WO2 nanoparticles with oxygen vacancies: a high-efficiency electrocatalyst for the conversion of nitrite to ammonia†
Abstract
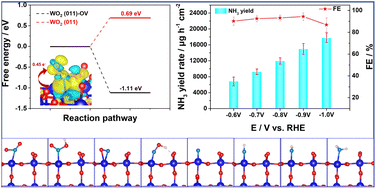
Electrocatalytic reduction of nitrite (NO2−) is considered an effective method to solve its environmental pollution issue as well as sustainable ammonia (NH3) synthesis under ambient conditions but still requires high-performance catalysts. Herein, self-supported monoclinic WO2 nanoparticles with abundant oxygen vacancies (OVs) on a tungsten plate (WO2/W) are demonstrated as a highly efficient electrocatalyst for selectively reducing NO2− to NH3. In 0.1 M NaOH solution containing 0.1 M NO2−, such an electrocatalyst achieves an excellent yield of 14 964.25 ± 826.06 μg h−1 cm−2 and high faradaic efficiency (FE) of 94.32 ± 1.15% at −0.9 V. Remarkably, the assembled Zn–NO2− battery delivers a high power density of 5.05 mW cm−2 and simultaneously offers an abundant NH3 yield of 1923.88 μg h−1 cm−2. Theoretical calculations reveal the critical role of OVs for WO2 in the nitrite electrocatalytic reduction process and its possible reaction pathway.
- This article is part of the themed collection: Journal of Materials Chemistry A HOT Papers


 Please wait while we load your content...
Please wait while we load your content...