Green liquid marble-based hydrogels as pesticidal pyrethrin slow-release carriers†
Qin
Li‡
a,
Changhong
Wang‡
a,
Jiayuan
He
a,
Dandan
Yang
a,
Ting
Li
a,
Huixian
Xu
a,
Weifeng
Shen
 a,
Liandi
Zhou
*b,
Saimeng
Jin
a,
Liandi
Zhou
*b,
Saimeng
Jin
 *a,
Qihui
Zhang
*a,
Qihui
Zhang
 *a and
James H.
Clark
*a and
James H.
Clark
 c
c
aSchool of Chemistry and Chemical Engineering, Chongqing University, Chongqing, 400044, China. E-mail: qhzhang@cqu.edu.cn; sj708@cqu.edu.cn
bChongqing College of Traditional Chinese Medicine, Chongqing 402760, China. E-mail: zhouliandi@cqctcm.edu.cn
cCirca Renewable Chemistry Institute, Green Chemistry Centre of Excellence, University of York, York, YO10 5DD, UK
First published on 30th January 2024
Abstract
Pyrethrins are effective agents against mosquito larvae, but their rapid degradation rate in water reduces their deinsectization efficiency and increases the cost of deinsectization. Based on liquid marbles, a biodegradable slow-release hydrogel carrier is prepared using green and low-cost materials for the slow release of natural pyrethrin on water surfaces to kill mosquito larvae. Green carriers with high stability and loading of high concentrations of pyrethrin are synthesized by cross-linking Ca2+ in 5% CaCl2 solution with sodium alginate. The superior stability of liquid marbles at the water surface and the electrostatic interaction between alginate and gelatin can effectively reduce the degradation rate of pyrethrin in water. Furthermore, the carrier system displayed a long sustained-release time (>144 h) and maintained larvicidal performance after 168 h. Good floating stability is demonstrated for the liquid marble hydrogels. The larvicidal activity of the obtained drug carrier reached 100% with different water volumes but the same dosage. This work provides a strategy to exterminate mosquito larvae with high efficiency by preparing a green hydrogel drug slow-release carrier, thus providing a new approach to expanding the application of liquid marbles in green chemistry.
1. Introduction
Mosquitoes are one of the main culprits in spreading malaria and other diseases, including dengue, chikungunya, yellow fever and Zika virus.1 In the open water system, the population of mosquito larvae can be controlled by various predators, including tadpoles, water bugs, fish, copepods and crabs.2 However, the smell from stagnant pools can attract mosquitoes to lay eggs, which hatch to release larvae.3 Therefore, mosquitoes often thrive in small stagnant waterholes.4 Organophosphates, microbial agents and insect growth regulators are normally applied to directly target mosquitoes; however, these agents are highly toxic or expensive.5 Some researchers have developed methods that use nanomaterials to block the mosquito life cycle.6,7 Parvin et al. developed a non-toxic nanoparticle coated with carbon to kill mosquito larvae after a long period of stagnation in the larval stage.8 As mosquito larvae (also known as worms) hang upside down below the surface of water and breathe at the surface with the end of their tails, the release of insecticides at the surface of water is an effective means of mosquito extermination. However, few studies have described a way to control mosquitoes based on the behaviour of the larvae.Pyrethrin is a green biocide derived from pyrethrum flowers and has been used against mosquitoes for a long time. Natural pyrethrin is effective in eradicating a wide range of insects and is considered less toxic to mammals and other non-target organisms.9 Compared to synthetic insecticides, natural pyrethrin is eco-friendly with a very short half-life in natural environment.10 Because of the degradation and deactivation of pyrethrin in sunlight and air,11 it does not pose a risk to humans and the environment. Despite its lipophilic properties, natural pyrethrin degrades rapidly in water and leaves no residue in water sediments or fish tissue.12 Rapid degradation leads to increased release frequency, dosage, and cost of pyrethrin, resulting in difficulty in mosquito control. Thus, a continuous and controlled mode for pyrethrin release needs to be developed to achieve an expected larvicidal efficacy and avoid the evaporation or early degradation of pyrethrin.
Early studies have provided some methods that are used to solve the above problems. Researchers prepared pyrethrin formulations in which pyrethrin was loaded with natural polymers,13 inorganic non-metallic materials14 and nano-mixed micelles.15–17 Among various natural polymers, alginates are used extensively in the preparation of hydrogel microspheres, micro-capsules and membranes for controlled drug release because of their biocompatible, biodegradable and nontoxic properties.18–20 In addition, alginates can be formed as ionotropic hydrogels in the presence of Ca2+, Ba2+, Fe3+ and other divalent or trivalent cations.21 Gelatin is a natural addition for preparing a drug carrier because of its unique characteristics, including nontoxic, edible, biodegradability and low cost.22 Some reports suggested that there exists electrostatic interaction between the groups of gelatin and alginates, which favors the stability of emulsion.23,24 Therefore, alginates and gelatin were selected as the aqueous phase to prepare the pyrethrin formulation.
Liquid marble is a natural phenomenon generally associated with the activity in that aphids transformed liquids into soft non-stick solids using wax-like substances.25 In 2001, Aussillous and Quere first named the shell–core type droplet as liquid marbles and reported on it systematically.26 Theoretically, the grains that encapsulate droplets can be any particle size less than a few hundred microns, such as silica particles, carbon nanotubes, and cellulose.27 The surface energy of the droplet will be reduced after covering the non-hydrophilic powder. Consequently, the globular liquid marbles maintain their original shapes, and the contact surface is not moistened, which makes liquid marbles stable in the air or water surface.28 Owing to these properties, such as easy preparation, wide source of raw materials, non-wetting and good elasticity, liquid marbles have gained increasing attention and are widely used in fields of cosmetics, microfluidics, microreactors and biopharmaceuticals.29–34 However, limited by their poor mechanical stability, up to now, liquid marbles have rarely been used in the agriculture sector to release pesticide.35
Herein, the advantages of liquid marbles and the habit of mosquito larvae were exploited, using green substances, such as cellulose, sodium alginate and gelatin, to design a hydrogel drug carrier to kill Aedes albopictus larvae continuously and chronically with fewer doses (Scheme 1). The sodium alginate solution and gelatin solution were mixed and then homogenized with pyrethrin to obtain a pyrethrin emulsion. Then, the pyrethrin emulsion was dropped onto the modified cellulose powder by pipette to prepare liquid marble. Subsequently, the liquid marble was placed on a 5% CaCl2 solution, and the liquid marble-based hydrogel was obtained by water exchange. The formation of hydrogels efficiently overcomes the mechanical instability of liquid marble. Simultaneously, the modified cellulose on the liquid marble-based hydrogels makes them float on the water, releasing pyrethrin to kill wigglers.
2. Materials and methods
2.1 Materials
Sodium alginate (SA, 98%) was obtained from Shanghai Yuanye Bio-Technology Co. (Shanghai, China). Gelatin (GE, 99%) was obtained from Damao Chemical Reagent Co. (Tianjin, China). Calcium chloride anhydrous (CaCl2, 96%) and vinyltriethoxysilane (VTES, 97%) were obtained from Aladdin Reagent Co. (Shanghai, China). Ethanol (EtOH, 99%) and sodium hydroxide (NaOH, 98%) were obtained from Kelong Chemicals Co. (Chengdu, China). Cellulose (99%) was obtained from Macklin Biochemical Co. (Shanghai, China). The pyrethrin technology (Py, 50.8%) was obtained from Nanbao Company Co. (Yunnan, China). Pyrethrin (80%) was obtained from Cato Research Chemicals, Inc. (America).The Aedes albopictus larvae were obtained from the Center for Disease Control and Prevention (Zhejiang, China).
2.2 Synthesis of composites
The water contact angles (WCAs) were measured by dropping 5 μL deionized water onto the cellulose powder surface, which was pressed on a glass slide. The average value of the three measurements at different spots was the final WCA of each group.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 5 minutes to obtain a pyrethrin emulsion (Py-SA-GE) at concentrations ranging from 2.1% to 12.5% (w/v). As the internal solution, the pyrethrin emulsion was dropped onto the modified cellulose powder to prepare the liquid marbles. Subsequently, liquid marbles loaded with pyrethrin were placed on the surface of the 5% CaCl2 solution to obtain liquid marble hydrogels loaded with pyrethrin (Py-LM-Gels). Finally, the liquid marble hydrogels were transferred into a freeze dryer for ice sublimation for 12 h. The blank liquid marble hydrogels (LM-Gels) were prepared without adding pyrethrin. For comparison with liquid marble hydrogels, conventional calcium alginate hydrogels were also prepared by dropping Py-SA-GE and SA-GE into a 5% CaCl2 solution.
000 rpm for 5 minutes to obtain a pyrethrin emulsion (Py-SA-GE) at concentrations ranging from 2.1% to 12.5% (w/v). As the internal solution, the pyrethrin emulsion was dropped onto the modified cellulose powder to prepare the liquid marbles. Subsequently, liquid marbles loaded with pyrethrin were placed on the surface of the 5% CaCl2 solution to obtain liquid marble hydrogels loaded with pyrethrin (Py-LM-Gels). Finally, the liquid marble hydrogels were transferred into a freeze dryer for ice sublimation for 12 h. The blank liquid marble hydrogels (LM-Gels) were prepared without adding pyrethrin. For comparison with liquid marble hydrogels, conventional calcium alginate hydrogels were also prepared by dropping Py-SA-GE and SA-GE into a 5% CaCl2 solution.
2.3 Pesticide loading capacity
To assess the percentage loading (PL%) and percentage entrapment efficiency (EE%) of the Py-LM-Gels, the content of pyrethrin in the Py-LM-Gels was detected using a T6 UV/VIS spectrophotometer. The Py-LM-Gels were ground in a mortar and then immersed in 5 mL of methanol. The suspension was sonicated for 1 h and centrifuged at 8000 rpm for 5 min. After that, the supernatant absorbance of UV analysis was recorded at 230 nm.36 The whole process was conducted under light-proof conditions. The drug carrier without pyrethrin has no influence on absorbance at 230 nm (Fig. S1†). The standard curve, which is used to calculate the amount of pyrethrin, is shown in Fig. S2.† The calculated formulas of PL% and EE% of the Py-LM-Gels are as follows: | (1) |
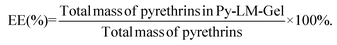 | (2) |
2.4 Release studies
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v
1, v![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) v) at a fixed temperature. All samples were shaken mildly in the dark. 100 μL of the released medium was removed at different time intervals (1, 2, 4, 6, 8, 10, 12, 24, 48, 72, 96, 120 and 144 h), and the absorbance using UV–VIS spectrometer was detected at 230 nm. An equal volume of fresh medium was added after sampling. The pyrethrin in the dissolved medium was calculated using the pyrethrin standard curve (Fig. S3†). For comparison, the release properties of the equivalent pyrethrin technology were investigated in the same way. The release rate of pyrethrin was calculated by applying eqn (3) as follows:
v) at a fixed temperature. All samples were shaken mildly in the dark. 100 μL of the released medium was removed at different time intervals (1, 2, 4, 6, 8, 10, 12, 24, 48, 72, 96, 120 and 144 h), and the absorbance using UV–VIS spectrometer was detected at 230 nm. An equal volume of fresh medium was added after sampling. The pyrethrin in the dissolved medium was calculated using the pyrethrin standard curve (Fig. S3†). For comparison, the release properties of the equivalent pyrethrin technology were investigated in the same way. The release rate of pyrethrin was calculated by applying eqn (3) as follows: | (3) |
 | (4) |
 | (5) |
 | (6) |
 | (7) |
2.5 Stability
 | (8) |
2.6 Application
3. Results and discussion
3.1 Characterization
As shown in Fig. 2(A), the WCA of modified cellulose is highest when the volume of NaOH is 9 mL. Therefore, 9 mL NaOH was selected as the optimal condition. Similarly, 90 min and 80% ethanol concentrations were selected as the optimum conditions for the modification of cellulose (Fig. 2(B) and (C)).
As shown in Fig. 2(D), the WCAs of cellulose with different ratios of cellulose-VTES were not significantly different. Considering environmental protection, cellulose-VTES (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, w
1, w![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) w) was selected as the optimum condition. The number of silane groups in silanized cellulose was 8.54 μmol g−1 determined by applying an Inductively Coupled Plasma Mass Spectrometer (ICP-MS).
w) was selected as the optimum condition. The number of silane groups in silanized cellulose was 8.54 μmol g−1 determined by applying an Inductively Coupled Plasma Mass Spectrometer (ICP-MS).
The contact angle is the basic measurement used to estimate the wettability of a surface. The WCAs correlate with surface energy, which determines whether the powder can be used to prepare liquid marbles. Fig. 2(E) shows the WCAs of the cellulose before and after modification (0° → 143°). The surface energy of silanized cellulose is 56.93 mJ m−2, which is calculated from the contact angle of silanized cellulose powder with formamide (31.831° ± 0.550) (Fig. S6†) and WCA. Fig. S7† illustrates the dispersion of silanized cellulose in different solvents, where it can be observed that the modified cellulose floats on the surface of the water owing to its excellent hydrophobicity while settling to the bottom in less polar solvents. Fig. 2(F) presents the images of liquid marbles with different volumes prepared by modified cellulose, demonstrating that modified cellulose is sufficiently hydrophobic to carry out subsequent experiments.
The FT-IR spectrum of the cellulose before and after modification is shown in Fig. 3(a) and (b). The bands at 3063 and 2960 cm−1 in Fig. 3(b) originate from the C–H stretching vibration and unsaturated alkene bonding. The band at 1602 cm−1 can be attributed to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibration. The band at 1409 cm−1 is attributed to the in-plane deformation vibration of the vinyl. In addition, the Si–O–C band at 763 cm−1 confirms the successful preparation of modified cellulose.37 The above results illustrated that the cellulose was modified by VTES. VTES can be hydrolyzed to yield vinylsilanetriol in an alkaline environment,38 following which the cellulose is silanized by vinylsilanetriol, as shown in Fig. 1. Silanized cellulose, meanwhile, has high hydrophobicity,39 interfacial bonding, and environmental durability40,41 owing to the introduction of silyl groups.
C stretching vibration. The band at 1409 cm−1 is attributed to the in-plane deformation vibration of the vinyl. In addition, the Si–O–C band at 763 cm−1 confirms the successful preparation of modified cellulose.37 The above results illustrated that the cellulose was modified by VTES. VTES can be hydrolyzed to yield vinylsilanetriol in an alkaline environment,38 following which the cellulose is silanized by vinylsilanetriol, as shown in Fig. 1. Silanized cellulose, meanwhile, has high hydrophobicity,39 interfacial bonding, and environmental durability40,41 owing to the introduction of silyl groups.
| Drug release carriers | Reaction condition | Sources of raw materials | Degradability | Ref. |
|---|---|---|---|---|
| MCM-48 | Room temperature, 24 h; 550 °C, 5 h; 110 °C, N2 reflux, 24 h. | CATB, Pluronic F127, TEOS, MPTMS, APTS, TMCS | Not degradable | 45 |
| LC@UiO-66 nanoparticle | 120 °C, 24 h; room temperature, 24 h. | ZrCl4, H2BDC | Not degradable | 46 |
| Ace@MSN-SS-C10 | 80 °C, 24 h; 600 °C, 5 h; room temperature, 24 h; 80 °C, 24 h. | CATB, Milli-Q, TEOS, MPTMS, GSH | Not degradable | 47 |
| PEG/MPTMS composite nanocarrier | Room temperature, 24 h; 85 °C, 24 h; 140 °C, 24 h. | h-BN, PEG, MPTMS | Not degradable | 48 |
| CAP@MIL-101(Fe)@silica | 120 °C, 12 h; room temperature, 24 h. | FeCl3, BDC, Na2SiO3·9H2O | Not degradable | 49 |
| Py-SA-GE | Room temperature, 1.5 h; 100 °C, 1 h; 60 °C, 12 h. | cellulose, VTES, sodium alginate | Degradable | This work |
As illustrated in Fig. 5(A), the appearance of the liquid marble hydrogels and conventional calcium alginate hydrogels was different. The cellulose shell makes both types of liquid marbles (loaded and unloaded pyrethrin) appear white. The micromorphology of LM-Gels, Py-LM-Gels, conventional calcium alginate hydrogels (Gels) and conventional calcium alginate hydrogels loaded pyrethrin (Py-Gels) were observed by SEM. The surfaces of the LM-Gels and Py-LM-Gels were covered by cellulose microspheres (Fig. 5(A)), which are hydrophobic and give them the ability to float on the water surface. The surfaces of the Gels have many folds, which may be caused by volume shrinkage after freeze-drying.24 Meanwhile, the surfaces of the Py-Gels were uneven, but no distinct folds were formed, which means that the addition of pyrethrin influenced the morphology of the hydrogels.
The FT-IR spectrum of pyrethrin technology showed a distinctive characteristic peak at 1717 cm−1 (Fig. 5(B, insert a)) attributed to the stretching vibration of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O. Compared to LM-Gels (Fig. 5(B, insert c)), FT-IR analysis confirmed the presence of C
O. Compared to LM-Gels (Fig. 5(B, insert c)), FT-IR analysis confirmed the presence of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O in Py-LM-Gels (Fig. 5(B, insert b)) at 1717 cm−1, which indicated that pyrethrin was successfully loaded into the material.
O in Py-LM-Gels (Fig. 5(B, insert b)) at 1717 cm−1, which indicated that pyrethrin was successfully loaded into the material.
The thermal decomposition processes of pyrethrin technology, LM-Gels, and Py-LM-Gels are depicted in Fig. 5(C). The weight loss of LM-Gels and Py-LM-Gels was below 100 °C because of the water loss. The first mass loss of pyrethrin technology occurred in the range of 120–200 °C. Mass loss of LM-Gels and Py-LM-Gels mainly occurred in the range of 200–500 °C. The thermal decomposition of SA was reported to occur in the range of 200–280 °C, primarily caused by the breakage of its uronic and glucuronic acid segments.24 In addition, the cellulose on the surface of LM-Gels and Py-LM-Gels is pyrolyzed in the range of 280–500 °C, undergoing dehydration, decarbonization and glycosidic bond breaking reactions. These processes generate small molecules of gas and large condensed volatile components, resulting in a rapid increase in mass loss. Compared to pyrethrin technology, Py-LM-Gels shift backwards in the range of 200–500 °C, indicating that the thermal decomposition temperature tends to be higher. In other words, Py-LM-Gels have a higher thermal stability than pyrethrin, which can mitigate the degradation of pyrethrin. Compared to LM-Gels, the lower inflection temperature of Py-LM-Gels at 200–300 °C suggested that the presence of pyrethrin may have caused minor changes to the internal structure of the liquid marble gels. From 500 to 800 °C, the mass losses of pyrethrin technology, LM-Gels, and Py-LM-Gels tend to be stable.
3.2 Optimization of preparation conditions
 | ||
| Fig. 6 (A) Effect of SA-GE concentration on PL% and EE%; (B) effect of pyrethrin concentration on PL% and EE%; (C) effect of CaCl2 concentration on PL% and EE%; (D) swelling study of Py-LM-Gels. | ||
| Concentration of SA-GE (%) | Concentration of CaCl2 (%) | Concentration of pyrethrin (%) | PL (%) | EE (%) |
|---|---|---|---|---|
| 1.50 | 5 | 2.5 | 6.8 ± 0.4 | 44.0 ± 0.5 |
| 1.75 | 5 | 2.5 | 11.5 ± 0.4 | 45.9 ± 2.2 |
| 2.00 | 5 | 2.5 | 11.7 ± 0.3 | 49.1 ± 1.0 |
| 2.25 | 5 | 2.5 | 10.6 ± 0.4 | 47.7 ± 1.0 |
| 2.50 | 5 | 2.5 | 8.8 ± 0.3 | 47.0 ± 0.5 |
| 2.00 | 3 | 2.5 | 9.4 ± 0.9 | 45.6 ± 1.6 |
| 2.00 | 5 | 2.5 | 11.6 ± 0.6 | 48.1 ± 0.2 |
| 2.00 | 7 | 2.5 | 7.7 ± 0.2 | 48.8 ± 0.5 |
| 2.00 | 9 | 2.5 | 6.0 ± 0.5 | 51.2 ± 0.9 |
| 2.00 | 11 | 2.5 | 6.3 ± 0.3 | 50.3 ± 0.4 |
| 2.00 | 5 | 2.5 | 12.3 ± 0.2 | 49.8 ± 0.5 |
| 2.00 | 5 | 5.0 | 12.6 ± 0.4 | 68.6 ± 2.6 |
| 2.00 | 5 | 7.5 | 15.5 ± 0.8 | 71.6 ± 0.8 |
| 2.00 | 5 | 10.0 | 19.2 ± 1.2 | 60.5 ± 6.5 |
| 2.00 | 5 | 12.5 | 20.7 ± 0.8 | 58.0 ± 6.1 |
The PL% increases with the increase in pyrethrin concentration (Fig. 6(B)). However, the EE% increased considerably when the concentration of pyrethrin was higher than 7.5%. Therefore, these concentrations (2.5%, 5% and 7.5%) were selected to prepare the liquid marble hydrogels for subsequent release experiments, as the optimized condition.
As illustrated in Fig. 6(C) and Table 2, when the concentration of the CaCl2 solution was lower than 5%, the cross-linking time was too long, leading to pesticide loss. When the CaCl2 concentration was more than 5%, the PL% began to decrease. This is because a high concentration of CaCl2 solution leads to excessive cross-linking in the unit time, resulting in a higher density. To confirm this hypothesis, the swelling aptitude of these samples made under the above 5 conditions was evaluated by gravimetry, as shown in Fig. 6(D). The swelling behavior of Py-LM-Gels is observed to be related to the concentration of the CaCl2 solution: the swelling equilibrium rates decreased as the concentration of the CaCl2 solution increased. This is consistent with the reported results.42 Furthermore, too many crosslinking agents may cause drug release to be inhibited. Therefore, 5% was selected as the optimum condition for the preparation of liquid marble hydrogels.
3.3 Release studies
| Model | Equation | R 2 |
|---|---|---|
| Zero-order | Y = 3.1998 + 0.7965t | 0.7631 |
| First-order | Y = 76.9936(1 − e−0.0366t) | 0.9883 |
| Higuchi | Y = 8.9104t1/2 | 0.9718 |
| Ritger–Peppas | Y = 3.9925t0.6527 | 0.9091 |
Among them, the first-order model was the best-fitted theoretical model (R2 = 0.9836). In addition, the release data correlated well with those of Ritger–Peppas and Higuchi. It is clear from the Ritger–Peppas model equations that the mechanism of drug release involves case II transport (n ≥ 0.85), non-Fickian transport (0.43 < n < 0.85) and Fickian diffusion (n ≤ 0.43).44 The calculated value of n is 0.65, suggesting that the release is caused by non-Fickian diffusion, including swelling of hydrogels by water absorption and slackening of the SA chains. Comparable results of the kinetic parameters were found at different temperatures and pyrethrin concentrations (Table S3†).
3.4 Stability
As shown in Fig. 8(A) and (B), owing to the hydrophobic cellulose on the surface, the Py-LM-Gels can float stably on water for more than 24 h. The floating rate did not decrease until after 48 h. In contrast, the Py-Gels without hydrophobic cellulose immediately sunk to the bottom. Additionally, Py-LM-Gels can be easily removed from water after use.In practical applications, the stability of storage is extremely crucial. Hence, the appearance and pyrethrin content of Py-LM-Gels over a period of 6 months were investigated. It is evident in Fig. 8(C) that the content of all samples remained stable without a significant decrease in concentration or significant changes in appearance.
3.5 Application
4. Conclusions
In this study, the habits of mosquito larvae and the characteristics of liquid marbles were exploited to design a green slow-release carrier by cross-linking Ca2+ and pyrethrin emulsion on the surface of the CaCl2 solution. The liquid marble-based hydrogels showed well-intended features, such as good floatation, long sustained-release time, stable release of pyrethrin in the needed range of temperature and good storage stability. In addition, hydrogel carrier efficient mosquito larvae killing was witnessed with the same dosage of pyrethrin in beakers containing different volumes of water because the materials can release pyrethrin on the water surface. Subsequent investigations could concentrate on improving the reuse efficiency of liquid marble hydrogel drug carriers and utilizing biomass cellulose as a source of material for slow-release drug carriers to improve the sustainability of the introduction strategy. In summary, as a green slow-release drug carrier, this carrier can effectively kill wigglers and provide a new clue for expanding the application of liquid marbles.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was financially supported by National Natural Science Foundation of China (81973451, 22208036), Fundamental and Frontier Research Fund of Chongqing (CSTB2023NSCQ-MSX0490, CSTC2019JSYJ-YZYSBAX0020, CSTC2018JCYJAX0661), the Science and Technology Research Program of Chongqing Municipal Education Commission (KJZD-K201800103), Fundamental Research Funds for the Central Universities (2019CDYGYB027, 2022CDJXY-003), Graduate Research and Innovation Foundation of Chongqing (CYS18033), Graduate Research and Innovation Project of Chongqing (CYB22041), Venture & Innovation Support Program for Chongqing Overseas Returnees (CX2019117).References
- F. Liu, Q. Wang, P. Xu, F. Andreazza, W. R. Valbon, E. Bandason, M. Chen, R. Yan, B. Feng, L. B. Smith, J. G. Scott, G. Takamatsu, M. Ihara, K. Matsuda, J. Klimavicz, J. Coats, E. E. Oliveira, Y. Du and K. Dong, Nat. Commun., 2021, 12, 2553 CrossRef CAS PubMed.
- K. Murugan, G. Benelli, S. Ayyappan, D. Dinesh, C. Panneerselvam, M. Nicoletti, J. S. Hwang, P. M. Kumar, J. Subramaniam and U. Suresh, Parasitol. Res., 2015, 114, 2243–2253 CrossRef PubMed.
- C. L. Silva-Inacio, A. A. Pereira De Paiva, J. M. Galvao De Araujo and M. D. F. Freire De Melo Ximenes, Rev. Soc. Bras. Med. Trop., 2020, 53, e20200502 CrossRef PubMed.
- Y. H. Zhou, Z. W. Zhang, Y. F. Fu, G. C. Zhang and S. Yuan, Front. Behav. Neurosci., 2018, 12, 00086 CrossRef PubMed.
- G. Benelli, Parasitol. Res., 2015, 114, 3201–3212 CrossRef PubMed.
- K. Murugan, D. Nataraj, P. Madhiyazhagan, V. Sujitha, B. Chandramohan, C. Panneerselva, D. Dinesh, R. Chandirasekar, K. Kovendan, U. Suresh, J. Subramaniam, M. Paulpandi, C. Vadivalagan, R. Rajaganesh, H. Wei, B. Syuhei, A. T. Aziz, M. S. Alsalhi, S. Devanesan, M. Nicoletti, A. Canale and G. Benelli, Parasitol. Res., 2016, 115, 1071–1083 CrossRef PubMed.
- H. A. E.-K. Bosly, N. Salah, S. A. Salama, R. A. Pashameah and A. Saeed, Acta Trop., 2023, 237, 106735 CrossRef CAS PubMed.
- N. Parvin, T. K. Mandal, P. C. Nagajyothi, P. M. Reddy, N. R. Reddy and S. W. Joo, J. Cluster Sci., 2021, 32, 1761–1767 CrossRef CAS.
- E. V. Minello, F. Lai, M. T. Zonchello, M. Melis, M. Russo, P. Cabras and J. Agric, Food Chem., 2005, 53, 8302–8305 CrossRef CAS PubMed.
- J. Shin, S.-M. Seo, I.-K. Park and J. Hyun, Carbohydr. Polym., 2021, 254, 117381 CrossRef CAS PubMed.
- B. L. Atkinson, A. J. Blackman, H. Faber and J. Agric, Food Chem., 2004, 52, 280–287 CrossRef CAS PubMed.
- N. Jeran, M. Grdisa, F. Varga, Z. Satovic, Z. Liber, D. Dabic and M. Biosic, Phytochem. Rev., 2021, 20, 875–905 CrossRef CAS.
- M. Fernández-Pérez, F. Flores-Céspedes, I. Daza-Fernández, F. Vidal-Peña and M. Villafranca-Sánchez, Ind. Eng. Chem. Res., 2014, 53, 13557–13564 CrossRef.
- M. Massaro, S. Pieraccini, S. Guernelli, M. L. Dindo, S. Francati, L. F. Liotta, G. C. Colletti, S. Masiero and S. Riela, Appl. Clay Sci., 2022, 230, 106719 CrossRef CAS.
- A. Kalaitzaki, N. E. Papanikolaou, F. Karamaouna, V. Dourtoglou, A. Xenakis and V. Papadimitriou, Langmuir, 2015, 31, 5722–5730 CrossRef CAS PubMed.
- N. E. Papanikolaou, A. Kalaitzaki, F. Karamaouna, A. Michaelakis, V. Papadimitriou, V. Dourtoglou and D. P. Papachristos, Environ. Sci. Pollut. Res., 2018, 25, 10243–10249 CrossRef CAS PubMed.
- Y. Zhang, W. Chen, M. Jing, S. Liu, J. Feng, H. Wu, Y. Zhou, X. Zhang and Z. Ma, Chem. Eng. J., 2019, 361, 1381–1391 CrossRef CAS.
- J. Xu, Y. He, Y. Li and Q. Ye, Mater. Lett., 2020, 260, 126968 CrossRef.
- Q.-Q. Wang, Y. Liu, C.-J. Zhang, C. Zhang and P. Zhu, Mater. Sci. Eng., C, 2019, 99, 1469–1476 CrossRef CAS PubMed.
- Y. Xiang, X. Lu, J. Yue, Y. Zhang, X. Sun, G. Zhang, D. Cai and Z. Wu, Sci. Total Environ., 2020, 722, 137811 CrossRef CAS PubMed.
- Z. Rahiminezhad, H. H. Gahruie, S. Esteghlal, G. R. Mesbahi, M.-T. Golmakani and S. M. H. Hosseini, LWT–Food Sci. Technol., 2020, 127, 109392 CrossRef CAS.
- A. Tanwar, P. Date and D. Ottoor, J. Colloid Interface Sci., 2021, 42, 100413 CrossRef CAS.
- J. Douliez, A. Perro, J. Chapel, B. Goudeau and L. Beven, Small, 2018, 14, 1803042 CrossRef PubMed.
- J. Xing, W. Dang, J. Li and J. Huang, Colloids Surf., B, 2021, 204, 111776 CrossRef CAS PubMed.
- S. Fujii, S.-I. Yusa and Y. Nakamura, Adv. Funct. Mater., 2016, 26, 7206–7223 CrossRef CAS.
- P. Aussillous and D. Quere, Nature, 2001, 411, 924–927 CrossRef CAS PubMed.
- M. Uda, S. Higashimoto, T. Hirai, Y. Nakamura and S. Fujii, Polymer, 2021, 212, 123295 CrossRef CAS.
- X. Lin, W. Ma, H. Wu, S. Cao, L. Huang, L. Chen and A. Takahara, Chem. Commun., 2016, 52, 1895–1898 RSC.
- Z. Zhao, S. Qin, D. Wang, Y. Pu, J.-X. Wang, J. Saczek, A. Harvey, C. Ling, S. Wang and J.-F. Chen, Chem. Eng. J., 2020, 391, 123478 CrossRef CAS.
- S. Yukioka, J. Fujiwara, M. Okada, S. Fujii, Y. Nakamura and S.-I. Yusa, Langmuir, 2020, 36, 6971–6976 CrossRef CAS PubMed.
- X. Luo, X. Zhang and Y. Feng, Acta Phys. Sin.-Ch. Ed., 2020, 36, 1–20 CrossRef PubMed.
- T. Boleslavská, O. Rychecký, M. Krov, P. Žvátora, O. Dammer, J. Beránek, P. Kozlík, T. Křížek, J. Hořínková, P. Ryšánek, J. Roušarová, N. K. Canová, M. Šíma, O. Slanař and F. Štěpánek, AAPS J., 2020, 22, 1–12 CrossRef PubMed.
- M. Arshadi, M. Azizi, H. Souzandeh, C. Tan, S. M. Davachi and A. Abbaspourrad, J. Mater. Chem. A, 2019, 7, 26456–26468 RSC.
- A. J. Leite, N. M. Oliveira, W. Song and J. F. Mano, Sci. Rep., 2018, 8, 15215 CrossRef PubMed.
- Y. Kim, S. Oh, H. Lee, D. Lee, M. Kim, H. S. Baek, W. S. Park, E. Kim and J.-H. Jang, Biomater. Sci., 2021, 9, 1639–1651 RSC.
- R. Yan, Q. Zhou, Z. Xu, Y. Wu, G. Zhu, M. Wang, Y. Guo, K. Dong and M. Chen, Pest Manage. Sci., 2021, 77, 3706–3712 CrossRef CAS PubMed.
- M. Beaumont, M. Bacher, M. Opietnik, W. G. Altmutter, A. Potthast and T. Rosenau, Molecules, 2018, 23, 23061427 CrossRef PubMed.
- Y. Wu, S. Jia, Y. Qing, S. Luo and M. Liu, J. Mater. Chem. A, 2016, 4, 14111–14121 RSC.
- H. Nakatani, K. Iwakura, K. Miyazaki, N. Okazaki and M. Terano, J. Appl. Polym. Sci., 2011, 119, 1732–1741 CrossRef CAS.
- E. E. Pérez, S. Domenek, N. Belgacem, C. Sillard and J. Bras, Biomacromolecules, 2014, 15, 4551–4560 CrossRef PubMed.
- B. P. Frank, D. P. Durkin, E. R. Caudill, L. Zhu, D. H. White, M. L. Curry, J. A. Pedersen and D. H. Fairbrother, ACS Appl. Nano Mater., 2018, 1, 7025–7038 CrossRef CAS.
- S. H. Ahn, M. Rath, C.-Y. Tsao, W. E. Bentley and S. R. Raghavan, ACS Appl. Mater. Interfaces, 2021, 13, 18432–18442 CrossRef CAS PubMed.
- J. Luo, Y. Gao, Y. Liu, J. Du, D.-X. Zhang, H. Cao, T. Jing, B.-X. Li and F. Liu, Nanoscale, 2021, 13, 15647–15658 RSC.
- G.-B. Li, J. Wang and X.-P. Kong, Carbohydr. Polym., 2020, 249, 116865 CrossRef CAS PubMed.
- S. Xiao, A. Shoaib, J. Xu and D. Lin, Sci. Total Environ., 2022, 831, 154914 CrossRef CAS PubMed.
- W. Menga, Z. Tiana, P. Yao, X. Fang, T. Wub, J. Cheng and A. Zoua, Colloids Surf., A, 2020, 604, 125266 CrossRef.
- Y. Ding, Z. Xiao, F. Chen, L. Yue, C. Wang, N. Fan, H. Ji and Z. Wang, Sci. Total Environ., 2023, 863, 160900 CrossRef CAS PubMed.
- L. Hao, L. Gong, L. Chen, M. Guan, H. Zhou, S. Qiu, H. Wen, H. Chen, X. Zhou and M. Akbulut, Chem. Eng. J., 2020, 396, 125233 CrossRef CAS.
- Y. Gao, Y. Liang, Z. Zhou, J. Yang, Y. Tian, J. Niu, G. Tang, J. Tang, X. Chen, Y. Li and Y. Cao, Chem. Eng. J., 2021, 422, 130143 CrossRef CAS.
Footnotes |
| † Electronic supplementary information (ESI) available: Supporting experimental procedures, supporting Figures, supporting tables. See DOI: https://doi.org/10.1039/d3gc03625a |
| ‡ These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2024 |








