“Every little thing that could possibly be provided helps”: analysis of online first-year chemistry resources using the universal design for learning framework
Charisse T.
Reyes
 ab,
Gwendolyn A.
Lawrie
ab,
Gwendolyn A.
Lawrie
 c,
Christopher D.
Thompson
c,
Christopher D.
Thompson
 a and
Sara H.
Kyne
a and
Sara H.
Kyne
 *a
*a
aSchool of Chemistry, Faculty of Science, Monash University, Clayton, VIC 3800, Australia. E-mail: sara.kyne@monash.edu
bFaculty of Education, University of the Philippines Open University, Los Baños, Laguna 4031, The Philippines
cSchool of Chemistry and Molecular Biosciences, The University of Queensland, Brisbane City, QLD 4072, Australia
First published on 22nd December 2021
Abstract
Rapid advancements in information and communication technologies (ICTs) have afforded numerous variations to traditional chemistry curricula where pedagogical strategies that have been employed have assumed “one-size-fits-all”. The translation of print-based instructional resources into multimodal online and digital forms enables greater accessibility, flexibility, and usability to support students in their understanding of complex chemistry concepts. To ideally offer an online learning environment that is accessible by all students to the greatest extent possible, this study employed the principles of the Universal Design for Learning (UDL) framework to analyse the design of online instructional resources for topics in first-year chemistry courses. Through application of UDL principles, students were provided with multiple means of representation of concepts, options for action and expression, and various avenues for engagement within the learning management system (LMS). This paper describes how the UDL framework was used to evaluate three separate independent Cases of tertiary first-year chemistry courses, including one university in the Philippines and two universities in Australia. Evaluation through surveys, focus groups, and individual interviews revealed students’ perceptions of the usefulness of the UDL-based features. Students cited benefit from multiple forms of content delivery, animations, interactive simulations, and video recordings because they facilitated processing of information, provided alternative ways of presenting the information, allowed for varying methods for response, navigation, and flexibility, and allowed for self-evaluation of their progress. These results suggest that applying the principles of the UDL framework in instructional design of an online environment in first-year chemistry courses can support and further enhance students’ learning irrespective of their individual contexts.
Introduction
Access to quality higher education and bridging the educational gap is an important aspect of social justice, and remains a persistent and challenging goal to achieve. Driven by an increasing diversity in learners, educators have explored ways to address the educational gap by providing learners with equitable access to quality education. In principle, all students should experience equitable access and opportunities for learning. Raycroft and Flynn (2020) argue that equity in education is a “democratic ideal”, and whilst requiring considerable attention, it results in superior learning outcomes. The Universal Design for Learning (UDL) framework is among many approaches that promote equity through the design of classrooms that intentionally incorporate strategies supporting marginalised students, at the same time benefiting all other students (Pilgrim and Ward, 2017). Through application of this framework, previous studies have demonstrated potential for learning to be improved by providing greater opportunities and access for a wider range of learners (Kumar and Wideman, 2014; Al-Azawei et al., 2016).The use of digital technology for instructional delivery should not only be engaging, but more importantly, should consider the individual learning needs of students and make content accessible to all types of learners, across multiple platforms. Kumar and Wideman (2014) argue students need to be able to access and comprehend learning materials, and must be offered accessible means to express their learning. An accessible learning environment incorporates easy-to-use learning materials and resources by removing or reducing the barriers to engaging with content and activities. Increasingly digital technologies are being used to design courses and teaching strategies to meet the needs of students from a variety of backgrounds, abilities and learning preferences (COU, 2020).
In chemistry education, learning involves accessing multiple representations of phenomena at different levels of thinking because it is not possible to observe atoms or molecules directly in most learning environments. The promotion of inclusive practices in teaching chemistry has benefited learning for students with disabilities (Mumba et al., 2015) through the use of multiple representations (i.e. single or multiple, multimodal, external representations). Adaptation of resources to increase accessibility is particularly important to include students with visual impairments (Ferreira and Lawrie, 2019; Teke and Sozbilir, 2019). Inclusive practices in chemistry have likewise facilitated the inclusion of gender and ethnicity-based learning materials in a multicultural student cohort (Bianchini et al., 2002) and the reduction of equity gaps in minoritised groups through active learning strategies in chemistry (White et al., 2021). Rather than attending to the needs of a particular group of students, applying the UDL framework enables inclusive practices that favour the shift of the “burden of adaptability from the shoulders of the students to the learning environment” (Rose and Meyer, 2002, as cited in Kumar and Wideman, 2014), resulting in wider accessibility for diverse students. This is because UDL advocates for changes in course design and delivery to ensure greater flexibility to benefit a broader representation of students. Whilst the UDL framework offers potential to achieve greater inclusivity amongst students, published studies employing this framework in chemistry education remain sparse particularly in the context of online learning environments.
Prior studies have explored instructors’ selection of various representations to address the challenge of providing students with means to facilitate an accurate and deeper understanding of complex chemistry topics (Ferreira and Lawrie, 2019; Robinson, 2000; Smiar and Mendez, 2016). Representations, as an integral aspect of chemistry, are utilised as a form of language to communicate highly abstract concepts of chemical structures such as the connectivity and three-dimensional arrangement of atoms. For a novice learner, representations play a key role in facilitating their shift from the macroscopic world of matter to operating in the sub-microscopic, microscopic, and symbolic level. Unlike expert learners, novice learners who have low prior knowledge of chemistry concepts frequently require scaffolded transitions between macroscopic and molecular representations (i.e. sub-microscopic, microscopic, and symbolic representations) because they experience difficulty linking these different levels of representations (Cook et al., 2008).
Rau (2017) further posits that students benefit from visual representations by acquiring “representational competencies,” for depicting content information. However, acquiring “representational competence in parallel with constructing conceptual understanding” (Lawrie and Bartle, 2013) remains a challenging aspect of teaching and learning chemistry, especially in a first-year course which delivers a broad range of introductory chemistry concepts in one semester. Whilst chemistry education researchers have developed understanding of how students engage with representations in traditional classrooms, this is less well understood in online environments.
Through the adoption of current technological affordances and online learning capabilities that are available to instructors, the UDL framework lends itself to addressing challenges in teaching first-year chemistry. The strategic adoption of curated multimedia and multimodal resources in online learning of chemistry that links multiple levels of macroscopic, sub-microscopic, microscopic, and symbolic representations (Mindayula and Sutrisno, 2021) has been employed in chemistry teaching. The application of UDL principles has the potential to bridge the gap across varying levels of understanding of chemistry and facilitate a more integrated understanding for each student.
Universal design for learning as a pedagogical framework
Advancements in technology have afforded numerous alternatives to traditional curricula and learning materials; historically most have been provided in print form only. As computers have become more accessible, and assistive technologies more common in schools, educators have adapted hardcopy resources into digital formats resulting in greater flexibility and usability. Given these affordances, King-Sears (2009) recommended the need to understand how technology can be used in conjunction with pedagogy to provide a universally designed instruction that is responsive to the needs of individual learners. Intentional application of universal design as part of instruction ensures that the learning environment is accessible by all students “to the greatest extent possible, without the need for adaptation or specialized design” (Burgstahler, 2020).The Center for Applied Special Technology (CAST) was co-founded by David Rose and Ann Meyer in 1984. This group began to define and extend the principles of universal design to the learning environment, culminating in the UDL framework that was widely disseminated in the 1990s. Almost three decades later, the global academic community has been actively implementing and evaluating the UDL framework across all levels of education. The application of the UDL framework generally aims to minimise barriers in order to maximise learning by creating a curriculum that is usable and can be understood by diverse learners regardless of their backgrounds, needs, interests, and goals (CAST, 2018). To achieve this, flexible pathways are offered to learners so they can progress in a way that best suits their individual needs. This framework is divided into two layers, namely the conceptual layer (comprised of three networks) and the implementation layer (comprised of three principles). The UDL framework helps us to understand how students learn through the three-network model of learning – recognition, strategic and affective networks, and how instructors can support students in their learning following the three principles of providing them with multiple means of representation, action and expression, and engagement. The UDL framework is operationalised through its 31 checkpoints as provided on the CAST website (https://www.cast.org/).
UDL strategies aim to develop educational environments that are “seamlessly and inherently functional” and they guide the integration of inquiry with teaching methods and learning technologies that support diverse learning needs (Curry, 2003; Curry et al., 2006). Furthermore, application of the UDL framework supports educational sectors in their shift from traditional education models to approaches that cater to the diversity that learners bring. The framework emphasises equitable access to the curriculum for all students. Hence, it provides guidance to the design of learning goals, materials, methods, and assessment in order to accommodate learner diversity.
UDL in the context of chemistry education
The application of UDL principles to create content, media, and adopt instructional technologies that are adaptive, customisable, flexible, usable, and accessible by the student may benefit science education in three distinct ways. These include enhancing student engagement and increased usability of science curricular materials (Basham and Marino, 2013), improving science education and increased inclusion among learners with disabilities (Watt et al., 2013), and improving student outcomes (Rappolt-Schlichtmann et al., 2013). Curry et al. (2006) suggested various means of incorporating inquiry-based learning with UDL in science courses by allowing learners greater access to content media, learner-centred instructional technologies, and to laboratory and field equipment. This instructional design lends itself to chemistry education in particular as a subdiscipline of science education.The American Chemical Society's (ACS) commitment to the removal of barriers that may deter students from undertaking chemistry programs and other careers in science (ACS, 2012) has led to the promotion of UDL in chemistry classrooms in the United States of America through several book publications and guidelines for UDL implementation. There have been exploratory studies in the high school chemistry context, for example King-Sears et al. (2015) reported receiving positive feedback from operationalising the principles of UDL in teaching high school chemistry students with and without disabilities using a workbook alongside a video clip, a unit organiser, and a mnemonic to solve mole concept problems. King-Sears and Johnson (2020) further suggested that teaching based on the UDL framework is important for students learning complex concepts including chemistry. The study of Baumann and Melle (2019) presented a significant increase in the performance of secondary school students with and without special educational needs in learning chemical reactions using software developed based on the UDL framework. Scanlon et al. (2018) investigated how three general chemistry curricula supported variations in learners’ abilities, interests, and needs through the lens of the 31 UDL checkpoints. Farrell and Seery (2016) suggested further strategies to implement UDL for meeting the needs of learners for language and literacy skills in chemistry, such as providing lesson overviews and opportunities to make links between students’ prior learning and the new lesson. However, studies evaluating the implementation of all three principles of the UDL (i.e. providing multiple means of representation, action and expression, and engagement) in online first-year chemistry have not been reported to date (Seok et al., 2018; Schreffler et al., 2019).
In this study, the implementation of the UDL framework to inform the design and evaluation of first-year chemistry online learning activities has been investigated through students’ perceptions of their learning experiences. The following questions guided this research:
1. How can UDL checkpoints be incorporated in the design and evaluation of an online first-year chemistry course?
2. What aspects of the student learning experience do students perceive as supportive based on the UDL-based features of the first-year chemistry course?
Research method
This study describes an exploratory multiple-case design with each of three Cases implementing the UDL framework in first-year chemistry courses from two universities in Australia and one in the Philippines. Due to the significant contextual variation across the three Cases, they are not directly compared in this study. Rather, the use of three cases permits broader examination of student experiences to develop deeper insight (Mill et al., 2012; Yin, 2013) and stronger support (Yin, 2013) of how UDL-based instructional design impacts the learning experiences of students in varied contexts (Baxter and Jack, 2008). This multiple-case study approach aims to “expand the generalizability of [our] findings to a broader array of contexts” (Yin, 2013) facilitating a wider exploration of how UDL can be implemented in different chemistry environments.Ethics approval was gained from respective ethics committees of each of the two participating universities in Australia. Due to the absence of an ethical research committee in the participating university from the Philippines, an approval to conduct the study was secured from the highest official of the university. Informed consent to participate was collected from participants through the survey and in interviews. All data were de-identified prior to the statistical analysis.
UDL of first-year chemistry topics in online environments
A guiding framework (Fig. 1) was employed for this multiple-case study that involved design, development, and evaluation of online learning materials with UDL-based features in first-year chemistry courses in the three participating universities as described in each of the separate Cases. UDL guidelines were enacted or reinforced in the online learning materials for five common topics in first-year chemistry courses (referred to as general chemistry in other contexts). These five topics are a subset of each whole course but are taught in a common sequence, namely, periodic table and trends, chemical bonding, Lewis structures, molecular shape, and polarity. In all three Cases, these five topics were regarded as foundational concepts delivered within the first five weeks of the semester.Survey of student perceptions of the impact of online learning materials with UDL-based features
A survey instrument (Appendix 1) was developed by the primary researcher (CR), informed by UDL literature, and administered in whole in Case 1 and Case 2. The instrument developed for this study was subjected to face and construct validity checks by a science education expert who was not a member of the research team. The reliability of the survey was evaluated through Cronbach's alpha (IBM SPSS 25.0) using pilot test data (Romero Martínez et al., 2020) with a sample size of 70 students yielding a value of α = 0.752. This is considered to be an acceptable level of internal consistency and hence reliability (Taber, 2018). A subset of items from this instrument (Appendix 1) were embedded into an existing evaluation instrument which is administered annually in Case 3 since overlapping questions existed. To determine the accuracy of each of the survey measurements, the margin of error (MOE) in the survey results for each Case was determined (Bartlett II et al., 2001; Taherdoost, 2017).In addition to students’ demographic information, previous secondary school background, and academic programs, surveys administered in Case 1 and Case 2 included questions on the perceived usefulness of the identified UDL-based features of the online learning materials.
Focus groups and individual interviews
At the end of the survey administered in each Case, respondents were asked to register their interest to voluntarily participate in focus groups or virtual interviews to elaborate on their survey responses. Volunteer participants were sent an email to choose their preferred date and time for the focus group/interview. Volunteers for the virtual interviews were offered to select their preferred online platform. A final email was sent to the participants to confirm the schedule and the venue/platform for the focus group/interview.In-person focus groups were conducted in Case 1 in 2019 to take advantage of the “synergy of the group interaction” allowing group members to feel comfortable with each other and to engage in the discussion (Rabiee, 2004). The focus groups were conducted in a small classroom within the chemistry building.
In 2020, in-person focus groups for Case 2 and Case 3 were not feasible due to restrictions to on-campus activities as a consequence of the COVID-19 pandemic. Therefore, individual interviews through video-conferencing using Zoom to enable access to remote participants were conducted. Traditional techniques of building rapport with participants through in-person conversations (such as welcoming non-verbal gestures and facial expressions, offering refreshments, etc.) became impossible during the remote interviews. Similar to techniques described by Pratt and Yezierski (2018), rapport between the interviewer (researcher, CR) and the participants was fostered by (1) sending emails thanking them for their voluntary participation in the interview and inquiring about their availability, (2) allowing participants to nominate their most convenient location and time to attend the interview and their preference for an online platform, and (3) giving them the choice to turn on or off their video camera during the interview. Pratt and Yezierski (2018) argue that these provisions for rapport building increases access to diverse participants.
For the three Cases, the same set of questions was asked, and the same protocol was used. Consent was obtained from the participants to audio-record the conversation for both focus groups and individual interviews. Transcripts were analysed for themes through inductive coding following the grounded theory approach (Pidgeon and Henwood, 1997). Through inductive thematic analysis, codes were generated from raw transcripts by identifying, arranging, and systematising the ideas and insights (Given, 2008) gathered from the focus groups and individual interviews. Codes were then organised into a higher-level categorisation to develop the themes. Interrater reliability measurements yielded a Krippendorff's alpha value of 0.782 signifying a reliable agreement among the three raters (Watts and Finkenstaedt-Quinn, 2021).
Description of the three cases
The participating universities were located in the Philippines and Australia. The first-year chemistry courses in all three participating universities were delivered in their respective first semesters in their academic year beginning in August (Case 1), March (Case 2) and February (Case 3). Table 1 summarises details of the instructional settings.| Particulars | Case descriptions | ||
|---|---|---|---|
| Case 1 | Case 2 | Case 3 | |
| Location | Philippines | Australia | Australia |
| Mode of delivery prior to this study | Traditional face-to-face | Blended | Blended |
| Online learning component employed prior to the study | Minimal to none; not widely and systematically employed | Well-established | Well-established |
| Mode of delivery employed in this study | Blended | Fully online (response due to COVID-19 pandemic) | Fully online (response due to COVID-19 pandemic) |
| LMS used | Moodle | Moodle | Blackboard and Edge |
| Semester involved in this study (inclusive months of the semester) | First Semester Academic Year 2019–2020 (August 2019–November 2019) | Semester 1, 2020 (March 2020–June 2020) | Semester 1, 2020 (February 2020–May 2020) |
| Survey questionnaire used (number of respondents) | Study survey (447) | Study survey (133) | Subset of study survey (107) |
| Focus group or individual interview conducted (number of participants) | Focus group (45) | Individual interview (11) | Individual interview (8) |
Up until the timepoint of this study in 2019, the mode of instruction in University 1 was traditional face-to-face teaching. Although there were efforts from the senior administration of University 1 to encourage the use of a learning management system (LMS) to integrate online learning, face-to-face teaching persisted and online teaching methodologies were not systemic. Individual teachers employed online platforms (i.e. shared drives) primarily for distribution of lecture presentation handouts, although this practice was not homogenous among all first-year chemistry teachers.
As part of this study, a blended mode of teaching and learning intervention was employed for the delivery of the five topics identified above over two weeks (weeks 4 to 5 of the semester). This blended delivery of the five chemistry topics was completed by 985 students enrolled in the course, via the Moodle learning management platform. For students enrolled in this course, it was the first time that they were introduced to blended learning with an online component delivered through Moodle.
Survey. Students were invited to respond to the survey one week after the end of the two-week blended learning implementation in September 2019. A pen-and-paper survey format was preferred over an online survey since students had been more familiar with this format and to promote a great response rate (Weigold et al., 2019). Completed surveys were returned by student respondents via their teachers, their laboratory instructors, or through submission to a designated box. A total of 447 complete and anonymous survey responses (45.4% response rate) were collected. At the 95% confidence level, the margin of error in survey responses from Case 1 is 4.6%.
Focus groups. 16 separate focus groups were completed with a total of 45 participating students (3 to 5 students per focus group session) during September 2019. Participants were anonymised and identified according to the session number they attended. Participants for each session were differentiated from each other through a letter designation. For example, the four participants in the first session were assigned a unique identity label: Participants 1a, 1b, 1c, and 1d. Focus groups lasted 30 to 45 minutes. To encourage a more comfortable and free-flowing discussion, students were encouraged to speak in both Filipino and/or English. Filipino words were later translated to English for the qualitative analysis of transcripts. The validity of the translated text was verified by a native Filipino speaker who has an expert knowledge of both Filipino and English.
The first-year chemistry course involved in this study is usually offered on-campus at the central campus of University 2 in Semester 1. There are two semesters in an academic year and each semester is made up of 12 teaching weeks, one revision week, and three weeks of examination. There was one course coordinator for this course who managed the delivery of the entire course for one semester and coordinated the teaching assignments among several academics.
In the years prior to this study, an online component had been effectively integrated into the blended mode of teaching and learning at this university. On a weekly basis, the blended learning activities for this course included an online independent study of pre-workshop learning materials delivered through Moodle, two one-hour on-campus workshop sessions, a one-hour on-campus tutorial session, and an on-campus laboratory session. The five topics included in this study were delivered during weeks 2 to 5 of the semester.
However, in 2020, the timepoint of this study, the course was shifted to fully online due to the COVID-19 pandemic response. On-campus workshop and laboratory sessions were substituted with weekly pre-recorded video materials and synchronous online sessions that were recorded and posted on Moodle. 1437 students were initially enrolled in this course, with 988 students remaining when the survey was administered.
Survey. Participants were invited to respond to the survey in Week 6 (one week after the delivery of the topics). Due to the restriction of on-campus activities, administration of an online version of the survey through Qualtrics was deemed appropriate. A total of 113 complete and anonymous responses were collected (11.4% response rate). At the 95% confidence level, the margin of error in survey responses from Case 2 is 5.9%.
Individual interviews. Individual interviews were conducted one week after the administration of the online survey. Participants were recruited through email invitations to arrange for their preferred schedule and online platform. Although options were provided to students, all 11 participants nominated Zoom as the preferred online video-conferencing platform. Each participant was assigned a unique identity label between Participant 17 to 27. Interviews lasted 20 to 30 minutes.
The hybrid mode of teaching and learning, where both on-campus (internal) and online (external) participation is accommodated, had been well-established at University 3 prior to this study. The first-year chemistry course at the timepoint of this study was offered internally only where all students attend on-campus activities. For this course, pre-lecture activities were provided through their Online Chemistry Learning Modules (OCLs) using the EdX Edge platform supporting students’ independent study prior to the weekly on-campus lecture sessions. Students attended on-campus laboratory sessions fortnightly.
The five topics included in this study were delivered during weeks 2 to 5 of the semester. On the first three weeks of Semester 1 2020, the Case 3 course was offered in the same manner as previously. However, when the pandemic situation in the state escalated, all on-campus activities were restricted resulting in a sudden shift into fully online teaching. On-campus lecture and laboratory sessions were replaced with weekly pre-recorded video materials that were posted in Blackboard and in Edge. 1100 students were enrolled in this course initially, but this dropped to 881 after restrictions began.
Survey. A subset of items from the survey were included into an existing evaluation instrument and administered at the end of the semester after the release of student final grades (i.e. June 2020). The same questions as in Cases 1 and 2 but different scale indicators were included in a Likert scale on student assessment of their learning gains (SALG) instrument which has been used in the course for several years. A total of 107 complete and anonymous responses were collected (12.1% response rate). At the 95% confidence level, the margin of error in survey responses from Case 3 is 6.2%.
Individual interviews. Interviews were conducted one week after the administration of the online survey. Emails were sent to participants who indicated their availability in the survey to arrange for their preferred interview schedule and online platforms. Zoom was nominated by eight participants from University 3, although one participant requested for the video to be turned off during the interview. Each participant was assigned a unique identity label between Participant 28 to 35. Interviews lasted 20 to 30 minutes.
Results and discussion
Three separate Cases were explored by analysing learning materials for online delivery of five first-year chemistry topics using the UDL principles to identify features that indicated effective practice. In accordance with the recommendations of Rao et al. (2018), examples of instructional design features that aligned with specific UDL guidelines are shared in Appendix 2. The intent to increase access to and decrease barriers to learning informed each aspect in the design of the online learning materials. Note that the combination of learning materials used in the three Cases were not identical, although there were some similarities based on the availability of the media formats in the resources used. Furthermore, not all UDL guidelines were integrated into each of the resources employed in the three Cases as discussed below.Case 1
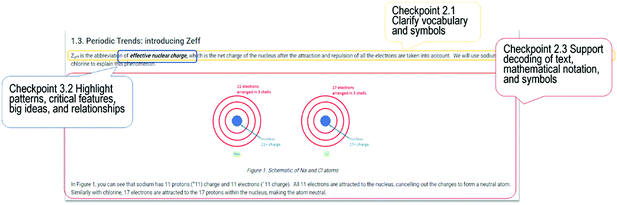
Prior to 2020, the Philippines uptake of ICT for education remained very low (UNESCO, 2018), although various strategies in applying ICT in education had already commenced in 1996 (Vergel de Dios, 2016). Because of this, a significant transformation in teaching practice in this course was needed in order for the UDL-based features of online learning resources to be implemented. Since the aim of this study primarily focuses on applying the UDL framework in online learning, a preliminary step of shifting the mode of teaching into an online platform was needed. This was the first time that enrolled students were introduced to blended learning with an online component. A resource-based learning (RBL) package (Hannafin and Hill, 2007; Hill, 2012; Reyes et al., 2022) was employed to facilitate the transition from the customary lecture-based intensive teaching to resource-based online learning modality. This transition is reported in another study (Reyes et al., 2022). UDL-based features were included in the design and development of the RBL package based on the 31 checkpoints prescribed by the UDL framework. Fig. 2 shows screenshots of selected examples. The only UDL checkpoint that was not enacted was Checkpoint 2.4 (promote understanding across languages). Whilst most students in Case 1 were non-native English speakers, English was the mandated language of instruction at University 1 and therefore all instructional materials for the five topics were presented in English.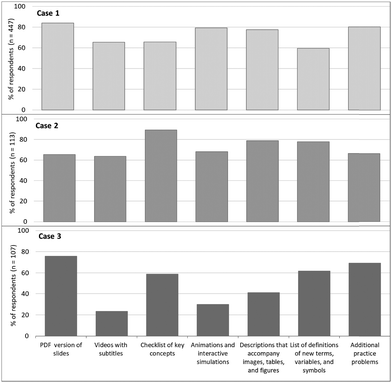 | ||
| Fig. 3 Percentage of respondents from the three Cases who reported these learning resources as being useful (survey data). | ||
Through their responses to the survey, participating students indicated that they had perceived animations and interactive simulations as helpful (79.4%). Further insight gained from analysis of the focus group discussion revealed that this was because the content had been presented in an alternative manner that allowed students to visualise the chemistry concepts that were not easily visible or that require imagination. As articulated by one focus group participant, when concepts were applied to the interactive simulation, they “did not need to imagine how chemical bonding occurred, [the student] could see it happening” (Participant 1a). Another focus group participant (Participant 5c) pointed out that molecular shapes were more easily understood because animations presented the “molecules as not flat, unlike in PowerPoints, the [bond] angles are more [visible], and now we know what three-dimensional molecules means”. Another student further elaborated, “I understand the lesson more because there are parts of the lesson that need to demonstrate moving [species]” (Participant 2a). Moreover, students perceived that animations and simulations provided in the Moodle site allowed for a more exciting way of learning because “there are more ways to present the information, unlike in lectures using just PowerPoints explained by [the lecturer]” (Participant 6c). Whilst the results reveal positive perceptions from the students, it is important to underscore that this was the first time this cohort was exposed to online learning in a constructive way. This was also the students first experience using web-based learning resources other than shared PDF notes to explore first-year chemistry concepts; hence, students’ perceptions may be impacted by these new experiences.
Students were also provided various forms of formative assessment to allow them to express their understanding of the chemistry concepts. Automated quizzes with in-built feedback were created within Moodle at the beginning or end of each topic. In some instances, web-based quizzes that could be accessed online external to the Moodle site were included to allow students further self-assessment of their learning. These quizzes were designed as ungraded formative assessments to serve as scaffolds for students’ learning. For some topics, problem sets with answer keys for feedback were also posted in the Moodle site for students to solve on their own. Guided step-by-step solutions were likewise provided to help students answer the problems given to them. Furthermore, students were encouraged to use an interactive simulation to answer problems in a worksheet. Students were also introduced to an online collaborative whiteboard where they could post their answer to a specific problem.
Survey respondents indicated that most of the UDL features that allowed them multiple means to act and express their learning helped them (Table 2). Of note, most respondents found that the step-by-step guides to solving problems (91.5%) and ability to use interactive chemistry simulations (87.9%) had positively impacted their learning. For example, an online resource demonstrating a step-by-step guide on how to draw the Lewis structure and an online interactive simulation exploring molecular shapes were employed in Case 1. In contrast to the traditional didactic lecture activity, students were taught the drawing of Lewis structures and molecular shapes using these available online resources. Despite being unfamiliar to these modalities, students found these helpful for their learning because stepwise worked solutions to the problems were provided to them to scaffold their learning. During the focus group, a student (Participant 6c) explained that in learning how to draw Lewis structures, “[t]hese are really helpful because to be able to see how many electrons are available, and which atoms they were assigned, where exactly electrons were put on which atoms, it is really helpful in that way. And I can almost use the step-by-step guide on any question about the same topic.”
| Percentage of respondents (%) | |||
|---|---|---|---|
| Positive (helped their learning) | None (had no effect on learning) | Negative (hindered them from learning) | |
| Detailed topic outline and learning objectives at the beginning of the lecture. | 80.3 | 18.6 | 1.1 |
| Checkpoints throughout the lecture/study guide | 78.3 | 19.2 | 2.5 |
| Self-assessment and reflective questions | 66.0 | 31.3 | 2.7 |
| Step-by-step guide and checklist in problem solving | 91.5 | 6.9 | 1.6 |
| Access to assistive technologies and tools such as text-to-speech (TTS), speech-to-text (STT), mouse, keyboard and alternative keys | 59.7 | 33.6 | 6.7 |
| Ability to choose your own time to take the quiz/submit answers to problem sets | 67.6 | 11.0 | 21.5 |
| Ability to choose pen and paper and/or computer tools (such as word, excel, PowerPoint) to answer problem sets | 77.2 | 17.9 | 4.9 |
| Ability to use interactive simulation to solve problems | 87.9 | 8.7 | 3.4 |
| Receiving feedback from quizzes and exercises | 85.7 | 11.9 | 2.5 |
Survey respondents reported that some aspects had no impact, or indeed negatively impacted their learning. 33.6% of the respondents reported that assistive technologies did not impact their learning because they “did not need to use [assistive technologies] in studying”, according to a focus group participant (Participant 1a). Whilst, 21.5% of respondents reported that the ability to choose their own time to study hindered their learning. A focus group participant said that this was because they “tend to procrastinate and put off their learning tasks including the quizzes and problem sets up to the deadline” (Participant 10b).
Table 3 shows that most of the student respondents self-reported their engagement with their learning by always setting specific goals in completing their tasks and requirements (42.3%). Furthermore, the majority of them always reviewed their lessons, and tried to figure out difficult concepts (60.9%). Only a small portion of the respondents reported not being able to pause and evaluate their learning (2.7%). As one student (Participant 8d) elaborated during the focus group discussion on why they self-evaluate, “I have been trying to make sure that I'm knowing topics before I move on to another topic, or making sure that I am concreting a fundamental before going into something a bit wider.”
| Percentage of respondents (%) | ||||
|---|---|---|---|---|
| Always | Sometimes | Once | Never | |
| I set myself specific goals to complete all the tasks and requirements | 42.3 | 52.1 | 4.0 | 1.6 |
| I had a good grasp of what I was required to learn for the past two weeks | 28.9 | 62.2 | 6.5 | 2.5 |
| I explored other contents that are socially relevant to my personal context such as my age, culture, gender, etc. beyond the given tasks | 17.2 | 41.8 | 13.2 | 27.7 |
| I created my own study routine for this unit/course that I try to keep it despite other priorities | 37.1 | 41.4 | 13.2 | 8.3 |
| I explored the range of resources, activities, and tasks for the past two weeks as I found them challenging but motivating | 44.3 | 42.7 | 10.3 | 2.7 |
| I took advantage of the opportunities to work on problem sets collaboratively with my classmates and/or teachers | 40.5 | 31.8 | 12.5 | 15.2 |
| I employ self-assessment questions at the end of each topic as a mechanism to reflect of my performance | 60.9 | 34.2 | 2.9 | 2.0 |
| I was very interested to receiving timely feedback encouraged me to persevere and enabled me to develop effective learning strategies and awareness of my own progress | 42.7 | 41.6 | 10.7 | 4.9 |
| When I encountered a difficulty in understanding a particular topic, I requested for assistance from my teacher and/or peers | 16.1 | 29.5 | 14.3 | 40.0 |
| I had been able to pause and evaluate my progress in the course | 47.0 | 43.2 | 7.2 | 2.7 |
Case 2
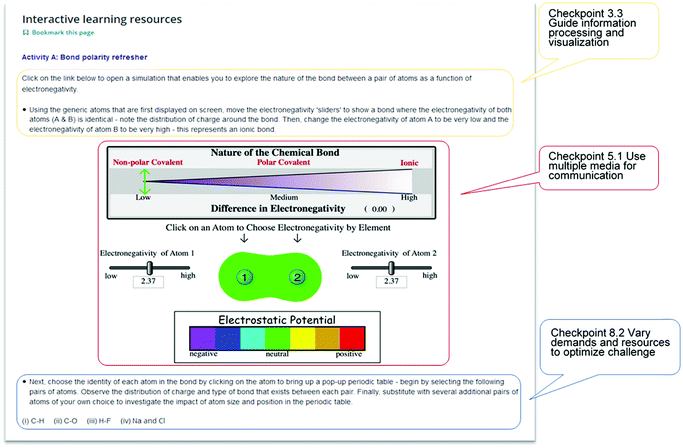
A review of the blended learning materials of University 2 (developed over several years of online delivery) found they already adhered with 25 of the 31 (80.6%) UDL checkpoints, and utilised a robust online learning platform. Hence, minimal modifications were required to enhance the accessibility features of the online learning materials, and resources were added to enact the missing UDL guidelines. Existing resources were enhanced by adding UDL-based features such as highlighting texts, adding list of definitions, and clarifying learning objectives.To enhance the UDL enactment in Case 2, some features were added to address lacking guidelines. For example, to address checkpoint 1.2 (offer alternatives for auditory information), transcripts were provided to accompany videos that could not be close-captioned and which students situated in other countries may have been unable to access. To address checkpoint 2.5 (illustrate through multiple media) and 6.3 (facilitate managing information and resources), simulations and animations with accompanying comprehensive instruction on the learning activities involving them were added. Fig. 5 illustrates some of these new enactments.
 | ||
| Fig. 5 Examples of specific application of UDL principles to the online learning materials for Case 2 introduced in this study. | ||
For Case 2, examples of checkpoint 2.4 (promote understanding across languages) were not observed in any aspect of the course. Whilst this could introduce a barrier for non-English speakers, English is the medium of instruction at University 2, and the more convenient language to teach chemistry concepts. Translating the chemistry content into multiple languages may be limiting due to language minorities where some linguistic components of the discipline cannot be translated effectively to languages other than English impacting on explanation of concepts. Strategies to incorporate this specific UDL guideline into teaching chemistry still requires further consideration.
A high percentage of survey respondents from Case 2 perceived the checklists of key chemistry concepts (89.4%) and the list of definitions of new terms, variables, and symbols (77.9%) as being useful (Case 2, Fig. 3). Individual interviews with students revealed that they perceived that these features highlighted the key elements of the concept being taught, supporting an easier grasp of the important concepts. This finding is consistent with the suggestion of Airey and Linder (2009) that in order for students to achieve fluency in discipline-specific discourse (i.e. chemistry topics), teachers must provide students guidance to actively engage students as they attempt “to make meaning such discourse for themselves.”
65.5% of respondents from Case 2 also found the PDF documents of the slides helpful for their learning. The PDF documents are aligned with checkpoints 1.1 (offer ways of customising the display of information) and 4.1 (vary the methods for response and navigation). Results suggest that students may have found PDF resources helpful because they were familiar with this format and they are readily customisable (for example, through digital annotation).
One student (Participant 20) elaborated during the interview that they found these UDL features “really helpful, especially when you have so much subjects to do. It is really helpful that someone's looking out for one you need to be responsible for. They help you organise things because then I can put that into a bigger list for myself to do and I can maintain everything I need to be doing.” Another student (Participant 19) thought that descriptions of new variables and symbols were likewise useful and “good in chemistry in particular because obviously [there are] a lot of equations and a lot of like subatomic things where it is sometimes hard to visualise if people are just talking about them, and having those diagrams and little notes, those could be useful.”
Video recordings, and animations and interactive simulations were also perceived as useful by 63.7% and 68.1% of the survey respondents, respectively. An interview participant (Participant 23) explained that, “The part of science that I really enjoy and that I reflect on is all the stuff that I do either kinaesthetically or visually. I think obviously in an online platform, you know you can't have that kinaesthetic learning so I find video recording is one of the next best things. I think that's one of the largest, I reckon probably the most key tool for me, I think video recording is huge. I think similarly with animation and interactive stuff, it sounded silly, but I think you know like the games or the drag-and-drop sort of style animations. I find them really good because it allows me to see what happens from within, like in the formation of bonds when small atoms come near each other.”
In Case 2, an open-source textbook was provided to all students as a substitute for a paid subscription to a copyrighted textbook. This free digital version of this textbook reduced the potential financial barrier to learning in the course. During their interview, Participant 17 highlighted this benefit of free access to materials: “I'm really grateful for it, because in some of the other [courses] I'm doing I did have to buy a textbook and that's on top of all the fees. It's just really very useful, something is free. And it's a very good book, it explains the concepts very well.”
| Percentage of respondents (%) | |||
|---|---|---|---|
| Positive (helped their learning) | None (had no effect on learning) | Negative (hindered them from learning) | |
| Detailed topic outline and learning objectives at the beginning of the lecture | 74.3 | 24.8 | 0.9 |
| Self-assessment and reflective questions | 90.3 | 8.0 | 1.8 |
| Step-by-step guide and checklist in problem solving | 71.7 | 23.9 | 4.4 |
| Access to assistive technologies and tools such as text-to-speech (TTS), speech-to-text (STT), mouse, keyboard and alternative keys | 32.7 | 61.9 | 5.3 |
| Ability to choose your own time to take the quiz/submit answers to problem sets | 88.5 | 8.8 | 2.7 |
| Ability to choose pen & paper and/or computer tools (such as Word, Excel, PowerPoint) to answer problem sets | 81.4 | 16.8 | 1.8 |
| Ability to use interactive simulation to solve problems | 69.9 | 23.9 | 6.2 |
| Receiving feedback from quizzes and exercises | 93.8 | 4.4 | 1.8 |
88.5% of the survey respondents likewise found the ability to choose their own time to complete their weekly learning activities helpful because of the flexibility that it offered them. A student explained during the interview, “I think this has to be one of the most helpful things because I really, really value having my own time. It just allows me to plan my days well, because some days, I might choose to either do less work and spend more with my family. Or choose days where I just study for the entire day. It just really, really helps to not just have the preparation quizzes that you have to do it in one day, and that you have a week to do it. That's definitely a positive” (Participant 17).
| Percentage of respondents (%) | ||||
|---|---|---|---|---|
| Always | Sometimes | Once | Never | |
| I set myself specific goals to complete all the tasks and requirements | 63.7 | 30.1 | 0.0 | 6.2 |
| I had a good grasp of what I was required to learn for the past four weeks | 34.5 | 55.8 | 5.3 | 4.4 |
| I explored other contents that are socially relevant to my personal context such as my age, culture, gender, etc. beyond the given tasks | 8.9 | 30.1 | 15.0 | 46.0 |
| I created my own study routine for this unit that I try to keep despite other priorities | 45.1 | 32.8 | 10.6 | 11.5 |
| I explored the range of resources, activities and tasks for the past four weeks as I found them challenging but motivating | 29.2 | 57.5 | 8.0 | 5.3 |
| I took advantage of the opportunities to work on problem sets collaboratively with my classmates, tutors and/or lecturers | 24.8 | 37.2 | 20.3 | 17.7 |
| I was very interested to receiving timely feedback that encouraged me to persevere and enabled me to develop effective learning strategies and awareness of my own progress | 69.0 | 24.8 | 2.7 | 3.5 |
| When I encountered a difficulty in understanding a particular topic, I requested for assistance from my teacher and/or peers | 17.7 | 41.5 | 20.4 | 20.4 |
| I had been able to pause and evaluate my progress in the unit | 35.4 | 39.9 | 15.0 | 9.7 |
To further provide opportunities for engaging students in their learning by allowing them to see the relevance and value of what they were learning, real-life contexts and applications of some topics were included in the online learning materials. Although these were provided, 46.0% of the respondents never explored these learning materials. According to one student who participated in the interview, “I don't always explore these contents, all the time. I enjoy exploring content which is relevant to this time in context with my studies. And I see them as beneficial as much as it is. I find that it's more of like a leisure thing as it helps stimulate the brain but not in overwhelming fashion” (Participant 27).
Case 3
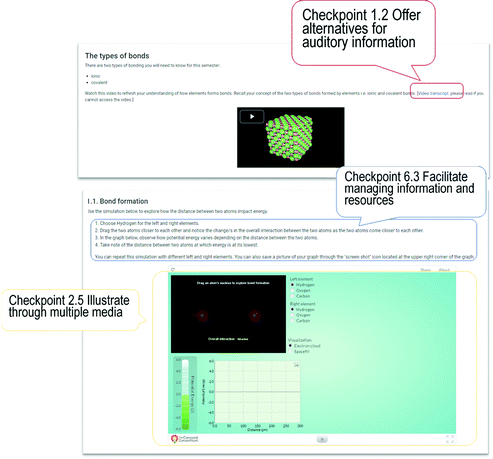
In Case 3, the online learning materials did not require substantial further alignment with UDL guidelines as the majority of features already aligned and were embedded across resources and activities resulting from an earlier course restructure.Similar to Case 2, Case 3 has a well-integrated online component in their hybrid delivery of the first-year chemistry course. Although Case 3 was designed without explicitly applying the UDL framework, inclusive practices had been intentionally applied in the development of online learning materials and hence included notable UDL features. Examples of these enactments are shown in Fig. 6. Learning resources and activities complemented each other which resulted in strong alignment with the UDL framework. A variety and diversity of learning resources were key features of the design of the course. Whilst no single resource could or did cover all of the 31 UDL checkpoints, a combination of the learning resources and activities combined to cover a large proportion of principles. A UDL checkpoint that was not effectively addressed was checkpoint 7.3 (minimise threats and distractions), because students are typically enrolled in four courses in parallel and may encounter external influences on their studies. Similar to Case 2, checkpoint 2.4 was not observed in any aspect of the course in Case 3 and is thus recognised as a potential barrier with around 15% enrolment being international students.
Survey respondents from Case 3 self-reported that they perceived the PDF version of PowerPoint lecture slides (75.7%) and additional practice problems with complete solutions (80.3%) had been useful for their learning (Case 3, Fig. 3). Since the UDL framework seeks to provide maximum learning opportunities for students to learn with minimum barriers, we posit that if given options to access and learn content, students may choose to access resources in formats that they are already familiar with. Similar to Case 2, students were familiar with PDF versions of slides, which may have enabled them easier access to the content, or a ready method to customise this resource. Interview Participant 31 elaborated that the PDF copies of the lecture enabled easier note-taking, “especially given the limited time when the lecturer is presenting structures of molecules and shapes or chemical equations, if we had to copy from the presentation to make our own notes, we might miss important details of the topic.” Interview Participant 32 explained that “[t]he practice problem was actually pretty good for exam revision … especially with chemistry there's just like definite solution, so it was a good starting point to have the solution.” Another student (Participant 35) further pointed out that “if you're given a solution, you can check to see whether you're correct or wrong.”
Furthermore, some respondents (29.9%) perceived the chemistry animations and interactive simulations helpful for their learning. During an interview, a student, who reported that they had never seen simulations like that before reflected that it was “very interesting to see what actually seems to go on with the molecules and things like that” (Participant 35). For this student, “the molecular shape, remembering them as you draw and add some bonding electrons or some nonbonding electrons to the molecule, it didn't help in any way to imagine molecules as compared to visualising them.” Interview Participant 33 particularly found the animations useful because “it allows another visual component of understanding.” This student thought that “like molecules or electrons and things that I cannot see, you will know what is happening at such a small level.”
| Percentage of respondents (%) | ||||||
|---|---|---|---|---|---|---|
| No help | A little help | Moderate help | Much help | Great help | Did not complete this | |
| Progression of chemistry concepts taught in lectures | 0.0 | 2.8 | 16.8 | 37.4 | 43.0 | 0.0 |
| Problem-solving activities embedded in lecture-based resources | 0.0 | 2.8 | 12.2 | 30.8 | 53.3 | 0.9 |
| Think.Chat.Learn discussions with other students | 2.8 | 30.2 | 23.6 | 21.7 | 17.0 | 4.7 |
| Padlet drawing tasks | 15.1 | 28.3 | 32.1 | 17.0 | 4.7 | 2.8 |
| Online quizzes | 0.0 | 3.7 | 11.2 | 15.9 | 68.2 | 0.9 |
| Formative feedback from the check questions | 7.5 | 22.6 | 23.6 | 14.2 | 24.5 | 7.6 |
These online quizzes reflected where students “did wrong and [they] could help students to understand which they did wrong” (Participant 35). Common errors in problem-solving were picked up and as one student pointed out, this “saved [them] a few times” (Participant 28) because if it were not pointed out, they would have started again working through the problem in the same way and getting the wrong answer again. Problem-solving activities likewise helped students to practice similar problems and apply worked solutions to new activities. The given hints were likewise helpful to get started. A student explained during the interview, “I see the exact same question repeated, I will try to approach each problem individually, drawing on what I know. I just read the question, and this is what we know, this is what we're trying to find out. These are things which I've used before that helped me to figure out the answers” (Participant 28). These findings align with the position of Airey and Linder (2009) that a repetitive or iterative practice is a necessary element of learning university science because this supports students to become fluent in disciplinary-specific discourse (such as learning the specific chemistry topics in this study).
In addition to online quizzes and problem sets with answer keys, 53.3% of students of Case 3 perceived that online chat and discussions with other students provided at least moderate help for their learning. An interview participant explained that discussions with other students allowed them to not only ask questions, but also because other students voluntarily shared their views of how to approach a problem. Participant 28 further elaborated that these discussions were “very, very useful for learning the content because it was not just what you're giving, someone's thought about it this way, or there would be three or four different people going this way, and eventually they get to an answer.”
To further encourage students’ interest in chemistry topics, real-life contexts and applications were integrated at important points within the topics. One student explained that because of what they were taught in the course, the student “would prefer to go into more biochemistry, like Doctor of Medicine, because [they] actually enjoy the chemistry part of it.” (Participant 34).
Moreover, students were provided with avenues for self-evaluation of progress through self-assessment questions, reflection points, and feedback from online quizzes and check questions. Students perceived these different features helpful for their learning because it allows them to “gain knowledge, and train [them] to make sure [they] actually do understand it and understand it correctly and somehow [they]'re gauging [their] progress by [themselves]” (Participant 30).
Students’ perceptions on the helpfulness of UDL-based features
Focus groups and individual interviews with students from each of the three Cases provided deeper insights into why students perceived these UDL-framed learning resources had been helpful to their learning. Through inductive coding, several themes were identified as significant from the discussions and are listed in the table in Appendix 3. Despite the differences in the actual resources and activities provided to students in the three Cases, parallel themes were found due to common benefits of implementing the UDL framework. For example, participants benefited from facilitated processing of information provided by the clear definitions of new terms and symbols, by highlighting relevant texts, and clarifying the learning objectives. Animations and interactive simulations, as well as videos and images provided students with better learning experiences by enhancing their visualisation and access of the concepts through different media. Moreover, participants also articulated how online quizzes and additional problem sets helped them by allowing opportunities for self-evaluation of their progress.Challenges in incorporating UDL-based features
Whilst there were promising affordances through implementing the UDL framework, some students experienced disadvantages resulting from incorporating several UDL-based features, representing limitations of this approach. As these features were delivered online they necessarily required internet connection to be accessible. For example, whilst there were undeniable benefits to the use of animations and simulations in content delivery, the media or format of the resources themselves could introduce a barrier in terms of accessibility to learning. For students from Case 1, this emerged as a significant barrier because of the limitations in the accessibility of ICT in the Philippines. Participant 5a explained in the focus group discussion that “accessing multimedia resources was sometimes problematic due to weak internet connection. Although animations looked exciting, there were instances when they failed to load in [their] laptop so [the student] was not able to experience it.” Further scaffolded support may be needed for these students to enable them to fully experience the benefits of UDL-based features such as animations and simulations.Some students chose not to use some UDL-based features, primarily because they felt that they did not need them. For example, assistive tools and technologies could be readily made available, but may not be useful for students who “didn't really know what that was” or if “[they were] not in a position where [they] needed any of them” as mentioned by Participants 26 and 27 from Case 2. Participant 29 from Case 3 did not use the simulations provided to them because for them, the “concepts were not hard enough that [they] had to go out of [their] way and use them so didn't really look at them much.”
The provision of multiple resources to provide students options may also present a barrier, rather than an opportunity, for some students who found the volume of resources overwhelming. Participant 4b in Case 1 elaborated in the focus group discussion that “it was more exhausting to read several pages than to listen to the teacher.” The same sentiments on the volume of resources were expressed by a Participant 29 from Case 3 who admitted not having used “heaps of the online chemistry learning resources because we had a lot of resources.” This student further explained that “[a]lthough that was great, I did not have to use all of them. I only use them if I found a particular concept slightly harder.”
Emerging considerations for applying UDL principles to inform the teaching and learning of first-year chemistry
The five foundation topics common to all three Cases included in this study can be considered as core concepts in chemistry, hence, a thorough understanding is critical as a foundation for future topics in chemistry (Hoffmann and Laszlo, 1989; Hamza et al., 2008). Since chemistry is a discipline that relies on multiple external representations for concepts, the first UDL principle of providing multiple means of representation was readily applicable to enhance chemistry teaching. The second and third UDL principles of providing options for action and expression, and options for engagement were addressed through the multiple learning resources and activities provided to students as shown in the learning designs of the three Cases.In the delivery of the chemical bonding topic, the use of video, animations, and/or simulations complemented the usual text-based content delivery in all three Cases. For example in Case 3, written chemical species, symbols, and equations presented in text format were illustrated in other multimedia formats that allowed better visualisation of the chemical phenomena occurring during bond formation (Fig. 6). The guided interactive simulations employed in the three Cases likewise allowed students to explore and generate knowledge through their own inquiry. In Case 1, enabling this in an online environment aimed for an enriched learning process by increasing not only students’ visualisation of the concept, but also by providing another means to gain knowledge, express their understanding and promote engagement with their own learning as demonstrated in Fig. 3 and Table 2. Increasing accessibility to learning chemical bonding was made feasible by addressing the barriers to understanding the concept resulting from a dominantly text-based instruction and the lack of visualisation.
Based on the principle of providing multiple means of engagement, students’ interest in the topics was fostered by giving additional context. For example in Case 2, the topic on the periodic table and trends included an online interactive periodic table that allowed students to explore commercial uses as well as common sources of each element. This interactive periodic table was accompanied by a YouTube video discussing the rare elements present in mobile phones, which further encourages students’ awareness of pressing global sustainability issues.
To promote the UDL principle of providing multiple means of action and expression, a PhET simulation to explore molecular shapes was employed across all three Cases, however different methods of expressing students’ understanding of the topic were utilised. This represents a common and shared value of this mode of representation in supporting learning of related concepts by the instructors at each institution. In Case 2, students were asked to complete a report posted in Moodle, whilst in Cases 1 and 3, students were asked to post pictures of their responses to questions on a Padlet. For all three Cases, students were first given scaffolding on how to navigate and explore the PhET simulation. It was followed by exploring the shapes of various model and actual molecules, then, answering some practice questions of varying levels of difficulty. With the guidance of UDL principles, a learning resource, online or otherwise, (i.e. simulation) can be integrated appropriately in a learning activity to maximise the use of the resource. As Frailich et al. (2009) argues, providing students with “clearly defined and focused activities [promote] an environment in which the students become active participants in the learning process.”
The focus group and individual interviews provided insights and themes that were common across the three Cases into why the students found the resources useful. Overall, the UDL-based features included in the design benefited the students in multiple ways. When asked if they could provide further comments about the UDL-based features, one participant (who felt capable of learning on their own nonetheless) expressed appreciation for the additional features and specifically referred to perceived barriers to learning: “Absolutely, if they are not there, it would be more difficult… But when you're facing extra barriers to learning, I think that every little thing that could possibly be provided helps” (Participant 25).
Limitations of the study
The findings in this study were based on three independent Cases of UDL implementation at three universities with different learning contexts. Whilst the perceived benefits of the UDL implementation presented were common amongst student participants, generalisation of these observations would not be appropriate. Although it may pose logistical challenges, future work may focus on implementing the same UDL-based features on similar instructional settings to verify the observations presented in the previous section.This study is also limited by the scope of implementation in terms of the number of topics covered. The UDL framework was evaluated in five topics in first-year chemistry, hence, further implementation on the whole course or to other chemistry courses may present a wider perspective on the applicability of the UDL framework in enhancing student online learning experiences of chemistry.
Particularly in Case 1, the introduction of a new learning modality, and the use of an LMS, could potentially limit the generalisability of the insights gained from this study. Given that the learning environment was new to the students, and that they were not adept at using the LMS at first, their perceptions about the learning materials provided might differ compared to students who were accustomed to this online learning modality. Hence, it is important to recognise that students are trained to make use of familiar resources and perhaps need orientation in how to best engage with new multiple options or modes. Where applicable to a learning activity, students should also be provided document formats that can be manipulated to allow them to adjust formatting (for example colour, font size, etc.) based on a student's individual preferences. Furthermore, whilst this study investigated ways to enhance online learning experiences through accessibility, the persistent issue of access to reliable and affordable internet in the Philippines could be a potential confounding factor despite the researchers’ efforts to employ digital resources that only require low-bandwidth (or something similar).
Further improvements in the instructional design to include other active learning strategies in the online component of the first-year chemistry could still be done in order to promote students’ learning experiences. For example, self-paced exploratory activities using simulations may be enriched further by a moderated discussion forum in the LMS to encourage students to elaborate on their learning at specific points. Discussion with peers enables students to generate representations of their understanding themselves and provides self-calibration through formative feedback (Ludvigsen et al., 2015; Gikandi and Morrow, 2016). Where applicable, cooperative learning may also be encouraged using context-based problem-solving activities within synchronous online sessions.
Conclusions from this study may likewise be limited since only students’ perceptions were explored, and it would be ideal to explore the teachers’ perception of the effectiveness of the UDL-based design implemented in this study. Future research could investigate teachers’ insights on how UDL-based instructional design could be employed to enhance students’ learning experiences in first-year chemistry.
A final possible and significant limitation in this study is that the data arising from Cases 2 and 3 were collected during the COVID-19 pandemic during 2020 when the usual program of learning was shifted online without time to intentionally adjust practice. Perceptions and insights from students may have been influenced by the adverse impacts of the pandemic on their learning experience. Since the pandemic restricted in-person learning, students were forced to engage only with online learning materials, and this could have introduced positive bias in students who found these online resources useful. In the future, findings from Cases 2 and 3 could be further affirmed as teaching and learning transitions into a post-pandemic format.
Implications for practice
Findings from this study suggest that there is substantial potential for designing a more accessible online learning environment by applying the UDL framework checkpoints in chemistry learning environments. Despite the contextual differences for the three Cases presented in this study, similar themes were observed in students’ perceived usefulness of the UDL-based features of the online learning materials. Although UDL features were incorporated to varying extents in the three different contexts, common positive outcomes were observed such as the benefits of facilitating information processing and multimodal presentation (and understanding) of chemistry concepts. Students from the three Cases likewise benefited from the provisions for flexibility, self-monitoring of progress, and free access to materials afforded by the UDL-based features.This study further suggests that the UDL framework could be “universally” applied to various contexts to increase access and decrease potential barriers to learning first-year chemistry topics. Whilst we may not be able to quantify the spectrum of our students’ diversity and address their every individual need, an accessible learning environment can be created through the UDL principles. As teachers and learning designers, being able to recognise potential impediments to learning and addressing these barriers will provide our students greater opportunities to maximise their learning of chemistry.
UDL principles could be integrated into the design of online learning materials for five first-year chemistry topics as demonstrated in this study. Common strategies included:
• Multiple means of presentation of content could benefit the delivery of topics such as periodic table and trends which are usually delivered in text and PowerPoint lecture slides. The use of an interactive periodic table would allow students to explore the characteristics of each element and in so doing, solidify their understanding of periodic trends.
• Aside from the typical lecture presentation on chemical bonding and Lewis structures, animations and simulations could be employed to enhance the visual conception of complex formation of bonds between atoms based on their electronic structures. This would not only boost students’ engagement with their learning, but may also potentially improve the quality of their conception of chemical bonding.
• Molecular shapes and polarity are two topics that should be demonstrated in three-dimensional space. These topics lend themselves easily to the abundance of readily available digital learning resources. Other than the typical two-dimensional flat drawings of molecules, animations, simulations, digital molecular models, and images in graphics interface format (gif) are among the many ways of presenting three-dimensional molecular shapes and illustrating their implications.
• An online learning environment designed with the UDL principles could also allow students other means to express their learning. For example, in addition to the summative examinations and quizzes, formative assessments that employ digital tools such as mobile phones and chemistry software, or the use of interactive quizzes with automated feedback, could be employed such that students may be given options to demonstrate their understanding.
Note, however, that the UDL framework may present an obstacle in learning design as it encourages provision for “multiple options”. The number of online learning resources and activities should be optimised given the amount of time students need to allot to study and to consider alternative options for supplementary and complementary resources. Careful consideration must be observed in curating the best resources that will allow maximum accessibility to learning without posing impediments in the actual learning process resulting from the enormous volume of resources. It is imperative to carefully look into the context, the value of the resources, and the benefits they provide towards increasing accessibility to learning.
Our students may not be able to acknowledge all the UDL-based features we put in their online learning environment, and how these features help them in their learning, but we should aim to make these accommodations and provisions available so they can access them whenever the need arises. In the end, these features may actually make a difference for a student who needs it but does not know it. After all, teaching chemistry in the most accessible way is the right thing to do.
Conclusions
The rapid increase in diversity of learners around the world has catalysed responses from various educational systems to provide equitable access to quality higher education. Previous studies have already established the viability of the UDL framework to address pedagogical requirements to accommodate diverse learners without the need to customise design for specific student groups. In this study, we operationalised the integration of UDL principles and guidelines in the design of first-year chemistry topics (periodic table and trends, chemical bonding, Lewis structures, molecular shape, and polarity) for online delivery, which has not previously been reported. The benefits of the UDL-based features were likewise presented in the three Cases of implementation in three universities with varied contexts. Although comparison of the three Cases was not intended, the outcomes of the study signify a universal indication of beneficial impacts on students’ learning experiences in chemistry.Seeking various means of representation, action and expression, and engagement requires design principles that heavily rely on the context of the instructional setting and pedagogical intentions. Despite differences in the level of ICT integration and availability of infrastructure among the three universities, online delivery of first-year chemistry was promoted with the guidance of the UDL framework. Resources readily available online were also useful in providing variety in learning resources as well as a wider range of learning activities that went beyond reading texts and listening to lectures. In particular, using learning materials with three-dimensional features was found to enrich the learning experience by providing visual appreciation of an abstract chemical phenomenon.
UDL-based features likewise enabled students to develop their autonomy for their own learning. Guides, lists, and instructions on the scope of the content, on navigating the learning resources, and processing the concept delivered by the resources empowered the students in building their own self-efficacy skills. Improving time management and study time approximations were likewise supported by the flexibility brought about by the UDL-based features. These benefits encompass not only students’ cognition, but of their metacognition as well.
Conflicts of interest
There are no conflicts to declare.Appendix 1: the survey instrument used in this study
Accessible design for digital learning environment in blended first-year chemistryPart A. Your background information
1. What age are you?
2. To which gender identity do you most identify?
| Female | Male | Non-binary | Rather not say | ||||
3. What is your status as a student?
| Full time | Part time |
4. Are you a recipient of a scholarship program or grant that supports you financially at University?
| Yes | No |
5. Do you identify as a person with a disability?
| Yes (please answer 7.1 & 7.2) | No |
7.1 How would you describe your disability? ______________________
7.2 With regard to your disability, what kind of assistance or support do you need for your study? ________________________________________________________________________
Part B. Your learning experiences
(Note: Items in bold face were not initially included in the original version of the existing survey which is annually administered in Case 3. These items were inserted in survey administered for this study. All other items have equivalent questions to the existing survey.)
The following questions seek to capture your experiences on learning the topics covered in the last four weeks (periodic trends, chemical bonding, Lewis structure, molecular shapes and polarity). To help you answer these questions, recall the way the lessons were delivered in the previous two weeks of your study i.e. Weeks 1 to 4.
Which of the following did you find useful for your study? (Tick all that apply)
| PDF printer-friendly version of power point notes | |
| Written transcription of the videos/audios/podcasts | |
| Text-to-speech (TTS) app (i.e. Speak for Word, Speech for Mac OS, other TTS app for android) | |
| Closed caption (CC) videos or videos with subtitles | |
| Multiple forms of information display (i.e. text, tables, illustrations, and diagram) | |
| Animation and interactive simulation supplementary to the lecture | |
| Highlighted text through bold faced, underlined and/or coloured fonts | |
| Additional practice problems of various types | |
| List of definitions of new terms in a text and new variables and symbols in a mathematical equation | |
| Descriptions that accompany images, tables and figures | |
| Discussions that relate background knowledge and current topics | |
| Concept maps and graphic organisers that accompany lecture notes | |
| Translation of words expressed in language/s other than English | |
| Checklist of key concepts at the beginning and end of each topic |
Identify which attributes impacted your study of the topics for the past two weeks positively (helped your learning), negatively (hindered you from learning), or none (had no effect).
| Attributes | Impact | ||
|---|---|---|---|
| Positive (helped your learning) | Negative (hindered you from learning) | None (had no effect) | |
| 1. Detailed topic outline and learning objectives at the beginning of the lecture. | |||
| 2. Checkpoints throughout the lecture/study guide | |||
| 3. Self-assessment and reflective questions at the end of the lecture/study guide | |||
| 4. Step-by-step guide and checklist in problem solving | |||
| 5. Access to assistive technologies and tools such as text-to-speech (TTS), mouse, keyboard and alternative keys | |||
| 6. Ability to choose your own time (within the two-week frame) to take the quiz/submit answers to problem sets | |||
| 7. Ability to choose pen & paper and/or computer tools (such as word, excel, powerpoint) to answer problem sets | |||
| 8. Ability to use interactive simulation to solve problems | |||
| 9. Receiving feedback from quizzes and exercises (based on performance) | |||
How often did you apply the following learning strategies while studying the topics for the past two weeks?
| Learning strategies | Always | Sometimes | Once | Never |
|---|---|---|---|---|
| 1. I set myself specific goals to complete all the tasks and requirements. | ||||
| 2. I had a good grasp of what I was required to learn for the past two weeks. | ||||
| 3. I explored other contents that are socially relevant to my personal context such as my age, culture, gender, etc. beyond the given tasks. | ||||
| 4. I created my own study routine for this unit/course that I try to keep it despite other priorities. | ||||
| 5. I explored the range of resources, activities, and tasks for the past two weeks as I found them challenging but motivating. | ||||
| 6. I wrote notes during lecture sessions and while I studied online learning materials. | ||||
| 7. I took advantage of the opportunities to work on problem sets collaboratively with my classmates and/or teachers. | ||||
| 8. I was very interested to receiving timely feedback encouraged me to persevere and enabled me to develop effective learning strategies and awareness of my own progress. | ||||
| 9. I employ self-assessment questions at the end of each topic as a mechanism to reflect of my performance. | ||||
| 10. If I found difficulty in understanding the concepts, I review my lessons and try to figure it out. | ||||
| 11. When I encountered a difficulty in understanding a particular topic, I requested for assistance from my teacher and/or peers thru various avenues such as “Ask us!” forums and private messaging. | ||||
| 12. I had been able to pause and evaluate my progress in the course/unit. |
Scale indicators for Case 3 only:
How much did each of the following activities help your learning?
| No help | A little help | Moderate help | Much help | Great help | Did not complete this | |
|---|---|---|---|---|---|---|
| Progression of chemistry concepts taught in lectures | ||||||
| Problem-solving activities embedded in lecture based resources | ||||||
| Think.Chat.Learn discussions with other students | ||||||
| Padlet drawing tasks | ||||||
| Online quizzes | ||||||
| Formative feedback from the check questions |
Appendix 2: UDL-based features included in the online learning materials used in first-year chemistry
| UDL-based features of the learning package | UDL checkpoint implemented | Barriers addressed/access increased |
|---|---|---|
| a Employed in Case 1 only. bEmployed in Case 2 and Case 3 only. | ||
| Study guide for each topic (Contained an introduction, list of learning objectives and key concepts, curated learning resources, and guided learning activities) | 6.1 Guide appropriate goal-setting | Access increased – enhances students’ ability to develop independent learning skills and learner autonomy |
| 6.3 Facilitate managing information and resources | ||
| 7.3 Minimise threats and distractions | ||
| PDF version of the study guide (A downloadable and printable form of the study guide)a | 1.1 Offer ways of customising the display of information | Access increased – content becomes accessible even if offline |
| Checklist of learning objectives or key concepts at the beginning and end of each topic | 6.1 Guide appropriate goal-setting | Access increased – providing scaffold to estimate difficulty of the scope of topics |
| PDF version of presentation slidesb | 1.1 Offer ways of customising the display of information | Access increased – content becomes accessible even if offline |
| 2.1 Clarify vocabulary and symbols | Barriers decreased – removes confusion at different aspects that hinders learning of the concept | |
| 2.2 Clarify syntax and structure | ||
| 2.3 Support decoding of text, mathematical notation, and symbols | ||
| 3.1 Activate or supply background knowledge | ||
| 3.2 Highlight patterns, critical features, big ideas, and relationships | ||
| 3.3 Guide information processing and visualisation | ||
| 3.4 Maximise transfer and generalisation | ||
| 5.3 Build fluencies with graduated levels of support for practice and performance | ||
| Written transcription of video | 1.2 Offer alternatives for auditory information | Barrier decreased – removes the difficulties for students who have auditory challenges |
| Access increased – ensures effective and accurate conveyance of information | ||
| Video-recording of workshops/lectures | 1.1 Offer ways of customising the display of information | Barriers decreased – provides students with the choice on how to best achieve their learning goals |
| 7.1 Optimise individual choice and autonomy | ||
| Videos with subtitles | 1.2 Offer alternatives for auditory information 2.5 Illustrate through multiple media | Barriers decreased – removes the difficulties for students who have auditory challenges; minimises competing interests and engagement by directing students where to focus |
| 3.1 Activate or supply background knowledge | ||
| 3.2 Highlight patterns, critical features, big ideas, and relationships | ||
| 3.3 Guide information processing and visualisation | ||
| 3.4 Maximise transfer and generalisation | ||
| 7. 2 Optimise relevance, value, and authenticity | ||
| 7.3 Minimise threats and distractions | ||
| 8.1 Heighten salience of goals and objectives | ||
| 8.2 Vary demands and resources to optimise challenge | ||
| Animations and interactive simulations | 5.2 Use multiple tools for construction and composition | Access increased – provides students immediate feedback based on their actions; builds up mastery of the concept |
| 8.4 Increase mastery-oriented feedback | ||
| Highlighted texts (bold-faced, underlined, and/or coloured fonts) | 2.3 Support decoding of text, mathematical notation, and symbols | Barrier decreased – removes the difficulty of identifying which aspects of the topic is critical or important |
| 3.2 Highlight patterns, critical features, big ideas, and relationships | ||
| Additional practice problems | 2.3 Support decoding of text, mathematical notation, and symbols | Barrier decreased – removes the difficulty of identifying which aspects of the topic is critical or important |
| 3.2 Highlight patterns, critical features, big ideas, and relationships | ||
| Definitions of new terms in text and new variables/symbols in mathematical equations | 2.1 Clarify vocabulary and symbols | Access increased – ensures accessibility of accurate and intended meaning to all; reduces misinterpretation |
| 2.2 Clarify syntax and structure | ||
| 2.3 Support decoding of text, mathematical notation, and symbols | ||
| Descriptions that accompany images, tables, figures | 1.3 Offer alternatives for visual information | Access increased – ensures accessibility of accurate and intended meaning to all; reduces misinterpretation |
| 2.3 Support decoding of text, mathematical notation, and symbols | ||
| 3.2 Highlight patterns, critical features, big ideas, and relationships | ||
| Self-assessment questions and reflective questions at the end of each topic | 6.4 Enhance capacity for monitoring progress | Access increased – allows students to be engaged in their own learning, and assessing their own progress |
| 7.1 Optimise individual choice and autonomy | ||
| 7. 2 Optimise relevance, value, and authenticity | ||
| 9.3 Develop self-assessment and reflection | ||
| Step-by-step guide and checklist in problem solving | 6.2 Support planning and strategy development | Barriers decreased – guides students to plan their strategy to solve a problem, and provides tools to achieve their goals. |
| Flexible study time | 7.1 Optimise individual choice and autonomy | Barriers decreased – provides students with the choice on how to best achieve their learning goals |
| Option for pen-and-paper or computer tools | 5.1 Use multiple media for communication | Barriers decreased – provides students with the choice on how to best achieve their learning goals; allows students the choice of optimal medium for navigating the content and processing the information |
| 5.2 Use multiple tools for construction and composition | ||
| Feedback from quizzes and exercises | 5.3 Build fluencies with graduated levels of support for practice and performance | Access increased – provides scaffolds to assist them as they build their mastery of the concept and independent learning |
Appendix 3: benefits of implementing the UDL framework in Cases 1, 2, and 3
| Themesa | Description (ways by which students found the UDL-based features helpful)b | Sample quotes |
|---|---|---|
| a Themes identified from focus group discussions and individual interviews from Cases 1, 2, and 3. bThe descriptions in this column were also the codes that were grouped to generate the higher-level categorisation into themes. 1Theme appeared in Case 1. 2Theme appeared in Case 2. 3Theme appeared in Case 3. | ||
| 1. Facilitates processing of information1,2,3 | Help in consolidating the information by: | “If there's a new term popping up, and it's not immediately obvious what it means from the context of the question, then it is nice to have a definition that you could just link to. And especially for some of the symbols as well, like an explanation of what symbols mean.” (Participant 30) |
| • Providing clear guidance and organised presentation | ||
| • Highlighting key elements | ||
| • Solidifying the concepts | ||
| • Ensures complete and accurate scope | ||
| 2. Alternative ways of presenting the information1,2,3 | Providing students with multiple alternatives to the accustomed formats by: | “With the video recording, you can listen to everything because it might be a little too much information if you just read it.” (Participant 21) |
| • Presenting concepts in multimedia formats to enhance visualisation | ||
| • Giving access to other resources that present information in another way | ||
| 3. Allows for varying methods for response or navigation1,2,3 | Making the resources online and digital permits the following advantages: | “Slow-learning students like me, [we] need an in-depth understanding at slow pace…I feel like I need to stop and go back several times for me to grasp it more. I recommend using Moodle for slow-learners like me.” (Participant 2b) |
| • Availability of tools to navigate the content and help students access content or fulfil tasks based on their ability | ||
| • Tools/technology and opportunities to access the content and to perform task more quickly | ||
| 4. Flexibility1,2,3 | Provision for flexibility in time, time management, and pacing | “In the class, it has a fixed timeframe unlike here in Moodle in which you are free to do the tasks anytime or when it is already the deadline…. With Moodle, you can put aside the stuff that are not urgent… In classrooms, of course, you will not be able to do that.” (Participant 9c) |
| 5. Allows for self-evaluation of progress1,2,3 | Opportunity to self-check the scope of assimilated information or measure of achievement such as: | “It's especially useful if you can check that it's right or wrong. … Go back and do it again and check, which was really helpful for concretising some of the lessons that we've been taught.” (Participant 28) |
| • Validating the correct conceptions and rectifying misconceptions | ||
| • Awareness of aspects that need improvement | ||
| 6. Access to free materials1,2 | Practically reducing financial costs to education by providing access to free materials | “When I saw in the resource section that [my university] provides free textbook I was quite happy because I did not know where to get it from. So that was quite helpful and I was happy with that.” (Participant 18) |
| 7. Relating learning to real-life agenda2,3 | Allowing students to connect the concepts with real-life contexts | “[I] look to my sister's chemistry high school chemistry textbook, to see if I could try explaining it to my little sister, and see if I understand it well enough to explain it to a 13-year old.” (Participant 30) |
| 8. Stress management1,2,3 | Helping students manage their expectations and stress by providing them guidance to: | “[T]his one I need to do next time I encounter something like this, that helped me a lot. The stress levels went down when I pause and evaluate my progress.” (Participant 18) |
| • Organise the work they need to complete | ||
| • Adjust their goals | ||
| • Prepare themselves | ||
Acknowledgements
The contributions made by participants from the first-year chemistry courses involved in this three-case study are gratefully acknowledged. This work is supported by the 2019–2021 One UP (University of the Philippines) Faculty Grant, Co-Funded Monash Graduate Scholarship, and the Monash Faculty of Science Dean's International Postgraduate Research Scholarship.References
- Airey J. and Linder C., (2009), A disciplinary discourse perspective on university science learning: Achieving fluency in a critical constellation of modes, J. Res. Sci. Teach., 46(1), 27–49 DOI:10.1002/tea.20265.
- Al-Azawei A., Serenelli F. and Lundqvist K., (2016), Universal design for learning (UDL): A content analysis of peer reviewed journals from 2012 to 2015, J. Scholarship Teach. Learn., 16(3), 39–56 DOI:10.14434/josotl.v16i3.19295.
- American Chemical Society (ACS), (2012), ACS Guidelines and Recommendations for the Teaching of High School Chemistry, ACS Office of Professional Training, Washington, DC, https://www.acs.org/content/dam/acsorg/education/policies/recommendations-for-the-teaching-of-high-school-chemistry.pdf.
- Bartlett II J. E., Kotrlik J. W. and Higgins C. C., (2001), Determining appropriate sample size in survey research, Inf. Technol., Learn., Perform. J., 19(1), 43–50, https://www.opalco.com/wp-content/uploads/2014/10/Reading-Sample-Size1.pdf.
- Basham J. D. and Marino M. T., (2013), Understanding STEM education and supporting students through universal design for learning, Teach. Except. Child., 45(4), 8–15.
- Baumann T. and Melle I., (2019), Evaluation of a digital UDL-based learning environment in inclusive chemistry education, Chem. Teach. Int., 1(2), 1–13 DOI:10.1515/cti-2018-0026.
- Baxter P. and Jack S., (2008), Qualitative case study methodology: Study design and implementation for novice researchers, Qual. Rep., 13(4), 544–559 DOI:10.46743/2160-3715/2008.1573.
- Bianchini J. A., Whitney D. J., Breton T. D. and Hilton-Brown B. A., (2002), Toward inclusive science education: University scientists’ views of students, instructional practices, and the nature of science, Sci. Educ., 86(1), 42–78 DOI:10.1002/sce.1043.
- Burgstahler S., (2020), Universal Design of Instruction (UDI): Definition, principles, guidelines, and examples, DO-IT, https://www.washington.edu/doit/universal-design-instruction-udi-definition-principles-guidelines-and-examples.
- CAST (Center for Applied Special Technology), (2018), The UDL guidelines, http://udlguidelines.cast.org.
- Cook M., Wiebe E. N. and Carter G., (2008), The influence of prior knowledge on viewing and interpreting graphics with macroscopic and molecular representations, Sci. Educ., 92(5), 848–867 DOI:10.1002/sce.20262.
- COU (Council of Ontario Universities), (2020), Introduction to accessible education, https://accessiblecampus.ca/tools-resources/educators-tool-kit/introduction-accessible-education/.
- Curry C., (2003), Universal design: Accessibility for all learners, Educ. Leadership, 61(2), 55–60.
- Curry C., Cohen L. and Lightbody N., (2006), Universal design in science learning, Sci. Teach., 73(3), 32–7.
- Farrell A. M. and Seery M., (2016), Making chemical language easy, Educ. Chem., https://eic.rsc.org/feature/making-chemical-language-easy/2000096.article.
- Ferreira J. E. V. and Lawrie G. A., (2019), Profiling the combinations of multiple representations used in large-class teaching: Pathways to inclusive practices, Chem. Educ. Res. Pract., 20(4), 902–923 10.1039/c9rp00001a.
- Frailich M., Kesner M. and Hofstein A., (2009), Enhancing students’ understanding of the concept of chemical bonding by using activities provided on an interactive website, J. Res. Sci. Teach., 46(3), 289–310 DOI:10.1002/tea.20278.
- Gikandi J. W. and Morrow D., (2016), Designing and implementing peer formative feedback within online learning environments, Technol., Pedag. Educ., 25(2), 153–170 DOI:10.1080/1475939X.2015.1058853.
- Given L. M., (2008), Codes and coding, in The SAGE Encyclopedia of Qualitative Research Methods, pp. 86–88 DOI:10.4135/9781412963909.n48.
- Hamza K., Wickman P. O. and Kelly G., (2008), Describing and analyzing learning in action: An empirical study of the importance of misconceptions in learning science, Sci. Educ., 92(1), 141–164 DOI:10.1002/sce.
- Hannafin M. J. and Hill J., (2007), Resource-based learning, in Spector M., Merrill M. D., van Merrienboer J. and Driscoll M. P. (ed.), Handbook of research on educational communications and technology, Erlbaum, pp. 525–536.
- Hill J. R., (2012), Resource-based learning, in Seel N. M. (ed.), Encyclopedia of the Sciences of Learning, Springer.
- Hoffmann R. and Laszlo P., (1989), Representation in chemistry, Diogenes, 37(147), 23–51 DOI:10.1177/039219218903714702.
- King-Sears M., (2009), Universal design for learning: Technology and pedagogy, Learn. Disabil. Q., 32(4), 199–201 DOI:10.2307/27740372.
- King-Sears M. E. and Johnson T. M., (2020), Universal design for learning chemistry instruction for students with and without learning disabilities, Remed. Spec. Educ., 41(4), 207–218 DOI:10.1177/0741932519862608.
- King-Sears M. E., Johnson T. M., Berkeley S., Weiss M. P., Peters-Burton E. E., Evmenova A. S., Menditto A. and Hursh J. C., (2015), An exploratory study of universal design for teaching chemistry to students with and without disabilities, Learn. Disabil. Q., 38(2), 84–96 DOI:10.1177/0731948714564575.
- Kumar K. L. and Wideman M., (2014), Accessible by design: Applying UDL principles in a first year undergraduate course, Can. J. High. Educ., 44(1), 125–147 DOI:10.47678/cjhe.v44i1.183704.
- Lawrie G. and Bartle E., (2013), Chemistry vlogs: A vehicle for student-generated representations and explanations to scaffold their understanding of structure–property relationships, Int. J. Innov. Sci. Math. Educ., 21(4), 27–45.
- Ludvigsen K., Krumsvik R. and Furnes B., (2015), Creating formative feedback spaces in large lectures, Comput. Educ., 88, 48–63 DOI:10.1016/j.compedu.2015.04.002.
- Mill A. J., Durepos G. and Wiebe E., (2012), Multiple-case designs, in Encyclopedia of Case Study Research, pp. 583–584 DOI:10.4135/9781412957397.
- Mindayula E. and Sutrisno H., (2021), Multiple representation: The teacher's perception in chemistry learning, J. Phys.: Conf. Ser., 1806, 012194 DOI:10.1088/1742-6596/1806/1/012194.
- Mumba F., Banda A., Chabalengula V. M. and Dolenc N., (2015), Chemistry teachers’ perceived benefits and challenges of inquiry-based instruction in inclusive chemistry classrooms, Sci. Educ. Int., 26(2), 180–194.
- Pidgeon N. and Henwood K., (1997), Using grounded theory in psychological research, in Hayes N. (ed.), Doing qualitative analysis in psychology, Psychology Press, Hove, UK, pp. 245–273.
- Pilgrim J. and Ward A., (2017), Addressing diversity through the universal design for learning lens, in Ortlieb E. and Cheek E. (ed.), Addressing Diversity in Literacy Instruction (Literacy Research, Practice and Evaluation, Vol. 8), Emerald Publishing Limited, pp. 229–249 DOI:10.1108/S2048-045820170000008011.
- Pratt J. M. and Yezierski E. J., (2018), A novel qualitative method to improve access, elicitation, and sample diversification for enhanced transferability applied to studying chemistry outreach, Chem. Educ. Res. Pract., 19(2), 410–430 10.1039/c7rp00200a.
- Rabiee F., (2004), Focus-group interview and data analysis, Proc. Nutr. Soc., 63(4), 655–660 DOI:10.1079/pns2004399.
- Rao K., Smith S.J., Edyburn D., Grima-Farrell C., Van Horn G. and Yalom-Chamowitz S., (2018), UDL Reporting Criteria, Developed by a working group of the Universal Design for Learning Implementation and Research (UDL-IRN) Research Committee, Retrieved from https://udl-irn.org/udl-reporting-criteria/.
- Rappolt-Schlichtmann G., Daley S. G., Lim S., Lapinski S., Robinson K. H. and Johnson M., (2013), Universal design for learning and elementary school science: Exploring the efficacy, use, and perceptions of a web-based science notebook, J. Educ. Psychol., 105(4), 1210–1225 DOI:10.1037/a00332171210.
- Rau M. A., (2017), Conditions for the effectiveness of multiple visual representations in enhancing STEM learning, Educ. Psychol. Rev., 717–761 DOI:10.1007/s10648-016-9365-3.
- Raycroft M. A. R. and Flynn A. B., (2020), What works? What's missing? An evaluation model for science curricula that analyses learning outcomes through five lenses, Chem. Educ. Res. Pract., 21(4), 1110–1131 10.1039/c9rp00157c.
- Reyes C. T., Kyne S. H., Lawrie G. A. and Thompson C. D., (2022), Implementing blended first-year chemistry in a developing country using online resources, Online Learning, [accepted manuscript].
- Robinson W. R., (2000), Learning about atoms, molecules, and chemical bonds: A case study of multiple-model use, J. Chem. Educ., 77(9), 1110–1111 DOI:10.1021/ed077p1110.
- Romero Martínez S. J., Ordóñez Camacho X. G., Guillén-Gamez F. D. and Bravo Agapito J., (2020), Attitudes toward technology among distance education students: Validation of an explanatory model, Online Learn., 24(2), 59–75.
- Rose D. H. and Meyer A., (2002), Teaching every student in the digital age: Universal Design for Learning, Alexandria, VA: ASCD.
- Scanlon E., Legron-Rodriguez T., Schreffler J., Ibadlit E., Vasquez E. and Chini J. J., (2018), Postsecondary chemistry curricula and universal design for learning: Planning for variations in learners’ abilities, needs, and interests, Chem. Educ. Res. Pract., 19(4), 1216–1239 10.1039/c8rp00095f.
- Schreffler J., Vasquez E., Chini J. and James W., (2019), Universal design for learning in postsecondary STEM education for students with disabilities: a systematic literature review, Int. J. STEM Educ., 6(1) DOI:10.1186/s40594-019-0161-8.
- Seok S., DaCosta B. and Hodges R. (2018), A systematic review of empirically based universal design for learning: Implementation and effectiveness of universal design in education for students with and without disabilities at the postsecondary level, Open J. Soc. Sci., 6(5), 171–189 DOI:10.4236/jss.2018.65014.
- Smiar K. and Mendez J. D., (2016), Creating and using interactive, 3D-printed models to improve student comprehension of the Bohr Model of the atom, bond polarity, and hybridization, J. Chem. Educ., 93(9), 1591–1594 DOI:10.1021/acs.jchemed.6b00297.
- Taber K., (2018), The use of Cronbach's alpha when developing and reporting research instruments in science education, Res. Sci. Educ., 48,1273–1296 DOI:10.1007/s11165-016-9602-2.
- Taherdoost H., (2017), Determining sample size; how to calculate survey sample size, Int. J. Econ. Manage. Syst., 2, 237–239.
- Teke D. and Sozbilir M., (2019), Teaching energy in living systems to a blind student in an inclusive classroom environment, Chem. Educ. Res. Pract., 20(4), 890–901 10.1039/c9rp00002j.
- UNESCO, (2018), Positioning ICT in Education to Achieve the Education 2030 Agenda in Asia and the Pacific: Recommendations for a Regional Strategy, https://unesdoc.unesco.org/ark:/48223/pf0000261661.
- Vergel de Dios B., (2016), Building and sustaining national ICT education agencies: Lessons from the Philippines, World Bank, Washington, DC, https://openknowledge.worldbank.org/handle/10986/26262.
- Watts F. M. and Finkenstaedt-Quinn S. A., (2021), The current state of methods for establishing reliability in qualitative chemistry education research articles, Chem. Educ. Res. Pract., 22, 565–578 10.1039/D1RP00007A.
- Watt S. J., Therrien W. J., Kaldenberg E. and Taylor J., (2013), Promoting inclusive practices in inquiry-based science classrooms, Teach. Except. Child., 45(4), 40–48.
- Weigold A., Weigold I. K. and Natera S. N., (2019), Response rates for surveys completed with paper-and-pencil and computers: Using meta-analysis to assess equivalence, Soc. Sci. Comput. Rev., 37(5), 649–668 DOI:10.1177/0894439318783435.
- White K. N., Vincent-Layton K. and Villarreal B., (2021), Equitable and inclusive practices designed to reduce equity gaps in undergraduate chemistry courses, J. Chem. Educ., 98(2), 330–339 DOI:10.1021/acs.jchemed.0c01094.
- Yin R., (2013), How to do better case studies: (With illustrations from 20 exemplary case studies), in Bickman L. and Rog D. (ed.), The SAGE Handbook of Applied Social Research Methods, pp. 254–282 DOI:10.4135/9781483348858.n8.
| This journal is © The Royal Society of Chemistry 2022 |