 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Droplet detachment force and its relation to Young–Dupre adhesion†
Dan
Daniel
 *a and
Xue Qi
Koh
b
*a and
Xue Qi
Koh
b
aDivision of Physical Sciences and Engineering, King Abdullah University of Science and Technology (KAUST), Thuwal 23955-6900, Saudi Arabia. E-mail: danield@kaust.edu.sa
bInstitute of Materials Research and Engineering, Agency for Science, Technology and Research (A*STAR), 138634, Singapore
First published on 20th October 2023
Abstract
Droplets adhere to surfaces due to their surface tension γ and understanding the vertical force Fd required to detach the droplet is key to many technologies (e.g., inkjet printing, optimal paint formulations). Here, we predicted Fd on different surfaces by numerically solving the Young–Laplace equation. Our numerical results are consistent with previously reported results for a wide range of experimental conditions: droplets subjected to surface vs. body forces with |Fd| ranging from nano- to milli-newtons, droplet radii R ranging from tens of microns to several millimetres, and for various surfaces (micro-/nano-structured superhydrophobic vs. lubricated surfaces). Finally, we derive an analytic solution for Fd on highly hydrophobic surfaces and further show that for receding contact angle θr > 140°, the normalized Fd/πR is equivalent to the Young–Dupre work of adhesion γ(1 + cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θr).
θr).
We encounter the effects of droplet adhesion every day: dew of droplets can be seen clinging to blades of grass in early morning, and droplets stuck to camera lenses affect image clarity on rainy/misty days.1,2 Despite the prevalence of the phenomenon and clear importance in various technologies (from inkjet printing3 to agricultural technologies4,5), there is still no consensus on how to link the vertical force Fd required to detach a droplet from a surface to its wetting properties.6–9 For example, Tadmor et al. (2017) proposed that Fd/2πr (where r is the contact radius) is equivalent to the Young–Dupre work of adhesion γ(1 + cos
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θr) where γ is the surface tension and θr is the receding contact angle,10 but others disagreed.11–13
θr) where γ is the surface tension and θr is the receding contact angle,10 but others disagreed.11–13
Extensive experimental data for droplet detachment exist in the literature,10,13–17 but their interpretation is complicated by the fact that various groups use very different methods to measure Fd (Fig. 1) with very different droplet volumes (V = 5 pL–10 μL) and detachment speeds (U = 10−6–10−3 m s−1). As a result, different groups reported force magnitudes that vary considerably from 38 nN (as measured using atomic force microscopy or AFM) to 1.8 μN (force microbalance),7 and 0.36 mN (centrifugal adhesion balance or CAB).10 We will show later that the various datasets are in broad agreement with one another once Fd is normalized by the droplet radius R. The various approaches described in the literature can be broadly categorized into surface vs. body force methods—an important distinction that is often ignored. Methods such as AFM and microbalance force sensor (Fig. 1A and B) apply a pulling force only at the top surface of the droplet, i.e., surface force. In contrast, in CAB (and other methods18), the centrifugal pulling force is applied to the entire droplet, i.e., body force.
 | ||
| Fig. 1 Different methods to measure droplet detachment force Fd using (A) atomic force microscope (volume V = 65 pL, U = 2 μm s−1, superhydrophobic surface), (B) microbalance force sensor (V = 5 μL, U = 5 μm s−1, superhydrophobic surface), and (C) centrifugal adhesion balance (V = 10 μL, hydrophobic). A is original unpublished data, while B and C are taken from Daniel et al. (2023)7 and Tadmor et al. (2017),10 respectively. | ||
In this paper, we show that the quasi-static approximation applies during the retraction process for slow detachment speeds, and that Fd can be predicted by numerically solving the Young–Laplace equation.19 Our numerical results are consistent with the experimental data collected independently by various research groups. We derive an analytic solution for Fd and further show that for a droplet of radius R the normalized Fd/πR (as opposed to Fd/2πr) is equivalent to the Young–Dupre work of adhesion γ(1 + cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θr) for highly hydrophobic surfaces with θr > 140°. In contrast, for less hydrophobic surfaces with θr < 140° there is no simple way to relate Fd to the Young–Dupre work of adhesion.
θr) for highly hydrophobic surfaces with θr > 140°. In contrast, for less hydrophobic surfaces with θr < 140° there is no simple way to relate Fd to the Young–Dupre work of adhesion.
Our analysis is general and applies for a wide range of experimental conditions (including those in Fig. 1) and different surfaces (flat, micro-/nano-structured superhydrophobic,20 and lubricated surfaces21,22).
1 Methods
1.1 Surface preparation
To make the superhydrophobic surface in Fig. 5A, we started with a 3 mm thick Sylgard 184 polydimethylsiloxane (PDMS) slab. We used a 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 monomer to linker ratio which was cured by heating the mixture at 70 °C for 3 hours. We then spraycoated the cured PDMS slab with a layer of hydrophobic nanoparticles (Glaco Mirror Coat Zero, Soft 99 Co.) before placing it vertically to dry for one hour before use. Note that the wetting properties of the resulting surface is dominated by the structured nanoparticles. We chose to use a PDMS slab as the base so that we can easily puncture the surface with a 34 Gauge needle (see Section 1.1.1). Scanning electron micrograph of the resulting nanostructures can be found in Fig. 1 of our previous publication.23
1 monomer to linker ratio which was cured by heating the mixture at 70 °C for 3 hours. We then spraycoated the cured PDMS slab with a layer of hydrophobic nanoparticles (Glaco Mirror Coat Zero, Soft 99 Co.) before placing it vertically to dry for one hour before use. Note that the wetting properties of the resulting surface is dominated by the structured nanoparticles. We chose to use a PDMS slab as the base so that we can easily puncture the surface with a 34 Gauge needle (see Section 1.1.1). Scanning electron micrograph of the resulting nanostructures can be found in Fig. 1 of our previous publication.23
To make the lubricated surface in Fig. 5B, we coated a thin layer of fluorinated oil (GPL Krytox 101, ∼3 μm thick, viscosity η = 30 mPa s−1) onto the nanostructured PDMS slab.
2 Results and discussions
Previously, different groups have suggested (implicitly6 or explicitly13,19) that detachment results when the droplet is so stretched that it can no longer be described by the Young–Laplace equation. In contrast, Tadmor et al. (2017) suggest that the detachment force should be related to the Young–Dupre work of adhesion,10 while others disagreed.11–13 In this paper, we aim to reconcile these different views.We start by numerically solving the axisymmetric Young–Laplace equation for a droplet subjected to either a surface force (Fig. 2) or a body force (Fig. 3). We can recast the Young–Laplace equation into its non-dimensional form by normalizing the various quantities with V1/3 and surface tension γ (e.g., ũ = u/V1/3, Δ![[P with combining tilde]](https://www.rsc.org/images/entities/i_char_0050_0303.gif) = ΔPV1/3/γ) to give
= ΔPV1/3/γ) to give
 | (1) |
For the surface force method, we can approximate the droplet geometry as being held by a circular disc with radius a at the top and further impose the boundary condition ũ(0) = ã (Fig. 2A) and assume that the Laplace pressure Δ![[P with combining tilde]](https://www.rsc.org/images/entities/i_char_0050_0303.gif) = Δ
= Δ![[P with combining tilde]](https://www.rsc.org/images/entities/i_char_0050_0303.gif) 0 is constant (i.e., we neglect the effects of gravity since the droplets are typically smaller than the capillary length). We performed numerical simulations for different θr = 40–160° and fixed ã = 0.45 (which approximates well the geometries used in various papers14,15 including in Fig. 1A and B) and plotted the force acting on the droplet
0 is constant (i.e., we neglect the effects of gravity since the droplets are typically smaller than the capillary length). We performed numerical simulations for different θr = 40–160° and fixed ã = 0.45 (which approximates well the geometries used in various papers14,15 including in Fig. 1A and B) and plotted the force acting on the droplet
![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) = −2π = −2π![[r with combining tilde]](https://www.rsc.org/images/entities/i_char_0072_0303.gif) ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sin sin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ + π θ + π![[r with combining tilde]](https://www.rsc.org/images/entities/i_char_0072_0303.gif) 2Δ 2Δ![[P with combining tilde]](https://www.rsc.org/images/entities/i_char_0050_0303.gif) 0 0 | (2) |
We terminate the simulations when we can no longer achieve numerical convergence; this is the point of droplet detachment and we can define the corresponding detachment force ![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) d which strongly depends on θr, i.e., the surface wetting properties. For hydrophilic surfaces, the detachment force is high (
d which strongly depends on θr, i.e., the surface wetting properties. For hydrophilic surfaces, the detachment force is high (![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) d = 1.6 for θr = 40°) but can become negligible for superhydrophobic surfaces (
d = 1.6 for θr = 40°) but can become negligible for superhydrophobic surfaces (![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) d = 0.1 for θr = 160°). For hydrophilic surfaces, the droplet is highly stretched at the point of detachment and resembles more of a capillary bridge (Fig. 2C). In contrast, the droplet retains its spherical cap shape till the point of detachment for highly hydrophobic surfaces (Fig. 2D and E).
d = 0.1 for θr = 160°). For hydrophilic surfaces, the droplet is highly stretched at the point of detachment and resembles more of a capillary bridge (Fig. 2C). In contrast, the droplet retains its spherical cap shape till the point of detachment for highly hydrophobic surfaces (Fig. 2D and E).
Interestingly, the droplet experiences a maximum force ![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) max >
max > ![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) d before detaching. For hydrophilic surfaces, the two force quantities can be quite different, with
d before detaching. For hydrophilic surfaces, the two force quantities can be quite different, with ![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) max/
max/![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) d = 2.2 for θr = 40° (Fig. 2B). In contrast,
d = 2.2 for θr = 40° (Fig. 2B). In contrast, ![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) max/
max/![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) d = 1.1 for θr = 160° (inset in Fig. 2B)
d = 1.1 for θr = 160° (inset in Fig. 2B)
We now repeat the numerical simulations for a droplet subjected to an effective acceleration geff and an equivalent non-dimensional body force.
![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) = V2/3ρgeff/γ. = V2/3ρgeff/γ. | (3) |
The droplet geometry still obeys the Young–Laplace equation in eqn (1), except with different boundary conditions ũ(0) = ũ′(0) = 0 and with the Laplace pressure Δ![[P with combining tilde]](https://www.rsc.org/images/entities/i_char_0050_0303.gif) = Δ
= Δ![[P with combining tilde]](https://www.rsc.org/images/entities/i_char_0050_0303.gif) 0 −
0 − ![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif)
![[z with combining tilde]](https://www.rsc.org/images/entities/i_char_007a_0303.gif) that varies with position
that varies with position ![[z with combining tilde]](https://www.rsc.org/images/entities/i_char_007a_0303.gif) (Fig. 3A).
(Fig. 3A).
We progressively stretch the droplets by increasing ![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) and noted the decrease in base radius
and noted the decrease in base radius ![[r with combining tilde]](https://www.rsc.org/images/entities/i_char_0072_0303.gif) for different θr = 2–160° (Fig. 3B). Note that
for different θr = 2–160° (Fig. 3B). Note that ![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) ; is the input variable here, whereas it is the output variable in Fig. 2B. As is before, we terminate the simulations when there is no longer numerical convergence, and we can define the corresponding detachment force
; is the input variable here, whereas it is the output variable in Fig. 2B. As is before, we terminate the simulations when there is no longer numerical convergence, and we can define the corresponding detachment force ![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) d (but no
d (but no ![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) max). For hydrophilic surfaces, the droplet resembles a spherical cap whose base is surrounded by a wetting skirt/meniscus of size
max). For hydrophilic surfaces, the droplet resembles a spherical cap whose base is surrounded by a wetting skirt/meniscus of size  the non-dimensional capillary length (see scale bar in Fig. 3C). The wetting skirt is a low pressure region and provides the suction required to hold onto the droplet. In contrast, for highly hydrophobic surfaces, the droplet retains its spherical cap geometry (Fig. 3D and E).
the non-dimensional capillary length (see scale bar in Fig. 3C). The wetting skirt is a low pressure region and provides the suction required to hold onto the droplet. In contrast, for highly hydrophobic surfaces, the droplet retains its spherical cap geometry (Fig. 3D and E).
We can check the validity of our simulation results by superimposing experimental data collected independently by five different research groups using different methodologies ranging from AFM,14 microbalance force sensors,15,16 and CAB.10,13 When the normalized detachment force Fd/γV1/3 is plotted against ε = 1 + cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θr, all the experimental data are consistent with results from our numerical simulations for both surface and body forces (blue and red curves in Fig. 4A, respectively). We have also included results collected by us (open symbols in Fig. 4A), including previously unpublished results in Fig. 1A and 5.
θr, all the experimental data are consistent with results from our numerical simulations for both surface and body forces (blue and red curves in Fig. 4A, respectively). We have also included results collected by us (open symbols in Fig. 4A), including previously unpublished results in Fig. 1A and 5.
 | ||
Fig. 4 (A) Plot of Fd/γV1/3 as a function of (1 + cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θr). Unfilled data points are our experimental results corresponding to Fig. 1A and 5A, B and from our previous paper,14 while filled data points are results from various groups.10,13,15,16 (B) Droplet geometry during the detachment process for high θr. Raw dataset can be found in ref. 27. θr). Unfilled data points are our experimental results corresponding to Fig. 1A and 5A, B and from our previous paper,14 while filled data points are results from various groups.10,13,15,16 (B) Droplet geometry during the detachment process for high θr. Raw dataset can be found in ref. 27. | ||
 | ||
| Fig. 5 Droplet detachment due to gravity. Water droplets at the point of detachment for (A) superhydrophobic and (B) lubricated surfaces. Raw dataset can be found in ref. 27. | ||
Interestingly, the two master curves approach each other in the limit of θr > 140°. In other words, Fd measurement is independent of the method chosen for highly hydrophobic surfaces with θr > 140°. In contrast, for less hydrophobic surfaces with θr < 140°, Fd measurement values depend strongly on the method chosen (surface vs. body forces), a point that is not well appreciated in the literature.
We have tabulated the simulation results in Table S1 (ESI†): we found that ![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) d values for body and surface forces differ by 10% or less for θr > 140°. For convenience, the numerical solutions of
d values for body and surface forces differ by 10% or less for θr > 140°. For convenience, the numerical solutions of ![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) d for surface and body force methods can be fitted globally with the polynomial functions
d for surface and body force methods can be fitted globally with the polynomial functions
 | (4) |
![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) d = 1.95ε for both body and surface forces. The slope of 1.95 has a simple physical interpretation which we will explore in the next section.
d = 1.95ε for both body and surface forces. The slope of 1.95 has a simple physical interpretation which we will explore in the next section.
Relation of Fd to Young–Dupre work of adhesion
We can derive an analytic expression for Fd in terms of ε = 1 + cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θr for ε ≪ 1, i.e., in the limit of θr → 180°. According to Young–Dupre, the energy required to detach the droplet is given by ΔEγ = πr2γε, which assumes that the droplet retains its spherical cap geometry after detachment. This is a reasonable assumption for a frozen water droplet detaching from a cold surface,28 but not for liquid droplets at room temperature. In reality, the detached droplet adopts a spherical shape (Fig. 4B); using simple geometrical arguments and power series expression, we can show that ΔEγ ≈ πR2γε2. At the same time, the droplet's centroid is raised by an amount δz = z′ − z ≈ Rε (See full derivation in Fig. S1, ESI†).
θr for ε ≪ 1, i.e., in the limit of θr → 180°. According to Young–Dupre, the energy required to detach the droplet is given by ΔEγ = πr2γε, which assumes that the droplet retains its spherical cap geometry after detachment. This is a reasonable assumption for a frozen water droplet detaching from a cold surface,28 but not for liquid droplets at room temperature. In reality, the detached droplet adopts a spherical shape (Fig. 4B); using simple geometrical arguments and power series expression, we can show that ΔEγ ≈ πR2γε2. At the same time, the droplet's centroid is raised by an amount δz = z′ − z ≈ Rε (See full derivation in Fig. S1, ESI†).
Since the work done by the detachment force Fdδz must be equal to ΔEγ,
 | (5) |
For highly hydrophobic surface, the detachment force Fd when normalized by πR is therefore equivalent to γ(1 + cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θr) the Young–Dupre work of adhesion (per unit area). This result is different from the scaling proposed by Tadmor et al. (2017) where the authors suggested that Fd/2πr = γ(1 + cos
θr) the Young–Dupre work of adhesion (per unit area). This result is different from the scaling proposed by Tadmor et al. (2017) where the authors suggested that Fd/2πr = γ(1 + cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θr).10 For hydrophilic surfaces, there is no simple way to relate Fd to cos
θr).10 For hydrophilic surfaces, there is no simple way to relate Fd to cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θr since the droplet is highly deformed and its geometry depends on the choice of experimental method (compare Fig. 2C and 3C).
θr since the droplet is highly deformed and its geometry depends on the choice of experimental method (compare Fig. 2C and 3C).
The results presented here are general and apply for a wide range of experimental conditions and surface types from superhydrophobic (Fig. 5A) to lubricated surfaces (Fig. 5B). In Fig. 5, we slowly increase the droplet volume until gravity (another example of body force) causes the droplet to detach from the surface. Eqn (5) correctly predicts the droplet weight Fd = Vρg at the point of detachment for both surfaces (see red open circles in Fig. 4). Note that there is a wrapping layer of the fluorinated oil for the droplet on lubricated surface (Fig. 5B),29,30 and we used an effective surface tension γ = 69 mN m−1 when calkculating Fd/γV1/3. Strictly speaking, our approach is true only for lubricated surfaces with vanishingly small wetting ridge and when viscous dissipation due to the wetting ridge can be ignored. These two conditions were met in our experiment since we used a relatively thin lubricant layer of about 3 μm and relatively non-viscous lubricant of viscosity η = 30 mPa s.
Validity of quasi-static approximation
In our analysis so far, we have used the quasi-static approximation and assumed that the body force applied or the gap h is increased gradually such that Fd is dominated by surface wetting properties (in particular θr) and not by dynamic effects (e.g., viscous dissipation). This is true for the cases described above and also explains why different groups reported that Fd is independent of U (at least for superhydrophobic surfaces).14,15There are however cases where this assumption is not valid. For example, viscous dissipation can dominate for highly viscous or non-Newtonian liquids.31 If the speed of detachment is faster than the speed at which the contact-line can recede, dynamical effects such as pinch-off instability can occur. Some underwater superoleophobic surface also exhibits θr = 180° (i.e., no contact line pinning) and Fd is always dominated by viscous dissipation even at low U ∼ μm s−1. Previously, we showed that Fd ∝ U3/5 for an oil droplet detaching from such a surface.24,32
Maximum vs. detachment force
As discussed previously, for surface force method (but not for body force), the droplet experiences a maximum force Fmax > Fd. There is much less data available for Fmax in the literature. Nevertheless, the experimental results from Samuel et al. (2011)16 and Zhu et al. (2022)17 are consistent with our numerical results (Fig. 6). | ||
Fig. 6 Comparison between numerical results for ![[F with combining tilde]](https://www.rsc.org/images/entities/i_char_0046_0303.gif) max (dashed blue line) and experimental results from Samuel et al. (2011)16 and Zhu et al. (2022).17 Results in Fig. 4 are also superimposed (in faint) for easy comparison. max (dashed blue line) and experimental results from Samuel et al. (2011)16 and Zhu et al. (2022).17 Results in Fig. 4 are also superimposed (in faint) for easy comparison. | ||
Our numerical simulations indicate that Fmax is linearly proportional to ε, i.e.,
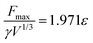 | (6) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θr) even for hydrophilic surfaces, though more experimental data is required to confirm this.
θr) even for hydrophilic surfaces, though more experimental data is required to confirm this.
Detachment vs. friction force
It is important to recognize that the lateral Ffric acting on a droplet can be very different in magnitude from the vertical Fd discussed in this paper. Furmidge proposed that the lateral friction force acting on the droplet is given by the relationFfric = πrγ(cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θr − cos θr − cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θa) θa) | (7) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θr).33 Hence, Fd/Ffric = R/r ∼ 10, i.e., it is easier to move a droplet on a superhydrophobic surface by applying a lateral force as compared to a vertical force.
θr).33 Hence, Fd/Ffric = R/r ∼ 10, i.e., it is easier to move a droplet on a superhydrophobic surface by applying a lateral force as compared to a vertical force.
More recently developed surfaces, such as lubricated21,22 and slippery covalently-attached liquid (SCAL) surfaces,34 typically exhibit θr ∼ 90°, r ∼ R, and 1 + cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θr ∼ 1, cos
θr ∼ 1, cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θr − cos
θr − cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θa ∼ 10−3.35–37 Hence, Fd/Ffric ∼ 103.
θa ∼ 10−3.35–37 Hence, Fd/Ffric ∼ 103.
3 Conclusions
Using a combination of numerical simulations and simple geometrical arguments, we were able to explain the observed detachment forces reported by various research groups spanning over a wide range of experimental parameters from micron- to millimetric-sized droplets and force magnitudes ranging from nano- to millinewtons. We also showed that for highly hydrophobic surfaces with θr > 140°, the normalized detachment force Fd/πR is equivalent to the Young–Dupre adhesion γ(1 + cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θr).
θr).
Author contributions
X. Q. K. performed the experiments here. D. D. conceptualized the project, performed numerical and theoretical analysis, supervised the research, and wrote the manuscript.Conflicts of interest
There are no conflicts to declare.Acknowledgements
We thank Sadullah and co-workers for sharing their data with us and for their valuable comments.Notes and references
- P.-G. Gennes, F. Brochard-Wyart and D. Quéré, Capillarity and wetting phenomena: drops, bubbles, pearls, waves, Springer, 2004 Search PubMed.
- D. Beysens, The physics of dew, breath figures and dropwise condensation, Springer, 2022, vol. 994 Search PubMed.
- B. Derby, Annu. Rev. Mater. Res., 2010, 40, 395–414 CrossRef CAS.
- C. Furmidge, J. Colloid Sci., 1962, 17, 309–324 CrossRef CAS.
- C. Cao, Y.-Y. Song, Z.-L. Zhou, L.-D. Cao, F.-M. Li and Q.-L. Huang, Soft Matter, 2018, 14, 8030–8035 RSC.
- H.-J. Butt, I. V. Roisman, M. Brinkmann, P. Papadopoulos, D. Vollmer and C. Semprebon, Curr. Opin. Colloid Interface Sci., 2014, 19, 343–354 CrossRef CAS.
- D. Daniel, M. Vuckovac, M. Backholm, M. Latikka, R. Karyappa, X. Q. Koh, J. V. I. Timonen, N. Tomczak and R. H. A. Ras, Commun. Phys., 2023, 6, 152 CrossRef CAS.
- Y. Jiang and C.-H. Choi, Adv. Mater. Interfaces, 2021, 8, 2001205 CrossRef.
- M. E. Schrader, Langmuir, 1995, 11, 3585–3589 CrossRef CAS.
- R. Tadmor, R. Das, S. Gulec, J. Liu, H. E. N’guessan, M. Shah, P. S. Wasnik and S. B. Yadav, Langmuir, 2017, 33, 3594–3600 CrossRef CAS PubMed.
- C. W. Extrand, Langmuir, 2017, 33, 9241–9242 CrossRef CAS PubMed.
- S. Gulec, S. Yadav, R. Das and R. Tadmor, Langmuir, 2017, 33, 13899–13901 CrossRef CAS PubMed.
- M. S. Sadullah, Y. Xu, S. Arunachalam and H. Mishra, arXiv, 2023, preprint, arXiv:2307.12527 DOI:10.48550/arXiv.2307.12527.
- D. Daniel, C. L. Lay, A. Sng, C. J. J. Lee, D. C. J. Neo, X. Y. Ling and N. Tomczak, Proc. Natl. Acad. Sci. U. S. A., 2019, 116, 25008–25012 CrossRef CAS PubMed.
- V. Liimatainen, M. Vuckovac, V. Jokinen, V. Sariola, M. J. Hokkanen, Q. Zhou and R. H. A. Ras, Nat. Commun., 2017, 8, 1798 CrossRef PubMed.
- B. Samuel, H. Zhao and K.-Y. Law, J. Phys. Chem. C, 2011, 115, 14852–14861 CrossRef CAS.
- Z. Zhu, Y. Jiang and J. W. Drelich, Langmuir, 2022, 38, 8456–8461 CrossRef CAS PubMed.
- M. Jamali, A. Moghadam, H. V. Tafreshi and B. Pourdeyhimi, Appl. Surface Sci., 2018, 456, 626–636 CrossRef CAS.
- J. F. Padday and A. R. Pitt, Philos. Trans. R. Soc., A, 1973, 275, 489–528 CrossRef.
- D. Quéré, Phys. A: Stat., 2002, 313, 32–46 CrossRef.
- T.-S. Wong, S. H. Kang, S. K. Y. Tang, E. J. Smythe, B. D. Hatton, A. Grinthal and J. Aizenberg, Nature, 2011, 477, 443–447 CrossRef CAS PubMed.
- A. Lafuma and D. Quéré, Europhys. Lett., 2011, 96, 56001 CrossRef.
- C. Thenarianto, X. Q. Koh, M. Lin, V. Jokinen and D. Daniel, Langmuir, 2023, 39, 3162–3167 CrossRef CAS PubMed.
- D. Daniel, S. S. Goh, T. N. B. Truong, X. Q. Koh and N. Tomczak, Adv. Mater. Interfaces, 2021, 8, 2001203 CrossRef CAS.
- D. Daniel, Codes for Solving Young–Laplace Equation, 2019, accessed: 2018-08-16, https://github.com/ddaniel331/laplace_solver Search PubMed.
- D. Daniel, Codes for Solving Young–Laplace Equation (Body force), 2023, accessed: 2023-08-18, https://github.com/ddaniel331/laplace_pendant Search PubMed.
- D. Daniel, Replication Data for: Droplet detachment force and its relation to Young–Dupre adhesion, 2023, accessed: 2023-08-16, https://doi.org/10.7910/DVN/3M6VQX Search PubMed.
- A. J. Meuler, J. D. Smith, K. K. Varanasi, J. M. Mabry, G. H. McKinley and R. E. Cohen, ACS Appl. Mater. Interfaces, 2010, 2, 3100–3110 CrossRef CAS PubMed.
- J. D. Smith, R. Dhiman, S. Anand, E. Reza-Garduno, R. E. Cohen, G. H. McKinley and K. K. Varanasi, Soft Matter, 2013, 9, 1772–1780 RSC.
- M. J. Kreder, D. Daniel, A. Tetreault, Z. Cao, B. Lemaire, J. V. I. Timonen and J. Aizenberg, Phys. Rev. X, 2018, 8, 031053 CAS.
- G. H. McKinley, HML Report No. 05-P-04, 2005.
- D. Daniel, A. Y. T. Chia, L. C. H. Moh, R. Liu, X. Q. Koh, X. Zhang and N. Tomczak, Commun. Phys., 2019, 2, 105 CrossRef.
- F. Schellenberger, N. Encinas, D. Vollmer and H.-J. Butt, Phys. Rev. Lett., 2016, 116, 096101 CrossRef PubMed.
- I. J. Gresham and C. Neto, Adv. Colloid Interface Sci., 2023, 102906 CrossRef CAS PubMed.
- D. Daniel, J. V. I. Timonen, R. Li, S. J. Velling and J. Aizenberg, Nat. Phys., 2017, 13, 1020–1025 Search PubMed.
- D. Daniel, J. V. I. Timonen, R. Li, S. J. Velling, M. J. Kreder, A. Tetreault and J. Aizenberg, Phys. Rev. Lett., 2018, 120, 244503 CrossRef CAS PubMed.
- A. Keiser, P. Baumli, D. Vollmer and D. Quéré, Phys. Rev. Fluids, 2020, 5, 014005 CrossRef.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d3sm01178j |
| This journal is © The Royal Society of Chemistry 2023 |



