Supramolecular biomaterials for enhanced cancer immunotherapy
Abstract
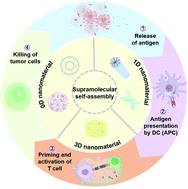
Cancer immunotherapy has achieved promising clinical results. However, many limitations associated with current cancer immunotherapy still exist, including low response rates and severe adverse effects in patients. Engineering biomaterials for the delivery of immunotherapeutic reagents has been suggested to be an effective strategy to improve cancer immunotherapy. Among different biomaterials, supramolecular biomaterials with flexible and versatile structures and functions have exhibited unparalleled advantages in promoting cancer immunotherapy. In recent years, various supramolecular formulations have been extensively explored as immunotherapeutic delivery platforms due to their high cargo-loading capacity/feasibility, facile immunization function, and excellent biocompatibility, which make them possible candidates for modular and personalized cancer immunotherapy. These nanoarchitectures with unique topologies possess distinguishing advantages in cancer immunotherapy, incarnating a structure–property relationship. Based on extensive state-of-the-art research, this minireview highlights recent advances in supramolecular biomaterials for cancer immunotherapy and discusses the possible mechanisms underlying how supramolecular biomaterials promote the development of cancer immunotherapy together with their potential for clinical translation.
- This article is part of the themed collection: Journal of Materials Chemistry B Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...