The need for hotspot-driven research
Philip G.
Jessop
 * and
Alex R.
MacDonald
* and
Alex R.
MacDonald

Department of Chemistry, Queen's University, 90 Bader Lane, Kingston, Ontario, Canada. E-mail: jessop@queensu.ca
First published on 31st October 2023
Abstract
The entire life cycle of a product, from the obtaining of the raw materials by mining or agriculture to the end of life (recycling or disposal), consists of many steps, some of which are much more harmful than the others. Whether we're considering global warming, ecotoxicity, or resource consumption, there are typically only one or two steps that cause the vast majority of the harm of the entire life cycle. These steps, known as hotspots, are the ones that we, as green chemists and chemical engineers, should be working on. There is little benefit to be obtained by fixing a step in the life cycle that is a minor contributor to harm. However, traditional practice in the field of green chemistry has been to spend time, energy, and materials to make a selected step greener without first checking whether the selected step is the one causing most of the harm. Therefore there is a need for greater utilization of life cycle assessments (LCA) of existing processes to identify the hotspots and for that identification to be the driver for the selection of new research projects and directions. Hotspot-driven research will maximize the environmental benefit of green chemistry research.
Introduction
Because green chemistry research takes time, requires money, and causes harm, we need to prioritize our efforts. Pressing environmental crises such as global warming and the rapid extinction of many species make it vital that green chemistry research pay environmental dividends quickly. Limited funding means we cannot solve everything. The environmental harm of chemical research means that we shouldn't try to solve everything. For all of these reasons, those of us who are motivated to do green chemistry research in order to reduce environmental harm should consider how we can maximize the potential environmental benefit of our research. We should prioritize fixing products and process steps that are the biggest contributors to harm. In this perspective paper, we propose a method to aid in the conscious selection of research projects which tackle these most critical issues: hotspot-driven research.A hotspot is a chemical, a process step, or a life stage that causes more harm than the others. For example, during the life cycle of beer, more global warming is caused by the manufacturing of the glass bottle than the agriculture, beer production, transport, and waste management steps combined.1 Thus, making the bottle is the global warming hotspot. Often the same hotspot will be the greatest source of harm for many impact metrics. For example, the glass bottle for beer is not only the global warming hotspot in beer production but the hotspot for most of the impacts studied, including abiotic resource depletion, acid rain formation, human toxicity, marine aquatic toxicity, freshwater aquatic toxicity, and smog formation. It is the second greatest source of harm in all but one of the remaining impacts.
Identifying and fixing hotspots is the key to maximizing the potential environmental benefit of green chemistry research. For example, if you want to reduce the global warming caused by beer, focus on the container. Replace it with a different container or find a greener way to make glass. If, instead, you spend research time and money reducing the global warming caused by one of the other steps, such as the brewing process, the potential benefit of your work (the decrease in global warming) is likely to be much smaller.
Choosing and greening a process step without first identifying the hotspot is risky because if that step is in fact a minor contributor to environmental harm then research efforts will result in little overall improvement. For example, one company made a replacement for the popular ammonia-based window cleaning fluid, hoping that by using greener chemicals they would lower the environmental harm of window-cleaning. They didn't realize that the cleaning solution wasn't the hotspot. The plastic bottle and a paper towel (if used) are much more harmful than the fluid inside the bottle.2 Designing and selling a greener fluid made only a small difference to the environmental impact of domestic window cleaning, much less than designing a greener bottle or cloth would have.
The types of hotspots
A direct hotspot is the process step which results in the most harm from the required inputs or outputs that are attributed to that process step. For example, a distillation step may be a hotspot due to the energy requirement, or a chemical reaction may be a hotspot due to the production of a harmful by-product. One can classify direct hotspots according to different impacts. For example, a global warming hotspot is the step that causes the most global warming. If the harm being considered is social harm, rather than environmental or health impacts, then the most harmful step is a social hotspot. Social harm can include problems with workers rights, human rights, and safety.3 One can also classify direct hotspots by ranking them in terms of amount of harm. A primary hotspot is the most harmful step of a process in terms of a particular impact such as global warming. A secondary hotspot is the second most harmful.Indirect hotspots may not be hotspots themselves, but they indirectly affect either the environmental impact of the hotspot or the amount of product obtained. Consider, for example, a process in which a reaction step gives an impure product, so a complex and environmentally harmful purification step is required. While the purification step may cause the most harm and therefore be the direct hotspot, the reaction step is an indirect hotspot. Fixing the reaction to obtain the product in better purity would simplify or even eliminate the need for the purification step. Similarly, modifying the reaction to obtain the product in greater yield, without any change in purity or overall environmental harm, would increase the number of grams of product and therefore decrease the harm per gram. Thus, the improvement that should be sought for an indirect hotspot is usually an increase in yield or purity, not a decrease in the environmental harm of that step. Attempting to make the indirect hotspot greener at the cost of lower yield or purity can increase the total environmental harm. For example, a study of the extraction of phenolics from pine needles with acetone showed that the hotspot is the electricity generation for the process. The extraction step itself is an indirect hotspot. Replacing the acetone solvent with ethanol might make the extraction step greener but it makes the total harm of the process worse because it lowers the yield of extract.4 For indirect hotspots, improving the yield or purity is a higher priority than reducing the harm caused by the step.
Corporate and societal hotspots (not to be confused with social hotspots): hotspots aren't always process steps. Hotspots can also be the most impactful product or activity in a company or a society. For anthropogenic causes of global warming, the hotspot is energy production.5 In terms of usage of volatile organic solvents, the hotspot is paints and coatings.6 The biggest contributor to microplastics in the oceans is tires.7 That means, if we want to solve global warming, reduce the use of volatile organic solvents, or eliminate microplastics in the oceans, we need to prioritize our research efforts on energy production, paints & coatings, or tires, respectively. Societal hotspots, such as the largest source of microplastics in the ocean, can be found from the literature on the topic, from government databases, or by research. For example, many countries maintain publicly available databases of industrial emissions that can be searched by chemical. Canada maintains the National Pollutant Release Inventory (NPRI),8 while the US EPA website hosts the Toxics Release Inventory.9 In Europe, emissions to air are tabulated in the Emissions Database published by the EMEP Centre on Emission Inventories and Projections.10 For example, one of us (PGJ) found some years ago, by searching the NPRI, that the largest source of emissions of hexane into the atmosphere in Canada is the vegetable oil industry (Fig. 1). That was great motivation for a research project aimed at developing a nonvolatile replacement for hexane in vegetable oil extractions.
How to do hotspot driven research
We propose the following step-by-step guide to hotspot identification and research project selection using the synthesis of 2,5-furandicarboxylic acid (FDCA) from forestry residues (Scheme 1)11 and bioethanol production as examples.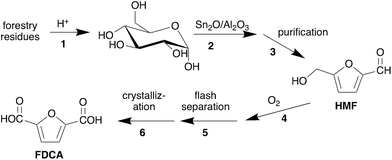 | ||
| Scheme 1 Synthesis of 2,5-furandicarboxylic acid (FDCA) from forestry residues.11 | ||
Step 1: reading life cycle data
Life cycle assessment (LCA) is a process in which the environmental harm of a process is systematically calculated and attributed to the process inputs and outputs. Through LCA data, we can identify hotspots in a lifecycle. When reading an LCA, the first step is to identify the goal, functional unit, and scope of the study. The goal might be “to identify the hotspots in the production of FDCA from forestry residues”. The functional unit quantitatively defines what process or product is being evaluated, such as “producing 1 kg of FDCA”. Both the goal and the functional unit should be clearly stated in the methods section of the study. Understanding the scope of the study is also essential to contextualizing the results. The scope of an LCA may be the entire lifespan of the product from production of raw materials to end of life (cradle to grave) or it may stop at the finished product stage and omit subsequent stages (cradle to gate). For the purposes of hotspot identification, cradle to grave is the best option when it is available. A cradle to gate LCA omits harm caused at the wholesale, retail, use and disposal stages, and therefore would miss any hotspots that occur in those life stages.Before looking at the impacts, you should also familiarize yourself with the steps of the process. A study by Bello et al.11 of the synthesis of 2,5-furandicarboxylic acid (FDCA) from hardwood forestry residues can serve as an example LCA study from which we can identify hotspots (Scheme 1). The synthesis consists of seven steps:
1. The sourcing and sulfuric acid-catalyzed hydrolysis of hardwood chips to glucose,
2. The dehydration of the glucose to 5-hydroxymethylfurfural (HMF) catalyzed by Sn2O-γAl2O3,
3. The recovery of HMF from the reaction mixture,
4. The oxidation of HMF to FDCA catalyzed by Pt/ZrO2,
5. The flash separation of FDCA from the reaction mixture,
6. The purification of the FDCA product by crystallization, and
7. The burning of natural gas and wastes from the process in a boiler for heat (not shown in the scheme).
Step 2: identifying the hotspot
An LCA will calculate the environmental harm for various impact metrics and typically include a bar graph showing the relative contributions of each process step to the impacts on the environment (Fig. 2). From this data, we can find the primary and secondary hotspots of this process by identifying the process steps responsible for the greatest amount of harm in each metric. As mentioned previously, it is often the case that one or two steps are the hotspots for most of the impacts studied. We can see from this particular study that step 3 (HMF recovery) is the hotspot for most impacts, including climate change (CC), ozone depletion (OD), terrestrial acidity (TA), marine eutrophication (ME), and photochemical oxidant or smog formation (POF). On the other hand, for freshwater ecotoxity (FE), human toxicity (HT), and freshwater and marine ecotoxicity (FET and MET), step 5 is the hotspot. For fossil fuel depletion, 3 and 5 are equal hotspots, and for agricultural land occupation (ALO), the sourcing of the biomass in step 1 is the hotspot. Thus HMF recovery is the primary hotspot in five of the eleven metrics and is secondary in three more. The flash separation is primary in four metrics and secondary in five. Therefore, greening either of these steps will likely negate much more harm than greening any of the other steps. | ||
| Fig. 2 Relative contributions of the seven process steps for the synthesis of 2,5-furandicarboxylic acid (FDCA) from forestry residues11 (reproduced from ref. 11 with permission from de Gruyter, copyright 2018). | ||
It is not necessary, in many cases, to decide whether one impact such as global warning is more important than another. Such subjective decisions are often made unnecessary by the fact that the hotspot for one impact is usually the hotspot for several other impacts as well. That is true in this example, where steps 3 and 5 are hotspots for many impacts. A step that is a hotspot for several impacts is inarguably worth improving, regardless of how we feel about the relative importance of the various impacts.
Step 3: evaluating the reliability of hotspot identification
Researchers may hesitate to devote resources to greening a hotspot that has been identified in only a single LCA study. This scepticism is warranted, as the numerical results of LCA studies tend to vary significantly even between studies with identical functional units. That's normal because each LCA is a snapshot of the situation in a single country, in a single year, with a specific set of assumptions. Another LCA that considered another country, was performed with data for a different year, or made different assumptions, would quite reasonably be expected to have quantitatively different results. Fortunately, for hotspot analysis the numerical results are irrelevant; the only result that is important is the identification of the most harmful step. Hotspot identification is less sensitive to country, year, and assumptions than numerical results. Nevertheless, there is still a risk that the hotspot may be misidentified in a study or may differ from study to study.To check the validity of their study, LCA authors perform sensitivity analysis. That procedure tests the study's data, assumptions, and models to determine the degree to which these factors affect the final answer. After determining the hotspot for a process, the authors will vary key variables or assumptions to see if that affects which step is the hotspot. For example, if the original calculation assumed that the biomass had to be trucked 100 km from the farm to the biorefinery, then the authors might change that distance to 20 km or 500 km to see whether those changes cause a different process step to become the hotspot. If the hotspot stays the same regardless of changes to key variables, then we can be confident that it is the true hotspot within the error limits of that study. Sensitivity analysis has limitations, however. It will not identify fundamental flaws in an LCA. If the LCA neglected some key factors, impacts, inputs, or outputs, then the hotspot may be incorrectly identified even if sensitivity analysis has been done correctly.
The bioethanol LCA literature can be used to illustrate a second method of testing the reliabiiity of hotspot identification: inter-study comparisons. If two or more LCA studies have been published on the same topic, then the reader can check to see if the studies have identified the same hotspots. For example, the LCA literature on bioethanol has tremendous variation from study to study, both in the conversion processes being studied and in the LCA methodology. Several biomass sources have been investigated as bioethanol feedstocks, though individual LCA studies typically focus on 1–3 feedstocks. Different studies also change the functional unit, system boundary, data source, LCA software, and impact assessment method. Despite these differences, the major hotspots are consistently identified as the post-fermentation distillation of ethanol from water and the agricultural production of the biomass.12–16 Therefore, we can be confident that these process steps are the ones that need our attention most and finding solutions to these problems will result in the greatest environmental return on investment. Comparing hotspot identification in different studies eliminates the uncertainty surrounding the methods or assumptions of any single study.
Step 4: greening the hotspot
Now that we've identified a hotspot, what do we do next? An LCA study will also contain a “life cycle inventory” which is a list of a system's input and output flows. We can consult the life cycle inventory to determine why the hotspot process steps are so harmful. For step 3 (HMF recovery), we find that large amounts of DCM are used, which causes the step to be harmful across several impacts. For step 5 (FDCA separation), the cause is excessive energy consumption. Consider ways you could reduce the harm of these steps. If you were to launch a research project based on this LCA, finding an alternative to DCM would be a good start.The best way to fix a hotspot is not necessarily by improving that step; it could be more effective to replace that step with a completely different technology. For example, let us consider the process of converting biomass to ethanol by fermentation. The two major hotspots of bioethanol production are the post-fermentation distillation of ethanol from water and the agricultural production of biomass.12–16 The distillation hotspot could be greened by improving the energy efficiency of the distillation, but as chemists we may find that to be outside our area of expertise or find that the process step has already been highly optimized to a point where further improvement appears unlikely. Green chemists instead have unique skills and knowledge which empower them to consider possible replacements for a problematic process step. Is distillation truly necessary, or can we identify greener alternatives for the separation of ethanol from water? This question could be the basis for a research project inspired by hotspots and informed by life cycle data.
Modifying an indirect hotspot is another possible strategy to lowering the overall environmental harm. Continuing with the example of bioethanol, one could instead choose to try and indirectly solve the distillation hotspot by improving the yield of the fermentation step. An increase in fermentation yield that does not affect the amount of harm caused by the overall process would cause a decrease in the harm per gram of product.
Step 5: evaluating the environmental effect of a process change
Finally, once a hotspot step has been identified and a greener replacement has been developed, it is critical to realize that the harm of the overall process may have actually increased, even though the hotspot itself is now greener. For example, if a synthesis step is altered to avoid the use of a harmful solvent, but that change necessitates a more difficult and harmful purification step, then the overall process may now cause more harm than before. Changes to a step that lead to changes in other steps should be evaluated based on their effect on the entire process, not just the hotspot. While we naturally tend to think in a reductionist approach, that each step is independent of the others, that approach is rarely valid. Every step affects other steps to some extent. This is the core concept within the approach known as systems thinking.17Modifications to a process or product, no matter how well-intentioned, may also create a new risk that wasn't a problem before. This is known as “risk migration”18 or, in toxicology, “regrettable substitutions”.19 A classic example is the replacement of tetrachloroethylene with hexane as the solvent in brake cleanser spray products. This substitution was made in order to avoid environmental harm associated with chlorine contamination of recovered waste oils. The hexane brake cleanser products were later changed to a 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 hexane/acetone mix because acetone, unlike hexane, doesn't contribute to ground-level ozone (smog). These changes, while well-intentioned, introduced a new risk; hexane/acetone mixes are highly neurotoxic. The new products led to vehicle repair shop workers suffering peripheral neuropathy, including the inability to control or even feel their limbs.20 Risk migration is an ever-present possibility that threatens to undermine the well-intentioned work of green chemists. Any modification to a product or process will come with some environmental burden. It remains the responsibility of green chemists to identify potential environmental harm that may be caused by a replacement technology. Advances in toxicology information, LCA software, and other methods of harm prediction have greatly improved our ability to anticipate such harm and should always be considered after a replacement process or product has been identified.
50 hexane/acetone mix because acetone, unlike hexane, doesn't contribute to ground-level ozone (smog). These changes, while well-intentioned, introduced a new risk; hexane/acetone mixes are highly neurotoxic. The new products led to vehicle repair shop workers suffering peripheral neuropathy, including the inability to control or even feel their limbs.20 Risk migration is an ever-present possibility that threatens to undermine the well-intentioned work of green chemists. Any modification to a product or process will come with some environmental burden. It remains the responsibility of green chemists to identify potential environmental harm that may be caused by a replacement technology. Advances in toxicology information, LCA software, and other methods of harm prediction have greatly improved our ability to anticipate such harm and should always be considered after a replacement process or product has been identified.
Is there value in greening a non-hotspot?
Is there any value to trying to green a step which is not a hotspot? Of course the answer is yes. All steps in a process cause harm, and reducing harm is the goal of green chemistry. However, the direct benefits to the environment is more limited if one greens a step which isn't a hotspot, simply because of the smaller harm related to that step.The bar graph in Fig. 2 gives us an estimate of the maximum reduction in harm that could be expected upon greening any particular step, neglecting effects on other steps. For example, the maximum reduction in global warming that one could obtain by greening the hotspot (step 3) is about 50% of the total for the entire process. Any attempt to green a step that is not a hotspot would result in less benefit. For example, attempts to reduce the carbon footprint of the HMF synthesis (step 2) would, at best, result in about a 4% reduction in the total global warming contribution. That's still a reduction in harm and is therefore worth doing, but because it is smaller it is not worth prioritizing.
On the other hand, creative solutions to green non-hotspots may serve as inspirations for other researchers to fix problematic steps in other processes. A new technology that makes little difference to one process may make an enormous difference when applied to a different process.
Additionally, the identification of a hotspot may change over time. If someone develops a much greener replacement for the current hotspot, then that step may be so improved that it is no longer the hotspot. In that case, the secondary hotspot will become the primary hotspot and all other steps will represent a greater fraction of the total harm. Thus past or future efforts to improve non-hotspots will have greater relative value after the actual hotspot has been greened.
Conclusions
While LCA experts, software, and published studies were rare in the early years of green chemistry, they are becoming more available. Thus while hotspot-driven research may have been more difficult in the past, it is now feasible and starting to be practiced.21–24 Adopting this approach should increase the environmental benefit of green chemistry and engineering research.Those researchers in green chemistry and engineering who are performing applied research with the objective of reducing environmental harm would be well advised to identify hotspots in existing processes or societies and focus their research efforts on greening those hotspots. Greening a step in a process, without checking whether it is a hotspot, may still lead to environmental harm reduction but the benefit of green chemistry research will be greater if we direct our efforts towards hotspots.
An exception to this strategy could be indirect hotspots, where, despite the low environmental impact of the step, efforts to improve the yield or purity of the step could result in significant reduction in the overall impacts of the process.
Author contributions
P. G. J.: conceptualization, project administration, investigation, supervision, writing – original draft, Writing – review and editing. A. R. M.: investigation, writing – original draft, writing – review and editing.Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors gratefully acknowledge funding from the Natural Sciences and Engineering Research Council of Canada (NSERC, grant RGPIN-2023-05700) and the Canada Research Chairs Program.References
- D. Amienyo and A. Azapagic, Int. J. Life Cycle Assess., 2016, 21, 492–509 CrossRef CAS.
- E. J. Beckman , personal communication.
- C. B. Norris, G. A. Norris and D. Aulisio, Sustainability, 2014, 6, 6973–6984 CrossRef.
- M. S. de Luna, G. Vetrone, S. Viggiano, L. Panzella, A. Marotta, G. Filippone and V. Ambrogi, ACS Sustainable Chem. Eng., 2023, 11, 4670–4677 CrossRef.
- H. Ritchie and M. Roser, Emissions by sector, https://ourworldindata.org/emissions-by-sector, (accessed 18 September 2023, 2023).
- ESIG Newsletter “Solutions”, Spring 1997, as reported by J. H. Clark, et al. , Int. J. Mol. Sci., 2015, 16, 17101 CrossRef CAS PubMed.
- P. Sundt, P.-E. Schulze and F. Syversen, Sources of microplastic- pollution to the marine environment, Report 1032, mepex, Norwegian Environment Agency, 2014 Search PubMed.
- National Pollutant Release Inventory, https://www.canada.ca/en/services/environment/pollution-waste-management/national-pollutant-release-inventory.html).
- Toxics Release Inventory (TRI) Program, 2023, https://www.epa.gov/toxics-release-inventory-tri-program.
- EMEP Centre on Emission Inventories and Projections, Convention on Long-range Transboundary Air Pollution, https://www.ceip.at, 2023.
- S. Bello, I. Salim, P. Méndez-Trelles, E. Rodil, G. Feijoo and M. T. Moreira, Holzforschung, 2019, 73, 105–115 CrossRef CAS.
- E. I. Wiloso, R. Heijungs and G. R. D. Snoo, Renewable Sustainable Energy Rev., 2012, 16, 5295–5308 CrossRef CAS.
- I. Tsiropoulos, A. P. C. Faaij, J. E. A. Seabra, L. Lundquist, U. Schenker, J.-F. O. Briois and M. K. Patel, Int. J. Life Cycle Assess., 2014, 19, 1049–1067 CrossRef CAS.
- S. Papong, C. Rewlay-ngoen, N. Itsubo and P. Malakul, J. Cleaner Prod., 2017, 157, 254–266 CrossRef.
- M. M. Parascanu, N. Sanchez, F. Sandoval-Salas, C. M. Carreto, G. Soreanu and L. Sanchez-Silva, Environ. Sci. Pollut. Res., 2021, 28, 64374–64393 CrossRef CAS PubMed.
- T. S. Angili, K. Grzesik, A. Rödl and M. Kaltschmitt, Energies, 2021, 14, 2939 CrossRef CAS.
- N. G. Leveson, Engineering a Safer World: Systems Thinking Applied to Safety, MIT Press, Cambridge, Massachusetts, 2012 Search PubMed.
- R. E. Alcock and J. Busby, Risk Anal., 2006, 26, 369–381 CrossRef PubMed.
- A. Maertens, E. Golden and T. Hartung, ACS Sustainable Chem. Eng., 2021, 9, 7749–7758 CrossRef CAS PubMed.
- M. P. Wilson, n-Hexane Exposure in the California Vehicle Repair Industry: Risk Assessment and Policy Analysis, PhD thesis, University of California, Berkeley, 2003 Search PubMed.
- B. Subramaniam, R. K. Helling and C. J. Bode, ACS Sustainable Chem. Eng., 2016, 4, 5859–5865 CrossRef CAS.
- X. Zhang, L. Zhang, K. Y. Fung, B. R. Bakshi and K. M. Ng, Chem. Eng. Sci., 2020, 217, 115508 CrossRef CAS.
- T. M. Hoogstad, S. M. Timmer, A. J. B. V. Boxtel, P. L. Buwalda, J. H. Bitter and L. Kiewidt, ChemistryOpen, 2022, 11, e202200029 CrossRef CAS PubMed.
- S. M. Heidari, E. Lee, B. Cecil and A. Anctil, Green Chem., 2023, 25, 4350–4361 RSC.
| This journal is © The Royal Society of Chemistry 2023 |

