Metal-free recycling of waste polyethylene terephthalate mediated by TBD protic ionic salts: the crucial role of anionic ligands†
Abstract
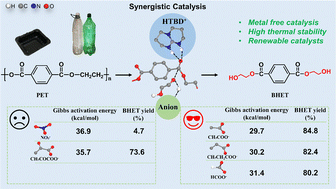
The structure–activity relationships of 1,5,7-triazabicyclo[4.4.0]dec-5-ene (TBD) based protic ionic salts for polyethylene terephthalate (PET) glycolysis by ethylene glycol (EG) were comprehensively investigated through theoretical prediction and experimental verification. The proton capture ability of the anionic ligand from EG is positively correlated with the activity of the catalyst, as the generation of EG− was crucial for the chain breaking reaction via nucleophilic attack on the carbonyl group. Furthermore, density functional theory calculations demonstrated that the HTBD cation and anionic ligands work in a cooperative manner in the PET glycolysis reaction, where the ligands abstract a proton from EG to generate EG− and provide a proton to produce the bis(hydroxyalkyl)terephthalate (BHET) product. The rate-determining step is the nucleophilic attack step, where the Gibbs energy barriers (ΔG≠) increase in the order of 29.7 kcal mol−1 (HTBD-OAc) < 30.2 kcal mol−1 (HTBD-CH3CH2COO) < 31.4 kcal mol−1 (HTBD-HCOO) < 35.7 kcal mol−1 (HTBD-CH3COCOO) < 36.9 kcal mol−1 (HTBD-NO3). This is confirmed from the experimental results that the BHET yields decrease in the order of 84.8% (HTBD-OAc) > 82.4% (HTBD-CH3CH2COO) > 80.2% (HTBD-HCOO) > 73.6% (HTBD-CH3COCOO) > 4.7% (HTBD-NO3). These findings offer valuable guidance for designing more efficient metal-free protic ionic salts, promoting sustainable PET recycling.
- This article is part of the themed collection: 2023 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...