Mechanochemical and aging-based reductive amination with chitosan and aldehydes affords high degree of substitution functional biopolymers†
Galen
Yang‡
 a,
Sophie
Régnier
b,
Noah
Huin
a,
Tracy
Liu
a,
Edmond
Lam‡
a,
Sophie
Régnier
b,
Noah
Huin
a,
Tracy
Liu
a,
Edmond
Lam‡
 *ab and
Audrey
Moores‡
*ab and
Audrey
Moores‡
 *ac
*ac
aCentre in Green Chemistry and Catalysis, Department of Chemistry, McGill, University, 801 Sherbrooke St. West, Montreal, QC H3A 0B8, Canada. E-mail: Audrey.moores@mcgill.ca
bNational Research Council Canada, Aquatic and Crop Resource Development Research Centre, 6100 Royalmount Avenue, Montreal, Quebec H4P 2R2, Canada
cDepartment of Materials Engineering, McGill University, 3610 University Street, Montreal, Quebec H3A 0C5, Canada
First published on 25th March 2024
Abstract
Chitosan is readily available from various biomass waste streams including crustaceans, cephalopods, insects, and fungus. The polymer possesses primary amine groups which are great handles for functionalization. Yet efficient functionalization with high degree of substitution is challenging to achieve via solvothermal methods due to limitations in chitosan solvation properties. Herein we report a mechanochemical and aging-based method directly addressing this point. Working in the solid phase helps stabilize the formation of Schiff bases from chitosan and aldehydes, affording a novel pathway to the green functionalization of chitosan by reductive alkylation, with unprecedentedly high degrees of substitution. The method showed great efficacy and compatibility for chitosan to be functionalized with 21 different aldehyde substrates and a low process mass intensity (PMI) of 36. This work also opens a new avenue for the development of mechanochemical and aging-based reductive amination transformations.
Introduction
Chitin is the second most abundant biopolymer and the most abundant nitrogen-containing biopolymer.1 Besides the great availability of polysaccharides, this class of natural polymers is also biodegradable and biocompatible, which makes it ideal for high-value biomedical applications.2,3 Chitin and its deacetylated derivative chitosan (Chs) can be extracted from various biomass waste streams, including crustaceans, cephalopods, insects, and fungi on a large scale annually.4 The primary amine groups of Chs not only impart unique antimicrobial ability on the biopolymer,5,6 but permits important chemical modifications for the introduction of new, tailor-made structures.7–9Amines are amenable to a wide range of organic transformations, such as reactions with acyl halide,10 sulfonyl chlorides,11 and alkyl halides.12 The coupling of amines with aldehydes to form Schiff bases13 is a popular and versatile functionalization approach. When subjected to subsequent reduction, Schiff bases form stable substituted amines and enamines, in an overall pathway called reductive amination.14 Despite these prospects, Chs functionalization faces several limitations, the most serious one being its poor solubility in most media.15,16 Indeed, while Chs dissolves well in aqueous acidic media, sides reactions including Chs hydrolysis occurs under these conditions.17,18 Additionally, aqueous solutions of Chs also tend to feature high viscosity, reducing mixability and processability,19,20 a problem that restricts the feasibility of some Chs chemical modifications. These complications are a shared conundrum in polysaccharide functionalization, leading to a common necessity of using harsh reaction conditions or using excess reagents to accomplish the target processes,21–23 impairing the competitiveness of natural polymers against synthetic polymers.24–26
Mechanochemistry is an emerging synthetic methodology27–30 which utilizes mechanical force to trigger chemical phenomena. It has been shown to reduce or eliminate the need for auxiliary substrates such as solvent, accelerate chemical reaction,31 reduce energy requirement30 and even afford reactivity and selectivity that have not been observed in solution-based chemistry.29,32 Besides reactions happening during mechanical treatment, the concept of aging has been introduced, in which reactions occur in the solid phase after an initial mixing phase.33 These solid-state methodologies are well suited in the context of biomass conversion and polysaccharide functionalization, especially as such starting materials tend to be insoluble,34 as demonstrated with chitin extraction,35,36 and conversion.36,37 Our group has developed a workflow using the combination of mechanochemistry and aging to extract chitin from crustacean wastes;35 deacetylate chitin into Chs;38 and hydrolyze Chs to achieve controlled molecular weight.39 Similarly, Van Poucke et al. recently reported a synthesis of water-soluble N-sulfonated Chs by mechanochemistry.40
Imine, or Schiff-base (SB), formation is one of the most studied functionalization of Chs and has led to applications that harness its bioactivities,41–45 capability as catalyst support for chemical synthesis,46–48 ability to absorb heavy metal ions,49 and sensing capability.50 The conventional synthesis of a Chs SB relies on the dissolution of Chs in an acetic acid solution or suspension in alcohol under reflux conditions.51–53 Although stoichiometric amount of aldehydes were used in these reactions, the formation of SB was not quantitatively reported in term of degree of substitution (DS).42–44,46–50 Mechanochemistry offers an interesting approach to SB synthesis,54 as illustrated by the example of Crawford et al. who reported the synthesis of a diamine from 4,4′-oxydianiline and 2 equivalents of ortho-vanillin by either ball milling or twin-screw extrusion.55 More recently, Fatika et al. reported a simple solid-state synthesis of air stable Chs SB via solvent-free mechanochemical grafting with a scope of three benzaldehyde derivatives.56
A SB is inherently unstable under acidic conditions and will regenerate into its carbonyl and the amine components.57,58 One simple yet effective strategy to solve this instability is to reduce the imine into an alkylated amine.59 This process, the reductive amination (reductive alkylation from the perspective of the amine), ensures access to a compound able to withstand a much broader range of chemical conditions.60,61 In general, reductive amination is also proposed as a greener way of making substituted amines since it avoids the use of reactive and potentially genotoxic alkylating reagents that are used in traditional SN2-type reactions.62,63 Reductive alkylation has been employed as an effective way to functionalize Chs to valuable materials.64–67 Bobu et al. reported a solution-based method to functionalize Chs with octanal into N-alkyl Chs via reductive amination, albeit with a low DS of 0.03, towards paper-making applications.68 In that sense, mechanochemistry appears as an exciting opportunity to improve Chs reductive alkylation and access higher levels of DS.
Herein we report a mechanochemical and aging-based approach to alkylate Chs via reductive amination, with a wide scope of functionalities. The resulting materials feature excellent stability. We investigated the kinetics of the solid-state SB formation and reduction by sodium borohydride (NaBH4) via aging. We also examined the versatility of this method with a range of carbonyl compounds from aromatic to aliphatic aldehydes. The method also allows for the production of Chs derivatives inaccessible by solvothermal methods. We characterized the special properties of some Chs derivatives and suggested potential applications for the materials. More importantly, we evaluated this method with green metrics and demonstrated that our process features improved process mass intensity (PMI) as compared to solution-based methods. This work is also a proof of concept of the deployment of reductive amination methodology through mechanochemistry and aging.
Results and discussion
Mechanochemical synthesis of chitosan furfural Schiff-bases with a mixer mill
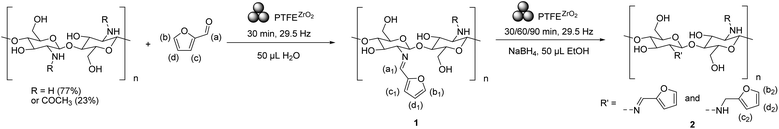
We first explored the preparation of Chs SB using furfural (a model aldehyde) by means of liquid-assisted grinding (LAG) mechanochemistry.69 A Retsch MM 400 mixer mill equipped with polytetrafluoroethylene (PTFE) SmartSnap grinding jars (15 mL) and one 7 mm zirconia ball in each jar was fitted with Chs and 1 equivalent of furfural (based on Chs primary amine units). As the process was optimized, we found that LAG favored the reaction, so we used 50 μL of deionized (DI) water and a milling time of 30 min under 29.5 Hz (Scheme 1). After set milling time, the reaction mixture was dispersed, washed, and filtered in ethanol (EtOH) and water before being dried.The resulting sample (1) was characterized by Fourier transform infra-red (FT-IR) spectroscopy, 13C magic angle spinning nuclear magnetic resonance (ssNMR) and 1H NMR. The appearance of a medium intensity peak at 1645 cm−1 was consistent with a C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N stretching absorption (see ESI† for complete characterization). The 13C ssNMR spectrum revealed the appearance of new peaks, especially one at 148.16 ppm characteristic of an imine carbon, as well as peaks for the introduction of a furan ring structure. Both FT-IR (Fig. S1†) and 13C ssNMR (Fig. S2†) were consistent with the formation of the desired compound 1.
N stretching absorption (see ESI† for complete characterization). The 13C ssNMR spectrum revealed the appearance of new peaks, especially one at 148.16 ppm characteristic of an imine carbon, as well as peaks for the introduction of a furan ring structure. Both FT-IR (Fig. S1†) and 13C ssNMR (Fig. S2†) were consistent with the formation of the desired compound 1.
Sample 1 was readily soluble at 1 wt% in 0.1% HCl solution, like untreated starting Chs, providing a homogeneous, viscous, and clear solution with a slight yellow hue. Solution state 1H NMR in 0.1% HCl in D2O was performed on 1 to calculate the degree of imination on Chs primary amine and the % conversion of the furfural as a reagent (see Fig. 1). Importantly, the starting Chs featured 23% acetylated monomers (calculated with 13C ssNMR), which cannot undergo SB formation and thus were used as an internal standard throughout this study for aldehyde % conversion and Chs degree of imination (see discussion in ESI†). 1H NMR of 1 revealed the presence of free furfural at 9.49 (a), 7.90 (b), 7.56 (c), and 6.75 ppm (d), and a set of four broad peaks at 8.80 (a1), 8.25 (b1), 7.98 (d1), and 6.99 ppm (c1), all with intensity at about 30% of the free furfural peaks. Together, the number and values of chemical shifts in this group, and the integration pattern confirm the formation of a 1. Overall, we measured 62.5% of free furfural and 34.3% of 1 (mass balance = 96.8%). A second 1H NMR spectrum was taken on the same NMR sample after 2 h and a complete disappearance of 1 was observed, with all the furfural type peaks being from free furfural (mass balance = 96.9%).
This analysis confirms that the condensation of a stoichiometric amount of furfural onto Chs was successful, although incomplete, and that under mildly acid liquid conditions (pH 5.5, RT), SB 1 is unstable and hydrolyzes back into free Chs and furfural. This instability is detrimental to the applicability of Chs SB because most applications would require a step of sample dissolution for dispersion or permeation. In those scenarios, the Chs SB would lose the desired properties obtained from the chemical modification and generate potentially hazardous free carbonyl compounds. This also explains why many synthetic methods rely on excess amounts of reagents to achieve Chs SB functionalization in a solution.42,43,46,49,52,53
Mechanochemical reductive alkylation of chitosan with furfural and NaBH4
One simple yet effective strategy to solve the SB instability conundrum is to reduce the imine into an alkylated amine, which cannot be hydrolyzed back into the aldehyde. We propose to reduce 1, using NaBH4 as a hydride ion source because of its relatively low air sensitivity, ease of handling, and its ability to selectively reduce the imines over acetamide groups. NaBH4 is also amenable to reactivity in the solid state,70 enabling the direct reduction of SB without the need for Chs dissolution.We first attempted to perform the SB condensation and reduction in one-pot, which is the conventional way in solution-based methods where Chs was dissolved in an acetic acid solution followed by addition of aldehyde and reducing agents (NaBH3CN or NaBH4) to the solution.66,68 In our solid-state experiments, Chs, one equivalent of furfural, one equivalent of NaBH4 (based on the theoretical imine groups) of NaBH4 and EtOH (50 μL) were subjected to 30 min milling, followed by the same work-up procedure (Scheme S1†). The resulting experimental sample was analyzed with 1H NMR and revealed that only unreacted Chs and furfuryl alcohol were present (Fig. S3†). The reduction of furfural took place too fast to allow for SB condensation to occur.
We then explored the telescoped version of this process. Chs and furfural were first milled for 30 min with LAG water, before NaBH4 (1 equivalent) and LAG EtOH (50 μL) were added,69 and milled for 30, 60, or 90 min, as described in Scheme 2. After set milling time, the reaction mixture underwent the previously mentioned work-up and drying procedures, and characterization by FT-IR, 13C ssNMR and 1H NMR.
After 30 min of milling during the reduction step, the FT-IR spectrum featured peaks characteristic of the furfural unit, revealing coupling occurred, yet the presence of a C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N stretching at 1645 cm−1, characteristic of an imine bond, showed that the reaction was not complete (Fig. S1†). The 13C ssNMR analysis also showed supporting evidence of coupling on Chs as the furan characteristic peaks were retained. However, the presence of the imine carbon peak of 1 indicated the NaBH4 treatment only led to a partial reduction of 1 (Fig. S2†). The sample was dissolved in 0.1% HCl D2O solvent, affording a homogeneous pale-yellow solution for 1H NMR characterization (see Fig. 2). In the spectrum, three broad peaks in the aromatic region at 7.64 (b2), 6.68 (d2), and 6.53 ppm (c2) were ascribed to the formation of furfuryl-Chs (2) with a DS of 39.38% (see ESI† for calculation). Free furfural was also observed in the spectrum, but no furfuryl alcohol. Since we demonstrated in the one-pot experiment that any residual uncoupled furfural should have reacted with NaBH4 during the second step of the reaction, free furfural in this spectrum was ascribed to SB hydrolysis during the NMR analysis. Thus, the telescoped reaction did afford the desired furfuryl-Chs 2, albeit moderate imine reduction % were observed. Another 1H NMR spectrum was taken on the same NMR sample after 12 h and as hypothesized, 2 remained stable, with no noticeable degradation.
N stretching at 1645 cm−1, characteristic of an imine bond, showed that the reaction was not complete (Fig. S1†). The 13C ssNMR analysis also showed supporting evidence of coupling on Chs as the furan characteristic peaks were retained. However, the presence of the imine carbon peak of 1 indicated the NaBH4 treatment only led to a partial reduction of 1 (Fig. S2†). The sample was dissolved in 0.1% HCl D2O solvent, affording a homogeneous pale-yellow solution for 1H NMR characterization (see Fig. 2). In the spectrum, three broad peaks in the aromatic region at 7.64 (b2), 6.68 (d2), and 6.53 ppm (c2) were ascribed to the formation of furfuryl-Chs (2) with a DS of 39.38% (see ESI† for calculation). Free furfural was also observed in the spectrum, but no furfuryl alcohol. Since we demonstrated in the one-pot experiment that any residual uncoupled furfural should have reacted with NaBH4 during the second step of the reaction, free furfural in this spectrum was ascribed to SB hydrolysis during the NMR analysis. Thus, the telescoped reaction did afford the desired furfuryl-Chs 2, albeit moderate imine reduction % were observed. Another 1H NMR spectrum was taken on the same NMR sample after 12 h and as hypothesized, 2 remained stable, with no noticeable degradation.
To increase imine reduction efficiency from 39.38% for 2, the milling time in the mechanochemical 1 reduction with NaBH4 was increased from 30 min to 60 and 90 min, while the milling time in the first step of 1 formation was unchanged as previously experiment already suggested satisfactory % conversion. The total % conversion of furfural increased from 90.73% for 30 min to 93.05% and 98.20% respectively, for 60 and 90 min (see Fig. S4.2†), only showing a marginal improvement. On the other hand, the DS with the furfuryl functionality increased from 39.38% with 30 min milling to 45.10% and 60.34% with 60- and 90-min milling (see Fig. S4.2†), respectively. Although there was a rising trend in the efficiency of the imine reduction, the prolonged milling up to 90 min caused a significant rise in instrument temperature, from room temperature (25.4 °C) to 40.6 °C after 90 min and 50.5 °C at 180 min, as seen in our previous work,39 which we thought would make the rationalization of mechanochemical vs. thermal effects difficult to disentangle beyond 90 min milling. Therefore, we turned to aging to seek improved DS.
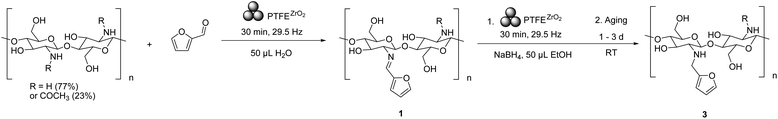
Mechanochemical and aging-based reductive alkylation of chitosan with furfural and NaBH4
We explored aging for 1 to 3 d under laboratory room conditions, on samples after the mechanochemical reductive alkylation with NaBH4 for 30 min, as described in Scheme 3. After set aging time, the reaction mixture underwent the previously mentioned work-up, drying procedures and characterization by 1H NMR in 0.1% HCl D2O (see Fig. S4.3†).While aging did not show a significant impact on the total % conversion of furfural into functionalization on Chs in either the form of imine or amine bonded furan, it had a clear positive effect on the imine reduction, with DS of 39.38% 78.5%, 82.6%, and 99.1% measured on 3 after 0, 1, 2, and 3 d of aging, respectively (Fig. 3). Previously, we observed similar positive effects for aging on polysaccharide transformations.38,39 This mechanochemical and aging-based reductive alkylation of Chs with furfural and NaBH4 showed great simplicity in achieving near complete DS, distinguishing 3 from 2, in which the functionalization resulted in a mixture of SB and reduced amine. The method eliminated the repeated dissolution, refluxing, and pH adjustment steps that were widely employed in previously reported solution-based methods of similar modifications.64–67 The mechanism behind this aging-extended 1 NaBH4 reduction was likely due to a homogeneous mixing and activation introduced by ball milling, followed by a continuance of the reaction during aging under ambient conditions.
The role of LAG solvent in mechanochemical and aging-based 3 reduction with NaBH4
The use of water in the LAG condition was crucial for the mechanochemical synthesis of 1 and consequently 3. Although SB formation is a condensation reaction and prefers anhydrous reaction conditions in the liquid phase,71,72 the presence of catalytic amounts of water was key to introduce reactivity to the system under mechanochemical conditions. This effect was tested by varying LAG conditions during the Chs furfural SB formation step (from starting material to 1, Scheme 3) while the LAG condition (50 μL EtOH) in the second step (formation of 3) was kept constant (see Table 2). The reaction between Chs and furfural, in absence of LAG water solvent, only led to 13.3% DS in 3, which was significantly lower than the one achieved with LAG condition of 99.1% (see Table 1). For further investigation, Chs was pre-dried at 117 °C to remove the 8.03% of water present in Chs as received from supplier, as measured by thermogravimetric analysis (TGA). The pre-dried Chs (DChs) and furfural reaction yielded even lower DS of 9.0%. Experiments were also conducted in which an equivalent of amount of water (20 μL) removed from Chs upon drying was added back to the DChs. This resulted in a comparable 17.0% DS to the experiment with Chs from the vendor bottle. Only when removed water was added back to dried Chs with the addition of the 50 μL LAG water, did a good DS of 90.0% was achieved.| Chs starting material | LAG solvent | Chs %DS |
|---|---|---|
| Chs | Water (50 μL) | 99.1 |
| Chs | none | 13.3 |
| DChs | none | 9.0 |
| DChs | Water (20 μL) | 17.0 |
| DChs | Water (20 + 50 μL) | 90.0 |
| DChs | EtOH (20 μL) | 19.0 |
| DChs | EtOH (20 + 50 μL) | 40.0 |
| Aldehyde (R′′CHO) | Chs %DS | % Isolated yield | |
|---|---|---|---|
| a Determined by 13C ssNMR. | |||
| Furfural | 99.1 | 77.2 | |
| a | 5-(Hydroxymethyl)furfural | 58.6 | 86.5 |
| b | 5-(1,2-Dioxolan-4-yl)furan-2-carbaldehyde | 83.0 | 74.1 |
| c | Isobutyladehyde | 38.4 | 74.5 |
| d | Pentanal | Virtually 100.0 | 80.6 |
| e | Hexanal | Virtually 100.0 | 76.2 |
| f | Heptaldehyde | Virtually 100.0 | 73.6 |
| g | Cyclohexanecarboxaldehyde | 97.2 | 76.1 |
| h | Octanala | 91.1 | 81.6 |
| i | Nonanala | 22.4 | 90.6 |
| j | Undecanala | 12.0 | 97.1 |
| k | Benzaldehyde | 93.2 | 77.2 |
| l | 4-Hydroxybenzaldehyde | 64.5 | 85.6 |
| m | 3,4-Dihydroxybenzaldehyde | 41.2 | 87.5 |
| n | 2-Chlorobenzaldehyde | 47.3 | 92.3 |
| o | 3-Bromobenzaldehyde | 45.7 | 97.8 |
| p | 4-Bromobenzaldehyde | 9.0 | 95.6 |
| q | 4-(Dimethylamino)benzaldehyde | 7.6 | 88.4 |
| r | 2-Pyridinecarboxaldehyde | 66.6 | 99.0 |
| s | 4-Nitrobenzaldehyde | 4.7 | 94.6 |
| t | 2-Hydroxy-1-naphthaldehyde | N/A | 74.2 |
The effect of LAG solvent was also studied by substituting water with EtOH. An equal volume of EtOH was used to replace the removed water from DChs and the experiment yielded 19.0% DS. When an additional 50 μL EtOH was added to the already present EtOH content, a 40.0% DS was measured, showing poorer results than with LAG water. This investigation revealed water as the optimal LAG solvent over EtOH in the mechanochemical synthesis of 1. The enhancement came from improved mobility for furfural molecules to diffuse into the Chs. Further experiments of substituting water with EtOH as the LAG solvent also promoted reactivity to 40.0% DS but was not as effective as water reaching 90.0% DS. The enhanced performance of water over EtOH as LAG solvent could stem from water acting as a Lewis acid, which is required for the protonation of the aldehyde to activate the nucleophilic attack of the primary amine of Chs. Such interaction is the key step in hemiaminal intermediate formation in the SB condensation. In contrast, EtOH is a Lewis base, incapable of protonating the aldehyde (Scheme S2†). As a result, the introduction of EtOH as a LAG solvent could only facilitate 1 formation through the enhancement of diffusion and was found less effective compared to water.
The aldehyde scope of the mechanochemical and aging-based chitosan reductive alkylation
Finally, we explored the versatility of this reported mechanochemical and aging-based Chs reductive alkylation functionalization by performing experiments on Chs with aldehyde compounds (see Scheme 4 and Table 2). The least amount of milling (30 min in each step) was selected to minimize the process's energy consumption and avoid comminution or undesired side reactions caused by increased temperature. A relatively long aging of 3 d was performed to achieve high reaction conversion and to reduce waste from unreacted reagents. | ||
| Scheme 4 The generic scheme of the mechanochemical and aging-based reductive alkylation of Chs into 3, 4a–4t with aldehyde R′′CHO (see Table 2 for scope). Typical experimental conditions: Chs (250 mg), 1 eq. of aldehyde compound a–t (0.99 mmol), and H2O (50 μL) are loaded into a PTFE jar with a 7 mm ZrO2 ball and milled for 30 min at 29.5 Hz. This was followed by the loading of 1 eq. of NaBH4 (32.4 mg) and EtOH (50 μL) into the PTFE jar containing the reaction mixture and milled for 30 min, and the sample mixture was transferred to a sealed vial and aged for 3 d at room temperature. | ||
The versatility and robustness of this mechanochemical and aging-based Chs reductive alkylation were investigated with a series of furfural derivatives, aliphatic aldehydes, and aryl aldehydes. The samples followed the mechanochemical and aging-based chitosan reductive alkylation with the selected scope of aldehydes were characterized in DS and isolated yield (see ESI† for calculation). There was only a small loss of samples in isolation and purification thanks to the omission of dissolution and precipitation in the procedure. In general, the isolated yield of samples, i.e. the percentage of recovered chitosan backbone, falls between 73.6 to 99.0%. Results revealed that the method worked well on furfural derivatives including 5-(hydroxymethyl)furfural (a) and synthesized 5-(1,2-dioxolan-4-yl)furan-2-carbaldehyde (b) without deprotecting the acetal group, with DS of 58.6% and 83.0% respectively. The reductive alkylation reaction between Chs and aliphatic aldehydes performed the best with aliphatic aldehydes consisting of alkyl chains or rings with 4–6 hydrocarbons. The calculated DS for 4d, 4e, and 4f were even slightly over 100% due to the intrinsic inconsistency of the starting Chs, thus the reductive alkylation with these aldehydes were seen as virtually complete. The cyclic structure of cyclohexanecarboxaldehyde (g) did not hinder the reaction as it still induced a high 97.2% DS. With isobutylaldehyde (c), octanal (h), nonanal (i), and undecanal (j), the DS from the reaction was significant but not as high as for other examples, ranging from 12.0 to 91.1%. Remarkably though, the properties of resulting alkylated Chs were significantly affected by this functionalization. The modification with aldehydes which have straight alkyl chains with more than 7 carbons significantly increased hydrophobicity of the materials, and samples 4h, 4i, and 4j were not soluble in the aqueous NMR solvent of 0.1% HCl acidified D2O. Thus, the characterization and DS calculations were carried out by 13C ssNMR, which confirmed the successful reaction, while more destructive techniques and conditions were explored to assist in acquiring 1H NMR spectra of the samples for more quantitativtive analysis(see discussion in ESI†). The long-chain aldehydes have a lower affinity with water, which could inhibit the protonation of the aldehydes by water, leading to lower DS as a result. Another factor of the low DS could be attributed to the poor diffusion of long-chain aldehydes onto Chs to enable the nucleophilic attack of Chs amine to the aldehyde substrates. Another interesting example is 4-(dimethylamino)benzaldehyde (q). Despite a low DS of 7.62%, 4q dissolves in DI water, affording a basic solution at pH 12. It is unusual for chitosan derivatives to be soluble at high pH, and this method complements the mechanochemical N-sulfonation of Chs as reported by Van Poucke et al.40 The completely distinct solvation properties of 4h, 4i, 4j to 4q suggested the reductive alkylation approach offers a powerful means for manipulating the properties of Chs such as solvation properties. Chs could be tailored to specific properties with only relatively conservative levels of DS
Finally, the reductive alkylation of Chs with aryl aldehydes was largely affected by the substituents. Benzaldehyde (k) afforded an excellent DS of 93%, as well as 2-pyridinecarboxaldehyde (r) with 66.6% DS. Electron donating groups para-substituted to the benzaldehyde (such as 4-hydroxybenzaldehyde, l) or electron withdrawing groups meta-substituted to the benzaldehyde (3-bromobenzaldehyde, o) seemed to favor good results with DS in the 45 to 65% range. Ortho substitution and disubstitution gave an average result with 3,4-dihydroxybenzaldehyde (m) and 2-chlorobenzaldehyde (n) affording results in the upper 40% range. With electron withdrawing groups para-substituted to benzaldehydes such as 4-bromobenzaldehyde (p) and 4-nitrobenzaldehyde (s), reaction efficiency was reduced to single digit DS, due to the reduced electron density on aldehyde oxygen inhibited its protonation. The para-substituted 4-(dimethylamino)benzaldehyde (q) did not provide a much better DS with 7.6%, although as mentioned above, the solvation property changes are quite drastic. 2-Hydroxy-1-naphthaldehyde (t) did not show any conversion, possibly because of the bulkiness of the naphthyl group.
Advantages of the method
We compared our novel mechanochemical and aging-based Chs reductive alkylation to the conventional solution-based methods,68,73,74 and to the solventless mechanochemical method for Chs SB recently reported by Fatika et al.56 In the perspective of the chemical bond formed in the current study and previously reported methods, our method allowed for more robust Chs functionalization compared to the mechanochemically synthesized Chs SB, affording alkylated Chs derivatives free from imine hydrolysis risk. When comparing our method to other Chs reductive alkylation systems, the present method does not require Chs dissolution in acidic aqueous media, providing a number of advantages: (1) this allows the functionalization on Chs to occur with acid-sensitive compounds as demonstrated with 5-(1,2-dioxolan-4-yl)furan-2-carbaldehyde (b) and (2) SB formation equilibrium is not favored under acidic conditions, meaning that when the reducing agent is added, only a fraction of the aldehyde is engaged in an imine bond, explaining the low DS reported before. A good example of this is provided with octanal (h), for which the mechanochemical and aging-based Chs reductive alkylation process achieved high DS at 49.3% compared to only 0.03% in the solution-based method reported by Bobu et al.68 In the work of Rabea et al. N-(butyl)chitosan (4d) was prepared by a similar solution-based approach.67 With 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mol ratio of pentanal (d) to Chs primary amine, they only achieved a DS of 36% compared to virtually 100% by our proposed method.67 Thus, the current method can perform similar chemical reactions with higher atom economy, reflected by the higher DS reported. The elimination of solvent in the reaction and simplification of the workup procedure also led to an impressively reduced calculated PMI of 36 (with furfural, 3) compared to 1300 calculated based on available information by Bobu's study and 602 from Rabea's study.67,68
1 mol ratio of pentanal (d) to Chs primary amine, they only achieved a DS of 36% compared to virtually 100% by our proposed method.67 Thus, the current method can perform similar chemical reactions with higher atom economy, reflected by the higher DS reported. The elimination of solvent in the reaction and simplification of the workup procedure also led to an impressively reduced calculated PMI of 36 (with furfural, 3) compared to 1300 calculated based on available information by Bobu's study and 602 from Rabea's study.67,68
Conclusion
In this study, we present a method to functionalize Chs by alkylation in the solid state via reductive amination by mechanochemical ball-milling combined with aging under ambient conditions. With water LAG conditions, Chs can form a SB with series of aldehyde compounds, followed by mechanochemical reduction by NaBH4 under EtOH LAG conditions to yield alkyl-Chs substituted on the amine Chs groups. The method enabled efficient reductive alkylation on Chs with furfural at 99.1% DS, close to total functionalization of the available Chs amine groups. A scope investigation proved the robustness and versatility of the presented method compatible with a wide range of both aliphatic and aromatic aldehydes. The mechanochemical approach eliminated the dissolution of Chs and allowed for the reaction to occur with aldehyde reagents that are unstable in solution. This method is simpler with reduced reaction substrates and steps leading to an impressively low PMI of 36. It affords Chs with new solvation properties. In conclusion, the mechanochemistry and aging-based reductive amination/alkylation of Chs provides a new avenue to design and effectively functionalize Chs into value-added material for applications such as bio-sensing, medication, food, cosmetic, and fibre making.Experimental
Materials
Chitosan (from shrimp shell, practical grade), furfural (99%), 5-(hydroxymethyl)furfural (≥99%, FG), benzaldehyde (ReagentPlus®, ≥99%), 4-hydroxybenzaldehyde (98%), 3,4-dihydroxybenzaldehyde (97%), 2-chlorobenzaldehyde (99%), 3-bromobenzaldehyde (97%), 4-bromobenzaldehyde (ReagentPlus®, 99%), 4-(dimethylamino)benzaldehyde (ACS reagent, 99%), 2-pyridinecarboxaldehyde (99%), 4-nitrobenzaldehyde (98%, GC), 2-hydroxy-1-naphthaldehyde (technical grade), isobutyraldehyde (≥99%), hexanal (98%), cyclohexanecarboxaldehyde (97%), heptaldehyde (95%), octanal (99%), nonyl aldehyde (95%), undecyl aldehyde (97%), sodium borohydride (99%) and deuterium oxide (99.9% D) were purchased from Sigma-Aldrich Co. L.L.C. (St. Louis, MO, USA). Pentanal (97%) and hydrochloric acid were purchased from Fischer Scientific L.L.C. (Ottawa, ON, Canada). EtOH for work-up and washing was purchased from Commercial Alcohols by Greenfield Global (Brampton, ON, Canada). For solution-state NMR characterization, 0.1% HCl acidified D2O was used to dissolve modified chitosan samples.Milling
In the following procedures, a Retsch MM 400 mixer mill was used, with polytetrafluoroethylene (PTFE) SmartSnap Jars (15 mL) manufactured by Form-Tech Scientific (Canada), equipped with 7 mm balls made of zirconia.Mechanochemical-based chitosan SB experiments
In a typical experiment, 250 mg of Chs with 1 equivalent of aldehyde to the glucosamine units were combined in the PTFE grinding jars and milled under a LAG condition with the addition of 50 μL of DI water for 30 min with one 7 mm zirconia ball.Mechanochemical and aging-based chitosan SB reduction experiments
In a typical experiment, the reaction mixture of Chs SB was combined with 1 equivalent of NaBH4 in the same PTFE grinding jars and milled under a new LAG condition with the addition of 50 μL of EtOH for 30 min with the same 7 mm zirconia ball. After milling, the reaction mixture was transferred into a sealed vial and aged for 3 d at RT. Following milling and aging, the reaction mixture was dispersed and washed in EtOH (40 mL), filtered, and washed repeatedly with DI water (80 mL) to remove all excess reagents, reaction by-products, and EtOH from the Chs samples during the first washing step. Finally, samples were collected and dried overnight in a vacuum oven at 50 °C for storage and characterization. The final alkylated Chs samples are fine powders with colours determined by the aldehyde used in the experiments. The same work-up procedure can also be applied after the mechanochemical-based Chs SB experiments to yield chitosan imine intermedia for reaction kinetic studies.Analysis – characterization – equipment details and methods
Abbreviations
| Chs | Chitosan |
| EtOH | Ethanol |
| SB | Schiff base |
| NaBH4 | Sodium borohydride |
| PMI | Process mass intensity |
| LAG | Liquid-assisted grinding |
| PTFE | Polytetrafluoroethylene |
| DI | Deionized |
| FT-IR | Fourier transform infra-red |
| ssNMR | Magic angle spinning nuclear magnetic resonance |
| DS | Degree of substitution |
| TGA | Thermogravimetric analysis |
Author contributions
The manuscript was written through the contributions of all authors. All authors have given approval to the final version of the manuscript.Conflicts of interest
There is no conflict to declare.Acknowledgements
We thank the Natural Science and Engineering Research Council of Canada (NSERC) – Discovery Grant and Discovery Accelerator Supplement, the Canada Foundation for Innovation (CFI), the Fonds de Recherche du Québec – Nature et Technologies (FRQNT) – the Centre for Green Chemistry and Catalysis (CGCC), the National Research Council (NRC) of Canada – Ocean program and McGill University including the Heather Munroe-Blum Fellowships in Green Chemistry for their financial support.References
- R. N. Tharanathan and F. S. Kittur, Crit. Rev. Food Sci. Nutr., 2003, 43, 61–87 CrossRef CAS PubMed.
- G. A. Martău, M. Mihai and D. C. Vodnar, Polymers, 2019, 11, 1837 CrossRef PubMed.
- M. C. Biswas, B. Jony, P. K. Nandy, R. A. Chowdhury, S. Halder, D. Kumar, S. Ramakrishna, M. Hassan, M. A. Ahsan, M. E. Hoque and M. A. Imam, J. Polym. Environ., 2022, 30, 51–74 CrossRef CAS.
- F. M. Kerton, Y. Liu, K. W. Omari and K. Hawboldt, Green Chem., 2013, 15, 860–871 RSC.
- R. C. Goy, D. D. Britto and O. B. Assis, Polimeros, 2009, 19, 241–247 CrossRef CAS.
- L.-Y. Zheng and J.-F. Zhu, Carbohydr. Polym., 2003, 54, 527–530 CrossRef CAS.
- V. K. Mourya and N. N. Inamdar, React. Funct. Polym., 2008, 68, 1013–1051 CrossRef CAS.
- I. F. Amaral, P. L. Granja and M. A. Barbosa, J. Biomater. Sci., Polym. Ed., 2005, 16, 1575–1593 CrossRef CAS PubMed.
- N. M. Alves and J. F. Mano, Int. J. Biol. Macromol., 2008, 43, 401–414 CrossRef CAS PubMed.
- C. A. G. N. Montalbetti and V. Falque, Tetrahedron, 2005, 61, 10827–10852 CrossRef CAS.
- S. Mondal and S. Malakar, Tetrahedron, 2020, 76, 131662 CrossRef CAS.
- E. A. Werner, J. Chem. Soc., Trans., 1918, 113, 899–902 RSC.
- M. A. Sprung, Chem. Rev., 1940, 26, 297–338 CrossRef CAS.
- O. I. Afanasyev, E. Kuchuk, D. L. Usanov and D. Chusov, Chem. Rev., 2019, 119, 11857–11911 CrossRef CAS PubMed.
- C. K. S. Pillai, W. Paul and C. P. Sharma, Prog. Polym. Sci., 2009, 34, 641–678 CrossRef CAS.
- V. Zargar, M. Asghari and A. Dashti, ChemBioEng Rev., 2015, 2, 204–226 CrossRef.
- A. V. Il'ina and V. P. Varlamov, Appl. Biochem. Microbiol., 2004, 40, 300–303 CrossRef.
- N. D. Aljbour, M. D. H. Beg and J. Gimbun, Chem. Eng. Technol., 2019, 42, 1741–1746 CrossRef CAS.
- D. P. Chattopadhyay and M. S. Inamdar, Int. J. Polym. Sci., 2010, 2010, 939536 Search PubMed.
- J. K. Hwang and H. H. Shin, Korea-Aust. Rheol. J., 2000, 12, 175–179 Search PubMed.
- X. Meng and K. J. Edgar, Prog. Polym. Sci., 2016, 53, 52–85 CrossRef CAS.
- K. Kurita, Prog. Polym. Sci., 2001, 26, 1921–1971 CrossRef CAS.
- G. M. Gübitz and A. C. Paulo, Curr. Opin. Biotechnol., 2003, 14, 577–582 CrossRef PubMed.
- S. Bhatia, in Natural Polymer Drug Delivery Systems: Nanoparticles, Plants, and Algae, Springer International Publishing, Cham, 2016, pp. 95–118, DOI:10.1007/978-3-319-41129-3_3.
- P. Ramesh and S. Vinodh, Int. J. Sustainable Eng., 2020, 13, 411–422 CrossRef.
- L. Shen and M. K. Patel, J. Polym. Environ., 2008, 16, 154–167 CrossRef CAS.
- S. L. James and T. Friščić, Chem. Soc. Rev., 2013, 42, 7494–7496 RSC.
- L. Takacs, Chem. Soc. Rev., 2013, 42, 7649–7659 RSC.
- J.-L. Do and T. Friščić, ACS Cent. Sci., 2017, 3, 13–19 CrossRef CAS PubMed.
- K. J. Ardila-Fierro and J. G. Hernández, ChemSusChem, 2021, 14, 2145–2162 CrossRef CAS PubMed.
- S. Lamour, S. Pallmann, M. Haas and O. Trapp, Life, 2019, 9, 52 CrossRef CAS PubMed.
- S. L. James, C. J. Adams, C. Bolm, D. Braga, P. Collier, T. Friščić, F. Grepioni, K. D. Harris, G. Hyett and W. Jones, Chem. Soc. Rev., 2012, 41, 413–447 RSC.
- M. J. Cliffe, C. Mottillo, R. S. Stein, D.-K. Bučar and T. Friščić, Chem. Sci., 2012, 3, 2495–2500 RSC.
- F. Hajiali, T. Jin, G. Yang, M. Santos, E. Lam and A. Moores, ChemSusChem, 2022, 15, e202102535 CrossRef CAS PubMed.
- F. Hajiali, J. Vidal, T. Jin, L. C. de la Garza, M. Santos, G. Yang and A. Moores, ACS Sustainable Chem. Eng., 2022, 10, 11348–11357 CrossRef CAS.
- G. Margoutidis and F. M. Kerton, in Biomass Valorization, 2021, pp. 343–365, DOI:10.1002/9783527825028.ch12.
- T. Jin, T. Liu, F. Hajiali, M. Santos, Y. Liu, D. Kurdyla, S. Régnier, S. Hrapovic, E. Lam and A. Moores, Angew. Chem., Int. Ed., 2022, 61, e202207206 CrossRef CAS PubMed.
- T. Di Nardo, C. Hadad, A. Nguyen Van Nhien and A. Moores, Green Chem., 2019, 21, 3276–3285 RSC.
- G. Yang, E. Lam and A. Moores, ACS Sustainable Chem. Eng., 2023, 11, 7765–7774 CrossRef CAS.
- C. Van Poucke, A. Vandeputte, S. Mangelinckx and C. V. Stevens, Green Chem., 2023, 25, 4271–4281 RSC.
- R. Antony, T. Arun and S. T. D. Manickam, Int. J. Biol. Macromol., 2019, 129, 615–633 CrossRef CAS PubMed.
- N. Nigam, S. Kumar, P. Dutta, S. Pei and T. Ghosh, RSC Adv., 2016, 6, 5575–5581 RSC.
- F. Chen, Z. Shi, K. Neoh and E. Kang, Biotechnol. Bioeng., 2009, 104, 30–39 CrossRef CAS PubMed.
- Y. Qin, J. Chen, Y. Bi, X. Xu, H. Zhou, J. Gao, Y. Hu, Y. Zhao and Z. Chai, Acta Biomater., 2015, 17, 201–209 CrossRef CAS PubMed.
- Z. Wei, D. M. Lewis, Y. Xu and S. Gerecht, Adv. Healthcare Mater., 2017, 6, 1700523 CrossRef PubMed.
- B. C. E. Makhubela, A. Jardine and G. S. Smith, Appl. Catal., A, 2011, 393, 231–241 CrossRef CAS.
- R. Antony, S. T. David, K. Karuppasamy, G. Sanjeev and S. Balakumar, Spectrochim. Acta, Part A, 2014, 124, 178–186 CrossRef CAS PubMed.
- S. W. Gong, H. F. He, C. Q. Zhao, L. J. Liu and Q. X. Cui, Synth. Commun., 2012, 42, 574–581 CrossRef CAS.
- D. H. K. Reddy and S.-M. Lee, J. Appl. Polym. Sci., 2013, 130, 4542–4550 CrossRef CAS.
- H. Xiao, W. Lu, X. Le, C. Ma, Z. Li, J. Zheng, J. Zhang, Y. Huang and T. Chen, Chem. Commun., 2016, 52, 13292–13295 RSC.
- S. Prasad, T. Gomathi and P. Sudha, Int. J. Novel Trends Pharm. Sci., 2017, 7, 57–63 Search PubMed.
- E. L. de Araújo, H. F. G. Barbosa, E. R. Dockal and É. T. G. Cavalheiro, Int. J. Biol. Macromol., 2017, 95, 168–176 CrossRef PubMed.
- H. R. Mardani, F. Ravari, A. Kalaki and L. Hokmabadi, J. Polym. Environ., 2020, 28, 2523–2538 CrossRef CAS.
- N. Krrushcheva, N. Loim and V. Sokolov, Russ. Chem. Bull., 1997, 46, 1952–1955 CrossRef.
- D. E. Crawford, C. K. G. Miskimmin, A. B. Albadarin, G. Walker and S. L. James, Green Chem., 2017, 19, 1507–1518 RSC.
- F. A. W. Fatika, M. Anwar, D. J. Prasetyo, W. A. Rizal, R. Suryani, P. Yuliyanto, S. Hariyadi, A. Suwanto, N. A. Bahmid, S. K. Wahono, F. H. Sriherfyna, C. D. Poeloengasih, B. Purwono, E. Agustian, R. Maryana and H. Hernawan, Int. J. Biol. Macromol., 2023, 247, 125759 CrossRef CAS PubMed.
- E. H. Cordes and W. P. Jencks, J. Am. Chem. Soc., 1962, 84, 832–837 CrossRef CAS.
- E. H. Cordes and W. P. Jencks, J. Am. Chem. Soc., 1963, 85, 2843–2848 CrossRef CAS.
- P. S. Reddy, S. Kanjilal, S. Sunitha and R. B. N. Prasad, Tetrahedron Lett., 2007, 48, 8807–8810 CrossRef CAS.
- G. Borg, D. A. Cogan and J. A. Ellman, Tetrahedron Lett., 1999, 40, 6709–6712 CrossRef CAS.
- A. Heydari, A. Arefi and M. Esfandyari, J. Mol. Catal. A: Chem., 2007, 274, 169–172 CrossRef CAS.
- K. N. Gusak, Z. V. Ignatovich and E. V. Koroleva, Russ. Chem. Rev., 2015, 84, 288 CrossRef CAS.
- M. A. Ayedi, Y. Le Bigot, H. Ammar, S. Abid, R. E. Gharbi and M. Delmas, Synth. Commun., 2013, 43, 2127–2133 CrossRef CAS.
- U. Janciauskaite, V. Rakutyte, J. Miskinis and R. Makuska, React. Funct. Polym., 2008, 68, 787–796 CrossRef CAS.
- H. E. Abdelwahab, S. Y. Hassan, G. A. Yacout, M. A. Mostafa and M. M. El Sadek, Polymers, 2015, 7, 2690–2700 CrossRef CAS.
- W. Yao, Y. Jiao, J. Luo, M. Du and L. Zong, Int. J. Biol. Macromol., 2012, 50, 821–825 CrossRef CAS PubMed.
- E. I. Rabea, M. E. Badawy, T. M. Rogge, C. V. Stevens, M. Höfte, W. Steurbaut and G. Smagghe, Pest Manage. Sci., 2005, 61, 951–960 CrossRef CAS PubMed.
- E. Bobu, R. Nicu, M. Lupei, F. Ciolacu and J. Desbrieres, Cellul. Chem. Technol., 2011, 45, 619 CAS.
- T. Friščić, S. L. Childs, S. A. A. Rizvi and W. Jones, CrystEngComm, 2009, 11, 418–426 RSC.
- J. Mack, D. Fulmer, S. Stofel and N. Santos, Green Chem., 2007, 9, 1041–1043 RSC.
- S. Perrone, A. Salomone, A. Caroli, A. Falcicchio, C. Citti, G. Cannazza and L. Troisi, Eur. J. Org. Chem., 2014, 5932–5938 CrossRef CAS.
- S. D. Montag, J. Chem. Educ., 2023, 100, 4456–4461 CrossRef CAS.
- Z.-W. Jing, Z.-W. Ma, C. Li, Y.-Y. Jia, M. Luo, X.-X. Ma, S.-Y. Zhou and B.-L. Zhang, Bioorg. Med. Chem. Lett., 2017, 27, 1003–1006 CrossRef CAS PubMed.
- H. C. B. Paula, R. B. C. Silva, C. M. Santos, F. D. S. Dantas, R. C. M. de Paula, L. R. M. de Lima, E. F. de Oliveira, E. A. T. Figueiredo and F. G. B. Dias, Int. J. Biol. Macromol., 2020, 163, 1591–1598 CrossRef CAS PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available: Additional methods information, and original spectroscopic data. See DOI: https://doi.org/10.1039/d4gc00127c |
| ‡ Present addresses: Centre in Green Chemistry and Catalysis, Department of Chemistry, McGill University, Montréal, Québec H3A 0B8, Canada |
| This journal is © The Royal Society of Chemistry 2024 |