Carbon-negative hydrogen: aqueous phase reforming (APR) of glycerol over NiPt bimetallic catalyst coupled with CO2 sequestration†
Leoncio
Santiago-Martínez
 ab,
Mengting
Li
a,
Paola
Munoz-Briones
ab,
Mengting
Li
a,
Paola
Munoz-Briones
 a,
Javiera
Vergara-Zambrano
a,
Javiera
Vergara-Zambrano
 a,
Styliani
Avraamidou
a,
James A.
Dumesic
a,
Styliani
Avraamidou
a,
James A.
Dumesic
 a and
George W.
Huber
a and
George W.
Huber
 *a
*a
aDepartment of Chemical and Biological Engineering, University of Wisconsin, Madison, WI 53706, USA. E-mail: gwhuber@wisc.edu
bDOE Center for Advanced Bioenergy and Bioproducts Innovation, University of Illinois at Urbana-Champaign, Urbana, IL, USA
First published on 17th May 2024
Abstract
Herein we report the production of high-pressure (19.3 bar), carbon-negative hydrogen (H2) from glycerol with a purity of 98.2 mol% H2, 1.8 mol% light hydrocarbons (mainly methane), and 400 ppm of CO. Aqueous phase reforming (APR) of 10 wt% glycerol solution was studied with a series of NiPt alumina bimetallic catalysts supported on alumina. The Ni8Pt1-450 catalyst had the highest hydrogen selectivity (95.6%) and the lowest alkanes selectivity (3.7%) of the tested catalysts. The hydrogen selectivity decreased in the order of Ni8Pt1-450 > Ni8Pt1-260 > Ni1Pt1-260 > Pt-260. The CO2 was sequestered with CaO adsorbent which formed CaCO3. We measured the adsorption capacity of the CaO adsorbent at different temperatures. Life cycle analysis showed that the APR of glycerol coupled with CO2 capture has net negative CO2 equivalent greenhouse gas emissions. The CO2 emissions are −9.9 kg CO2 eq./kg H2 and −50.1 kg CO2 eq./kg H2 when grid electricity and renewable electricity are used, respectively, and the CO2 is allocated respectively to the mass of products produced. The cost of this H2 (denoted as “green-emerald”) was estimated to be 2.4 USD per kg H2 when grid electricity is used and 2.7 USD per kg H2 when using renewable electricity. The cost of glycerol has the highest contribution of 1.71 USD per kg H2. Participation in the carbon credit markets can further decrease the price of the produced H2.
1. Introduction
Hydrogen (H2) is an important chemical in refining processes, ammonia production, energy storage, and as an alternative and cleaner fuel.1 Hydrogen is mainly produced from steam reforming of natural gas (grey H2), and coal gasification (black H2). These processes emit 8.5 to 12.9 kg CO2/kg H2 and 18 to 26 kg CO2/kg H2, respectively.2–7 To reduce the emissions associated with H2 production, an array of new technologies are under development, all of which produce H2 with a different emission level and energy source. Table 1 shows the H2 types and colors used to identify H2 types according to the Global Energy Infrastructure (GEI).8 Pink, turquoise, and green H2 have the lowest emissions ranging from 0 to 5.1 kg CO2/kg H2. Blue H2 is considered a low-carbon emissions hydrogen with as low as 0.7 kg CO2/kg H2, but could emit even 13 kg CO2/kg H2 due to fugitive methane emissions.5| Hydrogen color/type | Source | Hydrogen production process | CO2 emissions (kg CO2/kg H2) | Ref. |
|---|---|---|---|---|
| Green | Water | Electrolysis with renewable electricity (wind, solar, hydro) | 0–5.1 | 2–6 |
| Purple/pink/red | Electrolysis using nuclear energy | 0–0.6 | 2,4 and 6 | |
| Yellow | Electrolysis using mixed-origin grid energy | 14.5–28.6 | 4 and 5 | |
| Blue | Natural gas coal | Natural gas steam reforming or coal gasification with CCUS | 0.7–13 | 2–6 |
| Turquoise | Natural gas | Pyrolysis of natural gas | 1.9–4.8 | 2 |
| Grey | Natural gas steam reforming | 8.5–12.9 | 2–6 | |
| Brown | Lignite | Lignite gasification | 20–25.3 | 6 and 7 |
| Black | Coal | Coal gasification | 18.0–26 | 2–7 |
| Aqua | Oil sands or conventional oil fields | Oxygen injection into heavy oil reservoirs or oil sands deep underground | 0 | 6 and 9 |
| Gold | Underground deposits | Refers to hydrogen produced by fermenting microbes in depleted oil wells or naturally occurring H2 | Not available | 10–12 |
| White | Naturally occurring H2 | Not available | 13 | |
| Orange | Induced by injecting water in reactive formations | Not available | 13 | |
| Not classified | Biomass | Biomass (wood chips, and corn stover) gasification | 0.41–2.68 | 3 and 4 |
| Wood chips gasification with CCUS | −21–(−16) | 4 | ||
| Ethanol reforming | 9.19–14 | 3 | ||
| APR of glycerol | 3.88–4.11 | 14 |
The importance of sustainably produced H2 has been highlighted recently by the U.S. Department of Energy through the establishment of the 2020 H2 Program Plan15 to develop pathways to produce carbon-neutral H2 (0 kg CO2/kg H2) and carbon-negative H2 (<0 kg CO2/kg H2) at a low cost. The price target is 2.00 USD per kg of H2 production for transportation applications and 1.00 USD per kg for industrial and stationary power generation applications. The low-carbon H2 sources include H2 production from renewable sources like biomass or waste conversion, from water splitting using renewable electricity (green H2), and from natural gas steam reforming with carbon capture utilization and storage (CCUS) (blue H2).
Biomass captures CO2 from the atmosphere during the process of photosynthesis. Using biomass to produce H2 offers the advantage of producing H2 from a low-carbon source. Combining H2 production from biomass with carbon capture and storage during H2 production has the potential to produce carbon-negative H2.16 High-pressure H2 can be produced from biomass-derived sugar alcohols (ethylene glycol, glycerol, or sorbitol) through aqueous phase reforming (APR).17–22 Biomass-derived H2 can be classified as carbon-negative H2 when the CO2 is captured or sequestrated, as shown in Fig. 1. High-pressure CO2 and H2 are produced from APR. This CO2 can be chemically trapped in the carbonation process with the possibility of storing the carbon in geological Ca formations in the form of CaCO3. Thus, there is no direct CO2 emitted to the atmosphere during the process. Carbon capture usage and storage (CCUS) has been applied to the field of biomass conversion,23–27 primarily in the biomass gasification process with in situ CO2 adsorption. For example, Xu et al.26 reported the use of Fe/CaO catalyst for wood sawdust gasification to H2-rich syngas production, in which the presence of iron improves the CO2 adsorption capacity of CaO. Doranehgard et al.27 reported the use of CaO to improve tar cracking and reduce CO2 emissions in rice husk gasification. Han et al.24 found that pressurized sawdust gasification instead of atmospheric gasification in CaO, produces syngas with a higher H2 and lower CO2 content by promoting the gasification reactions and the CaO carbonation. However, gasification processes are typically carried out at low pressure, and they therefore require downstream compression of the H2.
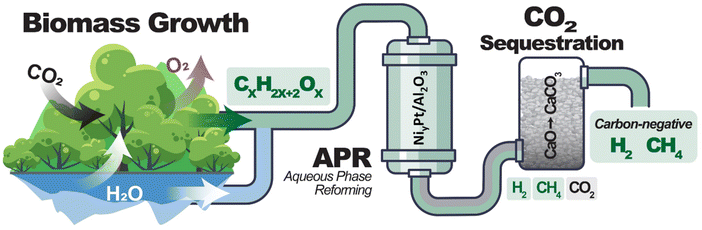 | ||
| Fig. 1 Schematic representation of a process to produce carbon-negative H2 from biomass-derived compounds through aqueous phase reforming (APR) technology and CO2 sequestration. | ||
Whole biomass,28,29 cellulose,30–33 and xylose34–36 have all been used as feedstocks for APR. Valenzuela et al.28 reported the APR of Southern pine sawdust, using sulfuric acid (2 M) and Pt/Al2O3 catalyst at 225 °C in a batch reactor. Wen et al.32 reported that the use of Pt/C can convert the cellulose efficiently into H2 in a one-pot APR at 260 °C, with 40% H2 selectivity. Later they used31 a Ce-modified RANEY® Ni catalysts at the same (260 °C) temperature and reported an increased H2 selectivity and decreased methane selectivity when large quantities of Ce were added at a Ce![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ni ratio of 0.054. All these studies were carried out in batch reactors with a biomass slurry feed. Industrially it is not practical to perform APR in batch reactors.
Ni ratio of 0.054. All these studies were carried out in batch reactors with a biomass slurry feed. Industrially it is not practical to perform APR in batch reactors.
Hydrogen selectivity in the APR depends on the reaction conditions and the catalysts used. Several studies18,20,37–44,45–49 reported reaction temperatures from 210 °C to 250 °C, with system pressures between 2 to 7 bar above the bubble point of water to maintain the non-volatile feedstock in the liquid phase. In some cases,37,50–54 the system pressure was varied from 10 to 21 bar above the bubble point of water. Davda et al.19 and Shabaker et al.17 reported that an increase in the total system pressure inhibits H2 production. Huber et al.18 studied the use of bimetallic PtNi over alumina, with a Pt![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ni molar ratio of 1
Ni molar ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, and 1
5, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 for the APR of ethylene glycol at 210 °C. Their findings indicate that a Pt
8 for the APR of ethylene glycol at 210 °C. Their findings indicate that a Pt![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ni ratio of 1
Ni ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 has a higher H2 selectivity (91.2%), and no alkane production compared to the pure Pt-based catalyst. El Doukkali et al.37 reported the use of a bimetallic PtNi catalyst over alumina, prepared by a sol–gel method containing 8 wt% Ni. This PtNi catalyst was more active for the APR process of glycerol than other bimetallic catalysts (PtFe, PtCo) or monometallic Ni or Pt over alumina. However, the H2 selectivity (40%) was lower than with the monometallic Pt catalyst. Other authors37,38,55,56 reported similar conclusions for the NiPt bimetallic catalysts for different Ni loadings, ranging from 0.75 wt% to 12 wt%.
5 has a higher H2 selectivity (91.2%), and no alkane production compared to the pure Pt-based catalyst. El Doukkali et al.37 reported the use of a bimetallic PtNi catalyst over alumina, prepared by a sol–gel method containing 8 wt% Ni. This PtNi catalyst was more active for the APR process of glycerol than other bimetallic catalysts (PtFe, PtCo) or monometallic Ni or Pt over alumina. However, the H2 selectivity (40%) was lower than with the monometallic Pt catalyst. Other authors37,38,55,56 reported similar conclusions for the NiPt bimetallic catalysts for different Ni loadings, ranging from 0.75 wt% to 12 wt%.
Several authors57–65 have reported the use of CaO as a sorbent for carbon mineralization. Hlaing et al.66 reported that CaO derived from aragonite reached an adsorption capacity of 0.125 mol CO2/mol CaO at 300 °C and 0.869 mol CO2/mol CaO at 700 °C. The carbonation temperature was the most important factor affecting the CaO adsorption capacity. The CO2 capture can be improved by the presence of steam61 in the flue gases during the carbonation at 650 °C or a pre-hydration step65 of the CaO for carbonation at room temperature. Using regenerable solvents in a water solution, such as monoethanolamine or sodium glycinate, can improve carbon mineralization by a single step of CO2 capture and carbon mineralization at low temperatures.64,67
G. Gadikota and A. A. Park68 reported that calcium and magnesium-bearing minerals such as wollastonite, olivine, or serpentine could be used for CO2 mineralization, since they contain non-carbonated Ca or Mg. There are two approaches for CO2 sequestration using minerals: in situ or ex situ carbon mineralization. The in situ carbon mineralization involves the injection of high-pressure CO2 in geological formations containing Ca and/or Mg minerals. For this approach, the CO2 needs to be separated from the hydrogen stream produced in the APR of biobased feedstocks and injected underground. Pressure swing adsorption (PSA)69 can be used to separate the CO2 and produce a high-purity H2 (>98%) stream. PSA has the advantage of low temperature (≈25 °C) operation, pressures between 20 and 40 bar, hydrogen recovery higher than 90%, and the adsorbent can last for several years. In ex situ mineralization, the minerals need to be mined, grounded, and reacted in adsorbers/reactors. The carbonated minerals can be deposited in geological formations or can be used in applications such as filler materials, coatings, construction materials, liming agents to neutralize soil acidity, or remediation of contaminated lands.68
In this study we decided to first use glycerol as a feedstock because of it ease to analyze in the process and to demonstrate integration with carbon capture. APR with carbon capture can have more of an impact when integrated with more abundant feedstocks such as biomass-derived sugar alcohols, xylose, cellulose, or whole biomass. According to the U.S. Department of Energy,15 over a billion tons of dry feedstock, including biomass and waste-stream resources, can be used for sustainable hydrogen production. Thus, future work should focus on how to integrate green-emerald hydrogen production with more abundant biomass feedstocks. The study focuses only on glycerol produced in a restricted area of the U.S. (Wisconsin area) to facilitate the techno-economic, and life cycle analysis. Improved catalysts, with increased stability, selectivity, and activity, are needed if APR is to be implemented commercially.
In this work, we report for the first time, the production of carbon-negative H2 from glycerol using APR coupled with CO2 sequestration by CaO carbonation, producing a high-pressure carbon-negative H2 stream with 98.2% of purity. We will denote this type of H2 as “green-emerald” to differentiate it from the other common types of H2 reported in Table 1. We perform an LCA and economic analysis using our experimental data to estimate the economics and environmental impacts of producing H2 by aqueous phase reforming coupled with CaO adsorption to separate H2 from CO2.
2. Methods
2.1. Catalyst preparation and characterization
Monometallic Pt/γ-Al2O3 and bimetallic NixPt1/γ-Al2O3 catalysts were prepared by incipient wetness impregnation, using Catapal B γ-Al2O3 of Sasol as the support. Catapal B was dried in a vacuum oven (VWR Symphony) at 60 °C for two hours before the impregnation. Pt/γ-Al2O3 with 3 wt% Pt (Pt-260) was prepared using 1 mL of tetraamine platinum(II) nitrate (Sigma-Aldrich) solution per gram of dried support. The catalyst was dried for 2–3 hours and calcined at 260 °C for two hours in a tube furnace using a heating rate of 1 °C min−1 and 100 mL min−1 of air. The calcined catalyst was milled in a mortar until fine powder and used without sieving. To prepare the bimetallic catalysts Ni1Pt1/γ-Al2O3 (Ni1Pt1-260) and Ni8Pt1/γ-Al2O3, a similar procedure was followed, using 1 mL of nickel nitrate hexahydrate solution to get a Ni to Pt atomic ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 8
1 and 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The impregnated samples were dried and calcined similarly as previously described.
1. The impregnated samples were dried and calcined similarly as previously described.
The prepared catalyst was reduced by flowing 100 mL min−1 of hydrogen for two hours at 260 °C, with a heating rate of 1 °C min−1. After the reduction, the samples were passivated for 30 min using a 1 vol% O2/Argon before the characterization with CO chemisorption. For the Ni8Pt1/γ-Al2O3 catalyst, two reduction temperatures were applied, 260 °C (Ni8Pt1-260) and 450 °C (Ni8Pt1-450).
2.2. Aqueous phase reforming (APR)
At the beginning of a run, 1.0 g of calcined catalyst was loaded into a 1/4′′ SS reactor. The catalyst was fixed at the center of the furnace using silica chips (35 mesh) and 0.1 g of fiberglass plugs to separate the silica beds and the catalyst bed. The reactor was kept at the operation temperature by monitoring the temperature with a K-type thermocouple (Omega) and using an aluminum insert in the furnace. The catalyst was reduced in situ at 260 °C for two hours with a heating rate of 1 °C min−1, with 100 mL min−1 of 10 vol% H2/Helium, except for Ni8Pt1/γ-Al2O3, which was reduced at 450 °C. After the two-hour reduction, the reactor was flushed with nitrogen; meanwhile the temperature decreased to the reaction temperature (209 °C). Then, the APR reaction system (Fig. 2) was pressurized at the operation pressure (typically 0.5 bar above the bubble point of water at the operation temperature) using nitrogen. The pressure was controlled by a backpressure regulator (TESCOM 26-1764-24). Once the pressure was stable, the feed solution consisting of 10 wt% glycerol/water was fed using an HPLC pump at a flow rate of 0.06 mL min−1. The product mixture was separated in the liquid–gas separator vessel, in which nitrogen was used as sweep gas at 45 to 50 mL min−1 and bubbled through the liquid phase inside the separator. The gaseous product mixture was then collected and analyzed using a GC (Shimadzu Refinery gas analyzer) equipped with an FID and two TCD detectors. The liquid product collected was analyzed by TOC (Shimadzu TOC-V CPH) to determine the carbon balance. For evaluating the catalysts at different temperatures and pressures, each data point reported in sections 3.1 and 3.2 corresponds to the mean value of three samples taken each two hours between 18 to 26 h of reaction.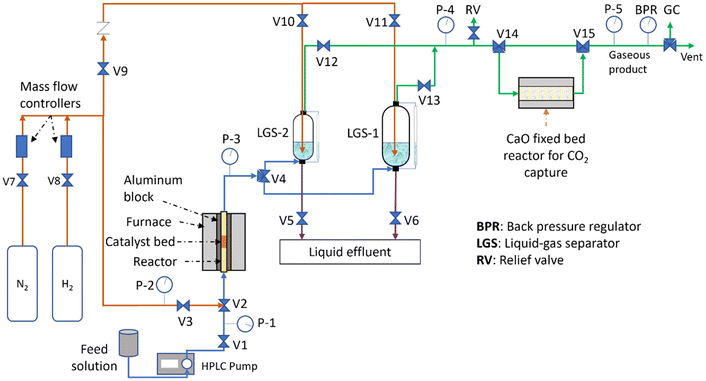 | ||
| Fig. 2 Scheme of the reactor system used for aqueous phase reforming of glycerol with CO2 sequestration. | ||
Hydrogen selectivity was calculated based on the number of molecules of H2 produced. The equations used to evaluate the catalyst performance are given in eqn (1)–(4), in which RR is the H2/CO2 reforming ratio (7/3 for glycerol).
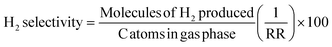 | (1) |
 | (2) |
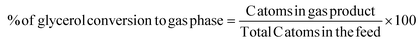 | (3) |
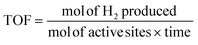 | (4) |
2.3. CO2 adsorption
CO2 adsorption experiments were carried out for the APR of 10 wt% glycerol/water at 209 °C and 19.3 bar over Ni8Pt1-450. For CO2 sequestration, the gas product mixture was passed through a fixed bed reactor packed with commercial CaO from Sigma-Aldrich (4 g, 15 g, and 50 g). The CaO was packed using a 1′′ SS tube in which the CaO was packed in beds of 5 g separated with quartz wool, as shown in Fig. 3; the pressure drop along the packed bed was negligible during the adsorption process.The CO2 adsorption experiment was carried out first with 50 g of CaO at isothermal conditions at 20, 200, 300, 400, 500, 600, and 700 °C, at the APR reaction system pressure. The adsorption started at room temperature until saturation; then the CO2 adsorber was heated to a higher isothermal temperature. On the other hand, for 15 g, and 4 g of CaO, the adsorption was carried out at isothermal conditions at 600 °C. For these experiments, the gas product was sampled at the inlet and the outlet of the CO2 adsorber for GC analysis.
2.4. CO2 adsorption modeling
Eqn (1) and (2) were applied to calculate the adsorption constant for CO2 adsorption over CaO. The CaO bed was considered as a plug-flow reactor. The feed to the bed is a mixture of CO2 and H2. The molar flow rate of H2 is constant, whereas the partial pressure of CO2 and the molar flow rate of CO2 decrease through the bed as CaO captures CO2. We assumed that the capture of CO2 is first order with respect to the partial pressure of CO2 as described in eqn (5), where k is the rate constant for CO2 capture (1/atm-min). The equation to calculate k is described in eqn (6), where FH2 is the molar flow rate of H2 (mol H2 per min). FN2 is the molar flow rate of N2 (mol N2 per min). PCO2 is the partial pressure of CO2 (atm). Ptot is the total pressure (atm). Stot is the total number of CaO active sites in the reactor (mol sites). Note that Stot is a function of time because the total number of active CaO sites decreases as sites become neutralized by the capture of CO2. TOF is the turnover frequency of reacting CO2 with CaO, defined as the rate of CO2 capture per CaO site per unit time (mol CO2 per mol site per min).| TOF = kPCO2 | (5) |
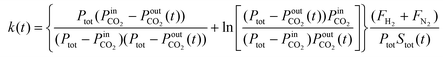 | (6) |
2.5. Life cycle assessment (LCA)
LCA is a tool to quantify the environmental impacts of technologies. LCA was conducted in this study to evaluate the system-wide greenhouse gas (GHG) emissions for hydrogen production. In this study we compared the process in which we coupled APR with CO2 sequestration using CaO at 600 °C, and the process of APR coupled with Pressure Swing Adsorption (PSA) to obtain a high-purity hydrogen stream. For this analysis, six stages (Fig. 4) were considered: (i) crop cultivation, (ii) seed drying, (iii) transportation, (iv) oil processing & refining, (v) glycerol production, and (vi) H2 production. Soybeans were used as the crop, as it is one of the most cultivated crops in Wisconsin (USA). Two scenarios for the electricity source are considered, one using the current Wisconsin grid electricity (Scenario A) and the other one using 100% renewable electricity (Scenario B). The methodology followed for carbon sequestration, and impact allocation, along with the list of assumptions and sources are discussed below.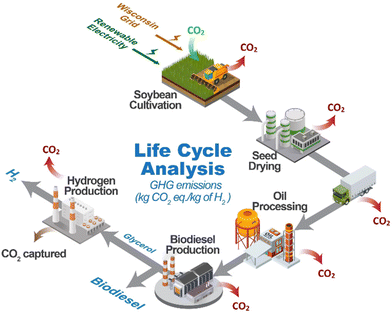 | ||
| Fig. 4 Hydrogen production pathway and types of GHG emissions. The GHG emissions (red arrows), uptake (green arrow), and capture (brown arrow) of each production stage. | ||
The second approach considered for allocation is based on the mass of carbon, which represents the actual carbon flow. It is suggested by Leinonen to be a logical choice of allocation method for forestry products.70 The carbon content of glycerol and biodiesel is determined and used to allocate the total carbon emissions of the production process to each product. Such a carbon allocation method is useful when analyzing the carbon footprint of bio-based products, such as soybean, where the carbon sequestration and storage of soybean are taken into account based on the quantity of biomass carbon stored in the products. Since not all products considered in the process are used directly to generate energy, the energy allocation was not performed, and carbon allocation is considered to be more appropriate.
(1) CO2 generated during H2 production is captured and sold for carbon credits.
(2) Direct land use refers to the physical occupation or transformation of land for the cultivation of soybean. Indirect land use takes into account the possible substitution of alternative land uses, including agricultural crops or natural ecosystems, as a result of the growing demand for H2 production from glycerol. These indirect uses were not employed here since the connection between land use and deforestation is not clearly explained and there is a lack of agreement on how to establish this connection.71 Therefore, land use calculations are not included.
(3) All processes considered (Fig. 4) are assumed to be located in Wisconsin, and it is assumed that the biodiesel and H2 facilities are located nearby. Therefore, only the transportation of soybean to the oil processing plant is considered. The study assumes that the soybeans are transported to the oil processing plant via diesel-fueled trucks.
(4) Seed yield is assumed to be 2889.6 kg (ha year)−1 and oil yield is assumed to be 547.4 kg (ha year)−1.75
(5) GHG emissions of soybean cultivation were based on data from conventional soybean production in the USA.75
(6) GHG emissions of biodiesel production were based on data from Dufour and Iribarren.74
(7) Two GHG emission allocation methods were considered, one based on mass and one on carbon content, as mentioned above.
(8) The CaO comes from geological formations, and emissions associated with its transportation are considered in the calculation.
(9) For the process of APR coupled with Pressure Swing Adsorption (PSA), we considered that the CO2 can be injected underground or sold to third parties.
An overview of the mass relationships between various feedstocks and products involved in the process is outlined in Table 2.
This study includes both primary and secondary data. Primary data collected from experiments and process simulations are used to evaluate the GHG emissions of H2 production using APR from glycerol. The OpenLCA software has been utilized. The data on materials were collected from the database Agribalyse. The ReCiPe 2016 midpoint (H) LCIA method converts the resource consumptions and emissions of the life cycle inventory data into global warming potential (GWP). Secondary data on other producing stages were collected from literature.71,74,75
3. Results and discussion
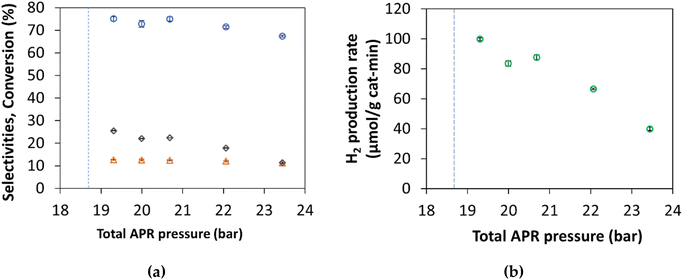
3.1. Effect of total pressure system on APR of glycerol
A Pt/γ-Al2O3 catalyst was used to study glycerol aqueous phase reforming (APR) as a function of temperature and pressure. Table 3 and Fig. 5 show that the H2 selectivity and production rate decrease as the total system pressure increases. The CO2 and C2–C3 alkanes selectivity increases with pressure. The catalyst was run for 480 h on stream with low deactivation. The H2 selectivity (75% at 209 °C) did not change during 523 h after different cycles between temperatures (209 °C–269 °C) and pressures (19.3 bar–57.9 bar); see Fig. S1 in ESI.†| P (bar) | Gas phase composition (mol%) | Selectivity (%) | Glycerol conversion to the gas phase (%) | H2 production rate | H2 TOF (min−1) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| H2 | CO2 | CH4 | C2H6 | C3H8 | H2 | Alkanes | (μmol μmolPt−1 min−1) | (μmol gcat−1 min−1) | |||
| 19.3 | 64.3 | 32.0 | 2.8 | 0.7 | 0.1 | 75.1 | 12.7 | 25.5 | 0.65 | 99.8 | 1.8 |
| 20.0 | 63.6 | 32.7 | 2.7 | 0.8 | 0.2 | 72.8 | 12.6 | 22.0 | 0.54 | 83.6 | 1.5 |
| 20.7 | 64.3 | 32.2 | 2.6 | 0.7 | 0.2 | 74.9 | 12.4 | 22.4 | 0.57 | 87.7 | 1.6 |
| 22.1 | 62.2 | 34.3 | 2.5 | 0.8 | 0.2 | 68.5 | 12.2 | 17.3 | 0.40 | 62.0 | 1.1 |
| 23.4 | 61.9 | 34.9 | 2.1 | 0.8 | 0.2 | 67.4 | 11.3 | 11.3 | 0.26 | 39.8 | 0.7 |
The effect of pressure for APR has been previously described by Davda et al.19 and Shabaker et al.17 with similar observations. Hydrogen inhibits the APR reaction. As shown in Table 3, the mol percent of H2 and methane decreases when the total pressure increases, while CO2, ethane, and propane mol percent increase. This behavior indicates that H2 is consumed in the hydrogenation of C2 and C3 intermediates.
As shown in Fig. 5 H2 selectivity and H2 production rate are highest when the system pressure is close to the bubble point of water. The remainder of the APR studies for this paper were fixed at 0.4–0.6 bar above the bubble point of water.
3.2. Aqueous phase reforming of glycerol with bimetallic NiPt catalysts
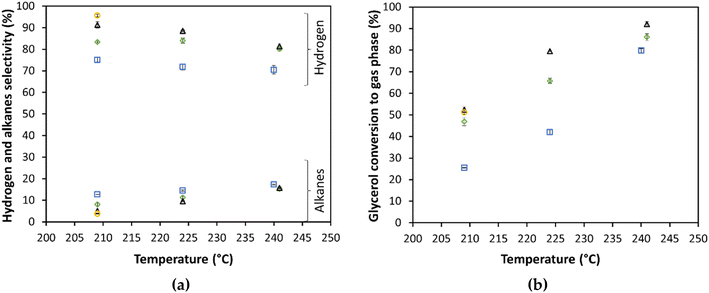
Bimetallic NiPt/alumina catalysts were prepared and studied for the APR of glycerol. Table 4 shows the APR of these catalysts at three different temperatures (209, 224, and 241 °C). As illustrated in Fig. 6, the H2 selectivity increases in the order Ni8Pt1 > Ni1Pt1 > Pt when the catalysts are reduced in situ at 260 °C. There is an additional increase in H2 selectivity when the Ni8Pt1-450 catalyst is reduced at 450 °C. These effects are more visible when APR is carried out at low temperature (209 °C), achieving a H2 selectivity of 95.7%, and a 3.7% of alkanes selectivity for Ni8Pt1-450. The Ni8Pt1-450 catalyst was selected for the APR of glycerol coupled with CO2 sequestration, detailed in the next section.| Catalyst | CO chemisorption (μmol g−1) | WHSV (h−1) | T (°C) | P (bar) | Gas phase composition (mol%) | Selectivity (%) | Glycerol conversion to gas phase (%) | H2 production rate | TOF (min−1) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| H2 | CO2 | CH4 | C2H6 | C3H8 | H2 | Alkanes | (μmol H2 gcat−1 min−1) | (μmol H2 μmolPt−1 min−1) | |||||||
| Pt-260 | 57.4 | 0.37 | 209 | 19.3 | 64.3 | 32.0 | 2.8 | 0.7 | 0.1 | 75.1 | 12.7 | 25.5 | 99.7 | 0.6 | 1.7 |
| 224 | 26.9 | 63.4 | 32.3 | 3.3 | 0.9 | 0.2 | 71.8 | 14.5 | 42.0 | 157.2 | 1.0 | 2.7 | |||
| 240 | 34.5 | 62.4 | 32.3 | 4.0 | 1.1 | 0.2 | 68.5 | 17.4 | 74.5 | 267.3 | 1.7 | 5.4 | |||
| Ni1Pt1-260 | 39.1 | 0.37 | 209 | 19.3 | 66.2 | 31.3 | 2.3 | 0.2 | 0.0 | 83.3 | 8.1 | 46.9 | 203.3 | 1.3 | 5.2 |
| 224 | 25.5 | 66.5 | 30.1 | 3.1 | 0.3 | 0.0 | 84.0 | 11.3 | 65.7 | 287.1 | 1.9 | 7.3 | |||
| 241 | 34.5 | 65.2 | 29.8 | 4.4 | 0.5 | 0.1 | 78.8 | 16.0 | 84.1 | 345.3 | 2.2 | 8.8 | |||
| Ni8Pt1-260 | 38.7 | 0.36 | 209 | 19.3 | 68.1 | 30.4 | 1.4 | 0.1 | 0.0 | 91.3 | 5.0 | 52.4 | 241.6 | 1.6 | 6.2 |
| 224 | 25.5 | 67.5 | 29.6 | 2.7 | 0.2 | 0.0 | 88.5 | 9.5 | 79.5 | 355.3 | 2.3 | 9.1 | |||
| 241 | 34.5 | 65.8 | 29.2 | 4.7 | 0.3 | 0.0 | 81.4 | 15.8 | 92.0 | 378.4 | 2.5 | 9.8 | |||
| Ni8Pt1-450 | 37.2 | 0.36 | 209 | 19.3 | 69.1 | 29.8 | 1.0 | 0.1 | 0.0 | 95.7 | 3.7 | 51.2 | 251.5 | 1.6 | 6.8 |
The gas phase H2 composition increased from 64.3 mol% at 209 °C with Pt/Al2O3 (Pt-260), to 69.1 mol% using the Ni8Pt1-450 bimetallic catalyst as shown in Table 4. The H2 to CO2 ratio was close to the stoichiometric ratio with only 1.1 mol% of alkanes. The CO concentrations were lower than 10 ppm under all the reaction conditions reported in Table 4.
Previous studies reported18,37,76 that the use of Ni promotes the water–gas shift reaction, improves the dehydrogenation of adsorbed glycerol, and decreases the strength of H2 and CO adsorption, preventing the blockage of active sites. Additional studies are needed to understand the effect of Ni addition and the interaction between Ni–Pt, and the support. Tanksale et al.77 found that Ni–Pt forms a metallic alloy over the support, resulting in a synergetic effect and enhanced activity on the APR of glycerol. The improved hydrogen selectivity of the Ni8Pt1-450 catalyst compared to Ni8Pt1-260 can be explained by the complete reduction of nickel oxide at 450 °C, affecting the crystal form and size.78 Rahman et al.76 reported that Ni6Pt, Ni8Pt, and Ni12Pt supported over Ce-doped alumina support, have similar hydrogen selectivities, with a higher selectivity over Ni6Pt with 83% and 86% yield on the APR of 1% of glycerol at 240 °C. This behavior is comparable to the selectivity we reported here for Ni8Pt1-260 at the same APR temperature (240 °C) but lower than our highest selectivity (95.7%) obtained for Ni8Pt1-450 in the APR at 209 °C. In addition, they reported 8 to 10 mol% of methane in the gas product. In this work, we reported (Table 4) that methane mol% is lower than 4.7% for Ni8Pt1-260 and as low as 1 mol% for Ni8Pt1-450.
Each catalyst shown in Table 4 was studied for APR of 10 wt% glycerol for at least 140 h on stream. The catalyst Pt-260 show no deactivation after 523 h on stream (Fig. S1 in ESI†). Ni1Pt1-260 and Ni8Pt1-260 show a 10% reduction in glycerol conversion and 5% reduction in H2 selectivity after 189 h and 140 h on stream, respectively (ESI Fig. S2 and S3†). Ni8Pt1-450 shows slow deactivation when the APR was carried out at 209 °C and 19.3 for 401 h on stream. This catalyst had a 15% reduction in glycerol conversion and 4% reduction of H2 selectivity during the first 40 h of stabilization, as shown in Fig. S4 in ESI.† After the first 40 h, the H2 selectivity was stable at 91%, as shown in Fig. 7. The gas composition was almost constant at 68% H2, 30.8% CO2, 1% methane, and no CO (<10 ppm).
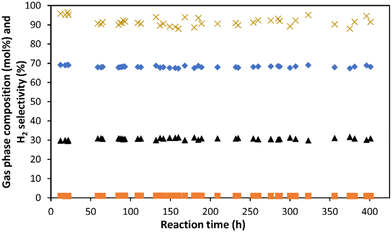 | ||
| Fig. 7 Gas phase composition (■ Methane, ▲ CO2, ✦ Hydrogen) and hydrogen selectivity (X) for the Ni8Pt1-450 catalyst, in the APR of 10 wt% of glycerol at 0.36 h−1 WHSV and 209 °C. | ||
3.3. CO2 sequestration
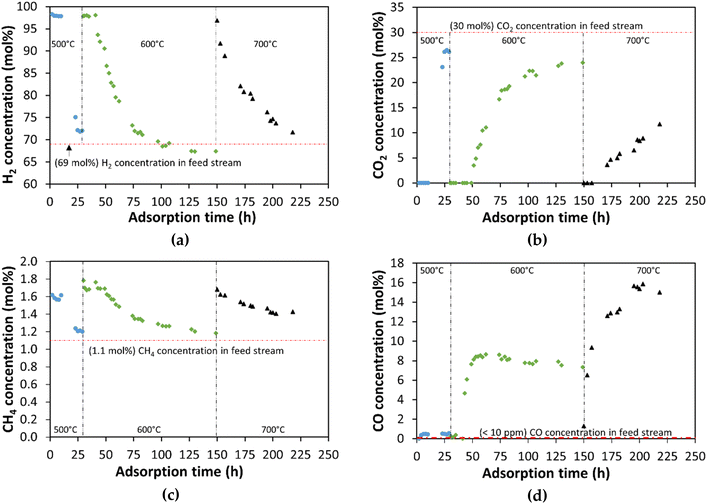
The APR of 10 wt% of glycerol over the Ni8Pt1-450 catalyst at 209 °C and 0.36 h−1 WHSV, was coupled with the CO2 sequestration over CaO to produce a high-pressure H2 stream with high purity. Sequestration experiments were conducted: (a) at low CO2 partial pressure (0.75 bar) in which nitrogen was used as a sweep gas for APR at adsorption temperatures from room temperature to 700 °C with different CaO loadings; and (b) at a higher CO2 partial pressure (5.3 bar) in which no nitrogen was used as sweep gas.The APR gas product contains less than 10 ppm of CO. From room temperature to 400 °C the CO concentration after the CaO bed was less than 150 ppm. At 500 °C the CO concentration increased to a maximum of 650 ppm at 29 h of adsorption time. As shown in Fig. 8d, the CO concentration increases rapidly during the first few hours of CO2 adsorption and tends to be constant (0.5 mol% at 500 °C, 8.4 mol% at 600 °C, and 15.5 mol% at 700 °C). CO emissions during the CO2 capture over CaO are caused due to the presence of H2. Hydrogen promotes the regeneration of CaO by the decomposition of CaCO3 producing CO and H2O.80
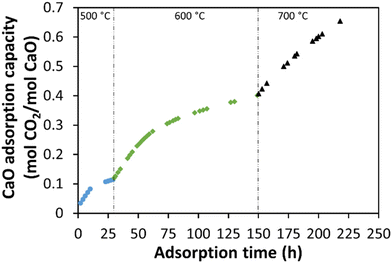
The CaO adsorption capacity was determined after each isothermal experiment, as shown in Fig. 9. The CaO adsorption capacity was 0.11 mol CO2/mol CaO at 500 °C, increasing to 0.40 mol CO2/mol CaO at 600 °C, and finally increasing to 0.65 mol CO2/mol CaO at 700 °C. These CaO adsorption capacities are lower than the values previously reported by Hlaing et al.66 of 0.87 mol CO2/mol CaO at 700 °C. However, the required time to obtain this adsorption capacity is around 225 h under these low CO2 partial pressure (0.75 bar) and total system pressure (19.3 bar). The limitation to achieve a higher CO2 adsorption capacity at this temperature (700 °C) CO is produced by the reverse water gas shift reaction. Nevertheless, the use of CaO as an adsorbent model indicates that calcium-bearing minerals could be used to capture CO2, as indicated by Gadikota and Park,68 by using an in situ or ex situ carbon mineralization approach.
The CO concentration at the outlet of the CO2 adsorber at 600 °C and an inlet CO2 partial pressure of 0.75 bar was 3.5 mol% and 4.5 mol% respectively for 4 g and 15 g of CaO packed in the adsorber (see Fig. S6 in ESI† for the whole data collected).
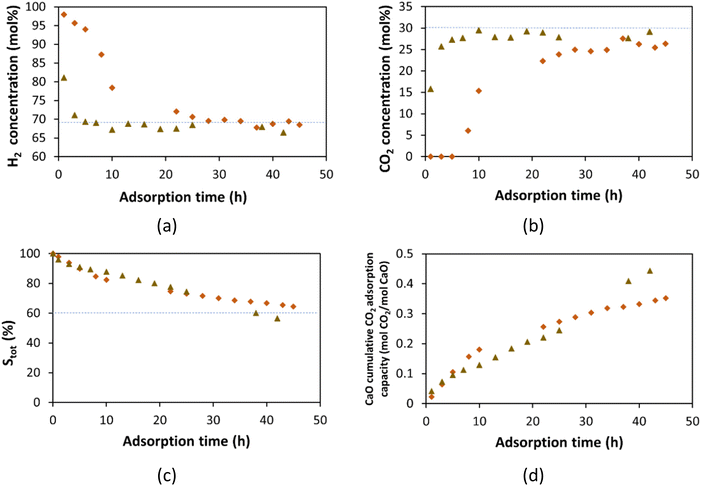
Fig. 12a and b show complete CO2 capture for the first 10 h of operation, producing a high-pressure (19.3 bar) gas stream with 95.5 mol% of H2 and 4 mol% of methane. There is an apparent CaO saturation after 50 h. The CO2 mol% in the outlet stream was around 10 mol% lower than the inlet stream at this saturation stage. Once the CaO is saturated, the outlet stream should have the same composition as the inlet unless the CO2 is reacting. Grasa and Abanades59 reported that CaO derived from natural limestones shows a residual constant conversion of about 7 to 8% after hundreds of cycles of carbonation/calcination, and seems to be independent of the CaO degradation by calcination temperature. The residual constant conversion is likely related to a catalytic conversion of CO2 in the CaO. Additionally, Fig. 12c shows that 50% of the initial CaO has reacted at 55 h of adsorption, for a total adsorption capacity of 0.45 mol of CO2/mol CaO (Fig. 12d). The total CaO active sites (STot) and the CaO sequestration capacity were calculated based on the amount of CO2 adsorbed. To understand this phenomenon, Fig. 13 shows the flow rate of gas phase components for both the inlet and outlet of the CaO bed.
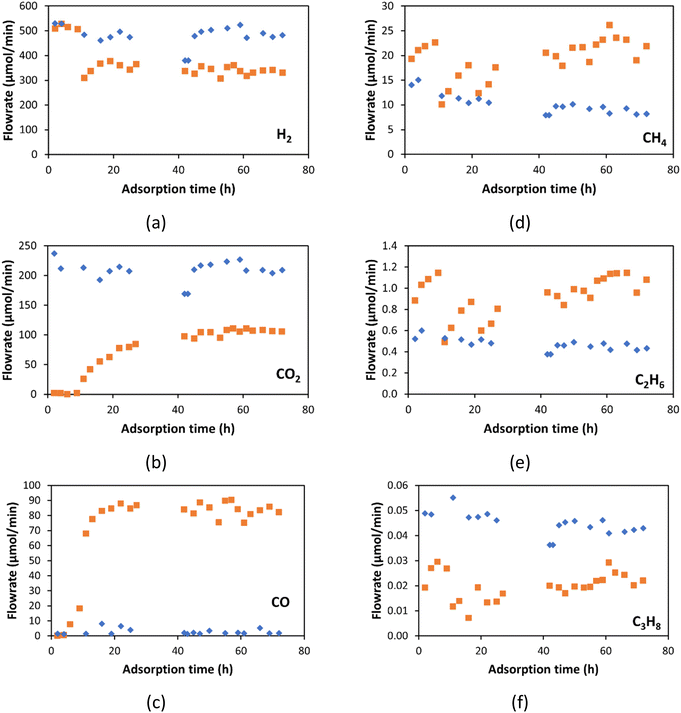
As shown in Fig. 13a, the H2 flow rate is the same at the inlet and the outlet for the first 9 h, where there is complete CO2 capture (Fig. 13b). After 9 h of operation, the H2 flow rate decreases, indicating that H2 is reacting in the CaO bed. Similarly, the propane (Fig. 13f) flow rate is lower in the outlet stream than in the inlet. The methane and ethane (Fig. 13d and e) flow rates are higher in the outlet stream than in the inlet. These changes in alkane flow rates indicate that propane is being cracked to methane and ethane. These findings indicate that the CaO acts as a catalyst bed at this temperature (600 °C) and pressure (19.3 bar). Table 5 shows the total mol of each component fed to the CaO bed for the 72 h of operation, and the total mol of each component obtained at the outlet stream. H2, CO2, and propane were consumed or reacted on the CaO bed, with reductions of 21.8, 60.4, and 54.9% respectively. Meanwhile, methane and ethane increased by around 100%. These changes by the catalytic effect of the CaO in the bed. The CaO catalytic pathways could include (a) CO and water emissions by the CaCO3 decomposition;81 (b) propane cracking into C1–C2 alkanes; and (c) CO2 hydrogenation to produce the same C1–C2 alkanes.
| Inlet (mol) | Outlet (mol) | Change (%) | |
|---|---|---|---|
| H2 | 2.01 | 1.57 | −21.8 |
| CO2 | 0.87 | 0.34 | −60.4 |
| CO | 0.0 | 0.32 | — |
| Methane | 0.04 | 0.08 | 100.9 |
| Ethane | 2.0 × 10−3 | 4.0 × 10−3 | 103.9 |
| Propane | 1.9 × 10−4 | 8.5 × 10−5 | −54.9 |
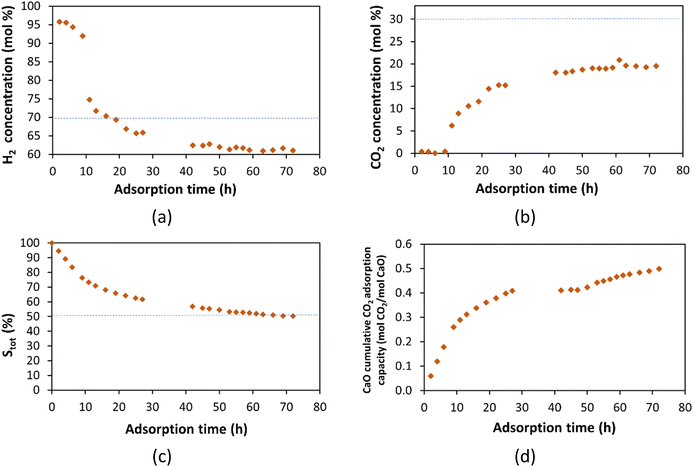
At this CO2 adsorption conditions (600 °C, 19.3 bar of total pressure, and 5.3 bar of CO2 partial pressure) the CO molar concentration increased from around 0.15 mol% (≈1500 ppm) in the adsorber inlet to around 16 mol% after 10 h of adsorption as can be seen in Fig. S6 of ESI.† The CO production in the CaO bed is affected by the CO2 partial pressure in the inlet stream in addition to the temperature as discussed previously in section 3.3.1. At 600 °C and 5.3 bar of CO2 partial pressure, the CO concentration is two times higher than the concentration at the same temperature and 0.75 bar of CO2 partial pressure, that was 8.4 mol%.
Fig. 15 shows the CO2 sequestration constant (k) calculated when the capture was carried out at 600 °C. The value of k was determined for 4 g, and 15 g of CaO at an inlet CO2 partial pressure of 0.75 bar; and for a higher CO2 inlet partial pressure of 5.3 bar with 25 g of CaO. As shown, the k-values have similar values independent of the amount of CaO packed or the CO2 inlet partial pressure. The k-value was constant at around 0.000165 1 atm per min from 10 to 57 min of adsorption. For the first 10 h of capture, there is a decrease from 0.000897 to 0.000189 1 atm per min, similar to the k-values determined at different temperatures (Fig. 14).
Using a k-value of 0.000165 1 atm per min we calculated the number of CaO active sites at different CO2 partial pressures (Fig. 16). 0.75 bar and 5.3 bar of inlet CO2 partial pressures was considered for the calculation at 600 °C.
CaO has been widely proposed for CO2 capture.57–65 The CaO sequestration capacity depends on the temperature. Nevertheless, the presence of H2 causes the reduction of calcium carbonate forming CO and water. As previously reported by Sun et al.80 this effect could be used to produce syngas from flue gases operating the CO2 adsorber at 600 or 700 °C. But, to keep low CO levels in the H2 stream while reducing the CO2 concentrations, it is recommendable to operate the CaO bed at low temperature such as 500 °C or lower, depending on the APR composition and the CO2 partial pressure. Pressure Swing Adsorption (PSA)69 was also considered for the LCA and TEA analysis presented below.
3.4. Life cycle assessment (LCA), and techno-economical analysis (TEA)
Fig. 17 presents the results of the LCA analysis in terms of GHG emissions using CaO adsorption (Fig. 17a and c), and PSA (Fig. 17b and d) for two scenarios: Scenario A using Wisconsin grid electricity, and Scenario B using 100% renewable electricity. Wisconsin's electricity generation comes from 44.2% natural gas, 28.8% coal, 17.3% nuclear, and 9.6% renewables.82 The results are presented considering two impact allocation methods: (i) mass allocation and (ii) carbon content allocation. This figure also shows the GHG contributions of each processing stage in different colors. Based on this analysis, we can observe that all the scenarios result in net negative GHG emissions, except for Scenario A considering allocation by carbon content when CaO is used for CO2 sequestration at 600 °C. However, CaO adsorption for carbon capture is not currently used industrially. If instead, PSA is used for the hydrogen purification, all the scenarios are carbon negative and with lower GHG emissions compared to the use of CaO at 600 °C. Replacing the electricity grid with renewable energy sources can decrease the GHG emissions of each production stage. Moreover, the allocation method plays a critical role in the resulting GHG emissions. Assumptions taken during the environmental impact analysis, including the allocation method, can significantly alter the analysis results.Fig. 18 presents a comparison of the APR of glycerol coupled with CCUS considering the mass allocation method compared to other H2 production technologies. All APR processes have net negative GHG emissions. The high biogenic uptake leads to negative CO2 emissions for APR processes that use glycerol derived from soybean, thereby contributing to the goal of achieving net-zero carbon emissions. A comparison of all scenarios and both impact allocation methods can be found in Fig. S7 in the ESI.† These results are in accordance with the GHG emissions (−18.5 kg CO2 eq./kg H2) reported previously4 for biomass gasification with CCUS. If Renewable energy is used instead of grid electricity the GHG emissions could be even lower than −50 kg CO2 eq./kg H2 under the mass allocation method.
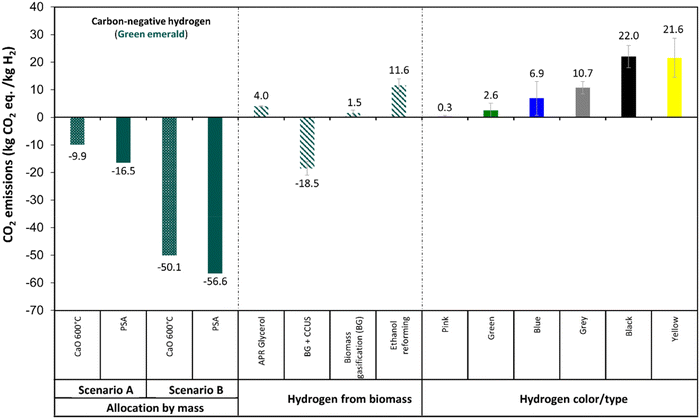 | ||
| Fig. 18 Comparison of GHG emissions of H2 production technology presented in this work with the technologies presented in Table 1. Scenario A: using Wisconsin grid electricity. Scenario B: using 100% renewable electricity. | ||
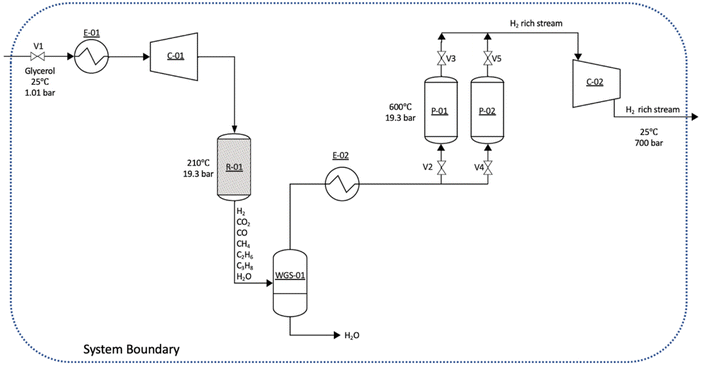
A preliminary techno-economic analysis83 was conducted to determine the H2 production costs, including raw material and utility costs. Fig. 19 shows the conceptual design of the industrial-scale APR process coupled with CO2 sequestration using CaO, which was modeled using the Aspen Plus program. The use of CaO for CO2 sequestration will need an additional step to obtain a high-purity H2, since the stream obtained still contains small amounts of alkanes and CO (1.8 to 4.4 mol% depending on the CO2 partial pressure). A small PSA unit could be coupled after the CO2 sequestration unit using CaO. The process includes four pieces of equipment: a reactor, a flash separator, a fluidized bed reactor, and a compressor. The conceptual design for the industrial-scale APR process coupled with PSA for H2 purification can be found in Fig. S8 in ESI,† which includes a reactor, a flash separator, a PSA unit, and a compressor. To provide a first estimation of the capital cost, Aspen Capital Cost Estimator was used, which considers various factors such as the size and complexity of each piece of equipment, required materials, and other expenses. These inputs generate calculations of material and energy balances, and capital costs. Based on the calculations, an APR plant producing about 823![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 kg of H2 per year would require a capital cost of USD 4.3 million and would consume 7112 tons per year of crude glycerol.
000 kg of H2 per year would require a capital cost of USD 4.3 million and would consume 7112 tons per year of crude glycerol.
Table 6 presents the material and energy costs associated with the operation of the plant based on two scenarios, along with the resulting estimated production cost of the H2. The transportation cost within Wisconsin is neglected. Results are compared to the target cost of $1 (USD) set by DOE to estimate the desired raw material cost. Based on rough estimates, the price of the 10 wt% glycerol solution needs to be less than 32.4 USD per ton for this technology to produce H2 at 1 USD per kg. Therefore, glycerol produced from waste oil processing facilities would be the most environmentally and economically favorable.
| Using PSA | Using CaO | The goal of 1 USD per kg H2 | |||
|---|---|---|---|---|---|
| Scenario A | Scenario B | Scenario A | Scenario B | ||
| Price of glycerol solution (USD per ton) | 198![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 84 84 |
198![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 84 84 |
198![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 84 84 |
198![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 84 84 |
32.4 |
| Raw material cost: glycerol (USD per kg H2) | 1.71 | 1.71 | 1.71 | 1.71 | 0.28 |
| Heat cost (USD per kg H2) | 0.13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85 85 |
0.13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85 85 |
0.22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85 85 |
0.22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85 85 |
0.22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85 85 |
| Electricity cost (USD per kg H2) | 0.50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 86 86 |
0.72![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 87 87 |
0.50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 86 86 |
0.72![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 87 87 |
0.50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 86 86 |
| H2 cost (USD per kg H2) | 2.3 | 2.6 | 2.4 | 2.7 | 1 |
The current study considers hydrogen production from soybean-based glycerol. However, other common feedstocks are corn stover or lignin-rich stream (LRS) (for example, from cellulosic ethanol production).88,89 The cost of hydrogen obtained is similar to a study suggesting a price of 1.5 € per kg H2 produced considering corn stover as feedstock and a hydrothermal liquefaction (HTL)-APR plant.88 However, when comparing multiple studies, LCA and TEA values can vary extremely depending on the system considerations, scope, and boundaries.14,88,90,91 Most studies assessing H2 production using APR technology don't perform LCA and TEA together. However, a recent study92 demonstrated a 60% reduction (from 20.9 to 8.2 g CO2 eq. per MJ) in GHG emissions (mass allocation method) on sustainable aviation fuel production, and a 17% reduction (2.20 to 1.84 $ per kg) in the minimum fuel selling price of SAF when hydrogen was produced in situ through APR of glycerol. This demonstrates that hydrogen produced through APR technology can help to reduce GHG emissions and costs when coupled with other processes.
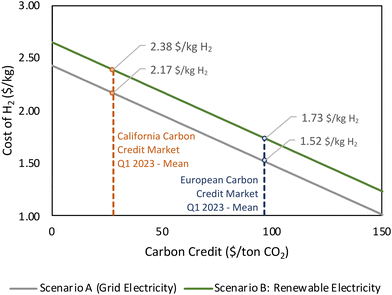
The cost of H2 production can be reduced by considering carbon credits. It should be noted that the market price of CO2 fluctuates over time. As shown in Fig. 20 for the H2 cost estimated for APR CO2 separation using PSA, if CO2 is sold back at the California carbon credit market price, the production cost of H2 is 2.38 USD per kg H2 with grid electricity and 2.17 USD per kg H2 with renewable electricity. The production cost is even lower if it is sold at the European market price; the production cost of H2 is 1.73 USD per kg H2 with grid electricity and 1.52 USD per kg H2 with renewable electricity.
This study was carried out considering the glycerol produced as a byproduct in the state of Wisconsin (USA), which was around 7112 tons and represents only around 1.5% of the total production in the United States in 2023. According to the U.S. Energy Information Administration93 the total biodiesel production in the United States in 2023 was around 6500 million litres, which generates 566![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tons of glycerol. This amount of glycerol can lead to the production of around 65
000 tons of glycerol. This amount of glycerol can lead to the production of around 65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ton of green-emerald H2 in the U.S., representing 0.5% of the total Hydrogen demand in 2020 in the U.S. according to the data of McKinsey & Company.94
000 ton of green-emerald H2 in the U.S., representing 0.5% of the total Hydrogen demand in 2020 in the U.S. according to the data of McKinsey & Company.94
The estimated global demand for biodiesel in 2027 will be 52 billion litres95 which corresponds to 4.6 million tons of glycerol. This amount of glycerol can produce 530![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tons of H2, which represents 0.6% of the global hydrogen demand. In addition to glycerol, this technology could be applied to other more abundant biomass feedstocks such as ethanol, xylitol, sorbitol, xylose, cellulose, or the whole biomass. This availability is especially important to achieve the goal of net-zero carbon emissions, and to diversify the renewable hydrogen sources.
000 tons of H2, which represents 0.6% of the global hydrogen demand. In addition to glycerol, this technology could be applied to other more abundant biomass feedstocks such as ethanol, xylitol, sorbitol, xylose, cellulose, or the whole biomass. This availability is especially important to achieve the goal of net-zero carbon emissions, and to diversify the renewable hydrogen sources.
4. Conclusions
Carbon-negative H2, or green-emerald H2, can be produced by coupling APR technology with CO2 sequestration. APR of glycerol at 209 °C using a Ni8Pt1-450 catalyst with a system pressure 0.5 bar above the bubble point of water had a H2 selectivity of 95.7%. The carbon-negative H2 stream obtained after CO2 sequestration had 98.2 mol% H2 purity and 1.8 mol% methane. For higher CO2 partial pressure and no nitrogen as sweep gas, the carbon-negative H2 purity was 95.6 mol%, with 3.8 mol% methane and 0.2% ethane. CO concentrations of 400 and 800 ppm were detected respectively. Calcium oxide had an adsorption capacity of 0.35–0.4 mol CO2/mol CaO at 600 °C for low CO2 partial pressure. The CaO can catalyze the formation of CO by reverse water gas shift reaction at higher adsorption capacity and CO2 partial pressures.From the LCA analysis, the total GHG emissions depend on the key assumptions and allocation method applied. The analysis showed that APR coupled with CCUS could produce H2 with net negative GHG emissions, especially considering the use of renewable energy. Future research will investigate different shares of renewable energy and their effect on GHG emissions and economic impacts. The GHG emissions can be as low as −50.1 kg CO2/kg H2 using CaO at 600 °C, and as low as −56.6 kg CO2/kg H2 using PSA when renewable electricity is used according to the allocation by mass in both cases. Our carbon-negative H2 has a lower carbon footprint than other reported methods to produce H2.2–7,9,14 Therefore, the APR of biomass-derived feedstocks coupled with CCUS can help to achieve the US DOE target of net-zero carbon emissions. TEA analysis indicates that the crude glycerol price needs to be lower than 32.4 USD per ton to produce H2 at 1 USD per kg H2.
Author contributions
Leoncio Santiago-Martínez contributed conceptualization, methodology, data curation, formal analysis, investigation, validation, visualization, writing – original draft preparation, writing – review and editing; Mengting Li contributed methodology, investigation, data curation, visualization, and writing – original draft preparation; Paola Munoz-Briones contributed investigation, data curation, and writing – review and editing; Javiera Vergara-Zambrano contributed investigation, data curation, visualization, and writing – review and editing; Styliani Avraamidou contributed conceptualization, resources, data curation, formal analysis, supervision, validation, investigation, methodology, funding acquisition, and writing – review and editing; James A. Dumesic contributed conceptualization, resources, formal analysis, supervision, funding acquisition, validation, methodology, project administration, and writing – review and editing; and George W. Huber contributed conceptualization, resources, formal analysis, supervision, funding acquisition, validation, methodology, project administration, writing – original draft preparation, and writing – review and editing.Conflicts of interest
The authors declare no conflict of interest.Acknowledgements
The authors gratefully thank Professor Bu Wang for his valuable comments on CO2 sequestration. This material is based upon work supported by the DOE Great Lakes Bioenergy Research Center (Award Number DE-SC0018409) and by the DOE Center for Advanced Bioenergy and Bioproducts Innovation (Award Number DE-SC0018420), U.S. Department of Energy, Office of Science, Office of Biological and Environmental Research.References
- International Energy Agency (IEA) , The Future of Hydrogen. Seizing today's opportunities. Report prepared by the IEA for the G20, Japan.
- M. M. Mohideen, B. Subramanian, J. Sun, J. Ge, H. Guo, A. V. Radhamani, S. Ramakrishna and Y. Liu, Renewable Sustainable Energy Rev., 2023, 174, 113153 CrossRef CAS.
- A. Mehmeti, A. Angelis-Dimakis, G. Arampatzis, S. J. McPhail and S. Ulgiati, Environments, 2018, 5, 1–19 CrossRef.
- International Energy Agency (IEA), Towards Hydrogen Definitions Based on their Emissions Intensity, France, 2023 Search PubMed.
- A. Ajanovic, M. Sayer and R. Haas, Int. J. Hydrogen Energy, 2022, 47, 24136–24154 CrossRef CAS.
- J. M. M. Arcos and D. M. F. Santos, Gases, 2023, 3, 25–46 CrossRef CAS.
- P. Burmistrz, T. Chmielniak, L. Czepirski and M. Gazda-Grzywacz, J. Cleaner Prod., 2016, 139, 858–865 CrossRef CAS.
- GEI (Global Energy Infrastructure), Hydrogen – data telling a story, https://globalenergyinfrastructure.com/articles/2021/03-march/hydrogen-data-telling-a-story/, (accessed 10 March 2023).
- M. Yu, K. Wang and H. Vredenburg, Int. J. Hydrogen Energy, 2021, 46, 21261–21273 CrossRef CAS.
- A. Willige, Spectra, Miitsubishi Heavy Ind. Gr., 2022 Search PubMed.
- E. Hand, Science, 2023, 379, 630–636 CrossRef PubMed.
- CEMVITA, Gold Hydrogen, https://www.cemvita.com/gold-hydrogen, (accessed 3 August 2023).
- F. Osselin, C. Soulaine, C. Fauguerolles, E. C. Gaucher, B. Scaillet and M. Pichavant, Nat. Geosci., 2022, 15, 765–769 CrossRef CAS.
- Y. F. Khalil, Clean Energy, 2021, 5, 387–402 CrossRef.
- S. McQueen, J. Stanford, S. Satyapal, E. Miller, N. Stetson, D. Papageorgopoulos, N. Rustagi, V. Arjona, J. Adams, K. Randolph, D. Peterson, L. Hill, M. Koleva, T. Reinhardt, E. Frye, R. Schrecengost, A. Kokkinos, J. Litynski, R. Conrad, G. Soloveichik, D. Tew, S. Litzelman, J. Vetrano, R. Onuschak, A. Hahn, E. Hsieh and R. Costa, Department of Energy Hydrogen Program Plan 2020, DOI:10.2172/1721803.
- C. D. Scown, Trends Biotechnol., 2022, 40, 1415–1424 CrossRef CAS PubMed.
- J. W. Shabaker, R. R. Davda, G. W. Huber, R. D. Cortright and J. A. Dumesic, J. Catal., 2003, 215, 344–352 CrossRef CAS.
- G. W. Huber, J. W. Shabaker, S. T. Evans and J. A. Dumesic, Appl. Catal., B, 2006, 62, 226–235 CrossRef CAS.
- R. R. Davda and J. A. Dumesic, Angew. Chem., Int. Ed., 2003, 42, 4068–4071 CrossRef CAS PubMed.
- R. D. Cortright, R. R. Davda and J. A. Dumesic, Nature, 2002, 418, 964–967 CrossRef CAS PubMed.
- R. R. Davda, J. W. Shabaker, G. W. Huber, R. D. Cortright and J. A. Dumesic, Appl. Catal., B, 2003, 43, 13–26 CrossRef CAS.
- J. W. Shabaker, G. W. Huber, R. R. Davda, R. D. Cortright and J. A. Dumesic, Catal. Lett., 2003, 88, 226–235 CrossRef.
- M. S. Masnadi, J. R. Grace, X. T. Bi, N. Ellis, C. J. Lim and J. W. Butler, Energy, 2015, 83, 326–336 CrossRef CAS.
- L. Han, Q. Wang, Z. Luo, N. Rong and G. Deng, Appl. Energy, 2013, 109, 36–43 CrossRef CAS.
- B. Li, H. Yang, L. Wei, J. Shao, X. Wang and H. Chen, Int. J. Hydrogen Energy, 2017, 42, 5840–5848 CrossRef CAS.
- C. Xu, S. Chen, A. Soomro, Z. Sun and W. Xiang, J. Energy Inst., 2018, 91, 805–816 CrossRef CAS.
- M. H. Doranehgard, H. Samadyar, M. Mesbah, P. Haratipour and S. Samiezade, Fuel, 2017, 202, 29–35 CrossRef CAS.
- M. B. Valenzuela, C. W. Jones and P. K. Agrawal, Energy and Fuels, 2006, 20, 1744–1752 CrossRef CAS.
- B. Meryemoglu, A. Hesenov, S. Irmak, O. M. Atanur and O. Erbatur, Int. J. Hydrogen Energy, 2010, 35, 12580–12587 CrossRef CAS.
- J. Zhang, W. Yan, Z. An, H. Song and J. He, ACS Sustainable Chem. Eng., 2018, 6, 7313–7324 CrossRef CAS.
- G. Wen, Y. Xu, Q. Liu, C. Wang, H. Liu and Z. Tian, Catal. Lett., 2011, 141, 1851–1858 CrossRef CAS.
- G. Wen, Y. Xu, Z. Xu and Z. Tian, Catal. Commun., 2010, 11, 522–526 CrossRef CAS.
- T. Soták, M. Hronec, I. Vávra and E. Dobročka, Int. J. Hydrogen Energy, 2016, 41, 21936–21944 CrossRef.
- G. Pipitone, G. Zoppi, A. Frattini, S. Bocchini, R. Pirone and S. Bensaid, Catal. Today, 2020, 345, 267–279 CrossRef CAS.
- C. H. Lee, M. K. Kim, S. Kim, S. W. Choi, H. W. Kim, J. Jae, H. Sohn, S. H. Choi, S. P. Yoon, K. B. Lee and H. C. Ham, Int. J. Energy Res., 2022, 46, 14478–14490 CrossRef CAS.
- Y. Kim, M. Kim, H. Jeong, Y. Kim, S. H. Choi, H. C. Ham, S. W. Lee, J. Y. Kim, K. H. Song, C. W. Yoon, Y. S. Jo and H. Sohn, Int. J. Hydrogen Energy, 2020, 45, 13848–13861 CrossRef CAS.
- M. El Doukkali, A. Iriondo, J. F. Cambra, L. Jalowiecki-Duhamel, A. S. Mamede, F. Dumeignil and P. L. Arias, J. Mol. Catal. A: Chem., 2013, 368–369, 125–136 CrossRef CAS.
- M. El Doukkali, A. Iriondo, N. Miletic, J. F. Cambra and P. L. Arias, Int. J. Hydrogen Energy, 2017, 42, 23617–23630 CrossRef CAS.
- L. Zhang, A. M. Karim, M. H. Engelhard, Z. Wei, D. L. King and Y. Wang, J. Catal., 2012, 287, 37–43 CrossRef CAS.
- A. Wawrzetz, B. Peng, A. Hrabar, A. Jentys, A. A. Lemonidou and J. A. Lercher, J. Catal., 2010, 269, 411–420 CrossRef CAS.
- Y. Guo, X. Liu, M. U. Azmat, W. Xu, J. Ren, Y. Wang and G. Lu, Int. J. Hydrogen Energy, 2012, 37, 227–234 CrossRef CAS.
- A. Ciftci, B. Peng, A. Jentys, J. A. Lercher and E. J. M. Hensen, Appl. Catal., A, 2012, 431–432, 113–119 CrossRef CAS.
- M. M. Rahman, T. L. Church, A. I. Minett and A. T. Harris, ChemSusChem, 2013, 6, 1006–1013 CrossRef CAS PubMed.
- A. J. Reynoso, U. Iriarte-velasco, M. A. Guti and J. L. Ayastuy, Catal. Today, 2021, 367, 278–289 CrossRef CAS.
- D. L. King, L. Zhang, G. Xia, A. M. Karim, D. J. Heldebrant, X. Wang, T. Peterson and Y. Wang, Appl. Catal., B, 2010, 99, 206–213 CrossRef CAS.
- N. Luo, X. Fu, F. Cao, T. Xiao and P. P. Edwards, Fuel, 2008, 87, 3483–3489 CrossRef CAS.
- A. Ciftci, D. A. J. M. Ligthart and E. J. M. Hensen, Green Chem., 2014, 16, 853–863 RSC.
- T.-W. Kim, H.-D. Kim, K.-E. Jeong, H.-J. Chae, S.-Y. Jeong, C.-H. Lee and C.-U. Kim, Green Chem., 2011, 13, 1718–1728 RSC.
- L.-J. Zhu, P.-J. Guo, X.-W. Chu, S.-R. Yan, M.-H. Qiao, K.-N. Fan, X.-X. Zhang and B.-N. Zong, Green Chem., 2008, 10, 1323–1330 RSC.
- F. Bastan, M. Kazemeini, A. Larimi and H. Maleki, Int. J. Hydrogen Energy, 2018, 43, 614–621 CrossRef CAS.
- A. S. Larimi, M. Kazemeini and F. Khorasheh, Appl. Catal., A, 2016, 523, 230–240 CrossRef CAS.
- A. S. Larimi, M. Kazemeini and F. Khorasheh, Int. J. Hydrogen Energy, 2016, 41, 17390–17398 CrossRef CAS.
- G. Wen, Y. Xu, H. Ma, Z. Xu and Z. Tian, Int. J. Hydrogen Energy, 2008, 33, 6657–6666 CrossRef CAS.
- J. Remón, J. Ruiz, M. Oliva, L. García and J. Arauzo, Chem. Eng. J., 2016, 299, 431–448 CrossRef.
- L. A. Dosso, C. R. Vera and J. M. Grau, Int. J. Hydrogen Energy, 2017, 42, 18853–18864 CrossRef CAS.
- C. He, J. Zheng, K. Wang, H. Lin, J. Y. Wang and Y. Yang, Appl. Catal., B, 2015, 162, 401–411 CrossRef CAS.
- Y. A. Criado, B. Arias and J. C. Abanades, Ind. Eng. Chem. Res., 2018, 57, 12595–12599 CrossRef CAS.
- F. D. M. Daud, K. Vignesh, S. Sreekantan and A. R. Mohamed, New J. Chem., 2016, 40, 231–237 RSC.
- G. S. Grasa and J. C. Abanades, Ind. Eng. Chem. Res., 2006, 45, 8846–8851 CrossRef CAS.
- M. Rydén and P. Ramos, Fuel Process. Technol., 2012, 96, 27–36 CrossRef.
- Z. Skoufa, A. Antzara, E. Heracleous and A. A. Lemonidou, Energy Procedia, 2016, 86, 171–180 CrossRef CAS.
- H. Sun, J. Wang, X. Liu, B. Shen, C. M. A. Parlett, G. O. Adwek, E. John Anthony, P. T. Williams and C. Wu, J. Mater. Chem. A, 2019, 7, 9977–9987 RSC.
- S. Turrado, B. Arias, J. R. Fernández and J. C. Abanades, Ind. Eng. Chem. Res., 2018, 57, 13372–13380 CrossRef CAS.
- M. Liu, A. Hohenshil and G. Gadikota, Energy Fuels, 2021, 35, 8051–8068 CrossRef CAS.
- F. N. Ridha, V. Manovic, A. Macchi and E. J. Anthony, Appl. Energy, 2015, 140, 297–303 CrossRef CAS.
- N. N. Hlaing, S. Sreekantan, H. Hinode, W. Kurniawan, A. A. Thant, R. Othman, A. R. Mohamed and C. Salime, AIP Conf. Proc., 2016, 1733, 020023 CrossRef.
- T. Yin, S. Yin, A. Srivastava and G. Gadikota, Resour., Conserv. Recycl., 2022, 180, 106209 CrossRef CAS.
- G. Gadikota and A. A. Park, in Carbon Dioxide Utilisation: Closing the Carbon Cycle: First Edition, ed. P. Styring, A. A. Quadrelli and K. Armstrong, Elsevier, 2015, pp. 115–137 Search PubMed.
- C. A. Grande, in Hydrogen Science and Engineering: Materials, Processes, Systems and Technology, ed. D. Stolten and B. Emonts, Wiley-VCH, Germany, 2016, pp. 489–508 Search PubMed.
- I. Leinonen, Int. J. Life Cycle Assess., 2022, 27, 1038–1043 CrossRef CAS.
- J. H. Schmidt, J. Cleaner Prod., 2015, 87, 130–138 CrossRef.
- Omni Tech International, Life Cycle Impact of Soybean Production and Soy Industrial Products, 2010 Search PubMed.
- N. Jungbluth, M. Faist Emmenegger, F. Dinkel, C. Stettler, G. Doka, M. Chudacoff, A. Dauriat, E. Gnansounou, M. Spielmann, J. Sutter, N. Kljun, M. Keller and K. Schleiss, Life cycle inventories of bioenergy, 2007 Search PubMed.
- J. Dufour and D. Iribarren, Renewable Energy, 2012, 38, 155–162 CrossRef CAS.
- T. D. Alcock, D. E. Salt, P. Wilson and S. J. Ramsden, Sci. Total Environ., 2022, 829, 154539 CrossRef CAS PubMed.
- M. M. Rahman, T. L. Church, M. F. Variava, A. T. Harris and A. I. Minett, RSC Adv., 2014, 4, 18951–18960 RSC.
- A. Tanksale, C. H. Zhou, J. N. Beltramini and G. Q. Lu, J. Inclusion Phenom. Macrocyclic Chem., 2009, 65, 83–88 CrossRef CAS.
- A. Morales-Marín, J. L. Ayastuy, U. Iriarte-Velasco and M. A. Gutiérrez-Ortiz, Appl. Catal., B, 2019, 244, 931–945 CrossRef.
- D. Alvarez and J. C. Abanades, Ind. Eng. Chem. Res., 2005, 44, 5608–5615 CrossRef CAS.
- S. Sun, Z. Lv, Y. Qiao, C. Qin, S. Xu and C. Wu, Carbon Capture Sci. Technol., 2021, 1, 1–4 Search PubMed.
- S. Sun, Z. Lv, Y. Qiao, C. Qin, S. Xu and C. Wu, Carbon Capture Sci. Technol., 2021, 1, 100001 CrossRef CAS.
- U.S. Energy, Information Administration (EIA), Wisconsin State Energy Profile, https://www.eia.gov/state/print.php?sid=WI, (accessed 12 March 2024).
- C. D. Scown, N. R. Baral, M. Yang, N. Vora and T. Huntington, Curr. Opin. Biotechnol., 2021, 67, 58–64 CrossRef CAS PubMed.
- Selina Wamucii, US Glycerol Prices, https://www.selinawamucii.com/insights/prices/united-states-of-america/glycerol/#:~:text=In2022%2C the approximate price, in New York and Washington, (accessed 13 December 2022).
- Natural Gas2022 data – 1990-2021 historical – 2023 forecast – price – quote – chart, https://tradingeconomics.com/commodity/natural-gas, (accessed 13 December 2022).
- Electricity Local, Wisconsin Electricity Rates and Consumption, https://www.electricitylocal.com/states/wisconsin/, (accessed 13 December 2022).
- IRENA, Int. Renewable Energy Agency, 2012, 1, 44 Search PubMed.
- E. Tito, G. Zoppi, G. Pipitone, E. Miliotti, A. Di Fraia, A. M. Rizzo, R. Pirone, D. Chiaramonti and S. Bensaid, J. Environ. Chem. Eng., 2023, 11, 109076 CrossRef CAS.
- G. Zoppi, E. Tito, I. Bianco, G. Pipitone, R. Pirone and S. Bensaid, Renewable Energy, 2023, 206, 375–385 CrossRef CAS.
- D. A. Sladkovskiy, L. I. Godina, K. V. Semikin, E. V. Sladkovskaya, D. A. Smirnova and D. Y. Murzin, Chem. Eng. Res. Des., 2018, 134, 104–116 CrossRef CAS.
- M. Khodabandehloo, A. Larimi and F. Khorasheh, Energy Convers. Manage., 2020, 225, 113483 CrossRef CAS.
- G. Pipitone, G. Zoppi, R. Pirone and S. Bensaid, J. Cleaner Prod., 2023, 418, 138141 CrossRef CAS.
- U.S. Energy, Information Administration (EIA), Monthly Energy Review, https://www.eia.gov/totalenergy/data/monthly/pdf/mer.pdf, (accessed 9 April 2024).
- G. Chiara, H. Bernd, N. Jesse and W. Maurits, Global Energy Perspective 2023: Hydrogen Outlook, https://www.mckinsey.com/industries/oil-and-gas/our-insights/global-energy-perspective-2023-hydrogen-outlook#/, (accessed 9 April 2024).
- International Energy Agency (IEA), Renewables 2022, Analysis and forecast to 2027, France, 2022 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4gc01896f |
| This journal is © The Royal Society of Chemistry 2024 |