Molecular engineering of α and β peripherally tri-halogenated substituted boron subphthalocyanines as mixed alloys to control physical and electrochemical properties for organic photovoltaic applications†
Abstract
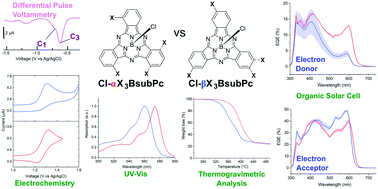
The chirality feature of C1 and C3 boron subphthalocyanines (BsubPcs) is an attractive property in material science and supramolecular chemistry. Normally in the field, enantiomeric mixtures are separated through a standard process. The goal of this study was to determine if the mixture of BsubPc enantiomers could form a mixed alloyed composition in the solid state, which is very relevant to their potential application in the organic electronic field. We present the synthesis and physical characteristics of a selection of four chiral BsubPc mixtures. These BsubPcs are specifically trichlorinated and trifluorinated in the periphery, such as Cl-βF3BsubPc, Cl-αF3BsubPc, Cl-βCl3BsubPc and Cl-αCl3BsubPc. The isomeric (C1/C3) ratios of each BsubPc were determined by NMR. Single-crystal XRD of all four BsubPc mixtures showed that the C1 and C3 isomers co-crystallized within their respective lattices forming solid-state mixed alloyed compositions. The structures of Cl-αF3BsubPc and Cl-βF3BsubPc crystallized in the same lattice as Cl-BsubPc, with some expansion of the unit cell volume, while the trichlorinated BsubPcs did not due to the large van der Waals radii of the chlorine atoms. This set of mixed alloyed BsubPcs was also integrated into organic solar cells/photovoltaics (OSCs/OPVs) as both non-fullerene electron acceptors and as electron donors. It was confirmed that these BsubPc isomers/enantiomers are applicable in OPVs as mixed alloyed compositions, with more promising functionality as non-fullerene electron acceptors given their significant contribution to the EQE spectra. Further molecular engineering of these materials will be made to enhance their OPV performance and to explore their bifunctional charge carrier mobility roles.


 Please wait while we load your content...
Please wait while we load your content...