Continuous-flow directed C–H amidation reactions using immobilized Cp*Rh(III) catalysts†
Yuki
Saito
,
Taro
Konno
and
Shū
Kobayashi
 *
*
Department of Chemistry, Graduate School of Science, The University of Tokyo, 7-3-1, Hongo, Bunkyo-ku, Tokyo, Japan. E-mail: shu_kobayashi@chem.s.u-tokyo.ac.jp
First published on 27th June 2024
Abstract
We report a green, continuous-flow C–H amidation methodology using heterogeneous Cp*Rh(III) catalysts. These catalysts enable halide- and salt-free reactions with excellent activity and durability across diverse substrates, demonstrated by a 200-hour, multi-gram scale reaction exceeding 290 TON (turnover number).
Transition metal-catalyzed C–H bond functionalization has emerged as a powerful and green synthetic method due to its ability to perform efficient chemical transformations under halogen-free and salt-free conditions.1,2 Recent studies have demonstrated the remarkable potential of this methodology for the synthesis of complex molecules like natural products and active pharmaceutical ingredients (APIs).3–6 Group 9 Cp*M(III) catalysts, in particular, have been extensively studied and utilized for various C–H functionalization reactions, showcasing their high activity and versatility.7–11 However, a significant challenge associated with these catalysts is their high cost, often requiring substantial catalyst loading.12 Additionally, potential contamination of products with toxic metals necessitates further purification steps, leading to increased waste and tedious procedures.
Heterogeneous catalysts offer a solution to these challenges. Their ease of separation and reusability via simple filtration promotes efficient syntheses and significantly reduces waste. When coupled with flow chemistry, this approach allows for highly efficient and continuous processes. Recent years have seen the development of diverse heterogeneous catalysts for continuous-flow reactions.13–16 However, a significant challenge remains: achieving heterogeneous transition metal-catalyzed C–H bond functionalizations in continuous flow. While success has been demonstrated for various continuous-flow reactions using homogeneous catalysts,17,18 only a few types of heterogeneous catalysts for flow C–H functionalizations have been reported.19–25 Furthermore, immobilization of Cp*M(III) catalysts, to the best of our knowledge, remains elusive due to the difficulties associated with the process. Notably, the high cost and waste generation associated with traditional methods for modifying the Cp* structure necessitate an immobilization approach that avoids ligand modification altogether.26
Building upon our prior development of heterogeneous chiral Rh(I) catalysts for continuous-flow hydrogenation, hydroacylation, enyne cyclization, and chiral Sc(III) catalysts for Friedel–Crafts reactions,25,27–29 we investigated a novel immobilization method for cationic transition metal complexes using non-covalent interactions. This method has been applied to develop continuous-flow reactions with heterogeneous catalysts. We hypothesized that Cp*Rh(III) could be immobilized using this method due to its cationic nature.
Here, we developed heterogeneous Cp*Rh(III) catalysts for continuous-flow C–H amidation of 2-phenylpyridines.30 These catalysts can be prepared in batch or flow mode by pumping a metal complex solution into a cartridge column. The catalysts exhibit high activity in C–H amidation for various substrates under continuous-flow conditions, maintaining high performance for at least 200 h of flow reaction without metal leaching.
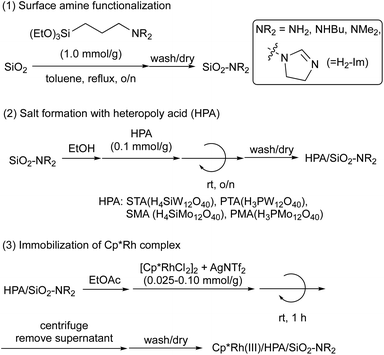
A three-step synthesis of heterogeneous CpRh(III) catalysts supported on silica is described in Scheme 1.25,27–29 In the first step, SiO2 was functionalized with amines (SiO2-NR2) using various silane coupling reagents containing primary, secondary, and tertiary amines, as well as dihydroimidazole (H2-Im). Subsequently, the obtained SiO2-NR2 was reacted with heteropoly acids (HPAs) – H3PW12O40 (PTA), H4SiW12O40 (STA), H3PMo12O40 (PMA), and H4SiMo12O40 (SMA) – to form surface-bound acid–base salts (HPA/SiO2-NR2). Finally, a Cp*Rh(NTf2)2 complex, synthesized from [Cp*RhCl2]2 and AgNTf2 based on reported procedures,30 was immobilized onto the HPA/SiO2-NR2 supports by stirring in ethyl acetate (EtOAc) for 1 h. This yielded the desired heterogeneous Cp*Rh(III) catalysts (Cp*Rh(III)/HPA/SiO2-NR2).
The impact of support structure on the catalytic C–H amidation of 2-phenylpyridine (1a) with dioxazolone (2a) under batch conditions was evaluated (Table 1). Initially, the effect of heteropoly acids (HPAs) was investigated using a simple primary amine (entries 1–4). STA emerged as the most effective HPA, affording an 85% yield of the desired product 3a. The highly negatively charged and soft STA4− anion appears to be well-suited for the cationic Cp*Rh(III) catalyst.31 However, a concerning 7.5% Rh leaching was observed after the reaction. To address Rh leaching, the amine unit structure was subsequently explored. While both primary and secondary amines resulted in lower yields and increased leaching (entries 5 and 6), the use of a dihydroimidazole unit (H2-Im) led to a gratifying increase in yield and a significant decrease in leaching (entry 7). Based on our prior research, it is hypothesized that the dihydroimidazole unit coordinates with the coordinatively unsaturated Rh(III) center, promoting stabilization and preventing leaching.29 Finally, the effect of catalyst loading was examined. Decreasing the loading concentration further enhanced the yield and completely suppressed Rh leaching (entries 8 and 9).
| Entry | Amine structure | HPA | Loading (mmol g−1) | Yielda (%) | Rh leachingb (%) |
|---|---|---|---|---|---|
| a Determined by 1H NMR analysis by using Durene as an internal standard. b Determined by ICP analysis. | |||||
| 1 | NH2 | STA | 0.05 | 85 | 7.5 |
| 2 | NH2 | SMA | 0.05 | 32 | ND |
| 3 | NH2 | PMA | 0.05 | 26 | ND |
| 4 | NH2 | PTA | 0.05 | 48 | ND |
| 5 | NHnBu | STA | 0.05 | 67 | 1.6 |
| 6 | NEt2 | STA | 0.05 | 19 | 1.8 |
| 7 | H2-Im | STA | 0.05 | 91 | 0.5 |
| 8 | H2-Im | STA | 0.10 | 90 | 2.3 |
| 9 | H2-Im | STA | 0.025 | 96 | UDL |
We characterized the prepared catalysts using both scanning transmission electron microscope (STEM) analysis and N2 adsorption/desorption isotherms. STEM revealed good dispersion of Rh, W, and N on the SiO2 surface, with no observed metal aggregation (Fig. S3†). N2 adsorption/desorption isotherms confirmed a stepwise decrease in surface area upon SiO2 functionalization, suggesting that the active Cp*Rh(III) species likely reside on the surface while maintaining the bulk structure of the material (Fig. S1, S2 and Tables S1, S2†).
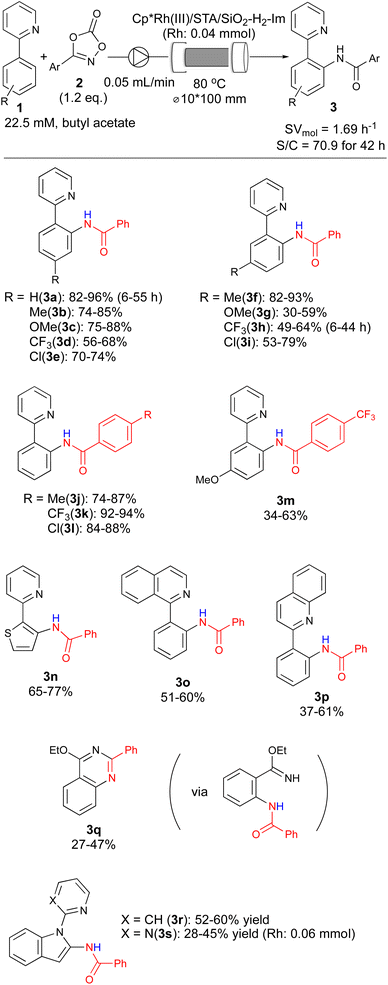
Leveraging the optimized support structure, we further optimized the reaction under continuous-flow conditions. To achieve this, we developed a continuous-flow immobilization method for heterogeneous catalyst preparation. A Cp*Rh(NTf)2 solution was pumped through a column reactor packed with STA/SiO2-H2-Im, immobilizing the catalyst on the fly. After washing with the reaction solvent, continuous-flow C–H amidation was performed. Initial temperature optimization under continuous-flow revealed a yield improvement to 70% at 80 °C using isoamyl acetate as the solvent (Fig. S4†). Next, we investigated the effect of the Cp* ligand structure (Fig. 1). While Cp and CpMe4 showed similar activity, replacing a methyl group with tBu and CF3 significantly decreased it. The Ind* ligand also exhibited lower activity and durability. These results suggest that catalyst immobilization via Cp ligand modification might compromise activity.
Optimizing catalyst and reagent amounts (Fig. 2) significantly increased the yield of 3a to over 90%. Notably, increasing the catalyst amount while reducing the amount of 2a from 2.0 to 1.2 equivalents maintained this high yield. Using 0.04 mmol of Rh catalyst and 1.2 equivalents of 2a achieved this result in a continuous-flow reaction over 24 hours. Importantly, no catalyst deactivation or Rh leaching was observed during the reaction.
With optimized flow conditions established, we investigated the substrate scope using a 42-hour continuous-flow reaction (Scheme 2). The effect of para-substituents on the aromatic ring was examined first. Electron-donating groups had minimal impact on reactivity (3b, c), while electron-withdrawing groups led to slightly lower yields for target products 3d and 3e. This suggests the importance of electron density in the directing pyridine for optimal reactivity. Next, the impact of meta-substituents was assessed. Interestingly, both electron-donating and electron-withdrawing groups negatively affected yields (3f–i). This observation implies that C–H bond acidity is another crucial factor in substrate reactivity. The scope of amidating reagents (3) was also explored. Both electron-donating and electron-withdrawing groups furnished the corresponding products (3j–l) in good to excellent yields. However, even a reactive amidating reagent failed to improve the yield for the electron-donating meta-substituted substrate (3m). This reinforces the notion that pyridine coordination and the subsequent CMD process are central to this reaction.32 C–H amidation of thiophene was explored, yielding product 3n in moderate to good yields. Finally, the applicability of other directing groups was tested. Quinoline and isoquinoline were successfully employed, affording products 3o and 3p, respectively. Interestingly, using an imidate as a directing group resulted in quinazoline product 3q, albeit in low to moderate yield. Finally, C–H amidations of indoles were also performed. Regardless of the directing group, the C2-amidtion products 3r and 3s were obtained in moderate yields.
Finally, an extended time continuous-flow experiment was performed to assess the catalyst's durability (Fig. 3). This experiment demonstrated that the target product 3a could be synthesized in >70% yield for over 200 h continuously. While gradual decrease in catalyst activity was observed over time, this experiment also revealed a total turnover number (TON) exceeding 290. We also attempted to clarify the cause of catalyst deactivation. First and the most importantly, no leaching of the catalyst was observed. STEM and EDS analyses revealed that there was no clear aggregation of Rh species (Fig. S5†). Washing the used catalyst revealed that some polar compounds were adsorbed on the catalyst; however, the activity was not recovered even after washing (Fig. S4, Scheme S7†). Considering the high TON of the homogeneous catalyst under batch conditions (Table S3†), the most plausible cause of the catalyst deactivation is the degradation of the catalyst caused by STA anion or surface amine structure.
In conclusion, we successfully developed heterogeneous Cp*Rh(III) catalysts enabling continuous-flow C–H amidation reactions. Investigation of the support structure revealed that both HPA structure and surface amine are crucial for catalyst activity and preventing leaching. Furthermore, detailed continuous-flow studies highlighted the critical role of the Cp structure in catalyst activity. The optimized reaction conditions resulted in high catalyst activity for various substrates. Notably, the reactions proceeded smoothly under halide-free and salt-free conditions. Finally, extended time flow reactions enabled multi-gram scale synthesis without catalyst leaching. Further investigations to clarify the cause of catalyst deactivation and improve the catalyst durability are under investigation.
Data availability
The data supporting this article have been included as part of the ESI.†Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was supported in part a The Grant-in-Aid for Transformative Research Areas (A) JP21A204/22H05345 Digitalization-driven Transformative Organic Synthesis (Digi-TOS) from the Ministry of Education, Culture, Sports, Science & Technology, Japan. We thank Dr Tei Maki (The University of Tokyo and JEOL) for STEM and EDS analyses.References
- D. Maiti, Handbook of CH-Functionalization, Wiley-VCH GmbH, 2022 Search PubMed.
- T. Punniyamurthy and A. Kumar, Transition-Metal-Catalyzed C-H Functionalization of Heterocycles, Wiley-VCH GmbH, 2023 Search PubMed.
- R. R. Karimov and J. F. Hartwig, Angew. Chem., Int. Ed., 2018, 57, 4234–4241 CrossRef CAS PubMed.
- D. J. Abrams, P. A. Provencher and E. J. Sorensen, Chem. Soc. Rev., 2018, 47, 8925–8967 RSC.
- M. Moir, J. J. Danon, T. A. Reekie and M. Kassiou, Expert Opin. Drug Discovery, 2019, 14, 1137–1149 CrossRef CAS PubMed.
- L. Guillemard, N. Kaplaneris, L. Ackermann and M. J. Johansson, Nat. Rev. Chem., 2021, 5, 522–545 CrossRef CAS.
- G. Song and X. Li, Acc. Chem. Res., 2015, 48, 1007–1020 CrossRef CAS.
- T. Yoshino and S. Matsunaga, Adv. Synth. Catal., 2017, 359, 1245–1262 CrossRef CAS.
- T. Piou and T. Rovis, Acc. Chem. Res., 2017, 51, 170–180 CrossRef PubMed.
- T. Yoshino, S. Satake and S. Matsunaga, Chem. – Eur. J., 2020, 26, 7346–7357 CrossRef CAS PubMed.
- S. Y. Hong, Y. Hwang, M. Lee and S. Chang, Acc. Chem. Res., 2021, 54, 2683–2700 CrossRef CAS.
- U. Dhawa, N. Kaplaneris and L. Ackermann, Org. Chem. Front., 2021, 8, 4886–4913 RSC.
- R. Munirathinam, J. Huskens and W. Verboom, Adv. Synth. Catal., 2015, 357, 1093–1123 CrossRef CAS.
- A. Tanimu, S. Jaenicke and K. Alhooshani, Chem. Eng. J., 2017, 327, 792–821 CrossRef CAS.
- K. Masuda, T. Ichitsuka, N. Koumura, K. Sato and S. Kobayashi, Tetrahedron, 2018, 74, 1705–1730 CrossRef CAS.
- W.-J. Yoo, H. Ishitani, Y. Saito, B. Laroche and S. Kobayashi, J. Org. Chem., 2020, 85, 5132–5145 CrossRef CAS PubMed.
- S. Santoro, F. Ferlin, L. Ackermann and L. Vaccaro, Chem. Soc. Rev., 2019, 48, 2767–2782 RSC.
- S. Govaerts, A. Nyuchev and T. Noel, J. Flow Chem., 2020, 10, 13–71 CrossRef CAS.
- T. Tagata, M. Nishida and A. Nishida, Adv. Synth. Catal., 2010, 352, 1662–1666 CrossRef CAS.
- H. P. L. Gemoets, G. Laudadio, K. Verstraete, V. Hessel and T. Noël, Angew. Chem., Int. Ed., 2017, 56, 7161–7165 CrossRef CAS.
- F. Ferlin, S. Santoro, L. Ackermann and L. Vaccaro, Green Chem., 2017, 19, 2510–2514 RSC.
- C. J. Yoo, D. Rackl, W. Liu, C. B. Hoyt, B. Pimentel, R. P. Lively, H. M. L. Davies and C. W. Jones, Angew. Chem., Int. Ed., 2018, 57, 10923–10927 CrossRef CAS PubMed.
- F. Ferlin, L. Luciani, S. Santoro, A. Marrocchi, D. Lanari, A. Bechtoldt, L. Ackermann and L. Vaccaro, Green Chem., 2018, 20, 2888–2893 RSC.
- F. Ferlin, I. Anastasiou, N. Salameh, T. Miyakoshi, O. Baudoin and L. Vaccaro, ChemSusChem, 2022, 15, 1–7 CrossRef PubMed.
- Y. Saito and S. Kobayashi, Angew. Chem., Int. Ed., 2023, 63, e202313778 CrossRef PubMed.
- J. M. Fraile, J. I. García and J. A. Mayoral, Chem. Rev., 2008, 109, 360–417 CrossRef PubMed.
- Y. Saito and S. Kobayashi, J. Am. Chem. Soc., 2020, 142, 16546–16551 CrossRef CAS.
- Y. Saito and S. Kobayashi, Angew. Chem., Int. Ed., 2021, 60, 26566–26570 CrossRef CAS PubMed.
- Y. Saito, Y. Sato and S. Kobayashi, ACS Catal., 2024, 14, 2202–2206 CrossRef CAS.
- Y. Park, K. T. Park, J. G. Kim and S. Chang, J. Am. Chem. Soc., 2015, 137, 4534–4542 CrossRef CAS PubMed.
- M. N. Timofeeva, Appl. Catal., A, 2003, 256, 19–35 CrossRef CAS.
- Y. Park, K. T. Park, J. G. Kim and S. Chang, J. Am. Chem. Soc., 2015, 137, 4534–4542 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4gc01747a |
| This journal is © The Royal Society of Chemistry 2024 |