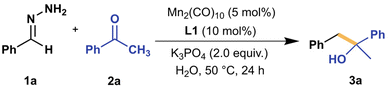Manganese-catalyzed nucleophilic addition of aldehydes to carbonyl compounds via hydrazone umpolung on water†
Jan Michael
Salgado
 ,
Durbis J.
Castillo-Pazos
,
Durbis J.
Castillo-Pazos
 ,
Juan D.
Lasso
,
Juan D.
Lasso
 ,
Konstantin L.
Stock
,
Konstantin L.
Stock
 and
Chao-Jun
Li
and
Chao-Jun
Li
 *
*
Department of Chemistry, and FRQNT Centre for Green Chemistry and Catalysis, McGill University, 801 Sherbrooke Street West, Montreal, QC H3A 0B8, Canada. E-mail: cj.li@mcgill.ca
First published on 24th May 2024
Abstract
Hydrazones as organometallic equivalents have emerged as a general and sustainable strategy to utilize naturally abundant aldehydes and ketones as feedstocks while only releasing water and nitrogen gas as byproducts. Yet the addition of these carbanion equivalents to carbonyl compounds has been limited to the use of precious metals as catalysts and hazardous solvents under an inert atmosphere. Herein, we report the development of a manganese-based catalyst system for the addition of aldehydes to carbonyl compounds producing secondary and tertiary alcohols with yields of up to 91%. Furthermore, this method has proven robust and reproducible under aqueous and aerobic conditions to employ an earth-abundant metal catalyst, hence advancing hydrazone umpolung chemistry to be more sustainable and operationally simple.
Introduction
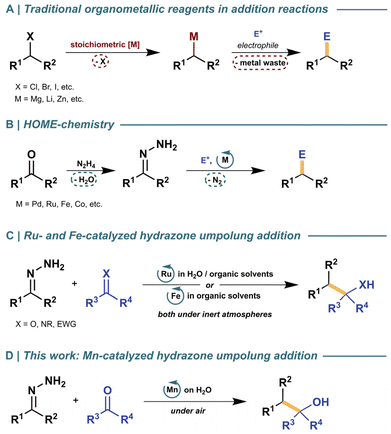
The use of organometallic reagents has long been a cornerstone in organic chemistry for the construction of complex molecules.1 Through controlled reactions with electrophiles, these reagents can facilitate the formation of crucial moieties in natural products, fine chemicals, materials, agrochemicals, and pharmaceuticals—notably, with carbonyl electrophiles to form alcohols (Scheme 1A).2,3 But despite their efficacy to form new C–C bonds, traditional organometallic reagents often require organic halide as feedstock and mandate stoichiometric amounts of metal. Moreover, besides the report of a few exceptions,4 these methodologies are generally intolerant to moisture and air, thus limiting their functional group compatibility and producing copious waste—challenges to overcome with the help of green chemistry.In response to these challenges, hydrazones as organometallic equivalents (HOME) have emerged as a sustainable approach to form new C–C bonds (Scheme 1B).5,6 The use of catalytic amounts of a transition metal—as opposed to the superstoichiometric addition of organometallic reagents—has facilitated a variety of transformations such as addition (Scheme 1C),7,8 olefination,9 and cross-coupling10,11 reactions, while only generating water and nitrogen gas as benign byproducts. However, HOME chemistry still presents two critical areas for improvement to showcase its applicability as a greener alternative to classical methods, first being the use of scarce and precious metals. This not only raises concerns for resource depletion, but also for having a higher system-wide environmental burden than first row metals.12 Therefore, it is crucial for the field to develop earth abundant metal (EAM) catalysts that can achieve the same transformations with equal or superior efficiency. The second opportunity for improvement comes from developing methods that are operationally simple, yet robust and highly tolerant to a wide diversity of functional groups and conditions. Additionally, development of robust chemical systems tolerant to oxygen and moisture that are compatible with green solvents like water are highly desirable for diverse scenarios, including medicinal chemistry campaigns and educational settings.13–19
While previous reports in our group have explored some of these improvements individually, there is still a gap for an operationally simple EAM-promoted HOME methodology capable of performing carbonyl additions in the presence of water and oxygen. In recent years, manganese as an EAM has become an attractive inexpensive and sustainable alternative to precious metals, where an increasing number of Mn-catalyzed systems have showcased their potential at promoting hydrogenation/dehydrogenation,20–25 hydrosilylation,26–30 hydroboration,31–34 and C–H activation35–40 reactions. In addition to its versatility, manganese has proven effective at catalyzing reactions in aqueous media,41,42 making it an ideal candidate to further explore in the context of HOME chemistry. Herein, we report an open-flask, on-water manganese-catalyzed nucleophilic addition of carbonyls for the synthesis of secondary and tertiary alcohols via hydrazone umpolung chemistry (Scheme 1D).
Results and discussion
Reaction development and optimization
Starting our investigation, we chose benzaldehyde hydrazone (1a) as the model nucleophile surrogate, adding to acetophenone (2a)—an unhindered aromatic ketone—as the model electrophile. We initially examined our model reaction (Scheme 2) in a controlled environment (under N2, inside a glovebox) and in an organic solvent, with toluene being our initial choice to allow the exploration of reaction conditions at higher temperatures. By screening different ligands (see Fig. S1† for full list) with Mn2(CO)10 as the initial catalyst of choice, we observed that the desired alcohol product 3a was only present when employing PNP-type ligands with L1 resulting in the highest yield. Mn(CO)5Br, another commonly utilized Mn pre-catalyst, gave similar yields with L1 under similar conditions but only when 12.5 mol% CsF was added to the mixture. Since Mn2(CO)10 does not need this to achieve consistent desirable yields, further optimizations were carried out with this pre-catalyst.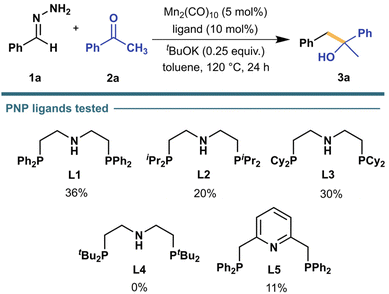 | ||
| Scheme 2 Investigation of PNP-type ligands in the nucleophilic addition of benzaldehyde hydrazone (1a, 0.25 mmol) to acetophenone (2a, 0.25 mmol) in 1.0 mL toluene under N2 atmosphere. Mn2(CO)10 (5.0 mol%), ligand (10 mol%), and tBuOK (25 mol%) were pre-mixed in 0.5 mL toluene for 1.5 h before adding 1a, 2a, and additional 0.5 mL toluene. 1H NMR yield was determined using 1,3,5-trimethoxybenzene as standard. See ESI† for more details. | ||
Initial investigations also revealed that prior mixing of the Mn pre-catalyst and ligand with the base before addition of the substrates was necessary to achieve reproducible results. This is most likely because the base participates in the formation of the active catalyst for the reaction as seen in previous related Mn-pincer catalyst systems.22,43 Knowing that the base plays an important role in the reaction, we decided to screen a series of organic and inorganic bases (Table S2†). All organic bases screened were ineffective, possibly because they can also act as ligands, hindering the formation of the active catalyst. On the other hand, inorganic bases provided better yields consistently, with K3PO4 being the most beneficial to the reaction. Further optimization allowed us to decrease the initial reaction temperature from 120 °C to 50 °C while retaining good yields (80%). However, conducting the reaction at room temperature significantly lowered the formation of 3a to 13%.
Having several conditions optimized while in a controlled environment, we decided to test under aerobic conditions employing water as a solvent (Table 1, entry 1), which gratifyingly provided a yield of 86%. Furthermore, we observed an increased rate of product 3a formation when employing water versus toluene (Fig. S6†), in compliance with the “on water” effect described by Sharpless and co-workers.18 Similarly to the reaction performed in organic solvents, both Mn2(CO)10 and L1 are essential for the reaction to proceed after generation of the active form of the catalyst (Table 1, entries 2–4). However, in contrast to previously reported Ru-catalyst systems in water,44,45 no special additives are needed to achieve good yields using Mn (Table 1, entries 5 and 6). Finally, attempts to lower the amount of pre-catalyst, ligand, and base needed revealed the optimized conditions shown on Table 1, entry 7—with yields above 80% (see ESI† for further details), even when performed under open-air (Table 1, entry 8).
| Entry | Deviation | 3a yield (%) |
|---|---|---|
| Reaction conditions: Mn2(CO)10 (5.0 mol%), L1 (10 mol%), and K3PO4 (2.0 equiv.) were pre-mixed in 0.5 mL H2O for 1.5 h (5 min for entries 7 and 8) before adding 1a (0.75 mmol), 2a (0.25 mmol), and additional 0.5 mL H2O. 1H NMR yield was determined using 1,3,5-trimethoxybenzene as standard.a Isolated yield. See ESI† for more details. | ||
| 1 | No deviations | 86 |
| 2 | No pre-catalyst | 0 |
| 3 | No ligand | 0 |
| 4 | No pre-catalyst and ligand | 0 |
| 5 | 0.4 equiv. 18-crown-6 additive | 86 |
| 6 | 2 wt% TPGS in H2O | 9 |
| 7 | 1.5 equiv. K3PO4, 2.5 mol% Mn2(CO)10, 5.0 mol% L1 | 85 (84)a |
| 8 | Same as entry 7 but open-air | 80 |
Substrate scope and limitations
With the optimized conditions in hand, we decided to explore the scope and limitations of this reaction (Scheme 3). All reactions for the scope were performed in the span of 16–24 hours to ensure completion. First, we started by probing the effects of substituents in aryl ketone electrophiles. Electron-withdrawing groups (3b–3e) proceeded with good to excellent yields while electron-donating substituents lowered the yield (3f and 3g), including trace amounts of product for 4′-hydroxyacetophenone—likely due to the phenolate's increased solubility in water, inhibiting its contact with the catalyst in the organic phase (3n). Interestingly, an aromatic heterocycle like furan (3h) proved compatible with this method. Next, we aimed to elucidate the steric limitations of aryl ketones. Generally, aryl ketones performed greatly as electrophiles even after increasing the chain length slightly (3i). However, the presence of a tert-butyl substituent (3m) revealed a limitation towards bulky substituents. Additionally, aliphatic ketones (3j) worked in moderate yields—if ring strain is present to drive the reaction forward. Switching to aldehydes revealed that aromatic aldehydes (3l) are too reactive in the presence of the hydrazone partner, predominantly forming the azine byproduct, even after performing the reactions at lower temperatures—room temperature and 0 °C to 23 °C. Such decrease in the yield for aldehyde-containing electrophiles was also previously reported for both Fe- and Ru-systems in organic solvents.7,8 Nonetheless, a less reactive aliphatic aldehyde (3k) proceeded with a moderate yield after increasing the catalyst loading.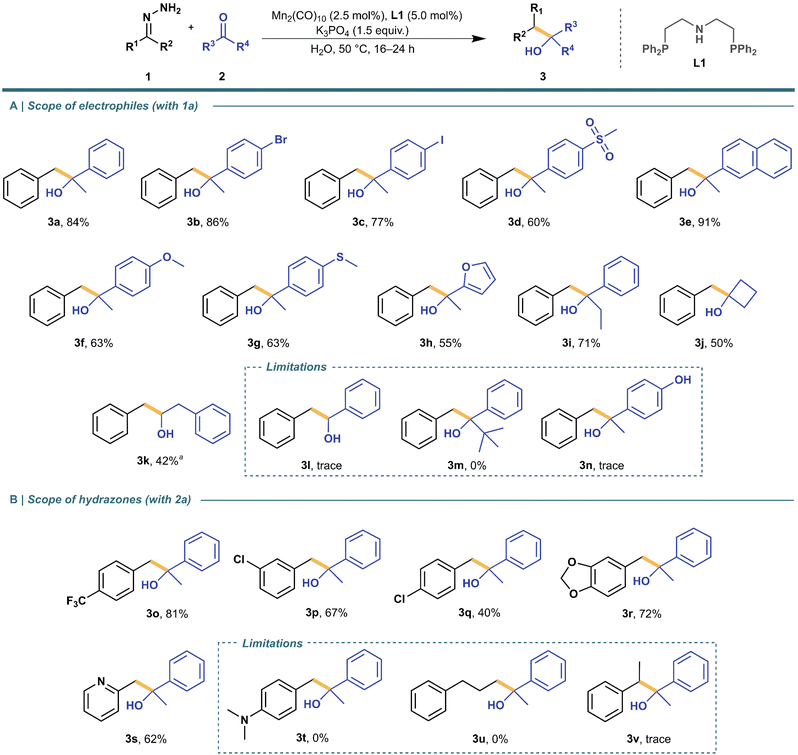 | ||
Scheme 3 Substrate scope of electrophiles and hydrazones. Reaction conditions: Mn2(CO)10 (2.5 mol%), L1 (5.0 mol%), and K3PO4 (1.5 equiv.) were pre-mixed in 0.5 mL H2O for 5 min before adding 1 (0.75 mmol), 2 (0.25 mmol), and additional 0.5 mL H2O. a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Condition variation for aldehydes: Mn2(CO)10 (5.0 mol%), L1 (10 mol%). Isolated yields are reported. See ESI† for more details. Condition variation for aldehydes: Mn2(CO)10 (5.0 mol%), L1 (10 mol%). Isolated yields are reported. See ESI† for more details. | ||
Subsequently, we shifted our focus to the exploration of hydrazone partners. For aromatic hydrazones, both electron-withdrawing and electron-donating groups (3o–3r) performed well under standard conditions. Additionally, there is a strong preference towards more electron-deficient aromatic aldehydes (like 3o) which may be due to better stabilization of the partial carbanion formed from its hydrazone precursor. In fact, this can also be observed by having the same substituent in different positions (3p and 3q). Consequently, aliphatic hydrazones (3u), having no aromatic ring to stabilize it, yielded no product. Furthermore, the presence of an amino substituent (3t) was not tolerated, possibly due to the combination of its weaker hydrazone carbanion stabilization and the affinity of nitrogen for metal coordination. Nevertheless, the reaction also proceeded with good yields in the presence of a pyridine-based substrate (3s). Like with the Ru-system,7 a ketone-derived hydrazone (1v) yielded its corresponding alcohol product (3v) poorly even under an elevated temperature (80 °C) to bypass steric limitations. However, the currently described method is still compatible with both aldehydes and ketones as substrates, with the first being more suitable as hydrazone carbanions, and the latter as electrophiles.
Proposed mechanism
A plausible mechanism is proposed (Scheme 4) based on previously reported hydrazone umpolung reactions7,46,47 and Mn-pincer catalyzed reactions.21,22,43,48 Grounded on reported literature,26,49–53 it is known that Mn2(CO)10 undergoes homolytic cleavage and subsequent oxidation reactions, therefore we propose the pre-activation step is required to form Mn(I) species A. The Mn-PNP complex (A) generated is then deprotonated by the base to form the active amido-Mn complex B. Hydrazone 1 adds to B, forming Mn-complex C which then undergoes a Zimmerman–Traxler-like transition state (D) to give the nucleophilic addition product (3) of 1 to 2 by releasing N2 as a byproduct, and regenerating the active species B.Comparison with existing HOME-chemistry methods
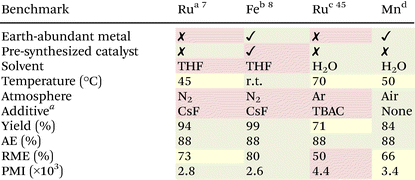
Finally, we present a comparative table highlighting the potential for this catalytic system to fulfil the areas of opportunity outlined at the beginning of this article. In contrast to previously reported Ru- and Fe-based nucleophilic additions via hydrazone umpolung,7,8,45 the advantages of the currently reported Mn-system are outlined in Table 2. Major advantages include the use of a non-precious metal catalyst and a greener solvent, with our method working in aqueous conditions even without the use of a phase-transfer catalyst. Previous methods were also all conducted in inert atmospheres whereas the currently reported method works even under open air, making it more practical and accessible.Catalysts used and 1a equivalences: a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [Ru(p-cymene)Cl2]2 (0.75 mol%)/dmpe (1.5 mol%)/1.25 equiv. 1a; b [Ru(p-cymene)Cl2]2 (0.75 mol%)/dmpe (1.5 mol%)/1.25 equiv. 1a; b![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe(dmpe)2Cl2 (5 mol%)/1.25 equiv. 1a; c Fe(dmpe)2Cl2 (5 mol%)/1.25 equiv. 1a; c![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [Ru(p-cymene)Cl2]2 (1.5 mol%)/PPh3 (6 mol%)/1.50 equiv. 1a; d [Ru(p-cymene)Cl2]2 (1.5 mol%)/PPh3 (6 mol%)/1.50 equiv. 1a; d![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Mn2(CO)10 (2.5 mol%)/L1 (5 mol%)/1.25 equiv. 1a.a Chemicals used aside from the catalyst system and base. TBAC: tetrabutylammonium chloride (a phase-transfer catalyst). AE: atom economy. RME: reaction mass efficiency. PMI: process mass intensity (including purification). Mn2(CO)10 (2.5 mol%)/L1 (5 mol%)/1.25 equiv. 1a.a Chemicals used aside from the catalyst system and base. TBAC: tetrabutylammonium chloride (a phase-transfer catalyst). AE: atom economy. RME: reaction mass efficiency. PMI: process mass intensity (including purification). |
|---|
|
In terms of scope, the systems observed similar general trends. Poor to no-yielding substrates for Mn on water such as aldehyde-based or bulky electrophiles, and ketone-based or aliphatic hydrazones also resulted in poor to moderate yields for Ru- and Fe-systems done in organic solvents.7,8 However, the Mn-promoted reaction allows for a wider scope than Ru-catalyzed reactions in water,45 with the latter having only moderate yields for electrophilic aromatic ketones.
Aside from these, the atom economy (AE), reaction mass efficiency (RME), and process mass intensity (PMI) were calculated for the model reaction of each methodology. All methods being compared has an 88% AE with the remaining atoms being extruded as benign byproducts—a major advantage of HOME-chemistry over using traditional organometallic reagents. Additionally, by lowering down the reaction volume (from 1.0 mL to 0.2 mL) of the Mn method, we were still able to achieve 84% yield of 3a even with just 1.25 equivalence of 1a. Thus, the currently described Mn-aqueous method has a higher RME than its Ru-aqueous counterpart. A feature worth mentioning is the capacity of this catalytic system to provide product in neat conditions (Table S4†). Although an increased 1a loading was needed to facilitate better mixing in this neat reaction, it provides an avenue for potential mechanochemistry adaptations in the future. Lastly, to capture all mass-based inputs including product isolation, we also computed the PMI of each method. It was found that the Mn method has a lower PMI than Ru– and Fe–organic systems, mainly due to the inherent work up involved in aqueous reactions. However, it was found to have a higher PMI than the Ru-aqueous method.
Conclusions
In summary, we have described the first earth-abundant metal catalyzed nucleophilic addition of hydrazones to carbonyls on water. By eliminating the use of organic solvents during the reaction and proceeding without the need for special additives or inert atmosphere, this method provides a platform for greener and more accessible HOME chemistry.Author contributions
JMS and CJL: conceptualization. JMS, DJCP, JDL, CJL: methodology and investigation. JMS and KLS conducted the experiments for optimization. JMS, DJCP, and JDL conducted the experiments for scope. JMS wrote the manuscript and all authors and coauthors contributed to data analysis and revising the manuscript. CJL supervised the work and provided mentorship.Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors acknowledge the Canada Research Chair (Tier I) foundation, the E.B. Eddy Endowment fund, Fonds de Recherche du Québec – Nature et Technologies (FRQNT), the Canada Foundation for Innovation, Centre en Chimie Verte et Catalyse, and the Natural Sciences and Engineering Research Council of Canada for support of our research. The authors also thank the McGill Magnetic Resonance facility and McGill analytical facility. JMS acknowledges the financial support granted by the FRQNT Doctoral Scholarship. DJCP acknowledges the financial support granted by the Vanier Canada Graduate Scholarship, CONACYT Mexico, and the Dr Lawrence Light Fellowship in Sustainability. JDL acknowledges and thanks the Walter C. Sumner Foundation and NuChem Sciences for their financial support.References
- R. Noyori and M. Kitamura, Angew. Chem., Int. Ed. Engl., 1991, 30, 49–69 CrossRef.
- N. Schneider, D. M. Lowe, R. A. Sayle, M. A. Tarselli and G. A. Landrum, J. Med. Chem., 2016, 59, 4385–4402 CrossRef CAS PubMed.
- S. D. Roughley and A. M. Jordan, J. Med. Chem., 2011, 54, 3451–3479 CrossRef CAS PubMed.
- M. J. Rodríguez-Álvarez, N. Ríos-Lombardía, S. E. García-Garrido, C. Concellón, V. del Amo, V. Capriati and J. García-Álvarez, Molecules, 2024, 29, 1422 CrossRef PubMed.
- X.-J. Dai, C.-C. Li and C.-J. Li, Chem. Soc. Rev., 2021, 50, 10733–10742 RSC.
- C.-J. Li, Pure Appl. Chem., 2023, 95, 465–474 CrossRef CAS.
- H. Wang, X.-J. Dai and C.-J. Li, Nat. Chem., 2017, 9, 374–378 CrossRef CAS PubMed.
- C.-C. Li, X.-J. Dai, H. Wang, D. Zhu, J. Gao and C.-J. Li, Org. Lett., 2018, 20, 3801–3805 CrossRef CAS PubMed.
- W. Wei, X.-J. Dai, H. Wang, C. Li, X. Yang and C.-J. Li, Chem. Sci., 2017, 8, 8193–8197 RSC.
- D. Cao, P. Pan, H. Zeng and C.-J. Li, Chem. Commun., 2019, 55, 9323–9326 RSC.
- R. Cheng, G. de Ruiter and C.-J. Li, Chem. Commun., 2022, 58, 11563–11566 RSC.
- P. Nuss and M. J. Eckelman, PLoS One, 2014, 9, e101298 CrossRef PubMed.
- D. Prat, A. Wells, J. Hayler, H. Sneddon, C. R. McElroy, S. Abou-Shehada and P. J. Dunn, Green Chem., 2015, 18, 288–296 RSC.
- M. Cortes-Clerget, J. Yu, J. R. A. Kincaid, P. Walde, F. Gallou and B. H. Lipshutz, Chem. Sci., 2021, 12, 4237–4266 RSC.
- C. J. Li, Chem. Rev., 1993, 93, 2023–2035 CrossRef CAS.
- C.-J. Li, Chem. Rev., 2005, 105, 3095–3166 CrossRef CAS PubMed.
- T. Kitanosono, K. Masuda, P. Xu and S. Kobayashi, Chem. Rev., 2018, 118, 679–746 CrossRef CAS PubMed.
- S. Narayan, J. Muldoon, M. G. Finn, V. V. Fokin, H. C. Kolb and K. B. Sharpless, Angew. Chem., Int. Ed., 2005, 44, 3275–3279 CrossRef CAS PubMed.
- A. Chanda and V. V. Fokin, Chem. Rev., 2009, 109, 725–748 CrossRef CAS PubMed.
- Y. Wang, M. Wang, Y. Li and Q. Liu, Chem, 2021, 7, 1180–1223 CAS.
- A. Mukherjee, A. Nerush, G. Leitus, L. J. W. Shimon, Y. Ben David, N. A. Espinosa Jalapa and D. Milstein, J. Am. Chem. Soc., 2016, 138, 4298–4301 CrossRef CAS PubMed.
- S. Elangovan, C. Topf, S. Fischer, H. Jiao, A. Spannenberg, W. Baumann, R. Ludwig, K. Junge and M. Beller, J. Am. Chem. Soc., 2016, 138, 8809–8814 CrossRef CAS PubMed.
- F. Kallmeier, T. Irrgang, T. Dietel and R. Kempe, Angew. Chem., Int. Ed., 2016, 55, 11806–11809 CrossRef CAS PubMed.
- U. K. Das, S. Chakraborty, Y. Diskin-Posner and D. Milstein, Angew. Chem., Int. Ed., 2018, 57, 13444–13448 CrossRef CAS PubMed.
- A. Bruneau-Voisine, D. Wang, T. Roisnel, C. Darcel and J.-B. Sortais, Catal. Commun., 2017, 92, 1–4 CrossRef CAS.
- J. Dong, X.-A. Yuan, Z. Yan, L. Mu, J. Ma, C. Zhu and J. Xie, Nat. Chem., 2021, 13, 182–190 CrossRef CAS PubMed.
- X. Yang and C. Wang, Angew. Chem., Int. Ed., 2018, 57, 923–928 CrossRef CAS PubMed.
- H. Liang, Y.-X. Ji, R.-H. Wang, Z.-H. Zhang and B. Zhang, Org. Lett., 2019, 21, 2750–2754 CrossRef CAS PubMed.
- J. Zheng, S. Chevance, C. Darcel and J.-B. Sortais, Chem. Commun., 2013, 49, 10010–10012 RSC.
- J. Zheng, S. Elangovan, D. A. Valyaev, R. Brousses, V. César, J.-B. Sortais, C. Darcel, N. Lugan and G. Lavigne, Adv. Synth. Catal., 2014, 356, 1093–1097 CrossRef CAS.
- G. Zhang, H. Zeng, J. Wu, Z. Yin, S. Zheng and J. C. Fettinger, Angew. Chem., Int. Ed., 2016, 55, 14369–14372 CrossRef CAS PubMed.
- C. Erken, A. Kaithal, S. Sen, T. Weyhermüller, M. Hölscher, C. Werlé and W. Leitner, Nat. Commun., 2018, 9, 4521 CrossRef PubMed.
- J. R. Carney, B. R. Dillon, L. Campbell and S. P. Thomas, Angew. Chem., Int. Ed., 2018, 57, 10620–10624 CrossRef CAS PubMed.
- S. Garhwal, A. A. Kroeger, R. Thenarukandiyil, N. Fridman, A. Karton and G. de Ruiter, Inorg. Chem., 2021, 60, 494–504 CrossRef CAS PubMed.
- R. Cano, K. Mackey and G. P. McGlacken, Catal. Sci. Technol., 2018, 8, 1251–1266 RSC.
- Y. Hu, B. Zhou and C. Wang, Acc. Chem. Res., 2018, 51, 816–827 CrossRef CAS PubMed.
- J. R. Carney, B. R. Dillon and S. P. Thomas, Eur. J. Org. Chem., 2016, 3912–3929 CrossRef CAS.
- W. Liu, J. Bang, Y. Zhang and L. Ackermann, Angew. Chem., Int. Ed., 2015, 54, 14137–14140 CrossRef CAS PubMed.
- Y. Kuninobu, Y. Nishina, T. Takeuchi and K. Takai, Angew. Chem., Int. Ed., 2007, 46, 6518–6520 CrossRef CAS PubMed.
- B. Zhou, Y. Hu and C. Wang, Angew. Chem., Int. Ed., 2015, 54, 13659–13663 CrossRef CAS PubMed.
- X. Kong, L. Lin and B. Xu, Adv. Synth. Catal., 2018, 360, 2801–2805 CrossRef CAS.
- S. L. Ko, E. Courtney, D. M. Light, D. D. Jones and D. G. P. McGlacken, Tetrahedron Green Chem, 2023, 2, 100019 CrossRef.
- Y. Wang, L. Zhu, Z. Shao, G. Li, Y. Lan and Q. Liu, J. Am. Chem. Soc., 2019, 141, 17337–17349 CrossRef CAS PubMed.
- Y.-Z. Wang, S.-D. Liu, L. Cheng, L. Liu and C.-J. Li, Org. Chem. Front., 2023, 10, 3021–3026 RSC.
- Y.-Z. Wang, Q. Liu, L. Cheng, S.-C. Yu, L. Liu and C.-J. Li, Tetrahedron, 2021, 80, 131889 CrossRef CAS.
- C.-C. Li, H. Wang, M. M. Sim, Z. Qiu, Z.-P. Chen, R. Z. Khaliullin and C.-J. Li, Nat. Commun., 2020, 11, 6022 CrossRef CAS PubMed.
- S.-S. Yan, L. Zhu, J.-H. Ye, Z. Zhang, H. Huang, H. Zeng, C.-J. Li, Y. Lan and D.-G. Yu, Chem. Sci., 2018, 9, 4873–4878 RSC.
- M. Peña-López, P. Piehl, S. Elangovan, H. Neumann and M. Beller, Angew. Chem., Int. Ed., 2016, 55, 14967–14971 CrossRef PubMed.
- A. E. Stiegman and D. R. Tyler, Inorg. Chem., 1984, 23, 527–529 CrossRef CAS.
- D. H. Nguyen, X. Trivelli, F. Capet, J.-F. Paul, F. Dumeignil and R. M. Gauvin, ACS Catal., 2017, 7, 2022–2032 CrossRef CAS.
- C. M. Hert, J. B. Curley, S. P. Kelley, N. Hazari and W. H. Bernskoetter, Organometallics, 2022, 41, 3332–3340 CrossRef CAS.
- A. T. Radosevich, J. G. Melnick, S. A. Stoian, D. Bacciu, C.-H. Chen, B. M. Foxman, O. V. Ozerov and D. G. Nocera, Inorg. Chem., 2009, 48, 9214–9221 CrossRef CAS PubMed.
- P. Schlichter and C. Werlé, Synthesis, 2022, 517–534 CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Detailed experimental procedures and characterization of all synthesized compounds. See DOI: https://doi.org/10.1039/d4gc01516a |
| This journal is © The Royal Society of Chemistry 2024 |