Towards a sustainable tomorrow: advancing green practices in organic chemistry
Sudripet
Sharma
a,
Fabrice
Gallou
 b and
Sachin
Handa
b and
Sachin
Handa
 *ac
*ac
a2320 S. Brook St University of Louisville, Louisville, KY 40292, USA
bNovartis Pharma AG, Basel 4056, Switzerland
c601 S College Ave, University of Missouri, Columbia, MO 65211, USA. E-mail: sachin.handa@missouri.edu
First published on 3rd May 2024
Abstract
The adoption of green chemistry principles has ushered in significant advancements in environmental safety and cost efficiency across various synthetic processes. One notable area of improvement lies in reducing the hazardous waste generated by using organic solvents in organic reactions. In contrast, the utilization of water as a solvent has emerged as a sustainable and environmentally friendly alternative. Micellar catalysis, driven by tailor-made surfactants, has played a pivotal role in enhancing water's efficacy as a solvent in organic synthesis. These designer surfactants boast unique structures that enhance the solubility of organic compounds in water and act as initiators or stabilizers for nanoparticle catalysts, facilitating efficient catalysis. Micelles function as nanoreactors, creating localized high concentrations of reactants that lead to unprecedented reaction rates and exceptional selectivity. This review underscores the plethora of sustainable protocols that have yielded outstanding results by leveraging aqueous micellar chemistry in pharmaceutical synthesis. Moreover, the review explores the integration of nanocatalysis using readily available first-row transition metals, with a particular emphasis on the role of surfactants in stabilizing the catalyst. The versatility of the proline-based surfactant PS-750-M as a ligand or capping agent, enabling ligand-free metal nanocatalysis, is also addressed. Lastly, the review addresses current challenges and future avenues in green chemistry, stressing the importance of ongoing research and innovation.
1. Introduction
Over the centuries, industrial growth has been deemed essential for modern civilization. Yet, alongside its beneficial effects, industrial advancement has had adverse consequences on the environment, encompassing soil degradation, deforestation, ozone depletion, climate change, and water and air pollution, leading to the loss of biodiversity.1–3 As we traverse the 21st century, we confront monumental challenges, notably global warming driven by a continual increase in CO2 levels, resulting in the melting of glaciers and a consequent rise in ocean levels at a rate of 3.42 mm per year.4,5 Furthermore, climate change, coupled with water and air pollution, significantly impacts human life quality.6,7 The irreversible human footprint on the environment underscores the imperative for “sustainable development”, entailing the more judicious use of natural resources than in the past. To tackle these challenges, green chemistry is spearheading a revolution in chemical processes and technologies towards a sustainable future. This entails the integration of non-toxic and environmentally friendly reactants and solvents, the reduction or elimination of toxic transition-metal catalysts, the enhancement of reaction efficiency through greener additives, and the provision of environmentally benign synthetic pathways with high atom economy.8–10The Brundtland Commission, established by the United Nations General Assembly in 1983, introduced the concept of sustainable development in their seminal report, “our common future”, published in 1987.11 Its goal is to maintain economic development while safeguarding the prospects of future generations to fulfill their needs.11 This laid the groundwork for the Rio Summit held in 1992 in Rio de Janeiro.12 Following this summit, the UN Commission on Sustainable Development was founded that same year.12,13 Notably, in 1985, a coalition of chemical industries collaborated to establish “the responsible care global charter”, a set of ethical guidelines to ensure the safe handling of chemicals throughout their life cycles.13–15 Additionally, the charter sought to underscore the chemical industry's role in promoting sustainable development and improving overall quality of life.14,15 Green chemistry gained prominence following the US Pollution Prevention Act of 1990.16 By 1991, the Environmental Protection Agency (EPA), in partnership with the US National Science Foundation (NSF), initiated research grants to encourage redesigning chemical processes for reduced environmental impact.17,18 The Gordon Conference on Green Chemistry in 1996 marked a milestone in advancing green chemistry, providing a platform for early-career chemists and scientists to exchange ideas.19,20 However, it wasn't until 1998 that clear guidelines emerged with the publication of the 12 principles of green chemistry in the book “Green chemistry: theory and practice” by P. T. Anastas and J. C. Warner.21–23 These principles revolutionized sustainable practices, offering a comprehensive framework for minimizing the environmental footprint of chemical processes. They emphasize waste prevention, atom economy, sustainable synthesis methodologies, the development of safer and biodegradable products, the use of green solvents and renewable raw materials, energy efficiency, catalytic reagents, biodegradability, real-time process analysis, and safer chemical processes (Fig. 1). Subsequently, the launch of the green chemistry journal by the Royal Society of Chemistry in 1999 and the establishment of the ACS Green Chemistry Institute (ACS GCI) by the American Chemical Society further bolstered global awareness through workshops, collaborations, conferences, and educational materials.24–32 Pharmaceutical industries also made strides in promoting Green Chemistry practices, with initiatives such as the ACS GCI's roundtable for pharmaceutical industries to accelerate the adoption of green and sustainable practices.13,33–38 These efforts have yielded positive outcomes, with numerous pharmaceutical companies embracing green chemistry principles in synthesizing important drug molecules.39,40
1.1. Impact of green practices on pharmaceutical industries
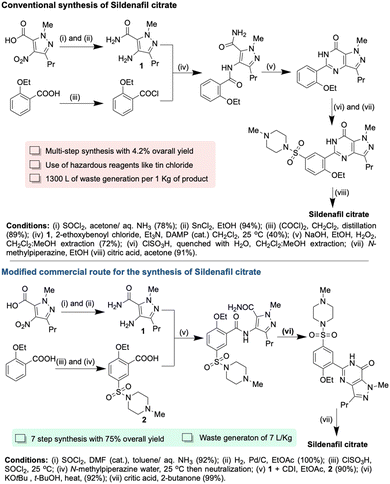
Green chemistry has significantly bolstered the pharmaceutical sector's environmental safety and cost efficiency.10,39–41 Recent years have witnessed a marked decrease in waste generation, a trend underscored by data from the Environmental Protection Agency (EPA), revealing a notable 27% reduction in chemical waste since 2011.42,43 This decline primarily stems from enhanced recycling of chemicals and organic solvents. Industries have also embraced various source reduction strategies, including adopting optimal operating practices and process modifications, resulting in a 36% decrease in waste and eliminating toxic reagents in favor of recyclable alternatives, leading to a 23% reduction in waste.43 Additionally, streamlining organic synthesis steps has contributed to waste reduction efforts. Such data vividly illustrates the undeniable advantages of green chemistry practices in curbing industrial waste.The transformative impact of green chemistry practices on waste reduction is exemplified in the synthesis of Pfizer's Sildenafil citrate, commonly known as Viagra, a medication for erectile dysfunction.44 Traditionally, its synthesis involved eleven steps, yielding a mere 4.2%, and necessitated using corrosive and toxic chemicals like tin chloride and thionyl chloride. Notably, the conventional process generated a substantial 1300 liters of waste per kilogram of product.45 In contrast, innovative green synthetic methods employed eco-friendly solvents such as butanol, EtOAc, and toluene, recycled throughout the process. The modified process dramatically increased the yield of the final three steps to 97%, resulting in a 75% overall yield while simultaneously slashing waste generation a mere 7 liters per kilogram of product44 (Scheme 1).
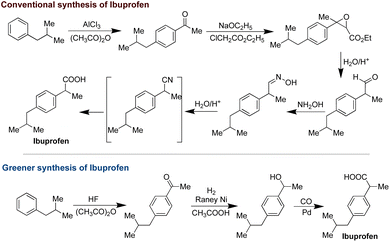
Another compelling illustration of the impact of green chemistry lies in the synthesis of ibuprofen, a widely used pain reliever. The conventional process, patented by the Boots company in 1960, comprised six stoichiometric steps, yielding a higher volume of waste and exhibiting only 40% atom efficiency.46 However, a notable advancement occurred with the development of a greener route by BHC (Boots-Hoechst-Celanese), which streamlined the process to three steps while achieving an impressive atom economy of 99%. This innovative approach involved the recycling and reuse of organic solvents and byproducts like acetic acid. Such strides in sustainability garnered significant acclaim, culminating in the receipt of the prestigious Presidential Green Chemistry Challenge Award in 1997 (Scheme 2).47,48
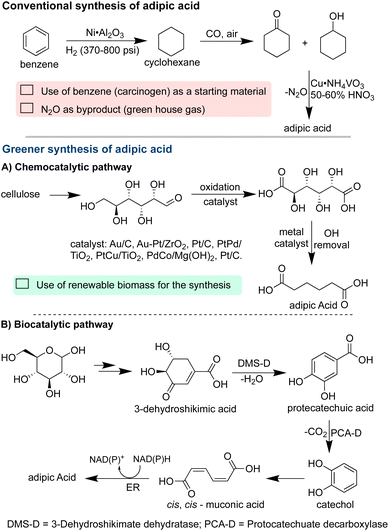
In a parallel vein, the synthesis of adipic acid exemplifies integrating green chemistry principles into the production of widely utilized precursors.49 It also serves as a fundamental component in the synthesis of resins, lubricants, Nylon-6,6, and plasticizers.50 Its global market value in 2021 was estimated at 5.45 billion US dollars, underlying its pervasive role as a foundational element across the chemical, food, and pharmaceutical industries.51,52
Nevertheless, the conventional synthesis of adipic acid relies on benzene as the starting material, a recognized carcinogen.52,53 This method also demands harsh reaction conditions, including the use of nitric acid, leading to the generation of nitrous oxide, a potent greenhouse gas.54 Embracing the principles of green chemistry, significant strides have been achieved, notably through the direct oxidation of cyclohexane, cyclohexanol, and cyclic hexanone to adipic acid using environmentally benign oxidants such as oxygen and hydrogen peroxide with water as a byproduct.55,56 Various recyclable and cost-effective catalysts, including metal oxides, carbon nanotubes, and polyoxometalates, have been explored.57,58 Particularly noteworthy is a synthetic pathway that leverages renewable feedstocks like glucose in combination with biocatalysts, offering a highly sustainable approach.59,60 The innovative methodology utilizes yeast to convert glucose into catechol and adipic acid, with water serving as the reaction medium under ambient pressure and temperature conditions (Scheme 3).59,60 Notably, in 2018, major industrial players, such as Bioamber, Rennovia, and Vedezyne, discontinued the production of adipic acid.61 However, a promising resurgence is evident as Genomatica has forged a partnership with Asahi Kasei, a Japanese-based manufacturer, for commercial synthesis.62 Toray Industries is scaling up a new fermentation process for adipic acid production.63
Pfizer introduced Zoloft, an antidepressant medication, in 1992.64,65 However, the traditional synthesis of its active ingredient, Sertraline, necessitates the use of significant amounts of toxic organic solvents like dichloromethane, THF, toluene, and hexane, leading to the production of approximately 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 liters of waste per 1000 kilograms of Zoloft.66 Moreover, the process involves the utilization of titanium tetrachloride (TiCl4) during the imine formation step, posing safety risks due to its corrosive nature and the emission of HCl fumes in the presence of moisture or air, particularly in large-scale operations. To address these challenges and promote sustainability, Pfizer revised the synthetic protocol by eliminating the usage of toxic organic solvents like THF, hexane, and toluene.67 The updated process incorporated ethyl acetate and EtOH as solvents, which were recycled throughout the process, saving 75
000 liters of waste per 1000 kilograms of Zoloft.66 Moreover, the process involves the utilization of titanium tetrachloride (TiCl4) during the imine formation step, posing safety risks due to its corrosive nature and the emission of HCl fumes in the presence of moisture or air, particularly in large-scale operations. To address these challenges and promote sustainability, Pfizer revised the synthetic protocol by eliminating the usage of toxic organic solvents like THF, hexane, and toluene.67 The updated process incorporated ethyl acetate and EtOH as solvents, which were recycled throughout the process, saving 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 liters of solvent per 1000 kilograms of Zoloft. Additionally, substituting palladium (Pd)/CaCO3 for corrosive TiCl4 led to more selective reductions, significantly boosting the isolated yield (Scheme 4). This greener approach is estimated to have reduced approximately 80 million gallons of waste generation to date.67,68
000 liters of solvent per 1000 kilograms of Zoloft. Additionally, substituting palladium (Pd)/CaCO3 for corrosive TiCl4 led to more selective reductions, significantly boosting the isolated yield (Scheme 4). This greener approach is estimated to have reduced approximately 80 million gallons of waste generation to date.67,68
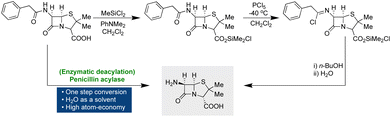
One of the 12 principles of green chemistry advocates for avoiding protection–deprotection or blocking group strategies in synthetic protocols.22 These unnecessary derivatizations increase the number of steps and contribute to waste generation, diminishing process efficiency. A prime illustration is found in the synthesis of 6-aminopenicillanic acid, a crucial intermediate in semisynthetic penicillin production.69 The traditional three-step synthetic route involves the protection–deprotection of functional groups and relies on hazardous reagents such as dichloromethane and PCl5 applied under harsh conditions.70,71 Moreover, to produce 1 kilogram of the product, 8.4 liters of CH2Cl2, 8.4 liters of n-BuOH, 1.2 kilograms of PCl5, and 0.6 kilograms of Me3SiCl, were utilized, resulting in a significant decrease in atom economy.70,71 However, a modern one-step enzymatic diacylation process conducted in water at 37 °C, employing NH3 to maintain the reaction's pH, has emerged as a sustainable alternative.72 By circumventing protection–deprotection strategies, this approach enhances reaction efficiency, yield, and atom economy (Scheme 5).
In the notable case study, Dow Chemical Company and BASF collaborated to develop a more environmentally friendly synthetic protocol for producing propylene oxide, a key compound among the industry's top 30 common intermediates.73 Its annual production surpasses 14 billion pounds, serving as a fundamental component in the synthesis of detergents, polyethylene, glycol ethers, and personal care products.74 The traditional manufacturing route suffered from undesired side reactions, leading to increased waste generation and reduced overall yield.75 To tackle this challenge, Dow and BASF pioneered an innovative approach known as the HPPO (hydrogen peroxide to propylene oxide) process.76–78 The method involves the reaction of propylene and hydrogen peroxide in the presence of a ZSM-5-type zeolite catalyst, yielding propylene oxide (PO) as a final product in excellent yield, while water is the only co-product (Scheme 6A).77 The new implementation of this process resulted in a remarkable 70–80% reduction in waste generation, earning it the Presidential Green Chemistry Challenge Award in 2010.79
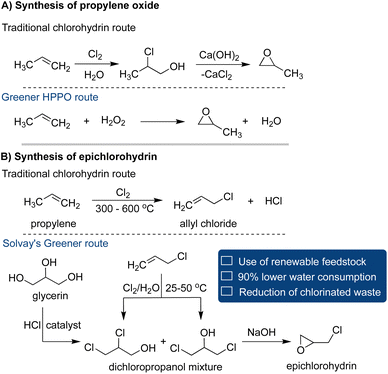 | ||
| Scheme 6 Traditional versus greener route for the synthesis of (A) propylene oxide and (B) epichlorohydrin. | ||
Epichlorohydrin stands as another high-volume commodity chemical for the synthesis of epoxy resins.80,81 The conventional production route relies on propene and chlorine gas as primary raw materials, leading to the formation of allyl chloride at elevated temperatures. Subsequently, hydrochlorination of allyl chloride yields a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of 1,3-dichlorohydrin and 2,3-dichlorohydrin, which, under basic conditions, transforms into epichlorohydrin. However, this process suffers from low chlorine atom efficiency and generates undesirable byproducts such as HCl, chloride anion, and chlorinated compounds. In contrast, Solvay has developed a more efficient and environmentally friendly approach utilizing glycerin, a renewable feedstock.81–83 This two-step process, devoid of solvents, exhibits superior sustainability characteristics. It boasts an enhanced atom economy, minimizes the generation of chlorinated byproducts, and consumes 90% less water than the traditional approach (Scheme 6B).
1 mixture of 1,3-dichlorohydrin and 2,3-dichlorohydrin, which, under basic conditions, transforms into epichlorohydrin. However, this process suffers from low chlorine atom efficiency and generates undesirable byproducts such as HCl, chloride anion, and chlorinated compounds. In contrast, Solvay has developed a more efficient and environmentally friendly approach utilizing glycerin, a renewable feedstock.81–83 This two-step process, devoid of solvents, exhibits superior sustainability characteristics. It boasts an enhanced atom economy, minimizes the generation of chlorinated byproducts, and consumes 90% less water than the traditional approach (Scheme 6B).
1.2. Toxic organic solvents and their alternatives
Organic solvents play a pivotal role in organic reactions,84 serving as the reaction medium and used for product extraction and purification.84,85 However, their extensive use, especially in manufacturing processes, poses significant concerns regarding accidental discharge, problematic synthetic protocols, and disposal.86,87 Efforts to fully recover and purify used solvents face challenges due to their volatility, which increases the risk of exposure and compromises worker safety.88 In response to growing awareness about the hazards posed by certain solvents, attempts were made in the 1990s to substitute highly toxic ones with structurally similar safer alternatives. For example, benzene, identified as a potential carcinogen, was replaced by toluene, which also has limitations.89,90 Similarly, carbon tetrachloride, restricted in 1989, gave way to chlorinated solvents such as dichloromethane or chloroform.91,92 However, later, dichloromethane and toluene were also found as harmful to unborn children and their organs.93,94 Subsequent findings by the World Health Organization (WHO) also highlighted the potential toxicity and carcinogenicity of chloroform and dichloromethane.90,95 Regulatory bodies like the European Union's REACH have imposed restrictions on the use of toluene, chloroform, and dichloromethane,96,97 along with solvents like DMF, DMAc, and NMP, which are slated for future bans.98,99 This underscores the urgent need for clear guidelines to facilitate the transition from toxic organic solvents to greener alternatives.Several pharmaceutical companies, including Pfizer, GlaxoSmithKline, and AstraZeneca, have developed solvent selection guides.100–103 These guides were consolidated into a unified structure by the American Chemical Society's Green Chemistry Institute Pharmaceutical Roundtable (GCI-PR) in 2010,104,105 streamlining the selection process based on criteria such as safety, health, and environmental impact (Fig. 2). However, the direct replacement of toxic organic solvents with greener alternatives in existing protocols to enhance sustainability is infrequent in the industry. This is mainly due to the challenges associated with regulatory changes, including lengthy lead times and additional costs related to renewing regulatory approvals and modifying large-scale production facilities. Therefore, prioritizing the adoption of greener alternatives from the onset to improve process efficiency, yield, and atom economy while simultaneously reducing overall cost is paramount.106,107
Solvents are often used in excess compared to reactants and products, significantly impacting the cost-effectiveness of any process. Adopting solvent-free methodologies or substituting more economical alternatives can lead to substantial cost reductions.84,85,106 While the ideal green method excludes solvents entirely, only a few processes can be performed in the gas phase without solvents.108,109 Mechanochemistry presents another alternative, heralded for its solvent-free, energy-efficient, and low-temperature approach.110–112 Despite its advantages, its widespread applications in large-scale industrial processes remain limited. Additionally, organic solvents are still necessary for product isolation and purification, allowing only a marginal reduction in solvent usage.110
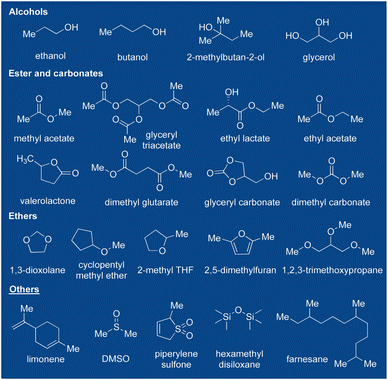
From a green chemistry perspective, the optimal green solvent should meet several criteria: (1) it should be readily available with a secure long-term source of supply. (2) Its performance should be on par or exceed that of conventional organic solvents. (3) High stability is essential. (4) It should be minimally or non-flammable. (5) Recyclability and affordability are crucial. (6) It should be non-toxic or should have acceptable ecotoxicity. (7) Biodegradability is desirable. (8) Easy handling is also important.113 Some alcohols, esters, carbonates, ethers, and other alternatives have been identified as greener options than commonly used solvents shown in Scheme 7.103,114 However, further toxicological and ecological data are needed for many biomass-based solvents. Regarding biomass-based solvents, questions still remain about cost efficiency, bulk availability, and possible recyclability.113
An emerging area within advanced green solvents encompasses ionic liquids,115,116 supercritical fluids,117,118 and deep eutectic solvents.119 Each of these solvent types comes with its own set of advantages and disadvantages. Ionic liquids are salts consisting of poorly coordinating ions, placing them in a category of polar non-coordinating solvents. Despite their efficacy, they often suffer from non-biodegradability and high costs, limiting their industrial-scale application.119–121 Conversely, deep eutectic solvents have gained traction as substitutes for ionic liquids due to their affordability, ease of synthesis, biocompatibility, biodegradability, and low toxicity. While they find utility across various reaction types, challenges such as high viscosity, hygroscopicity, and compatibility issues necessitate further refinement.121,122
Supercritical fluids represent a promising alternative to traditional organic solvents.117,118 These fluids exist in a state where gas and liquid coalesce under specific temperatures and pressures, known as critical conditions. Supercritical CO2 stands out for its non-toxic, non-flammable nature, cost-effectiveness, and extensive application in processes like decaffeinating coffee and tea. Nevertheless, challenges persist, including the limited solubility of polar compounds and the necessity of specialized, often costly equipment, making their utilization primarily capital-intensive.117,118
The utilization of water as a green and sustainable reaction medium has captivated chemists for decades.123–125 Water presents an obvious choice due to its appealing attributes: inexpensive, abundantly available, non-flammable, and non-toxic.123,124 However, its industrial applications still require refinement due to several factors, such as the limited solubility of organic compounds and the instability of water-sensitive intermediates or catalysts within the reaction medium.126 Despite these challenges, in academia, chemists persistently endeavor to pioneer new processes leveraging water as the sole solvent.127,128 Reactions employing water as a solvent are primarily categorized into “in water” and “on water”.124,128–130 The term “in water” denotes homogeneous reaction systems where reactants are entirely soluble, while “on water” refers to reactions conducted under heterogeneous conditions.124,128–130 In the 1980s, Breslow and coworkers first showcased the use of water in Diels–Alder reactions, harvesting the hydrophobic effect.131 It was observed that heating water to high temperatures under critical or supercritical conditions mimics its polarity with organic solvents like ethanol or acetone, thereby facilitating higher reaction rates.131–133 Water's unique properties, such as high surface tension due to hydrogen bonding ability and high dielectric constant, can enhance reaction rates and selectivity in organic transformations.132,133 However, elevated temperatures, additives, and phase transfer catalysis often augment the solubility of organic compounds.125,134,135 Notably, the reactants or products can decompose at higher temperatures. Furthermore, special water-compatible ligands and catalysts are required in catalytic processes, limiting water's broad applicability.127,136
1.3. Aqueous micellar chemistry as a sustainable alternative
Surface active agents, commonly referred to as surfactants, play a crucial role in organic transformations by harnessing the unique properties of water and enhancing its solubility profile.137,138 Surfactant molecules consist of distinct hydrophilic (polar head) or ionic components, along with hydrophobic (non-polar tail) fragments. When dissolved in water, the polar head interacts strongly with water, while the non-polar portion remains insoluble.139,140 As the concentration of the surfactant in water increases to a specific threshold, known as the critical micelle concentration (CMC), molecules aggregate to form micelles.139,140 In this micellization process, the non-polar tails are sequestered inside the micelles while the polar heads orient themselves towards the outer regions. This process is driven by entropy and results in the formation of micelles. In this process, the hydrophilic parts are surrounded by water molecules, while the interior of the micelle largely remains lipophilic.139 One key advantage of micelles is their ability to enhance the solubility of the organic species. Non-polar compounds are either encapsulated within the micelles or reside at the micelle's polar-nonpolar interface, experiencing significantly higher local concentrations and thus promoting faster reaction rates.139–141 Micellar catalysis is an often-used term to describe surfactant-forming micelles as solubilizing nanoparticles. However, as stated years ago by Romsted, Bunton, and Yao, “micellar catalysis a useful misnomer”,142 correctly indicating that usually micelles do not participate in the reactions taking place, and hence, are technically not functioning as catalysts. Regardless, it presents an intriguing opportunity for recycling both the reaction media and the catalysts in organic transformations.143,144 After the reaction completion, the micellar solution containing the metal catalyst confined within the micelles can be easily recycled through simple filtration of the final product or extraction using water-immiscible organic solvents in minimal amounts.The most widely used surfactants are typically classified into three main categories: anionic, cationic, and non-ionic surfactants. In the latter category, PEG (polyethylene glycol) is predominantly employed as the hydrophilic part. Anionic surfactants feature an anionic head group paired with a long aliphatic tail.145–147 Sodium dodecyl sulfate (SDS) stands out as one of the most utilized anionic surfactants, renowned for its high solubility of organic species.145,148,149 Furthermore, charged metal species or metal nanoparticles (NPs) can interact with anionic micelles through ionic interactions, thereby enhancing stability and accelerating reaction rates (Scheme 8).150
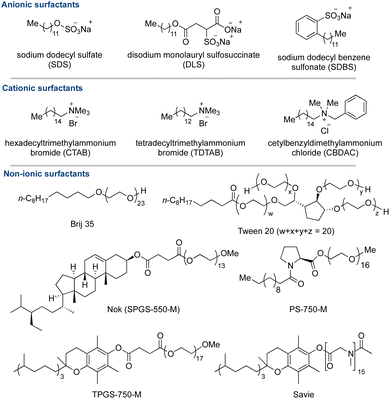
Cationic micelles, while possessing catalytic potential, suffer from limited applicability due to their propensity to bind strongly with metals, thereby diminishing catalytic activity.150,151 To mitigate this issue and prevent inactivation of catalysis, bulky ligands become necessary to shield the metals, albeit at the expense of constraining their utility in metal-catalyzed reactions.151,152 In contrast, non-ionic surfactants represent a more prevalent class, esteemed for their stability and compatibility with biocatalysis.153,154 These surfactants often contain polyethylene glycol (PEG) as a hydrophilic component.127,129,135,155 Brij, Triton, and Tween stand out for their widespread adoption owing to their cost-effectiveness and efficacy in organic transformations.156–159 However, Triton's toxicity led to its prohibition in Europe in 2020.160–162 In response to sustainability imperatives, a new wave of designer surfactants has emerged,163–168 offering promising alternatives and leveraging micellar catalysis to facilitate diverse organic transformations (Scheme 8).169–172
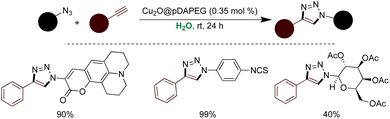
Lipshutz's pioneering contributions have significantly advanced the realm of micellar catalysis, mainly through innovative methodologies facilitating the utilization of newly designed PEG-based surfactants across diverse organic transformations,173 notably in cross-coupling chemistry126,135,154,172 These surfactants, meticulously crafted to incorporate readily available, non-toxic, and biodegradable components, have garnered widespread acclaim. Among the notable iterations are the first-generation PTS, the second-generation TPGS-750-M, and the third-generation SPGS-550-M, also known as Nok, which have found extensive applications within the Lipshutz group.155,168,172,174 Concurrently, the Handa group introduced a benign proline-based surfactant, PS-750-M, engineered to emulate polar solvents like DMAc or DMF, proving instrumental in various organic transformations, particularly in transition metal-catalyzed cross-couplings and nanocatalysis (Scheme 8).169,175–182 Noteworthy is the prevalent use of polyethylene glycol (PEG) as a hydrophilic constituent in surfactants; however, these polyethers pose challenges due to their limited biodegradability and potential peroxide formation upon prolonged air exposure. In response, the Lipshutz group developed ‘Savie’, a surfactant derived from vitamin E and polysarcosine, distinguished by its biodegradability and absence of peroxide by-products. The broad spectrum of applications of these micellar media in organic transformations is illustrated in Fig. 3.183 Notably, corrosive reagents are still needed for Savie's synthesis.
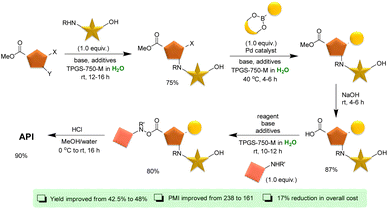
The field of micellar catalysis has experienced remarkable expansion over the past two decades, attributable to its outstanding catalytic performance and application in various industrially significant organic transformations.184–186 In a milestone achievement, Novartis reported the first large-scale synthesis of an active pharmaceutical ingredient (API) utilizing surfactant technology in 2016.187 The five-step synthesis encompassed a range of chemical reactions, including SNAr reaction for N-arylation in the presence of a free hydroxy group, Pd-catalyzed Suzuki–Miyaura cross-coupling, and in situ hydrolysis of ester to acid, followed by acid-amine couplings, all conducted under an aqueous medium. The final step involved base-mediated ester hydrolysis in a water/MeOH system, yielding the API with an overall yield of 48%. This approach improved the overall yield by 5.5% compared to the traditional route, while exhibiting a lower process mass intensity (PMI) of 161. Furthermore, the surfactant process led to a significant cost reduction of 17% (Scheme 9).
The Lipshutz group, in collaboration with Novartis, presented a compelling demonstration of employing a micellar medium in synthesizing a bioactive molecule.188 The focus was on Boscalid, an active fungicide marketed by BASF with an annual production of 1000 metric tons.189,190 However, the traditional synthetic route involves the high Pd loading in Suzuki–Miyaura couplings and the use of expensive Pt/C for nitro reductions under elevated hydrogen pressure, all conducted in organic solvents.189 The Lipshutz group developed a sustainable 3-step, 1-pot methodology, employing a minimal amount of THF as a co-solvent. This approach entails C–C bond formations utilizing only 700 ppm of Pd catalyst within aqueous micelles of 2 wt% TPGS-750-M, under mild heating of 55 °C. Subsequently, the nitro reduction was carried out in the same pot using carbonyl iron powder (CIP), followed by Schotten Baumann's reaction, resulting in the final product with an impressive overall yield of 83% (Scheme 10).188
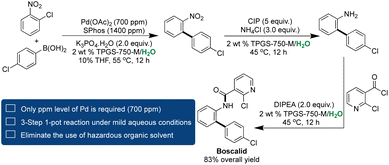 | ||
| Scheme 10 A sustainable 1-pot, 3-step synthesis of Boscalid using ppm Pd catalysis in aqueous micellar medium. | ||
1.4. Adoption of micellar catalysis by the pharmaceutical industry
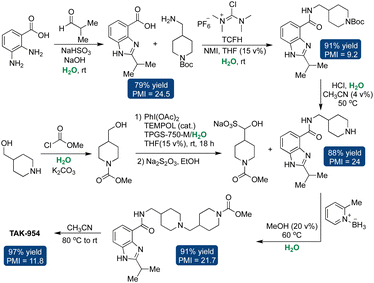
Novartis showcased the application of micellar catalysis in kilogram-scale synthesis of LSZ10, a bioactive drug recognized as an estrogen receptor-degrader effective against breast cancer.191,192 This endeavor involved a Pd-catalyzed Suzuki–Miyaura coupling conducted on a 600-gram scale, utilizing only 1.5 mol% Pd catalyst in aqueous micelles of 2 wt% TPGS-750-M.193 The additive LiBr was used to minimize unwanted hydrodebromination. The resulting product boasted high purity, and reduced PMI from 116 to 68 (Scheme 11).Takeda modified the production process of its API TAK-954, a receptor agonist utilized in treating post-operative gastrointestinal dysfunction.193,194 The conventional approach relied on multistep synthesis employing hazardous solvents, resulting in substantial waste generation. The overall yield was only 35%, accompanied by a PMI of 350. However, by integrating water as the primary reaction medium throughout all synthetic stages, organic solvent usage plummeted by 94%, leading to a notable enhancement in yield, rising from 35% to 56%. Remarkably, this adjustment substantially reduced the process's PMI from 350 to 79. Furthermore, the utilization of organic solvents for product purification and isolation was significantly reduced (Scheme 12).193
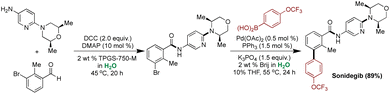
In 2019, Lipshutz and coworkers developed an innovative synthetic approach for producing Sonidegib, an anti-cancer agent.195,196 The conventional method posed challenges due to its reliance on organic solvents and high Pd loading (10 mol%) for the Suzuki–Miyaura coupling step.197 The modified greener method involved the amide coupling performed utilizing DCC (N,N′-dicyclohexylcarbodiimide) and catalytic DMAP (10 mol%) in aqueous solution of 2 wt% TPGS-750-M, resulting in 82% isolated yield. Subsequently, the Suzuki–Miyaura coupling was executed with only 5000 ppm (0.5 mol%) of Pd loading, compared to 10 mol% in the traditional method, under mild micellar conditions.195 The final product Sonidegib, was obtained with an overall yield of more than 80%. The new methodology exhibited a fivefold reduction in E-factor compared to the conventional method (Scheme 13).
In a notable application of micellar catalysis in medicinal chemistry, AbbVie reported a direct comparison between micellar catalysis and chemistry in a traditional organic solvent to synthesize bioactive molecules.198,199 Surprisingly, certain reactions that yielded no product in organic solvents proved successful in micellar media. For instance, Sonogoshira couplings employing a 5 mol% Pd and 10 mol% CuI system in DMSO failed to produce the desired arylated alkyne product. However, employing aqueous micellar conditions with a lower catalyst loading of 1 mol% Pd and no Cu resulted in a 48% isolated yield of the desired product (Scheme 14A).198,199 Similarly, the Rh-catalyzed synthesis of dihydroquinolinones was performed in dioxane and aqueous micelles of TPGS-750-M, which showed significant differences. When the reaction was conducted with [RhCl(COD)]2, KOH as a base, and 1,4-dioxane as solvent under reflux conditions, no product was obtained. Conversely, employing micellar conditions at room temperature yielded the desired product in a 78% yield. These examples showcased the superiority of micellar catalysis over traditional methodologies (Scheme 14B).198,199
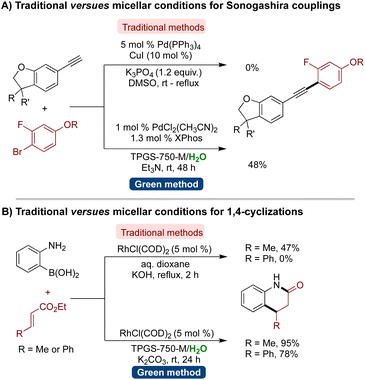 | ||
| Scheme 14 AbbVie's comparison of conditions for (A) Sonogashira-couplings, and (B) 1,4-cyclization (traditional versus micellar catalysis). | ||
In a recent endeavor to scale up a challenging C(sp2)–C(sp3) cross-electrophilic coupling, Novartis executed a kilo-scale synthesis of an intermediate for their drug candidate employing dual Ni/Cu catalysis under micellar conditions.200 Traditionally, such methods relied on toxic organic solvents, primarily DMF, DMAc, and NMP. However, this report introduces the first cross-electrophile coupling in water utilizing 2 wt% aqueous TPGS-750-M as a reaction medium at a kilo-scale. Remarkably, the desired product was obtained with an excellent yield of 85% and 98% purity through a single isolation process (Scheme 15).
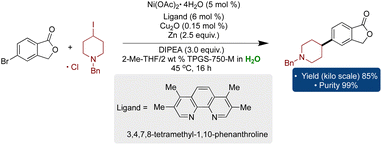 | ||
| Scheme 15 The first example of kilo-scale cross-electrophilic coupling using dual Ni/Cu catalysis in the aqueous micellar environment by Novartis. | ||
1.5. Sustainable nanocatalysis in an aqueous environment
Organometallic catalysts serve as a cornerstone in several organic transformations.201 Catalysis is broadly categorized into homogeneous and heterogeneous catalysis.202 Homogeneous catalysis is a type of catalytic reaction in which both the catalyst and the reactants are in the same phase, typically in a solution or a gaseous state. The catalyst molecules interact directly with the reactant molecules to facilitate the reaction, and the catalytic cycle occurs entirely within the same phase. This type of catalysis often involves coordination complexes or organometallic compounds as catalysts, where the active species undergo reversible coordination with reactants to lower the activation energy of the reaction.203 On the other hand, heterogeneous catalysis refers to a type of catalytic reaction where the catalyst exists in a different phase from the reactants. Typically, the catalyst is in a solid phase, while the reactants are either in the gas or liquid phase. In heterogeneous catalysis, the reactant molecules adsorb onto the catalyst's surface, where the catalytic reaction occurs. Unlike homogeneous catalysis, where the catalyst and reactants are uniformly mixed, the catalyst remains distinct from the reaction mixture in heterogeneous catalysis.203,204 While homogeneous catalysis finds widespread use in the chemistry community due to the enhanced interactions between metal complexes and reactants,205–207 a notable drawback is the challenge of separating metal catalysts from final products, leading to metal contamination—a significant concern in the pharmaceutical industry.208 In contrast, heterogeneous catalysis involves immobilizing the metal on solid supports, minimizing or eliminating metal leaching, and enhancing recyclability, albeit with lower catalytic activity due to fewer accessible active sites.204,206 To address such challenges inherent in traditional organometallic catalysis, NP catalysis has emerged as a promising field, combining characteristics of both homogeneous and heterogeneous catalytic systems.209–211 NPs offer advantages such as large surface area, high catalytic activity, and selectivity.212 Their poor solubility in some organic solvents facilitates easy separations and recyclability, thus mimicking the benefits of homogeneous catalysis while overcoming the limitations of accessing fewer catalytic sites (Fig. 4).209,211Despite their promising attributes, NP catalysis faces several sustainability challenges. These include the utilization of costly ligands and metals, the propensity of NPs to aggregate, leading to diminished catalytic activity,213 instability in the aqueous medium,214 scalability and reproducibility concerns in their synthesis,215 potential toxicity,216 difficulties in post-reaction catalyst recovery,217 bench-stability, and the risk of metal leaching.218 To overcome these challenges, it is imperative to devise innovative processes that leverage earth-abundant metals and micellar catalysis. This approach aims to enhance the robustness and recyclability of resulting catalysts while mitigating the need for expensive ligands.
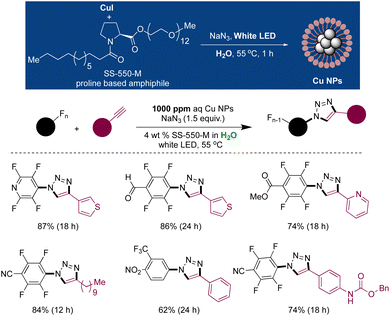
Recently, Handa and coworkers developed Cu(II) nano aggregates that in situ produce Cu(I) NPs upon light irradiation in the presence of sodium azide, enabling ppm level (1000 ppm) Cu(I) catalysis in an aqueous micellar environment.177 It was demonstrated that the SS-550-M, a modification of PS-750-M with a lower chain length of mPEG (500-M), played a crucial role in generating and stabilizing Cu NPs through its tertiary amide core. Mixing CuI with aqueous micelles of SS-550-M and NaN3 led to the formation of amphiphile-bound Cu-azide nanomaterial. High-resolution transmission electron microscopy (HRTEM) and scanning electron microscopy (SEM) analysis revealed the presence of nano-rings with a size range of 100–120 nm. The binding of Cu with the tertiary amide core of SS-550-M was confirmed using 13C NMR, 15N NMR, and IR spectroscopy. XAS analysis confirmed the oxidation state of Cu in nanomaterial as +2. However, exposure of NPs to white LED conditions resulted in a reduction of Cu(II) to Cu(I), as verified by X-ray absorption spectroscopy (XAS) analysis. Density functional theory (DFT) calculations, in conjunctions with XAS analysis, suggested an azide-to-Cu charge transfer mechanism responsible for the generation of Cu(I) species. This unique photoactive Cu nanomaterial was employed in [3 + 2]-cycloadditions of in situ generated perfluoroazide and alkyne under white LED irradiation with catalyst loading of 1000 ppm. The methodology demonstrated a broad substrate scope, and the nanomaterial exhibited high stability for up to 3 months (Scheme 17).
Khan and coworkers demonstrated the use of CuO NPs in catalyzing click reaction for one-pot synthesis of substituted 1-alkyl-1,2,3-triazole-4-methanol, followed by oxidation with PCC (pyridinium chlorochromate) to access 1-alkyl-1,2,3-triazole-4-carbaldehyde in an aqueous medium.241 Various alkyl halides were reacted with propargyl alcohol in the presence of sodium azide, 5 mol% Cu NPs, and 10 mol% sodium ascorbate to yield various triazoles, which were subsequentially oxidized. Notably, the final triazole-4-carbaldehydes underwent further reaction with phenylacetylene and aminopyridine, forming nitrogen-rich substituted heterocycles (Scheme 18).241
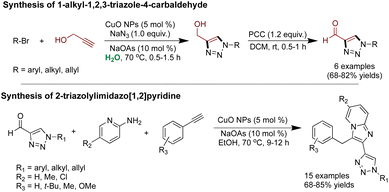 | ||
| Scheme 18 Cu NPs catalyzed for the synthesis of 1-alkyl-1,2,3-triazole-4-carbaldehyde and 2-triazolyl-imidazo[1,2]pyridine. | ||
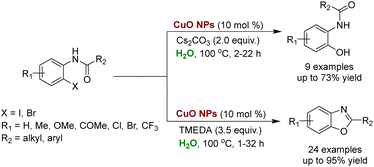
CuO NPs found application in synthesizing benzoxazole, a common moiety present in many bioactive natural products responsible for their anticancer activities.242,243 Patel and coworkers demonstrated the synthesis of O-hydroxyphenyl benzamides and 2-arylbenzoxazoles utilizing CuO NPs in an aqueous medium.244O-Hydroxyphenylbenzamides were synthesized using non-ligated CuO NPs in combination with Cs2CO3 as a base, resulting in moderate-to-good yields. However, when CuO NPs were ligated with TMEDA (tetramethylethylenediamine), high selectivity towards 2-arylbenzoxazoles was achieved with excellent yields. The catalyst exhibited good recyclability for up to 5 cycles (Scheme 19).244
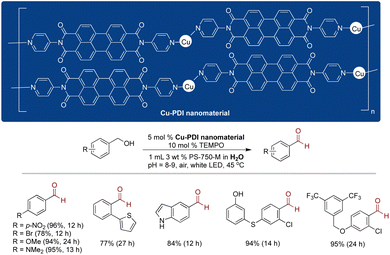
Handa and coworkers introduced a novel copper-perylene diimide (PDI)-based polymeric nanomaterial for the sustainable aerobic oxidation of alcohols in an aqueous micellar medium.245 The synthesis of this nanomaterial involved the complexation of Cu(CH3CN)4PF6 with PDI at 80 °C in dimethoxyethane for 14 h. Characterization by SEM confirmed the porous nature of the resulting material with nanochannels, while X-ray photoelectron spectroscopy (XPS) validated the +1 oxidation state of Cu in the nanomaterial. Under white light irradiation, utilizing 5 mol% Cu nanomaterial, 10 mol% TEMPO (2,2,6,6-tetramethyl-1-piperidinyloxy), and aqueous micelles of PS-750-M, enabled the oxidation of benzylic alcohols to their corresponding aldehydes with broad substrate scope and high selectivity. The Cu-PDI nanomaterial exhibited high recyclability, retaining its activity for up to two cycles without significant loss. Mechanistic insights, derived from time dependent DFT calculations, suggested that visible light triggered a charge transfer forming a triplet state, subsequently quenched by molecular oxygen, thereby responsible for the redox processes (Scheme 20).
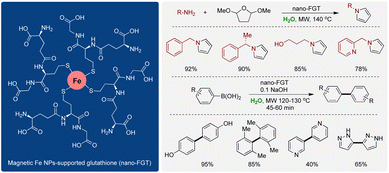 | ||
| Scheme 21 Nano ferrous-glutathione (nano-FGT) for Paal–Knorr reaction to synthesize pyrroles from an amine. | ||
Fe nanocatalysis remains a relatively underdeveloped field within organometallic catalysis, which is characterized by limited applicability to industrial settings.257,258 Handa and Lipshutz have made significant strides in this area by developing doped Fe NPs containing trace metals such as Pd, Cu, or Ni at ppm levels to facilitate various organic transformations.259–265 The NPs are prepared from inexpensive FeCl3 using MeMgCl as a reductant in THF. A diverse array of transformations, including Suzuki couplings, cycloadditions, reductions, Negishi couplings, Heck reactions, and Sonogeshira couplings, can all be achieved using ppm levels of Pd or Ni or Cu and different phosphine ligands alongside Fe as a base metal.259–267
In 2015, Handa and Lipshutz introduced a new class of Fe-based NPs featuring a ppm level of ligated Pd for sustainable C–C couplings in an aqueous medium.265 The reaction proceeded under mild aqueous conditions (rt–45 °C) with an exceptionally low loading of 350–400 ppm Pd. The versatility of this technology was demonstrated across numerous substrates with good-to-excellent yields. Control experiments revealed the importance of both metals in achieving the desired reactivity, as no reaction occurred in the presence of Pd or Fe alone. TEM analysis unveiled needle-shaped NPs comprising both Fe and ligated Pd. Subsequently, the efficiency of this technique was linked to the nano-to-nano effect.267 Cryo-TEM analysis confirmed that mPEG acted as a stabilizer, facilitating the delivery of reactants from nanomicelles to the NPs for efficient catalysis (Scheme 22A).
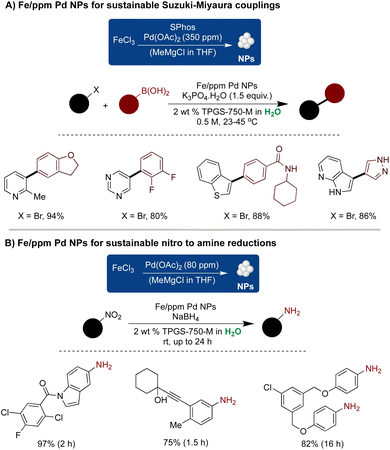 | ||
| Scheme 22 Fe/ppm Pd NPs for sustainable (A) Suzuki–Miyaura couplings, and (B) nitro to amine reductions in aqueous micellar conditions. | ||
The modification of Fe ppm Pd NPs utilizing only 80 ppm of Pd(OAc)2 relative to FeCl3 under ligand-free conditions was reported for nitro reductions under micellar conditions. The reaction conditions were mild, employing NaBH4 as a reductant at room temperature, and proved applicable to a broad range of substrates.259 The representative examples are depicted in Scheme 22B. Subsequently, Fe NPs doped with ppm levels of Pd and Ni were synthesized, exhibiting enhanced activity and broad functional group tolerance towards nitro reductions.264
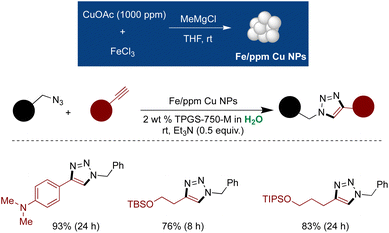
Extending on this technology, new Fe ppm Cu NPs were synthesized using 1000 ppm Cu(OAc)2 and applied to cycloaddition of azides and alkynes to form substituted triazole-products.268 A variety of 1,4-disubstituted triazoles were synthesized with moderate-to-excellent yields. The trace Cu impurity in the isolated product was <1 ppm, as detected by inductively coupled plasma mass spectrometry (ICP-MS) analysis. Notably, the aqueous mixture and the NPs were recyclable up to 5 times, demonstrating the robustness of the methodology (Scheme 23).
This Fe ppm Pd technology was further extended to various cross-couplings. For example, Fe NPs synthesized using XPhos as ligand with 500 ppm Pd were applied on Cu-free Sonogashira couplings under mild aqueous micellar conditions (Scheme 24A).262 Several diverse substrates were synthesized using this methodology, including direct application in the synthesis of bioactive molecules. A recycling study demonstrated that both the aqueous medium and the NP catalyst could be recycled five times, achieving a remarkably low E-factor of 4.1.262 On similar grounds, by replacing the ligand from XPhos to AmPhos and employing 2500 ppm Pd loading, new Fe ppm Pd NPs were developed and utilized in water-sensitive Negishi couplings in aqueous micellar medium (Scheme 24B).269 The approach proved compatible with a diverse range of highly functionalized (hetero)aromatic bromides, yielding coupling products with good-to-excellent yields. Notably, a head-to-head comparison with traditional Negishi couplings used in the industry showcased the superiority of the Fe ppm Pd NPs technology, offering higher yields, remarkably low catalyst loading (ppm Pd with no Ni and Ir), and eliminating the need for toxic organic solvents as reaction media.269
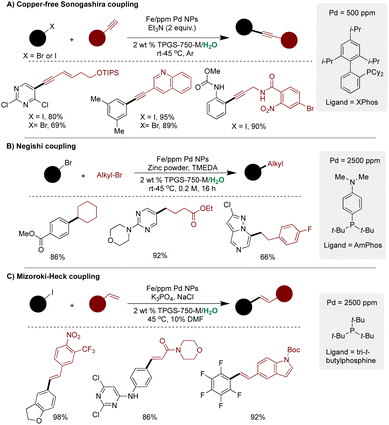 | ||
| Scheme 24 Fe/ppm Pd NPs for; (A) Cu-free Sonogeshira couplings, (B) Negishi coupling, and (C) Mizoroki–Heck couplings in an aqueous micellar medium. | ||
In 2021, the Lipshutz group extended this technology to Mizoroki–Heck couplings in an aqueous medium (Scheme 24C).270 NPs derived from FeCl3 and ppm Pd ligated with t-Bu3P proved highly active for the coupling reaction under mild conditions, demonstrating a broad substrate scope. Intriguingly, no residual Pd was detected in the purified products. The methodology was further extended to gram-scale reactions, achieving an impressively low E-factor of only 0.62.270
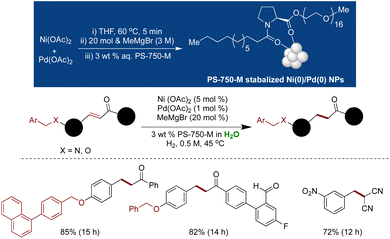
Due to the inherent instability of Ni(0) and its susceptibility to oxidation under aqueous conditions, Handa and coworkers devised a strategy to stabilize Ni(0) at the nanoscale through doping with Pd.180 Only a minimal amount of Pd, in conjunction with a proline-based surfactant, was found to be indispensable for maintaining the stability of Ni(0) NPs. The synthesis involved the combination of 1 mol% Pd(OAc)2, 10 mol% Ni(OAc)2 ligated with 3,4,7,8-(Me)4-1,10-phenanthroline, and 20 mol% MeMgBr as a reductant in THF, followed by the swift addition of the proline surfactant PS-750-M. HRTEM analysis confirmed the formation of microballs of Ni/Pd NPs, while XPS confirmed the zero oxidation state of Ni and Pd within these microballs. These Ni(0) NPs exhibited exceptional selectivity for carbamate deprotection in the presence of benzyl ethers under ambient hydrogen pressure with mild heating at 45 °C under aqueous micellar conditions. This methodology demonstrated broad applicability for Cbz deprotection in the presence of O-benzyl, N-benzyl, olefins, and nitriles groups. The NP catalyst displayed high stability, with the one-month-old catalyst retaining effectiveness comparable to that of the fresh one. However, the presence of ligand remained necessary to stabilize Ni(0) species in water (Scheme 26).180
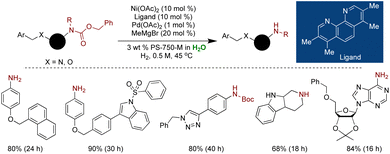 | ||
| Scheme 26 Ni(0)Pd(0) NPs for selective carbamate deprotection in the presence of benzyl ethers under aqueous micellar medium. | ||
To synthesize ligand-free Ni(0) NPs in water, Handa and coworkers developed a method involving the stabilization of Pd-doped Ni NPs using a designer amphiphile, PS-750-M possessing a tertiary amide core.176 The binding of micelles to NPs through the proline linker of the surfactant was confirmed via surface-enhanced Raman spectroscopy, revealing a shift in the carbonyl signals of the amphiphile when bound to the metal NPs. The stable Ni(0)/Pd(0) NPs displayed remarkable selectivity in 1,4-reductions of enones, enamides, enenitriles, and ketoamides under ambient hydrogen pressure at 45 °C within an aqueous micellar environment. The methodology demonstrated applicability across a wide range of substrates with high selectivity. However, the NPs were sensitive to air, leading to a reduction in selectivity primarily due to the aerobic oxidation of Ni(0) to Ni(II) (Scheme 27).176
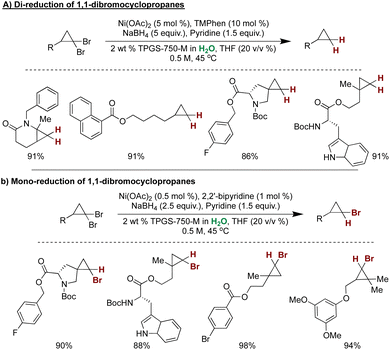
Lipshutz and coworkers harnessed the unique selectivity of Ni NPs for the selective mono- and di-hydrodehalogenative reductions of gem-dibromocyclopropanes.282 Employing 5 mol% Ni(OAc)2 with 3,4,7,8-(Me)4-1,10-phenthroline as a ligand, NaBH4 as the hydrogen source, and pyridine as a base, resulted in selective dehalogenation of dibromocyclopropanes with good functional group tolerance. Furthermore, reducing the loading of Ni(OAc)2 to 0.5 mol% ligated with 2,2′-bipyridine resulted in highly selective debromination, affording mono-brominated cyclopropanes as the sole product (Scheme 28).282
1.6. (Nano)-palladium catalysis in aqueous media
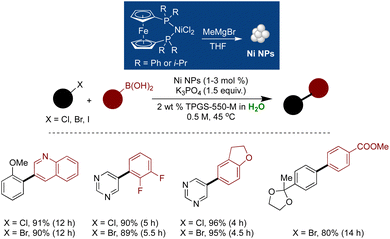
Pd stands as the most potent and extensively employed metal in cross-coupling chemistry.190,219,283,284 Given its broad utility, developing Pd NPs with heightened catalytic efficiency and recyclability represents a burgeoning frontier in catalysis.285–288 Pd(II), in the presence of mild reductants, readily undergoes reduction to Pd(0), subsequently aggregating to form Pd(0) NPs.286,288,289 These Pd NPs have exhibited remarkable activity and enhanced selectivity in the cross-couplings.286,290 For example, in 2014, Lipshutz and coworkers demonstrated the synthesis of Pd NPs through the admixture of Pd(OAc)2 with NaBH4 under aqueous micellar conditions. These NPs displayed high selectivity in reducing alkynes to Z-selective alkenes at room temperature.291 The reaction proceeded under mild conditions, with hydrogen gas at ambient pressure, and showcased applicability across a wide range of functionalized alkynes. Both the NPs and the micellar medium were recyclable (Scheme 29).291The Handa group has demonstrated the unique properties of Pd NPs (with or without ligand) in combination with designer proline-based surfactant PS-750-M. This surfactant serves dual roles as an NP stabilizer and a ligand for cross-coupling reactions.169,178,179,292 Achieving Buchwald–Hartwig aminations, which are challenging due to NP deactivation caused by their binding with amine substrates, was made possible through the synthesis of Pd NPs ligated with cBRIDP, doped with Cu, and adsorbed on an activated carbon surface.179 These tailored NPs facilitated highly efficient C–N bond formations. Micelles of PS-750-M played a crucial role in achieving the desired reactivity, and the methodology proved applicable across a wide range of substrates, including nitrogen-rich heterocycles. HRTEM analysis revealed an average NP size of 2.5 nm. 31P NMR analysis of NPs indicated the binding of the ligand (cBRIDP) with both Cu and Pd, a finding corroborated by XAS analysis. However, no direct interaction between Cu and Pd was observed, suggesting that the ligand was a bridge between both metals. The catalyst exhibited high stability and recyclability, with no significant loss of reactivity observed over five cycles (Scheme 30).179
In pursuit of efficient C–N bond formations in an aqueous medium, the collaboration between the Braje and Handa groups unveiled a novel technology enabling the instantaneous formation of Pd NPs within the hydrophobic pockets of environmentally benign, cost-effective and biodegradable cellulose-based polymer hydroxypropyl cellulose (HPMC).130,293 It consists of both hydrophobic and hydrophilic regions, serving as a reaction medium for ultrafast amination in water. Notably, the hydrophobic pockets of HPMC, generated by its alkyl ether side chains and cyclic groups, play a crucial role in initiating the formation of metal NPs and providing surfaces for their stabilization. Furthermore, the high concentration of reactants within these hydrophobic pockets accelerates reaction rates. Mixing (cinnamyl)PdCl2 with t-BuXPhos in 2 wt% aqueous HPMC for 5 minutes at 45 °C yielded ultrasmall ligated Pd NPs with an average size of 1.5 nm, as determined by HRTEM analysis. The activity of these NPs was assessed in rapid aminations in water, revealing unprecedentedly short reaction times under standard conditions, along with excellent functional group tolerance and high isolated yields. The scalability of this protocol was successfully demonstrated on gram-scales (Scheme 31).293
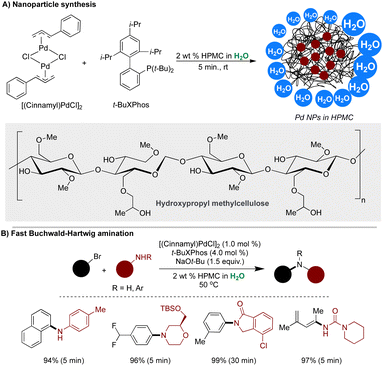 | ||
| Scheme 31 (A) Synthesis of ligated Pd NPs in HPMC, and (B) Pd NPs catalyzed fast Buchwald–Hartwig amination. | ||
Handa and coworkers devised a unique protocol to explore the reactivity of carbenes in water, involving use of Pd NPs ligated with triphenylphosphine and shielded by nanomicelles of PS-750-M.182 This approach enabled metal–carbene migratory insertion for the synthesis of terminal olefins. These Pd NPs exhibited broad generality in coupling reactions with high recyclability. Characterization of the NPs was conducted through 31P NMR, HRTEM, and XPS analysis. Dynamic light scattering (DLS) experiments revealed that the size of the nanomicelles of PS-750-M increased from 200 nm to 520 nm upon the addition of NPs to the micellar solution, indicating that the NPs remained encapsulated within the micelles and were shielded from water molecules. The nanocatalyst demonstrated stability for up to 30 days with no significant decrease in activity (Scheme 32A).182
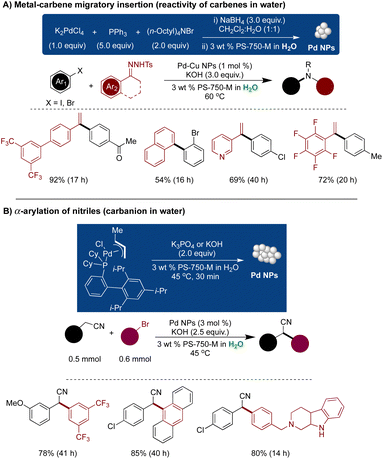 | ||
| Scheme 32 Pd NPs catalyzed; (A) metal–carbene migratory insertion for the synthesis of terminal olefins (reactivity of carbenes in water) (data taken from ref. 148), and (B) α-arylation of nitriles (carbanion in water). | ||
The shielding effect of nanomicelles was harnessed for the α-arylation of phenyl acetonitriles with aryl halide through the generation of carbanion intermediate.296 Traditional methods demand extremely dry reaction conditions, high catalyst loading, and the use of toxic organic solvents under reflux conditions. The micellar methodology employed pre-complexed [XPhosPd(crotyl)Cl], which, in the presence of a base, instantaneously generated Pd(0) species through reductive eliminations of crotyl chloride. This led to the aggregation and formation of ultrasmall-ligated Pd NPs. The choice of ligand was critical, with the electron-rich XPhos ligand enhancing the NP's lipophilicity, thereby facilitating more effective interaction with the micellar core and ensuring high stability. The NPs were characterized by HRTEM, XPS, and 31P NMR analysis. The coupling reactions were conducted using a 3 mol% Pd catalyst, along with KOH as a base and 3 wt% aqueous PS-750-M at 45 °C, which in situ generated active Pd NPs. The substrate scope revealed high functional and protecting group tolerance and superior compatibility towards heterocycles. A mechanistic investigation involving trapping the carbanion species with aldehyde or allyl bromide provided evidence for the existence of carbanion-type species in the reaction mechanism (Scheme 32B).296
Handa and coworkers devised a novel strategy to impart stability to the non-ligated Pd NPs through the designer surfactant PS-750-M.169 Ultrasmall Pd(II) NPs were synthesized and stabilized in 3 wt% aqueous PS-750-M at 45 °C using Pd(OAc)2 as a metal precursor.292 The stirring rate proved crucial, as stirring at 800 rpm yielded nano-aggregates sized 50–60 nm, while vigorous stirring at 1500 rpm produced ultrasmall NPs of 2 nm size. The surfactant PS-750-M played a pivotal role in stabilizing Pd NPs, as confirmed by HRMS and IR spectroscopy. IR studies revealed a shift in the carbonyl stretches of NPs-bound PS-750-M compared to unbound PS-750-M, confirming NP binding through the surfactant's tertiary amide core. Subsequent HRMS analysis corroborated the accurate mass of 1156.582 Da corresponding to the Pd(II) bound PS-750-M. The activity of these highly stable Pd(II) NPs was evaluated in the oxidative Mizoroki–Heck type couplings in aqueous micellar conditions. Optimized conditions included 3 mol% Pd(II) NPs, benzoquinone as oxidant, and 3 wt% aqueous PS-750-M at rt with stirring at 1500 rpm. The methodology was applied to a wide range of substrates, including perfluoro styrene, acrylates, alkyl styrene, and heterocycles. However, the method was not compatible with N-heterocycles (Scheme 33).292
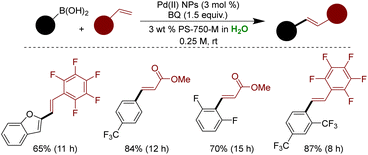 | ||
| Scheme 33 Ligand-free ultrasmall Pd(II) NPs for oxidative Mizoroki–Heck couplings in aqueous micellar medium. | ||
Handa and coworkers advanced ligand-free technology to synthesize and stabilize Pd(0) NPs from Pd(II) precursors using phenylboronic acid as a mild reducing agent under basic conditions.178 The process involved base-assisted Pd(II) transmetallation by phenyl nucleophile, followed by reductive elimination, generating biphenyl and Pd(0) species. Rapid nucleation produced Pd(0) NPs, stabilized by PS-750-M. Comprehensive characterization using HRTEM, IR, NMR, and surface enhanced Raman spectroscopy (SERS) confirmed the properties of the NPs. HRTEM analysis revealed ultrasmall Pd(0) NPs with an average size of 2.4 nm, while XPS confirmed the presence of Pd(0) NPs. IR analysis elucidated the binding of Pd NPs with the carbonyls of the amphiphile, showing new carbonyl signals in Pd(0) NP-bound PS-750-M compared to unbound PS-750-M. 13C NMR analysis exhibited multiple downfield signals in the carbonyl region (183 to 180 ppm) in PS-750-M-bound Pd NPs compared to unbound PS-750-M. SERS further confirmed the binding of Pd NPs through the carbonyl of the surfactant. These highly stable Pd(0) NPs were then utilized in cross-couplings of boronic acids with water-sensitive acid chlorides under mild aqueous conditions. The acid chloride's high solubility in the hydrophobic micellar core prevented hydrolysis under basic pH conditions, facilitating highly efficient cross-couplings. A wide range of acid chlorides, including alkyl acid chloride and (hetero)arylboronic acids, were compatible under standard reaction conditions. Notably, these ligand-free Pd(0) NPs were also effective in Suzuki–Miyaura cross-couplings under mild aqueous conditions, demonstrating broad substrate scope and excellent functional group tolerance (Scheme 34).178
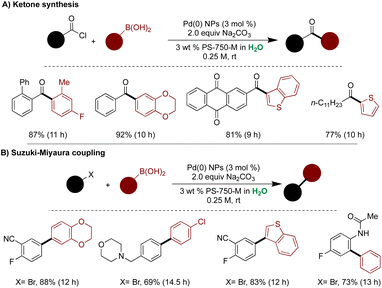 | ||
| Scheme 34 Ligand-free ultrasmall Pd(0) NPs for, (A) biaryl ketone synthesis, and (B) Suzuki–Miyaura couplings in aqueous micellar medium. | ||
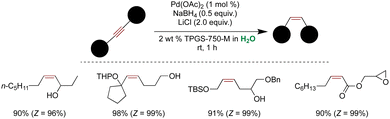
A spontaneous synthesis of Pd(0) NPs from Pd2dba3, an air and water-sensitive Pd precursor, was successfully demonstrated.303 The NPs were synthesized by stirring Pd2db3 in a micellar solution of 3 wt% PS-750-M under basic conditions. Various analytical techniques, including 1H NMR, IR, mass spectrometry, HRTEM, and XPS analysis, were employed to characterize these NPs. These NPs, stabilized by metal-micellar binding, exhibited high activity in cross-couplings of water-sensitive triazine adducts of carboxylic acid with aryl/heteroaryl boronic acids, facilitating the synthesis of biaryl ketones with a broad scope and excellent functional group tolerance. Additionally, Pd(0) NPs were synthetically synthesized by stirring Pd2(dba)3 in an aqueous micellar medium under basic pH and hydrogen pressure or by using MeMgBr as a reductant instead of hydrogen. However, these NPs were less effective than naturally formed NPs when no reductant was used (Scheme 35A).303
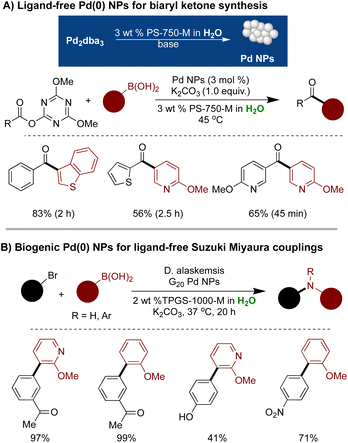 | ||
| Scheme 35 (A) Ligand-free Pd NPs for biaryl ketone formation in aqueous micellar environment; (B) biogenic Pd(0) NPs in ligand-free Suzuki–Miyaura couplings. | ||
Horsfall and coworkers have also showcased the synthesis of biogenic Pd(0) NPs generated by Desulfovibrio alaskensis G20, a sulfate-reducing bacterium, and their application in ligand-free Suzuki–Miyaura couplings.304 The preparation of these NPs (DaPdNPs) involves the anaerobic cultures of D alaskensis G20, grown with Na2PdCl4 as Pd salt for 20 h at 30 °C. The NPs were then isolated via centrifugation with a 97% isolated yield. X-ray diffraction (XRD) analysis confirmed the presence of Pd in a zero oxidation state in Pd NPs. The activity of these NPs was demonstrated on Suzuki–Miyaura cross-coupling with low Pd loading (0.5 mol%) in micelles of TPGS-1000-M, resulting in access to C–C coupled products with good-to-excellent yields (Scheme 35B).
1.7. Organic solvent-free couplings in aqueous medium
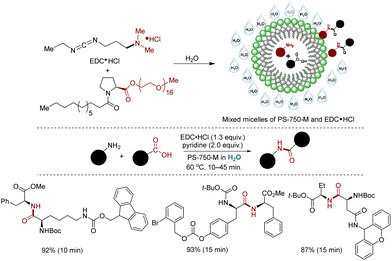
The primary critique of micellar catalysis lies in the continued requirement of organic as an additive or during workup processes.137,138 Simply replacing the organic solvent with water as a reaction medium while still relying on organic solvents for product extraction and purification raises sustainability concerns. A study by GSK on amide coupling reactions highlighted that approximately 80% of waste generation stems from organic solvents, with a significant portion attributed to workup and purification solvents, notably dichloromethane.305,306 However, recent advancements in micellar catalysis methodologies have tackled this issue by minimizing or eliminating the need for organic solvents in product extractions.307–310 One notable green alternative, devised by Handa and coworkers, involves a rapid amide coupling reaction within an aqueous micellar environment, obviating the need for organic solvents (reaction time: 10 to 45 minutes).310 A standout feature of this approach is its solvent-free process, with the final amide products easily precipitating from the micellar medium and separable through filtration. By employing the inexpensive and safe coupling reagent EDC (3-dimethylamino-propyl)-ethyl-carbodiimide, water-soluble urea byproducts are formed, facilitating straightforward product separation. This method demonstrates broad applicability across numerous amino acids, exhibiting excellent functional group and protecting group tolerances, scalability, and extension to bioactive molecule synthesis with impressive isolated yields. However, another notable advantage is the absence of epimerization in the final product, a common drawback in many amide couplings. Mechanistic studies unveiled the pivotal role of EDC·HCl as an ionic amphiphile that forms mixed micelles with the surfactant PS-750-M. These mixed micelles, rich in EDC, efficiently activate acids for coupling reactions, thus accounting for the accelerated reaction rates (Scheme 36).311 Nonetheless, this amidation strategy may not always be favored, particularly in pharmaceutical industry processes where regulatory control points are necessary, as a pyridine base is required in this synthetic process.The organic solvent-free technology was expanded to Cu-catalyzed cycloaddition reactions. In this approach, the shielding effect and hydrophobic pockets of HPMC were leveraged to produce highly stable Cu(I) NPs without the need for reducing agents or supports.307 Stirring CuI in aqueous HPMC at 60 °C for 10 minutes led to the formation of ultrasmall Cu(I) NPs of an average size of 3.6 nm, as confirmed with HRTEM analysis. XPS analysis further validates the presence of Cu in +1 oxidation state. Once generated, these Cu(I) NPs were utilized for ultrafast cycloaddition reactions using benzyl bromide, alkynes, and NaN3. Upon reaction completion, the triazole product precipitated and was easily separated by filtration. This methodology demonstrated broad substrate compatibility with short reaction times of 10–45 minutes and excellent isolated yields. HPLC analysis corroborated the high purity of the final products, reaching up to 98% (Scheme 37).307
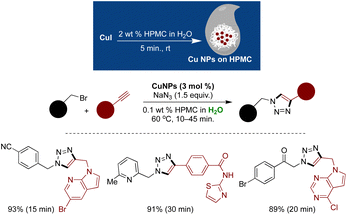 | ||
| Scheme 37 Cu(I) NPs stabilized on HPMC for fast and completely organic-solvent free cycloadditions in aqueous micellar medium. | ||
1.8. Other sustainable organic transformations in micellar medium
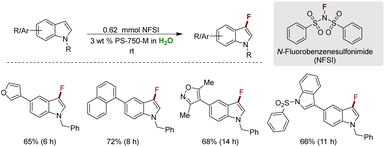
In 2019, Handa and coworkers reported a method for monofluorination of indoles, operating under mild micellar conditions.322 This approach capitalizes on the protective influence of micelles, effectively shielding the indole substrate from undesired side reactions such as difluorinations or unwanted oxidations. Key to this process is the utilization of N-fluorobenzenesulfonimide (NFSI) as the electrophilic fluorine source within the micelles of PS-750-M. The methodology exhibited broad applicability, accommodating a diverse array of functional groups including aldehydes, nitro, ester, ether, and sulfonyl. It extends its versatility to the monofluorination of arenes with exceptional selectivity. Through control experiments, a radical mechanism was elucidated and subsequently corroborated by trapping the radical intermediate using butylated hydroxytoluene (BHT). Importantly, this method demonstrates scalability to gram-scale production, with the added benefit of recyclability of the aqueous mixture up to three times, boasting an overall E-factor of 6.2 (Scheme 38).323
In 2015, Lipshutz and coworkers used an approach utilizing mild aqueous micellar conditions for SNAr reactions, accommodating oxygen, nitrogen, and sulfur nucleophiles.325 The method employed nanomicelles of TPGS-750-M, enabling SNAr reaction across a diverse spectrum of substrates, including various heterocycles, while exhibiting remarkable tolerance towards functional groups. A comparative analysis with DMF as the organic solvent underscored the superiority of the micellar medium over conventional organic solvent-based methodologies. It is important to note that the method was not been demonstrated with weak anionic nucleophiles, such as sulfinate salts (Scheme 39).
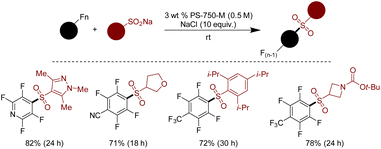
In 2018, Handa and coworkers reported the sulfonylation of perfluoroarenes using sulfinate salts, culminating in synthesizing valued (hetero)arylsulfone scaffolds.326 The driving force for this transformation was the polar inner core of proline-based surfactant PS-750-M, which adeptly accommodates the polar sulfone moieties, facilitating their effective interaction with perfluoroarenes. Adding acetone as a co-solvent ensures solubility, while NaCl acting as a supplementary agent, further propels the sulfinate salts into the micelles. This micellar technology showcased versatility across a broad range of substrates, including heterocyclic moieties, delivering products with yields ranging from good to excellent. The recycling of the reaction medium was demonstrated with E-factor of approximately zero (Scheme 40).
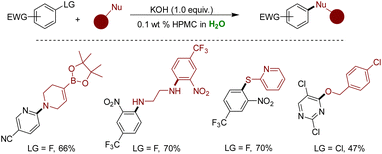
Recently, the Braje and Handa group delved into utilizing benign HPMC for the SNAr reaction, presenting a scalable, versatile, and efficient method for constructing C–N, C–O, and C–S bonds.327 Their work revealed remarkable functional group tolerance under optimized conditions, yielding high yields in short reaction times. Notably, base-sensitive functional groups, such as oxetanes, esters, vinyl boronic acids, and alkyl boronic esters, were well accommodated under these conditions. Additionally, the authors showcased a direct application of this methodology in synthesizing bioactive molecules. However, it's worth noting that the approach wasn't universally applicable for O-nucleophiles (Scheme 41).
1.9. Emulsion polymerization
The emulsion polymerization method, a widely utilized technique employing a micellar medium, is instrumental in producing synthetic latexes and resins by polymerizing monomers in water.328–330 These polymeric materials find application in diverse fields, such as adhesives, paints and coatings, and textiles.328,329 The process starts by emulsifying the monomer, typically insoluble in water, into a solution with a surfactant. Upon reaching a critical concentration, known as the CMC, the surfactant forms micelles that encapsulate the monomer within their hydrophobic cores. However, only a fraction of the monomer enters the micelle's interior, with the majority dispersed as droplets stabilized by surfactant molecules on their surfaces. The introduction of a water-soluble radical initiator triggers polymerization within the micellar interior. Micelles serve as a focal point for the organic monomer and water-soluble initiator, offering a preferable reaction site due to their high monomer concentration and surface-to-volume ratio. As the polymerization progresses, monomer diffusion from droplets aids in maintaining a consistent monomer concentration within the micelles.Compared to monomer droplets, micelles serve as favored reaction sites due to their elevated monomer concentration and superior surface-to-volume ratio.329–331 Moreover, the initiators utilized remain insoluble in organic monomers. During the polymerization process, monomers diffuse from droplets to maintain a consistent concentration within the micelles. As the polymerization progresses, monomer droplet size gradually diminishes until complete disappearance (Fig. 5). A significant advantage of surfactant employment is the compartmentalization of propagating chains, effectively hindering termination steps. Consequently, this leads to accelerated polymerization rates and the production of high molecular weight polymers.330 Another benefit lies in substituting organic solvents with water as a dispersion medium, facilitating excellent heat dissipation during polymerization.332 However, drawbacks include challenges associated with surfactant removal from the polymer and the energy-intensive process of water removal from polymers.
2. Future directions
Sustainability in organic synthesis has emerged as a pivotal challenge for the chemistry community, driven by increasing global awareness and a concerted effort to reduce waste generation.10,38,39,207,333 The past decade has witnessed significant strides in integrating green chemistry principles, encompassing strategies such as enhancing atom economy, devising alternative synthetic routes for feedstocks, promoting sustainable biocatalysis, utilizing eco-friendly solvents—preferably water—designing safer chemicals, and prioritizing waste management with a focus on recyclability.10,17,21,23,333,334 Additionally, the burgeoning field of nanotechnology holds promise for revolutionizing synthetic chemistry, mainly through utilizing novel NPs catalysis, which can offer enhanced efficiency and selectivity compared to traditional methodologies.212,266 Leveraging active metals in aqueous micellar conditions presents a viable option;321 however, challenges such as low reactivity and selectivity, particularly with non-precious metals, must be addressed.228 Advancements towards achieving catalysis at the parts per million level using precious metals like Pd in aqueous media represent significant strides toward sustainability.259,262,264,265,269,335The integration of organometallic catalysts with designer surfactants has yielded promising outcomes in micellar catalysis,137,138,185 with further potential seen in extending micellar catalysis with nanocatalysis to address environmental concerns.169 Notably, the recyclability of catalysts and reaction media, alongside the enhanced stability of NPs within the micellar core, presents exciting prospects for reducing toxic-organic waste and unlocking unique reactivities unattainable in organic solvents.135,184,267 However, it's imperative to consider potential risks associated with metal contamination of water in aqueous chemistry, as well as the toxicity of designer surfactants.162,336 Careful attention to surfactant molecules’ biodegradability and toxicity profiles is essential in their design. Notably, some of the commercially available surfactants, like alkyl benzene sulfonate-based anionic surfactants, quaternary ammonium ethoxylated, and alcohol ethoxylates, cause harmful effects on aquatic/terrestrial ecosystems.162,337 Also, PEG ethers are suspected to impact skin toxicity significantly.338 Therefore, while designing a surfactant molecule, its biodegradability and toxicity should be carefully considered. The Lipshutz group has tackled the problem of toxicity caused by PEG ethers by creating a surfactant known as Savie,183 which is based on polysarcosine and vitamin E. However, any further modifications to this surfactant must maintain its necessary benign properties while addressing its toxicity concerns to avoid potential toxicological issues. Notably, due to the high solubility profile, toxic contaminants or organic pollutants are highly soluble in micellar solutions resulting in the need to tackle wastewater activities professionally.339 Furthermore, effective management of wastewater activities is crucial, with Novartis presenting practical strategies tailored to TPGS-750-M, which can be adapted for other wastewater systems—various approaches, including physiochemical processes, membrane filtration, and electrocoagulation, merit assessment for wastewater pre-treatment. Addressing sustainability challenges in organic synthesis necessitates a comprehensive and multi-faceted approach, integrating innovative technologies, responsible design practices, and proactive waste management strategies.162
Micellar catalysis, often presented as an organic solvent-free technique, isn't entirely devoid of solvents. In fact, the process of isolating and purifying products using organic solvents generates significantly more waste, ranging from 10 to 30 times, than the reaction medium itself. Hence, the imperative to develop methodologies that eschew organic solvents entirely from synthesis is paramount. Within our research group, we've identified several technologies where product isolation can be achieved solely through filtration, completely bypassing the need for organic solvents at any stage of the synthesis process.307,310,311 These methodologies represent a paradigm shift towards what we term as “truly organic solvent-free” practices. However, the product filtration approach is not applicable to every transformation.
Moreover, the concern regarding solvent use can be mitigated by integrating novel techniques to minimize environmental footprints. One such avenue lies in advancing benign chiral surfactants for stereo-controlled synthesis. Additionally, there's burgeoning interest in engineering micelles with extended conjugation for applications in photocatalysis, presenting exciting prospects for enhancing reaction efficiency.
Mechanochemistry stands out as another highly promising and sustainable methodology. It harnesses mechanical force, such as grinding, milling, and pounding, to drive chemical and physicochemical transformations.340–343 Notably, it offers distinct advantages, including the capability to circumvent solubility issues of reactants and the feasibility of executing reactions in a solvent-free environment.342,343 Nonetheless, there remains a significant knowledge gap concerning the physiochemical intricacies of mechanochemistry, particularly its interplay with thermodynamics and kinetics. Further investigations are imperative to elucidate the correlation between the applied force magnitude and specific chemical transformations, thereby averting uncontrolled side reactions or agglomeration phenomena.
3. Conclusions
Over the past decade, there has been a notable surge in the adoption of greener chemical processes. Yet, significant challenges persist, particularly regarding the widespread integration of sustainable technologies within chemical industries. One promising avenue that has emerged is the utilization of chemistry in water facilitated by designer surfactants. This approach has demonstrated cost and energy efficiency, particularly in synthesizing pharmaceutical intermediates. Another noteworthy advancement involves the application of organometallic catalysis within nanomicelles, showcasing heightened reactivity and exceptional selectivity across various cross-coupling reactions. Micellar catalysis leveraging heterogeneous NPs has also unlocked novel reactivities with superior selectivities. Within this framework, the micellar core serves as a stabilizing or capping agent, yielding stable and highly active NPs with uniform sizes, thereby streamlining the otherwise complex process of nanocatalyst synthesis. As this field continues to evolve, it is poised to garner broader recognition and adoption within the fine chemicals production landscape, heralding a promising future for sustainable chemistry practices.Author contributions
The manuscript was written through the contributions of all authors.Conflicts of interest
There are no conflicts to declare.Acknowledgements
S. H. is grateful to the U.S. National Science Foundation for financial support (CHE 2044778, 2345856). We warmly acknowledge the partial financial support from Novartis Institutes for Biomedical Research.References
- R. Patnaik , IOP Conf. Ser. Earth Environ. Sci., 2018, 120, 12016.
- O. A. Aluko, E. E. Osei Opoku and M. Ibrahim, J. Environ. Manage., 2021, 281, 111892 CrossRef PubMed.
- Y. Li, S. Zhou, Z. Jia, L. Ge, L. Mei, X. Sui, X. Wang, B. Li, J. Wang and S. Wu, Int. J. Environ. Res. Public Health, 2018, 15 Search PubMed.
- K.-H. Kim, E. Kabir and S. Ara Jahan, J. Environ. Sci. Health, Part C: Environ. Carcinog. Ecotoxicol. Rev., 2014, 32, 299–318 CrossRef PubMed.
- For details, see the United Nations report on climate change, (last accessed 25th January, 2024), https://www.un.org/en/climatechange/reports.
- I. Manisalidis, E. Stavropoulou, A. Stavropoulos and E. Bezirtzoglou, Front. Public Health, 2020, 8 Search PubMed.
- R. Fuller, P. J. Landrigan, K. Balakrishnan, G. Bathan, S. Bose-O'Reilly, M. Brauer, J. Caravanos, T. Chiles, A. Cohen, L. Corra, M. Cropper, G. Ferraro, J. Hanna, D. Hanrahan, H. Hu, D. Hunter, G. Janata, R. Kupka, B. Lanphear, M. Lichtveld, K. Martin, A. Mustapha, E. Sanchez-Triana, K. Sandilya, L. Schaefli, J. Shaw, J. Seddon, W. Suk, M. M. Téllez-Rojo and C. Yan, Lancet Planet. Health, 2022, 6, e535–e547 CrossRef PubMed.
- K. N. Ganesh, D. Zhang, S. J. Miller, K. Rossen, P. J. Chirik, M. C. Kozlowski, J. B. Zimmerman, B. W. Brooks, P. E. Savage, D. T. Allen and A. M. Voutchkova-Kostal, Environ. Sci. Technol. Lett., 2021, 8, 487–491 CrossRef CAS.
- I. T. Horváth and P. T. Anastas, Chem. Rev., 2007, 107, 2169–2173 CrossRef PubMed.
- S. Sharma, J. Das, W. Braje and S. Handa, ChemSusChem, 2020, 13, 2859–2875 CrossRef CAS PubMed.
- Ed. G. Brundland, Report of the World Commission on Environment and Development: Our Common Future, https://sustainabledevelopment.un.org/content/documents/5987our-common-future.pdf. (Last accessed 31 March, 2024).
- Earth Summit, Rio de Janeiro, https://www.eea.europa.eu/help/glossary/chm-biodiversity/earth-summit-rio-de-janeiro#:~:text=Popularly%20known%20as%20the%20%E2%80%99Earth,Stockholm%2C%20Sweden%2C%20in%201972. (Last accessed 31 March, 2024).
- R. Höfer, in Sustainable Solutions for Modern Economies, ed. R. Höfer, The Royal Society of Chemistry, 2009 Search PubMed.
- CEFIC, Responsible Care: An ethical framework towards safe chemicals management and performance excellence, https://cefic.org/responsible-care/. (Last accessed 31 March, 2024).
- Responsible Care Global Charter, https://icca-chem.org/resources/responsible-care-global-charter/. (Last accessed 31 March, 2024).
- US Senate. Pollution Prevention Act of 1990, https://www.epw.senate.gov/PPA90. (Last accessed 31st March, 2024).
- B. A. de Marco, B. S. Rechelo, E. G. Tótoli, A. C. Kogawa and H. R. N. Salgado, Saudi Pharm. J., 2019, 27, 1–8 CrossRef PubMed.
- Please visit, https://www.acs.org/greenchemistry/what-is-green-chemistry/history-of-green-chemistry.html. (Last accessed 31st March 2024).
- Gordon Conference 1996, https://www.grc.org/environmentally-benign-organic-synthesis-conference/1996/. (Last accessed 31st March 2024).
- P. Anastas, R. Kazlauskas and G. Sheldrake, Green Chem., 2006, 8, 677–678 RSC.
- P. T. Anastas and J. C. Warner, Green Chemistry: Theory and Practice, Oxford University Press, Oxford, 1998 Search PubMed.
- 12 Principles of Green Chemistry, https://www.acs.org/greenchemistry/principles/12-principles-of-green-chemistry.html. (Last accessed 31st March 2024).
- P. T. Anastas and N. Eghbali, Chem. Soc. Rev., 2010, 39, 301–312 RSC.
- J. B. Manley, P. T. Anastas and B. W. Cue, J. Cleaner Prod., 2008, 16, 743–750 CrossRef.
- J. H. Clark, Green Chem., 2006, 8, 17–21 RSC.
- Green Chemistry Webinars and Videos, https://www.acs.org/greenchemistry/students-educators/webinar-and-videos.html. (Last accessed 31st March 2024).
- ACS Summer School on Green Chemistry & Sustainable Energy, https://www.acs.org/greenchemistry/students-educators/summer-school.html. (Last accessed 31st March 2024).
- ACS Green Chemistry Academic Programs, https://www.acs.org/greenchemistry/students-educators/academicprograms.html. (Last accessed 31st March 2024).
- A. Iles and M. J. Mulvihill, Environ. Sci. Technol., 2012, 46, 5643–5649 CrossRef CAS PubMed.
- ACS Green Chemistry Education, https://www.acs.org/greenchemistry/students-educators.html. (Last accessed 31st March 2024).
- Beyond Benign. Green Chemistry Commitment, https://www.beyondbenign.org/he-green-chemistry-commitment/. (Last accessed 31st March 2024).
- ACS Green Chemistry Textbooks & Printed Resources, https://www.acs.org/greenchemistry/students-educators/printed-resources.html. (Last accessed 31st March 2024).
- B. W. Cue and J. Zhang, Green Chem. Lett. Rev., 2009, 2, 193–211 CrossRef CAS.
- N. Winterton, Clean Technol. Environ. Policy, 2021, 23, 2499–2522 CrossRef PubMed.
- E. S. Beach, Z. Cui and P. T. Anastas, Energy Environ. Sci., 2009, 2, 1038–1049 RSC.
- M. J. Raymond, C. S. Slater and M. J. Savelski, Green Chem., 2010, 12, 1826–1834 RSC.
- P. J. Dunn, A. S. Wells and M. T. Williams, in Green Chemistry in the Pharmaceutical Industry, 2010, pp. 333–355 Search PubMed.
- S. Koenig, Scalable Green Chemistry: Case Studies from the Pharmaceutical Industry, CRC Press, 2013 Search PubMed.
- W. Zhao, Natl. Sci. Rev., 2018, 5, 953–956 CrossRef CAS.
- S. Kar, H. Sanderson, K. Roy, E. Benfenati and J. Leszczynski, Chem. Rev., 2022, 122, 3637–3710 CrossRef CAS PubMed.
- F. Roschangar, R. A. Sheldon and C. H. Senanayake, Green Chem., 2015, 17, 752–768 RSC.
- Chemical & Engineering News Archive, 2015, 93, 32–33.
- For details, visit the EPA website, https://www.epa.gov/trinationalanalysis/source-reduction-activities-0. (Last accessed 31st March 2024).
- P. J. Dunn, S. Galvin and K. Hettenbach, Green Chem., 2004, 6, 43–48 RSC.
- H. W. Hamilton, D. F. Ortwine, D. F. Worth and J. A. Bristol, J. Med. Chem., 1987, 30, 91–96 CrossRef CAS PubMed.
- N. J. Stuart and A. S. Sanders, United States Patent, 3385886, 1961 Search PubMed.
- Presidential Green Chemistry Challenge: 1997 Greener Synthetic Pathways Award, https://www.epa.gov/greenchemistry/presidential-green-chemistry-challenge-1997-greener-synthetic-pathways-award. (Last accessed 31st March 2024).
- A. P. Dicks, Green Organic Chemistry in Lecture and Laboratory, CRC Press, 2011 Search PubMed.
- W. Yan, G. Zhang, J. Wang, M. Liu, Y. Sun, Z. Zhou, W. Zhang, S. Zhang, X. Xu, J. Shen and X. Jin, Front. Chem., 2020, 8 Search PubMed.
- W. Deng, L. Yan, B. Wang, Q. Zhang, H. Song, S. Wang, Q. Zhang and Y. Wang, Angew. Chem., Int. Ed., 2021, 60, 4712–4719 CrossRef CAS PubMed.
- Adipic Acid Market – Global Industry Analysis, Size, Share, Growth, Trends, and Forecast, 2022–2028, https://www.marketwatch.com/press-release/adipic-acid-market–global-industry-analysis-size-share-growth-trends-and-forecast-2022–2028-2022-12-01. (Last accessed 31st March 2024).
- A. Castellan, J. C. J. Bart and S. Cavallaro, Catal. Today, 1991, 9, 237–254 CrossRef CAS.
- M. J. Gilkey, A. v. Mironenko, D. G. Vlachos and B. Xu, ACS Catal., 2017, 7, 6619–6634 CrossRef CAS.
- T. J. Griffis, Z. Chen, J. M. Baker, J. D. Wood, D. B. Millet, X. Lee, R. T. Venterea and P. A. Turner, Proc. Natl. Acad. Sci. U. S. A., 2017, 114, 12081–12085 CrossRef CAS PubMed.
- S. Chatterjee, P. Bhanja, L. Paul, M. Ali and A. Bhaumik, Dalton Trans., 2018, 47, 791–798 RSC.
- F. Cavani, L. Ferroni, A. Frattini, C. Lucarelli, A. Mazzini, K. Raabova, S. Alini, P. Accorinti and P. Babini, Appl. Catal., A, 2011, 391, 118–124 CrossRef CAS.
- R. B. N. Baig and R. S. Varma, Chem. Soc. Rev., 2012, 41, 1559–1584 RSC.
- J. Luo, Y. Huang, B. Ding, P. Wang, X. Geng, J. Zhang and Y. Wei, Catalysts, 2018, 8 CAS.
- T. P. Fedorchuk, A. N. Khusnutdinova, E. Evdokimova, R. Flick, R. Di Leo, P. Stogios, A. Savchenko and A. F. Yakunin, J. Am. Chem. Soc., 2020, 142, 1038–1048 CrossRef CAS PubMed.
- K. Raj, S. Partow, K. Correia, A. N. Khusnutdinova, A. F. Yakunin and R. Mahadevan, Metab. Eng. Commun., 2018, 6, 28–32 CrossRef PubMed.
- An update on biobased adipic acid synthesis, https://biorrefineria.blogspot.com/2021/05/Biobased-adipic-acid.html. (Last accessed 31st March 2024).
- Asahi Kasei partners with Genomatica on renewably sourced nylon-6,6, https://www.genomatica.com/news-content/genomatica-and-asahi-kasei-nylon-partnership/. (Last accessed 31st March 2024).
- Toray: From sugar to bio-based PA 66, https://www.textiletechnology.net/fibers/news/toray-from-sugar-to-bio-based-pa-66-32758. (Last accessed 31st March 2024).
- G. MacQueen, L. Born and M. Steiner, CNS Drug Rev., 2001, 7, 1–24 CrossRef CAS PubMed.
- A. L. McRae and K. T. Brady, Expert Opin. Pharmacother., 2001, 2, 883–892 CrossRef CAS PubMed.
- W. M. Welch, A. R. Kraska, R. Sarges and B. K. Koe, J. Med. Chem., 1984, 27, 1508–1515 CrossRef CAS PubMed.
- G. P. Taber, D. M. Pfisterer and J. C. Colberg, Org. Process Res. Dev., 2004, 8, 385–388 CrossRef CAS.
- Presidential Green Chemistry Challenge: 2002 Greener Synthetic Pathways Award, https://www.epa.gov/greenchemistry/presidential-green-chemistry-challenge-2002-greener-synthetic-pathways-award. (Last accessed 31st March 2024).
- G. J. M. Herschbach, C. P. van der Beek and P. W. M. van Dijck, The penicillins: properties, biosynthesis, and fermentation, Marcel Dekker AG, 1984 Search PubMed.
- G. J. M. Hersbach, Antonie van Leeuwenhoek, 1983, 49, 93–94 CrossRef.
- C. P. van der Beek and J. A. Roels, Antonie van Leeuwenhoek, 1984, 50, 625–639 CrossRef CAS PubMed.
- M. A. Wegman, M. H. A. Janssen, F. van Rantwijk and R. A. Sheldon, Adv. Synth. Catal., 2001, 343, 559–576 CrossRef CAS.
- H. Baer, M. Bergamo, A. Forlin, L. H. Pottenger and J. Lindner, in Ullmann's Encyclopedia of Industrial Chemistry, 2012 Search PubMed.
- Z. Zhao, J. Jiang and F. Wang, J. Energy Chem., 2021, 56, 193–202 CrossRef CAS.
- E. M. Jorge, Chlorohydrin Process, U.S. Patent, 6043400A, 1996 Search PubMed.
- P. L. Short, Chem. Eng. News, 2009, 87, 21 Search PubMed.
- P. Bassler, M. Weidenbach and H. Goebbel, Chem. Eng. Trans., 2010, 21, 571–576 Search PubMed , SE-Research articles.
- M. G. Clerici , Oil Gas European Magazine, 2006, 122, 77–82.
- Presidential Green Chemistry Challenge: 2010 Greener Synthetic Pathways Award, https://www.epa.gov/greenchemistry/presidential-green-chemistry-challenge-2010-greener-synthetic-pathways-award. (Last accessed 31st March 2024).
- T. W. Abraham and R. Hofer, Lipid-Based Polymer Building Blocks and Polymers, ed. K. Matyjaszewski and M. Moller, Elsevier, Amsterdam, 2012, pp. 15–57 Search PubMed.
- Agrobiobase: Epichlorohydrin (via EPICEROL process), https://www.agrobiobase.com/en/database/bioproducts/plastics-composites-rubber/epichlorohydrin-via-epicerol-process. (Last accessed 31st March 2024).
- B. M. Bell, J. R. Briggs, R. M. Campbell, S. M. Chambers, P. D. Gaarenstroom, J. G. Hippler, B. D. Hook, K. Kearns, J. M. Kenney, W. J. Kruper, D. J. Schreck, C. N. Theriault and C. P. Wolfe, Clean, 2008, 36, 657–661 CAS.
- J. E. McGrath, M. A. Hickner and R. Hofer, Introduction: Polymers for a Sustainable Environment and Green Energy, ed. K. Matyjaszewski and M. Moller, Elsevier, Amsterdam, 2012, pp. 1–3 Search PubMed.
- S. Abou-Shehada, J. H. Clark, G. Paggiola and J. Sherwood, Chem. Eng. Process., 2016, 99, 88–96 CrossRef CAS.
- J. H. Clark, T. J. Farmer, A. J. Hunt and J. Sherwood, Int. J. Mol. Sci., 2015, 16, 17101–17159 CrossRef CAS PubMed.
- S. W. Breeden, J. H. Clark, D. J. Macquarrie and J. R. Sherwood, Green solvents, John Wiley and Sons, Chichester, UK, 2012 Search PubMed.
- T. Sahoo, J. Panda, J. Sahu, D. Sarangi, S. K. Sahoo, B. B. Nanda and R. Sahu, Curr. Org. Synth., 2020, 17, 426–439 CrossRef CAS PubMed.
- F. M. Kerton and R. Marriott, Altern. Solvents Green Chem, 2013, pp. 31–50 Search PubMed.
- International Labour Organization (1971) Benzene convention: convention concerning protection against hazards of poisoning arising from benzene, https://www.ilo.org/dyn/normlex/en/f?p=NORMLEXPUB:12100:0::NO::P12100_ILO_CODE:C136. (Last accessed 31st March 2024).
- World Health Organization (2015) IARC monographs on the evaluation of carcinogenic risks to human, https://monographs.iarc.fr/ENG/Classification/index.php. (Last accessed 31st March 2024).
- United Nations Environment Programme (1987) The Montreal protocol on substances that deplete the ozone layer, https://ozone.unep.org/en/treaties-and-decisions/montreal-protocol-substances-deplete-ozone-layer. (Last accessed 31st March 2024).
- Q. Liang, P. A. Newman, J. S. Daniel, S. Reimann, B. D. Hall, G. Dutton and L. J. M. Kuijpers, Geophys. Res. Lett., 2014, 41, 5307–5315 CrossRef CAS.
- Finnish Safety and Chemicals Agency. (2013) Toluene substance evaluation report (under REACH), https://echa.europa.eu/documents/10162/a58633d6-1620-4764-b3bf-6308cad42e8b. (Last accessed 31st March 2024).
- European Chemicals Agency (ECHA) (2015) Classification and labelling inventory, https://echa.europa.eu/information-on-chemicals/cl-inventory-database. (Last accessed 31st March 2024).
- R. Hossaini, M. P. Chipperfield, S. A. Montzka, A. Rap, S. Dhomse and W. Feng, Nat. Geosci., 2015, 8, 186–190 CrossRef CAS.
- European Chemicals Agency (ECHA) (2015) Guidance on REACH, https://echa.europa.eu/guidance-documents/guidance-on-reach. (Last accessed 31st March 2024).
- European Chemicals Agency (2015) List of restrictions, https://echa.europa.eu/addressing-chemicals-of-concern/restrictions/list-of-restrictions. (Last accessed 31st March 2024).
- European Chemicals Agency (ECHA) (2015) Candidate list of substances of very high concern for authorisation, https://echa.europa.eu/candidate-list-table. (Last accessed 31st March 2024).
- Please visit, https://echa.europa.eu/view-article/-/journal_content/title/9109026-58. (Last accessed 31st March 2024).
- R. K. Henderson, C. Jiménez-González, D. J. C. Constable, S. R. Alston, G. G. A. Inglis, G. Fisher, J. Sherwood, S. P. Binks and A. D. Curzons, Green Chem., 2011, 13, 854–862 RSC.
- D. Prat, O. Pardigon, H.-W. Flemming, S. Letestu, V. Ducandas, P. Isnard, E. Guntrum, T. Senac, S. Ruisseau, P. Cruciani and P. Hosek, Org. Process Res. Dev., 2013, 17, 1517–1525 CrossRef CAS.
- K. Alfonsi, J. Colberg, P. J. Dunn, T. Fevig, S. Jennings, T. A. Johnson, H. P. Kleine, C. Knight, M. A. Nagy, D. A. Perry and M. Stefaniak, Green Chem., 2008, 10, 31–36 RSC.
- F. P. Byrne, S. Jin, G. Paggiola, T. H. M. Petchey, J. H. Clark, T. J. Farmer, A. J. Hunt, C. Robert McElroy and J. Sherwood, Sustainable Chem. Processes, 2016, 4, 7 CrossRef.
- American Chemical Society (ACS) (2015) The ACS GCI pharmaceutical roundtable solvent selection guide, https://www.acs.org/content/acs/en/greenchemistry/research-innovation/research-topics/solvents.html. (Last accessed 31st March 2024).
- J. B. Manley and C. R. Hargreaves, Collaboration to deliver a solvent selection guide for the pharmaceutical industry. ACS GCI pharmaceutical roundtable, https://www.acs.org/content/dam/acsorg/greenchemistry/industriainnovation/roundtable/solvent-selection-guide.pdf. (Last accessed 31st March 2024).
- D. H. Adam, M. N. S. Hasibuan, R. Syahputra and L. H. Pasaribu, Int. J. Sci. Technol. Res., 2020, 09, 471–473 Search PubMed.
- Pharma goes green to cut costs | News – Chemistry World, https://www.chemistryworld.com/news/pharma-goes-green-to-cut-costs/3003155.article. (Last accessed 31st March 2024).
- K. R. Ryan and I. C. Plumb, Crit. Rev. Solid State Mater. Sci., 1988, 15, 153–200 CrossRef CAS.
- W. Herbst and K. Hunger, Industrial organic pigments: production, properties, applications, John Wiley & Sons, 2006 Search PubMed.
- S. L. James, C. J. Adams, C. Bolm, D. Braga, P. Collier, T. Friščić, F. Grepioni, K. D. M. Harris, G. Hyett, W. Jones, A. Krebs, J. Mack, L. Maini, A. G. Orpen, I. P. Parkin, W. C. Shearouse, J. W. Steed and D. C. Waddell, Chem. Soc. Rev., 2012, 41, 413–447 RSC.
- M. A. P. Martins, C. P. Frizzo, D. N. Moreira, L. Buriol and P. Machado, Chem. Rev., 2009, 109, 4140–4182 CrossRef CAS PubMed.
- B. Rodríguez, A. Bruckmann, T. Rantanen and C. Bolm, Adv. Synth. Catal., 2007, 349, 2213–2233 CrossRef.
- C. J. Clarke, W.-C. Tu, O. Levers, A. Bröhl and J. P. Hallett, Chem. Rev., 2018, 118, 747–800 CrossRef CAS PubMed.
- C. Capello, U. Fischer and K. Hungerbühler, Green Chem., 2007, 9, 927–934 RSC.
- S. Mallakpour and M. Dinari, Ionic Liquids as Green Solvents: Progress and Prospects, ed. A. Mohammad and D. Inamuddin, Springer Netherlands, Dordrecht, 2012, pp. 1–32 Search PubMed.
- T. Welton, Green Chem., 2011, 13, 225 RSC.
- M. Poliakoff and P. Licence, Philos. Trans. R. Soc., A, 2015, 373, 20150018 CrossRef PubMed.
- Ž. Knez, M. Pantić, D. Cör, Z. Novak and M. Knez Hrnčič, Chem. Eng. Process., 2019, 141, 107532 CrossRef.
- E. L. Smith, A. P. Abbott and K. S. Ryder, Chem. Rev., 2014, 114, 11060–11082 CrossRef CAS PubMed.
- S. S. de Jesus and R. Maciel Filho, Renewable Sustainable Energy Rev., 2022, 157, 112039 CrossRef CAS.
- A. K. Halder and M. N. D. S. Cordeiro, ACS Sustainable Chem. Eng., 2019, 7, 10649–10660 CrossRef CAS.
- J. Wang, S. Zhang, Z. Ma and L. Yan, Green Chem. Eng., 2021, 2, 359–367 CrossRef.
- F. Zhou, Z. Hearne and C.-J. Li, Curr. Opin. Green Sustainable Chem., 2019, 18, 118–123 CrossRef.
- A. Chanda and V. V. Fokin, Chem. Rev., 2009, 109, 725–748 CrossRef CAS PubMed.
- C.-J. Li and L. Chen, Chem. Soc. Rev., 2006, 35, 68–82 RSC.
- M. Cortes-Clerget, J. Yu, J. R. A. Kincaid, P. Walde, F. Gallou and B. H. Lipshutz, Chem. Sci., 2021, 12, 4237–4266 RSC.
- T. Kitanosono, K. Masuda, P. Xu and S. Kobayashi, Chem. Rev., 2018, 118, 679–746 CrossRef CAS PubMed.
- J. E. Klijn and J. B. F. N. Engberts, Nature, 2005, 435, 746–747 CrossRef CAS PubMed.
- B. H. Lipshutz, S. Ghorai and M. Cortes-Clerget, Chem. – Eur. J., 2018, 24, 6672–6695 CrossRef CAS PubMed.
- T. Kitanosono and S. Kobayashi, Chem. – Eur. J., 2020, 26, 9408–9429 CrossRef CAS PubMed.
- D. C. Rideout and R. Breslow, J. Am. Chem. Soc., 1980, 102, 7816–7817 CrossRef CAS.
- C. J. Cramer and D. G. Truhlar, in Structure and Reactivity in Aqueous Solution, American Chemical Society, 1994, vol. 568, p. 1 Search PubMed.
- W. Blokzijl and J. B. F. N. Engberts, Angew. Chem., Int. Ed. Engl., 1993, 32, 1545–1579 CrossRef.
- M.-O. Simon and C.-J. Li, Chem. Soc. Rev., 2012, 41, 1415–1427 RSC.
- B. H. Lipshutz, Curr. Opin. Green Sustainable Chem., 2018, 11, 1–8 CrossRef.
- Y. Zhang, Y. Chen, P. Westerhoff, K. Hristovski and J. C. Crittenden, Water Res., 2008, 42, 2204–2212 CrossRef CAS PubMed.
- T. Shen, S. Zhou, J. Ruan, X. Chen, X. Liu, X. Ge and C. Qian, Adv. Colloid Interface Sci., 2021, 287, 102299 CrossRef CAS PubMed.
- G. La Sorella, G. Strukul and A. Scarso, Green Chem., 2015, 17, 644–683 RSC.
- L.-J. Chen, S.-Y. Lin and C.-C. Huang, J. Phys. Chem. B, 1998, 102, 4350–4356 CrossRef CAS.
- A. Patist, S. G. Oh, R. Leung and D. O. Shah, Colloids Surf., A, 2001, 176, 3–16 CrossRef CAS.
- M. P. Andersson, J. Mol. Liq., 2023, 383, 122169 CrossRef CAS.
- L. S. Romsted, C. A. Bunton and J. Yao, Curr. Opin. Colloid Interface Sci., 1997, 2, 622–628 CrossRef CAS.
- Q. Zhang, X.-Z. Shu, J. M. Lucas, F. D. Toste, G. A. Somorjai and A. P. Alivisatos, Nano Lett., 2014, 14, 379–383 CrossRef CAS PubMed.
- M. Schwarze, Chem. Ing. Tech., 2021, 93, 31–41 CrossRef CAS.
- H. W. Stache, Anionic surfactants: organic chemistry, CRC Press, 1995, vol. 56 Search PubMed.
- D. C. Ghosh, P. K. Sen and B. Pal, J. Phys. Chem. B, 2020, 124, 2048–2059 CrossRef CAS PubMed.
- H. Azira and A. Tazerouti, J. Surfactants Deterg., 2007, 10, 185–190 CrossRef CAS.
- R. Gava, P. Ballestín, A. Prieto, A. Caballero and P. J. Pérez, Chem. Commun., 2019, 55, 11243–11246 RSC.
- E. H. Wanderlind, C. R. Bittencourt, A. M. Manfredi, A. P. Gerola, B. S. Souza, H. D. Fiedler and F. Nome, J. Phys. Org. Chem., 2019, 32, e3837 CrossRef.
- A. B. Mirgorodskaya, E. I. Yackevich, V. v. Syakaev, L. Ya. Zakharova, S. K. Latypov and A. I. Konovalov, J. Chem. Eng. Data, 2012, 57, 3153–3163 CrossRef CAS.
- P. A. Hassan and J. v. Yakhmi, Langmuir, 2000, 16, 7187–7191 CrossRef CAS.
- F. P. Ballistreri, R. M. Toscano, M. E. Amato, A. Pappalardo, C. M. A. Gangemi, S. Spidalieri, R. Puglisi and G. Trusso Sfrazzetto, Catalysts, 2018, 8 Search PubMed.
- D. Goswami, Appl. Biochem. Biotechnol., 2020, 191, 744–762 CrossRef CAS PubMed.
- M. Cortes-Clerget, N. Akporji, J. Zhou, F. Gao, P. Guo, M. Parmentier, F. Gallou, J.-Y. Berthon and B. H. Lipshutz, Nat. Commun., 2019, 10, 2169 CrossRef PubMed.
- P. Klumphu and B. H. Lipshutz, J. Org. Chem., 2014, 79, 888–900 CrossRef CAS PubMed.
- M. Bu, G. Lu, J. Jiang and C. Cai, Catal. Sci. Technol., 2018, 8, 3728–3732 RSC.
- X. Hao, Z. Xu, H. Lu, X. Dai, T. Yang, X. Lin and F. Ren, Org. Lett., 2015, 17, 3382–3385 CrossRef CAS.
- B. Zhang, T. Liu, Y. Bian, T. Lu and J. Feng, ACS Sustainable Chem. Eng., 2018, 6, 2651–2655 CrossRef CAS.
- X.-H. Li, C. Mi, X.-H. Liao and X.-G. Meng, Catal. Lett., 2017, 147, 2508–2514 CrossRef CAS.
- Regulations in the European Union for the Use of Triton X-100 in the Pharmaceutical Industry, https://www.bdo.com/insights/industries/life-sciences/regulations-in-the-european-union-for-the-use-of-triton-x-100-in-the-pharmaceutical-industry. (Last accessed 31st March 2024).
- R. Ravindran, S. Juliet, A. K. K. Gopalan, A. K. Kavalimakkil, S. A. Ramankutty, S. N. Nair, P. M. Narayanan and S. Ghosh, J. Parasit. Dis., 2011, 35, 237–239 CrossRef PubMed.
- S. O. Badmus, H. K. Amusa, T. A. Oyehan and T. A. Saleh, Environ. Sci. Pollut. Res., 2021, 28, 62085–62104 CrossRef CAS PubMed.
- X. Zhang, A. F. Cardozo, S. Chen, W. Zhang, C. Julcour, M. Lansalot, J.-F. Blanco, F. Gayet, H. Delmas, B. Charleux, E. Manoury, F. D'Agosto and R. Poli, Chem. – Eur. J., 2014, 20, 15505–15517 CrossRef CAS PubMed.
- F. Fabris, M. Illner, J.-U. Repke, A. Scarso and M. Schwarze, Molecules, 2023, 28 Search PubMed.
- N. Compagno, R. Profeta and A. Scarso, Curr. Opin. Green Sustainable Chem., 2023, 39, 100729 CrossRef CAS.
- R. Adamik, A. R. Herczegh, I. Varga, Z. May and Z. Novák, Green Chem., 2023, 25, 3462–3468 RSC.
- N. A. Isley, Utilizing Micellar Catalysis for Organic Synthesis: A Desk Reference, https://www.acsgcipr.org/wp-content/uploads/Micelle-catalysis-guide-sigma-aldrich.pdf. (Last accessed 31st March 2024).
- J. F. Rathman, Curr. Opin. Colloid Interface Sci., 1996, 1, 514–518 CrossRef CAS.
- G. Kaur, K. Kaur and S. Handa, Curr. Opin. Green Sustainable Chem., 2022, 38, 100690 CrossRef CAS.
- S. M. K. Reddy, J. Kothandapani, M. Sengan, A. Veerappan and S. Selva Ganesan, Mol. Catal., 2019, 465, 80–86 CrossRef CAS.
- M. P. Andersson, F. Gallou, P. Klumphu, B. S. Takale and B. H. Lipshutz, Chem. – Eur. J., 2018, 24, 6778–6786 CrossRef CAS PubMed.
- B. H. Lipshutz, S. Ghorai, A. R. Abela, R. Moser, T. Nishikata, C. Duplais, A. Krasovskiy, R. D. Gaston and R. C. Gadwood, J. Org. Chem., 2011, 76, 4379–4391 CrossRef CAS PubMed.
- Merck: The Lipshutz Portfolio of Surfactants, https://www.sigmaaldrich.com/US/en/technical-documents/technical-article/chemistry-and-synthesis/cross-coupling/lipshutz-portfolio-surfactants. (Last accessed 31st March 2024).
- B. H. Lipshutz, Synlett, 2021, 32, 1588–1605 CrossRef CAS.
- S. Hazra, F. Gallou and S. Handa, ACS Sustainable Chem. Eng., 2022, 10, 5299–5306 CrossRef CAS.
- D. Ogulu, P. P. Bora, M. Bihani, S. Sharma, T. N. Ansari, A. J. Wilson, J. B. Jasinski, F. Gallou and S. Handa, ACS Appl. Mater. Interfaces, 2022, 14, 6754–6761 CrossRef CAS PubMed.
- S. Sharma, S. Parmar, F. Ibrahim, A. H. Clark, M. Nachtegaal, J. B. Jasinski, F. Gallou, P. M. Kozlowski and S. Handa, Adv. Funct. Mater., 2022, 33, 2204459 CrossRef.
- T. N. Ansari, S. Sharma, S. Hazra, J. B. Jasinski, A. J. Wilson, F. Hicks, D. K. Leahy and S. Handa, JACS Au, 2021, 1, 1506–1513 CrossRef CAS PubMed.
- T. N. Ansari, A. Taussat, A. H. Clark, M. Nachtegaal, S. Plummer, F. Gallou and S. Handa, ACS Catal., 2019, 9, 10389–10397 CrossRef CAS.
- M. Bihani, P. P. Bora, M. Nachtegaal, J. B. Jasinski, S. Plummer, F. Gallou and S. Handa, ACS Catal., 2019, 9, 7520–7526 CrossRef CAS.
- T. N. Ansari, S. Sharma, S. Hazra, F. Hicks, D. K. Leahy and S. Handa, ACS Catal., 2022, 12, 15686–15695 CrossRef CAS.
- U. T. Duong, A. B. Gade, S. Plummer, F. Gallou and S. Handa, ACS Catal., 2019, 9, 10963–10970 CrossRef CAS.
- J. R. A. Kincaid, M. J. Wong, N. Akporji, F. Gallou, D. M. Fialho and B. H. Lipshutz, J. Am. Chem. Soc., 2023, 145, 4266–4278 CrossRef CAS PubMed.
- M. Cortes-Clerget, J. R. A. Kincaid, N. Akporji and B. H. Lipshutz, in Supramolecular Catalysis, 2022, pp. 467–487 Search PubMed.
- A. Steven, Synthesis, 2019, 51, 2632–2647 CrossRef CAS.
- P. Sar, A. Ghosh, A. Scarso and B. Saha, Res. Chem. Intermed., 2019, 45, 6021–6041 CrossRef CAS.
- F. Gallou, N. A. Isley, A. Ganic, U. Onken and M. Parmentier, Green Chem., 2016, 18, 14–19 RSC.
- B. S. Takale, R. R. Thakore, R. Mallarapu, F. Gallou and B. H. Lipshutz, Org. Process Res. Dev., 2020, 24, 101–105 CrossRef CAS.
- H. Mayer, D. Golsch, H. Isak and J. Schröder, U.S. Patent, 7241896B2, 2007 Search PubMed.
- C. Torborg and M. Beller, Adv. Synth. Catal., 2009, 351, 3027–3043 CrossRef CAS.
- G. S. Tria, T. Abrams, J. Baird, H. E. Burks, B. Firestone, L. A. Gaither, L. G. Hamann, G. He, C. A. Kirby, S. Kim, F. Lombardo, K. J. Macchi, D. P. McDonnell, Y. Mishina, J. D. Norris, J. Nunez, C. Springer, Y. Sun, N. M. Thomsen, C. Wang, J. Wang, B. Yu, C.-L. Tiong-Yip and S. Peukert, J. Med. Chem., 2018, 61, 2837–2864 CrossRef CAS PubMed.
- M. Parmentier, M. Wagner, R. Wickendick, M. Baenziger, A. Langlois and F. Gallou, Org. Process Res. Dev., 2020, 24, 1536–1542 CrossRef CAS.
- J. D. Bailey, E. Helbling, A. Mankar, M. Stirling, F. Hicks and D. K. Leahy, Green Chem., 2021, 23, 788–795 RSC.
- C. Chen, L. Zhang, C. Almansa, M. Rosario, M. Cwik, S. K. Balani and R. Lock, Clin. Pharmacol. Drug Dev., 2022, 11, 142–149 CrossRef CAS PubMed.
- B. S. Takale, R. R. Thakore, F. Y. Kong and B. H. Lipshutz, Green Chem., 2019, 21, 6258–6262 RSC.
- N. R. Lee, A. A. Bikovtseva, M. Cortes-Clerget, F. Gallou and B. H. Lipshutz, Org. Lett., 2017, 19, 6518–6521 CrossRef CAS PubMed.
- J. S. Bajwa, M. de L. Cruz, S. K. Dodd, L. M. Waykole and R. Wu, Salts of N-[6-cis-2,6-Dimethylmorpholin-4yl)pyridine-3yl]-2-methyl-4′-(trifluoromethoxy)[1,1′-biphenyl]-3-carboxamide, WO 2010033481A1, 2010 Search PubMed.
- A. M. Linsenmeier and W. M. Braje, Tetrahedron, 2015, 71, 6913–6919 CrossRef CAS.
- B. H. Lipshutz, J. Org. Chem., 2017, 82, 2806–2816 CrossRef CAS PubMed.
- B. Wu, N. Ye, K. Zhao, M. Shi, J. Liao, J. Zhang, W. Chen, X. Li, Y. Han, M. Cortes-Clerget, M. L. Regnier, M. Parmentier, C. Mathes, F. Rampf and F. Gallou, Chem. Commun., 2024, 60, 2349–2352 RSC.
- C. A. Busacca, D. R. Fandrick, J. J. Song and C. H. Senanayake, Adv. Synth. Catal., 2011, 353, 1825–1864 CrossRef CAS.
- A. Behr, in Ullmann's Encyclopaedia of Industrial Chemistry, 2000 Search PubMed.
- O. Deutschmann, H. Knözinger, K. Kochloefl and T. Turek, in Ullmann's Encyclopedia of Industrial Chemistry, 2009 Search PubMed.
- C. M. Friend and B. Xu, Acc. Chem. Res., 2017, 50, 517–521 CrossRef CAS PubMed.
- D. J. Cole-Hamilton, Science, 2003, 299, 1702–1706 CrossRef CAS PubMed.
- C. Copéret, M. Chabanas, R. Petroff Saint-Arroman and J.-M. Basset, Angew. Chem., Int. Ed., 2003, 42, 156–181 CrossRef PubMed.
- R. T. Baker and W. Tumas, Science, 1999, 284, 1477–1479 CrossRef CAS.
- L. Luo, B. Wang, J. Jiang, M. Fitzgerald, Q. Huang, Z. Yu, H. Li, J. Zhang, J. Wei, C. Yang, H. Zhang, L. Dong and S. Chen, Front. Pharmacol., 2021, 11 Search PubMed.
- A. Dandia, S. Parihar, R. Sharma, K. S. Rathore and V. Parewa, Green Sustainable Process for Chemical and Environmental Engineering and Science, ed. Inamuddin, R. Boddula and S. Asiri, Elsevier, 2020, pp. 71–103 Search PubMed.
- R. Narayanan, Green Chem. Lett. Rev., 2012, 5, 707–725 CrossRef CAS.
- R. Santonocito and G. Trusso Sfrazzetto, Green Nanocatalysts in Organic Synthesis, ed. G. Anilkumar and S. Saranya, Springer Singapore, Singapore, 2021, pp. 221–236 Search PubMed.
- L. L. Chng, N. Erathodiyil and J. Y. Ying, Acc. Chem. Res., 2013, 46, 1825–1837 CrossRef CAS PubMed.
- M. Zahmakıran and S. Özkar, Nanoscale, 2011, 3, 3462–3481 RSC.
- J. Faria, M. P. Ruiz and D. E. Resasco, Adv. Synth. Catal., 2010, 352, 2359–2364 CrossRef CAS.
- H. Lee, S. E. Habas, S. Kweskin, D. Butcher, G. A. Somorjai and P. Yang, Angew. Chem., Int. Ed., 2006, 45, 7824–7828 CrossRef CAS PubMed.
- F. D. Guerra, M. F. Attia, D. C. Whitehead and F. Alexis, Molecules, 2018, 23, 1760 CrossRef PubMed.
- O. Myakonkaya, C. Guibert, J. Eastoe and I. Grillo, Langmuir, 2010, 26, 3794–3797 CrossRef CAS PubMed.
- L. D. Pachón and G. Rothenberg, Appl. Organomet. Chem., 2008, 22, 288–299 CrossRef.
- C. C. C. Johansson Seechurn, M. O. Kitching, T. J. Colacot and V. Snieckus, Angew. Chem., Int. Ed., 2012, 51, 5062–5085 CrossRef CAS PubMed.
- R. M. Bullock, Catalysis without precious metals, John Wiley & Sons, 2011 Search PubMed.
- J. D. Hayler, D. K. Leahy and E. M. Simmons, Organometallics, 2019, 38, 36–46 CrossRef CAS.
- Daily Metal Prices, https://www.dailymetalprice.com/. (Last accessed 31st March 2024).
- For details, Please visit, https://matthey.com/products-and-markets/pgms-and-circularity/pgm-management/. (Last accessed 31st March 2024).
- P. A. Lusty and G. A. Gunn, Geol. Soc. London, Spec. Publ., 2015, 393, 265–276 CrossRef.
- P. Kushwaha, Curr. Pharm. Anal., 2021, 17, 960–968 CrossRef CAS.
- E. B. Bauer, Iron Catalysis: Historic Overview and Current Trends, ed. E. Bauer, Springer International Publishing, Cham, 2015, pp. 1–18 Search PubMed.
- J. E. Zweig, D. E. Kim and T. R. Newhouse, Chem. Rev., 2017, 117, 11680–11752 CrossRef CAS PubMed.
- E. P. Beaumier, A. J. Pearce, X. Y. See and I. A. Tonks, Nat. Rev. Chem., 2019, 3, 15–34 CrossRef PubMed.
- M. B. Gawande, A. Goswami, F.-X. Felpin, T. Asefa, X. Huang, R. Silva, X. Zou, R. Zboril and R. S. Varma, Chem. Rev., 2016, 116, 3722–3811 CrossRef CAS PubMed.
- B. C. Ranu, R. Dey, T. Chatterjee and S. Ahammed, ChemSusChem, 2012, 5, 22–44 CrossRef CAS PubMed.
- H. C. Kolb, M. G. Finn and K. B. Sharpless, Angew. Chem., Int. Ed., 2001, 40, 2004–2021 CrossRef CAS PubMed.
- H. C. Kolb and K. B. Sharpless, Drug Discovery Today, 2003, 8, 1128–1137 CrossRef CAS PubMed.
- C. D. Hein, X.-M. Liu and D. Wang, Pharm. Res., 2008, 25, 2216–2230 CrossRef CAS PubMed.
- P. Wu, A. K. Feldman, A. K. Nugent, C. J. Hawker, A. Scheel, B. Voit, J. Pyun, J. M. J. Fréchet, K. B. Sharpless and V. V. Fokin, Angew. Chem., Int. Ed., 2004, 43, 3928–3932 CrossRef CAS PubMed.
- Nobel Prize in Click Chemistry, https://www.nobelprize.org/prizes/chemistry/. (Last accessed 31st March 2024).
- J. E. Hein and V. V. Fokin, Chem. Soc. Rev., 2010, 39, 1302–1315 RSC.
- F. Himo, T. Lovell, R. Hilgraf, V. V. Rostovtsev, L. Noodleman, K. B. Sharpless and V. V. Fokin, J. Am. Chem. Soc., 2005, 127, 210–216 CrossRef CAS PubMed.
- B. H. Lipshutz and B. R. Taft, Angew. Chem., Int. Ed., 2006, 45, 8235–8238 CrossRef CAS PubMed.
- C. Girard, E. Önen, M. Aufort, S. Beauvière, E. Samson and J. Herscovici, Org. Lett., 2006, 8, 1689–1692 CrossRef CAS PubMed.
- D. Clarisse, P. Prakash, V. Geertsen, F. Miserque, E. Gravel and E. Doris, Green Chem., 2017, 19, 3112–3115 RSC.
- P. R. Bagdi, R. S. Basha and A. T. Khan, RSC Adv., 2015, 5, 61337–61344 RSC.
- J. Easmon, G. Pürstinger, K.-S. Thies, G. Heinisch and J. Hofmann, J. Med. Chem., 2006, 49, 6343–6350 CrossRef CAS PubMed.
- T. R. Kau, F. Schroeder, S. Ramaswamy, C. L. Wojciechowski, J. J. Zhao, T. M. Roberts, J. Clardy, W. R. Sellers and P. A. Silver, Cancer Cell, 2003, 4, 463–476 CrossRef CAS PubMed.
- N. Khatun, S. Guin, S. K. Rout and B. K. Patel, RSC Adv., 2014, 4, 10770–10778 RSC.
- U. Duong, T. N. Ansari, S. Parmar, S. Sharma, P. M. Kozlowski, J. B. Jasinski, S. Plummer, F. Gallou and S. Handa, ACS Sustainable Chem. Eng., 2021, 9, 2854–2860 CrossRef CAS.
- I. Bertini, H. B. Gray, S. J. Lippard and J. S. Valentine, Bioinorganic chemistry, University Science Books, 1994 Search PubMed.
- B. Plietker, Iron catalysis: fundamentals and applications, Springer Science & Business Media, 2011, vol. 33 Search PubMed.
- C. Bolm, J. Legros, J. Le Paih and L. Zani, Chem. Rev., 2004, 104, 6217–6254 CrossRef CAS PubMed.
- I. Bauer and H.-J. Knölker, Chem. Rev., 2015, 115, 3170–3387 CrossRef CAS PubMed.
- J. M. Hoyt, K. T. Sylvester, S. P. Semproni and P. J. Chirik, J. Am. Chem. Soc., 2013, 135, 4862–4877 CrossRef CAS PubMed.
- P. J. Chirik, Acc. Chem. Res., 2015, 48, 1687–1695 CrossRef CAS PubMed.
- A. M. Abu-Dief and S. M. Abdel-Fatah, Beni Suef Univ. J., Basic Appl. Sci., 2018, 7, 55–67 Search PubMed.
- E. B. Bauer, Curr. Org. Chem., 2008, 12, 1341–1369 CrossRef CAS.
- D. L. Huber, Small, 2005, 1, 482–501 CrossRef CAS PubMed.
- V. Polshettiwar and R. S. Varma, Tetrahedron, 2010, 66, 1091–1097 CrossRef CAS.
- R. B. Nasir Baig and R. S. Varma, Green Chem., 2013, 15, 398–417 RSC.
- E. Nakamura, T. Hatakeyama, S. Ito, K. Ishizuka, L. Ilies and M. Nakamura, in Organic Reactions, 2014, pp. 1–210 Search PubMed.
- B. D. Sherry and A. Fürstner, Acc. Chem. Res., 2008, 41, 1500–1511 CrossRef CAS PubMed.
- J. Feng, S. Handa, F. Gallou and B. H. Lipshutz, Angew. Chem., Int. Ed., 2016, 55, 8979–8983 CrossRef CAS PubMed.
- S. Handa, J. D. Smith, M. S. Hageman, M. Gonzalez and B. H. Lipshutz, ACS Catal., 2016, 6, 8179–8183 CrossRef CAS.
- C. M. Gabriel, M. Parmentier, C. Riegert, M. Lanz, S. Handa, B. H. Lipshutz and F. Gallou, Org. Process Res. Dev., 2017, 21, 247–252 CrossRef CAS.
- B. Jin, F. Gallou, J. Reilly and B. H. Lipshutz, Chem. Sci., 2019, 10, 3481–3485 RSC.
- R. R. Thakore, K. S. Iyer and B. H. Lipshutz, Curr. Opin. Green Sustainable Chem., 2021, 31, 100493 CrossRef CAS.
- H. Pang, F. Gallou, H. Sohn, J. Camacho-Bunquin, M. Delferro and B. H. Lipshutz, Green Chem., 2018, 20, 130–135 RSC.
- S. Handa, Y. Wang, F. Gallou and B. H. Lipshutz, Science, 2015, 349, 1087–1091 CrossRef CAS PubMed.
- B. H. Lipshutz, J. C. Caravez and K. S. Iyer, Curr. Opin. Green Sustainable Chem., 2022, 38, 100686 CrossRef CAS.
- B. H. Lipshutz, Johnson Matthey Technol. Rev., 2017, 61, 196–202 CrossRef CAS.
- A. Adenot, E. B. Landstrom, F. Gallou and B. H. Lipshutz, Green Chem., 2017, 19, 2506–2509 RSC.
- Y. Hu, M. J. Wong and B. H. Lipshutz, Angew. Chem., Int. Ed., 2022, 61, e202209784 CrossRef CAS PubMed.
- H. Pang, Y. Hu, J. Yu, F. Gallou and B. H. Lipshutz, J. Am. Chem. Soc., 2021, 143, 3373–3382 CrossRef CAS PubMed.
- V. P. Ananikov, ACS Catal., 2015, 5, 1964–1971 CrossRef CAS.
- S. Ogoshi, Nickel Catalysis in Organic synthesis: Methods and Reactions, John Wiley & Sons, 2019 Search PubMed.
- S. Z. Tasker, E. A. Standley and T. F. Jamison, Nature, 2014, 509, 299–309 CrossRef CAS PubMed.
- M. Luescher, B. H. Lipshutz and F. Gallou, ChemRxiv., 2024, preprint, DOI:10.26434/chemrxiv-2024-tc9hm.
- M. D. Hossain, R. A. Mayanovic, S. Dey, R. Sakidja and M. Benamara, Phys. Chem. Chem. Phys., 2018, 20, 10396–10406 RSC.
- E. A. Standley and T. F. Jamison, J. Am. Chem. Soc., 2013, 135, 1585–1592 CrossRef CAS PubMed.
- Y. Hou, H. Kondoh, T. Ohta and S. Gao, Appl. Surf. Sci., 2005, 241, 218–222 CrossRef CAS.
- K. Oh, C. Mériadec, B. Lassalle-Kaiser, V. Dorcet, B. Fabre, S. Ababou-Girard, L. Joanny, F. Gouttefangeas and G. Loget, Energy Environ. Sci., 2018, 11, 2590–2599 RSC.
- N. D. Clement, K. J. Cavell, C. Jones and C. J. Elsevier, Angew. Chem., Int. Ed., 2004, 43, 1277–1279 CrossRef CAS PubMed.
- Ö. Metin and S. Özkar, Int. J. Hydrogen Energy, 2011, 36, 1424–1432 CrossRef.
- S. Handa, E. D. Slack and B. H. Lipshutz, Angew. Chem., Int. Ed., 2015, 54, 11994–11998 CrossRef CAS PubMed.
- A. B. Wood, M. Cortes-Clerget, J. R. A. Kincaid, B. Akkachairin, V. Singhania, F. Gallou and B. H. Lipshutz, Angew. Chem., Int. Ed., 2020, 59, 17587–17593 CrossRef CAS PubMed.
- A. R. Muci and S. L. Buchwald, Practical Palladium Catalysts for C-N and C-O Bond Formation, ed. N. Miyaura, Springer Berlin Heidelberg, Berlin, Heidelberg, 2002, pp. 131–209 Search PubMed.
- A. Biffis, P. Centomo, A. del Zotto and M. Zecca, Chem. Rev., 2018, 118, 2249–2295 CrossRef CAS PubMed.
- S. McCarthy, D. C. Braddock and J. D. E. T. Wilton-Ely, Coord. Chem. Rev., 2021, 442, 213925 CrossRef CAS.
- M. Aksoy, H. Kilic, B. Nişancı and Ö. Metin, Inorg. Chem. Front., 2021, 8, 499–545 RSC.
- A. Reina, T. Dang-Bao, I. Guerrero-Ríos and M. Gómez, Nanomaterials, 2021, 11 Search PubMed.
- M. Iqbal, Y. V. Kaneti, J. Kim, B. Yuliarto, Y.-M. Kang, Y. Bando, Y. Sugahara and Y. Yamauchi, Small, 2019, 15, 1804378 CrossRef PubMed.
- H. Chen, G. Wei, A. Ispas, S. G. Hickey and A. Eychmüller, J. Phys. Chem. C, 2010, 114, 21976–21981 CrossRef CAS.
- X. Zhao, Y. Chang, W.-J. Chen, Q. Wu, X. Pan, K. Chen and B. Weng, ACS Omega, 2022, 7, 17–31 CrossRef CAS PubMed.
- E. D. Slack, C. M. Gabriel and B. H. Lipshutz, Angew. Chem., Int. Ed., 2014, 53, 14051–14054 CrossRef CAS PubMed.
- T. N. Ansari, J. B. Jasinski, D. K. Leahy and S. Handa, JACS Au, 2021, 1, 308–315 CrossRef CAS PubMed.
- D. Petkova, N. Borlinghaus, S. Sharma, J. Kaschel, T. Lindner, J. Klee, A. Jolit, V. Haller, S. Heitz, K. Britze, J. Dietrich, W. M. Braje and S. Handa, ACS Sustainable Chem. Eng., 2020, 8, 12612–12617 CrossRef CAS.
- A. Bhattacharjya, P. Klumphu and B. H. Lipshutz, Nat. Commun., 2015, 6, 7401 CrossRef PubMed.
- B. S. Takale, R. R. Thakore, G. Casotti, X. Li, F. Gallou and B. H. Lipshutz, Angew. Chem., Int. Ed., 2021, 60, 4158–4163 CrossRef CAS PubMed.
- M. Bihani, T. N. Ansari, L. Finck, P. P. Bora, J. B. Jasinski, B. Pavuluri, D. K. Leahy and S. Handa, ACS Catal., 2020, 10, 6816–6821 CrossRef CAS.
- F. Diederich and P. J. Stang, Metal-catalyzed cross-coupling reactions, John Wiley & Sons, 2008 Search PubMed.
- C. Amatore, A. Jutand and G. Le Duc, Chem. – Eur. J., 2011, 17, 2492–2503 CrossRef CAS PubMed.
- R. Martin and S. L. Buchwald, Acc. Chem. Res., 2008, 41, 1461–1473 CrossRef CAS PubMed.
- A. Kumar, G. K. Rao, S. Kumar and A. K. Singh, Dalton Trans., 2013, 42, 5200–5223 RSC.
- I. Hussain, J. Capricho and M. A. Yawer, Adv. Synth. Catal., 2016, 358, 3320–3349 CrossRef CAS.
- Y. Wang, Y. Liu, W. Zhang, H. Sun, K. Zhang, Y. Jian, Q. Gu, G. Zhang, J. Li and Z. Gao, ChemSusChem, 2019, 12, 5265–5273 CrossRef CAS PubMed.
- G. Kaur, J. B. Jasinski, F. Gallou and S. Handa, ACS Appl. Mater. Interfaces, 2022, 14, 50947–50955 CrossRef CAS PubMed.
- Y. Era, J. A. Dennis, S. Wallace and L. E. Horsfall, Green Chem., 2021, 23, 8886–8890 RSC.
- D. J. C. Constable, C. Jimenez-Gonzalez and R. K. Henderson, Org. Process Res. Dev., 2007, 11, 133–137 CrossRef CAS.
- M. T. Sabatini, L. T. Boulton, H. F. Sneddon and T. D. Sheppard, Nat. Catal., 2019, 2, 10–17 CrossRef CAS.
- S. Sharma, J. B. Jasinski, W. M. Braje and S. Handa, ChemSusChem, 2023, 16, e202201826 CrossRef CAS PubMed.
- H. C. Hailes, Org. Process Res. Dev., 2007, 11, 114–120 CrossRef CAS.
- G. Hedouin, D. Ogulu, G. Kaur and S. Handa, Chem. Commun., 2023, 59, 2842–2853 RSC.
- S. Sharma, N. W. Buchbinder, W. M. Braje and S. Handa, Org. Lett., 2021, 25, 1960–1965 CAS.
- S. Sharma, G. Kaur and S. Handa, Org. Process Res. Dev., 2021, 25, 1960–1965 CrossRef CAS.
- J. Wang, M. Sánchez-Roselló, J. L. Aceña, C. del Pozo, A. E. Sorochinsky, S. Fustero, V. A. Soloshonok and H. Liu, Chem. Rev., 2014, 114, 2432–2506 CrossRef CAS PubMed.
- Y. Zhou, J. Wang, Z. Gu, S. Wang, W. Zhu, J. L. Aceña, V. A. Soloshonok, K. Izawa and H. Liu, Chem. Rev., 2016, 116, 422–518 CrossRef CAS PubMed.
- S. Purser, P. R. Moore, S. Swallow and V. Gouverneur, Chem. Soc. Rev., 2008, 37, 320–330 RSC.
- W. K. Hagmann, J. Med. Chem., 2008, 51, 4359–4369 CrossRef CAS PubMed.
- T. C. Barden, Indoles: Industrial, Agricultural and Over-the-Counter Uses. In Heterocyclic Scaffolds II (Topics in Heterocyclic Chemistry, Vol 26), ed. G. W. Gribble, Springer Berlin Heidelberg, Berlin, Heidelberg, 2010, pp. 31–46 Search PubMed.
- R. Lin, S. Ding, Z. Shi and N. Jiao, Org. Lett., 2011, 13, 4498–4501 CrossRef CAS PubMed.
- Y. Takeuchi, T. Tarui and N. Shibata, Org. Lett., 2000, 2, 639–642 CrossRef CAS PubMed.
- A. Arcadi, E. Pietropaolo, A. Alvino and V. Michelet, Org. Lett., 2013, 15, 2766–2769 CrossRef CAS PubMed.
- L. Yang, Y. Ma, F. Song and J. You, Chem. Commun., 2014, 50, 3024–3026 RSC.
- B. Alcaide, P. Almendros, S. Cembellín, T. Martínez del Campo and A. Muñoz, Chem. Commun., 2016, 52, 6813–6816 RSC.
- P. P. Bora, M. Bihani, S. Plummer, F. Gallou and S. Handa, ChemSusChem, 2019, 12, 3037–3042 CrossRef CAS PubMed.
- D. G. Brown and J. Boström, J. Med. Chem., 2016, 59, 4443–4458 CrossRef CAS PubMed.
- M. Baumann and I. R. Baxendale, Beilstein J. Org. Chem., 2013, 9, 2265–2319 CrossRef PubMed.
- N. A. Isley, R. T. H. Linstadt, S. M. Kelly, F. Gallou and B. H. Lipshutz, Org. Lett., 2015, 17, 4734–4737 CrossRef CAS PubMed.
- J. D. Smith, T. N. Ansari, M. P. Andersson, D. Yadagiri, F. Ibrahim, S. Liang, G. B. Hammond, F. Gallou and S. Handa, Green Chem., 2018, 20, 1784–1790 RSC.
- N. Borlinghaus, T. N. Ansari, L. H. Braje, D. Ogulu, S. Handa, V. Wittmann and W. M. Braje, Green Chem., 2021, 23, 3955–3962 RSC.
- P. Gurnani and S. Perrier, Prog. Polym. Sci., 2020, 102, 101209 CrossRef CAS.
- M. J. Barandiaran, J. C. de la Cal and J. M. Asua, in Polymer Reaction Engineering, 2007, pp. 233–272 Search PubMed.
- P. A. Lovell and F. J. Schork, Biomacromolecules, 2020, 21, 4396–4441 CrossRef CAS PubMed.
- A. N. M. B. El-hoshoudy, Emulsion Polymerization Mechanism, ed. N. Cankaya, IntechOpen, Rijeka, 2018, ch. 1 Search PubMed.
- C. S. Chern, Prog. Polym. Sci., 2006, 31, 443–486 CrossRef CAS.
- P. Marion, B. Bernela, A. Piccirilli, B. Estrine, N. Patouillard, J. Guilbot and F. Jérôme, Green Chem., 2017, 19, 4973–4989 RSC.
- R. A. Sheldon, Green Chem., 2016, 18, 3180–3183 RSC.
- D. Wang and D. Astruc, Chem. Soc. Rev., 2017, 46, 816–854 RSC.
- C. Krell, R. Schreiber, L. Hueber, L. Sciascera, X. Zheng, A. Clarke, R. Haenggi, M. Parmentier, H. Baguia, S. Rodde and F. Gallou, Org. Process Res. Dev., 2021, 25, 900–915 CrossRef CAS.
- M. Hampel, A. Mauffret, K. Pazdro and J. Blasco, Environ. Monit. Assess., 2012, 184, 6013–6023 CrossRef CAS PubMed.
- E. Lémery, S. Briançon, Y. Chevalier, C. Bordes, T. Oddos, A. Gohier and M.-A. Bolzinger, Colloids Surf., A, 2015, 469, 166–179 CrossRef.
- F.-J. Zhu, W.-L. Ma, T.-F. Xu, Y. Ding, X. Zhao, W.-L. Li, L.-Y. Liu, W.-W. Song, Y.-F. Li and Z.-F. Zhang, Ecotoxicol. Environ. Saf., 2018, 153, 84–90 CrossRef CAS PubMed.
- S. Pagola, Crystals, 2023, 13, 124 CrossRef CAS.
- C. Len, V. Duhan, W. Ouyang, R. Nguyen and B. Lochab, Front. Chem., 2023, 11, 1 Search PubMed.
- K. J. Ardila-Fierro and J. G. Hernández, ChemSusChem, 2021, 14, 2145–2162 CrossRef CAS PubMed.
- C. Espro and D. Rodríguez-Padrón, Curr. Opin. Green Sustainable Chem., 2021, 30, 100478 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2024 |