Nanoporous high-entropy alloys for highly stable and efficient catalysts†
Abstract
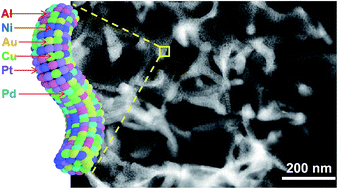
Controllably incorporating multiple immiscible metal elements into one single nanostructure has immeasurable technological and scientific potential, but it remains a challenge for the conventional bottom-up synthetic methods. Herein, we presented a general and scalable route to prepare multi-component nanostructured alloys referred to as nanoporous high-entropy alloys (np-HEAs) by combining bulk melting, fast cooling, and dealloying. To demonstrate this concept, we synthesized senary AlNiCuPtPdAu, octonary AlNiCuPtPdAuCoFe, and senary all-non-noble metal AlNiCuMoCoFe np-HEA with ligament sizes of ∼2–3 nm and precisely controlled composition by dealloying the designed precursor alloys. With a naturally formed thin oxide layer of spinel γ-Al2O3, AlNiCuPtPdAu np-HEA exhibited greatly enhanced high-temperature stability (up to 600 °C) and CO oxidation activity. Interestingly, with the removal of the surface oxide layer, np-HEA still showed good resistance to coarsening at 200 °C for 10 h due to its intrinsically low diffusivity originating from the multiple-principal-element mixing effect. For electrocatalysis, np-HEA with a low Pt loading amount exhibited 10 times the mass activity of Pt/C for oxygen reduction reaction and maintained 92.5% of its initial activity after 100k electrochemical cycles.


 Please wait while we load your content...
Please wait while we load your content...