Studies on chemoselective synthesis of 1,4- and 1,2-dihydropyridine derivatives by a Hantzsch-like reaction: a combined experimental and DFT study†
Abstract
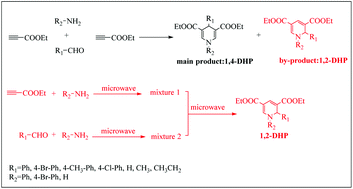
In the experimental process of preparing diethyl 3,5-dicarboxylate-1,4-dihydropyridine (1,4-DHP) by a Hantzsch-like reaction, it was found that a by-product named diethyl 3,5-dicarboxylate-1,2-dihydropyridine (1,2-DHP) was produced in the reaction. To discuss this phenomenon, the effects of the reaction conditions on the yield ratio of 1,4-DHP and 1,2-DHP were studied by using aromatic amines, aromatic aldehydes and ethyl propiolate as raw materials. The mechanisms for the formation of 1,4-DHP and 1,2-DHP were proposed based on the isolated intermediate named diethyl 4-((phenylamino)methylene)pent-2-enedioate generated by the Michael addition of aniline and ethyl propiolate. The transition state structures were optimized and the reaction energy barriers of intermediates in the speculated mechanisms were calculated by DFT calculations at the M062X/def2TZVP//B3LYP-D3/def-SVP level. It was found that the reaction energy barriers and dominant configurations of intermediates IM2 and IM3′ are the determinants for the chemoselectivity. Together, these results demonstrate a high chemoselectivity in the synthesis of 1,4-DHPs and 1,2-DHPs by a Hantzsch-like reaction and that 1,4-DHPs and 1,2-DHPs can be easily obtained under different conditions.
- This article is part of the themed collection: Mechanistic, computational & physical organic chemistry in OBC


 Please wait while we load your content...
Please wait while we load your content...