Cationic dye adsorption and separation at discrete molecular level: first example of an iron cluster with rapid and selective adsorption of methylene blue from aqueous system†
Abstract
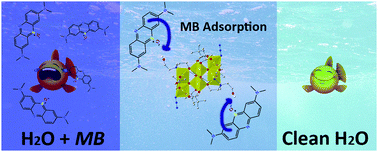
A hexanuclear iron(III) complex, [Fe6III(μ3-O)(edteH)2(piv)6(N3)2] (1), was prepared by the reaction of FeCl2·4H2O with N,N,N′,N′-tetrakis(2-hydroxyethyl)ethylenediamine (H4edte) and pivalic acid (Hpiv) in the presence of NaN3 in MeCN/CH2Cl2. Its X-ray structure determination demonstrated a centrosymmetric complex with two triangular [Fe3(μ3-O)]7+ units. To verify the importance of this material, the adsorption study for 1 was performed. The use of 1 as a possible dye adsorbent was evaluated in an aqueous medium on two model dyes: methylene blue (MB) and methyl orange (MO). The results obtained indicated that 1 showed remarkable adsorption properties for MB (88.60%). This Fe cluster is the first example of a homometallic coordination cluster that presents an exciting adsorption property. The adsorption factor was that free O–H groups in the coordination cluster delivered O–H⋯π interactions through the MB dye, which was attributed to enhanced adsorption. The effects of various parameters, such as pH and temperature, were studied. The MB adsorption efficiency by adsorbent was ∼89% at neutral pH; however, for MO, it was ∼48% at the same pH. Also, MB adsorption at a higher temperature (40 °C) was ∼96%, whereas ∼61% for MO. The kinetic data provided a good confirmation of the adsorption pathways, i.e. the pseudo-second-order. Overall, this study disclosed an easy-to-prepare, stable, and reusable iron(III) cluster as a cost-effective, eco-friendly and efficient adsorbent for hazardous water-soluble aromatic dyes. This type of adsorbents can be fine-tuned further for the desired efficiency and dye selectivity.


 Please wait while we load your content...
Please wait while we load your content...