Co-adsorption of tetracycline and Cu(ii) onto a novel amino-functionalized biochar: adsorption behavior and mechanism
Abstract
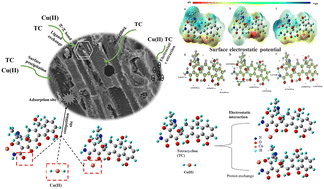
In this work, a novel amino-functionalized biochar (NS-700) was prepared successfully by pretreating biomass with (NH4)2S2O8, and could be used for the simultaneous and efficient removal of tetracycline (TC) and Cu(II). NS-700 was analyzed by Fourier-transform infrared spectroscopy (FTIR), elemental analysis, X-ray photoelectron spectroscopy (XPS) and other characterization techniques. Results showed that NS-700 contained a variety of oxygen-containing and nitrogen-containing functional groups, as well as a more developed pore structure. The specific surface area of NS-700 was up to 739.9 m2 g−1. Through the effects of changing the solution pH, adsorption kinetics and isotherm model arguments, the adsorption behavior and performance of NS-700 for TC and Cu(II) in different systems were investigated. The maximum adsorption capacity of NS-700 for Cu(II) and TC was 49.07 mg g−1 and 323.26 mg g−1, respectively. The existence of TC could slightly reduce the adsorption capacity for Cu(II). A low concentration of Cu(II) could increase the adsorption of TC, but a high Cu(II) concentration inhibited the TC adsorption on NS-700. Combined with theoretical calculations, the adsorption mechanism of NS-700 in different systems was explored. The results demonstrated that the amide groups on NS-700 were combined with TC through complexation, and the protonated amino groups could be ion-exchanged with Cu(II). Cu(II) could interact with O22–O26 or O22–N25 on TC to form a complex, and the complexation caused by the interaction of O22–N25 on TC and Cu(II) played an important role in the adsorption of Cu(II) and TC by NS-700. This study puts forward a guideline and practical method for the application of biochar in the pollution control of heavy metals and antibiotics.


 Please wait while we load your content...
Please wait while we load your content...