Nano porous hydroxyapatite as a bi-functional catalyst for bio-fuel production†
Abstract
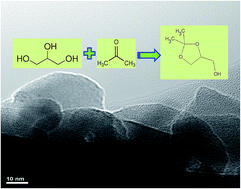
Nano porous hydroxyapatite possessing acid–base functionality has been successfully synthesized by a simple hydrothermal method using P123 tri-block co-polymer as a structure directing agent. The material exhibited excellent catalytic activity for the production of solketal (100% selectivity) from bio-glycerol (90% conversion).

 Please wait while we load your content...
Please wait while we load your content...