Deep eutectic solvents as green solvents for materials preparation
Dongkun
Yu
a,
Depeng
Jiang
b,
Zhimin
Xue
 *c and
Tiancheng
Mu
*c and
Tiancheng
Mu
 *d
*d
aDepartment of Applied Physics, KTH Royal Institute of Technology, Hannes Alfvéns väg 12, 114 19, Stockholm, Sweden
bShanghai Institute of Space Power-Sources, Shanghai 200245, China
cCollege of Materials Science and Technology, Beijing Forestry University, Beijing 100083, P. R. China. E-mail: zmxue@bjfu.edu.cn
dDepartment of Chemistry, Renmin University of China, Beijing 100872, P. R. China. E-mail: tcmu@ruc.edu.cn
First published on 16th May 2024
Abstract
The concept of sustainability has progressively evolved into a consensus for achieving long-term development, and the significance of solvents in regard to green chemistry and sustainable processes cannot be underestimated. Hydrogen-bonding interactions and heterogeneity at the nanoscale determine the outstanding solvating properties of deep eutectic solvents (DESs). A relatively wide electrochemical window is the prerequisite for the preparation of more materials (especially active metals) by electrodeposition in these eutectic systems. The role of DESs in the fabrication process may be chemical or physical, although the line between the two is blurred. At times, DESs function as a reducing agent, while in other cases they solely provide a liquid environment to facilitate the reaction. This review provides a set of methodologies to guide the preparation of materials with specific morphology and functions.
1. Introduction
As a new class of green solvents, deep eutectic solvents (DESs) have attracted much attention in recent years due to their low toxicity, biodegradability and tunable properties. DESs are usually defined as mixtures composed of two or more solid or liquid components that form eutectics at room temperature and can be used as solvents, as can be known from their names.1 The formation of DESs is based on the eutectic principle, i.e., forming the eutectic mixture with a lower melting point than each single component, in which hydrogen bonding interactions are attributed as the intrinsic driving force.2,3 Later on, concepts such as eutectic molecular liquids, type V DESs, halogen bonding DESs, supramolecular DESs, and eutectogels were proposed as well.4–8The most common method for preparing DESs involves mixing hydrogen bond donors (HBDs) and hydrogen bond acceptors (HBAs) in specific ratios.9 HBDs are usually quaternary ammonium salts or choline derivatives, while HBAs are carboxylic acids, metal salts, or polyols (Scheme 1).10 When mixed, HBDs and HBAs form hydrogen bonds, resulting in the formation of a eutectic mixture with a lower melting point than either individual component. However, with the deepening of relevant basic research, some other principles have also been discovered, such as the effect of entropy increase.3,11 The unique properties and potential applications of DESs make them a promising class of green solvents applicable to various fields, including the extraction and separation of biomass, materials preparation, organic chemical synthesis, pharmaceutics, genetic science, etc.12–16
Currently, DESs occupy a significant position in the synthesis of nanoparticles and nanomaterials. The unique hydrogen-bonding network between components in DESs creates a favorable environment for the nucleation and controlled growth of nanoparticles.17 This, furthermore, facilitates precise tuning of the size, morphology, and surface properties of nanoparticles, offering great potential for applications in fields such as catalysis and drug delivery, where the properties of nanoparticles play a key role.
Fig. 1 summarizes representative studies of materials prepared using DESs since 2004. Notably, early interest among researchers was directed towards the synthesis of Metal Organic Frameworks (MOFs), which may be related to the hot topics at that time.18–20 Some endeavours were focused on nanoparticle preparation as well. For instance, Liao et al. manipulated the morphology of gold nanoparticles by modulating the water proportion within the DES.21 It was not until 2017 that research on the mechanism of materials growth began to appear, which may be related to the research progress of neutron scattering technology in DES structure investigation.22,23 Subsequently, the topic of materials prepared by DES entered a new stage.
 | ||
| Fig. 1 Schematic graph showing milestone work of materials synthesized using DESs. ZIF analogues SIZ-2,18 reproduced with permission from ref. 18. Copyright 2004, Nature Publishing Group. Aluminophosphate Materials,19 reproduced with permission from ref. 19. Copyright 2006, Wiley-VCH. Gold nanoparticles,21 reproduced with permission from ref. 21. Copyright 2008, Wiley-VCH. Trivalent MOFs,20 reproduced with permission from ref. 20. Copyright 2009, Wiley-VCH. Layered zinc phosphates,28 reproduced with permission from ref. 28. Copyright 2010, Wiley-VCH. MPO4 (M = Co, Fe, Mn) framework,29 reproduced with permission from ref. 29. Copyright 2012, Royal Society of Chemistry. Ceria,22 reproduced with permission from ref. 22. Copyright 2017, Nature Publishing Group. Selenidostannates,30 reproduced with permission from ref. 30. Copyright 2018, Royal Society of Chemistry. Iron oxide,27 reproduced with permission from ref. 27. Copyright 2021, Royal Society of Chemistry, BiVO4 with Bi vacancies,31 reproduced with permission from ref. 31. Copyright 2022, Wiley-VCH. | ||
Researchers are deeply exploring the possibility of combining DESs with other cutting-edge synthesis techniques such as microwave-assisted synthesis and continuous-flow synthesis.24,25 This convergence could potentially enable unprecedented efficiencies and scalability in materials production. Furthermore, the application range of DESs has been extended to the synthesis of complex polymers and functional materials, taking full advantage of their intrinsic heterogeneity and macromolecular structures.
As the field advances, a critical focus will be on understanding the fundamental mechanisms underlying materials synthesis within DESs.26,27 These mechanisms enable researchers to not only harness the current capabilities more effectively but also to design novel DESs tailored specifically for unique materials synthesis challenges. The marriage of green chemistry principles, meticulous engineering, and innovative materials design is poised to reshape industries and technologies in ways that are sustainable, efficient, and at the forefront of scientific innovation. This review aims to summarize the application of DESs in the materials preparation process, starting from their unique and significant properties, combined with specific examples to propose how to design solvent systems according to various demands and purposes. At the end of the article, the authors also put forward the prospects for DESs in the materials preparation process.
2. Mechanisms and properties
2.1. Coulomb interactions and hydrogen bonding
The interaction form in DESs is quite complex because of the diversity of their single components. However, the main interactions are Coulomb interaction and hydrogen bonding interaction, and even multiple interaction modes. Despite some controversy, these non-covalent interactions are generally believed to be the cause of the lower melting point of the system. Hammond et al. studied a series of lanthanide nitrate hydrate-based DESs.32 Due to the excessive number of species in the liquid mixtures, the observed nanostructures of intercalated hydrogen bonds containing water and urea were considered as a special structure, the lubricating molecular pseudophase. This bipartite structure observation helps explain some unusual physical properties, such as low viscosity and high surface tension in type IV DESs. In another work, Ce3+ showed a different structure in the choline chloride and urea system from the above system.22 The intermolecular coordination number shows that chloride ions dominate the cerium ligands. The coordination ability of urea is enhanced, while the coordination ability of nitrate is weakened. The results show that the choline chloride (in reline) significantly changes the environment around cerium ions, which further determines the kinetics of their hydrolysis process.Similarly, urea can coordinate chlorides with strong hydrogen bonds. This forms a complementary choline shell around urea and chloride. In this way, due to the smaller spatial volume of the urea molecule, the binding probability is increased. In short, the results showed that reline has a strong and complex hydrogen bond network between species, which is consistent with previous NMR results.33 A cage structure was proposed to illustrate the hydrogen bond interactions of various strengths in the DES. The same complex hydrogen bonding networks were also reported in others’ work.34,35
To compare with the above work, Holbrey et al. conducted a series of neutron diffraction work on DESs under 338 K.36 The results from EPSR showed that the generated model is consistent with the experimental data at these moderately elevated temperatures. In reline, the role of choline's hydroxyl group as a hydrogen bond donor or acceptor is less important than previously reported at 303 K. The same hydrogen bond network also exists in other types of DES. In some DESs containing alcohols (especially polyols), this phenomenon may be particularly noticeable.37,38 Additionally, studies have shown that the structural network of glyceline is completely determined by its single content, glycerol, although the viscosity between the two is very different.39 The network dynamics in glyceline are defined by the glycerol–glycerol dynamic correlation, which may be due to the poly-hydroxyl structure of glycerol. In contrast, the choline–glycerol dynamic correlation function in glyceline shows largely uncorrelated fluctuations. These findings indicate that glyceline is actually a highly correlated hydrogen bond network of glycerol molecules, similar to the choline chloride–urea system. The difference is that the choline ion in glyceline occupies the interstitial space and has almost no structural or dynamic correlation with the glycerol network. Therefore, the local transport and kinetic properties of glyceline are determined by relatively loosely bound choline ions because they dominate the structure and collective dynamics of glyceline. A comprehensive study based on ethylene glycol–DES (ethaline) has also reached similar conclusions.40
The hydrogen bonding supramolecular structure of a DES can be studied by DFT calculations, and the orientation of HBD and HBA can be well visualized.41 A variety of information including electrostatic interactions, heat of formation, charge mobility, interaction energy, electron energy, zero-point energy, dipole moment, heat capacity, entropy, bond angles and dihedral angles can be provided. Among them, parameters such as bond length, bond angle, dipole moment and molecular energy play an important reference role. These properties mainly depend on the concentration of electron density to form nucleophilic sites, while electrophilic sites allow us to understand the properties that lead to physical and chemical phenomena. In addition, DFT calculations are often combined with molecular dynamics simulations to study the structural properties of DES.
When the HBD changes to a more polar carboxylic acid, a special structure, called “ionic domain”, will form in the bulk of the DES.42 In the malic acid–choline chloride system, the synergistic effect of them clamps the chloride anion through hydrogen bonding and electrostatic interaction, and then forms an ion cluster that is composited with the eutectic stoichiometry. This has been seen before in urea-based DES reline. However, the malic acid molecules with richer hydrogen bonding functionality (both hydrogen bonding donor and acceptor abilities) yield a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 eutectic. It further showed the malic acid-based DES is more resilient to dissociation into clusters of the HBD molecules than is seen in reline. This structure is stabilized to a certain extent by the hydrogen bond interaction between HBD and choline molecules. This charge delocalization cage is surrounded by 6–7 other such cages, which is very similar to the radial structure of ionic liquids forming variable charges. This provides a basis for the extension of the related structure and properties of ionic liquids to DESs. Understanding these interaction modes and structures of components in DESs is vitally significant for the crystal nucleation when designing and preparing materials.
1 eutectic. It further showed the malic acid-based DES is more resilient to dissociation into clusters of the HBD molecules than is seen in reline. This structure is stabilized to a certain extent by the hydrogen bond interaction between HBD and choline molecules. This charge delocalization cage is surrounded by 6–7 other such cages, which is very similar to the radial structure of ionic liquids forming variable charges. This provides a basis for the extension of the related structure and properties of ionic liquids to DESs. Understanding these interaction modes and structures of components in DESs is vitally significant for the crystal nucleation when designing and preparing materials.
2.2. Polarity and acidity
The solubility of solvents is widely believed to be related to their polarity, where the principle of similar compatibility (like dissolves like) is the proof. In addition to the solvation process, dissolution sometimes involves chemical reactions, for example, in the case of calcium carbonate dissolved in hydrochloric acid. The capability to dissolve many substances (various precursors) is a prerequisite for the synthesis process in these green solvents. DESs are believed to have excellent dissolving properties. There is even research on the solvation of nanoparticles in DESs, which shows their excellent solvation properties.43 Generally, there are two types of dissolution processes. One does not destroy the structural unit of the substance itself. The other completely dissociates substances into corresponding ions. Solutes in the former process are mostly molecular compounds, such as some natural products; the latter are mostly ionic compounds, such as metal oxides.44–47 It is worth noting that in some dissolution processes, the formation of small molecules (such as water) is also involved. Therefore, the polarity of a DES largely determines the scope of substances to be dissolved.Although they are considered to be substitutes for volatile organic solvents and have great potential in various applications, detailed knowledge of the dipolarity afforded by DESs was lacking before 2014. Understanding the polarity of DESs is essential if they are to be used as green alternatives to various volatile organic compounds in industry and academia. In terms of composition, conventional DESs are composed of molecular compounds (i.e., urea) and ionic compounds (i.e., choline chloride). Therefore, the polarity of DESs should be higher than that of ordinary polar solvents and similar to the polarity of ionic liquids, which is why they are called ionic liquid analogues. So, how polar are DESs? Pandey and coworkers explored the behaviour of well-established solvent polarity probes dissolved in four choline chloride-based DESs.48 Fluorescence dye maxima of emission and excitation along with the Stokes shifts in chosen DESs and several other common and popular solvents are compared. Normalized dipolarity can be determined by eqn (1) and (2),48
 | (1) |
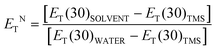 | (2) |
Such solvation properties of DESs are believed to be difficult to define precisely and impossible to express quantitatively, whereas Kamlet–Taft solvatochromic parameters gained popularity later. Among them, α represents the hydrogen bond donor ability, β represents the hydrogen bond acceptor ability, and π* (determined by the shift of the absorption band of different probes) represents the polarity of solvents.50 Marrucho et al. studied the polarity of different families of DESs by using solvatochromic responses of UV-vis absorption probes.51 The results were similar to those obtained by normalized polarity.48 HBA dominates the polarity of DESs, while the hydrogen bonding basicity in their studied DESs does not vary much within the same HBA. Further results showed that the DES displays a high capacity to donate and accept protons when compared with common solvents (including ILs). They concluded that the polarity of DESs can be easily designed by choosing their single components.
In addition to optical spectroscopy, chromatography is also a good method for studying molecular interactions in DESs. Such method has been proved to perform well in solvents such as ionic liquids, so it has been extended to the field of eutectic systems.52 Anderson et al. conducted the first study to measure chromatographically solute–solvent interactions of some carboxylic acid-based DESs.
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) SP = log SP = log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K = c + eE + sS + aA + bB + lL K = c + eE + sS + aA + bB + lL | (3) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) SP refers to some property of a series of solutes in a fixed phase (or phases). For example, SP could be L, the gas–liquid partition coefficient for a series of solutes in a given liquid.53 Thus in eqn (3), k refers to the retention factor of each probe molecule for the DES stationary phase at a specific temperature. The retention factor is calculated chromatographically by measuring the retention time of each solute as well as the column dead volume. Factors represented by capital letters (E, S, A, B, and L) are solute descriptors: E, excess molar refraction calculated from the solute's refractive index; S, solute dipolarity/polarizability; A, solute hydrogen bond acidity; B, solute hydrogen bond basicity; and in Anderson's work, and L is solute gas–hexadecane partition coefficient at 298 K.50 Retention factors and solute descriptors are used to measure the system constants (e, s, a, b, l) that characterize the multiple solvation interactions of the solvent: e is the ability of the solvent to interact with π- and n-electron pairs of the solute; s describes the dipolarity/polarizability of the solvent; a is the measure of the hydrogen bond basicity; b is the hydrogen bond acidity; and l describes the difference between the hydrophobicity or lipophilicity of the two phases.50,53
SP refers to some property of a series of solutes in a fixed phase (or phases). For example, SP could be L, the gas–liquid partition coefficient for a series of solutes in a given liquid.53 Thus in eqn (3), k refers to the retention factor of each probe molecule for the DES stationary phase at a specific temperature. The retention factor is calculated chromatographically by measuring the retention time of each solute as well as the column dead volume. Factors represented by capital letters (E, S, A, B, and L) are solute descriptors: E, excess molar refraction calculated from the solute's refractive index; S, solute dipolarity/polarizability; A, solute hydrogen bond acidity; B, solute hydrogen bond basicity; and in Anderson's work, and L is solute gas–hexadecane partition coefficient at 298 K.50 Retention factors and solute descriptors are used to measure the system constants (e, s, a, b, l) that characterize the multiple solvation interactions of the solvent: e is the ability of the solvent to interact with π- and n-electron pairs of the solute; s describes the dipolarity/polarizability of the solvent; a is the measure of the hydrogen bond basicity; b is the hydrogen bond acidity; and l describes the difference between the hydrophobicity or lipophilicity of the two phases.50,53
It was found that DESs composed of larger molar ratios of low pKa HBAs result in systems with higher hydrogen bond acidity. Further optical spectroscopy experiments showed that an increase in temperature led to reduced acidity of HBDs in choline chloride-based DESs.54 However, the dipolarity/polarizability and the basicity of HBAs did not change with the temperature of DES.
Some nuclear magnetic resonance probes (such as pyridin-N-oxide) can also be used to determine the solvatochromic parameters of DESs.55 The understood strong intrinsic interaction (hydrogen bonding interaction) determined by these solvation studies can be used to interpret the excellent extraction performance of DESs. More importantly, it makes it possible to predict and understand the dissolving properties of DESs, and finally, guide the synthesis of task-specific solvents.
Some other properties of DES are determined by polarity as well. Jackson et al. investigated the structures of proteins in a DES and its water mixtures by circular dichroism and small-angle neutron scattering.56 Based on the results of protein conformation in the DES, they put forward a hypothesis: the conformation of protein depends on the solvation environment of amino acids that promote protein folding, and these amino acids are likely to be responsible for their enzymatic activity. In DESs, HBDs replace the water surrounding the amino acid in the active site, which promotes a partially folded conformation and reduces activity through a combination of specific binding and reduced solvophobic effect. In addition, in the case of hydrated DESs, water may form a shell around these active sites, allowing the protein to fold in the buffer. The solvated electron has also been observed in DES, and this provides the possibility of enzyme-catalyzed reactions in DESs.57
According to a publication by Milano et al., the photosynthetic reaction center of bacteria (Rhodobacter sphaeroides) in DESs can generate a charge-separation state and even perform its natural light cycle.58 This discovery not only verified the good maintenance of the protein structure in DESs, but also implied that DESs possess excellent electron solvation and transport properties.57 When considered for electrochemical applications, fast electron transfer in DESs is a strong advantage. Different types of solvent systems are critical to energy storage density. In Hapiot's work, the electron transfer rate constant measured in ethaline is close to that reported for the oxidation of ferrocene or ferrocyanide in water in an organic solvent.59 This shows that from the point of view of electron transport rate, DES (at least ethaline) seems to be more similar to molecular solvents than ionic liquids. In addition, the report of ultra-stable free radicals in DESs illustrates their great potential applied in electrochromic devices.60
2.3. Heterogeneity and nanostructure of DESs
The permanent dipoles of polar solvents give them polarization ability, which tends to concentrate the charge of covalent bonds, or separates ionic compounds into solvated ions. The value of the dielectric constant (ability to separate charge) increases with the increase in the permanent dipole of the molecule. Given that HBDs are usually polyols, poly-acids or amides, DESs theoretically belong to a class of protic solvents. However, the presence of ionic regions in DESs allows the local polarity to reach the level of ionic compounds. Therefore, they cannot be simply classified into any conventional solvent class.Heterogeneous and amphiphilic nanostructures have been shown to play a pivotal role in ionic liquids, which had not been experimentally proved in DESs before 2017.61 Through the application of some experimental techniques such as neutron diffraction and ultrafast spectroscopy, these properties were gradually unveiled.62–65 In this way, DESs can be better utilized by designing the characteristics related to their nanostructures.
Kuroda et al. conducted a thorough investigation on a series of amide-based DESs, trying to find evidence of molecular heterogeneities in such systems.66 In principle, the ultrafast kinetics is attributed to the formation and breaking of hydrogen bonds between amides. A slow dynamic component observed in the time evolution of the photon echo signal was considered to be related to the asymmetry and polarity of the HBA. Interestingly, it was found that the level of heterogeneity in apolar domains increased with the asymmetry in the geometry and the strength of interaction between HBA and HBD. Domains created by heterogeneity slowed the formation and breakage of hydrogen bonding interactions, thereby rationalizing the overall dynamics.
Neutron diffraction has proved to be an effective technique for studying the molecular-level structure of a liquid system. The experiment conducted by Atkin et al. revealed the phenomenon of cationic alkyl chain association in DESs.67 As the chain length increases, this association becomes stronger, indicating the higher the degree of amphiphilicity of DESs. Understanding this amphiphilic ability contributes to the design of DESs, and to dissolving more diverse solutes.
In addition, some ingenious experimental methods from ionic liquids have been carried out in DESs, which benefits the study of their spatial heterogeneity.68 The electrostatic field can be estimated from infrared experiments based on the Stark effect, which can be used as an effective method to illustrate the microscopic heterogeneous solvation of DESs. Bagchi et al. investigated four choline chloride-based DESs (the HBDs are diols with different lengths of alkyl chains).69 The results showed that the total electrostatic field can be decomposed into contributions from HBA and HBD constituents to obtain an in-depth understanding of the nanoscale heterogeneity of the DESs around the solute molecule. Compared with neutron scattering and ultrafast spectroscopy, infrared spectroscopy experiments are easy to operate, and the results can also be supported by molecular dynamics simulations.
Hydrophobic DESs have received widespread attention since their proposal. The most typical example of these novel DESs is DL-menthol and lauric acid. Kashyap et al. performed atomic and molecular dynamics simulations to understand the bulk phase morphology that is present in the hydrophobic eutectic solvent based on DL-menthol.70 The pre-peak in simulated total X-ray scattering structure function indicated the presence of intermediate-range structural heterogeneity in the studied hydrophobic DESs. This further indicated the existence of polar regions composed of only polar groups, and these domains were distributed in a non-polar environment formed by the non-polar groups of DL-menthol and the alkyl tail of the HBD. That is to say, the reason for the hydrophobicity of these DESs is that the intrinsic “huge” non-polar regions hinder the interaction between the polar regions and water, thereby hindering the solvation process. In addition, the results of real-space correlations and angular-resolved radial distribution function indicated that the structures of these DESs were dominated by strong hydrogen bonding interactions between all species. All these balance correlations control the overall stability of the DES, thus hindering the crystallization of the system. This is consistent with the results from neutron diffraction.23,71 In addition, studies have shown that there is also time heterogeneity in DESs, which is still significant even at temperatures 100–160 K higher than their glass transition temperatures.72,73
Before 2017, although there were some comprehensive studies on the ontological properties of DESs, there was almost no research on their structure and properties at the interface. Understanding the behaviour of these solvents on the energized interface will facilitate their application in electrochemistry. Kaur et al. applied all-atom molecular dynamics simulations to explore the morphology and nanostructure of reline in bulk and interfaces.74 Studies have shown that urea molecules exhibit different orientations near the positively charged and negatively charged electrodes, which may be related to the carbonyl group (π electrons) in urea. There is a significant short-range interaction between the neutral graphene electrode and choline in reline. The existence of the multilayer structure of the reline near the charged electrode is also reflected in the electrostatic potential and electric field distribution. More importantly, the calculated differential capacitance reveals the dependence of electrostatic properties on the shape and size that molecules exhibit near the electrodes. Within a year, a similar work was reported. The results of De Proft et al. further indicated that the distinct orientation of urea in the electric double layer is determined by the charge on the surface.75 The difference of ±0.32 C m−2 on the electrified surface causes a 90° rotation of urea. These results provided new insights into the electric double layer structure of DESs, whose interface structure can be adjusted and designed at the molecular level.
Fig. 2 shows two pioneering works illustrating the heterogeneity of DESs. Conducted by Kaur and colleagues, the simulation efforts encompassed a molecular dynamics study investigating the X-ray and neutron scattering structure of DES (acetamide + LiClO4, molar ratio = 81![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19).61Fig. 2a reveals that the studied DESs exhibit distinct spatial heterogeneity at the molecular length scale. The result clearly indicated that both LiClO4–LiClO4 correlations and the RCONH2–RCONH2 term significantly influence the pre-peak phenomenon, underlining the substantial role played by potent electrostatic interactions in shaping the structural characteristics of these systems. Conversely, the principal peak predominantly stems from the self-correlations of RCONH2. However, both LiClO4–LiClO4 and RCONH2–LiClO4 contributions moderately impact the principal peak. The accompanying figure illustrates the consistent nature of the electrolyte's influence on the pre-peak, irrespective of variations in alkylamide tail length. Similarly, the alkylamide contribution remains unaltered despite tail modifications. The nanoscale-sized extended domains of LiClO4 (highlighted in red) are readily discernible within the equilibrium snapshots of the depicted DES in Fig. 2b. The system exhibits pronounced heterogeneity, marked by the uneven distribution of alkylamide molecules and ions. Since then, a number of articles investigating microscopic heterogeneity have emerged.
19).61Fig. 2a reveals that the studied DESs exhibit distinct spatial heterogeneity at the molecular length scale. The result clearly indicated that both LiClO4–LiClO4 correlations and the RCONH2–RCONH2 term significantly influence the pre-peak phenomenon, underlining the substantial role played by potent electrostatic interactions in shaping the structural characteristics of these systems. Conversely, the principal peak predominantly stems from the self-correlations of RCONH2. However, both LiClO4–LiClO4 and RCONH2–LiClO4 contributions moderately impact the principal peak. The accompanying figure illustrates the consistent nature of the electrolyte's influence on the pre-peak, irrespective of variations in alkylamide tail length. Similarly, the alkylamide contribution remains unaltered despite tail modifications. The nanoscale-sized extended domains of LiClO4 (highlighted in red) are readily discernible within the equilibrium snapshots of the depicted DES in Fig. 2b. The system exhibits pronounced heterogeneity, marked by the uneven distribution of alkylamide molecules and ions. Since then, a number of articles investigating microscopic heterogeneity have emerged.
 | ||
| Fig. 2 Simulation and experiment investigation of heterogeneity in DESs. (a) Partial X-ray scattering S(q) for DES (acetamide + LiClO4). (b) Equilibrium snapshots of the DES (the Li+/ClO4− ion pairs are shown as a red isosurface in all of the systems, while the acetamide molecules are shown in green).61 Reproduced with permission from ref. 61. Copyright 2016, American Chemical Society. (c) Lateral force vs. normal force of mica in air (black), ChCl/BuOOH (blue), and ChCl/HeOOH (red). The friction coefficient (m) is derived from the slope of the plot within the boundary regime. The inset illustrates a representative schematic depicting the shift from the multilayer regime to the boundary regime when considering ChCl/HeOOH on mica. (d) 5 nm by 5 nm AM–AFM phase image of mica immersed in ChCl/HeOOH. Insert: the 2D fast Fourier transform image and radial average value.76 Reproduced with permission from ref. 76. Copyright 2022 Elsevier. | ||
In Buzolic's work, the structure of DESs formed by mixing ChCl with butyric acid (ChCl–BuOOH) and hexanoic acid (ChCl/HeOOH) at a molar ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 was studied, attempting to investigate the effect of carbon chain length on heterogeneity.76 With the aliphatic chain increases from C4 to C6, the nanostructure becomes larger and more defined, and the AFM force curves show that the interfacial nanostructures extend farther from the surface (Fig. 2c and d). The AFM force curves depict an expansion of the interfacial nanostructures away from the surface. The self-assembled nanostructures in these systems emerge from the convergence of choline cations, chloride anions, and acid-alcohol groups, yielding polar domains through electrostatic attraction and hydrogen bonding. Within this context, alkyl chains endowed with carboxylic groups are effectively excluded from polar domains, congregating instead within nonpolar domains.
4 was studied, attempting to investigate the effect of carbon chain length on heterogeneity.76 With the aliphatic chain increases from C4 to C6, the nanostructure becomes larger and more defined, and the AFM force curves show that the interfacial nanostructures extend farther from the surface (Fig. 2c and d). The AFM force curves depict an expansion of the interfacial nanostructures away from the surface. The self-assembled nanostructures in these systems emerge from the convergence of choline cations, chloride anions, and acid-alcohol groups, yielding polar domains through electrostatic attraction and hydrogen bonding. Within this context, alkyl chains endowed with carboxylic groups are effectively excluded from polar domains, congregating instead within nonpolar domains.
The phase behaviour of both bulk and confined DESs was also investigated. Gardas et al. used non-ionic surfactant (Tween-80) and cyclohexane to study the reverse micellar system based on DES (benzyltripropylammonium chloride![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethylene = 1
ethylene = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3).77 All the vibrational modes of HBA were found to be significantly red-shifted in the DES compared with pure substance, which indicated the strong intermolecular association between HBAs and HBDs during the formation of the eutectic system. Laman spectra indicated a disruption of hydrogen bonding interactions between HBAs and HBDs when forming a reverse micellar system. In another work, the phase behaviours of both bulk and confined DESs in controlled pore glasses were also investigated.78 Lan et al. conducted a thorough test on the thermodynamic properties of reline, the most typical DES. Differential scanning calorimetry revealed that under nanoconfinement, the melting points and enthalpies are reduced, while the cold crystallization temperatures become higher. A new crystal structure of reline in the 50 nm pores was proved by X-ray diffraction analysis. Similar to the reverse micellar result, the thermodynamic and kinetic properties of reline manifested the significant role played by the hydrogen bonding interactions when forming the eutectic system. Understanding the structures and thermodynamic properties of DESs beyond their bulk lays the foundation for expanding their new applications.
3).77 All the vibrational modes of HBA were found to be significantly red-shifted in the DES compared with pure substance, which indicated the strong intermolecular association between HBAs and HBDs during the formation of the eutectic system. Laman spectra indicated a disruption of hydrogen bonding interactions between HBAs and HBDs when forming a reverse micellar system. In another work, the phase behaviours of both bulk and confined DESs in controlled pore glasses were also investigated.78 Lan et al. conducted a thorough test on the thermodynamic properties of reline, the most typical DES. Differential scanning calorimetry revealed that under nanoconfinement, the melting points and enthalpies are reduced, while the cold crystallization temperatures become higher. A new crystal structure of reline in the 50 nm pores was proved by X-ray diffraction analysis. Similar to the reverse micellar result, the thermodynamic and kinetic properties of reline manifested the significant role played by the hydrogen bonding interactions when forming the eutectic system. Understanding the structures and thermodynamic properties of DESs beyond their bulk lays the foundation for expanding their new applications.
Quaternary ammonium salts such as choline chloride are usually used as HBA to construct DESs. The length of the aliphatic chain not only affects the melting point and other properties of the system, but also determines the solvent kinetics of DESs. One of the reasons why ionic liquids are liquid at room temperature is that their asymmetric structure hinders the crystallization process. As the analogues of ionic liquids, do DESs follow similar laws? Samanta et al. investigated the liquid structure of different DESs (HBAs with various chain lengths) by monitoring the fluorescence response of probe molecules.79 The results indicated that the spatial and dynamic heterogeneity in DESs were more pronounced with longer alkyl chains, which was consistent with the recently reported results.80 The translational and rotational diffusion dynamics of selected probe molecules indicated that heterogeneity arises through the formation of molecular-scale domains, which becomes more pronounced in solvents with longer chain lengths.
Similarly, several works exploring the effect of alkyl chains in HBD were also reported. Bagchi et al. conducted 2-dimensional IR spectroscopy research on three DESs (HBAs are 1,2-ethanediol, 1,3-propanediol, and 1,4-butanediol).81 The result showed that as the length of the hydrocarbon chain increases, a reasonable reason for the slower solvent kinetics is the difference in viscosity of the DES. The fast component in the solvation kinetics was attributed to the fluctuation of the solvent in the first solvation shell, whereas the centre of mass of the molecule did not change significantly. Therefore, the time scale of the diffusion motion depends on the DES viscosity, which is consistent with what Samanta et al. reported.79 In another work, Samanta and Hossain studied the kinetics of hydroxyl localization (on HBD) on their diffusion.80 Compared with the length of the alkyl chain, the spatial and dynamic heterogeneity changes with the position of the hydroxyl group are insignificant.
Vis et al. developed a statistical model combining theoretical and experimental results to predict the non-ideal behavior of eutectic mixtures.82 Studies have shown the existence of a strong correlation between the non-ideality of the system and the volume of the quaternary ammonium salt, and smaller ionic bonds will lead to stronger interactions. Due to the shorter side chain the distance between N+ and Br− is reduced, and Br− will experience a stronger interaction than the –OH of carboxylic acid or polyol. Understanding the solvation properties and solvent dynamics of DESs is conducive to designing task-specific solvent systems for various applications. Knowing the interaction between components at the molecular level lays the foundation for the synthesis of molecules and materials in DESs.
2.4. Stability and reactivity
The relatively high thermal stability of DESs is of great value because it directly affects the sustainable application of these solvents at high temperature and the stability of the materials synthesis processes. High temperature is necessary in many synthetic reactions and industrial applications, so the thermal stability of DESs is crucial to ensure the efficiency and quality of synthesis.On the one hand, the thermal stability of DESs is related to their reliability as reaction media. Some separation and synthesis processes need to be performed at relatively high temperatures to achieve the desired product conversions and selectivities.83,84 If DESs decompose at undesired temperatures, it will lead to reaction failure or product with low purity. Therefore, DESs need to be stable at high temperature to ensure the smooth progress of the reaction. On the other hand, thermal stability is crucial for the control of materials synthesis. The preparation of many advanced materials requires high-temperature conditions to achieve the desired crystal growth, phase transition or other physicochemical changes. If DESs decompose during the synthesis process, it may affect the structure and properties of the material, and even lead to unpredictable results. Therefore, the use of DESs with good thermal stability during materials preparation helps to ensure the consistency and controllability of the synthesized materials.
Chen et al. studied the thermal stability of 40 DESs and found that the thermal stability of HBD has a crucial influence on the Tonset of the system.85 In general, the DES will first break down into HBD and HBA because of the weakness of hydrogen bonding interactions at high temperature. Subsequently, HBD, which is less stable, starts to volatilize or decompose; HBA also volatilizes or decomposes, but at a higher temperature. Hydrogen bonding plays an important role in the thermal stability of the DES, and it hinders the “escape” of molecules, so that it requires greater energy to decompose than pure HBA and HBD, which causes the Tonset of the DES to shift to a higher temperature. For the first time, Janković et al. presented the physicochemical elucidation of the overall thermal decomposition mechanism and the effect of enthalpy–entropy compensation on the emerging reaction mechanism for investigating type II DES (MgCl2·6H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ChCl).86 It was found that the thermodynamic principle of the enthalpy–entropy compensation is the source of the kinetic compensation effect, which is the consequence of the molecular interaction effect during the thermal decomposition of type II DES. The key phenomenon in the complex multi-step process is the parallel dehydration steps of MgCl2·6H2O in DES, corresponding to the above work by Chen and coworkers.
ChCl).86 It was found that the thermodynamic principle of the enthalpy–entropy compensation is the source of the kinetic compensation effect, which is the consequence of the molecular interaction effect during the thermal decomposition of type II DES. The key phenomenon in the complex multi-step process is the parallel dehydration steps of MgCl2·6H2O in DES, corresponding to the above work by Chen and coworkers.
For some DESs containing carboxylic acids (especially dicarboxylic acids), the decomposition temperature is even lower than the preparation temperature of the DES.87 This puts forward new requirements for the preparation method of the DES, and the grinding method and ultrasonic method came into being. Although many impressive results have been obtained, research on thermal stability has not stopped. High-resolution TGA experiments were performed by Liu et al. which provide a clearer view than both the slow and fast heating rate in conventional TGA.88 The long-term thermal stability of DESs was also investigated.89
Electrochemical stability is very significant as well. The electrochemical stability of solvents, especially those with broad electrochemical windows, is of great significance in practical production and wide applications. The electrochemical window refers to the potential range in which electrochemical reactions can be performed in a solution, and a wide electrochemical window is beneficial for:
1. Realizing multiple reactions in the same system
2. Improving the control and stability of the reaction
3. Providing a wider working potential range.
Li et al. investigated the electrochemical potential window of 24 choline-based DESs, and the choline–methyl urea system has the largest value, which is 4.72 V.91 The broad electrochemical window of DESs makes their application in electrochemistry more diverse and flexible. It not only promotes the integration of various electrochemical reactions, but also provides support for high efficiency, stability and innovation in actual production, and promotes technological development in the field of materials preparation. In addition, the radiation stability of DESs was investigated, as was the study of ionic liquids.92,93 There are also some publications about the corrosivity of DESs to containers or electrodes, which is very important.94
2.5. Effect of temperature and water content
The influence of temperature on the properties of solvents is huge. For example, it will change the viscosity and then change some transport properties. Temperature also has a great influence on the intrinsic polarity and internal noncovalent interactions of DESs. It was reported that an increase in temperature leads to reduced acidity of HBDs in choline chloride-based DESs.95 However, due to various reasons, such as the instability of the probe in a DES, it seems difficult to achieve a unified result on polarity parameters of DESs.54 Unlike the polarity parameter (which contains many parameters), the result of heterogeneity is relatively straightforward. Kashyap et al. explored the molecular-level structures of DESs and the variation with temperature by molecular dynamics simulation on X-ray scattering and neutron diffraction.61,90 The result showed that increasing temperature leads to the increase of nanoscale heterogeneity because of ion-pair segregation in the studied DESs. In the X-ray scattering results (Fig. 3a), the left peak in a curve, called pre-peak or first sharp diffraction peak, represents nanoscale structural organization, and corresponds to self-segregated domains of Li+/ClO4−. The results of radial distribution function (Fig. 3b and c) show that whether it is ions of the same charge or ions of different charges, as the temperature increases, the nearest neighbouring peak is slightly shorter. In addition, as the temperature increases, the height of the peak increases significantly, which means stronger ion pairing and ion-pair segregation at higher temperatures. In the acetamide/lithium perchlorate system, the molecular-ion behaviour is exactly the opposite (Fig. 3d). In short, increasing temperature led to the increase of nanoscale heterogeneity because of the ion-pair segregation in the studied DESs. | ||
| Fig. 3 (a) Temperature dependence of X-ray scattering structure functions for acetamide + Li+/ClO4−. Temperature-dependent radial distribution functions for (b) Li+–Cl, (c) Li+–Li+, and (d) Li+–OR. OR is the oxygen atom in acetamide, and Cl is in ClO4−.90 Reproduced with permission from ref. 90. Copyright 2018, American Chemical Society. | ||
Similarly, the hydrophobicity of DES also changes with temperature. Longeras et al. reported a thermal-switchable smart DES.96 When the temperature of the system is higher than 26 °C, the original DES–water system will separate to two phase. In other words, the hydrophilicity of DES decreases with increasing temperature. Considering that both ammonium and oleate ions are more soluble than their neutral form in water, this phase separation phenomenon was believed to be induced by a decrease in ionicity. It is worth mentioning that the value of the DES–H2O system lies not only in the regulation of viscosity and polarity, but also in reducing the cost of solvents used for synthesis.
3. Solvothermal method
The solvothermal method based on DESs is not limited by water, achieving a wider range of chemical environments, which is helpful for the synthesis of different materials. For example, product crystal structure and morphology can be controlled at higher temperatures and pressures. In addition, DESs are generally considered to have good solubility and miscibility, which facilitates dissolving the reactants, thereby enabling processes that cannot be carried out in aqueous systems.3.1. Metal organic frameworks
In 2009, Zhang and coworkers reported the structure-directing roles of DESs, followed by investigating their performance in gas capture.20 As shown in Fig. 4, the first role observed in compound 1 is the introduction of cations and anions. In 1, the cationic choline ion acts as the extra framework structural directing agents (SDAs), while the chloride ion binds to the polymer layer. The second role was observed in the three isomorphic compounds, in which the chloride ions are the extra framework SDAs. The neutral urea molecule is bonded to the framework. The third effect observed in compound 3 is opposite to the second one, because choline ions bind to the framework through their hydroxyl groups, and the molecule acts as an SDA outside the framework. Finally, the fourth role observed in compounds 4–6 is the direct binding of neutral molecules to the polymer backbone. TGA showed the main structure of selected MOF (compound 6) possesses a pretty high thermal stability (almost 0 weight loss up to 550 °C), and the N2 adsorption/desorption study revealed its microporous structure. Further tests showed the BET and Langmuir surface areas are 186.3 and 261.5 m2 g−1, respectively. Since 2006, many synthetic paradigms and methodological works have been reported in the development of synthesizing framework compounds in DESs.29,97,98 | ||
| Fig. 4 The coordination shell of the metal central and the guest component in (a) 1, (b) 2, (c) 3, (d) 4, (e) 5, (f) 6, where O is in black, N is in gray, and O1w represents water oxygen atom. (g) Chemical structure of different ligands.20 Reproduced with permission from ref. 20. Copyright 2009, Wiley-VCH. | ||
3.2. Nanoparticles
It has been demonstrated that the choice of solvents used to synthesize nanoparticles can affect their surface chemical composition, morphology, and fluorescence characteristics. Genç et al. used a multiple-solvent system to prepare hydrophilic and hydrophobic carbon dots through solvothermal synthesis.99 In this method, carbon dots were prepared in a DES, while the traditional solvent system was used for separation. The waste products of centrifugation can be minimized, and the particle recovery rate was increased by 90%. Moreover, the photoluminescence and electronic characteristics of the synthesized carbon dots were adjustable. The hydrophobic and hydrophilic carbon dots reported here can be used in functional textiles, electronic products, and biomedical imaging.Nitrogen fixation is an important reaction that causes significant energy consumption every year. Therefore, it is very valuable to find catalysts with low cost and high efficiency. Electrocatalysis of nitrogen to ammonia at ambient temperature is a desired way for tackling the issue. Zhao and coworkers reported the DES-solvothermal synthesis of nanostructured Fe3S4via a one-pot reaction.100 The obtained Fe3S4 catalyst exhibits high NH3 yield (75.4 μg h−1 mg−1 cat.) and faradaic efficiency (6.45%) at −0.4 V vs. reversible hydrogen electrode.
Synthesizing other particles with adjustable size in DESs is also possible. Jiang et al. reported for the first time the use of PEGylated DES for the solvothermal synthesis of nickel–cobalt sulfide.101 Compared with the traditional vulcanization route, this route is cost-effective and energy-efficient, combining solvothermal synthesis and vulcanization processes. The structure and composition of the synthesized sulfide can be adjusted by adjusting the ratio of the reactants. PEGylated DES was believed to play multiple roles, including as a solvent, shape control agent and sulfur source in the synthesis of sulfides. The prepared sulfide exhibited excellent oxygen evolution reaction performance, almost no deactivation after 2000 cycles, while the Faraday efficiency was higher than 95%. This research provides a new method for synthesizing layered-structured metal sulfides for energy conversion and storage applications. Torrente-Murciano et al. proposed a new method for preparing V2O5 with different morphologies using a DES (reline).102 This work overcame the dependence of current synthesis methods on shape-directing agents (such as surfactants). Selective formation of V2O5 3D beads, 2D nanosheets and 1D randomly arranged nanowires can be achieved by tuning the water content in reline. The results of electrospray ionization mass spectrometry proved the formation of alkylamine-based ionic species during the heating process, and these ionic species play a significant role in the resulting material morphology with template and exfoliation characteristics. The nanostructured V2O5 cathode showed a fast charge–discharge response, which makes it a potential electrode for batteries.
3.3. Composite materials
DESs have been used as green and sustainable media, acting as solvents and structure inducers when synthesizing composite nanomaterials. Pal et al. reported a facile approach for the fabrication of anisotropic bimetallic gold–copper nanostructures by using reline.103 The precursor (CuCl2) was found to impact the reduction kinetics of the reaction, and further affect the morphology and elemental composition of final product. The fabricated nanoparticles revealed a high SERS signal for crystal violet molecules adsorbed at their surfaces. Zhang et al. developed an electrochemical activation process to trigger a reversible phase change reaction and surface reconstruction to synthesize highly active and stable self-supporting Ag nanoparticles grown on a Cu substrate.104 Activated Ag NPs/Cu have a larger surface area, more accessible active sites, and strong metal–support interaction with tailored atomic/electronic structure, thereby improving electrochemical catalytic activity. The electrode optimized by the subsequent ion-exchange method shows ORR performance equal to or better than that of the most advanced Pt/C and has excellent electrochemical stability and methanol tolerance.Fu et al. reported the use of DES (ethaline) to synthesize tungsten oxide–molybdenum oxide composite materials.105 The synthesized material is composed of agglomerated particles, and the crystal size ranges from ∼56.0 nm to ∼68.0 nm, depending on the reaction temperature. UV-vis irradiation test showed the composite material possesses photochromic properties with good reversibility and cyclability. Compared with the conventional method of preparing metal oxides, the synthesis temperature of the above work is much lower.106 In another work, a flower structure g-C3N4/BiOCl/BiVO4 with a ternary heterojunction was synthesized by a solvothermal method in DES.107 The synthesized nanoflowers showed excellent catalyst stability and extraordinary durability on oxygen evolution reaction. Such ternary heterojunction was demonstrated to extend the life of the photogenerated charge, and improve the separation efficiency of photogenerated electron–hole pairs. The above work provides new insights into the design of catalysts using green solvents. Similarly, a Ni–Fe layered double hydroxide nanosheets/sulfur-doped graphite carbon nitride heterostructure can be prepared in DESs.108 The combination of the layered structure provides a synergistic quantum confinement effect and promotes abundant active sites. The development of advanced architectures with lower energy requirements and minimal by-products signifies the superior characteristics of synthetic methods within the guidelines of green chemistry.
3.4. Specific structures
Gan et al. demonstrated an efficient method for preparing N/O self-doped hollow carbon nanorods from ternary DES.110 The open hollow nanorod structure provides high capacity and long-term cycling stability. Similarly, Pérez-García and coworkers reported a work in which hydrophobic DESs were used to synthesize polyacrylamide macroporous materials. The γ-Fe2O3 nanoparticles precipitated on the porous surface of high internal-phase emulsions, thereby providing the final materials with magnetic properties and electrostatic performance.111 Dye adsorption experiments showed that the synthesized material has a strong adsorption capacity (>350 mg g−1) and good recycling performance. This functional material is expected to be used for precision-controlled cleaning robots.Materials with high surface area, abundant active sites, and enhanced electrical conductivity are highly desired for supercapacitors. Ding et al. reported a method of K cation-intercalated birnessite-type MnO2via a facile room-temperature wet-chemical process.109 The DES used is a quaternary component system: monoethanolamine (MEA), diethanolamine, and triethanolamine are used as HBDs, and ChCl is used as HBA. During the preparation, K+ can be pre-intercalated into the birnessite-type MnO2 layer by adding ethanolamine-based DES (Fig. 5a). In addition, the morphology, crystallinity, and porosity of manganese oxides can be effectively tuned by changing the type of HBD in the DES (Fig. 5b–e). The as-prepared MnOx material exhibited the best electrochemical performance, including high specific surface area (220 m2 g−1), high specific capacitance (290 F g−1), and high stability (92.5% retention over 8000 cycles). In another work, a DES-assisted method of fabricating BiVO4 photoanode with bismuth vacancies was proposed, which can significantly enhance the charge diffusion coefficient by 5.8 times, thereby enhancing the charge transport efficiency (Fig. 5f–k).31 As is shown in Fig. 5f, the hydrogen bond network between ethylene glycol and choline chloride is the driving force in the preparation process. At the solid–liquid interface, the crystal lattice energy is weakened and Bi atoms are leached out by Cl− ions. The charge transfer efficiency was further enhanced by loading the CoBi cocatalyst, achieving 4.5 mA cm−2 at 1.23 V compared with the reversible hydrogen electrode under AM 1.5 G illumination. The above work refines cation defect engineering to enable the same potential for charge transport properties of different types of semiconductor materials for solar fuel conversion.
 | ||
| Fig. 5 Inserting and leaching processes conducted in DESs. (a) Schematic illustration for electrochemical reaction process of K inserted birnessite-type MnO2 electrodes. (b) XRD patterns of MnOx–CM, MnOx–CD, MnOx–CT, and MnOx. (c) SEM, (d) TEM images and (e) EDX mappings of MnOx–CM.109 Reproduced with permission from ref. 109. Copyright 2023 Elsevier. (f) Schematical diagram of Bi leaching from BiVO4 lattices. (g) Top-view SEM and (h) HRTEM images of Bi1−xVO4. (i) Ultraviolet photoelectron spectroscopy (UPS) spectra of BiVO4 and Bi1−xVO4 and corresponding band alignments (j). (k) HAADF–STEM–EDX element mapping of Bi1−xVO4.31 Reproduced with permission from ref. 31. Copyright 2022, Wiley-VCH. | ||
As mentioned above, to some extent, the introduction of water does not change the structural properties of DESs.112 However, transport properties such as viscosity vary significantly with water content. Some solvothermal reactions can be carried out in DES–water systems. Similar to “pure” DESs, hydrated DESs were believed to play a triple role (solvent, reactant and template) in synthesis processes. The added water in these eutectic systems is responsible for adjusting viscosity, pH, polarity, etc. In some research work, the precursors were first dissolved in water and then mixed with DESs for further reactions. In others, water was added to the mixture of the precursors and DESs. Wang et al. prepared slender BiVO4 microtubes with a monoclinic structure on a large scale by the solvothermal method.113 A certain amount of water was added to the solution to adjust the viscosity of Reline. The length of the BiVO4 microtube is about 800 nm, and the wall thickness is about 100 nm. The openings of the synthesized microtubes exhibit a jagged structure. The formation mechanism of slender microtubules is attributed to complex collaboration, which involves the reaction-crystallization process controlled by BiOCl and the nucleation growth process of nanosheets induced by the reline–water system.
The rapid, controllable, and green synthesis of TiO2 is still a challenge for its large-scale preparation and application. Tang et al. compared a series of TiO2 prepared with different DES systems (Fig. 6).114 The photocatalytic water-splitting performance of the prepared catalyst is 13.13 mmol h−1 g−1, which is 18 times that of commercial P25 TiO2. The higher activity is attributed to the Crassula perforata-like shape with surface heterojunctions and defects. This research provided a new way for a green and fast way for synthesizing other photocatalysts.
 | ||
| Fig. 6 SEM images for the TiO2 particles synthesized in different DESs. (a) ChCl/urea, (b) ChCl/glycerinum, (c) ChCl/EG, (d) ChCl/oxalic acid, (e) betaine/oxalic acid, (f) chlorocholine chloride/oxalic acid.114 Reproduced with permission from ref. 114. Copyright 2018 Elsevier. | ||
DESs sometimes participate in the reaction as a reducing agent, which may be because of the reductive activity of single components. A typical example is the DES which contains ethylene glycol. Karimi and Eshraghi reported a facile method for synthesizing Mn3O4 nanoparticles.115 Microscopic elemental studies showed spherical nanoparticles with an average particle size of 25 nm. The magnetic characteristics of the synthesized Mn3O4 nanoparticles reveal their paramagnetic behavior at room temperature. In addition, manganese oxides with different element ratios and different morphologies can also be synthesized in the DES–water system.116
4. Electrodeposition method
Benefiting from a wide electrochemical window, DESs have many advantages in the electrodeposition process of metals, alloys, compounds and composite materials. For example, high potentials can be applied so that electrodeposition can be performed at higher current densities to achieve fast reaction rates and efficient electrodeposition.117 In a multi-component system, precise electrodeposition can be realized for preparing complex alloy materials, on account of the inherent high selectivity.As mentioned above, the intrinsic structure of DESs significantly diverges from that of water, thereby rendering the solvation shell of metal ions within DESs more intricate (depicted in Fig. 7). In an aqueous environment, metal ions are solvated by water molecules, mainly through coordination. Although the interaction in DESs is the same as that in water, there are different ligands (e.g., HBD and HBA), resulting in a dramatic effect on ion mobility. We can simplify electrodeposition into a three-step process:17
1. The solvated ion M+ is adsorbed on the electrode surface. Such a process occurs around the electric double layer.
2. Once the ion M+ reaches the inner Helmholtz phase, it turns to get electrons from the anode.
3. The ions M+ are immediately reduced due to the potential across the electrodes.
 | ||
| Fig. 7 Interfaces of anode in an aqueous solution (A) and a DES (B). The hydrogen evolution side reaction is hindered by applying DESs in the electrodeposition process.17 Reproduced with permission from ref. 17. Copyright 2022, Cell press, Elsevier. | ||
By comparing the two different processes, we can know the most significant side reactions and avoid the hydrogen evolution reaction during the deposition process. As mentioned above, this advantage is related to the wide electrochemical window of DES, which is caused by its chemical composition. It is worth mentioning that in water–DES systems, the electroplating products are consistent with those in DESs. In other words, the electroplating solution can be diluted by adding water to reduce the viscosity and further increase the reaction rate. This is attributed to the hydrogen bonding network formed by DES with metal ions being stronger than that of water and the latter. However, when the water content reaches a certain level, the solution will lose the properties of a DES and become an ordinary aqueous solution. The limiting water content varies depending on the type of DES, and researchers are also actively exploring it.
4.1. Metals and alloys
Some metals are widely used in everyday materials, so their reduction and purification are of great value. Jovićević et al. studied aluminum deposition under constant potential and constant current mechanisms.118 The shape and size of the deposited aluminum crystal grains depend on the deposition time. The size ranges from nano to micrometers, and from regular crystals to needle-like or sheet-like structures in shape. In addition, the use of the new DES can realize the electrodeposition of aluminum using Al2O3 as the raw material.119 The study by Harb et al. showed that by adding fluoride ions to DESs, the structures of Mo complexes were significantly changed.120 Mo was thus successfully deposited from ethaline onto the nickel substrate at a constant potential. Cyclic voltammetry proved that fluoride changed the electrochemical behaviour of Mo in solution, which is consistent with previous reports. This work paved the way for the possible development of the Mo electrolytic refining process.In another work, concave-disdyakis triacontahedral (C-DTH) palladium nanocrystals were prepared by shape-controlled electrodeposition in reline.121 Results showed that the urea adsorbate plays a vital role in the shape evolution, especially in the formation of the specific structure of Pd nanoparticles. The electrical behaviour of Pd is believed to arise through changes in the coordination environment of metal ions. To better explore the growth mechanism of nanocrystals, SEM was used to monitor and characterize the growth process. In the first 10 minutes, the Pd nanoparticles grew to a size of about 45 nm. As the growth time increased, preliminary DTH appeared. Then, it further grew into a truncated C-DTH at 30 minutes and formed a perfect C-DTH shape at 45 minutes. Finally, as time increases, no new structure will appear, but only an increase in size. In addition, there are many elemental metals and alloys that can be electrodeposited in DES, which will be introduced below. All in all, the intervention of DESs can not only realize some specific electrodeposition processes but also accurately control the morphology of synthesized materials.
Ruthenium and its compounds have received increasing attention in recent years because of their high catalytic activity.122 Magagnin et al. reported for the first time the electrodeposition process of ruthenium from ethaline.123 It was found that sulfamic acid can facilitate the electrodeposition process. The electrochemical experiments carried out proved that the main consequence of the existence of sulfamic acid is the oxidation of Ru(III) to Ru(IV), which indicates the reduction of Ru occurs from Ru(IV) species. The optimized plating conditions can produce a uniform and crack-free Ru coating with a thickness of up to 800 nm.
In addition to transition metals, Sn–Sb alloy powders can be electrodeposited in ethaline.124 The reduction of Sb(III) and Sn(II) in DES involved an irreversible process controlled by diffusion. When Sb(III) and Sn(II) species coexist, the value of the diffusion coefficient decreases slightly, which may be mainly due to their synergistic effect and the change of metal ion activity coefficient. The electrodeposition potential plays a central role in adjusting the alloy composition but makes little difference to its surface morphology.
Table 1 summarizes metals and alloys deposited in DESs as well as the corresponding operating conditions. As is shown in Table 1, the most favored counter electrode is platinum, while the substrate (working electrode) is variable, depending on the metal deposited. Galvanostatic and potentiostatic modes are desirable for achieving the reducing process. Among the above works, there are several strategies to promote the electrodeposition process, such as increasing temperature and diluting the electrolyte. In some cases there is a single compartment cell, while others involve a binary cell along with a separator. Most of the authors applied the same process when carrying out cyclic voltammetry (CV), linear sweep voltammetry (LSV) and final electrodeposition, and a minority of them used a two-electrode system when electrodepositing.
| Metal/alloy | DES | Electrode | Operation | Ref. | ||
|---|---|---|---|---|---|---|
| HBA | HBD | Ratioa | W/R/Cb | |||
a Molar ratio of HBA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HBD.
b W|R|C refers to working, reference, and counter electrode, respectively.
c More than one HBD.
d Means more than one ratio.
e Electrolyte contains additional water; DBU: 1,5-diazabicyclo[5.4.0]-5-undecene, MTU: methylthiourea, GC: glassy carbon. HBD.
b W|R|C refers to working, reference, and counter electrode, respectively.
c More than one HBD.
d Means more than one ratio.
e Electrolyte contains additional water; DBU: 1,5-diazabicyclo[5.4.0]-5-undecene, MTU: methylthiourea, GC: glassy carbon.
|
||||||
| Ag | ChCl | EG | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Pt|Ag|Pt | The silver was deposited potentiostatically at −35 mV for times ranging from 10 to 6000 s | 129 |
| Al | DBU | MTU | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1c 1c |
Cu|Al|Pt | All processes were performed in an atmospheric environment with a scan rate of 50 mV s−1 | 119 |
| Au | ChCl | EG | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Pt|Ag|Pt | Au coatings were deposited from their respective solutions by immersing the Ni-coated sample in the solution at 80 °C for 45 min | 130 |
| Bi | ChCl | EG | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Cu|Ag|Pt | Cu atoms were displaced by Bi atoms via the consumption of Bi(III) complex ions and the formation of Cu(I) species | 131 |
| Cd | ChCl | Thiouread | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Steel|Ag/AgCl|Pt | Cd was electrodeposited by employing the DES solution (−0.85 V vs. Ag/AgCl, 10 min) on a carbon steel surface | 132 |
| Co | ChCl | Uread | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Cu|—|Pt | Current densities between 3 and 30 mA cm−2 were applied for 30 and 120 min | 133 |
| Cr | ChCl | CrCl3·6H2O | 2.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1e 1e |
Steel|—|Pt | The electrodeposition of Cr was performed under a constant 50 mA cm−2 and at 40 °C | 134 |
| Cu | ChCl | Urea | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Au|Cu|Au | Copper electrodeposition process was found to be taking place through the formation of an underpotential deposition adlayer | 135 |
| Fe | ChCl | EG | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 4 |
Pt|Ag/AgCl|Fe | The electrodeposition process was operated in the Fe-containing DES under a current density of 50 mA cm−2 | 136 |
| Ga | ChCl | Urea | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Mo|Ag|Pt | Electrodeposition of the Ga films was performed under −1.1 V (60 °C) at a rotation speed of 300 rpm for 30 min | 137 |
| In | ChCl | Urea | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Mo|—|Pt | Bulk electrodeposition of indium was carried out at 80 °C in the In-containing DES at 10, 30, and 50 mA cm−2 for 60 min | 138 |
| Mn | ChCl | EG | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Cu|Ag/AgCl|C | Electrodeposition was carried out at 50 °C under a constant potential of −1.50 V | 139 |
| Ni | ChCl | EG | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Al|Ag|Pt | Galvanic deposition process of Ni was carried out in Ni-containing DES at 50 °C for 4 h | 140 |
| Pb | ChCl | Urea | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Cu|Ag|Pb | Electrodeposition process was conducted via potentiostatic or galvanostatic electrolysis in the baths steadily stirred under 90 °C | 141 |
| Pd | ChCl | Urea | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Au|Ag|Pt | The electrodeposition of Pd on Au was operated around −0.2 V and an underpotential deposition around 0 V at 60 °C | 142 |
| Pt | ChCl | Urea | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
GC|Pt|Pt | Potential scan range of −1.5 to −0.2 V (80 °C), scan rate of 50 mV s−1 and CV number of 80 cycles | 143 |
| Re | ChCl | Urea | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Cu|Ag|Pt | Electrodeposition was carried out under galvanostatic (0.3 and 0.5 A d−1 m−2) and potentiostatic (−1.2 and −1.6 V) modes under stirring for 30 min | 144 |
| Ru | ChCl | EG | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Cu|Pt|C | Both galvanostatic and potentiostatic deposition from Ru-containing DES was performed under 80 °C | 123 |
| Sb | ChCl | EG | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Ti|Ag|Pt | The electrodeposition process was carried out from 10 to 50 mA cm−2 for 5 h at different temperature | 145 |
| Sm | ChCl | Urea | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
C|Ag/AgCl|Pt | Metal and alloy deposits were obtained during a voltammetric hold potential at −1.9 V under stirred conditions for 30 min | 146 |
| Sm–Co | ||||||
| Sn | ChCl | EG | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Ni|-|Sn | At different temperature (40–80 °C), the constant current was supplied by a DC power at 20 mA cm−2 for 1 h | 147 |
| Zn | ChCl | Uread | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Steel|Ag|Pt | The deposition occurred in Zn-containing DESs at 50 °C, on mild steel at I = 3 mA cm−2 | 148 |
| As–In | ChCl | EG | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Cu|Ag|Pt | Constant potentials (−0.5 to −1.03 V) were applied at 80 °C with the deposition charge density of 2 C cm−2 | 149 |
| Au–Mn | ChCl | Urea | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Au|Au|Pt | Au–Mn was electrodeposited potentiostatically (−0.7 to −1.9 V) for 1 hour | 150 |
| Cd–Zn | ChCl | EG | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Cu|Ag/Ag+|Pt | The electrodepositions were conducted at the constant potentials in the range from −1.3 to −1.7 V for 6 C cm−2 at 50 to 90 °C | 151 |
| Ce–Ni | ChCl | EG | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Steel|Pt|Pt | Electrodeposition process was carried out using the chronopotentiometry method (10 mA cm−2) up to a charge of 100 mA h | 152 |
| Co–Cr | ChCl | EG | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Brass|—|C | Electrodeposition of Co–Cr alloys was obtained by using direct current and pulsed electrodeposition techniques | 153 |
| Co–La | NaBr | Urea | — | Steel|Hg/HgO|Pt | La–Mo alloy was deposited from different current density 10 to 40 mA cm−2 at 130 °C | 154 |
| CoSO4 | ||||||
| LaCl3 | ||||||
| Co–Nd | ChCl | Urea | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Cu|Ag|W | Co–Nd alloy was obtained by electrodepositing under constant current density (50 to 125 mA cm−2 at 100 °C) | 155 |
| Cu–Mn | ChCl | EG | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Cu|—|Pt | Pulse current electrodeposition (30 min) was conducted in which each cycle consisted of 1 s of low current, followed by 1 s of high current | 156 |
| Cu–Ni | ChCl | Urea | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Ni|Ag|Pt | Cu–Ni alloy was electrodeposited from −0.9 V to −1.2 V in the leached electrolyte at 70 °C for 1 h | 157 |
| Cu–Zn | ChCl | Urea | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Fe|Ag|Pt | Electrodeposition was conducted at the range of potentials −0.80 to −1.15 V, and 70 °C for 2 h | 158 |
| Fe–Co | ChCl | EG | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.24 2.24 |
Cu|—|Fe | The alloy was electroplated using a direct current 67 mA cm−2 for 2 to 20 min (at 100 °C) | 159 |
| Fe–Cr | ChCl | EG | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 3 |
Brass|—|Cr | Fe–Cr alloy was prepared by electrodeposition in DES by varying cell voltage (2.4 to 2.8 V) and temperature (60 to 80 °C) | 160 |
| La–Ni | Acetamide | Urea | — | Cu|—|C | The alloys were obtained by potentiostatic electrolysis under an Ar atmosphere for 25 min (at 80 °C) | 161 |
| Sm–Fe | NaBr | |||||
| Gd–Co | KBr | |||||
| Ni–P | ChCl | EG | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Brass|Ag|Ni | Ni–P alloys were obtained at a potential of −0.7 V for 10 min (30 ± 2 °C) | 162 |
| Ni–Ta | ChCl | Urea | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Cu|Ag|Ti | Electrodeposition was performed at potentiostatic (−0.3 V) and galvanostatic (1 mA cm−2) conditions for a duration of 30 minutes with stirring at 400 rpm | 163 |
| Pd–Ag | ChCl | Urea | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Cu|Pt|Pd | Alloys were obtained by pulse plating and potentiostatic deposition for 15 or 30 min | 164 |
| Pd–Pt–Ag | ||||||
| Sn–Bi | ChCl | EG | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Au|Ag|Pt | Different coatings were deposited potentiostatically on substrates at different potentials (−0.4 to −0.6 V) | 165 |
| Sn–In | ChCl | EG | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Cu|—|Pt | Galvanostatic (1 and 25 mA cm−2) deposition was performed for 20 min at 25 to 60 °C | 166 |
| Zn–Mn | ChCl | Urea | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Steel|Hg/Hg2Cl2|Zn | Zn–Mn alloy was deposited by a charge density of 20 C cm−2, at 70 °C | 167 |
| Zn–Ti | ZnCl2 | Urea | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 3 |
Cu|Pt|C | Zn–Ti alloy was prepared at deposition potential of −1.3 to −1.8 V for 2 h (80 °C) | 168 |
| Pr–Mg–Co | ChCl | Urea | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Cu|Ag|Pt | The optimum coating was obtained at −1.05 V for 20 min | 169 |
| Zn–Ni–P | ChCl | Urea | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Cu|Ag|Ni | Ni–Sn–P alloys were obtained by using constant potentials (−0.6 to −0.8 V) with total charge density of 4 C cm−2 at 60 °C | 170 |
4.2. Compounds
Compared with alloys, the types and structures of metal compounds are much more complicated. Moreover, the functions of the compounds are more diverse. For example, some materials have excellent electronic band structures and are suitable as photocatalysts. Metal oxides are among the simplest compounds. Wang et al. directly prepared nanostructured NiO films through simple and effective electrodeposition.125 It was proved that the surface morphology and density of the film are related to the operating temperature. The higher the temperature, the lower the viscosity. Therefore, nucleation precedes growth at higher temperatures, so the particle size becomes smaller when deposited at higher temperatures. The light transmittance of the film increases with the increase of the electrodeposition temperature because the purity of the oxide in the product is higher at higher temperatures. Zhang et al. introduced a two-step method to prepare metal sulfides. In the first step, Ni nanospheres were deposited on the foamed nickel framework.126 Subsequently, the resulting metal composite material undergoes a vulcanization reaction at 80 °C. It is worth mentioning that the solvent system used in both steps is a DES (ethaline). The decomposition process of thiourea in ethaline drives time-dependent surface reorganization and phase transitions on Ni substrates, leading to the in situ formation of a Ni3S2 layer with a controllable structure. The prepared material has a high specific surface area, shows a high specific capacitance of 5.633 mF cm−2 at a current density of 10 mA cm−2 with good durability, and is expected to be practically applied in high-performance electrochemical devices.Numerous materials are designed and synthesized to solve energy problems. Some of the work starts with preparing the materials and is then followed by testing their catalytic activity. The design and synthesis of high-efficiency and low-cost bifunctional electrocatalysts for hydrogen evolution reactions and oxygen evolution reactions are essential for electrochemical water splitting related to clean and renewable energy technologies. In the study of Zhang et al., a one-step electrodeposition method of FexCo3−x(PO4)2 nanosheets filled with 3D microspheres was introduced.127 The excellent performance can be attributed to the fact that Fe doping changes the active electronic properties of O in the phosphate, thereby achieving the best hydrogen adsorption at the active site. In addition, the unique 3D microsphere structure combined with the internal structure of the 2D nanosheet provides a rich catalytic interface with more active sites and favorable transfer kinetics. The integrated electrode exhibits excellent HER electrocatalytic performance in a wide pH range and has robust catalytic activity for overall water decomposition in alkaline media. In another work, the electrodeposition of P–Co nanoparticles on nickel foam was reported.128 The prepared catalyst showed excellent performance for both hydrogen evolution reactions and oxygen evolution reactions. It is believed that the above materials will boost the development of hydrogen energy in the future.
There are several conclusions obtained from Table 1:
• Electrode, especially the substrate (cathode), is vitally important and should be considered carefully.
• Thickness and morphology are usually dominated by time, which needs to be controlled based on the target.
• Conditions of CV or LSV experiment and practical deposition processes can be different.
• Galvanostatic and potentiostatic deposition are two different methods, which can be adjusted according to the actual situation during the production process.
• Dilution with water and increasing temperature are two ordinary means to lower the viscosity and accelerate mass transfer.
4.3. Specific structures
Fabricating nanoparticle-modified electrodes is very challenging because it usually involves a multi-step route, such as chemical synthesis, purification, and final embedding on the electrode surface. Moreover, it always requires high temperatures. By using DESs as electrolytes, electrodeposition can be operated at room temperature, achieving an all-in-one process. Renjith and Lakshminarayanan reported a single-step electrochemical preparation and the in situ deposition of thin clusters of cobalt nanoparticles on the electrode surface in maline.171 The thickness of the nanoparticle clusters ranges from 20 to 90 nm. Although the catalyst loading is very low, the cobalt nanoparticles on the dense graphite flakes can obtain an oxygen evolution reaction (OER) current of 10 mA cm−2 at a very low overpotential of 350 mV, which indicates that it is a good potential anode material for the oxygen evolution reaction.As mentioned above, the two precursors in DES are not necessarily reduced in the form of alloys. Mohan et al. synthesized multilayer Ag–Co corn-shaped material by a pulse technique.172 The synthesized materials exhibited a maximum giant magnetoresistance value of 9.94% at room temperature. In some cases, DES can achieve material composites that are difficult to complete in water systems. It has proved difficult to co-deposit SiO2 particles into a metal matrix in an aqueous solution. Liang and coworkers demonstrated the pulse electrical co-deposition of nano-sized SiO2 particles and a Ni matrix in ethaline.173 SiO2 nanoparticles exhibit excellent dispersion stability in DES, which is very important. In addition, the presence of SiO2 nanoparticles has a significant impact on the nucleation mechanism of Ni. Increasing the content of SiO2 in the composite material can significantly improve the corrosion resistance of the coating.
A simple one-step electrochemical deposition strategy of self-supporting Co–O and Co–Se hybrid films in reline has been reported.174 DES was believed to play an important role in forming porous nanostructures, and these pores significantly promote the intrinsic catalytic activity of the material. The Co–Se component in the hybrid film was inherently unstable under OER conditions and transformed into Co–O species with enhanced surface oxygen vacancies. Therefore, the prepared material shows high catalytic performance and robust durability for both HER and OER.
The core–shell structure is a more complex structure than ordinary composites. It contains an inner core and a uniform outer thin layer. The synthesis of this special structure material can be achieved by electrodeposition. Pd@Pd(OH)2 nanoparticles were electroplated onto the glassy carbon electrode in reline.175 The core–shell structure is so distinct that it shows clearly in scanning electron microscopy. Interestingly, the small amount of water (0.15% by Karl Fischer coulometric titration) inherent in DES plays a significant role in forming the outer shell. The corresponding chemical reaction formula is as follows.
| Pd2+ + e− → Pd | (4) |
| 2H2O + 2e− → H2 + 2OH | (5) |
| Pd2+ + 2OH− → Pd(OH)2 | (6) |
4.4. Coating and etching
Etching and coating are two reversed processes, which can both be achieved by using appropriate DES (Fig. 8). In Xu's work, an in situ DES etching strategy was developed to prepare NiP encapsulated in P,O co-doped carbon nanosheets.177 DES composed of tetrabutylphosphonium chloride and urea (molar ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) serves not only an etchant to extract Ni2+ from nickel foam, but also a phosphorus source to form nickel phosphide uniformly embedded in the carbon film in situ to produce a sheet-like structure. Another example showcases the typical process in the semiconductor industry, realizing nanoscale surface roughness with desirable flatness.178 Hu and coworkers proposed a smooth etch strategy by in-depth study of the interfacial reactions of GaN photoanodes in DES and other aqueous electrolytes. With the increase of the anodic potential, hydroxyl anions and H2O molecules first react with holes (h+) on the GaN interface, initiating GaN oxidation by generating hydroxyl radicals (OH˙). It was found that etching is the result of the synergy of interfacial reactions, including GaN oxidation, oxide film formation and dissolution, and side reactions. This method allows the preparation of groove structures most typical of GaN-based devices with nanometer-scale surface flatness, such as insulated-gate bipolar transistors.
1) serves not only an etchant to extract Ni2+ from nickel foam, but also a phosphorus source to form nickel phosphide uniformly embedded in the carbon film in situ to produce a sheet-like structure. Another example showcases the typical process in the semiconductor industry, realizing nanoscale surface roughness with desirable flatness.178 Hu and coworkers proposed a smooth etch strategy by in-depth study of the interfacial reactions of GaN photoanodes in DES and other aqueous electrolytes. With the increase of the anodic potential, hydroxyl anions and H2O molecules first react with holes (h+) on the GaN interface, initiating GaN oxidation by generating hydroxyl radicals (OH˙). It was found that etching is the result of the synergy of interfacial reactions, including GaN oxidation, oxide film formation and dissolution, and side reactions. This method allows the preparation of groove structures most typical of GaN-based devices with nanometer-scale surface flatness, such as insulated-gate bipolar transistors.
 | ||
| Fig. 8 Etching and coating processes conducted in DESs. (a) Schematic of t-Ni–P@POC synthesis process. (b) and (c) SEM images of t-Ni–P@POC at different magnifications. (d) TEM image of t-Ni–P@POC.177 Reproduced with permission from ref. 177. Copyright 2022, Wiley-VCH. (e) The DES-assisted etching process. (f) and (g) AFM images of bare and oxidized GaN substrates. (h) SEM image of patterned GaN after etching. (i) Laser scanning microscopy image of grooves prepared at −0.2 V.178 Reproduced with permission from ref. 178. Copyright 2020, IOP publishing. (j) Schematic representation of planar diffusion mechanism in the case of Pt(1 1 1). (k) In situ STM-image of layered Cu on Pt(1 1 1) at −0.27 V. (l) The cross-section profile was obtained along white dashed line in (k). (m) Mechanism of forming hemispherical plane on a Pt(poly). (n) In situ STM-image of semispherical Cu on Pt(poly). (o) The cross-section profile was obtained along white dashed line in (n).179 Reproduced with permission from ref. 179. Copyright 2022 Elsevier. | ||
In contrast to the etching process, Zaytsev et al. studied the electrodeposition of Cu in DES, on single crystal Pt(1 1 1) and polycrystalline Pt(poly) planes.179 The underpotential deposition process of Cu on Pt(1 1 1) surface in DES was observed for the first time, in which the surface morphology and (co)adsorption of solution components played important roles. Furthermore, the mechanism of Cu electrodeposition in DES was found to be determined by the surface structure: STM data demonstrated the layer-by-layer growth of 2D Cu deposits on Pt(1 1 1), while the resulting Cu deposits on Pt(poly) had characteristics of grain-like morphology.
We usually use platinum as a counter electrode because of its stable chemical properties, but is this the case? A novel work showed that the dissolution behaviour of the electrode in the composite electrodeposition process still needs further study. Lu et al. reported the dissolution mechanism of a platinum electrode and silver reference electrode during nickel–carbon composite electrodeposition in DESs.180 When the pre-prepared nickel–carbon particles were added to ethaline with Pt as the counter electrode and Ag as the reference electrode, a platinum-based composite material with a “cauliflower” morphology could be deposited. The excellent catalytic performance is attributed to the dissolution–electrodeposition of the Pt and Ag electrodes during the composite electrodeposition process. Density functional theory calculations further show that Pt/Ag interact with chlorine atoms on their surface, and the added activated carbon particles have a significant effect on the dissolution process. Lu's work provides a new method for preparing Pt-based composite materials, and also indicates that the dissolution behaviour of the Pt counter electrode and the Ag reference electrode should be taken into consideration when designing the electrodeposition reaction.
DFT calculation is a very effective way to study the properties of a DES, such as showing the internal structure, predicting viscosity, etc. Zhang et al. investigated the molecular structure of DES (ChCl and MgCl2·6H2O).181 The result shows that the main chemical species of the above system is [MgClm(H2O)6−m]2−m and [ChxCly]x+y clusters. Additionally, the active magnesium complexes in the electrochemical process were determined, and the electrochemical process mechanism between choline chloride and magnesium chloride hexahydrate was explained.
5. Calcination
The composition of DESs is remarkably diverse, encompassing more than 40 metallic elements, and there is even a report on a uranium-based DES.182 On the other hand, DESs provide a homogeneous and stable liquid environment in regard of precursors. Furthermore, the heterogeneity of DESs makes them suitable for tuning the morphology of the product at the nanoscale. Based on the thermal stability studies mentioned above, the calcination method is often adopted to prepare the target materials.5.1. Carbon materials
In some cases, the DES itself serves as the carbon source, and no other precursor is needed. Cai et al. reported a direct calcination method to prepare N-doped porous carbon.183 The DES used in this work was composed of glucose and urea, which is low cost and green. Miniature supercapacitors based on N-doped carbon nanosheets exhibit a specific capacitance of 23.2 mF cm−2 at 0.2 mA cm−2. In addition, such calcination methods can be applied to prepare mesoporous silica.184 Compared with those based on surfactant or ionic liquid, DES-templated synthesis is cheaper and more environmentally friendly. Porous carbon turns to form a coating layer, which is beneficial for the fabrication of multilayer structures and core–shell structure materials.185,186 Wen et al. synthesized NaTi2(PO4)3/C by calcinating choline chloride/triethanolamine DES.187 The 3D layered porous material that has abundant macropores was an anode material for sodium-ion batteries.Graphitic carbon nitride (g-C3N4) as a kind of metal-free n-type semiconductor has been considered a promising light photocatalyst. However, the photocatalytic performance of g-C3N4 is far from satisfactory, because the random transfer of charge carriers in the plane leads to a low conversion rate. Therefore, some oxides, such as ZrO2 and CuO, can be used as cocatalysts to improve the photocatalytic performance of the composites.188,189 During calcination, the urea in the DES decomposes to produce g-C3N4 accompanied by the generation of ZrO2, making the composites extremely stable. The prepared material shows high activity for OER and NRR reactions. However, in the case of CuO, the metal oxide and C3N4 were not generated from the DES at the same time. The DES was used to dope Cl− into the CuO lattice structure, and then the Cl−-doped CuO was combined with the pre-prepared C3N4via an ultrasonication and mixing method. In addition to g-C3N4, borides can also be prepared from DESs by calcination.
5.2. Metal-based materials
In addition to amorphous carbon materials and their composite materials, compounds with crystal structures can be prepared by calcination in DESs as well.190,191 Furthermore, designing the reacting conditions makes it possible to control the morphology of the synthesized products. George et al. reported metal oxide particles as precursors to prepare target materials, which is unusual in the synthesis based on DESs.192 They smartly used the decomposition of the carboxylic acid component in the DES to synthesize co-oxides. The electrode modified by nano-MgFe2O4 showed a high sensitivity for detecting nitrofurantoin and 4-nitrophenol.In order to enhance the photocatalytic efficiency of g-C3N4, Mou et al. synthesized g-C3N4/ZrO2 layered materials with the assistance of DES (ZrOCl2·8H2O/urea).189 Studies have shown that when the ZrO2 content in the composite material is 30 wt%, the optimal NH4+ yield can reach 1446 μmol L−1 h−1. The apparent quantum efficiency at 400 nm exceeds 2.14%, which is 7.9 times higher than the original g-C3N4, and 27.5 times that of ZrO2. In addition, the introduction of amorphous ZrO2 inhibits the generation of hydrogen and can promote rapid photogenerated electron transfer.
The materials synthesized by calcining DESs can also be used in traditional catalytic reactions. The selective oxidation of alcohols to the corresponding aldehydes and ketones is of great significance in academic and industrial fields. Tao et al. prepared an excellent nanostructured catalyst of cobalt nanoparticles supported on nitrogen-doped carbon (Co–N–C).193 The results show that the optimized Co–N–C exhibits excellent catalytic performance in the selective oxidation of aryl alcohols and alkyl alcohols, enabling the corresponding aldehydes and ketones to reach >99% yield. Such high catalytic activity is believed to be due to the synergistic interaction between the nitrogen-doped carbon support and the Co–N species. Interestingly, the magnetically recyclable Co–N–C catalyst can be easily separated from the reaction system by using an external magnetic field.
Doping and compounding are common means of modifying materials, and this section starts by presenting two related studies in the context of calcination (Fig. 9). Ying et al. designed Mn-doped Bi2O3 nanosheets, which were fabricated by using a DES for the first time.194 The results showed that the introduction of Mn into Bi2O3 can significantly enhance the faradaic efficiency of the nitrogen reduction reaction by suppressing the unfavorable hydrogen generation. Wei et al. investigated the effects of chain length, functional group substitution, and molar ratio on the thermodynamic stability of DESs based on different amino acids.195 Ni and composite Ni@Ni(OH)2 samples were prepared by a one-step annealing process, which exhibited quite good oxygen evolution reaction performance in alkaline solutions.
 | ||
| Fig. 9 Doping and compositing conducted in DES. (a) Schematic illustration of the Mn-doped Bi2O3 nanosheets synthesis. (b) XRD patterns of 10% Mn–Bi2O3, 5% Mn–Bi2O3 and Bi2O3. (c), (d) TEM images, (e) HRTEM, and (f) SEM and corresponding EDX mappings of 5% Mn–Bi2O3. (g) XPS spectra of 10% Mn–Bi2O3, 5% Mn–Bi2O3 and Bi2O3 (Mn 2p). (h) N2 adsorption–desorption isotherms of Bi2O3, 5% Mn–Bi2O3 and 10% Mn–Bi2O3.194 Reproduced with permission from ref. 194. Copyright 2022, American Chemical Society. (i) Schematic diagram of the formation of Ni nanoparticles and a 2D Ni/Ni(OH)2 heterostructure in amino acid-based DESs, respectively. SEM images of (j) Ni nanoparticles and (k) Ni/Ni(OH)2 nanosheets. (l) Raman spectra of Ni/Ni(OH)2 and Ni. (m) HRTEM image of Ni/Ni(OH)2 nanosheets, inserted: the related small-angle X-ray diffraction image. (n) XPS spectra of the Ni and Ni/Ni(OH)2 samples (Ni 2p).195 Reproduced with permission from ref. 195. Copyright 2022, Royal Society of Chemistry. | ||
5.3. Bottom-up and top-down
In the above content, we mainly introduced solvothermal, electrochemical and calcination methods for synthesizing materials. To finalize this section, two opposite processes are compared, illustrating strategies for designing task-specific DESs to prepare target materials (Fig. 10). In Shinde's study, nanoparticle-decorated ultrathin Ni1−xMnxCo2O4 nanosheets (NPs@NFs) were bottom-up synthesized by adjusting the molar ratio between Mn and Ni in the DES.196 The unique and highly porous NPs@NFs nanostructure helps to increase the overall surface area of the material, while Mn, Ni, and Co ions constitute its redox activity ability, thereby enhancing the electrochemical activity of the material. This Ni–Mn–Co2O4 hybrid nanostructure exhibits excellent performance with a high specific capacity of 761 mA h g−1 at a relatively high current density of 30 mA cm−2 and a cycle-retained rate of 92.86%. Furthermore, the hybrid asymmetric supercapacitor (Ni0.8Mn0.2Co2O4//AC) device exhibits an extended potential window of 1.5 V, yielding an ultrahigh energy density of 66.2 W kg−1. Top-down preparation of few-layer hydroxylated boron nitride nanosheets (OH-BNNS) from bulk material is highly valued and promising, although an efficient method still remains challenging. Shan and co-workers developed a one-step preparation process of OH-BNNS assisted by an aqueous DES electrolyte-assisted exfoliation and oxidation process.197 The results showed that the obtained OH-BNNS had an average lateral dimension of 625 nm and a thickness of six layers. Adding OH-BNNSs and DES into polyvinyl alcohol (PVA) substrates creates composite gel polymer electrolytes for solid-state flexible supercapacitors. Moreover, a symmetric solid-state flexible supercapacitor composed of activated carbon electrodes and PVA/DES/OH-BNNSs gel polymer electrolyte emerged with a wide voltage window of 2.3 V and a high specific capacitance of 151.22 F g at 0.1 A g for 1500 cycles with a 98% maintenance of capacity. | ||
| Fig. 10 Comparison of bottom-up and top-down methods for synthesizing materials in DESs. (a) Schematic diagram of the formation of Ni1−xMnxCo2O4 composite nanoflakes. HR-TEM image (b) and high-resolution XPS spectra (c) of the Ni0.8Mn0.2Co2O4 composite, Co 2p. (d) Specific capacitance with respect to various current densities of solid-state hybrid Ni0.8Mn0.2Co2O4//AC (activated carbon). (e) Nyquist plots of hybrid supercapacitor after and before cycling stability of the hybrid Ni0.8Mn0.2Co2O4//AC device.196 Reproduced with permission from ref. 196. Copyright 2022, Wiley-VCH. (f) Schematic representation of the mechanism of electrochemical exfoliation in DES. SEM images of bulk h-BN (g) and hydroxylated boron nitride nanosheets (h), inset: the histogram of statistical size. XRD patterns (i) and FT-IR spectra (j) for bulk h-BN and PVA/DES/OH-BNNSs. (k) Galvanostatic charge–discharge behaviors of supercapacitors with PVA/DES/OH-BNNSs. (l) Cycling stability of supercapacitors with PVA/DES/OH-BNNSs and PVA/DES gel polymer electrolytes.197 Reproduced with permission from ref. 197. Copyright 2022, American Chemical Society. | ||
6. Supramolecules and polymers in DES
With the development of eutectic systems, concepts such as eutectogel and supramolecular DES were proposed.7,8 DES-based gel, further referred to as eutectogels, serves as a promising candidate for solid composite electrolytes. These robust composite electrolytes demonstrate satisfactory thermal stability up to more than 100 °C, along with an extensive electrochemical stability range and elevated ionic conductivity at ambient temperatures. In a work, Liu et al. reported a complex of natural guanosine and H3BO3, which showed excellent gelling ability in DESs.198 In this case, the reactants guanosine, H3BO3, and KOH in DES ChCl/MPG turn to produce an anionic guanosine boronic acid diester complex (GB), which is further templated by K+ to form a hydrogen bond G-quartet structure (G4). The G4-quadruplex spontaneously stacks into a column G-quadruplex structure generated by physically entangled fibres that constitute the ChCl/MPG eutectogel (Fig. 11a). Conversely, mixing guanosine, H3BO3, and KOH in a glycerol-based DES resulted in GB monoester and GB diester complexes. This further leads to a more complex G-quadruplex structure with a different supramolecular chirality (Fig. 11a). | ||
| Fig. 11 (a) The scheme of a possible mechanism for gelation process via formation of guanosine/boric acid (GB) borate esters and subsequently G4-quartets. Inset: the CD spectra. (b) Photographs of the ChCl/MPG ECD at −0.7 V for colouring and 0 V for bleaching. (b) UV-vis spectra at various voltages for the ECD based on the hybrid GB/MPG eutectogel. (c) Transmittance responses of the ChCl/MPG ECD at −0.7 V for colouring and 0 V for bleaching. (d) In situ transmittance response of the ChCl/MPG ECD over 100 coloring-bleaching cycles.198 Reproduced with permission from ref. 198. Copyright 2020, Wiley-VC. (e) In situ transmittance response of the ChCl/MPG ECD over 100 cycles, coloring for 60 seconds with a potential at 0.7 V, and bleaching for 150 seconds with 0 V applied. | ||
Sometimes, eutectogels combined with other sustainable materials (such as biomass) exhibit enhanced mechanical properties. A polymerized high-performance sensor incorporating polymerized DES into delignified wood has attracted public attention.199 The retained orientation and the strong hydrogen bonding interactions between the cellulose-rich template and the eutectogel give the wood excellent mechanical properties. The prepared material has high transparency (90% light transmittance), good stretchability (strain up to 80%) and high conductivity (0.16 S m−1).
Additionally, this proved that the existence of a solid-like nanofiber network does not significantly inhibit ion transport in DES. The high room temperature ionic conductivity of ChCl/MPG eutectogel places it in the top echelon of gel electrolyte materials and is high enough for possible applications. The hybrid ChCl/MPG eutectogel film exhibits excellent electrochromic activity, making it a good material for fabricating electrochromic devices (ECDs). When the potential increased from 0 to −0.7 V, it changed from light yellow to uniform blue (Fig. 11b). The in situ UV-vis transmission spectra of ChCl/MPG ECD at different potentials showed its transmittance decreases with the increase of potential and reaches saturation at −0.7 V when the maximum optical contrast (ΔTmax) is 59% (Fig. 11c). The lowest working voltage for ChCl/MPG ECD colouring is −0.4 V, which is much lower than the other semi-solid ECD devices. As is shown in Fig. 11d, the colouring time of ChCl/MPG ECD is about 15 seconds, and the bleaching time is about 30 seconds (time to reach 90% ΔTmax). Moreover, ChCl/MPG ECD showed strong cycling performance, maintaining 94.2% of the initial ΔTmax after 100 cycles (Fig. 11e). The low working voltage, fast response time and good cycling stability of the prepared ECD prove the possibility of supramolecular eutectogels as next-generation electrolytes for flexible electronic devices.
The above showcased that DES was used to synthesize gel for solid electrolytes, whereas the synthesis method of eutectogel may be different. For example, Smith et al. prepared a series of supramolecular eutectogels with ultra-high ionic conductivity by self-assembly.200 In other cases, the monomers polymerized in DES have changed, leading to the synthesis of diverse eutectogels.201,202 And there are other methods of forming a framework network, such as the condensation of silica precursors.8
Since 2020, the concept of supramolecular DES (SUPRADES) has attracted attention from researchers, while host–guest DES originated earlier.7,203 This new system retained its inclusion capacity in the resulting solvent, raising a new family of solvent systems. In the case of cyclodextrins (CDs), mixtures of functionalized CDs with hydrogen bond donor species enable an extended liquid-state window. These solvents maintain CD's inclusion capability while also enhancing solvation effectiveness through a prolonged hydrogen bonding network.
Early work focused on the dissolution of CDs in DES, along with their potential applications.204 For example, Xu et al. demonstrated for the first time that hydrophobic DES (methyltrioctylammonium chloride![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) octanoic acid) can be used for chiral separations in capillary electrophoresis.205 Optimal conditions were explored: pH 2.5, phosphate buffer containing 1% carboxymethyl-β-CD and 0.15% DES, applied voltage 20 kV. Through this approach, separations of drugs that cannot be accomplished solely within the CD system are successfully attained.
octanoic acid) can be used for chiral separations in capillary electrophoresis.205 Optimal conditions were explored: pH 2.5, phosphate buffer containing 1% carboxymethyl-β-CD and 0.15% DES, applied voltage 20 kV. Through this approach, separations of drugs that cannot be accomplished solely within the CD system are successfully attained.
After 2020, CDs have transitioned from their role as solutes to that of solvents.7,206,207 Guan et al. designed CD-based DES (methyl-β-cyclodextrin: formic acid) for catalytic desulfurization of fuel oil.208 Theoretical calculations and experimental findings have indicated that peroxygen compounds serve as the primary active species, and the remarkable catalytic efficacy of CD-based DESs can reduce the reaction barrier by 81% in the EODS process, achieving an impressive sulfur-removal rate of 99.6%.
7. Outlooks and conclusions
The wide range of materials synthesized using DESs also poses a challenging issue when it comes to scaling up production. In this regard, there have also been some explorations. Cheng et al. provided a fast heat exchange and uniform mass transfer process, in which Pd nanocrystals with mainly exposed Pd (100) faces can be synthesized efficiently (Fig. 12a).209 The expanded hydrogen network and abundant ion species in DES were believed to stabilize the Pd crystal plane and limit the nanocrystal size, which is consistent with reports on the properties of the solvent system.210 Pd NCs supported on porous carbon exhibited a size distribution of ∼13 ± 3.5 nm and a high electrochemical active surface area (46.0 m2 g−1). Further characterization showed these composite materials have high catalytic performance and excellent stability for the oxidation of formic acid. | ||
| Fig. 12 One-step flow and shape-controlled synthesis of nano particles in DES. (a) Schematic illustration of combining DES solvothermal and microfluidic technology to continuously produce nano-sized Pd@N-carbon.209 Reproduced with permission from ref. 209. Copyright 2020, American Chemical Society. (b) Shape-controlled PtPd nanocrystal synthesis in flowing ethaline.25 Reproduced with permission from ref. 25. Copyright 2023, American Chemical Society. | ||
The above works demonstrate the extraordinary future of DES as a scalable, sustainable, and high-performance fluid in the flow process to enhance nanomanufacturing efforts. Kim et al. synthesized an amorphous RuCo2O4 nanofiber in a reline–water system.211 It is worth mentioning that the entire reaction was carried out at 40 °C. Urea underwent a decomposition reaction, and the purported ammonia acted as a reducing agent to react with two salts, which is shown in the following equations:
| CO(NH3)2 → NH3 + HCNO | (7) |
| RuCl3 + NH3 → Ru(NH3)Cl3 | (8) |
| Co(NO3)2 + NH3 → Co(NH3)(NO3)2 | (9) |
 | (10) |
Reline and water can lower the total surface energy of the particles produced. The synthesized bimetallic oxide possesses an effective surface area of 245 m2 g−1, and they were expected to have potential in high-efficiency materials, such as supercapacitors. With the reduction of operating temperature, the cost of the microfluidic method has also been reduced, and shows great application potential in the future.
Another work showed the possibility of low-cost, scalable, controlled size-consistent preparation of nano-catalysts. Li et al. used a continuous flow reactor and ethaline to synthesize intermetallic PtPd nanoparticles with controlled shape in 6 minutes.25 The synthesized PtPd nanocrystals exhibited octahedral shape with mainly exposed (111) facets and an average diameter of 12.8 nm (Fig. 12b). Furthermore, PtPd nanoparticles synthesized by this method are more uniform than particles produced using conventional flask reactors using similar synthesis conditions.
Although DES preparation materials have exhibited some advancements, there are also some problems that need to be solved in this field. First, the kinetic study of the solvothermal growth of materials, including the effects of different temperatures, synthesis time, and monomer composition on the product has been neglected. Second, insight into the transformation process from liquid phase to solid phase during calcination is missing. Third, solvent recycling when preparing materials by electrodeposition is a problem. In addition, AI-assisted research on the properties of DES is considered to have a guiding role in their application.212
In 1998, 12 principles of green chemistry were proposed. The description of atom economy is “Synthetic methods should be designed to maximize incorporation of all materials used in the process into the final product”.213 Environmental factor (E-factor), which was defined as the following, is believed to be a more accurate measurement of the environmental acceptability of a chemical manufacturing process:214
Despite this, there are relatively few reports on the E factor regarding DESs. Researchers have generally discussed the atom economy of reactions involved in DES, which may be because these reactions are mainly at the laboratory scale and in the early stage of industrialization. We believe that as this field develops, atom economics and E-factor will become increasingly significant when evaluating cases in DES-chemical manufacturing.
To summarize, DESs participate in the preparation of materials physically or/and chemically. The physical aspect includes solvating, exfoliating, dispersing and confining, while the chemical part includes reacting, composing, polymerizing and modifying (Fig. 13). This review first highlights the heterogeneity of the DES, a characteristic property of this solvent, and explains the origin of good solubility and templating effect. In addition, polarity and solvation properties are discussed as they relate to materials prepared by solvothermal methods; studies of thermal decomposition kinetics are a fundamental aspect of materials prepared by calcination methods, and electrochemical stability is a prerequisite for electrodepositing materials such as metals. Some specific processes in the field of materials preparation are compared, such as intercalation and leaching, doping and compositing, etching and coating. The two opposite processes, top-down and bottom-up, can serve as valuable references when synthesizing materials in a nonaqueous system. Finally, the micro-fluidic method may provide a possible solution to the scaled-up production of nanomaterials in DESs.
Conflicts of interest
The authors declare no competing interests.Acknowledgements
This work was supported by the National Natural Science Foundation of China (22238011 and 22073112). D. Y. is thankful for the support from the China Scholarship Council (202006360037). D. J. is thankful for the support from the National Natural Science Foundation of China (U1937601).References
- A. P. Abbott, G. Capper, D. L. Davies, R. K. Rasheed and V. Tambyrajah, Chem. Commun., 2003, 70–71 RSC.
- E. L. Smith, A. P. Abbott and K. S. Ryder, Chem. Rev., 2014, 114, 11060–11082 CrossRef CAS PubMed.
- D. Yu, Z. Xue and T. Mu, Chem. Soc. Rev., 2021, 50, 8596–8638 RSC.
- D. O. Abranches, M. A. R. Martins, L. P. Silva, N. Schaeffer, S. P. Pinho and J. A. P. Coutinho, Chem. Commun., 2019, 55, 10253–10256 RSC.
- D. Yu and T. Mu, J. Phys. Chem. B, 2019, 123, 4958–4966 CrossRef CAS PubMed.
- R. Shi, D. Yu, F. Zhou, J. Yu and T. Mu, Chem. Commun., 2022, 58, 4607–4610 RSC.
- T. El Achkar, T. Moufawad, S. Ruellan, D. Landy, H. Greige-Gerges and S. Fourmentin, Chem. Commun., 2020, 56, 3385–3388 RSC.
- B. Joos, T. Vranken, W. Marchal, M. Safari, M. K. Van Bael and A. T. Hardy, Chem. Mater., 2018, 30, 655–662 CrossRef CAS.
- A. P. Abbott, D. Boothby, G. Capper, D. L. Davies and R. K. Rasheed, J. Am. Chem. Soc., 2004, 126, 9142–9147 CrossRef CAS PubMed.
- B. B. Hansen, S. Spittle, B. Chen, D. Poe, Y. Zhang, J. M. Klein, A. Horton, L. Adhikari, T. Zelovich, B. W. Doherty, B. Gurkan, E. J. Maginn, A. Ragauskas, M. Dadmun, T. A. Zawodzinski, G. A. Baker, M. E. Tuckerman, R. F. Savinell and J. R. Sangoro, Chem. Rev., 2021, 121, 1232–1285 CrossRef CAS PubMed.
- P. Y. Chew and A. Reinhardt, J. Chem. Phys., 2023, 158, 030902 CrossRef CAS PubMed.
- Q. Liu, T. Yuan, Q.-J. Fu, Y.-Y. Bai, F. Peng and C.-L. Yao, Cellulose, 2019, 26, 9447–9462 CrossRef CAS.
- T. Zhang, T. Doert, H. Wang, S. Zhang and M. Ruck, Angew. Chem., Int. Ed., 2021, 60, 22148–22165 CrossRef CAS PubMed.
- C. Vidal, J. Garcia-Alvarez, A. Hernan-Gomez, A. R. Kennedy and E. Hevia, Angew. Chem., Int. Ed., 2016, 55, 16145–16148 CrossRef CAS PubMed.
- H. Vanda, Y. Dai, E. G. Wilson, R. Verpoorte and Y. H. Choi, C. R. Chim., 2018, 21, 628–638 CrossRef CAS.
- I. Mamajanov, A. E. Engelhart, H. D. Bean and N. V. Hud, Angew. Chem., Int. Ed., 2010, 49, 6310–6314 CrossRef CAS PubMed.
- D. Yu, Z. Xue and T. Mu, Cell Rep. Phys. Sci., 2022, 3, 100809 CrossRef CAS.
- D. Nakamura, I. Gunjishima, S. Yamaguchi, T. Ito, A. Okamoto, H. Kondo, S. Onda and K. Takatori, Nature, 2004, 430, 1009–1012 CrossRef CAS PubMed.
- E. R. Parnham, E. A. Drylie, P. S. Wheatley, A. M. Z. Slawin and R. E. Morris, Angew. Chem., 2006, 118, 5084–5088 CrossRef.
- J. Zhang, T. Wu, S. Chen, P. Feng and X. Bu, Angew. Chem., Int. Ed., 2009, 48, 3486–3490 CrossRef CAS PubMed.
- H. G. Liao, Y. X. Jiang, Z. Y. Zhou, S. P. Chen and S. G. Sun, Angew. Chem., Int. Ed., 2008, 47, 9100–9103 CrossRef CAS PubMed.
- O. S. Hammond, K. J. Edler, D. T. Bowron and L. Torrente-Murciano, Nat. Commun., 2017, 8, 14150 CrossRef CAS PubMed.
- O. S. Hammond, D. T. Bowron and K. J. Edler, Green Chem., 2016, 18, 2736–2744 RSC.
- M. Zhang, Y.-H. Liu, Z.-R. Shang, H.-C. Hu and Z.-H. Zhang, Catal. Commun., 2017, 88, 39–44 CrossRef CAS.
- B. Li, H. Zhang, J. Kaelin, S. Gao, F. Xia, B. An, K. Lu and Y. Cheng, ACS Appl. Nano Mater., 2023, 6, 3184–3190 CrossRef CAS.
- S. Datta, J. Mahin, E. Liberti, I. Manasi, K. J. Edler and L. Torrente-Murciano, ACS Sustainable Chem. Eng., 2023, 11, 10242–10251 CrossRef CAS PubMed.
- O. S. Hammond, R. S. Atri, D. T. Bowron, L. de Campo, S. Diaz-Moreno, L. L. Keenan, J. Doutch, S. Eslava and K. J. Edler, Nanoscale, 2021, 13, 1723–1737 RSC.
- P. C. Jhang, N. T. Chuang and S. L. Wang, Angew. Chem., Int. Ed., 2010, 49, 4200–4204 CrossRef CAS PubMed.
- B. T. Yonemoto, Z. Lin and F. Jiao, Chem. Commun., 2012, 48, 9132–9134 RSC.
- K. Y. Wang, D. Ding, S. Zhang, Y. Wang, W. Liu, S. Wang, S. H. Wang, D. Liu and C. Wang, Chem. Commun., 2018, 54, 4806–4809 RSC.
- Y. Lu, Y. Yang, X. Fan, Y. Li, D. Zhou, B. Cai, L. Wang, K. Fan and K. Zhang, Adv. Mater., 2022, 34, e2108178 CrossRef PubMed.
- O. S. Hammond, D. T. Bowron and K. J. Edler, ACS Sustainable Chem. Eng., 2019, 7, 4932–4940 CrossRef CAS.
- C. D'Agostino, R. C. Harris, A. P. Abbott, L. F. Gladden and M. D. Mantle, Phys. Chem. Chem. Phys., 2011, 13, 21383–21391 RSC.
- M. E. Di Pietro, O. Hammond, A. van den Bruinhorst, A. Mannu, A. Padua, A. Mele and M. Costa Gomes, Phys. Chem. Chem. Phys., 2021, 23, 107–111 RSC.
- D. V. Wagle, G. A. Baker and E. Mamontov, J. Phys. Chem. Lett., 2015, 6, 2924–2928 CrossRef CAS PubMed.
- M. Gilmore, L. M. Moura, A. H. Turner, M. Swadzba-Kwasny, S. K. Callear, J. A. McCune, O. A. Scherman and J. D. Holbrey, J. Chem. Phys., 2018, 148, 193823 CrossRef PubMed.
- K. Biernacki, H. K. S. Souza, C. M. R. Almeida, A. L. Magalhães and M. P. Gonçalves, ACS Sustainable Chem. Eng., 2020, 8, 18712–18728 CrossRef CAS.
- P. Kalhor, K. Ghandi, H. Ashraf and Z. Yu, Phys. Chem. Chem. Phys., 2021, 23, 13136–13147 RSC.
- A. Faraone, D. V. Wagle, G. A. Baker, E. C. Novak, M. Ohl, D. Reuter, P. Lunkenheimer, A. Loidl and E. Mamontov, J. Phys. Chem. B, 2018, 122, 1261–1267 CrossRef CAS PubMed.
- Y. Zhang, D. Poe, L. Heroux, H. Squire, B. W. Doherty, Z. Long, M. Dadmun, B. Gurkan, M. E. Tuckerman and E. J. Maginn, J. Phys. Chem. B, 2020, 124, 5251–5264 CrossRef CAS PubMed.
- Z. Naseem, R. A. Shehzad, A. Ihsan, J. Iqbal, M. Zahid, A. Pervaiz and G. Sarwari, Chem. Phys. Lett., 2021, 769, 138427 CrossRef CAS.
- O. S. Hammond, D. T. Bowron, A. J. Jackson, T. Arnold, A. Sanchez-Fernandez, N. Tsapatsaris, V. Garcia Sakai and K. J. Edler, J. Phys. Chem. B, 2017, 121, 7473–7483 CrossRef CAS PubMed.
- M. Atilhan and S. Aparicio, J. Phys. Chem. C, 2018, 122, 18029–18039 CrossRef CAS.
- A. Triolo, F. Lo Celso and O. Russina, J. Phys. Chem. B, 2020, 124, 2652–2660 CrossRef CAS PubMed.
- H. Yu, Z. Xue, X. Lan, Q. Liu, R. Shi and T. Mu, Cellulose, 2020, 27, 6175–6188 CrossRef.
- Q. Liu, X. Zhao, D. Yu, H. Yu, Y. Zhang, Z. Xue and T. Mu, Green Chem., 2019, 21, 5291–5297 RSC.
- W. Chen, J. Jiang, X. Lan, X. Zhao, H. Mou and T. Mu, Green Chem., 2019, 21, 4748–4756 RSC.
- A. Pandey, R. Rai, M. Pal and S. Pandey, Phys. Chem. Chem. Phys., 2014, 16, 1559–1568 RSC.
- M. B. Taysun, E. Sert and F. S. Atalay, J. Chem. Eng. Data, 2017, 62, 1173–1181 CrossRef CAS.
- M. Q. Farooq, G. A. Odugbesi, N. M. Abbasi and J. L. Anderson, ACS Sustainable Chem. Eng., 2020, 8, 18286–18296 CrossRef CAS.
- C. Florindo, A. J. S. McIntosh, T. Welton, L. C. Branco and I. M. Marrucho, Phys. Chem. Chem. Phys., 2017, 20, 206–213 RSC.
- J. L. Anderson, J. Ding, T. Welton and D. W. Armstrong, J. Am. Chem. Soc., 2002, 124, 14247–14254 CrossRef CAS PubMed.
- M. H. Abraham, Chem. Soc. Rev., 1993, 22, 73–83 RSC.
- A. Valvi, J. Dutta and S. Tiwari, J. Phys. Chem. B, 2017, 121, 11356–11366 CrossRef CAS PubMed.
- A. R. R. Teles, E. V. Capela, R. S. Carmo, J. A. P. Coutinho, A. J. D. Silvestre and M. G. Freire, Fluid Phase Equilib., 2017, 448, 15–21 CrossRef CAS PubMed.
- A. Sanchez-Fernandez, K. J. Edler, T. Arnold, D. Alba Venero and A. J. Jackson, Phys. Chem. Chem. Phys., 2017, 19, 8667–8670 RSC.
- L. Das and S. Adhikari, J. Phys. Chem. B, 2018, 122, 8900–8907 CrossRef CAS PubMed.
- F. Milano, L. Giotta, M. R. Guascito, A. Agostiano, S. Sblendorio, L. Valli, F. M. Perna, L. Cicco, M. Trotta and V. Capriati, ACS Sustainable Chem. Eng., 2017, 5, 7768–7776 CrossRef CAS.
- F. Zhen, L. Percevault, L. Paquin, E. Limanton, C. Lagrost and P. Hapiot, J. Phys. Chem. B, 2020, 124, 1025–1032 CrossRef CAS PubMed.
- J. A. McCune, M. F. Kuehnel, E. Reisner and O. A. Scherman, Chem, 2020, 6, 1819–1830 CAS.
- S. Kaur, A. Gupta and H. K. Kashyap, J. Phys. Chem. B, 2016, 120, 6712–6720 CrossRef CAS PubMed.
- V. Alizadeh, D. Geller, F. Malberg, P. B. Sanchez, A. Padua and B. Kirchner, ChemPhysChem, 2019, 20, 1786–1792 CrossRef CAS PubMed.
- A. Felipe, C. A. Lovenduski, J. L. Baker and G. E. Lindberg, Phys. Chem. Chem. Phys., 2022, 24, 13720–13729 RSC.
- S. Spittle, D. Poe, B. Doherty, C. Kolodziej, L. Heroux, M. A. Haque, H. Squire, T. Cosby, Y. Zhang, C. Fraenza, S. Bhattacharyya, M. Tyagi, J. Peng, R. A. Elgammal, T. Zawodzinski, M. Tuckerman, S. Greenbaum, B. Gurkan, C. Burda, M. Dadmun, E. J. Maginn and J. Sangoro, Nat. Commun., 2022, 13, 219 CrossRef CAS PubMed.
- R. Maglia de Souza, M. Karttunen and M. C. C. Ribeiro, J. Chem. Inf. Model., 2021, 61, 5938–5947 CrossRef CAS PubMed.
- Y. Cui and D. G. Kuroda, J. Phys. Chem. A, 2018, 122, 1185–1193 CrossRef CAS PubMed.
- S. McDonald, T. Murphy, S. Imberti, G. G. Warr and R. Atkin, J. Phys. Chem. Lett., 2018, 9, 3922–3927 CrossRef CAS PubMed.
- S. Zhang, R. Shi, X. Ma, L. Lu, Y. He, X. Zhang, Y. Wang and Y. Deng, Chem. – Eur. J., 2012, 18, 11904–11908 CrossRef CAS PubMed.
- S. Chatterjee, T. Haldar, D. Ghosh and S. Bagchi, J. Phys. Chem. B, 2020, 124, 3709–3715 CrossRef CAS PubMed.
- A. Malik and H. K. Kashyap, Phys. Chem. Chem. Phys., 2021, 23, 3915–3924 RSC.
- L. Percevault, A. Jani, T. Sohier, L. Noirez, L. Paquin, F. Gauffre and D. Morineau, J. Phys. Chem. B, 2020, 124, 9126–9135 CrossRef CAS PubMed.
- N. Subba, E. Tarif, P. Sen and R. Biswas, J. Phys. Chem. B, 2020, 124, 1995–2005 CrossRef CAS PubMed.
- S. Banerjee, P. K. Ghorai, S. Das, J. Rajbangshi and R. Biswas, J. Chem. Phys., 2020, 153, 234502 CrossRef CAS PubMed.
- S. Kaur, S. Sharma and H. K. Kashyap, J. Chem. Phys., 2017, 147, 194507 CrossRef PubMed.
- M. H. Mamme, S. L. C. Moors, H. Terryn, J. Deconinck, J. Ustarroz and F. De Proft, J. Phys. Chem. Lett., 2018, 9, 6296–6304 CrossRef CAS PubMed.
- J. J. Buzolic, H. Li, Z. M. Aman, G. G. Warr and R. Atkin, J. Colloid Interface Sci., 2022, 616, 121–128 CrossRef CAS PubMed.
- S. Panda, K. Kundu, J. Kiefer, S. Umapathy and R. L. Gardas, J. Phys. Chem. B, 2019, 123, 3359–3371 CrossRef CAS PubMed.
- X. Lan, X. Wang, W. Du, T. Mu and X. Z. Lan, Phys. Chem. Chem. Phys., 2021, 23, 13785–13788 RSC.
- S. S. Hossain, S. Paul and A. Samanta, J. Phys. Chem. B, 2019, 123, 6842–6850 CrossRef CAS PubMed.
- S. S. Hossain and A. Samanta, Phys. Chem. Chem. Phys., 2018, 20, 24613–24622 RSC.
- S. Chatterjee, D. Ghosh, T. Haldar, P. Deb, S. S. Sakpal, S. H. Deshmukh, S. M. Kashid and S. Bagchi, J. Phys. Chem. B, 2019, 123, 9355–9363 CrossRef CAS PubMed.
- L. Kollau, R. Tuinier, J. Verhaak, J. den Doelder, I. A. W. Filot and M. Vis, J. Phys. Chem. B, 2020, 124, 5209–5219 CrossRef CAS PubMed.
- Y. Hou, B. Zhang, M. Gao, S. Ren and W. Wu, J. Chem. Thermodyn., 2023, 179, 106999 CrossRef CAS.
- C. Vidal, J. Garcia-Alvarez, A. Hernan-Gomez, A. R. Kennedy and E. Hevia, Angew. Chem., Int. Ed., 2014, 53, 5969–5973 CrossRef CAS PubMed.
- W. Chen, Z. Xue, J. Wang, J. Jiang, X. Zhao and T. Mu, Acta Phys.-Chim. Sin., 2018, 34, 904–911 CAS.
- B. Janković, N. Manić, I. Perović, M. Vujković and N. Zdolšek, J. Mol. Liq., 2023, 374, 121274 CrossRef.
- C. Florindo, F. S. Oliveira, L. P. N. Rebelo, A. M. Fernandes and I. M. Marrucho, ACS Sustainable Chem. Eng., 2014, 2, 2416–2425 CrossRef CAS.
- S. Liu, D. Yu, Y. Chen, R. Shi, F. Zhou and T. Mu, Ind. Eng. Chem. Res., 2022, 61, 14347–14354 CrossRef CAS.
- Y. Chen, D. Yu, L. Fu, M. Wang, D. Feng, Y. Yang, X. Xue, J. Wang and T. Mu, Phys. Chem. Chem. Phys., 2019, 21, 11810–11821 RSC.
- S. Kaur and H. K. Kashyap, J. Phys. Chem. B, 2018, 122, 5242–5250 CrossRef CAS PubMed.
- Q. Li, J. Jiang, G. Li, W. Zhao, X. Zhao and T. Mu, Sci. China: Chem., 2016, 59, 571–577 CrossRef CAS.
- L. Das, S. Mukherjee, D. Kumar Maity and S. Adhikari, J. Mol. Liq., 2022, 360, 119377 CrossRef CAS.
- Z. Xue, L. Qin, J. Jiang, T. Mu and G. Gao, Phys. Chem. Chem. Phys., 2018, 20, 8382–8402 RSC.
- A. Kityk, P. Švec, J. Šoltys, V. Pavlik and M. Hnatko, J. Mol. Liq., 2023, 375, 121316 CrossRef CAS.
- A. Pandey and S. Pandey, J. Phys. Chem. B, 2014, 118, 14652–14661 CrossRef CAS PubMed.
- O. Longeras, A. Gautier, K. Ballerat-Busserolles and J.-M. Andanson, ACS Sustainable Chem. Eng., 2020, 8, 12516–12520 CrossRef CAS.
- S. Chen, J. Zhang, T. Wu, P. Feng and X. Bu, Dalton Trans., 2010, 39, 697–699 RSC.
- J. Qiu, P. Guan, Y. Zhao, Z. Li, H. Wang and J. Wang, Green Chem., 2020, 22, 7537–7542 RSC.
- S. D. Çalhan, M. Ö. Alaş, M. Aşık, F. N. D. Kaya and R. Genç, J. Mater. Sci., 2018, 53, 15362–15375 CrossRef.
- X. Zhao, X. Lan, D. Yu, H. Fu, Z. Liu and T. Mu, Chem. Commun., 2018, 54, 13010–13013 RSC.
- J. Jiang, C. Yan, X. Zhao, H. Luo, Z. Xue and T. Mu, Green Chem., 2017, 19, 3023–3031 RSC.
- S. Datta, C. Jo, M. De Volder and L. Torrente-Murciano, ACS Appl. Mater. Interfaces, 2020, 12, 18803–18812 CrossRef CAS PubMed.
- S. Kumar-Krishnan, R. Esparza and U. Pal, ACS Omega, 2020, 5, 3699–3708 CrossRef CAS PubMed.
- H. Lei, S. Singh Siwal, X. Zhang and Q. Zhang, J. Colloid Interface Sci., 2020, 571, 1–12 CrossRef CAS PubMed.
- O. Oderinde, M. Kang, M. Kalulu, F. Yao and G. Fu, Superlattices Microstruct., 2019, 125, 103–112 CrossRef CAS.
- P. Jittiarporn, L. Sikong, K. Kooptarnond, W. Taweepreda, S. Stoenescu, S. Badilescu and V.-V. Truong, Surf. Coat. Technol., 2017, 327, 66–74 CrossRef CAS.
- D. P. Jaihindh, A. Manikandan, Y. L. Chueh and Y. P. Fu, ChemSusChem, 2020, 13, 2726–2738 CrossRef CAS PubMed.
- B. Sriram, J. N. Baby, S.-F. Wang, M. R. Ranjitha, M. Govindasamy and M. George, ACS Sustainable Chem. Eng., 2020, 8, 17772–17782 CrossRef CAS.
- L. Ding, Z. Li, L. Chen and Z. Qi, Electrochim. Acta, 2023, 455, 142424 CrossRef CAS.
- D. Xue, D. Zhu, H. Duan, Z. Wang, Y. Lv, W. Xiong, L. Li, M. Liu and L. Gan, Chem. Commun., 2019, 55, 11219–11222 RSC.
- M. T. Vallejo-Macías, C. L. Recio-Colmenares, J. B. Pelayo-Vázquez, S. Gómez-Salazar, F. Carvajal-Ramos, J. F. Soltero-Martínez, M. Vázquez-Lepe, J. D. Mota-Morales and M. G. Pérez-García, ACS Appl. Nano Mater., 2020, 3, 5794–5806 CrossRef.
- O. S. Hammond, D. T. Bowron and K. J. Edler, Angew. Chem., Int. Ed., 2017, 56, 9782–9785 CrossRef CAS PubMed.
- W. Liu, Y. Yu, L. Cao, G. Su, X. Liu, L. Zhang and Y. Wang, J. Hazard. Mater., 2010, 181, 1102–1108 CrossRef CAS PubMed.
- Q. Wang, B. Dong, Y. Zhao, F. Huang, J. Xie, G. Cui and B. Tang, Chem. Eng. J., 2018, 348, 811–819 CrossRef CAS.
- M. Karimi and M. J. Eshraghi, J. Alloys Compd., 2017, 696, 171–176 CrossRef CAS.
- K. Aruchamy, R. N. Maalige, M. M. Halanur, A. Mahto, R. Nagaraj, D. Kalpana, D. Ghosh, D. Mondal and S. K. Nataraj, Chem. Eng. J., 2020, 379, 112327 CrossRef.
- M. Starykevich, A. N. Salak, D. K. Ivanou, K. A. Yasakau, P. S. André, R. A. S. Ferreira, M. L. Zheludkevich and M. G. S. Ferreira, J. Electrochem. Soc., 2017, 164, D88–D94 CrossRef CAS.
- V. S. Cvetković, N. M. Vukićević, N. Jovićević, J. S. Stevanović and J. N. Jovićević, Trans. Nonferrous Met. Soc. China, 2020, 30, 823–834 CrossRef.
- J. Wang, P. Wang, Q. Wang, H. Mou, B. Cao, D. Yu, D. Wang, S. Zhang and T. Mu, ACS Sustainable Chem. Eng., 2018, 6, 15480–15486 CrossRef CAS.
- E. Gunnell, A. Avery, L. Gunnell, M. Spotts, M. Stoddard and J. N. Harb, J. Electrochem. Soc., 2021, 168, 046501 CrossRef CAS.
- L. Wei, C.-D. Xu, L. Huang, Z.-Y. Zhou, S.-P. Chen and S.-G. Sun, J. Phys. Chem. C, 2015, 120, 15569–15577 CrossRef.
- A. Bordet, S. El Sayed, M. Sanger, K. J. Boniface, D. Kalsi, K. L. Luska, P. G. Jessop and W. Leitner, Nat. Chem., 2021, 13, 916 CrossRef CAS PubMed.
- R. Bernasconi, A. Lucotti, L. Nobili and L. Magagnin, J. Electrochem. Soc., 2018, 165, D620–D627 CrossRef CAS.
- Z. Su, C. Xu, Y. Hua, J. Li, J. Ru, M. Wang, L. Xiong and Y. Zhang, Int. J. Electrochem. Sci., 2016, 11, 3311 CrossRef CAS.
- G.-F. Cai, J.-P. Tu, C.-D. Gu, J.-H. Zhang, J. Chen, D. Zhou, S.-J. Shi and X.-L. Wang, J. Mater. Chem. A, 2013, 1., 4286–4292 RSC.
- L. Chen, J. Zeng, M. Guo, R. Xue, R. Deng and Q. Zhang, J. Colloid Interface Sci., 2021, 583, 594–604 CrossRef CAS PubMed.
- C. Yang, T. He, W. Zhou, R. Deng and Q. Zhang, ACS Sustainable Chem. Eng., 2020, 8, 13793–13804 CrossRef CAS.
- K. Li, T.-Z. Ren, Z.-Y. Yuan and T. J. Bandosz, Int. J. Hydrogen Energy, 2018, 43, 10448–10457 CrossRef CAS.
- A. P. Abbott, M. Azam, K. S. Ryder and S. Saleem, Trans. IMF, 2018, 96, 297–303 CrossRef CAS.
- A. D. Ballantyne, G. C. Forrest, G. Frisch, J. M. Hartley and K. S. Ryder, Phys. Chem. Chem. Phys., 2015, 17, 30540–30550 RSC.
- S.-P. Wang, Y.-Y. Hsieh, P.-Y. Chen and I. W. Sun, J. Electrochem. Soc., 2019, 166, D768–D775 CrossRef CAS.
- E. Gutierrez, J. A. Rodriguez, J. Cruz-Borbolla, Y. Castrillejo and E. Barrado, Int. J. Electrochem. Sci., 2018, 13, 11016–11023 CrossRef CAS.
- L. Anicai, S. Costovici, A. Cojocaru, A. Manea and T. Visan, Trans. IMF, 2016, 93, 302–312 CrossRef.
- V. S. Protsenko, L. S. Bobrova, S. A. Korniy, A. A. Kityk and F. I. Danilov, Funct. Mater., 2018, 25, 539–545 CrossRef CAS.
- P. Sebastián, E. Gómez, V. Climent and J. M. Feliu, Electrochem. Commun., 2017, 78, 51–55 CrossRef.
- M. A. Miller, J. S. Wainright and R. F. Savinell, J. Electrochem. Soc., 2017, 164, A796–A803 CrossRef CAS.
- M. Steichen, M. Thomassey, S. Siebentritt and P. J. Dale, Phys. Chem. Chem. Phys., 2011, 13, 4292–4302 RSC.
- R. Bernasconi and L. Magagnin, J. Optoelectron. Adv. Mater., 2015, 17, 122–126 RSC.
- M. W. Guo, C. B. Sun, W. Q. Yang, L. Chen, H. Lei and Q. B. Zhang, Electrochim. Acta, 2020, 341, 136017 CrossRef CAS.
- R. Kang, M. An, F. Cui, J. Wang and J. Liang, Mater. Res. Express, 2019, 6, 1165a6 CrossRef.
- Y.-S. Liao, P.-Y. Chen and I. W. Sun, Electrochim. Acta, 2016, 214, 265–275 CrossRef CAS.
- F. Soma, Q. Rayée, M. Bougouma, C. Baustert, C. Buess-Herman and T. Doneux, Electrochim. Acta, 2020, 345, 136165 CrossRef CAS.
- L. Wei, Y.-J. Fan, H.-H. Wang, N. Tian, Z.-Y. Zhou and S.-G. Sun, Electrochim. Acta, 2012, 76, 468–474 CrossRef CAS.
- M. Jayakumar, C. Murugesan, C. Andrew, K. Natarajan and R. Sekar, J. Electrochem. Soc., 2022, 169, 032510 CrossRef CAS.
- J. Bu, J. Ru, Z. Wang, Y. Hua, C. Xu, Y. Zhang and Y. Wang, Adv. Powder Technol., 2019, 30, 2859–2867 CrossRef CAS.
- E. Gómez, P. Cojocaru, L. Magagnin and E. Valles, J. Electroanal. Chem., 2011, 658, 18–24 CrossRef.
- Z. Wang, J. Ru, Y. Hua, D. Wang and J. Bu, J. Electrochem. Soc., 2020, 167, 082504 CrossRef CAS.
- A. P. Abbott, J. C. Barron, G. Frisch, S. Gurman, K. S. Ryder and A. Fernando Silva, Phys. Chem. Chem. Phys., 2011, 13, 10224–10231 RSC.
- P.-K. Wang, W.-J. Lin and I. W. Sun, J. Electrochem. Soc., 2019, 166, D374–D380 CrossRef CAS.
- B. Bozzini, B. Busson, C. Humbert, C. Mele and A. Tadjeddine, Electrochim. Acta, 2016, 218, 208–215 CrossRef CAS.
- Y. S. Gao, W. C. Hu, X. Q. Gao and B. X. Duan, Surf. Eng., 2013, 28, 590–593 CrossRef.
- K. Kopczyński and G. Lota, Electrochem. Commun., 2019, 107, 106538 CrossRef.
- G. Saravanan and S. Mohan, J. Alloys Compd., 2012, 522, 162–166 CrossRef CAS.
- S.-L. Wang, J.-B. Lin, Q.-Y. Cai and Y. Zhang, J. Alloys Compd., 2008, 450, 142–147 CrossRef CAS.
- A. Liu, Z. Shi and R. G. Reddy, Metall. Mater. Trans. B, 2020, 51, 1162–1168 CrossRef CAS.
- W.-S. Chiang, J.-Q. Huang, P.-C. Chen, P.-W. Wu, A. Joi and Y. Dordi, J. Alloys Compd., 2018, 742, 38–44 CrossRef CAS.
- S. Geng, H. Dong, Y. Lu, S. Wang, Y. Huang, X. Zou, Y. Zhang, Q. Xu and X. Lu, Sep. Purif. Technol., 2020, 242, 116779 CrossRef CAS.
- X. Xie, X. Zou, X. Lu, Q. Xu, C. Lu, C. Chen and Z. Zhou, J. Appl. Electrochem., 2017, 47, 679–689 CrossRef CAS.
- T. Yanai, K. Shiraishi, Y. Watanabe, T. Ohgai, M. Nakano, K. Suzuki and H. Fukunaga, J. Appl. Phys., 2015, 117, 17A925 CrossRef.
- Z. Wang, T. Wu, J. Ru, Y. Hua, J. Bu and D. Wang, Surf. Coat. Technol., 2021, 406, 126636 CrossRef CAS.
- J. Li, H. Lai, B. Fan, B. Zhuang, L. Guan and Z. Huang, J. Alloys Compd., 2009, 477, 547–551 CrossRef CAS.
- Y. You, C. Gu, X. Wang and J. Tu, J. Electrochem. Soc., 2012, 159, D642–D648 CrossRef CAS.
- V. R. C. Thanu, C. Andrew and M. Jayakumar, Surf. Interfaces, 2020, 19, 100539 CrossRef CAS.
- M. Manolova, R. Böck, I. Scharf, T. Mehner and T. Lampke, J. Alloys Compd., 2021, 855, 157462 CrossRef CAS.
- L. Vieira, J. Burt, P. W. Richardson, D. Schloffer, D. Fuchs, A. Moser, P. N. Bartlett, G. Reid and B. Gollas, ChemistryOpen, 2017, 6, 393–401 CrossRef CAS PubMed.
- L. Anicai, A. Petica, S. Costovici, C. Moise, O. Brincoveanu and T. Visan, Coatings, 2019, 9, 800 CrossRef CAS.
- M. Bučko, D. Culliton, A. J. Betts and J. B. Bajat, Trans. IMF, 2017, 95, 60–64 CrossRef.
- C. Xu, Q. Wu, Y. Hua and J. Li, J. Solid State Electrochem., 2014, 18, 2149–2155 CrossRef CAS.
- M. Li, B. Q. Chen, T. T. Xiong, L. X. Gao, C. Du, Y. N. Zhu and S. M. Zhang, J. Dispersion Sci. Technol., 2019, 41, 941–947 CrossRef.
- S. Fashu, L. Mudzingwa, R. Khan and M. Tozvireva, Trans. IMF, 2017, 96, 20–26 CrossRef.
- A. Renjith and V. Lakshminarayanan, ACS Appl. Nano Mater., 2020, 3, 2705–2712 CrossRef CAS.
- S. Kumaraguru, R. Pavulraj, J. Vijayakumar and S. Mohan, J. Alloys Compd., 2017, 693, 1143–1149 CrossRef CAS.
- R. Li, Y. Hou and J. Liang, Appl. Surf. Sci., 2016, 367, 449–458 CrossRef CAS.
- W. Q. Yang, Y. X. Hua, Q. B. Zhang, H. Lei and C. Y. Xu, Electrochim. Acta, 2018, 273, 71–79 CrossRef CAS.
- L. Juárez-Marmolejo, B. Maldonado-Teodocio, M. G. Montes de Oca-Yemha, M. Romero-Romo, M. T. Ramírez-Silva, E. M. Arce-Estrada, P. Morales-Gil, J. Mostany and M. Palomar-Pardavé, J. Electrochem. Soc., 2020, 167, 112509 CrossRef.
- A. Renjith and V. Lakshminarayanan, J. Mater. Chem. A, 2015, 3, 3019–3028 RSC.
- D. Xu, Y. Yang, B. Zhang, Z. Yang, S. Liu and T. Mu, ChemSusChem, 2022, 15, e202200822 CrossRef CAS PubMed.
- H. Hu, S. Guo, Y. Wang and K. Shi, J. Electrochem. Soc., 2020, 167, 166512 CrossRef.
- O. I. Zaytsev, M. R. Ehrenburg, E. B. Molodkina, P. Broekmann and A. V. Rudnev, J. Electroanal. Chem., 2022, 926, 116940 CrossRef CAS.
- S. Wang, X. Xiong, X. Zou, K. Ding, Z. Pang, Q. Xu, Z. Zhou and X. Lu, J. Mater. Chem. A, 2020, 8, 4354–4361 RSC.
- C. Zhang, Y. Jia, Y. Jing, H. Wang and K. Hong, J. Mol. Model., 2014, 20, 2374 CrossRef PubMed.
- R. Gupta, J. Gamare, M. Sahu, K. Pandey and S. K. Gupta, J. Mol. Liq., 2021, 329, 115550 CrossRef CAS.
- R. Zhou, C. Xu, J. Yang, D. Guan and J. Cai, Chem. Lett., 2020, 49, 585–588 CrossRef CAS.
- D.-W. Lee, M.-H. Jin, J.-H. Park, Y.-J. Lee, Y.-C. Choi, J. Chan Park and D. H. Chun, ACS Sustainable Chem. Eng., 2018, 6, 12241–12250 CrossRef CAS.
- H. Mou, J. Wang, D. Yu, D. Zhang, F. Lu, L. Chen, D. Wang and T. Mu, J. Mater. Chem. A, 2019, 7, 13455–13459 RSC.
- G. M. Thorat, H. S. Jadhav, W.-J. Chung and J. G. Seo, J. Alloys Compd., 2018, 732, 694–704 CrossRef CAS.
- F. Jiang, Y. Zhou, J. Su, Y.-F. Long, X.-Y. Lv and Y.-X. Wen, Ionics, 2020, 26, 5553–5563 CrossRef CAS.
- A. Verma, D. P. Jaihindh and Y. P. Fu, Dalton Trans., 2019, 48, 8594–8610 RSC.
- H. Mou, J. Wang, D. Yu, D. Zhang, W. Chen, Y. Wang, D. Wang and T. Mu, ACS Appl. Mater. Interfaces, 2019, 11, 44360–44365 CrossRef CAS PubMed.
- J. Wang, H. Mou, R. Li, Y. Li, D. Wang, Z. Xue and T. Mu, Green Chem., 2018, 20, 5266–5270 RSC.
- R. Boston, P. Y. Foeller, D. C. Sinclair and I. M. Reaney, Inorg. Chem., 2017, 56, 542–547 CrossRef CAS PubMed.
- J. N. Baby, B. Sriram, S.-F. Wang and M. George, ACS Sustainable Chem. Eng., 2020, 8, 1479–1486 CrossRef CAS.
- X. Zhao, Y. Zhou, A.-L. Jin, K. Huang, F. Liu and D.-J. Tao, New J. Chem., 2018, 42, 15871–15878 RSC.
- H. Ying, J. Bi, H. Xu, G. Wu, X. Wu, J. Hao and Z. Li, ACS Sustainable Chem. Eng., 2022, 10, 6766–6774 CrossRef CAS.
- Y. Wei, J. Jiang, J. Dong, Y. Xu, J. Fu and Q. Xu, Green Chem., 2022, 24, 8014–8020 RSC.
- S. K. Shinde, S. S. Karade, N. C. Maile, H. M. Yadav, A. D. Jagadale, M. B. Jalak and D. Y. Kim, Int. J. Energy Res., 2022, 46, 16693–16715 CrossRef CAS.
- Q. Shan, Q. Ding, X. Wang and W. Wu, Langmuir, 2022, 38, 8169–8178 CrossRef CAS PubMed.
- C. Gu, Y. Peng, J. Li, H. Wang, X. Q. Xie, X. Cao and C. S. Liu, Angew. Chem., Int. Ed., 2020, 59, 18768–18773 CrossRef CAS PubMed.
- M. Wang, R. Li, G. Chen, S. Zhou, X. Feng, Y. Chen, M. He, D. Liu, T. Song and H. Qi, ACS Appl. Mater. Interfaces, 2019, 11, 14313–14321 CrossRef CAS PubMed.
- J. Ruiz-Olles, P. Slavik, N. K. Whitelaw and D. K. Smith, Angew. Chem., Int. Ed., 2019, 58, 4173–4178 CrossRef CAS PubMed.
- M. W. Logan, S. Langevin, B. Tan, A. W. Freeman, C. Hoffman, D. B. Trigg and K. Gerasopoulos, J. Mater. Chem. A, 2020, 8, 8485–8495 RSC.
- C. Wang, H. Zhang, S. Dong, Z. Hu, R. Hu, Z. Guo, T. Wang, G. Cui and L. Chen, Chem. Mater., 2020, 32, 9167–9175 CrossRef CAS.
- D. Yu, H. Mou, H. Fu, X. Lan, Y. Wang and T. Mu, Chem. – Asian J., 2019, 14, 4183–4188 CrossRef CAS PubMed.
- P. Janicka, M. Kaykhaii, J. Płotka-Wasylka and J. Gębicki, Green Chem., 2022, 24, 5035–5045 RSC.
- Y. Xu, A. Li, S. Xue, S. Ding and Q. Zhang, Talanta, 2023, 260, 124556 CrossRef CAS PubMed.
- S. Wu, C. Cai, F. Li, Z. Tan and S. Dong, Angew. Chem., Int. Ed., 2020, 59, 11871–11875 CrossRef CAS PubMed.
- Z.-H. Cai, J.-D. Wang, L. Liu, L.-D. Ruan, Q. Gu, X.-Y. Yan, L.-N. Fu, P.-Q. Zhao, S. Zhang and Y.-J. Fu, Chem. Eng. J., 2023, 457., 141333 CrossRef.
- S. Guan, Z. Li, B. Xu, J. Wu, N. Wang, J. Zhang, J. Han, T. Guan, J. Wang and K. Li, Chem. Eng. J., 2022, 441., 136022 CrossRef.
- H. Zhang, K. Lu, B. Li, Y. Liu, Y. Su, R. Wang and Y. Cheng, ACS Appl. Mater. Interfaces, 2020, 12, 42704–42710 CrossRef CAS PubMed.
- C. R. Ashworth, R. P. Matthews, T. Welton and P. A. Hunt, Phys. Chem. Chem. Phys., 2016, 18, 18145–18160 RSC.
- S. S. Karade, S. Lalwani, J.-H. Eum and H. Kim, Sustainable Energy Fuels, 2020, 4, 3066–3076 RSC.
- D. Shi, F. Zhou, W. Mu, C. Ling, T. Mu, G. Yu and R. Li, Phys. Chem. Chem. Phys., 2022, 24, 26029–26036 RSC.
- P. T. Anastas and J. C. Warner, Green Chemistry: Theory and Practice, Oxford University Press, New York, 1998 Search PubMed.
- R. A. Sheldon, Green Chem., 2017, 19, 18–43 RSC.
| This journal is © The Royal Society of Chemistry 2024 |