Catalytic self-transfer hydrogenolysis of lignin over Ni/C catalysts†
Xuelei
Mei
ab,
Huizhen
Liu
 *cd,
Haihong
Wu
*cd,
Haihong
Wu
 *ab,
Wei
Wu
ab,
Bingxiao
Zheng
ab,
Yani
Liu
ab,
Xinrui
Zheng
ab,
Yaqin
Wang
ab,
Wanying
Han
ab and
Buxing
Han
*ab,
Wei
Wu
ab,
Bingxiao
Zheng
ab,
Yani
Liu
ab,
Xinrui
Zheng
ab,
Yaqin
Wang
ab,
Wanying
Han
ab and
Buxing
Han
 *abcd
*abcd
aShanghai Key Laboratory of Green Chemistry and Chemical Processes, State Key Laboratory of Petroleum Molecular & Process Engineering, School of Chemistry and Molecular Engineering, East China Normal University, Shanghai 200062, China. E-mail: liuhz@iccas.ac.cn; hhwu@chem.ecnu.edu.cn; hanbx@iccas.ac.cn
bInstitute of Eco-Chongming, Shanghai 202162, China
cSchool of Chemistry and Chemical Engineering, University of Chinese Academy of Sciences, Beijing 100049, China
dBeijing National Laboratory for Molecular Sciences, CAS Key Laboratory of Colloid and Interface and Thermodynamics, CAS Research/Education Center for Excellence in Molecular Sciences, Institute of Chemistry, Chinese Academy of Sciences, Beijing 100190, China
First published on 20th February 2024
Abstract
Lignin is composed of phenylpropyl alcohol through the C–O and C–C bonds, where β-O-4 accounts for the majority. Self-transfer hydrogenolysis (STH) is a promising method to produce valuable chemicals and fuels from lignin by cleaving the β-O-4 bond without exogenous hydrogen, but all the reported work used noble metal-based catalysts. In this work, a highly efficient Ni/C catalyst was derived from a Ni-containing metal–organic framework (Ni-MOF), and its self-transfer hydrogenolysis performance towards ether bonds in lignin model compounds was evaluated using 2-phenoxy-1-phenylethanol as a model compound in detail. It was found that the catalyst pyrolyzed under a nitrogen atmosphere at 500 °C (Ni-NDC-500) was very efficient for the reaction. Moreover, it could also catalyze the reaction of native lignin into monomers effectively without exogenous hydrogen. In addition, Ni-NDC-500 was recycled three times without an obvious reduction of the activity.
1. Introduction
Due to the depletion of fossil resources, utilization of biomass as a sustainable, carbon-neutral supply of fuels and chemicals is an immediate need.1–8 Lignocellulose is an important biomass, of which lignin is interesting due to its high aromatic content.9 Lignin is a complex polymer connected by the C–O and C–C bonds.10 The β-O-4 bonds, which occupy more than half of the linkage structure in lignin, are a main object of study. The cleavage of β-O-4 ether bonds of lignin compounds using hydrogen is usually referred to as hydrogenolysis.11The liquid-phase hydrogenolysis reaction typically requires high H2 pressures (1–20 MPa) to ensure better dissolution of H2 into the reaction medium.12 However, hydrogen cannot be naturally produced and is usually industrially produced by steam-reforming fossil fuels like coal, natural gas, and oil, leading to an increased carbon footprint during production and transportation. In addition to hydrogen gas, hydrogen donors are also utilized for the depolymerization and hydrogenolysis of lignin.13 Lignin possesses abundant aliphatic hydroxyl groups capable of undergoing dehydrogenation (Cα–OH to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) to obtain hydrogen. Studies have shown that lignin can depolymerize into monomers without requiring additional hydrogen sources, utilizing the hydroxyl groups within lignin itself as the hydrogen donor. This approach is termed the self-hydrogen transfer hydrogenolysis (STH) process.14 For the STH of lignin model compounds, Cai and coworkers used MIL-100(Fe) supported Pd–Ni bimetal nanoparticles.15 The STH of native lignin to monomers over a Pd–PdO/TiO2 catalyst was reported by Wang et al.16
O) to obtain hydrogen. Studies have shown that lignin can depolymerize into monomers without requiring additional hydrogen sources, utilizing the hydroxyl groups within lignin itself as the hydrogen donor. This approach is termed the self-hydrogen transfer hydrogenolysis (STH) process.14 For the STH of lignin model compounds, Cai and coworkers used MIL-100(Fe) supported Pd–Ni bimetal nanoparticles.15 The STH of native lignin to monomers over a Pd–PdO/TiO2 catalyst was reported by Wang et al.16
In addition, the –OCH3 group in lignin can provide a hydrogen source for the STH process through dehydrogenation or aqueous phase reforming. Wang's group used NiAl2O4 loading with various noble metals to produce alkylphenol from lignin by self-reforming-driven depolymerization and hydrogenolysis, and found that demethoxylation is the rate-determining step in the whole process.17–19 Rong and Li reported the highly selective conversion of guaiacol to phenol in pure water by a versatile nano-porous Ni catalyst, and the mechanism showed that the initial hydrogen source came from water splitting on the surface of the Ni catalyst, and the subsequent aqueous phase reforming of methanol generated more hydrogen and further accelerated the hydrodeoxygenation process.20 Our group proposed self-supported hydrogenolysis (SSH) of aromatic ethers to produce arenes using the methoxy group as the hydrogen source over RuW/SiO2.21 The mechanism study showed that the aliphatic C–H bond in anisole was firstly activated and followed by the selective hydrogenolysis of Caryl–OMe to generate benzene. And the RuW/HY30 catalyst could deconstruct the Csp2–Csp3 and Csp2–O bonds in the lignin structure in an orderly manner, allowing the production of benzene exclusively from lignin.22 These articles used water as the reaction medium, which is a cheap, non-toxic and environmentally friendly reaction medium.15–22
The reported works on the STH process are based on precious metal catalysts. However, the resources of precious metals are very limited, and mining and purification release large amounts of CO2 compared with non-precious metals.23 Therefore, it is highly promising to replace noble metals with abundant and inexpensive transition metals for the self-transfer hydrogenolysis and upgradation of lignin model compounds. Many research studies show that Ni-based catalysts could efficiently and selectively cleave the β-O-4 ether bonds of lignin.24–26 MOF-derived catalysts prepared by controlled pyrolysis provide a route to synthesize catalysts containing high surface area and highly dispersed metal nanoparticles, which might facilitate the hydrogenolysis of lignin.27 There have been many reports on MOF-derived catalysts for lignin hydrogenolysis, but the reaction was carried out under a hydrogen atmosphere.28–30
Herein, we report a simple method to obtain a Ni/C catalyst derived from Ni-MOF with high catalytic activity in the STH of lignin. The active sites and reaction mechanism of the catalysts were investigated in depth. This is the first work on inexpensive transition metals for the self-transfer hydrogenolysis process.
2. Experiment section
2.1 Catalyst preparation
Ni-MOF was prepared by the co-precipitation method reported in the literature.31 Fig. S1† is a schematic of the preparation procedures of the Ni/C catalyst. Ni(NO3)2·6H2O (4.06 g) and 1,4-naphthalenedicarboxylic acid (NDC, 2.94 g) were dissolved in N,N-dimethylformamide (DMF) (48 mL), ethanol (6 mL) and H2O (6 mL). The mixture was mixed well using an ultrasonic process for 30 minutes, and then 12 mL of triethylamine was added to the mixture. After continuous ultrasound for four hours at room temperature, the precursors were collected by centrifugation and washed three times with ethanol and distilled water. The product was then dried overnight in a blast dryer at 120 °C. Ni-MOF (Ni-NDC) was obtained. Ni-NDC was then calcined in a tube furnace under nitrogen at 400, 500, 600 and 700 °C. The obtained catalysts are respectively denoted as Ni-NDC-400, Ni-NDC-500, Ni-NDC-600 and Ni-NDC-700.2.2 Catalyst characterization
The phase and unit cell structures of synthesised catalysts were analyzed by powder XRD technique. XRD analysis was carried out on an XRD-Ultima IV diffractometer manufactured by Rigaku Corporation, Japan. The obtained XRD pattern was compared with a standard card to obtain the phase composition of the catalyst. X-ray photoelectron spectroscopy (XPS) was mainly used to analyze the surface element composition, valence charge and species of the catalyst. The K-Alpha type XPS produced by Thermo Fisher Company of the United States used an Al anode (energy of 1486.6 eV) as the excitation source and C 1s (284.6 eV binding energy) as the standard to calibrate the gas element. The morphology and particle size distributions of the catalysts were studied using HRTEM (FEI Tecnai F30). The N2 adsorption–desorption method was used to analyze the specific surface areas of catalysts on a TriStar II Model 3020 adsorption desorber. The relevant specific surface area of the catalysts in this study was also obtained by the BET method on the instrument. Inductively coupled plasma atomic emission spectrometry (ICP-AES, Agilent Technologies, 5100) was adopted to measure the Ni content. The NMR spectra were acquired on a Bruker Avance II300 400 MHz spectrometer. The lignin sample was dissolved in deuterated dimethyl sulfoxide (DMSO-d6). The central solvent peak at δC/δH 39.5/2.49 was used as an internal reference. Two-dimensional HSQC NMR experiments were used for structural characterization and assignment authentication. HSQC cross-peaks were assigned by combining the results and comparing them with the literature.2.3 Catalyst activity tests
The catalytic reactions were conducted in a Teflon-lined stainless-steel autoclave (15 mL) with the desired dosages of the substrate, catalyst, internal standard (n-dodecane or n-hexadecane), and solvent. In a typical experiment, 64.2 mg (0.3 mmol) of 2-phenoxy-1-phenylethanol, 40 mg of Ni-NDC-500 catalyst, and 5 mL of distilled water were added to the reactor. The reactor was purged with N2 three times to remove the air at room temperature. Then N2 was charged to the desired pressure and the reactor was placed in a furnace at the desired temperature under an agitation speed of 800 rpm. After the reaction, the reactor was cooled down to room temperature and the gas was released. The liquid phase was carefully extracted with 2 mL of ethyl acetate. The qualitative analysis of reaction products was carried out on a GC-MS system (Agilent 7890A—5975C), and the quantitative analysis was executed on a GC system (Agilent 8890B) equipped with an HP-5 column and an FID detector.3. Results and discussion
3.1 Characterization of catalysts
The crystallinity and crystalline phases of the prepared catalysts were investigated using X-ray diffraction. As shown in Fig. 1a, Ni-NDC exhibited a distinct characteristic peak at 7.9°, suggesting the successful synthesis of Ni-MOF.31 The XRD pattern of Ni-NDC-400 revealed a mixed phase of NiO and Ni, suggesting incomplete reduction by carbon at a calcination temperature of 400 °C. The XRD patterns of Ni-NDC-500, Ni-NDC-600, and Ni-NDC-700 catalysts exhibited good agreement with the standard face-centred cubic structure of Ni0 (JCPDS No. 70-0989), with diffraction peaks at 44.5°, 51.8°, and 76.3° corresponding to (111), (200), and (220) crystal planes, respectively. The findings suggested that the majority of Ni2+ ions in the Ni-NDC-500, Ni-NDC-600, and Ni-NDC-700 catalysts were reduced to Ni0. Additionally, as the calcination temperature increased, the intensity of XRD peaks also increased, indicating larger crystalline grains and improved crystallinity.XPS (X-ray photoelectron spectroscopy) was employed to examine the valence state of surface elements in the catalysts. Fig. 1b shows the Ni 2p XPS spectra of the calcined catalysts. The peaks observed at 870.1 eV and 852.6 eV correspond to Ni0, while the peaks at 871.7 eV and 854.1 eV correspond to Ni2+. Notably, Ni-NDC-500 exhibited the highest content of surface Ni0.
The N2 adsorption–desorption isotherms were used to determine the specific surface area and pore structure of the Ni-NDC catalysts. The results are presented in Fig. S2.† Ni-NDC exhibited a type II isotherm in the Brunauer classification and an obviously H3 hysteresis loop, which confirms its microporous characteristics. Notably, Ni-NDC-400 and Ni-NDC-500 also displayed a type II isotherm and H3 hysteresis loop, indicating the presence of a microporous structure. However, Ni-NDC-600 and Ni-NDC-700 exhibited an H4 hysteresis loop, indicating the presence of a mesoporous structure in the catalyst. The surface areas of the catalysts were calculated using the multi-point Brunauer–Emmett–Teller (BET) method, and the values are listed in Fig. S2.† The BET surface areas of Ni-NDC-400, Ni-NDC-500, Ni-NDC-600, and Ni-NDC-700 were 32.96, 122.19, 144.56, and 157.31 m2 g−1, respectively. The significant specific surface area facilitated the substrate mass transfer and exposed catalytically active sites.
Table 1 presents the ICP results, and the Ni contents in Ni-NDC, Ni-NDC-400, Ni-NDC-500, Ni-NDC-600, and Ni-NDC-700 were 21.3%, 40.8%, 54.5%, 57.9%, and 60.5%, respectively. As the calcination temperature increased, the bulk Ni content also increased, indicating that a higher temperature resulted in carbon loss. Additionally, the lower surface Ni content observed through XPS suggests that a significant portion of Ni is encapsulated by carbon.
| Entry | Catalysts | Conv. (%) | Yield (%) | Bulk | Surface | ||||
|---|---|---|---|---|---|---|---|---|---|
| 1b | 1c | 1d | 1e | 1f | Nia % | Nib % | |||
| Reaction conditions: 40 mg catalysts, 0.3 mmol of 2-phenoxy-1-phenylethanol, 5 mL of distilled water, 220 °C, 2 h, initial pressure 2 MPa nitrogen, and stirring at 800 rpm. Yields and conversions were determined by GC relative to an internal standard.a Bulk Ni content was obtained by ICP-OES.b Surface Ni content was obtained by XPS. | |||||||||
| 1 | No catalysts | 0 | — | — | — | — | — | — | — |
| 2 | Ni-NDC | 8.5 | 1.5 | — | — | — | — | 21.3 | 1.01 |
| 3 | Ni-NDC-700 | 24.9 | 14.4 | 6.0 | — | 3.6 | — | 60.5 | 1.18 |
| 4 | Ni-NDC-600 | 92.8 | 80.3 | 60.1 | 1.9 | 8.8 | 4.5 | 57.9 | 1.31 |
| 5 | Ni-NDC-500 | 99.9 | 99.9 | 77.9 | 3.0 | — | — | 54.5 | 1.54 |
| 6 | Ni-NDC-400 | 14.8 | 4.4 | 0.7 | — | 4.8 | 0.9 | 48.0 | 0.59 |
TEM and HR-TEM were employed to observe the morphology and spatial structure of the prepared catalysts. In Fig. 2a and b, the uncalcined catalyst did not exhibit any visible Ni metal particles. However, after calcination, uniformly dispersed Ni nanoparticles with an average size of approximately 10 nm were observed within the carbon framework (Fig. 2c–j). The HRTEM images revealed the detailed structure of these Ni nanoparticles, demonstrating high crystallinity and clear lattice fringes. The lattice spacings of 1.76 Å and 2.03 Å corresponded to the (200) plane and (111) plane of nickel, respectively, as per the JCPDS 70-0989 reference. These TEM observations aligned with the XRD results, showing that the average particle size of Ni increased gradually with the calcination temperature.32
 | ||
| Fig. 2 TEM and HRTEM images of Ni-NDC (a and b), Ni-NDC-400 (c and d), Ni-NDC-500 (e and f), NDC-600 (g and h), and Ni-NDC-700 (i and j). | ||
3.2 Effect of calcination temperature
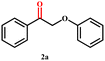
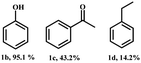
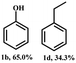
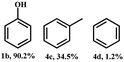
2-Phenoxy-1-phenylethanol (1a) is a dimeric model compound of lignin and was used as a probe molecule to study the STH process of β-O-4 linkage. Phenol (1b), acetophenone (1c), ethylbenzene (1d), 2-phenoxyacetophenone (1e) and phenoxyethylbenzene (1f) were produced during the reaction process (Scheme 1). The performances of Ni-MOF and catalysts pyrolyzed at different temperatures are listed in Table 1. The substrate remains unconverted in the absence of a catalyst (Table 1, entry 1). However, upon adding catalysts, the conversions of 1a significantly improved. The activity of Ni-NDC was very low and the conversion of 1a was only 8.5% (Table 1, entry 2). Ni-NDC-400 also exhibited low activity due to the predominant presence of Ni2+ rather than active Ni0 species. Ni-NDC-500 showed the best catalytic performance among the catalysts checked and the conversion of 1a could reach 99.9% and the yields of phenol and acetophenone were 99.9% and 77.9%, respectively. A higher calcination temperature results in lower activity. Further increasing the calcination temperature led to decreased activity. Over Ni-NDC-600, the yields of phenol and acetophenone were 80.3% and 60.1%, respectively. However, these yields decreased significantly to 14.4% and 6.0% over Ni-NDC-700. The XRD, XPS, and TEM results indicated that excessively high temperatures caused Ni metal agglomeration and a decrease in Ni0 content on the surface, while excessively low temperatures resulted in incomplete reduction of Ni to the metallic state. The optimal catalytic activity of Ni-NDC-500 could be attributed to its high surface Ni0 content and the appropriate size of Ni nanoparticles. | ||
| Scheme 1 Cleavage of 2-phenoxy-1-phenylethanol in the absence of external hydrogen over Ni-based catalysts. | ||
3.3 Optimization of reaction conditions over Ni-NDC-500
The optimization of reaction conditions was performed using 2-phenoxy-1-phenylethanol as the probe molecule and Ni-NDC-500 as the catalyst. Initially, the impact of temperature on the STH reaction was investigated (Fig. 3a). Higher temperatures led to an increased conversion of 1a, with a complete conversion at 220 °C. At this temperature, the yields of phenol and acetophenone were 99.9% and 77.9%, respectively. However, when the temperature rose to 240 °C, the yield of acetophenone decreased while the yield of ethylbenzene increased. The effect of reaction time was examined at 220 °C, as shown in Fig. 3b. With a reaction time of 2 h, the substrate was completely converted into acetophenone and phenol. Subsequently, as the reaction time extended, acetophenone underwent further hydrogenation to produce ethylbenzene. The reduction of acetophenone occurred through hydrogen derived from water, leading to the oxidation of the catalyst Ni0 to Ni2+. Additionally, Fig. 3c and d demonstrate the influence of solvent amount and catalyst quantity on the STH reaction. 40 mg of the catalyst yielded relatively satisfactory results for the STH reaction. The amount of solvent also had a discernible effect on the reaction outcome, as an increase in solvent amount improved the reaction efficiency.3.4 Reuse of the catalyst
The catalytic stability of the Ni-NDC-500 catalyst was assessed by conducting recycling experiments. The catalyst was recovered from the reaction mixture through magnetic separation (Fig. S3†), followed by washing and drying before being reused in the subsequent cycles. The performance of the catalyst did not change obviously over three cycles (Fig. 4). After four cycles, there was a slight decrease in reactivity. Comparison of the XPS spectra and XRD patterns (Fig. S4 and S5†) between the catalyst after four cycles and the fresh catalyst revealed no alterations in crystal structure. The surface Ni0/(Ni0 + Ni2+) content decreased only from 71% to 57% after the four reaction cycles. Additionally, Fig. S6† shows the TEM image of the used catalyst, which closely resembles that of the original catalyst (Fig. 2e and f). These results further confirm that Ni-NDC-500 exhibits both a well-preserved spatial structure and robust catalytic stability during the STH reaction.3.5 STH process of lignin models and native lignin
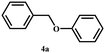
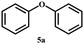
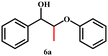
After establishing a reliable procedure, we investigated the substrate scope of the novel Ni-NDC-500-catalyzed hydrogenolysis reaction of lignin model compounds without external hydrogen. Notably, the catalyst effectively cleaved ether bonds in β-O-4 model substrates containing hydroxyl groups (6a–8a, Table 2, entries 5–7). Interestingly, it was also observed that the Ni-NDC-500 catalyst facilitated the cleavage of ether bonds in substrates lacking Cα–OH groups (2a–5a, Table 2, entries 1–4). This suggests that the catalyst could utilize hydrogen derived from H2O to enable the cleavage of ether bonds. The X-ray diffraction (XRD) pattern in Fig. S7† demonstrates the emergence of NiO following the catalysis of the 2a and 3a transformations using Ni-NDC-500. This suggests that in the absence of Cα–OH in the substrate, hydrogen from water reduces the substrate, leading to the oxidation of Ni0 to NiO.20 Meanwhile 2-phenoxy-1-acetophenone (2a) exhibited lower reactivity compared to 2-phenoxy-1-phenylethanol (Scheme S1†), undergoing transformation into phenol and acetophenone using the Ni-NDC-500 catalyst, despite the lower ether bond energy in 2-phenoxy-1-acetophenone (2a, 227.8 kJ mol−1) compared to 2-phenoxy-1-phenylethanol (1a, 274.0 kJ mol−1).33 This suggests that Ni-NDC-500 demonstrates higher activity for self-transfer hydrogenolysis using Cα–OH as the hydrogen source compared to transfer hydrogenolysis with H2O as the hydrogen source, indicating that the STH process governs the reaction.The self-transfer hydrogenolysis (STH) process of native lignin, extracted from poplar, eucalyptus, and sequoia wood powder using the Ni-NDC-500 catalyst, was carried out following established methods.34 The obtained results, presented in Fig. 5 and Fig. S8,† revealed lignin monomer yields of 5.5%, 10.4%, and 7.4% by weight for poplar, eucalyptus, and metasequoia, respectively. Analysis conducted using 2D-HSQC and HMBC (Fig. S9–S11†) confirmed the efficient cleavage of β-O-4 bonds in lignin by the Ni-NDC-500 catalyst, with the resulting monomer retaining the methoxy group in the lignin unit structure.
3.6 Main reaction pathway
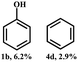
Control experiments were conducted to investigate the reaction mechanism described in Scheme 2. When compound 1a was transformed with Ni-NDC-500 at 150 °C for 12 hours, yields of 92.3% phenol (1b) and 63.6% acetophenone (1c) were obtained. However, no products were detected when (1-methoxy-2-phenoxyethyl)benzene (9a) was used under the same reaction conditions. These results highlight the importance of Cα–OH groups in the transformation process. Interestingly, at 220 °C, (1-methoxy-2-phenoxyethyl)benzene (9a) exhibited 39% conversion, resulting in 26.9% phenol (1b), 15.1% (1-methoxyethyl)benzene (9b) and 7.8% ethylbenzene (1d). This suggests that the catalyst can facilitate ether bond cleavage by utilizing hydrogen derived from H2O at 220 °C, while self-transfer hydrogenolysis utilizing Cα–OH as the hydrogen source process occurred at 150 °C. Furthermore, when 1,2-diphenylethanol (10a) was used as the substrate, the dehydration reaction took place and stilbene (10d) was produced in the absence of a catalyst, while the dehydrogenation reaction proceeded with Ni-NDC-500 and 1,2-diphenylethan-1-one (10b) was produced.Based on the obtained results, a proposed pathway and the reaction mechanism are presented in Scheme 3. The reaction initiates with the dehydrogenation of 2-phenoxy-1-phenylethanol, resulting in the formation of 2-phenoxy-1-acetophenone and the adsorption of chemically bound hydrogen (“hydrogen pool”) on Ni nanoparticles.35,36 Subsequently, the presence of atomic hydrogen on Ni-NDC-500 facilitates the cleavage of the β-O-4 linkage to form acetophenone and phenol.
 | ||
| Scheme 3 Reaction mechanism of the lignin β-O-4 model cleavage to produce phenol and acetophenone over Ni-NDC-500 catalysts without external hydrogen. | ||
Isotopic labeling was used to further validate the reaction mechanism. When the Cα position of 2-phenoxy-1-phenylethanol is replaced by deuterium (11a), the reaction rate slows down due to the isotopic effect.37 The reactions were performed in H2O and D2O. The mass spectrometry of 1a and hydrogenolysis products shows deuterium labelled phenol and acetophenone under the reaction conditions. It is worth noting that deuterium labelled 1a was also detected after 12 h at 150 °C even in the absence of catalyst (Fig. 6d, Fig. S12–S14†). The NMR data showed that an H/D exchange occurred between Cα–OH and D2O and Cα–OH changed to Cα–OD even in the absence of catalyst (Fig. S15†). In the presence of catalyst, 1a with a molecular weight of 218 was detected, implying that more H/D exchange of the substrate occurred in the presence of Ni.38 A primary isotope effect was observed (the ratio of reaction rates using H2O and D2O, kH2O/kD2O = 1.51, Fig. S17†) at 150 °C, suggesting that the dehydrogenation (Cα–OH into Cα![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) is the rate-determining step.
O) is the rate-determining step.
4. Conclusions
Ni-MOF was used as a precursor to prepare the Ni/C catalyst, which could catalyze the transformation of lignin model compounds and lignin into monomers. The performance of the catalysts depended strongly on the calcination temperature, and the Ni-NDC-500 catalyst showed the best performance among the catalysts pyrolyzed at different temperatures, leading to an efficient cleavage of various lignin model compounds and native lignin into monomers with high yields. In addition, the catalyst was reused three times without obvious reduction in activity. Based on the findings from control experiments, possible reaction pathways have been proposed. The suggested reaction mechanism involves two steps: the dehydrogenation of Cα–OH on Ni-NDC-500 to generate a Cα![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O intermediate and a “hydrogen pool”, followed by the hydrogenolysis of the ether bond in the Cα
O intermediate and a “hydrogen pool”, followed by the hydrogenolysis of the ether bond in the Cα![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O intermediate facilitated by the “hydrogen pool”. This study represents a novel work on the utilization of non-precious metal-based catalysts for the STH process of lignin.
O intermediate facilitated by the “hydrogen pool”. This study represents a novel work on the utilization of non-precious metal-based catalysts for the STH process of lignin.
Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors thank the National Natural Science Foundation of China (22293012, 22179132, 22293015, and 22121002) and the Research Funds of Happiness Flower ECNU (2020ST2203) for support.References
- C. O. Tuck, E. Pérez, I. T. Horváth, R. A. Sheldon and M. Poliakoff, Science, 2012, 337, 695–699 CrossRef CAS PubMed.
- G. Liu, A. W. Robertson, M. M. Li, W. C. H. Kuo, M. T. Darby, M. H. Muhieddine, Y. C. Lin, K. Suenaga, M. Stamatakis, J. H. Warner and S. C. E. Tsang, Nat. Chem., 2017, 9, 810–816 CrossRef CAS PubMed.
- B. Zheng, J. Song, H. Wu, S. Han, J. Zhai, K. Zhang, W. Wu, C. Xu, M. He and B. Han, Green Chem., 2021, 23, 268–273 RSC.
- Y. Yang, Y. Wang, S. Li, X. Shen, B. Chen, H. Liu and B. Han, Green Chem., 2020, 22, 4937–4942 RSC.
- S. Li, M. Dong, M. Peng, Q. Mei, Y. Wang, J. Yang, Y. Yang, B. Chen, S. Liu, D. Xiao, H. Liu, D. Ma and B. Han, Innovation, 2022, 3, 100189 CAS.
- Y. Liao, S.-F. Koelewijn, G. Van den Bossche, J. Van Aelst, S. Van den Bosch, T. Renders, K. Navare, T. Nicolaï, K. Van Aelst, M. Maesen, H. Matsushima, J. M. Thevelein, K. Van Acker, B. Lagrain, D. Verboekend and B. F. Sels, Science, 2020, 367, 1385–1390 CrossRef CAS PubMed.
- Y. Queneau and B. Han, Innovation, 2022, 3, 100184 CAS.
- H. Liu, T. Jiang, B. Han, S. Liang and Y. Zhou, Science, 2009, 326, 1250–1252 CrossRef CAS PubMed.
- M. Cao, B. Chen, T. Ruan, X. Ouyang and X. Qiu, Acta Phys.-Chim. Sin., 2022, 38(10), 2204037 Search PubMed.
- H. Zhou, Y. Jing and Y. Wang, Acta Phys.-Chim. Sin., 2022, 38(10), 2203016 Search PubMed.
- J. Zhang, Green Energy Environ., 2018, 3, 328–334 CrossRef.
- A. Bjelić, M. Grilc, M. Huš and B. Likozar, Chem. Eng. J., 2019, 359, 305–320 CrossRef.
- C. Espro, B. Gumina, T. Szumelda, E. Paone and F. Mauriello, Catalysts, 2018, 8, 313 CrossRef.
- J. M. Nichols, L. M. Bishop, R. G. Bergman and J. A. Ellman, J. Am. Chem. Soc., 2010, 132, 12554–12555 CrossRef CAS PubMed.
- J.-w. Zhang, G.-p. Lu and C. Cai, Green Chem., 2017, 19, 4538–4543 RSC.
- Z. Dou, Z. Zhang and M. Wang, Appl. Catal., B, 2022, 301, 120767 CrossRef CAS.
- L. Li, L. Dong, D. Li, Y. Guo, X. Liu and Y. Wang, ACS Catal., 2020, 10, 15197–15206 CrossRef CAS.
- L. Li, T. Zhang, Z. Guo, X. Liu, Y. Guo, Y. Huang and Y. Wang, Ind. Eng. Chem. Res., 2021, 60, 11699–11706 CrossRef CAS.
- Z. Guo, L. Li, Y. Guo, X. Liu and Y. Wang, Catal. Sci. Technol., 2022, 12, 5143–5151 RSC.
- X. Ren, Z. Sun, J. Lu, J. Cheng, P. Zhou, X. Yu, Z. Rong and C. Li, Green Chem., 2023, 25, 1955–1969 RSC.
- Q. Meng, J. Yan, H. Liu, C. Chen, S. Li, X. Shen, J. Song, L. Zheng and B. Han, Sci. Adv., 2019, 5, eaax6839 CrossRef CAS PubMed.
- Q. Meng, J. Yan, R. Wu, H. Liu, Y. Sun, N. Wu, J. Xiang, L. Zheng, J. Zhang and B. Han, Nat. Commun., 2021, 12, 4534 CrossRef CAS PubMed.
- X. Shen, C. Zhang, B. Han and F. Wang, Chem. Soc. Rev., 2022, 51, 1608–1628 RSC.
- X. Shen, Y. Xin, H. Liu and B. Han, ChemSusChem, 2020, 13, 4367–4381 CrossRef CAS PubMed.
- Q. Song, F. Wang, J. Cai, Y. Wang, J. Zhang, W. Yu and J. Xu, Energy Environ. Sci., 2013, 6, 994–1007 RSC.
- H. Luo, I. M. Klein, Y. Jiang, H. Zhu, B. Liu, H. I. Kenttämaa and M. M. Abu-Omar, ACS Sustainable Chem. Eng., 2016, 4, 2316–2322 CrossRef CAS.
- A. Shivhare, D. Jampaiah, S. K. Bhargava, A. F. Lee, R. Srivastava and K. Wilson, ACS Sustainable Chem. Eng., 2021, 9, 3379–3407 CrossRef CAS.
- Q. Wang, L.-P. Xiao, Y.-H. Lv, W.-Z. Yin, C.-J. Hou and R.-C. Sun, ACS Catal., 2022, 12, 11899–11909 CrossRef CAS.
- X.-G. Si, Y.-P. Zhao, Q.-L. Song, J.-P. Cao, R.-Y. Wang and X.-Y. Wei, React. Chem. Eng., 2020, 5, 886–895 RSC.
- W. Wu, H. Liu, H. Wu, B. Zheng, S. Han, K. Zhang, X. Mei, C. Xu, M. He and B. Han, ACS Sustainable Chem. Eng., 2021, 9, 11862–11871 CrossRef CAS.
- M.-Y. Zong, C.-Z. Fan, X.-F. Yang and D.-H. Wang, Mol. Catal., 2021, 509, 111609 CrossRef CAS.
- Y. Guo, X. Gao, C. Zhang, Y. Wu, X. Chang, T. Wang, X. Zheng, A. Du, B. Wang, J. Zheng, K. Ostrikov and X. Li, J. Mater. Chem. A, 2019, 7, 8129–8135 RSC.
- C. Zhang, J. Lu, X. Zhang, K. MacArthur, M. Heggen, H. Li and F. Wang, Green Chem., 2016, 18, 6545–6555 RSC.
- L. Dong, L. Lin, X. Han, X. Si, X. Liu, Y. Guo, F. Lu, S. Rudić, S. F. Parker, S. Yang and Y. Wang, Chem, 2019, 5, 1521–1536 CAS.
- M. V. Galkin, C. Dahlstrand and J. S. Samec, ChemSusChem, 2015, 8, 2187–2192 CrossRef CAS PubMed.
- N. Luo, M. Wang, H. Li, J. Zhang, T. Hou, H. Chen, X. Zhang, J. Lu and F. Wang, ACS Catal., 2017, 7, 4571–4580 CrossRef CAS.
- X. Zhao, J. Wang, L. Lian, G. Zhang, P. An, K. Zeng, H. He, T. Yuan, J. Huang, L. Wang and Y.-N. Liu, ACS Catal., 2023, 13, 2326–2334 CrossRef CAS.
- M. Gómez-Gallego and M. A. Sierra, Chem. Rev., 2011, 111, 4857–4963 CrossRef PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Materials, experimental procedure scheme of catalysts, lignin extraction process, synthesis of lignin model compounds, characterization of catalysts, GC profile, GC-MS data and NMR data. See DOI: https://doi.org/10.1039/d3gc04217k |
| This journal is © The Royal Society of Chemistry 2024 |