiO2 after the reaction (1,4-AHERY, 463 K, 1,2-DME solvent, a similar turnover number),50 a similar combustion signal was observed (peak temperature: 510 K) with a lower weight loss (2.9% weight loss around the exothermic peak), in addition to the coke combustion (1.6% weight loss around the exothermic peak at 736 K). The MoOx–Cu–Na/TiO2 catalyst produces almost the same amount of total organic deposited species as the case of MoOx–dpAu/TiO2, but the coke amount was lower. The lower amount of coke which cannot be removed by the regeneration (calcination at 573 K) could lead to the higher selectivity to 2,5-DHF over the regenerated MoOx–Cu–Na/TiO2 catalysts than regenerated MoOx–dpAu/TiO2 for four-times reuse (98% vs. 90%).50
Kinetic study on the DODH reaction of 1,4-AHERY over the MoOx–Cu–Na/TiO2 catalyst
The effects of H2 pressure and substrate amount on the DODH reaction of 1,4-AHERY over the MoOx–Cu–Na/TiO2 catalyst are shown in Fig. 5 and 6 (their detailed data are summarized in Tables S13 and S14,† respectively). The reaction order with respect to the H2 pressure was 0.13 in the range from 2 MPa to 8 MPa (Fig. 5). This small reaction order suggests that the steps in which H2 participates are fast, including the reduction of catalyst (copper oxide to copper metal). Similar little dependence on the H2 pressure has been also observed in the case of MoOx–dpAu/TiO2. The reaction order with respect to the concentration of the substrate (1,4-AHERY) was 0.10 (Fig. 6). The small reaction order suggests that the adsorption of the substrate onto the catalyst surfaces is fast and saturated. Based on the generally accepted mechanism of DODH and the currently observed kinetics, the reaction proceeds via the fast reduction of MoOx species, fast adsorption of the substrate to form Mo diolate intermediates, and extrusion of the olefinic product from the Mo diolate. The extrusion of the olefinic product is the rate-determining step. This mechanism is similar to the proposed ones for MoOx–Au/TiO2 and ReOx–M/CeO2 (M = Pd, Au, Ag) catalysts.32,34,35,50 | ||
Fig. 5 Dependence of the initial reaction rate over the MoOx–Cu–Na/TiO2 catalyst on H2 pressure. (a) Raw data. (b) Double logarithmic plot of the reaction rate as a function of H2 pressure. Reaction rate (mmol (gcat h)−1) = yield2,5-DHF (mmol)/(reaction time (h) × catalyst amount (g)). Reaction conditions: 1,4-AHERY (0.5 g), 1,2-DME (4 g), n-dodecane (internal standard) (0.1 g), MoOx–Cu–Na/TiO2 (1 wt% Mo, 2 wt% Cu, Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Na = 100 Na = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15) (0.15 g), H2 (2–8 MPa), 423 K, 0–2 h. The details are listed in Table S13.† 15) (0.15 g), H2 (2–8 MPa), 423 K, 0–2 h. The details are listed in Table S13.† | ||
 | ||
Fig. 6 Dependence of the initial reaction rate over the MoOx–Cu–Na/TiO2 catalyst on the substrate concentration. (a) Raw data. (b) Double logarithmic plot of the reaction rate as a function of the substrate concentration. Reaction rate (mmol (gcat h)−1) = yield2,5-DHF (mmol)/(reaction time (h) × catalyst amount (g)). Reaction conditions: 1,4-AHERY 0.5 g, 1,2-DME 2–9.5 g, n-dodecane (internal standard) (0.1 g) MoOx–Cu–Na/TiO2 (1.0 wt% Mo, 2.0 wt% Cu, Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Na = 100 Na = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15) (0.15 g), H2 (8 MPa), 423 K, 0–2 h. The details are listed in Table S14.† 15) (0.15 g), H2 (8 MPa), 423 K, 0–2 h. The details are listed in Table S14.† | ||
Catalyst characterization
The MoOx–Cu/TiO2 catalysts were characterized by X-ray diffraction (XRD), scanning-transmission electron microscopy with energy-dispersive X-ray spectroscopy (STEM-EDX), temperature-programmed reduction with H2 (H2-TPR) and X-ray absorption spectroscopy (XAS).The XRD patterns of the MoOx–Cu–Na/TiO2 catalysts after their catalytic use (Fig. S6†) did not show any peaks related to Mo species or Cu species regardless of the Cu amounts except for the case of the sample with a high Cu loading (5 wt%). The absence of MoOx peaks has been also reported for MoOx–dpAu/TiO2 catalysts with the same Mo loading amount, which was due to the low Mo amount and high dispersion of Mo species.50 The absence of XRD peaks of 0.5–3 wt% Cu metal suggests the high dispersion of Cu species; however, the different possibilities of low crystallinity and/or low reduction degree of Cu species cannot be ruled out. In the XRD pattern of the catalysts with 5 wt% Cu, the peaks assignable to the fcc Cu metal, which was formed by the reduction of Cu species in the as-prepared catalyst under the reaction conditions, were observed. The particle size of Cu in the 5 wt% Cu catalyst was calculated from XRD to be 18 nm.
The STEM-EDX images of the MoOx–Cu–Na/TiO2 catalysts are shown in Fig. 7 and S7.† The Mo species were distributed uniformly in all the catalysts, agreeing with the XRD results. The uniform distribution of Mo species has also been reported for MoOx–Au/TiO2.50 The Cu species were present as particles with ca. 20 nm in diameter. In the catalyst with 1 wt% Cu, the spherical Cu particles were sparsely present. The lower activity of 1 wt% Cu catalyst could be due to the too small number of Cu particles by their aggregation; the portions in the absence of Cu particles do not have activity. The number of Cu particles was larger in the Cu 2 wt% catalyst without a significant change of the particle size of Cu species. On the other hand, in the Cu 5 wt% catalyst, the shape of Cu particles became block-like, and Cu species were also widely distributed on the support. Considering the XRD results, these Cu particles in the 5 wt% Cu catalyst were fcc Cu0 crystals. The structural change can be related to the selectivity change found in Fig. 2.
 | ||
Fig. 7 STEM-EDX images of MoOx–Cu–Na/TiO2 (1 wt% Mo, 2 wt% Cu, Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Na = 100 Na = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15), after the reaction (4 h in Fig. 1). 15), after the reaction (4 h in Fig. 1). | ||
In the H2-TPR profiles (Fig. S9†), MoOx/TiO2 showed a reduction signal between 673 and 773 K with a peak maximum at 725 K. The H2 consumption amount calculated from this signal was 0.27 mmol g−1, which was about 3 equivalents to the Mo amount (1 wt% Mo = 0.10 mmol-Mo g−1). Considering that the complete reduction of MoO3 to Mo metal with H2 is difficult, both MoO3 and surface TiO2 were reduced at the same time. The simultaneous reduction of MoO3 and TiO2 support was also reported for MoOx/TiO2 (P-25);50 however, the current MoOx/TiO2 (anatase) showed the smaller total H2 consumption amount and higher starting temperature of the reduction than the reported MoOx/TiO2 (P-25) catalyst. The anatase TiO2 support in MoOx/TiO2 seems more difficult to be reduced than the rutile TiO2 support. The addition of Cu species to MoOx/TiO2 drastically shifted the reduction signal toward the low-temperature side. In the case of MoOx–Cu/TiO2, no peak appeared at around 725 K, and a peak at 481 K was observed. Although the peak-top temperature (481 K) was higher than the standard reaction temperature (463 K), the reduction started below 463 K. For MoOx–Ni/TiO2, MoOx–Co/TiO2 and MoOx–Fe/TiO2, the reduction did not begin below 593 K. The difficulty in the reduction of MoOx species could be related to the low catalytic activity (Table 1). For MoOx–Ag/TiO2 with the similar activity to MoOx–Cu(–Na)/TiO2, the main reduction signal was located at a similarly high temperature to those of MoOx–Ni/TiO2 and MoOx–Fe/TiO2, while the weak reduction signal was present at around the standard reaction temperature of 463 K. Both Cu and Ag can promote the reduction of MoOx species. The H2-TPR profiles for MoOx–Cu–Na/TiO2 with the different Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Na molar ratios are also shown in Fig. S9.† The addition of Na only slightly shifted the reduction peak by ca. 10 K. This observation suggests that the reduction of Mo with H2 was not affected by the Na additive. These results agreed with that the activity of MoOx–Cu–Na/TiO2 was almost independent on the Na amount (Table S2†). Based on the H2-TPR data of the catalysts with the different Mo
Na molar ratios are also shown in Fig. S9.† The addition of Na only slightly shifted the reduction peak by ca. 10 K. This observation suggests that the reduction of Mo with H2 was not affected by the Na additive. These results agreed with that the activity of MoOx–Cu–Na/TiO2 was almost independent on the Na amount (Table S2†). Based on the H2-TPR data of the catalysts with the different Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Na ratios, we assumed that the Na+ ion did not interact with MoOx and rather basically bound on the TiO2 support surface. The H2-TPR profiles for the MoOx–Cu–Na/TiO2 catalysts (Mo
Na ratios, we assumed that the Na+ ion did not interact with MoOx and rather basically bound on the TiO2 support surface. The H2-TPR profiles for the MoOx–Cu–Na/TiO2 catalysts (Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Na = 100
Na = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15) with the different Mo loading amounts are shown in Fig. S8.† Cu/TiO2 and MoOx–Cu–Na/TiO2 with Mo 0.5 wt% and Cu 1 wt% showed similar profiles with the reduction signal at around 473 K, and the first peaks (293 K–540 K) exhibited higher H2 consumption than that required for CuO reduction, which suggested that Mo, Cu and a part of surface Ti species were simultaneously reduced. The reduction is suggested to proceed well at the reaction temperature of 463 K. However, when the Mo loading amount increased over 1 wt%, the reduction signal of Cu was significantly shifted toward the high-temperature side. For the MoOx–Cu–Na/TiO2 catalyst with the Mo loading amount of 2 wt%, the peak of reduction temperature appeared at 553 K, and the reduction did not start below 463 K. The reduction of Cu species was thus suppressed by the excess amount of Mo, and this behavior is related to the low activity of MoOx–Cu–Na/TiO2 catalysts with the high Mo loadings (Fig. S9†).
15) with the different Mo loading amounts are shown in Fig. S8.† Cu/TiO2 and MoOx–Cu–Na/TiO2 with Mo 0.5 wt% and Cu 1 wt% showed similar profiles with the reduction signal at around 473 K, and the first peaks (293 K–540 K) exhibited higher H2 consumption than that required for CuO reduction, which suggested that Mo, Cu and a part of surface Ti species were simultaneously reduced. The reduction is suggested to proceed well at the reaction temperature of 463 K. However, when the Mo loading amount increased over 1 wt%, the reduction signal of Cu was significantly shifted toward the high-temperature side. For the MoOx–Cu–Na/TiO2 catalyst with the Mo loading amount of 2 wt%, the peak of reduction temperature appeared at 553 K, and the reduction did not start below 463 K. The reduction of Cu species was thus suppressed by the excess amount of Mo, and this behavior is related to the low activity of MoOx–Cu–Na/TiO2 catalysts with the high Mo loadings (Fig. S9†).
The results of Cu K-edge X-ray absorption near-edge structure (XANES) and extended X-ray absorption fine structure (EXAFS) in both k-space and R-space are shown in Fig. 8. The XANES spectrum of MoOx–Cu–Na/TiO2 after its catalytic use was similar to that of Cu0 foil. The EXAFS spectrum of the same catalyst can be fitted well with the Cu–Cu metal bond with a coordination number (CN) of 10.7 (Table S15†). These results agreed well with the STEM-EDX results where the Cu species were present as relatively large metallic particles.
 | ||
Fig. 8 (A) Cu K-edge XANES spectra of catalysts and standard compounds. (B) k3-Weighted EXAFS oscillations shown in the k-space. (C) k3-Weighted EXAFS oscillations shown in the R-space; the data were not phase-corrected. (D) Filtered spectra (solid lines) and fitting (dotted lines). Fourier-filtering range is listed in Table S15.† (a) Cu foil, (b) MoOx–Cu–Na/TiO2 (2 wt% Cu, 1 wt% Mo, Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Na = 100 Na = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15) after the reaction for 4 h, (c) CuO. 15) after the reaction for 4 h, (c) CuO. | ||
The Mo K-edge XANES and EXAFS data are shown in Fig. 9. The XANES spectrum of MoOx–Cu–Na/TiO2 after the reaction (f) was similar to those of MoO2 (b) and MoOx–Au/TiO2 after the reaction (i). The EXAFS spectrum and curve-fitting result (Table S16†) of MoOx–Cu–Na/TiO2 after the reaction (f) were also similar to those of MoOx–Au/TiO2 after the reaction (i). The results demonstrate that the structure of Mo species in MoOx–Cu–Na/TiO2 during the reaction was similar to that in MoOx–Au/TiO2 where MoIV oxide cluster species are dominant.50 A recent DFT study of MoOx species on TiO2 surfaces also suggests that the dimeric Mo species are good active sites for DODH when H2 is sufficiently activated.73 On the other hand, Cu-free MoOx–Na/TiO2 after the reaction (g) provided a similar spectrum to that of MoO3 (c). Although the structure was different from MoOx–Au/TiO2 before the reaction where AHM-like Mo K-edge EXAFS spectrum was obtained ((h) and (e)),50 the valence of Mo was +6, demonstrating that Cu species promoted the reduction of Mo species like Au.
 | ||
Fig. 9 (A) Mo K-edge XANES spectra of catalysts and standard compounds. (B) k3-Weighted EXAFS oscillations shown in the k-space. (C) k3-Weighted EXAFS oscillations shown in the R-space, the data were not phase-corrected. (D) Filtered spectra (solid lines) and fitting (dotted lines). Fourier-filtering range is listed in Table S16.† (a) Mo foil, (b) MoO2, (c) MoO3, (d) Na2MoO4, (e) (NH4)6Mo7O24·4H2O (AHM), (f) MoOx–Cu–Na/TiO2 (2 wt% Cu, 1 wt% Mo, Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Na = 100 Na = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15) after use for 4 h, (g) MoOx–Na/TiO2 (1.0 wt% Mo, Mo 15) after use for 4 h, (g) MoOx–Na/TiO2 (1.0 wt% Mo, Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Na = 100 Na = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15) after use for 4 h, (h) MoOx–dpAu/TiO2 (P-25 support, Au 0.3 wt%, Mo 1 wt%) before the reaction; (i) MoOx–dpAu/TiO2 (P-25 support, Au 0.3 wt%, Mo 1 wt%) after use for 24 h. The data of (h), (i) and standard compounds have been reported.50 15) after use for 4 h, (h) MoOx–dpAu/TiO2 (P-25 support, Au 0.3 wt%, Mo 1 wt%) before the reaction; (i) MoOx–dpAu/TiO2 (P-25 support, Au 0.3 wt%, Mo 1 wt%) after use for 24 h. The data of (h), (i) and standard compounds have been reported.50 | ||
Conclusions
MoOx–Cu/TiO2 catalysts exhibited a good performance in the DODH reaction of various diols. The use of a small amount of the basic Mo precursor Na2MoO4 (MoOx–Cu–Na/TiO2) enhanced the selectivity to DODH products, achieving the highest yield of 2,5-dihydrofuran (2,5-DHF) at 81% from 1,4-anhydroerythritol (1,4-AHERY). The Cu loading amount much affects the performance of MoOx–Cu–Na/TiO2: the loading amount less than 2 wt% decreased the activity and that above 3 wt% much decreased the selectivity to the target product. In comparison with a more expensive MoOx–Au/TiO2 catalyst, a higher yield and higher initial rate were obtained by MoOx–Cu–Na/TiO2. This catalyst maintained its performance through at least three reuse cycles without significant drops in the conversion or selectivity when the spent catalyst was calcined before each run. The MoOx–Cu–Na/TiO2 catalyst displayed broad substrate scope including both linear and cyclic diols. Notably, this catalyst also worked well for tartaric ester, which is difficult to be converted to the corresponding DODH product in the reported catalysts with H2 as the reductant. The kinetic study with the MoOx–Cu–Na/TiO2 catalyst revealed that the release of the alkene is the rate-determining step, which is consistent with the general DODH mechanism with a sufficient supply of the reductant. Through various characterization studies, it was observed that Cu existed as metallic particles in the optimized catalyst (Cu 2 wt%, Mo 1 wt%), while MoVI was present on the surface of the catalyst. The Mo active site underwent the transition from the original oxidation state of +6 to +4 upon the activation of H2 facilitated by Cu, in a similar way to MoOx–M/TiO2 (M: noble metal) catalysts.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was supported by KAKENHI 23H05404, 23K20034 and 23K26451. J. G. appreciates the support from the “Amano Institute of Technology Scholarship” and “Global Hagi Scholarship”. The authors also appreciate the assistance in STEM-EDS measurement by Mr Yuichiro Hayasaka (The Electron Microscopy Center, Tohoku University).References
- A. Corma, S. Iborra and A. Velty, Chem. Rev., 2007, 107, 2411–2502 CrossRef CAS.
- J. J. Bozell and G. R. Petersen, Green Chem., 2010, 12, 539–554 RSC.
- M. J. Climent, A. Corma and S. Iborra, Green Chem., 2011, 13, 520–540 RSC.
- M. Besson, P. Gallezot and C. Pinel, Chem. Rev., 2014, 114, 1827–1870 CrossRef CAS.
- B. M. Stadler, C. Wulf, T. Werner, S. Tin and J. G. de Vries, ACS Catal., 2019, 9, 8012–8067 CrossRef CAS.
- T. A. Ewing, N. Nouse, M. van Lint, J. van Haveren, J. Hugenholtz and D. S. van Es, Green Chem., 2022, 24, 6373–6405 RSC.
- K. Tomishige, Y. Nakagawa and M. Tamura, Green Chem., 2017, 19, 2876–2924 RSC.
- K. Tomishige, M. Yabushita, J. Cao and Y. Nakagawa, Green Chem., 2022, 24, 5652–5690 RSC.
- Y. Nakagawa, M. Yabushita and K. Tomishige, RSC Sustainability, 2023, 1, 814–837 RSC.
- K. Avasthi, A. Bohre, M. Grilc, B. Likozar and B. Saha, Catal. Sci. Technol., 2020, 10, 5411–5437 RSC.
- D. J. Ward, D. J. Saccomando, G. Walker and S. M. Mansell, Catal. Sci. Technol., 2023, 13, 2638–2647 RSC.
- C. Boucher-Jacobs and K. M. Nicholas, in Selective Catalysis for Renewable Feedstocks and Chemicals, ed. K. M. Nicholas, 2014, vol. 353, pp. 163–184 Search PubMed.
- N. N. Tshibalonza and J.-C. M. Monbaliu, Green Chem., 2020, 22, 4801–4848 RSC.
- F. C. Jentoft, Catal. Sci. Technol., 2022, 12, 6308–6358 RSC.
- S. Raju, J. T. Jastrzebski, M. Lutz, L. O. Witteman, J. R. Dethlefsen, P. Fristrup, M.-E. Moret and R. J. K. Gebbink, Inorg. Chem., 2015, 54, 11031–11036 CrossRef CAS.
- A. R. Petersen and P. Fristrup, Chem. – Eur. J., 2017, 23, 10235–10243 CrossRef CAS PubMed.
- J. R. Dethlefsen and P. Fristrup, ChemSusChem, 2015, 8, 767–775 CrossRef CAS PubMed.
- G. K. Cook and M. A. Andrews, J. Am. Chem. Soc., 1996, 118, 9448–9449 CrossRef CAS.
- B. Wozniak, Y. Li, S. Tin and J. G. de Vries, Green Chem., 2018, 20, 4433–4437 RSC.
- I. Ahmad, G. Chapman and K. M. Nicholas, Organometallics, 2011, 30, 2810–2818 CrossRef CAS.
- J. Yi, S. Liu and M. M. Abu-Omar, ChemSusChem, 2012, 5, 1401–1404 CrossRef CAS PubMed.
- E. Arceo, J. A. Ellman and R. G. Bergman, J. Am. Chem. Soc., 2010, 132, 11408–11409 CrossRef CAS PubMed.
- M. Shiramizu and F. D. Toste, Angew. Chem., Int. Ed., 2012, 51, 8082–8086 CrossRef CAS.
- G. W. Huber and A. Corma, Angew. Chem., Int. Ed., 2007, 46, 7184–7201 CrossRef CAS.
- S. Kim, E. E. Kwon, Y. T. Kim, S. Jung, H. J. Kim, G. W. Huber and J. Lee, Green Chem., 2019, 21, 3715–3743 RSC.
- X. Yao, T. J. Strathmann, Y. Li, L. E. Cronmiller, H. Ma and J. Zhang, Green Chem., 2021, 23, 1114–1129 RSC.
- Y. S. Yun, C. E. Berdugo-Díaz and D. W. Flaherty, ACS Catal., 2021, 11, 11193–11232 CrossRef CAS.
- M. Žula, M. Grilc and B. Likozar, Chem. Eng. J., 2022, 444, 136564 CrossRef.
- V. Canale, L. Tonucci, M. Bressan and N. d'Alessandro, Catal. Sci. Technol., 2014, 4, 3697–3704 RSC.
- J. E. Ziegler, M. J. Zdilla, A. J. Evans and M. M. Abu-Omar, Inorg. Chem., 2009, 48, 9998–10000 CrossRef CAS.
- N. Ota, M. Tamura, Y. Nakagawa, K. Okumura and K. Tomishige, Angew. Chem., Int. Ed., 2015, 54, 1897–1900 CrossRef CAS PubMed.
- N. Ota, M. Tamura, Y. Nakagawa, K. Okumura and K. Tomishige, ACS Catal., 2016, 6, 3213–3226 CrossRef CAS.
- S. Tazawa, N. Ota, M. Tamura, Y. Nakagawa, K. Okumura and K. Tomishige, ACS Catal., 2016, 6, 6393–6397 CrossRef CAS.
- Y. Nakagawa, S. Tazawa, T. Wang, M. Tamura, N. Hiyoshi, K. Okumura and K. Tomishige, ACS Catal., 2018, 8, 584–595 CrossRef CAS.
- K. Yamaguchi, J. Cao, M. Betchaku, Y. Nakagawa, M. Tamura, A. Nakayama, M. Yabushita and K. Tomishige, ChemSusChem, 2022, e202102663 CrossRef CAS PubMed.
- K. Yamaguchi, Y. Nakagawa, C. Li, M. Yabushita and K. Tomishige, ACS Catal., 2022, 12, 12582–12595 CrossRef CAS.
- L. Hills, R. Moyano, F. Montilla, A. Pastor, A. Galindo, E. Álvarez, F. Marchetti and C. Pettinari, Eur. J. Inorg. Chem., 2013, 2013, 3352–3361 CrossRef CAS.
- K. Beckerle, A. Sauer, T. P. Spaniol and J. Okuda, Polyhedron, 2016, 116, 105–110 CrossRef CAS.
- M. Stalpaert and D. De Vos, ACS Sustainable Chem. Eng., 2018, 6, 12197–12204 CrossRef CAS.
- R. Tran and S. M. Kilyanek, Dalton Trans., 2019, 48, 16304–16311 RSC.
- J. Li, M. Lutz and R. Gebbink, ChemCatChem, 2020, 12, 6356–6365 CrossRef CAS.
- R. Tran, C. A. Canote and S. M. Kilyanek, Organometallics, 2023, 42, 1190–1197 CrossRef CAS.
- N. Wagner, W.-C. Tang, J. Wagner, B. Nguyen, J. Y. Lam, S. Gibbons-Stovall, A. Matias, S. Martinez, T. Trieu-Tran, G. Clabaugh, C. Navarro, I. Abboud, F. Flores, K. M. Nicholas and A. John, Catal. Sci. Technol., 2024 10.1039/D4CY00441H.
- R. S. Srivastava, J. Organomet. Chem., 2024, 1003, 122942 CrossRef.
- J. R. Dethlefsen, D. Lupp, B. C. Oh and P. Fristrup, ChemSusChem, 2014, 7, 425–428 CrossRef CAS PubMed.
- J. R. Dethlefsen, D. Lupp, A. Teshome, L. B. Nielsen and P. Fristrup, ACS Catal., 2015, 5, 3638–3647 CrossRef CAS.
- C. A. Navarro and A. John, Inorg. Chem. Commun., 2019, 99, 145–148 CrossRef CAS.
- L. Sandbrink, K. Beckerle, I. Meiners, R. Liffmann, K. Rahimi, J. Okuda and R. Palkovits, ChemSusChem, 2017, 10, 1375–1379 CrossRef CAS PubMed.
- J. Sebastian, C. Mebrahtu and R. Palkovits, Catal. Sci. Technol., 2023, 13, 1087–1097 RSC.
- S. Hacatrjan, L. Liu, J. Gan, Y. Nakagawa, J. Cao, M. Yabushita, M. Tamura and K. Tomishige, Catal. Sci. Technol., 2022, 12, 2146–2161 RSC.
- S. Albarracin-Suazo, L. Freitas de Lima e Freitas, B. MacQueen, A. Heyden, J. A. Lauterbach, E. Nikolla and Y. J. Pagán-Torres, ACS Sustainable Chem. Eng., 2022, 10, 5719–5727 CrossRef CAS.
- B. E. Sharkey, A. L. Denning, F. C. Jentoft, R. Gangadhara, T. V. Gopaladasu and K. M. Nicholas, Catal. Today, 2018, 310, 86–93 CrossRef CAS.
- Y. Xi, J. Lauterbach, Y. Pagan-Torres and A. Heyden, Catal. Sci. Technol., 2020, 10, 3731–3738 RSC.
- M. H. Li, S. Y. Chen, Q. K. Jiang, Q. L. Chen, X. Wang, Y. Yan, J. Liu, C. C. Lv, W. P. Ding and X. F. Guo, ACS Catal., 2021, 11, 3026–3039 CrossRef CAS.
- G. A. Filonenko, R. van Putten, E. J. M. Hensen and E. A. Pidko, Chem. Soc. Rev., 2018, 47, 1459–1483 RSC.
- G. Kyriakou, M. B. Boucher, A. D. Jewell, E. A. Lewis, T. J. Lawton, A. E. Baber, H. L. Tierney, M. Flytzani-Stephanopoulos and E. C. H. Sykes, Science, 2012, 335, 1209–1212 CrossRef CAS PubMed.
- S. De, J. G. Zhang, R. Luque and N. Yan, Energy Environ. Sci., 2016, 9, 3314–3347 RSC.
- M. Behrens, F. Studt, I. Kasatkin, S. Kuhl, M. Havecker, F. Abild-Pedersen, S. Zander, F. Girgsdies, P. Kurr, B. L. Kniep, M. Tovar, R. W. Fischer, J. K. Norskov and R. Schlogl, Science, 2012, 336, 893–897 CrossRef CAS PubMed.
- F. Cannizzaro, E. J. M. Hensen and I. A. W. Filot, ACS Catal., 2023, 13, 1875–1892 CrossRef CAS PubMed.
- B. Ravel and M. Newville, J. Synchrotron Radiat., 2005, 12, 537–541 CrossRef CAS PubMed.
- Y. Nakagawa, T. Kasumi, J. Ogihara, M. Tamura, T. Arai and K. Tomishige, ACS Omega, 2020, 5, 2520–2530 CrossRef CAS PubMed.
- T. Wang, S. Liu, M. Tamura, Y. Nakagawa, N. Hiyoshi and K. Tomishige, Green Chem., 2018, 20, 2547–2557 RSC.
- T. Wang, M. Tamura, Y. Nakagawa and K. Tomishige, ChemSusChem, 2019, 12, 3615–3626 CrossRef CAS PubMed.
- W. I. Bengough and G. B. Park, Eur. Polym. J., 1978, 14, 889–894 CrossRef CAS.
- F. Bovey and I. Kolthoff, Chem. Rev., 1948, 42, 491–525 CrossRef CAS PubMed.
- A. L. Denning, H. Dang, Z. Liu, K. M. Nicholas and F. C. Jentoft, ChemCatChem, 2013, 5, 3567–3570 CrossRef CAS.
- A. Nacy, L. F. de Lima e Freitas, S. Albarracín-Suazo, G. Ruiz-Valentín, C. A. Roberts, E. Nikolla and Y. J. Pagán-Torres, ChemCatChem, 2021, 13, 1294–1298 CrossRef CAS.
- M. Shiramizu and F. D. Toste, Angew. Chem., Int. Ed., 2013, 52, 12905–12909 CrossRef CAS PubMed.
- X. Li, D. Wu, T. Lu, G. Yi, H. Su and Y. Zhang, Angew. Chem., Int. Ed., 2014, 53, 4200–4204 CrossRef CAS PubMed.
- W. Deng, L. Yan, B. Wang, Q. Zhang, H. Song, S. Wang, Q. Zhang and Y. Wang, Angew. Chem., Int. Ed., 2021, 60, 4712–4719 CrossRef CAS PubMed.
- J. H. Jang, J. T. Hopper, I. Ro, P. Christopher and M. M. Abu-Omar, Catal. Sci. Technol., 2023, 13, 714–725 RSC.
- X. Luo, R. Lu, H. Jiang, X. Si, J. Xu and F. Lu, Ind. Eng. Chem. Res., 2021, 60, 4510–4515 CrossRef CAS.
- D. Asada, T. Ikeda, K. Muraoka, Y. Nakagawa, K. Tomishige and A. Nakayama, J. Phys. Chem. C, 2022, 126, 20375–20387 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4gc02006e |
| This journal is © The Royal Society of Chemistry 2024 |

