Concise synthesis of bicyclic iminosugars via reductive functionalization of sugar-derived lactams and subsequent RCM reaction†
Abstract
An efficient method for the synthesis of bicyclic iminosugars has been developed. The strategy is based on the partial reduction of sugar-derived lactams by Schwartz's reagent and tandem stereoselective nucleophile addition dictated by Woerpel's model which affords polyhydroxylated cyclic amines as key intermediates. Introduction of a vinyl or allyl group to the iminosugar produces diene derivatives that can be subjected to the ring-closing metathesis reaction (RCM) to furnish polyhydroxylated pyrrolizidine, indolizidine and quinozilidine derivatives in good to excellent yields. This sequence of reactions has been applied to the formal synthesis of hyacinthacine A2, a polyhydroxylated pyrrolizidine alkaloid.
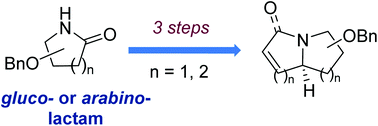
- This article is part of the themed collection: Synthetic methodology in OBC


 Please wait while we load your content...
Please wait while we load your content...