Open Access Article
Open Access ArticleAn isoindoline bridged [M(η6-arene)2]+ (M = Re, 99mTc) ansa-arenophane and its dinuclear macrocycles with axial chirality†
Joshua
Csucker
 ,
Da Kyung
Jo
,
Qaisar
Nadeem
,
Olivier
Blacque
,
Da Kyung
Jo
,
Qaisar
Nadeem
,
Olivier
Blacque
 ,
Thomas
Fox
,
Henrik
Braband
,
Thomas
Fox
,
Henrik
Braband
 and
Roger
Alberto
and
Roger
Alberto
 *
*
Department of Chemistry, University of Zurich, Winterthurerstrasse 190, CH-8057 Zurich, Switzerland. E-mail: ariel@chem.uzh.ch
First published on 1st June 2022
Abstract
This work presents a straightforward method for the preparation of an isoindoline bridged [M(arene)2]+ (M = Re, 99mTc) ansa-[3]arenophane. This intramolecular formation of an ansa-complex is accompanied by the intermolecular formation of a pair of isoindoline bridged macrocyclic dinuclear sandwich complexes, one of which exhibits axial chirality.
Introduction
Cyclopentadienyl-based ansa-metallocenes and arene-based [n]arenophanes (n = number of bridging atoms) are the backbone of olefin polymerization and other catalytic processes.1Ansa-derivatives of ferrocene and group 4 elements have been studied particularly extensive due to their impressive chemical and catalytic diversity.1–4 There is a plethora of literature precedence for late transition metal ansa-complexes5 including group 7 elements.6Ansa-rhenocene complexes with ReIII were reported by Heinekey et al.7 and Conway and coworkers.8 They reported the synthesis of C(CH3)2 and Si(CH3)2 bridged rhenocenes respectively. These interesting compounds suffer though from shortcomings; e.g. the Si(CH3)2 bridged compound is thermally unstable at room temperature (t1/2 = 2 h) and decomposes under methane elimination. The C(CH3)2 linked complex can only be isolated in very low yields. Ansa-[n]arenophanes are far less known and mainly reported since their discovery by Schneider et al. in the 1990s9 for group 6 and for group 8 elements.10 They are unknown at all for rhenium and technetium thus far and represent an unanswered challenge. Given the existence of arenophanes with group 6 ansa-metallocenes and ansa-[n]arenophane with group 8 elements, stable group 7 ansa-[n]arenophanes would fill a knowledge gap.An attractive entry into ansa-chemistry is the [M(η6-arene)2]+ (M = Re, 99mTc) family of compounds. These cationic, arene-based sandwich complexes are water-soluble and air- and moisture stable. Alkyl derivatized [Re(η6-arene)2]+ complexes can directly be synthesized from Na[ReO4] under Fischer–Hafner conditions.11,12 Lithiation of [Re(η6-C6H6)2]+ and quenching with electrophiles leads to an array of mono- and bis-substituted compounds.13–18 Alternatively, naphthalene in [Re(η6-napht)2]+ can be exchanged by functionalized arenes with a high degree of functional group tolerance.15,18
Technetium chemistry is developed in tandem with the one of rhenium. 99mTc complexes are of special interest for diagnostic, medicinal chemistry due to their potential application as radio-pharmaceuticals.19,20 A distinct advantage of [99mTc(η6-arene)2]+ complexes is their direct synthesis in water and under mild conditions with vast substrate scope.15,18,21
We present in this report the first example of stable [M(η6-arene)2]+ (M = Re, 99mTc) type ansa-complex, featuring an isoindoline unit as the bridge between the two arene ligands, and an uncommon dinuclear macrocycle formed by intermolecular isoindoline formation starting from [Re(η6-C6H5-NH2)2]+.
Results and discussion
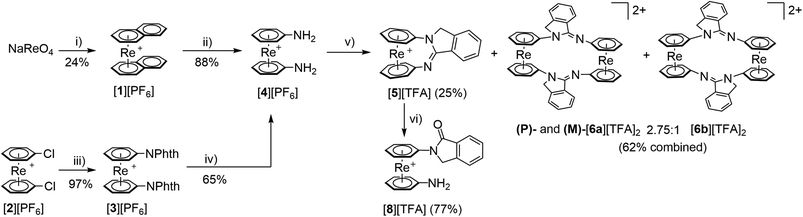
Since the aforementioned silyl- or carbon-bridged ansa-metallocenes of ReIII are temperature, oxygen and moisture sensitive, we hypothesized that ansa-[n]arenophanes would be more persistent. We targeted to exploit the literature reported condensation reaction between 2 eq. of aniline or amino pyridines and ortho-phenyldialdehyde (OPA) which is known to form heterocyclic isoindolines units in a straight-forward manner (Scheme 1, left).22–24 We rationalized that a 1,1′-diamino sandwich complex might replace the individual anilines in the organic template reaction to form the corresponding ansa-complex. With [M(aniline)2]+ (M = Re: [4]+; M = 99mTc: [7]+) as synthetic precursor an isoindoline-bridged ansa-[3]arenophane should be accessible by treatment with OPA (Scheme 1). Complex [4][PF6] was prepared via two different synthetic routes (Scheme 2). Starting from [1][PF6], [4][PF6] can be obtained in 21% yield over two steps according to a literature procedure by arene exchange of naphthalene with aniline.15 The literature yields were improved from 39 to 88% by employing a large excess of aniline as compared to the literature report. An alternative route is an adapted Gabriel amine synthesis inspired by the preparation of 1,1′-diaminoferrocene as reported by Abdulmalic and Rüffer.25 Bis-chlorinated [2][PF6]17 was treated with potassium phthalimide (KPhth) in the presence of copper to provide [3][PF6] in excellent yields (97%). The X-ray structure of [3][PF6] was elucidated (ESI, Fig. S24†). Hydrazine mediated phthalimide elimination delivered [4][PF6] in a satisfactory yield of 65% (Scheme 2). | ||
| Scheme 1 Concept for the synthesis of novel isoindoline bridged ansa-complexes. OPA = ortho-phenyldialdehyde; M = Re, 99mTc. | ||
Complex [4][PF6] was treated with stoichiometric amounts of OPA (Scheme 2), which provided the ansa-complex [5][TFA] in 25% yield. Additionally, two dinuclear macrocyclic complexes, [6a][TFA]2 and [6b][TFA]2 were isolated in a combined yield of 62% and in a 2.75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio according to 1H NMR data (ESI Fig. S7–S15†). Increasing the dilution of the reagents distinctly favors the formation of [5]+, albeit with sustained observation of the dinuclear species. At extremely high dilution, as in case of 99mTc (vide infra), exclusively the monomer is observed. All products were characterized by NMR, FT-IR, HR-ESI-MS, elemental analysis (EA) and single crystal X-ray diffraction (ESI, Fig. S17–S23 and S25–27†). Due to the hygroscopic nature of the TFA salts, the compounds were converted to the respective PF6− salts for EA measurements.
1 molar ratio according to 1H NMR data (ESI Fig. S7–S15†). Increasing the dilution of the reagents distinctly favors the formation of [5]+, albeit with sustained observation of the dinuclear species. At extremely high dilution, as in case of 99mTc (vide infra), exclusively the monomer is observed. All products were characterized by NMR, FT-IR, HR-ESI-MS, elemental analysis (EA) and single crystal X-ray diffraction (ESI, Fig. S17–S23 and S25–27†). Due to the hygroscopic nature of the TFA salts, the compounds were converted to the respective PF6− salts for EA measurements.
The formation of [5]+ was monitored over the course of the reaction by UPLC-ESI-MS. Based on results thus obtained, we propose a step-by-step mechanism shown in Scheme 3. This proposal is based on a similar pathway as described by Chebolu et al. for the synthesis of 1,2-disubstituted benzimidazoles.22 Two possible routes lead to the ansa-species [5]+, namely formation of a symmetrical di-imine followed by cyclization or a sequence of mono-imine formation followed by a sigmatropic rearrangement. According to our data, the second pathway is more likely to be at play. Imine formation between [4]+ and OPA (observed as [M + H2O]+; m/z = 507.05) followed by nucleophilic attack of the second aniline nitrogen generates the A ring. Subsequent imine formation between the secondary amine and the remaining aldehyde moiety results in the fused A–B–C ring system, supported by the observation of the key intermediate I. Elimination of water and rapid [1,3] sigmatropic rearrangement finally delivers [5]+. The dinuclear species [6a]2+ and [6b]2+ are the result of intermolecular imine formations instead of an intramolecular pathway. A detailed overview of all observed intermediates and pathways is provided in the ESI (Scheme S1†).
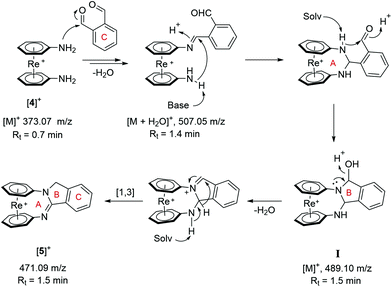 | ||
| Scheme 3 Mechanistic reaction scheme of the formation of [5]+ from [4]+. Indicated m/z values and retention times (Rt) correspond to observed intermediates. | ||
Complex [5][PF6] crystallized in the monoclinic space group P21/c with one CH3CN solvent molecule in the asymmetric unit (Fig. 1). The centroid–Re–centroid angle (169.66(7)°) is significantly smaller than the optimal 180° but still larger than the one reported by Heinekey et al. for their ansa-rhenocene (145.2(16)°).7 The angle between the η6-C6H6 planes is 15.23(9)°, indicating an exposure of the rhenium center. The arene ligands are in an almost perfectly eclipsed conformation. The bridging isoindoline unit is arranged perpendicular to the π-surfaces of the sandwich scaffold.
 | ||
| Fig. 1 ORTEP representation of the cations [5]+ of the crystal structure [5][PF6]·CH3CN (left), and [6b]2+ of the crystal structure [6b][TFA]2·2H2O (right). Thermal ellipsoids represent 50% probability. Hydrogens and counterions were omitted for clarity.‡ | ||
We were interested in reactivities of [5]+, particularly in the question if oxidative addition to the exposed rhenium center would occur, given the strain imposed on the arene ligands by the bridging isoindoline. Such reactivities would be desirable if the ansa-complexes were supposed to enter some catalytic processes. For their application in radiopharmacy, however, any metal-based reactivity has to be omitted. Treatment of [5][TFA] with CH3I however did not lead to an oxidative addition and reaction with HBF4 did not lead to rhenium protonation. Clean hydrolysis of the imine was observed with aqueous acids (77% yield, see ESI†) to yield compound [8][TFA]. This reaction is however, ligand-rather than metal-based. The strain of the arene ligands is thus too small to expose the rhenium center and facilitating e.g. oxidative addition reactions.
Analytically pure samples of [6a][TFA]2 were obtained by fractional crystallization. Complex [6b][TFA]2 could not be fully separated from [6a][TFA]2. A structure elucidation revealed axial chirality26 in [6a]2+, thus both (P)- and (M)-enantiomers are present in the crystals as evident from a combination of NMR and crystallographic data. Moreover, classical coalescence behavior of the 1H NMR signals was observed between 270 K and 330 K (ESI, Fig. S28†). This coalescence process describes the rapid interconversion between (P)- and (M)-enantiomers of [6a]2+. The process has a free activation energy barrier of ΔG‡ = 64.0 ± 0.4 kJ mol−1, which corresponds to a first order rate constant k = 65.76 s−1 at 298 K (ESI chapter 4†). The best estimate for the coalescence temperature is 323 K. Thus, the solution structure of [6a]2+ at room temperature is described as a rapid equilibrium between its enantiomers. In combination with ROESY correlation data (ESI, Fig. S31†), we assessed that only [6a]2+ but not [6b]2+ is involved in the coalescence behavior. The crystallographic data of [6a][TFA]2 confirmed the axial chirality and the presence of a racemate (Fig. 2). It crystallized in the centrosymmetric space group C2/c and the two rhenium centers lie on a two-fold axis. The asymmetric unit features both helical enantiomers. We assigned the (P)- and (M)-enantiomers based on rotational direction of the head-to-head oriented helices.26 Bond lengths and angles of the isoindoline units are in the same range as those of [5][PF6]·CH3CN. The crystal structure of [6b][TFA]2 shows a flytrap-like head-to-tail geometry of the two isoindoline-units bridging the two [Re(η6C6H6)2]+ scaffolds (Fig. 1).
 | ||
| Fig. 2 Side-by-side ORTEP representations of the cations (P)-[6a]2+ (left) and (M)-[6a]2+of the crystal structure [6a][TFA]2·H2O. Counterions, hydrogen atoms and labels were omitted for clarity.‡ The black bar represents the mirror plane relating the two enantiomers. | ||
It was tempting to investigate, if 99mTc would yield the same ansa-[n]arenophane complex directly in water and despite the presence of water, which affects imine formation. To verify this hypothesis, the same route as with rhenium was employed. Aqueous [99mTcO4]− was treated with aniline and zinc (impossible for Re) in saline which produced exclusively [99mTc(η6-aniline)2]+ ([7]+, Scheme 4).15 To remove excess aniline, the crude reaction mixture was purified via HPLC and peaks containing [7]+ collected. The resulting solution was treated with OPA which gave the 99mTc homologue ansa-complex [99mTc][5]+ in a clean reaction. Complex [99mTc][5]+ was isolated in excellent radiochemical purity of >98% after HPLC purification. Its chemical identity was confirmed by chromatographic coinjection with the rhenium homologue [5][TFA] (Fig. 3). Complex [99mTc][5]+ represents the first example of a technetium ansa-complex. Although a two-step reaction, it is amazing that this kind of structurally diverse complexes can be prepared in water and in good yields. Analysis of the reaction solution did not indicate the formation of dinuclear species, analogous to [6a]2+. The high dilution of 99mTc (around 10−8 M) would require an extremely fast process to form dinuclear species, which appeared not to be the case with the reaction between [7]+ and OPA.27
 | ||
| Scheme 4 Synthesis of the 99mTc ansa-complex [99mTc][5]+. Reaction conditions: (i) zinc, aniline, 100 °C, 30 min, microwave; (ii) OPA, 80 °C, 40 min. | ||
 | ||
| Fig. 3 Coinjection of [99mTc][5]+ (red) with [5]+ (black). The difference in retention times is associated with the dead volume between UV cell and radio detector. | ||
Especially the well-established synergy between rhenium and technetium renders the presented compounds interesting candidates for the development of chemical and radioactive probes.
Conclusions
We report the first examples of ansa-[M(η6-arene)2]+ (M = Re, 99mTc) complexes, featuring an isoindoline as the bridging unit. The high degree of stability coupled with the biorelevant properties of the isoindoline unit24 renders [5][TFA] an attractive candidate as building block for bioorganometallic chemistry. The 1,2-dialdehyde unit can thereby act as an anchoring group to an active pharmaceutical or be even part of it. The fact that the analogous 99mTc complex is readily available further underlines the potential of the system in a radiopharmaceutical context. We assess that further exposure of the rhenium is required to explore eventual catalytic applications of [5]+ and therefore, a more straining bridging unit is required.Moreover, two dinuclear macrocyclic complexes, featuring comparably rare axial chirality for dinuclear species, were isolated and fully characterized. Perspective studies of the dinuclear species point to the preparation of heteronuclear Re–99mTc dimers to probe them as potential theranostic pairs.
Author contributions
JC wrote and edited the manuscript, conceptualized the project and performed experiments, DKJ performed experiments and edited the initial manuscript, QN performed 99mTc chemistry, OB performed all crystallographic measurements, TF recorded NMR spectra of [6a][TFA]2 at various temperatures, HB advised 99mTc experiments, RA revised the manuscript and initiated the project.Conflicts of interest
There are no conflicts to declare.Acknowledgements
The authors acknowledge financial support from the University of Zurich.Notes and references
- B. Wang, Coord. Chem. Rev., 2006, 250, 242–258 CrossRef CAS.
- M. A. Bau, S. Wiesler, S. L. Younas and J. Streuff, Chem. – Eur. J., 2019, 25, 10531–10545 CrossRef CAS PubMed.
- L. N. Jende, A. Vantomme, A. Welle, J.-M. Brusson, J.-F. Carpentier and E. Kirillov, J. Organomet. Chem., 2018, 878, 19–29 CrossRef CAS.
- J. Kuwabara, D. Takeuchi and K. Osakada, ChemComm, 2006, 3815–3817 RSC.
- T. Arnold, H. Braunschweig, A. Damme, C. Hörl, T. Kramer, I. Krummenacher and J. Mager, Organometallics, 2014, 33, 1659–1664 CrossRef CAS.
- H. Braunschweig, A. Damme, K. Dück, M. Fuß, C. Hörl, T. Kramer, I. Krummenacher, T. Kupfer, V. Paprocki and C. Schneider, Chem. – Eur. J., 2015, 21, 14797–14803 CrossRef CAS PubMed.
- D. M. Heinekey and C. E. Radzewich, Organometallics, 1999, 18, 3070–3074 CrossRef CAS.
- S. L. J. Conway, L. H. Doerrer, J. C. Green, M. L. H. Green, A. Scottow and A. H. H. Stephens, J. Chem. Soc., Dalton Trans., 2000, 329–333 RSC.
- J. J. Schneider, N. Czap, D. Spickermann, C. W. Lehmann, M. Fontani, F. Laschi and P. Zanello, J. Organomet. Chem., 1999, 590, 7–14 CrossRef CAS.
- D. Seyferth, Organometallics, 2002, 21, 2800–2820 CrossRef CAS.
- E. O. Fischer and A. Wirzmüller, Chem. Ber., 1957, 90, 1725–1730 CrossRef CAS.
- E. A. Trifonova, D. S. Perekalin, K. A. Lyssenko and A. R. Kudinov, J. Organomet. Chem., 2013, 727, 60–63 CrossRef CAS.
- G. Meola, H. Braband, P. Schmutz, M. Benz, B. Spingler and R. Alberto, Inorg. Chem., 2016, 55, 11131–11139 CrossRef CAS PubMed.
- G. Meola, H. Braband, D. Hernández-Valdés, C. Gotzmann, T. Fox, B. Spingler and R. Alberto, Inorg. Chem., 2017, 56, 6297–6301 CrossRef CAS PubMed.
- Q. Nadeem, G. Meola, H. Braband, R. Bolliger, O. Blacque, D. Hernández-Valdés and R. Alberto, Angew. Chem., Int. Ed., 2020, 59, 1197–1200 CrossRef CAS PubMed.
- D. Hernández-Valdés, R. Fernández-Terán, B. Probst, B. Spingler and R. Alberto, Helv. Chim. Acta, 2020, 103, e2000147 CrossRef.
- D. Hernández-Valdés, F. Avignon, P. Müller, G. Meola, B. Probst, T. Fox, B. Spingler and R. Alberto, Dalton Trans., 2020, 49, 5250–5256 RSC.
- Q. Nadeem, F. Battistin, O. Blacque and R. Alberto, Chem. – Eur. J., 2022, 28, e202103566 CrossRef CAS PubMed.
- A. G. Jones, Radiochim. Acta, 1995, 70–71, 289–298 CrossRef.
- U. Mazzi, R. Schibli, H.-J. Pietzsch, J.-U. Künstler and H. Spies, in Technetium-99m Pharmaceuticals: Preparation and Quality Control in Nuclear Medicine, ed. I. Zolle, Springer Berlin Heidelberg, Berlin, Heidelberg, 2007, pp. 7–58 Search PubMed.
- M. Benz, H. Braband, P. Schmutz, J. Halter and R. Alberto, Chem. Sci., 2015, 6, 165–169 RSC.
- R. Chebolu, D. N. Kommi, D. Kumar, N. Bollineni and A. K. Chakraborti, J. Org. Chem., 2012, 77, 10158–10167 CrossRef CAS PubMed.
- S. S. Al-Shihry, Molbank, 2005, 2005, M447 CrossRef.
- I. Sović, S. Kraljević Pavelić, E. Markova-Car, N. Ilić, R. Nhili, S. Depauw, M.-H. David-Cordonnier and G. Karminski-Zamola, Eur. J. Inorg. Chem., 2014, 87, 372–385 Search PubMed.
- M. A. Abdulmalic and T. Rüffer, Bull. Chem. Soc. Jpn., 2013, 86, 724–728 CrossRef CAS.
- M. Rickhaus, M. Mayor and M. Juríček, Chem. Soc. Rev., 2016, 45, 1542–1556 RSC.
- R. Bolliger, A. Frei, H. Braband, G. Meola, B. Spingler and R. Alberto, Chem. – Eur. J., 2019, 25, 7101–7104 CrossRef CAS PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available. CCDC 2142820–2142823. For ESI and crystallographic data in CIF or other electronic format see DOI: https://doi.org/10.1039/d2dt00743f |
| ‡ The crystallographic data is available in the electronic ESI.† |
| This journal is © The Royal Society of Chemistry 2022 |