Adsorptive catalysis of hierarchical porous heteroatom-doped biomass: from recovered heavy metal to efficient pollutant decontamination†
Abstract
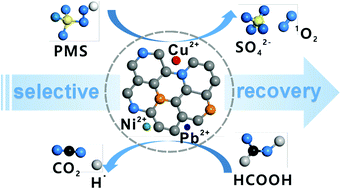
Extraction of heavy metals from wastewater serves to meet the global challenges of toxic metal waste remediation and valuable metal recycling. Herein, by utilizing a cheap and abundant biowaste of defatted soybean residues as a N-rich biomass precursor, a heteroatom-doped macroporous carbon biosorbent (denoted CKS) was formed through a one-pot pyrolysis method in the presence of potassium oxalate (activating agent) and calcium sulfate (hard template). Increased surface polarity with high sulfur doping (10.73 at%) results in the as-prepared material having a strong affinity for heavy metals due to soft acid-soft base interactions, and the optimized CKS-800 (pyrolyzed at 800 °C) shows high selectivity toward Cu2+ and Ni2+ with high distribution coefficients (6.7 × 105 mL g−1 for Cu2+ and 9.8 × 104 mL g−1 for Ni2+). Detailed isotherm studies demonstrate the excellent adsorption capacities of CKS toward Cu2+ (1366.67 mg g−1), Ni2+ (1250.21 mg g−1) and Pb2+ (619.23 mg g−1). Moreover, the prepared CKS–metals (CKS–M) after metal remediation display superior catalytic capability both for organic pollutant degradation with peroxymonosulfate as an oxidant and catalytic reduction of toxic CrVI to nontoxic CrIII with formic acid as a reducing agent. The synergistic effects of crystalline carbon and heteroatom doping render the low-cost hierarchical CKS biomass an effective adsorptive catalysis material for efficient water remediation and sustainable utilization in homogeneous catalysis, which avoids further disposal and valorizes the metal complex itself in a “circular economy” model for multi-pollutant decontamination.


 Please wait while we load your content...
Please wait while we load your content...